Guidelines for Canadian Drinking Water Quality: Guideline Technical Document – Benzo[a]pyrene
Endnotes
Table of Contents
- Part I. Overview and Application
- Part II. Science and Technical Considerations
- 4.0 Identity, use and sources in the environment
- 5.0 Exposure
- 6.0 Analytical methods
- 7.0 Treatment technology and distribution system considerations
- 8.0 Kinetics and metabolism
- 9.0 Health effects
- 10.0 Classification and assessment
- 11.0 Rationale
- 12.0 References
- Appendix A: List of acronyms
Benzo[a]pyrene
Part I. Overview and Application
1.0 Guideline
The maximum acceptable concentration (MAC) for benzo[a]pyrene (BaP) in drinking water is 0.000 04 mg/L (0.04 µg/L).
2.0 Executive summary
Levels of benzo[a]pyrene (BaP) in Canadian drinking water are generally very low. There are no industrial or commercial uses for BaP in Canada, but BaP is commonly found in the environment as a product of the incomplete combustion of organic matter. Natural sources of BaP emissions (e.g., forest fires, volcanic activity) have been documented, but human-made sources, such as combustion of fossil fuels, residential wood burning, creosote-treated wood and coke oven emissions, are the major sources of BaP emissions in the environment. Although BaP can be found in both surface water and groundwater, its presence in drinking water is primarily due to its leaching from the linings of water storage tanks and distribution lines.
This guideline technical document reviews and assesses all identified health risks associated with BaP in drinking water. It incorporates new studies and approaches and takes into consideration the availability of appropriate analytical methods and treatment technologies. Based on this review, the guideline for BaP in drinking water is a maximum acceptable concentration (MAC) of 0.000 04 mg/L (0.04 µg/L).
2.1 Health effects
BaP has been classified by the International Agency for Research on Cancer (IARC) in Group 1: carcinogenic to humans, based on sufficient evidence in animals and strong evidence that the cancer mechanisms observed in animals also apply to humans. Limited studies in humans suggest increased cancer risks in individuals exposed to mixtures of substances that included BaP, namely consumers of charred meats, coke oven workers and smokers. Long-term exposure of rodents to BaP induces various types of tumours affecting the forestomach, liver, esophagus, lung, tongue, larynx, oral cavity, skin and mammary gland. The cancer risk assessment was based on forestomach tumours (used as a surrogate for all cancer endpoints) in female mice.
Various non-cancer health effects related to exposure to BaP were observed in rodents, including neurological and neurodevelopmental effects occurring at low levels of exposure. Both cancer and non-cancer risk assessments were considered. The cancer risk assessment was used to establish the MAC since it resulted in a more conservative value and there is greater confidence in the known carcinogenic effects of BaP than in non-cancer effects.
2.2 Exposure
Canadians can be exposed to BaP through its presence in food, air, consumer products, drinking water, soil and some occupational settings. The main source of exposure to BaP is food (e.g., charred meat). BaP is not frequently found in Canadian drinking water supplies, and levels detected are generally low. As BaP is neither volatile nor readily absorbed through the skin, the contributions of the dermal and inhalation routes of exposure to overall exposure from drinking water are considered to be negligible.
2.3 Analysis and treatment
The establishment of a drinking water guideline must take into consideration the ability to both measure the contaminant in and remove it from drinking water supplies. Existing analytical methods are available to reliably measure BaP in drinking water below the MAC.
Treatment technologies are available at both municipal and residential scales to remove BaP from water to below the MAC. The best available technology for removing BaP is granular activated carbon. At the municipal scale, currently available technologies expected to be able to consistently achieve BaP concentrations below the MAC are conventional treatment, granular activated carbon and powder activated carbon.
At the residential level, although there are no treatment units currently certified to remove BaP, treatment devices using technologies such as activated carbon may remove BaP from drinking water to achieve concentrations below the MAC.
3.0 Application of the guideline
Note: Specific guidance related to the implementation of drinking water guidelines should be obtained from the appropriate drinking water authority in the affected jurisdiction.
BaP is a human carcinogen, and exposure to any concentration from any medium of exposure, including drinking water, may increase the risk of cancer. The drinking water guideline is based on lifetime exposure (70 years) to BaP from drinking water.
The guideline for a carcinogen is normally established at a level at which the increased cancer risk is "essentially negligible" when a person is exposed at that concentration in drinking water over a lifetime. In the context of drinking water guidelines, Health Canada has defined the term "essentially negligible" as a range from one new cancer above background levels per 100 000 people to one new cancer above background levels per 1 million people (i.e., 10-5-10−6). An increased cancer risk of 10−6 is targeted in cases where intake from sources other than drinking water are significant. In the case of BaP, where food is the main source of exposure, the MAC of 0.000 04 mg/L is established at a concentration that would present an "essentially negligible" risk of one new cancer above background per million people (i.e., 10−6) exposed to BaP in drinking water at the MAC over a lifetime.
For drinking water supplies that occasionally experience short-term exceedances above the MAC, it is suggested that a plan be developed and implemented to address these situations. For more significant, long-term exceedances that cannot be addressed through treatment, it is suggested that alternative sources of drinking water be considered.
3.1 Monitoring
Water sources should be characterized to determine if BaP is present. Quarterly monitoring should be conducted when surface waters are impacted by municipal sewage effluent, petroleum refinery wastewater and oil spills. In addition, surface water sources that are or may have been impacted by the deposition of polycyclic aromatic hydrocarbons (PAHs) (e.g., forest fires) should be monitored. Groundwater that is impacted by petroleum or PAH contamination should also be monitored. Utilities that have baseline data indicating that BaP is not present may conduct less frequent monitoring.
Although components and coatings for distribution systems are now required to meet standards that limit the leaching of contaminants, distribution pipes and storage tanks installed prior to the 1980s may leach BaP if coal tar (bitumen) or asphaltic products were used to coat them. It is recommended that distribution systems with these linings be monitored for BaP and other PAHs. Quarterly monitoring for BaP should be undertaken in the areas of the distribution system where coal tar linings may have been used, especially at locations with the maximum residence time (e.g., dead ends) or when there are hydraulic disturbances in these locations (e.g., water main replacement). Utilities that have baseline data indicating that BaP is not present within the distribution system may conduct less frequent monitoring. Jurisdictions may consider reduced sampling if an appropriate flushing protocol is in place.
3.2 Mitigation
Distribution system operations designed to promote water quality and minimize water age will minimize the release of BaP. The following actions are recommended to help reduce the concentration of BaP originating from the distribution system:
- Maintain the dose of disinfectants within the range required for their application.
- Avoid changes in the hydraulic regime and water hammer. Differences in water pressure of more than 3 bar (45 psi) can result in the mobilization of particles containing PAHs.
- Reduce the residence time of water in the distribution system. Stagnation periods of more than 7 hours can lead to elevated PAH concentrations.
- Systematically flush the distribution system to ensure the removal of particles.
Part II. Science and Technical Considerations
4.0 Identity, use and sources in the environment
Benzo[a]pyrene (BaP) (Chemical Abstracts Service registry number 50-32-8) is a relatively large molecule of the polycyclic aromatic hydrocarbon (PAH) class of compounds. It is composed of five unsubstituted rings and is the most well studied of the PAHs. It has the chemical formula C20H12 and a molecular weight of 252.3 g/mol. BaP and other PAHs are formed during the incomplete combustion or pyrolysis of organic matter. At ambient pressures and temperatures, BaP exists as a relatively stable, pale yellow solid with a faint aromatic odour. BaP is not readily volatile or reactive. It has a melting point that ranges from 176.5°C to 179.3°C, a boiling point ranging from 310°C to 495°C and a Henry's law constant of 4.57 × 10−7 at 25°C. The reported octanol-water partition coefficients (log Kow) of BaP are relatively high, ranging from 5.97 to 6.35 (IARC, 2010; OEHHA, 2010; HSDB, 2011; CHEMFATE, 2013; U.S. EPA, 2013). The solubility of BaP in water is limited (1.62 µg/L at 25°C), whereas BaP is readily soluble in organic solvents, including chloroform, benzene, toluene and xylene (IARC, 2010; HSDB, 2011; CHEMFATE, 2013; U.S. EPA, 2013). No data on the odour and taste thresholds for BaP were identified (ATSDR, 1995; OEHHA, 2010).
PAHs are found ubiquitously in the environment, typically at low concentrations, although levels can be high in the proximity of PAH sources. They have no industrial uses and are not manufactured. However, they occur naturally during forest fires and volcanic eruptions and, to a greater extent, anthropogenically through the combustion of fossil fuels, in coke oven emissions and through agricultural management practices, among other sources. The largest single source is the burning of wood in homes; additional sources of PAHs affecting surface waters include atmospheric deposition, oil spills, petroleum processing, municipal effluents and urban runoff (ATSDR, 1995).
4.1 Environmental fate
The chemical properties of BaP strongly favour its sorption onto particles, soil and sediment (Mackay and Paterson, 1991). As such, approximately 82% of BaP released in the environment is estimated to partition into soil, 17% into sediment, 1% into water and < 1% into air (Hattemer-Frey and Travis, 1991). The evaporation half-life of BaP in water is estimated to be 43 days, assuming a depth of 1 m, a flow rate of 1 m/s and a wind velocity of 3 m/s (Irwin, 1997). Adsorption to soil and sediment will limit evaporation. Degradation can also occur by photolysis near the surface as well as by bacterial degradation in natural systems. The half-life in soil can range between 2 days and 1.9 years (U.S. EPA, 2007). BaP is reasonably stable in air and can be transported over considerable distances. Degradation of BaP in air occurs through photolysis and reactions with ozone and nitrogen dioxide.
5.0 Exposure
Food is an important source of exposure to BaP. For non-smokers with no occupational exposure to BaP, it is estimated that 70% of exposure to PAHs, including BaP, is attributable to diet, with cereals and vegetables being the most important sources, except where there is high consumption of meat cooked over an open flame (Phillips, 1999). Exposure through inhalation of combustion products can also be an important source, whereas ingestion of drinking water and soil contribute to a lesser extent to total BaP exposure.
5.1 Water
There is a paucity of information on concentrations of BaP in groundwater. Because of its strong association with soil, BaP is not expected to leach into groundwater. Nevertheless, concentrations as high as 0.32 µg/L have been reported in groundwater in proximity to a wood preserving plant in New Brunswick (WMS et al., 1989).
In the Great Lakes, average concentrations of BaP range from 0.03 to 0.7 ng/L (Government of Canada, 1991).
Concentrations of BaP in Canadian drinking water are generally very low. BaP was generally not detected in drinking water across Ontario (Ontario Ministry of the Environment, 2013), British Columbia (British Columbia Ministry of Health, 2013) or Saskatchewan (Saskatchewan Water Security Agency, 2013). For these provinces, the Method Detection Limits (MDLs) ranged from 0.001 to 1 µg/L and any sample detections were typically at or below the MDL. In Québec, 97,6% of the 6678 samples collected in distribution systems between 2002 and 2012 were below the detection limit. (range of 0.002 - 0.01 µg/L). For the results above the detection limit, the median was 0.004 µg/L, with 16 results at or above 0.04 µg/L (Ministère du Développement durable, de l'Environnement, de la Faune et des Parcs du Québec, 2013). Four results of the 5711 samples taken in New Brunswick (MDL 0.001 - 0.5 µg/L) (New Brunswick Department of Health and Wellness, 2013) and one of 268 samples taken in Nova Scotia (MDL 0.009 - 0.01 µg/L) (Nova Scotia Department of Environment and Labour, 2013) were above 0.04 μg/L.
BaP concentrations in drinking water in the United States have been reported to range from 0 to 1 µg/L (1000 ng/L) (ATSDR, 1995), with typical concentrations around 0.000 55 µg/L (0.55 ng/L) (Santodonato et al., 1981). Kolpin et al. (2002) undertook a nationwide study to assess the occurrence of a variety of pharmaceuticals, hormones and organic contaminants in 139 streams in 30 U.S. states; the authors found BaP in 85 samples, with a maximum concentration of 0.24 µg/L and a median concentration of 0.04 µg/L. International levels of BaP in drinking water range from < 0.04 to 914 ng/L, as reported by the World Health Organization (WHO, 2003).
5.1.1 Leaching from coal tar linings
Concentrations of BaP in treated water can be higher than those in the untreated source water because of the leaching of BaP due to PAH-containing coal tar coatings on drinking water distribution pipes and storage tanks (Basu et al., 1987;WHO, 2003; OEHHA, 2010). Prior to the 1980s, asphaltic or bituminous linings were commonly used as a coating material to protect against corrosion in cast iron or ductile iron, concrete and steel pipes used to convey drinking water and in storage tanks used to store finished drinking water (Basu et al., 1987; WHO, 2003). Although this practice has largely been discontinued, pipes, reservoirs and storage tanks may still be lined with this material and have been shown to significantly increase BaP levels in drinking water (Vu Duc and Huynh, 1981; Basu et al., 1987; OEHHA, 2010). The presence of BaP at high concentrations in drinking water samples is indicative of the presence of particulate matter from the deterioration of coal tar and asphaltic coatings.
A Dutch nationwide study that investigated drinking water samples at 120 locations with cast iron drinking water mains coated with coal tar and bitumen found elevated levels of BaP in drinking water during flushing (up to 22 μg/L), with a rapid drop after flushing (maximum concentration of 2.1 μg/L); when repairs were made to the water mains, PAH levels above 0.01 μg/L, the Dutch drinking water quality standard, were detected for up to 40 days in some of the sampling locations (Blokker et al., 2013).
The extent of leaching of BaP is determined by a number of factors, including water age, hydraulic disturbances and the condition of the pipe or tank coating. Studies of factors affecting the mobilization of PAHs, including BaP, demonstrated that the rapid disturbances in the hydraulic regime (e.g., water hammer, rapid increase in flow velocity) resulting in enhanced contact time of the water with the coal tar lining, especially under stagnant, anaerobic conditions, can significantly increase the leaching of BaP (Maier et al., 2000). The results from this study are presented in greater detail in Section 7.1.6.
5.2 Food
The primary dietary source of BaP is charred meat. Charbroiled steak is estimated to contain on average 9000 ng of BaP per kilogram of meat. Canadian meat, poultry and fish (raw) contain up to 11.0 ng of BaP per kilogram (Government of Canada, 1991). However, BaP is also found in grains, oils, fruits and vegetables. A study of 200 food items available in the United States reported BaP concentrations of up to 0.51 ng/g in non-meat foods, including dairy products, breads, grains, sweets and produce, whereas cooked meat products contained up to 4.86 ng of BaP per gram (Kazerouni et al., 2001). It is estimated that an average 50 kg teenager is exposed to 80 ng of BaP per kilogram of body weight (bw) per day from smoked meat and 380 ng/kg bw per day from canola oil (Shi et al., 2010).
5.3 Air
Most BaP in air is readily adsorbed onto airborne particulate matter, although it can also be present in vapour form. A study of 120 cities in the United States reported concentrations of BaP in air ranging from 0.2 to 19.3 ng/m3 (Pucknat, 1981). Concentrations up to 460 ng/m3 have been detected near an aluminum smelter in Shawinigan, Quebec (Ringuette et al., 1993). Ambient air levels of PAHs in general are highest during colder months, as a result of increased heating (WHO, 1998). PAH concentrations ranging from 1 to 30 ng/m3 have been recorded in various urban cities, but can reach 200 ng/m3 in larger cities with heavy motor vehicle traffic (WHO, 1998). Tobacco smoke, gas cooking and heating appliances are important sources of BaP in indoor air. Concentrations of BaP in cigarette-polluted areas range from 400 to 760 000 ng/m3 (OEHHA, 2010).
5.4 Consumer products
Some consumer products such as coal tar-based shampoos contain BaP. Cigarette smoking constitutes a major source of BaP. Domestic Canadian cigarettes are estimated to contain 6.1 ng BaP per cigarette, whereas imported cigarettes contain approximately 5.7 ng BaP per cigarette (Hammond and O'Connor, 2008). It is estimated that the BaP content in mainstream smoke ranges from 5 to 80 ng per cigarette, with higher levels in sidestream smoke (25-200 ng BaP per cigarette; OEHHA, 2010).
5.5 Soil
BaP can accumulate in soil due to its hydrophobic properties. Typical background concentrations of BaP in Ontario soils are estimated to be 0.30 and 0.039 mg/kg in urban and rural parkland soils, respectively (CCME, 2010). In New England, background soil BaP concentrations were found to range from 0.04 to 13 mg/kg (Bradley et al., 1994). However, soil concentrations of BaP can be considerably higher at or near industrial sites. For example, BaP concentrations as high as 1300 mg/kg have been found at the site of a former coking facility in Lasalle, Quebec (CCME, 2010), and WHO (1998) reported levels as high as 1000 mg/kg.
5.6 Multi-route exposure from drinking water
The multi-route exposure approach for drinking water put forth by Krishnan and Carrier (2008) is not suitable for high molecular weight compounds like BaP. However, a significant contribution of dermal exposure to total exposure from drinking water is unlikely, as BaP is a hydrophobic molecule typically associated with soil particles, with limited absorption and distribution to systemic tissues following dermal exposure (see Section 8.1 and 8.2). Moreover, BaP is not readily volatile; thus, exposure through inhalation during bathing and showering is considered negligible. Thus, the contribution of dermal and inhalation exposure to overall exposure is assumed to be negligible.
6.0 Analytical methods
The U.S. Environmental Protection Agency (EPA) currently has three approved analytical methods (Method 525.2, Method 550 and Method 550.1) for the analysis of BaP in drinking water (U.S. EPA, 2009a). These methods are general methods for the determination of synthetic organic compounds and use either a liquid-liquid extraction (LLE) or liquid-solid extraction (LSE) followed by gas chromatography/mass spectrometry (GC/MS) or high-performance liquid chromatography (HPLC) to separate the analytes:
- Method 525.2 uses an LSE method followed by capillary column GC/MS. The method detection limit (MDL) values range from 0.032 to 0.23 µg/L (U.S. EPA, 1995, 2003). Originally, the U.S. EPA had approved Method 525.1, but it was replaced by Method 525.2, which is more sensitive, in 1995 (U.S. EPA, 2003, 2009a).
- Method 550 uses LLE and HPLC equipped with ultraviolet (UV) and fluorescence detection. This method has an MDL of 0.029 µg/L (U.S. EPA, 1990, 2009a).
- Method 550.1 uses LSE and HPLC equipped with UV and fluorescence detection. This method has an MDL of 0.016 µg/L (U.S. EPA, 1990, 2009a).
Standard Method 6440B can also be used for the analysis of BaP in drinking water (APHA et al., 2012). This method is based on LLE and HPLC and has an MDL of 0.02 µg/L.
The practical quantitation level (PQL) for U.S. EPA-approved methods is 0.0002 mg/L (0.2 µg/L) and is based on the capability of laboratories to measure BaP within reasonable limits of precision and accuracy at the time of regulation (U.S. EPA, 1992). Current PQL assessments are based on two approaches: (1) the lowest value for which 75% of laboratories can quantitate within prescribed accuracy limits based on actual performance data, if the data are sufficient; or (2) multiplying the upper levels of the MDLs to account for the variability inherent to test methods and instruments used for analyses, when data are insufficient. In establishing the PQL, the U.S. EPA considers and prefers the laboratory performance data for methods approved at the time of the review over the MDL approach.
In both the first and second Six-Year Review of existing National Primary Drinking Water Regulations, the U.S. EPA determined that it could not lower the PQL for BaP (U.S. EPA, 2003, 2009a, 2009b). In the analysis of the first and second Six-Year Review data sets, it was determined that there was a lack of data below the current PQL and that the variability of the second Six-Year Review's proficiency test results did not support reducing the PQL (U.S. EPA, 2009b).
The U.S. EPA used two other approaches to determine if an estimated quantitation level (EQL) below the current PQL could be identified: a minimum reporting level (MRL) approach and an MDL multiplier approach based on the approved analytical methods. The EQL is an estimate of the possible lower bound for a PQL. It is based on statistical analysis of proficiency test results for a method or the MRL from the data set. The MRLs represent laboratory analytical limits for a large pool of laboratories in the United States. Using the MRLs of more than 55 487 records collected from 1998 to 2005, it was determined that only 55.4% of these values were less than or equal to the modal MRL of 0.000 02 mg/L. As the criterion of 80% of MRL values being equal to or less than the modal MRL was not met, the U.S. EPA did not base the EQL on the modal MRL (U.S. EPA, 2009b). Application of a multiplier of 10 to the median MDL of the three approved analytical methods (Methods 525.2, 550 and 550.1) resulted in a possible PQL of 0.0003 mg/L. As this value was higher than the current PQL, the U.S. EPA determined that there was no potential to lower the PQL for BaP; the PQL remains 0.0002 mg/L (0.2 μg/L; U.S. EPA, 2009b).
There is no equivalent centralized program for the collection and rigorous statistical analysis of analytical data in Canada. Although establishing a PQL for Canadian laboratories is not possible, lower quantification levels may be achievable by some laboratories.
As part of the guideline review process, a number of commercial laboratories were surveyed by several provinces in order to determine the MDLs and minimum reporting levels that were achievable for the analysis of BaP. The MDLs provided from 15 commercial laboratories ranged from 0.001 µg/L to 1.9 µg/L (n>850 samples) using a variety of analytical methods including EPA Methods 525.2; 846; 3510; 3541 and 8270; and SM6410B. The minimum reporting levels for BaP (equivalent to the MRL described above) provided by these laboratories ranged from 0.001 to 6.5 µg/L. Of the 12 minimum reporting levels provided, 11 of the values were equal to or less than 0.01 µg/L.
Based on the limited data above, analytical methods are available to reliably measure concentrations of BaP equal to or less than 0.01 µg/L with the analytical methods currently in use. As a result, analytical methods are not expected to be a limiting factor for the MAC for BaP. Responsible authorities should establish performance measures when specifying which analytical method to use (i.e. sample volume, sampling requirements, sample preservation and storage and MDL).
7.0 Treatment technology and distribution system considerations
7.1 Municipal scale
Municipal water filtration plants that rely on conventional treatment techniques (coagulation, sedimentation, filtration and chlorination) can reduce concentrations of PAHs, including BaP, in drinking water sources (Sorrell et al., 1980; Westerhoff et al., 2005; Snyder et al., 2007). As a result of its hydrophobic nature, BaP in raw water will adsorb to particulate matter and can subsequently be removed by conventional treatment processes (Sorrell et al., 1980). Limited full-scale data exist on the removal of BaP from drinking water using conventional treatment. However, the data indicate that removal of BaP to levels below 0.01 µg/L can be achieved using conventional treatment.
The U.S. EPA lists granular activated carbon (GAC) as the Best Available Technology (BAT) for the removal of BaP from drinking water to achieve concentrations below the Maximum Contaminant Level of 0.0002 mg/L (0.2 µg/L) (U.S. EPA, 1992). Small system compliance technologies for the removal of BaP from drinking water include GAC, point-of-use GAC and powder activated carbon (PAC) (U.S. EPA, 1992). The available data indicate that removal of BaP to levels well below 0.01 µg/L can be achieved using GAC or PAC.
BaP is primarily of concern as a potential contaminant from cast iron or ductile iron, concrete and steel pipes lined with asphaltic or coal tar coatings, mainly installed prior to the 1980s (Basu et al., 1987). BaP leaching from pipes and storage tanks can be addressed by replacing the pipes and storage tanks, limiting changes in the hydraulic regime, avoiding severe flow disturbances (i.e., water hammer) and reducing the residence time of water in the distribution system (Maier et al., 2000). Managing hydraulic fluctuations to minimize biofilm sloughing or scale release will also help to minimize the release of BaP. In general, distribution system operations designed to promote water quality and minimize water age will also help to minimize the release of BaP.
7.1.1 Conventional coagulation/filtration and lime softening processes
In a review of the efficiency of PAH removal by full-scale treatment plants, Sorrell et al. (1980) found that BaP concentrations appeared to be substantially reduced by conventional treatment (i.e., coagulation/sedimentation/filtration) followed by chlorination. Effective removal appeared to be closely related to particulate removal; consequently, it was concluded that conventional treatment was generally effective for the removal of BaP. However, it was also noted that some of the studies indicated the use of activated carbon in their conventional treatment processes, which could account for an increased removal of BaP. The authors found that when BaP was detected (raw water concentrations ranging from 4-13 ng/L), it was reduced to concentrations below 1 ng/L (75-92% removal) at all sites except Jefferson Parish, LA, where the concentration was less than 2 ng/L in the finished water.
Snyder et al. (2007) investigated a range of endocrine-disrupting compounds, pharmaceuticals and personal care products, including BaP, to assess their removal by drinking water and water reuse treatment processes. Concentrations of BaP were analyzed using gas chromatography with tandem mass spectrometry (GC/MS/MS). The reporting limit was 10 ng/L, with an MDL of 2.35 ng/L and an instrument detection limit of 4.3 pg/L (Trenholm et al., 2006). All 20 full-scale drinking water treatment plants had influent concentrations of BaP lower than the reporting limit of 10 ng/L (Snyder et al., 2007). Only 13 of the 20 treatment plants used coagulation with lime softening. The authors found that the combination of coagulation with alum or ferric chloride and lime softening was capable of removing 50-80% of BaP, but did not specify the number of plants or the influent concentrations. Very limited data were provided specifically for BaP removal in this study.
A bench-scale study of conventional drinking water treatment processes, using three natural waters of varying quality and a synthetic (model) water spiked with a natural organic matter (NOM) isolate, was conducted at a temperature of 20°C, pH 5.5 and pH 8.5, alkalinity of 38 mg/L as calcium carbonate (CaCO3) and a dissolved organic carbon concentration of 3.5 mg/L (Westerhoff et al., 2005; Snyder et al., 2007). Conventional treatment was simulated by conducting 1 minute of rapid mix followed by 20 minutes of flocculation and 60 minutes of settling. Coagulation was achieved using alum (dose of 30, 35 or 40 mg/L) or ferric chloride (doses ranging from 8.3 to 38 mg/L). Lime softening used calcium hydroxide at doses ranging from 53 to 238 mg/L and soda ash at doses ranging from 0 to 177 mg/L, to a final pH of 11.3. Initial BaP concentrations ranged from 39 to 115 ng/L. Treated water concentrations achieved with alum coagulation were calculated to range between 10 and 34 ng/L (removal of 68% to > 74%) for the natural waters and between 10 and 51 ng/L (removal of 43-91%) for the synthetic water. Calculated treated water concentrations for ferric coagulation were similar, ranging from 10 to 33 ng/L (67-80% removal) for the natural waters and reaching 34 ng/L (57% removal) for the synthetic water. Calculated lime softening concentrations were also comparable, ranging from 10 to 11 ng/L (> 74% to > 90% removal) for the natural waters and reaching 46 ng/L (47% removal) for the synthetic water. In conclusion, conventional treatment was capable of reducing BaP concentrations of up to 105 ng/L to less than 34 ng/L in natural waters at bench scale. Removal of BaP in synthetic water appeared to be less effective at higher pH. Overall, alum achieved an average 72% removal of BaP (Westerhoff et al., 2005). Ferric chloride achieved 74% removal of BaP; ferric chloride was not significantly different from lime softening, which achieved BaP removals of 82% (Snyder et al., 2007).
7.1.2 Activated carbon
Activated carbon is used in water treatment processes either as GAC or as PAC. The adsorption capacity of activated carbon to remove organic compounds is affected by a variety of factors, including competition from other contaminants, NOM, temperature and the physical/chemical properties of the organic compounds and the carbon media (Speth, 1990).
7.1.2.1 Granular activated carbon
GAC is the most widely used technology for the removal of BaP in small water treatment systems because of its simplicity, ease of operation, practicality and affordability (Snoeyink, 1990; U.S. EPA, 1998). In the GAC process, the contaminants diffuse into the adsorbent granules and accumulate on the inner surface within the pores as water passes through the GAC contactor.
Early studies demonstrated the effectiveness of activated carbon in removing PAHs, such as BaP. Basu and Saxena (1978) reported concentrations of BaP ranging from 0.3-42.1 ng/L in surface waters used as drinking water sources in four U.S. cities (Huntington, WV; Buffalo, NY; and Pittsburgh and Philadelphia, PA). These four full-scale water treatment plants included the addition of a coagulant, activated carbon (GAC, PAC or both) and chlorine in the treatment train. In addition, three of the treatment plants used lime. No operational details on the various processes (e.g., coagulant, carbon or chlorine dosage, contact time) were provided. Results for the full-scale water treatment plant samples at these four sites showed finished water BaP concentrations ranging from 0.2 to 0.5 ng/L (68% to > 99% removal) using conventional treatment and GAC. It was noted that the site with the lowest removal rate (Buffalo, NY) also had the lowest influent concentration of BaP and did not appear to use lime in the treatment process. In a review of full-scale studies to remove PAHs, Sorrell et al. (1980) found that the use of activated carbon for the removal of low concentrations of PAHs, such as BaP, may not be appropriate, as removal effectiveness may be reduced at lower concentrations.
Stackelberg et al. (2004) studied a variety of compounds, including BaP, in the source, filtered and finished water of a 62 MGallon/day (235 ML/day) drinking water treatment plant that used conventional treatment, PAC (for taste and odour) and GAC (contact time of 1.5-3 minutes). BaP was analyzed by continuous LLE GC/MS, and the reporting level was 0.5 µg/L (detection limits were not specified). The authors reported that BaP was present in 42% of the 12 samples collected from the streams feeding the treatment plant and the sampling points in the raw water supply of the plant. The concentrations of BaP in these samples were estimated graphically to range between 0.04 and 0.09 µg/L, but the authors did not detect BaP in the finished water (detection limits were not specified).
In a follow-up study, Stackelberg et al. (2007) analyzed a variety of compounds, including BaP, in the source, clarified, filtered and finished water of the same treatment plant as in the 2004 study. Although the treatment plant still used conventional treatment and GAC, modifications were made prior to the 2007 study: PAC addition was discontinued; clarification (flocculation/sedimentation) was enhanced using microsand; primary disinfection was moved after clarification; and 9% of the filter backwash decant water was recycled to the head of the plant. The authors did not detect BaP in the source, clarified, filtered or finished water. However, elevated concentrations of BaP were detected in the dried solids of the sludge from the sedimentation tank (48.5 µg/kg) and the filter backwash sediments from the GAC filters (15 µg/kg). A decrease in the BaP concentration of 69% (w/w) was seen in the solids from the sedimentation to the filtration step. The authors attributed the presence of BaP in the solids without a concurrent presence of BaP in the water to temporal variations in sampling. That is, the presence of BaP in the source water and its subsequent removal from the source water occurred during a period when the water was not being sampled for the study.
7.1.2.2 Powder activated carbon
In the same bench-scale study described in Section 7.1.1, Westerhoff et al. (2005) examined the effectiveness of PAC for the removal of BaP using a dose of 5 mg/L and a contact time of 4 hours. The source water contained a dissolved organic carbon concentration of 3.5 mg/L, with an alkalinity of 128 mg/L as CaCO3 and an initial pH of 7.9. Concentrations of BaP were analyzed using GC/MS/MS. The reporting limit was 10 ng/L, with an MDL of 2.35 ng/L and an instrument detection limit of 4.3 ng/L (Trenholm et al., 2006). Initial BaP concentrations ranged from 66 to 155 ng/L, and treated water concentrations achieved with PAC were calculated to range between 9 and 36 ng/L (removal from 69% to > 94%) for all waters. Under these conditions, the average initial BaP concentration of 110 ng/L was reduced by 83% for all four sample waters (calculated finished water concentration of 30 ng/L) (Westerhoff et al., 2005; Snyder et al., 2007). It was also determined that removal of compounds such as BaP by PAC was independent of the initial concentration of BaP, but removal could be improved by increasing the PAC dosage (Westerhoff et al., 2005; Snyder et al., 2007). The study also found that the Kow value was a reasonable indicator of removal efficiency under controlled PAC test conditions for many of the compounds (Westerhoff et al., 2005).
7.1.3 Oxidation processes
The formation of by-products, including disinfection by-products, from the oxidation of BaP or other organic compounds in the source water, should be considered when using oxidation processes. The types and concentrations of by-products formed will be dependent on the source water quality, concentrations and types of oxidants and reaction contact time. For example, advanced oxidation processes (AOPs) such as ozone/hydrogen peroxide and ozone/UV can produce bromate in waters containing bromide ion (Fronk, 1987; Crittenden et al., 2005). The presence of by-products may require additional treatment following AOPs and/or process optimization to minimize by-product formation.
7.1.3.1 Chlorination/chloramination and mixed oxidants
In bench-scale investigations of BaP removal undertaken by Westerhoff et al. (2005), enough chlorine was dosed to establish a chlorine residual of 1 mg/L after 24 hours. The residual chlorine was quenched with ascorbic acid after sampling to prevent further reaction. Chlorine doses ranging from 2.5 to 6.75 mg/L at pH 5.5 in filtered water oxidized BaP concentrations between 30 and 115 ng/L at rates above 75%. Oxidation rates were above 80% when ambient pH was 7.0 or higher for a chlorine dose of 3 mg/L. The study also investigated the differences between chlorine and chloramine under identical water quality conditions at BaP concentrations of 83-95 ng/L. Chloramine doses of 2 and 3 mg/L were evaluated, and samples were collected after 24 and 168 hrs of contact time to simulate distribution system conditions (Westerhoff et al., 2005; Snyder et al., 2007). Oxidation of BaP clearly increased with chloramine dose and contact time, with oxidation rates ranging from 65% to > 89% (calculated final concentration range of 15-33 ng/L).
The same study (Westerhoff et al., 2005; Snyder et al., 2007) used mixed oxidants and household bleach to generate free and combined chlorine mixtures. Mixed oxidants may contain additional oxidants (e.g., hydroxyl radicals), and the study was conducted simultaneously with chlorine for comparison. Chlorine doses of 2 and 3 mg/L were added to a BaP concentration of 97 ng/L for 24 hours of contact time, to simulate distribution system conditions. Mixed oxidants at doses ranging from 5 to 20 mL/L for up to 20 minutes of contact time were not effective at oxidizing BaP, achieving only 30% or less oxidation.
7.1.3.2 Ozonation and advanced oxidation processes
AOPs refer to the use of combinations of chemical oxidants (e.g., ozone), UV light and catalysts (e.g., hydrogen peroxide) to generate highly reactive radicals, such as hydroxyl radicals, which are strong oxidants and react rapidly and non-selectively with organic contaminants. Physical and chemical properties of the water to be treated, such as alkalinity, pH, NOM, iron and manganese, and turbidity, have a major impact on AOPs as these processes scavenge hydroxyl radicals or absorb UV light used to produce the radicals (Crittenden et al., 2005).
Butković et al. (1983) determined that the reaction of BaP with an excess of ozone in distilled water followed first-order reaction kinetics. The half-life of an initial BaP concentration of 18 µg/L in the presence of 10−4 M ozone at pH 7 was 1 second. Based on this half-life, the authors estimated that ozonation of drinking water with a dose of 1 mg/L would completely oxidize and ultimately destroy BaP. A recent study on the reaction kinetics of BaP in ozonation processes supported this finding (Jin et al., 2012).
A bench-scale study by Yerushalmi et al. (2006) using ozone at a concentration of 0.5 or 2.5 mg/L and biodegradation either sequentially (with ozone as a pre-treatment) or simultaneously found that increasing the contact time from 10 to 60 minutes improved the oxidation of BaP. At 0.5 mg/L, little difference was seen when the dose was applied either sequentially or simultaneously, regardless of the contact time for the applied dose. However, a dose of 2.5 mg/L significantly improved BaP removal from 26% for a contact time of 20 minutes to 63% for a contact time of 60 minutes.
Snyder et al. (2007) examined the effectiveness of ozonation for BaP oxidation. The average removal of BaP in the four sample waters tested exceeded 80% when filtered water was ozonated with an ozone dose of 2.5-8.0 mg/L, based on a target residual ozone concentration of 0.2-0.3 mg/L present after 3 minutes and no residual present after 10 minutes (equivalent to a disinfectant concentration-time value for primary disinfection of viruses or Giardia).
7.1.3.3 UV and advanced oxidation processes
In a bench-scale study by Snyder et al. (2007), UV and UV with hydrogen peroxide were evaluated for the decrease of BaP (influent concentration 104-332 ng/L) using a medium-pressure lamp at doses of 40 and 1000 mJ/cm2, in a variety of water qualities. Treatment with UV at a dose of 40 mJ/cm2 (typical disinfection dose) did not achieve any appreciable removal of BaP. However, treatment with UV at 1000 mJ/cm2 resulted in BaP decreases of 63-82%. The use of an AOP (addition of hydrogen peroxide at 5 mg/L prior to UV at 1000 mJ/cm2) did not significantly improve the removal of BaP.
Sanches et al. (2011) tested low-pressure UV lamps for their efficiency at decreasing the BaP concentration in laboratory-grade water, groundwater and surface water. With a UV dose of 1500 mJ/cm2, BaP was efficiently degraded. The highest percent removal was obtained in groundwater (93%), in comparison with laboratory-grade water (79%; influent concentration of 5.6 µM) and surface water (48%). The authors concluded that NOM and dissolved organic material likely impacted the removal efficiency.
7.1.4 Membrane processes
Snyder et al. (2007) undertook a bench-scale study of nanofiltration (NF) and ultrafiltration (UF) membranes for the removal of BaP. Commercially available thin film composite NF and UF membranes were studied using dead-end stirred-cell membrane units, which are effective in assessing relative compound rejection. The membranes were made of ultrathin polyimide coated by either an aromatic polyamide or a sulfonated polyethersulfone. Membrane filtration was performed on three natural waters and one synthetic water, at ambient pH (6.8-8.2) for the natural waters and at pH 7.5 for the synthetic water. An initial volume of 300 mL of the sample was passed through the membrane, and 200 mL of permeate was collected; this was repeated until a total of 1 L of permeate had been collected. Influent concentrations of BaP ranged from 40 to 155 ng/L, and the rejection rates achieved varied between > 75% and > 94% (calculated permeate concentrations of 9-20 ng/L). The rejection rates for BaP did not differ between the NF and UF membranes.
7.1.5 Other technologies
An experimental technology has been shown to have potential for removing PAHs, including BaP, from water, but there is not sufficient information available at present to fully evaluate the technology:
- Inorgano-organo-clay adsorption media: Srinivasan and Folger (1990) compared the performance of inorgano-organo-clays (IOCs) with GAC for the removal of BaP from industrial wastewater. IOCs are clay adsorbents that have been modified using inorganic polycations and cationic surfactants. Based on their earlier work, the authors hypothesized that the surface orientation of the adsorbed organic carbon and the amount of organic adsorbent are both important factors affecting the performance of the sorbent. BaP is hydrophobic and has a relatively low aqueous solubility and therefore is readily adsorbable by the lipophilic surfaces of IOCs.
7.1.6 Distribution system considerations
The main source of BaP in drinking water is usually coal tar linings used in the distribution system rather than the source water (WHO, 2003). Although new pipe is no longer installed with this type of lining, old pipes with coal tar linings remain in service (Maier et al., 2000). Coal tar-lined pipes can be rehabilitated or replaced; the preferred approach will be determined from site-specific conditions (e.g., pipe material, evidence of external corrosion, estimated remaining service life, pipe diameter). Until pipes with coal tar linings can be replaced, there are operational strategies available to manage BaP release in the distribution system.
Maier et al. (2000) investigated the factors that influenced the mobilization of 13 PAHs (including BaP) from coal tar linings of water mains. Extensive monitoring was conducted in 22 water supply zones representing 834 km of pipe installed prior to 1972. This monitoring program indicated that increases in PAH concentrations were influenced by reduced activity of the microbial biofilm on the internal pipe walls, sloughing of biofilm and the related release of particles from encrustations, disturbances of the hydraulic regime and stagnation (retention) time. These factors were further investigated in laboratory, pipe rig and field investigations.
Field investigations in a water supply zone that provided the opportunity to switch from a disinfected to a non-disinfected water supply examined BaP removal. Five experiments were carried out, and the effects of chlorine and chlorine dioxide on the leaching of PAHs were studied at doses ranging from 0.025 to 0.3 mg/L for up to 444 days. Although the authors found that increases in the chlorine and chlorine dioxide doses resulted in increased levels of several PAHs in the distribution system, BaP concentrations were not affected.
A pipe rig study using three 10 m sections of distribution system consisting of cast iron pipe lined with coal tar exposed to three water quality conditions under different flow, disinfection and general hydraulic conditions was undertaken by Maier et al. (2000). Sampling was done at 10 locations in the rig, including before and after the point of addition for the disinfectant. The authors confirmed that PAH concentrations increased with chlorine and chlorine dioxide doses. In addition, they found that reduced oxygen in the distribution system decreased the microbial activity of the biofilm, resulting in increased PAH levels.
Maier et al. (2000) found that increased stagnation time increased PAH levels and turbidity. In addition, the authors showed that a pH below 5 also significantly increased PAH concentrations in the distribution system. Hydraulic disturbances increased the turbidity, with concurrent increased concentrations of BaP (68 ng/L) and other PAHs. Filtration of the turbidity resulted in BaP concentrations below the analytical detection limit.
Blokker et al. (2013) measured PAHs, including BaP, under different operational conditions to help identify those that were associated with increases in PAH concentrations in drinking water. The authors sampled drinking water at 120 locations during undisturbed operations, during and after flushing of pipes and after removal of a section of pipe. The samples were collected from each location 8 times over a 2.5-week period. Of the samples collected, 49% of the low-velocity flush and 61% of the high-velocity flush hydrant samples contained BaP at concentrations exceeding 0.01 µg/L. The maximum BaP concentrations for each operational condition ranged from 0.003 to 22 µg/L. The lowest concentrations were noted in the undisturbed (normal operations) sample locations, and the highest concentrations were seen in hydrant samples during flushing. The concentrations decreased rapidly after flushing. High PAH levels were also seen in samples collected after water main repairs, and it took up to 40 days after the repairs for the levels to decrease below the standard of 0.01 μg/L. The authors found that PAH levels were not correlated with turbidity or with flushing velocities.
In the laboratory experiments conducted by Maier et al. (2000), stainless steel coupons coated with coal tar were exposed to drinking water for 4-32 weeks. The coal tar surface represented the substrate for the growth of biofilm. These coupons were subsequently studied in batch experiments to investigate the rate of leaching over 24 hours under various conditions. The authors found that biofilm had a protective effect and that significantly lower levels of PAH leaching were observed when biofilm was present on the coupons. Based on their findings, the authors recommended strategies for reducing the concentration of PAHs, including BaP, in drinking water originating from the distribution system. Maintaining disinfectant doses within the range required for their application, avoiding water hammer and changes in the hydraulic regime and reducing residence time/stagnation of water in the distribution system will help keep PAH concentrations from increasing. Distribution system flushing should be conducted in a systematic way to ensure that all particles are removed (Vreeburg, 2010). This is supported by the findings of Maier et al. (2000) and Blokker et al. (2013) on the presence of particulates containing PAHs during disturbances and flushing.
Materials currently used in the distribution system that meet NSF International/American National Standards Institute (NSF/ANSI) Standard 61 (NSF/ANSI, 2013) would be expected to leach very small amounts of BaP into drinking water. In order for drinking water materials to be certified to this standard, they cannot leach more than 0.02 µg/L BaP into water. This acceptance criterion is the single product allowable concentration, which is derived using the contaminant regulatory values from the U.S. EPA (one tenth of the regulatory value of 0.2 µg/L) (NSF/ANSI, 2013).
7.2 Residential scale
It is not generally recommended that residential drinking water treatment devices be used to provide additional treatment to municipally treated water. However, the migration of BaP from distribution system pipes lined with coal tar coatings was reported to be a possible source of BaP in drinking water distribution systems. In such cases, as well as where an individual household obtains its drinking water from a well, a private residential drinking water treatment device may be an option for reducing BaP concentrations in drinking water.
Health Canada does not recommend specific brands of drinking water treatment devices, but it strongly recommends that consumers use devices that have been certified by an accredited certification body as meeting the appropriate NSF/ANSI drinking water treatment unit standards. These standards have been designed to safeguard drinking water by helping to ensure the material safety and performance of products that come into contact with drinking water. Certification organizations provide assurance that a product conforms to applicable standards and must be accredited by the Standards Council of Canada (SCC). In Canada, the following organizations have been accredited by the SCC to certify drinking water devices and materials as meeting NSF/ANSI standards (SCC, 2015):
- CSA International (www.csa-international.org);
- NSF International (www.nsf.org);
- Water Quality Association (www.wqa.org);
- Underwriters Laboratories Inc. (www.ul.com);
- Bureau de normalisation du Québec (www.bnq.qc.ca); and
- International Association of Plumbing & Mechanical Officials (www.iapmo.org).
An up-to-date list of accredited certification organizations can be obtained from the SCC (2015).
There is little information in the published literature regarding the effectiveness of GAC treatment technology at the residential scale, and no certified residential treatment devices are currently available for the removal of BaP in drinking water. Carbon filters may be installed at the faucet (point of use) or at the location where water enters the home (point of entry). Some point-of-entry or point-of-use treatment technologies such as activated carbon may remove BaP from the drinking water below the MAC. No drinking water treatment device is certified specifically for BaP removal at this time, as BaP is not currently included in any of the NSF/ANSI drinking water treatment unit standards.
Before a treatment device is installed, the water should be tested to determine general water chemistry and to verify the concentration of BaP. Periodic testing by an accredited laboratory should be conducted on both the water entering the treatment device and the finished water to verify that the treatment device is effective. Products that use adsorption technology can lose removal capacity through usage and time and need to be maintained and/or replaced. Consumers should verify the expected longevity of the adsorption media in their treatment device as per the manufacturer's recommendations and service it when required.
8.0 Kinetics and metabolism
8.1 Absorption
In rats, fecal analysis revealed that oral administration of BaP at doses above 250 mg/kg bw per day through food resulted in approximately 60% BaP uptake (Chang, 1943). Absorption of BaP from the gastrointestinal tract, as assessed through BaP fluorescence in the walls of the glandular stomach of mice and cats, appears to be facilitated by its solubilization by bile salts (Ermala et al., 1951). The oral bioavailability of BaP in rats has been estimated to be 10% (Foth et al., 1988) or 40% (Ramesh et al., 2001b), depending on the BaP dose administered (3 µg/kg bw or 100 mg/kg bw, respectively). Discrepancies in the amount of bioavailable BaP may be due to the higher doses of BaP employed in the latter study, which may impair extraction of BaP by gastrointestinal fluids (Ramesh et al., 2001b). BaP absorption following oral administration is also influenced by the oil and fat content of the gastrointestinal tract (ATSDR, 1995) and is enhanced by solvents containing both hydrophilic and hydrophobic constituents (Ekwall et al., 1951). This non-linear relationship between dose and bioavailability of BaP, as well as differences in the vehicle employed, will affect the absorption of BaP following oral exposure.
Approximately 90% of inhaled radioactively labelled BaP was cleared from the lungs of Fischer 344 rats within 30 minutes (Sun et al., 1982). Inhalation of BaP by laboratory animals can lead to rapid absorption from the lungs into the systemic circulation. Maximal BaP concentrations in plasma were achieved 1 hour after nose-only inhalational exposure of rats to BaP–carbon black aerosol (0, 0.1, 1.0 and 2.5 mg/m3 for 4 hours); only trace amounts were detectable 5 hours later (Ramesh et al., 2001a). Similarly, following intratracheal instillation of 3H-labelled BaP dissolved in triethylene glycol at a dose of 1 μg/kg bw in rats, approximately 40% of the BaP was cleared from the lungs within 5 minutes of exposure; only about 5% of the initial dose remained 6 hours later (Weyand and Bevan, 1986). Thus, BaP is rapidly absorbed following inhalation. As has been observed following oral administration, vehicle is an important factor governing the absorption of BaP following inhalation exposure; this includes whether the BaP is associated with particles and the size of the particles (Creasia et al., 1976).
In vitro dermal exposure to BaP using skin samples of mice, rats, rabbits, guinea pigs, marmosets and humans results in absorption levels ranging from 0.1% to 10%. Induction of drug-metabolizing enzymes in the skin increases the permeation of topically applied BaP, suggesting that BaP metabolism is important for its dermal absorption (Kao et al., 1985). The vehicle in which BaP is delivered is another important factor in determining the dermal absorption of BaP; in rhesus monkeys, BaP absorption was 51 ± 22% when applied in acetone and 13.2 ± 3.4% when applied in soil (Wester et al., 1990). In vitro experiments with the skin of cadavers mounted over human serum receptor fluid showed that approximately 1.4% of the total dose applied permeated the skin and 0.01% entered the serum receptor fluid when BaP was applied in soil; when acetone was used as the vehicle, approximately 24% and 0.1% of the total applied dose were detected in the skin and serum, respectively (Wester et al., 1990).
8.2 Distribution
Oral administration of BaP can lead to rapid distribution to blood, liver and other organs. For example, rats given an oral BaP dose of 100 mg/kg bw exhibited the highest amount of BaP in serum (~67% of the total, unmetabolized dose) 8 hours post-exposure. Liver showed a biphasic pattern of BaP retention, with peaks at approximately 10% of the total metabolized dose 2 and 24 hours after exposure (Ramesh et al., 2001b). Similarly, BaP was distributed rapidly to the circulation in orally exposed rats, exhibiting measurable levels within 1 hour of administration (Ramesh et al., 2002).
Experimental animals exposed to BaP by inhalation exhibited elevated BaP levels in the lungs shortly after exposure, followed by distribution to various organs via the systemic circulation. Following intratracheal instillation of radiolabelled BaP in rats, the majority of the administered dose was distributed in the lungs (59.5%), carcass (14.4%) and liver (12.5%) within 5 minutes of the initial exposure. Six hours following exposure, the intestine contained the majority of BaP (45% in the contents and 15% in the tissue), followed by the carcass (21.5%), stomach (5.8%), lungs (5.2%), liver (4.6%) and kidneys (2.0%). Blood contained about 4% of the total dose in the first 15 minutes and approximately 1-2% at 0.5-6 hours post-exposure (Weyand and Bevan, 1986). The nature of the vehicle (i.e., whether BaP was administered adhered to particles or in its vaporized form) was an important factor in the distribution of BaP following inhalation (Creasia et al., 1976).
Following topical application of 3H-labelled BaP at a dose of 50 mg/kg bw in mice, BaP was detected primarily in skin, with negligible amounts also found in the liver, lungs and stomach (Carlson et al., 1986). Moreover, BaP was detected in the urine and white blood cells of patients topically treated with coal tar ointment (Godschalk et al., 1998). Approximately 13-51% of administered BaP, depending on the vehicle used, penetrated the skin of rhesus monkeys, and 1-24% penetrated human skin (Wester et al., 1990). These findings suggest that BaP can penetrate, but mostly remains within, skin.
The systemic distribution of BaP following intravenous exposure has been examined. Five minutes following exposure of rats to 14C-labelled BaP at a dose of 15 mg/kg bw, the lungs contained the highest amount of radioactivity, followed by liver, heart, kidney, blood, brain, spleen, testes and adipose tissue (Moir et al., 1998). A more recent study (Marie et al., 2010) confirmed BaP distribution to various organs and tissues in rats following intravenous administration. Two hours after administration, the largest proportion of the administered BaP was found in the lungs (17.0%), followed by skin (7.6%), adipose tissue (2.5%), liver (2.3%) and kidney (0.5%).
Increased levels of BaP have also been found in rat fetuses following oral administration of BaP in corn oil to pregnant dams. The highest BaP concentration in fetal tissues was detected 2-3 hours after administration of 200 mg/kg bw to dams, with about 1.5% of the total dose distributed to the fetus (Shendrikova and Aleksandrov, 1974). Another study determined that transplacental transfer of BaP occurs upon maternal BaP inhalation, with metabolites being present in rat pups up to 15 days after their gestational exposure (Wu et al., 2003).
8.3 Metabolism
The metabolism of BaP has been well studied and was reviewed by IARC (2010). The aryl hydrocarbon receptor (AhR) is the principal transcription factor governing the activation of phase I and phase II enzymes. After BaP binds to the AhR, the BaP-AhR complex translocates to the nucleus of the cell, where it dimerizes with the AhR nuclear translocator protein and binds to the promoters of genes containing AhR response elements, thus activating enzyme production (Michaelson et al., 2011). As such, BaP induces its own metabolism by acting as both an inducer and a substrate of metabolic enzymes.
In phase I metabolism, BaP is converted to an epoxide primarily by cytochrome P450 (CYP) enzymes (CYP1A1 and CYP1B1), but also by other enzymes, including prostaglandin synthase (Trush et al., 1982). BaP epoxides undergo hydration by epoxide hydrolase to form diols. Aldo-keto reductases convert BaP diols to catechols (Lan et al., 2004). Secondary epoxidation by CYPs yields diol epoxides. BaP-7,8-diol-9,10-epoxide (BPDE) is the most mutagenic and well-studied BaP diol epoxide, forming covalent adducts with deoxyribonucleic acid (DNA) at the N2 position of deoxyguanosine (Fang et al., 2001). Alternatively, CYPs can also catalyze one-electron oxidation of BaP to form a BaP radical cation (Cavalieri and Rogan, 1992). These metabolites are direct genotoxic compounds that form unstable adducts with purinic bases (Straif et al., 2005). In addition, CYPs can undergo uncoupling of their catalytic reactions, leading to the production of superoxide (·O2−) and other reactive oxygen species, including hydrogen peroxide and hydroxyl radicals (Shertzer et al., 2004).
During phase II metabolism, BaP metabolites are conjugated with hydrophilic moieties (glutathione, glucuronic acid or sulphate) to enhance their solubility in water, thus allowing urinary and fecal excretion (Garg et al., 2008; Meinl et al., 2008; Bock and Bock-Hennig, 2010). Phase II metabolism enzymes include glutathione S-transferases, uridine diphosphate-glucuronosyl transferases and sulphotransferases.
It should be noted that male and female rats appear to metabolize BaP differently; significantly lower amounts of metabolites were detected in feces and urine of females relative to males following oral exposure (Van de Wiel et al., 1993) though it is not know if such sex-specific differences occur in other species. 3-Hydroxybenzo[a]pyrene is the predominant metabolite detected in the feces and urine of rats, constituting approximately 6-12% of the total BaP administered orally (Van de Wiel et al., 1993).
8.4 Excretion
Following ingestion of BaP, fecal excretion has been reported to account for 60-80% of elimination in rats (Foth et al., 1988; Van de Wiel et al., 1993). In comparison, the urinary route is a relatively minor contributor to BaP excretion, accounting for less than 1% of excretion after oral exposure. In rats treated with BaP by intraduodenal infusion, the intestine absorbed about 40% of the compound, and the liver contributed to 40% of the first-pass extraction. Approximately 10% of this original dose is thought to undergo pulmonary extraction (Foth et al., 1988).
Excretion following the inhalation route of exposure is primarily through feces, with some excretion in urine. Nose only inhalation in rats of 4.8 mg/m3 radioactively labelled BaP (single or over 4 weeks) resulted in approximately 96% excretion through feces (Wolff et al., 1989).
BaP is not readily internalized following dermal exposure, and thus excretion is not applicable.
8.5 PBPK models
The currently available physiologically based pharmacokinetic (PBPK) models are not adequately developed to describe the series of animal experiments studying the pharmacokinetics of BaP. Only one publication (Crowell et al., 2011) presents a PBPK model to describe available time series data from the scientific literature on BaP in rats and mice. However, this preliminary PBPK model fails to use appropriate physiological values. In particular, the cardiac output in the model does not match the reference values cited by Brown et al. (1997). The blood flow values in Crowell et al. (2011) are 10-100 times lower than the actual values referred to in Brown et al. (1997). Even after correcting the blood flow values, the PBPK model predictions are not consistent with data presented and validated in Crowell et al. (2011). This inconsistency makes it difficult to confirm the model structure and pharmacokinetic processes suggested by this work.
A publication by Haddad et al. (1998) describes a PBPK model for the pharmacokinetics of pyrene, another PAH. However, extrapolation of that PBPK model to BaP yielded inconsistent results because of the uncertainty related to the tissue binding constants used in the model. A publication by Heredia-Ortiz et al. (2011) presented a toxicokinetic compartmental model to describe the pharmacokinetics of BaP in rats. Although the model is consistent with experimental data on rats, it uses rate constants instead of physiological parameters. These rate constants are consistent with the fitted data only and cannot be extrapolated to other species, such as humans and mice, or to dose conditions beyond those observed in the study.
Further research on pharmacokinetic parameters such as blood to tissue partitioning and model development using data on the pharmacokinetics of BaP from the available literature will be needed to properly relate the BaP exposure dose to internal dose metrics.
9.0 Health effects
9.1 Effects in humans
9.1.1 Acute toxicity
No data relating to acute toxicity were identified in humans.
9.1.2 Subchronic and chronic toxicity and carcinogenicity
The epidemiological data for BaP are limited to co-exposures to other substances, including other PAHs. Oral exposure was examined primarily via consumption of charred meats, whereas exposure via inhalation was assessed through occupational exposures in coke oven, aluminum and iron and steel workers as well as in smokers. In each case, BaP exposure occurred as part of a mixture. Thus, it is not possible to draw firm conclusions regarding the carcinogenicity of BaP or other chronic effects specific to BaP exposure in humans. Although no conclusions can be drawn from a single study, it should be noted that the majority of studies suggested a positive association between BaP exposure by both ingestion and inhalation and cancer in humans.
9.1.2.1 Tumorigenesis and carcinogenesis
Increased incidences of tumours and cancers were the most common correlate of BaP exposure in humans. Exposure through diet was associated with colorectal cancers and adenoma (Butler et al., 2003; Gunter et al., 2005; Sinha et al., 2005a, 2005b; Ferrucci et al., 2012), pancreatic cancer (Anderson et al., 2005; Li et al., 2007) and lung cancer (Tram et al., 2009). Other studies that have investigated the association of oral BaP exposure with prostate cancer, non-Hodgkin's lymphoma, breast cancer and esophageal cancer have reported negative or inconclusive findings (Cross et al., 2005, 2006; Hakami et al., 2008; Fu et al., 2011). The lowest BaP doses at which effects were observed following oral exposure were reported in a study by Anderson et al. (2005). This study investigated heterocyclic amine and BaP intakes in 193 cases of pancreatic cancer and 674 controls according to usual meat intake and meat preparation methods. An increase in cancer incidence was observed, as shown by odds ratios of 2.0 (95% confidence interval [CI] 1.1-3.7) and 2.2 (95% CI 1.2-4.0), which were reported for estimated median BaP doses of 10.4 and 53.7 ng/day, respectively. It should be noted that studies of meat intake involve exposure to various mutagenic and carcinogenic compounds and that BaP concentration estimates are based on recall of past meat consumption, which can lead to biased results. As such, it is difficult to interpret the results of the aforementioned studies because of the lack of precise information on ingested doses.
Studies have also associated BaP inhalation with carcinogenic outcomes. Smokers and occupationally exposed workers were reported to have increased levels of BPDE-DNA adducts, which are specific to BaP exposure (Haugen et al., 1986; Perera et al., 1988; Hemminki et al., 1990; Izzotti et al., 1991; De Flora et al., 1993; Mumford et al., 1993; Pan et al., 1998). The primary carcinogenic effects observed with occupational exposures involving BaP were related to lung cancer (Armstrong et al., 1994; Friesen et al., 2007; Armstrong and Gibbs, 2009; De Stefani et al., 2009). A lung cancer odds ratio of 1.6 (95% CI 1.2-2.3) was associated with cumulative BaP exposure levels of 0.85-1.96 μg/m3·year in a Chinese iron-steel complex, although co-exposure to other PAHs and dust also occurred (Xu et al., 1996). Bladder cancer incidences were also estimated to increase by 2.3% per μg/m3·year assuming a 10-year latency period in aluminum smelter workers exposed to BaP and benzene-soluble matter (Armstrong et al., 1986).
Thus, there is some evidence suggesting that BaP exposure is associated with excess cancer risk.
9.1.3 Reproductive and developmental toxicity
There is some information that suggests an association between exposure to PAHs and adverse reproductive and developmental effects. One study examined dietary exposure to BaP, among other PAHs, during pregnancy and reported a significant association with reductions in birth weight and length. However, these effects were significant only in women with low dietary intake of vitamin C (Duarte-Salles et al., 2012). Studies of inhalation exposure in pregnant women near a coal-fired power plant and near fires that occurred at the World Trade Center have associated cord blood BaP-DNA adducts with reduced fetal weight (Perera et al., 2005; Tang et al., 2006), whereas no significant effects on birth outcome were noted in a study of inner-city minorities exposed to BaP and other PAHs (Perera et al., 2004).
9.2 Effects on experimental animals
There is a large database of information on animal responses to BaP. As such, primarily studies with adequate dose-response information are reported below.
9.2.1 Acute toxicity
Little information regarding acute toxicity in animals was available. The acute oral toxicity of BaP is considered to be fairly low, with a median lethal dose (LD50) in excess of 1600 mg/kg bw in mice (Awogi and Sato, 1989). Intraperitoneal injection of BaP in mice resulted in an LD50 of 232 mg/kg bw (Salamone, 1981). In rats, subcutaneous injection resulted in an LD50 of 50 mg/kg bw (RTECS, 1994).
9.2.2 Short-term exposure
9.2.2.1 Renal and hepatic toxicity
Increased liver weights were observed in male and female rats gavaged with BaP at a dose of 30 mg/kg bw per day in both sexes as well as 10 mg/kg bw in males (5 days/week, for 90 days) (Kroese et al., 2001) and in male rats exposed to 90 mg/kg bw per day (5 days/week, for 35 weeks) (De Jong et al., 1999). A broad dose-response study was performed on male and female F344 rats given BaP in their diets at a dose of 0, 5, 50 or 100 mg/kg bw per day for 90 days. A significant increase in the liver to body weight ratio was observed in the high-dose males (100 mg/kg bw per day), but not in females at any dose (Knuckles et al., 2001).
In a study in which male and female F344 rats were given BaP in their diet at 0, 5, 50 or 100 mg/kg bw per day for 90 days, an increase in tubular casts in the kidneys of males was observed at the middle and high doses, but no effects were observed in females at any dose (Knuckles et al., 2001).
Thus, the overall evidence for an impact of BaP on hepatic and renal function is limited.
9.2.2.2 Cardiovascular toxicity
Two studies have examined susceptibility to atherosclerosis following exposure to BaP. An increase in mean atherosclerotic lesion size in the aortic tree and root was observed in atherosclerosis-prone (apoliprotein E [ApoE]-null) mice gavaged with BaP at a dose of 2.5 mg/kg bw per week for 24 weeks compared with olive oil controls (Yang et al., 2009). Atherosclerosis was also observed in ApoE-null mice gavaged with BaP at 5 mg/kg bw twice in 1 week compared with tricaprylin controls (Knaapen et al., 2007).
Additional cardiovascular effects observed included increased heart weight and markers of hypertrophy. Increases in heart to body weight ratio and markers of hypertrophy were observed in Sprague-Dawley rats injected intraperitoneally with BaP at 20 mg/kg bw per day for 7 days compared with corn oil controls (Aboutabl et al., 2009, 2011). Adverse cardiovascular effects of BaP may also be transmitted to progeny.One study suggests that in utero exposure to BaP at 0.6 or 1.2 mg/kg bw on embryonic days 14-17 (through gavage of pregnant Long-Evans hooded rats) predisposes offspring to functional deficits in cardiovascular development and cardiac dysfunction later in life (Jules et al., 2012).
9.2.2.3 Neurotoxicity
The primary neurological effects observed following BaP exposure pertain to impaired spatial learning and memory. Xia et al. (2011) dosed Sprague-Dawley rats with BaP (0, 1.0, 2.5 or 6.25 mg/kg bw per day) daily by intraperitoneal injection for 13 weeks and then subjected the animals to testing in the Morris water maze. Delayed performance of BaP-treated rats was evident at 2.5 mg/kg bw per day, with no significant effects found at the 1.0 mg/kg bw per day dose. This study further demonstrated that various other endpoints, including neurotransmitter levels and oxidative stress parameters in the hippocampus, were also sensitive to BaP, even at the lowest dose. Other neurobehavioural effects reported following BaP exposure included aggressive behaviour and increased sexual activity (Bouayed et al., 2012), reduced anxiety (Grova et al., 2008; Bouayed et al., 2012) and impaired motor activity (Saunders et al., 2006).
9.2.2.4 Immunotoxicity
Exposure to BaP through various routes has been associated with immunosuppression, as shown by reductions in antibody production and thymus weight, inhibited lymphocytic responses and other immunological alterations.
Thymus weight was reduced with a dose-response trend in rats exposed by oral gavage following a 3-month exposure, with significant findings occurring at BaP doses exceeding 15 g/kg bw in males and 50 g/kg bw in females (Kroese et al., 2001). Reductions in antibody production have been demonstrated in several studies. Adult B6C3F1 mice exposed to 10 daily subcutaneous injections of BaP in corn oil (0, 50, 200 or 400 mg/kg bw) over 14 days exhibited a reduced number of antibody plaque-forming cells, even at the lowest exposure dose (Dean et al., 1983). Reductions in antibody production at low BaP doses were further demonstrated in female B6C3F1 mice exposed to BaP in corn oil at 0, 5, 20 or 40 mg/kg bw by subcutaneous injection. Again, changes in antibody production were apparent at the lowest exposure dose (White and Holsapple, 1984). Furthermore, in utero BaP exposure by intraperitoneal injection of 150 mg/kg bw per day to pregnant mice from gestational days 11 to 17 also resulted in immunosuppression, as measured by a reduced ability to produce plaque-forming cells in offspring at 18 months of age (Urso and Gengozian, 1984).
Inhibition of lymphocytic responses has also been noted following BaP exposure. Eight-week-old C57 and C3H mice exposed to BaP by intraperitoneal injection (0, 0.5, 5 or 50 mg/kg bw) exhibited inhibited lymphocyte binding at the two highest doses (Wojdani and Alfred, 1984). In utero exposure of pregnant mice to BaP at 150 mg/kg bw per day by intraperitoneal injection from gestation days 11 to 17 also resulted in a reduced ability of lymphocytes to undergo a mixed lymphocytic response in offspring at 18 months of age (Urso and Gengozian, 1984).
Other immunotoxicities included effects on the thymus and immunological cells other than lymphocytes. In a study by De Jong et al. (1999), Wistar rats were orally exposed to BaP (0, 3, 10, 30 or 90 mg/kg bw per day) for 35 days. At all but the lowest dose, rats exhibited a reduced number of red blood cells, reduced numbers of B cells in the spleen and decreased thymus weights, which are all indicative of immunotoxicity. Wistar rats fed BaP at a dose of 150 mg/kg bw per day for 30 days also had reduced bone marrow and spleen cell numbers (Fischer et al., 2011).
9.2.3 Long-term exposure and carcinogenicity
BaP is classified by the International Agency for Research on Cancer (IARC, 2012) as carcinogenic to humans (Group 1), based on sufficient evidence in animals and strong evidence that the cancer mechanisms observed in animals also apply to humans. All routes of exposure were shown to cause local and systemic carcinogenic effects. Evidence of carcinogenesis in animals is presented according to tumour site below.
9.2.3.1 Digestive tract tumours
The most sensitive site for tumour formation following oral exposure was the forestomach. Female A/J mice (30 per dose) fed a diet containing BaP at a concentration of 0, 16 or 98 mg/kg (total dose 0, 11 and 67 mg) for 260 days developed forestomach tumours at all exposure doses (Weyand et al., 1995). In another study, female B6C3F1 mice (48 per dose) fed a diet containing BaP at 0, 5, 25 or 100 mg/kg (corresponding to approximately 0, 0.7, 3.3 and 13.0 mg/kg bw per day) for 2 years developed forestomach tumours at 25 and 100 mg/kg diet and esophageal tumours at 100 mg/kg diet. Combined incidences of forestomach papilloma and carcinoma were 1/48, 3/47, 36/46 and 46/47 for mice exposed to 0, 5, 25 and 100 mg/kg diet, respectively. Tongue tumours were also observed at the highest dose. Similar tumour potency was noted for pure BaP and a complex coal tar mixture containing the same amount of BaP (Culp et al., 1998). In another study, male and female CFW mice fed a diet containing BaP at 0, 1, 10, 20, 30, 40, 45, 50, 100 or 250 mg/kg (corresponding to approximately 0, 0.1, 1.3, 2.6, 3.9, 5.2, 5.9, 6.5, 13.0 and 32.5 mg/kg bw per day) developed papillomas and squamous cell carcinomas of the forestomach in all dose groups above 40 mg/kg diet relative to controls, with a dose-response trend. However, the groups in this study combined animals of both sexes and of various ages, and varying exposure durations were used, thus making interpretation of the results difficult (Neal and Rigdon, 1967). In another chronic study, male and female Wistar rats (52 of each sex per dose) were administered BaP by gavage at a dose of 0, 3, 10 or 30 mg/kg bw per day, 5 days/week, for 104 weeks. Here, males were more sensitive to forestomach tumours than females, as shown by tumour occurrence at 3 mg/kg bw per day in males and 10 mg/kg bw per day in females. Combined incidences of forestomach papilloma and carcinoma were 0/52, 7/52, 18/52 and 17/52 in males and 1/52, 6/51, 30/51 and 50/52 in females for the 0, 3, 10 and 30 mg/kg bw per day doses, respectively (Wester et al., 2012). In one lifetime study of rats, administration of BaP by gavage at 0.15 mg/kg bw either 5 days/week or every 3rd and 9th days caused an increase in forestomach tumours. However, administration of the same dose through diet instead of by gavage (0.15 mg/kg bw 5 times per week or every 9 days) did not cause a significant increase in forestomach tumours (Brune et al., 1981).
In another ingestion study, female B6C3F1 mice (48 per dose) fed a diet containing BaP at 0, 5, 25 or 100 mg/kg bw for 2 years developed tumours of the tongue at the highest dose (Culp et al., 1998). Tumours of the digestive tract were also noted after inhalation exposures. Male hamsters (24 per group) were administered BaP via inhalation (0, 2.2, 9.5 or 46.5 mg/m3) 4.5 hours/day for 10 weeks and then 3 hours/day for the rest of their lifetimes (total average dose of 0, 29, 127 and 383 mg BaP per animal). Hamsters treated with the two highest doses showed an increased incidence of papillomas and squamous cell carcinomas in the upper digestive tract at doses as low as 9.5 mg/m3 (Thyssen et al., 1981).
9.2.3.2 Liver tumours
Exposure of male and female Wistar rats (52 of each sex per dose) to BaP by oral gavage at a dose of 0, 3, 10 or 30 mg/kg bw per day, 5 days/week, for 104 weeks caused tumours in the livers of both males and females at the two highest doses (Wester et al., 2012).
9.2.3.3 Respiratory tract tumours
Respiratory tract tumours were observed following both oral and inhalation administration of BaP. Female A/J mice (30 per dose) fed a diet containing BaP at 0, 16 or 98 mg/kg (total dose 0, 11 and 67 mg) for 260 days developed lung tumours at all doses (Weyand et al., 1995). In an inhalation study, male hamsters (24 per group) were dosed with BaP at 0, 2.2, 9.5 or 46.5 mg/m3 for 4.5 hours/day over 10 weeks and then for 3 hours/day for the rest of their lifetimes (total average doses of 0, 29, 127 and 383 mg BaP per animal). Hamsters treated with the two highest doses showed an increased incidence of papillomas and squamous cell carcinomas in the upper respiratory tract (Thyssen et al., 1981).
9.2.3.4 Skin tumours
Dermal application of BaP also resulted in tumour formation in rodents. Mice are especially sensitive and exhibit benign (squamous cell papillomas and keratoacanthomas) and malignant (primarily squamous cell carcinomas) tumours when BaP is applied to the skin for an extended period of time (Poel, 1959, 1963; Roe et al., 1970; Habs et al., 1980, 1984; Sivak et al., 1997). The lowest dose at which dermal tumours were observed was in a study of C3H/HeJ mice. Mice exposed to BaP at levels of 0.5 and 5.0 µg in cyclohexanone/acetone to shaved dorsal skin twice per week developed skin tumours at rates of 20% (5/30) and 90% (27/30), respectively (Sivak et al., 1997). Skin tumour development was also shown in studies of acute dermal exposure to BaP followed by repeated exposures to tumour promoters (Hoffmann et al., 1972; Slaga et al., 1978, 1980; Wood et al., 1980; Cavalieri et al., 1981, 1991; El-Bayoumy et al., 1982; LaVoie et al., 1982; Weyand et al., 1992).
9.2.3.5 Other tumours
Mammary gland tumours have also been observed upon ingestion of BaP. Rats gavaged with 50 µmol BaP once per week for 8 weeks exhibited increased numbers of mammary gland adenocarcinomas. However, the lowest dose at which this effect can occur is not known (El-Bayoumy et al., 1995).
9.2.4 Genotoxicity
BaP has consistently been shown to cause point mutations in vitro in Salmonella typhimurium and chromosome damage in mammalian cells, including human cells, upon metabolic activation. For these reasons, BaP is often used as a positive control for assays on other compounds. The 2010 IARC monograph reviewed all of the in vivo cases of BaP-DNA adducts in mouse skin after topical treatment and in internal organs and blood lymphocytes following oral, inhalation and intraperitoneal administration of BaP (IARC, 2010). Of all the in vivo studies reviewed, the one by Garner et al. (1985) administered the lowest dose that caused adducts. In this study, male Wistar rats were given a single dose of BaP (2 mg/kg bw) by intraperitoneal injection, and DNA adducts were measured in lungs and liver. Because of the extensive evidence of BaP genotoxicity, in vivo findings are the focus here.
Various studies have demonstrated that BaP induces mutations. BaP induced three types of Ki-ras codon 12 mutations in lung adenomas of A/J mice: GGT→TGT (56.3%), GGT→GTT (25%) and GGT→GAT (19%) (Mass et al., 1993). BaP also induced codon 13 (DiGiovanni et al., 1993) and codon 61 (Chakravarti et al., 1995) mutations in c-Ha-ras in Sencar mouse skin papillomas. BaP caused mutations in murine embryonic fibroblasts from human TP53 knock-in (Hupki) mice that were similar to those found in smoking-related lung cancers in humans (Liu et al., 2005). Male Muta™Mouse mice exposed to BaP at 25, 50 or 75 mg/kg bw per day for 28 days and sacrificed 3 days after the final exposure had increased lacZ transgene mutant frequencies in the lungs (Labib et al., 2012), liver, glandular stomach, small intestine and bone marrow (Lemieux et al., 2011) at all doses. A dose-dependent increase in mutant Pig-a phenotypes was also observed in these mice (Lemieux et al., 2011; Labib et al., 2012). A meta-analysis of historical in vivo genotoxicity studies used the benchmark dose (BMD) approach to calculate the dose representing a specified 10% change in effect in exposed animals compared with controls (BMD10) from dose-response transgenic rodent mutation assay data. BMD10 values as low as 2.08 mg/kg bw were observed for mutations (Hernandez et al., 2011).
Exposure to BaP was also shown to increase the frequency of micronuclei. Awogi and Sato (1989) exposed male MS/Ae mice to single BaP doses of 62.5, 125, 250 or 500 mg/kg bw by intraperitoneal injection and measured bone marrow micronucleus frequencies. Increased micronuclei were found for all of the doses tested except the lowest. In another study, male HRA/SKh mice were topically exposed to 0.5, 5, 50, 100 or 500 µg BaP alongside controls with a 24-hour post-treatment sampling time. Increased numbers of micronuclei were found at all of the doses in the cultured keratinocytes (He and Baker, 1991). Muta™Mouse males exposed to BaP at 25, 50 or 75 mg/kg bw per day for 28 days by oral gavage and sacrificed 3 days after the final exposure had significant increases in the frequency of micronucleated reticulocytes and normochromatic erythrocytes at all doses (Lemieux et al., 2011). A meta-analysis of historical in vivo genotoxicity studies using the BMD approach to calculate a BMD10 from dose-response data on micronucleus frequency in blood found a BMD10 of 1.28 mg/kg bw (Hernandez et al., 2011).
There is clear evidence that BaP induces other genotoxic effects, including DNA single- and double-strand breaks, sister chromatid exchanges and chromosomal aberrations (ATSDR, 1995). For example, male and female Chinese hamsters given BaP by intraperitoneal injection at 450 mg/kg bw per day on 2 consecutive days exhibited a significant increase in the number of bone marrow cell metaphases with chromosomal aberrations and sister chromatid exchanges (Roszinsky-Kocher et al., 1979). Male DBA/2 mice given two BaP doses of either 10 or 100 mg/kg bw intragastrically experienced significant increases in the frequency of sister chromatid exchanges in bone marrow cells at both doses, whereas male C57BL/6J mice exhibited only a small effect at 100 mg/kg bw (Wielgosz et al., 1991). A single exposure to BaP at 62.5 mg/kg bw intragastrically in adult Brown Norway rats did not cause induction of unscheduled DNA synthesis. However, a significant increase in DNA single-strand breaks relative to controls was observed (Mullaart et al., 1989).
9.2.5 Reproductive and developmental toxicity
9.2.5.1 Reproductive toxicity
Effects on fertility have been observed in experimental animals of both sexes. In male Sprague-Dawley rats, exposure to BaP by oral gavage at 1 or 5 mg/kg bw per day for 90 days caused decreases in testosterone and changes in testicular macrophages in comparison with control animals (Zheng et al., 2010). Various effects were observed in male Swiss albino rats exposed orally to BaP at 50 mg/kg bw per day for 10 days (Arafa et al., 2009) and in male Sprague-Dawley rats exposed orally to BaP at 5 mg/kg bw per day for 12 weeks (Chen et al., 2011), including decreases in sperm count, increases in the number of abnormal sperm and changes in testosterone and testicular enzyme levels. Ovotoxicity has also been found in adult rodents exposed to BaP. For example, production of the sex hormone 17β-estradiol was suppressed in female rats following intragastric exposure to BaP at 5 or 10 mg/kg bw on alternating days for 60 days (Xu et al., 2010).
Experimental animals that were exposed to BaP in utero also exhibited adverse reproductive effects in their adult life. In one study, reproductive effects observed in offspring were seen at lower doses than in the directly exposed adults. Decreases in pregnancy and litter viability in CD-1 mice were observed at a BaP dose of 160 mg/kg bw per day, whereas gestational exposure to BaP doses as low as 10 mg/kg bw per day on days 7-16 resulted in decreased fertility in male offspring and marked effects on gonadal tissues, including atrophic seminiferous tubules and increased numbers of interstitial cells in the testes of males, as well as considerable reductions of ovarian tissue in female offspring. Further evidence of transgenerational reproductive effects include ovotoxicity in offspring of NMRI mice treated with BaP by gavage at 10 mg/kg bw per day on gestational days 7-16 (Kristensen et al., 1995) and reduced testicular weight and sperm count in male mice after treatment with BaP at 2 or 10 mg/kg bw per day on gestational days 7-16 (Nakamura et al., 2012).
Overall, these adverse reproductive effects were observed at BaP doses as low as 1 mg/kg bw per day and 2 mg/kg bw per day in adults and offspring, respectively.
9.2.5.2 Developmental toxicity
Developmental toxicity included some evidence of reduced fetal survival and birth weight, a predisposition to cancer in adulthood, neurodevelopmental effects, malformations as well as immunotoxicity.
Reduced body weight 20 and 42 days after birth has been shown in the offspring of CD-1 mice treated with BaP at 40 or 160 mg/kg bw per day via oral gavage on days 7 through 16 of gestation, but not at 10 mg/kg bw per day (Mackenzie and Angevine, 1981). Decreased fetal survival resulting from resorptions and stillbirths as well as an increased incidence of fetal malformations were found in mice exposed gestationally to BaP at 50-300 mg/kg bwvia intraperitoneal injection of their pregnant mothers (Shum et al., 1979). Pregnant rats that inhaled BaP at a concentration of 25, 50 or 100 µg/m3 for 4 hours/day over 10 days exhibited up to 70% fetal resorptions (Wu et al., 2003).
Exposure to BaP during gestational development results in higher incidences of lung, liver and ovarian cancers in adulthood (reviewed by U.S. EPA, 2007). The number of tumours for each site was significantly increased even in unexposed F2 to F5 generations derived from F1 mice exposed in utero via subcutaneous injection, although only at a very high BaP dose of 150 mg/kg bw per day (Urso and Gengozian, 1982, 1984; Turusov et al., 1990). An additional study notes that oral exposure of pregnant rats to BaP at 0.6 and 1.2 mg/kg bw per day can predispose offspring to cardiac dysfunction later in life (Jules et al., 2012).
Several studies have examined the effects of BaP on neurodevelopment in rodents. Neurodevelopment has been examined in mice exposed through maternal milk of BaP-exposed dams. Maternal exposure to 2 and 20 mg/kg bw during the first 14 days of lactation significantly decreased reflexes in the pups and altered neuronal gene expression. Altered behaviour was observed in the elevated plus maze test several days after the termination of exposure (Bouayed et al., 2009). The study by Chen et al. (2012) is the most complete neurodevelopmental study, as it employed a broader range of BaP concentrations (0, 0.02, 0.2 and 2 mg/kg bw per day) and examined the consequences of exposure early in life on anxiety-like behaviour and spatial learning into adulthood. BaP was administered by oral gavage to Sprague-Dawley rat neonates of both sexes for 6 days from postnatal days 5 to 11, and neurodevelopment was assessed on various days up to postnatal day 75 (early adulthood). Although few effects on developmental milestones and sensory responses were observed in exposed juvenile rats, more apparent neurotoxicity was found in young adult rats. In the elevated plus maze test (measure of anxiety-like behaviour), significant anxiolytic-like effects were seen at doses as low as 0.2 mg/kg bw per day in males and 0.02 mg/kg bw per day in females. In the Morris maze test (swim maze to measure spatial learning), effects were seen at doses of 0.2 mg/kg bw per day and higher in both male and female rats. A lowest-observed-adverse-effect level (LOAEL) of 0.02 mg/kg bw per day, the lowest dose tested, can be identified from this study. However, few other studies are available to support neurodevelopmental effects, especially at such low doses of BaP.
Furthermore, in utero BaP exposure by intraperitoneal injection of 150 mg/kg bw per day to pregnant mice from gestational days 11 to 17 also resulted in immunosuppression, as measured by a reduced ability to produce plaque-forming cells as well as alterations in the function of lymphocytes in offspring at 18 months of age (Urso and Gengozian, 1984).
9.3 Toxicogenomics
Numerous studies have used whole genome gene expression profiles (toxicogenomics) to identify early molecular changes predictive of cancer and other effects. These analyses allow the identification and quantification of cellular responses to chemical exposure by analysis of gene expression activity. Most of these studies have been conducted in vitro using liver (Hockley et al., 2006, 2009; Michaelson et al., 2011; Lizarraga et al., 2012), colon (de Waard et al., 2008; Hockley et al., 2008), mammary (Gwinn et al., 2005; Keshava et al., 2005; Gwinn and Weston, 2008), lung epithelial (Hirano et al., 2013), prostate (Hrubá et al., 2011), amnion (Lu et al., 2009, 2010) and oral epithelial (Li et al., 2008) cells. Some of these studies were done in untransformed normal human cells and in cells relevant to the affected organs. These, along with in vivo studies conducted in various tissues, were considered to be the most relevant and are therefore reported below.
Two studies have investigated pulmonary and hepatic gene expression profiles of 5-week-old male B6C3F1 mice (five per dose group) that received BaP in corn oil by oral gavage at a concentration of 0 (corn oil alone), 5, 50, 150 or 300 mg/kg bw per day for 3 consecutive days (Halappanavar et al., 2011; Yauk et al., 2011). Samples were collected for gene expression analysis 4 or 24 hours after the last exposure. Hepatic gene expression analysis in mice exposed to BaP at 150 or 300 mg/kg bw per day for 3 days revealed approximately 836 and 1541 genes, respectively, that were altered relative to controls (either upregulated or downregulated with fold change [FC] differences ≥ 1.5 and false discovery rate [FDR] adjusted p-value ≤ 0.05). In the lungs of these mice, 558 and 1267 genes were differentially expressed (FC ≥ 1.5 and FDR p ≤ 0.05) following 150 and 300 mg/kg bw per day BaP exposure compared with controls, respectively. The overall gene expression response in the liver was consistent with the known physiological function of the organ, including its primary role in the detoxification of xenobiotic compounds. The pulmonary gene expression response to BaP included changes in biological pathways involved in B cell receptor signalling, inflammation, DNA damage responses and immunosuppression. Overall, these pathways indicate substantive changes in metabolism, DNA repair and oxidative stress occurring in lung and liver following gavage exposure. All of the aforementioned molecular pathways support the carcinogenic properties of BaP.
Three additional studies have investigated pulmonary, hepatic and forestomach gene expression responses in adult (25-week-old) Muta™Mouse males (five per group) exposed to BaP in olive oil by oral gavage at a concentration of 0, 25, 50 or 75 mg/kg bw per day for 28 consecutive days (Labib et al., 2012; Malik et al., 2012). Mice were sacrificed 72 hours after the final exposure. Analyses revealed significant changes at the transcript levels (FC ≥ 1.5 and FDR p-value ≤ 0.05) of 6, 7 and 121 genes in the livers, 20, 145 and 373 genes in the lungs and 9, 135 and 408 genes in the forestomachs of mice in the 25, 50 and 75 mg/kg bw per day exposure groups, respectively. In the liver, the genes were primarily associated with biological processes such as xenobiotic metabolism and p53 signalling indicative of DNA damage. In the lung, there was also significant enrichment of p53 signalling and other cancer-related pathways. Although the p53 signalling pathway was also perturbed in the forestomach, the most significantly enriched processes were involved in antigen processing and presentation, immune response, chemotaxis and keratinocyte differentiation, thus suggesting inflammatory, immune and DNA damage responses. The results in the lung and liver are consistent with expected changes in gene expression in response to a genotoxic carcinogen, whereas the forestomach data point to additional mechanisms relevant to carcinogenesis.
Molecular pathways identified in vitro in treated human cells can provide insight on the relevance of these responses in human tissues. For example, pathways involved in cell cycle regulation, apoptosis/anti-apoptosis, chromatin assembly and oxidative stress were identified in both breast and liver carcinoma cell lines (MCF-7 and HepG2 cells) at levels that induced minimal cytotoxicity (Hockley et al., 2006). Untransformed human amnion cells treated with BPDE also responded by changes in cell cycle regulation, growth and apoptosis (Lu et al., 2009, 2010). These findings support the human relevance of carcinogenic outcomes observed in experimental animals.
9.4 Mode of action
Mode of action (MOA) analyses allow the identification of mechanisms leading to toxic outcomes and their relevance to humans. As a result of the overwhelming evidence of tumour formation in experimental animals and limited evidence of other toxic effects, the MOA analysis was focused on tumour development. The predominant mechanism of tumour initiation is related to BaP metabolite formation following binding to the AhR and is described below.
BaP metabolites have been consistently shown to be genotoxic and mutagenic in vivo and in vitro, and exposure to BaP at environmentally relevant concentrations in experimental animals leads to DNA strand breaks, DNA adducts and chromosomal aberrations (see Section 9.2.4). As such, genotoxicity resulting from BaP metabolites is considered to be the primary MOA for BaP-induced tumorigenesis. Binding of BaP to the AhR is the critical event leading to metabolite formation. For this reason, mice devoid of AhR have been shown not to develop tumours following BaP exposure (Shimizu et al., 2000), although it should be noted that the presence of other chemicals that can induce the specific metabolic enzymes noted below may in part contribute to additional metabolism of BaP. BaP binding to the AhR also occurs in humans, although binding affinity can vary greatly (Harper et al., 2002), thus affecting interindividual sensitivity to exposure.
Binding to the AhR results in the induction of P450 enzymes, including CYP1A1 and CYP1B1, which are responsible for metabolizing BaP to genotoxic carbon cations or epoxides such as BPDE (Cavalieri and Rogan, 1995). The requirement of these enzymes for BaP metabolism is supported by knockout studies in animals. For example, BaP blood levels in CYP1A1−/− and CYP1A1−/− CYP1B1−/− knockout mice are 25- and 75-fold greater than in control animals, respectively, and these mice are less sensitive to many BaP-related toxicities (Uno et al., 2006). There is also clear evidence that BaP metabolism to reactive metabolites occurs in humans. The BaP metabolite BaP-7,8-dihydrodiol, a precursor to the genotoxic metabolite BPDE, is produced by the microsomes of human liver and lung donors (Shimada et al., 1989). Similarly, 3-OH BaP is readily detectable in the urine of occupationally exposed humans in various workplaces, including coking plants and graphite electrode production facilities (Forster et al., 2008).
BaP metabolites, most notably BPDE, form covalent adducts directly with DNA, as well as protein. As a parallel but secondary mechanism, reactive oxygen species produced as by-products of BaP metabolism (from BaP catechol-quinone redox cycling) can also oxidize DNA (Lan et al., 2004). Increased adduct formation leads to heritable changes in DNA, as observed by mutations in various test systems (see Section 9.2.4) as well as in BaP-exposed non-smokers (DeMarini et al., 2001). Thus, the evidence suggests that the genotoxicity of BaP metabolites is the primary reason for tumorigenesis and carcinogenesis in experimental animals. The weight of evidence is sufficiently strong to conclude that this MOA is relevant to humans, as also concluded by IARC (2010).
10.0 Classification and assessment
10.1 Cancer risk assessment
The International Agency for Research on Cancer has classified BaP as carcinogenic to humans (group 1), based on sufficient evidence in animals and strong evidence that the mechanisms of carcinogenesis observed in animals also operate in humans (IARC, 2010). Carcinogenic outcomes in humans are supported by increased cancer risk in consumers of charred meats, coke oven workers and smokers. Rodents chronically exposed to BaP develop both tumours related to the point of entry as well as tumours related to systemic distribution of BaP. Affected tissues include the forestomach, liver, esophagus, lung, tongue, larynx, oral cavity, skin and mammary gland. MOA analyses and both in vivo and in vitro genotoxicity data implicate BaP metabolites in the direct damage of DNA.
Two studies that evaluated tumour induction in mice and rats were considered in deriving the health-based value (HBV) for BaP in drinking water (Culp et al., 1998; Wester et al., 2012). Both studies were chronic in duration, tested for multiple doses and employed a sufficient number of animals in each group. Furthermore, both studies were relevant to the oral route of exposure to BaP. In female B6C3F1 mice, increased incidences of papillomas and carcinomas of the forestomach, esophagus and larynx were found (Culp et al., 1998). In male and female Wistar rats, increased incidences of forestomach, liver and oral cavity papillomas and carcinomas were found (Wester et al., 2012). All endpoints were considered in calculating the HBV. BMD modelling was conducted using the software BMDS Version 2.2 (U.S. EPA, 2011) to estimate the 95% lower confidence limit on the BMD10 (BMDL10) for each cancer endpoint. In both studies, the forestomach was determined to be the most sensitive tissue. As humans do not have a forestomach, the application of rodent forestomach tumour data for predicting cancer risk in humans for a particular chemical should meet certain criteria, including a carcinogenic MOA and induction of tumours at multiple sites and in various species (Proctor et al., 2007). BaP meets these criteria because it acts through a genotoxic MOA, it causes tumours at multiple sites by oral exposures in various species and in both sexes and its MOA in carcinogenesis is understood and considered applicable to humans. The lowest BMDL10 identified from the two studies was 0.5389 mg/kg bw per day (Moffat et al., 2015), which was observed in female mice using the best fit model (log logistic). This value was used as a point of departure for development of an HBV for BaP.
Current PBPK models are not adequately developed for use in developing a drinking water guideline (see Section 8.5), and information on interspecies toxicokinetic and toxicodynamic differences necessary to apply chemical-specific adjustment factors was not available. Thus, the point of departure was adjusted using allometric scaling, in order to more properly represent human exposures:
Equivalent human dose = 0.5389 mg/kg bw per day × (0.03 kg/70 kg)¼
= 0.07758 mg/kg bw per day
where:
- 0.5389 mg/kg bw per day is the lowest BMDL10 identified for forestomach tumours in female mice (Culp et al., 1998; Moffat et al., 2015);
- 0.03 kg is the default average body weight of a mouse;
- 70 kg is the default average body weight of a human adult; and
- (0.03 kg/70 kg)¼ is the allometric scaling factor to account for interspecies differences in susceptibility to BaP.
The equivalent human dose was used to derive the cancer slope factor (CSF):
CSF = 0.1 / 0.07758 mg/kg bw per day
= 1.2890 (mg/kg bw per day)−1
where:
- 0.1 is the 10% benchmark response; and
- 0.07758 mg/kg bw per day is the equivalent human dose calculated above.
The dose associated with each lifetime risk is calculated by dividing the corresponding lifetime risk by the CSF calculated above.
| Lifetime risk | Dose (mg/kg bw per day) |
|---|---|
10−4 |
0.776 × 10−4 |
10−5 |
0.776 × 10−5 |
10−6 |
0.776 × 10−6 |
The formula to convert from dose to concentration in drinking water is:
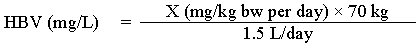
Text Description
The HBV in mg/L is calculated by multiplying the dose in mg/kg bw per day by 70 kg (the default average body weight for a human adult). This product is then divided by 1.5 L/day (the average daily intake of water by an adult).
where:
- X is the dose calculated above;
- 70 kg is the default average body weight of a human adult; and
- 1.5 L/day is the average daily intake of water by an adult.
Based on the calculated unit risks, the estimated concentrations corresponding to excess lifetime human cancer risks of 10−4, 10−5 and 10−6 are as follows:
| Lifetime risk | Estimated concentrations in drinking water (µg/L) |
|---|---|
| 10−4 | 4 |
| 10−5 | 0.4 |
| 10−6 | 0.04 |
An excess lifetime cancer risk of 10−6 or below is used when intake from other sources is considerable (Krishnan and Carrier, 2013). As there are other significant sources of exposure to BaP (i.e., food), the excess lifetime cancer risk of 10−6 was used to derive an HBV of 0.04 µg/L.
10.2 Non-cancer risk assessment
There is some evidence of non-cancer effects of BaP exposure, including adverse neurological and neurodevelopmental effects that can occur at low doses. Delayed performance and markers of neurotoxicity in the hippocampus were apparent in rats injected with 2.5 mg/kg bw per day for 13 weeks (Xia et al., 2011). One study of neonatal rats exposed orally from postnatal days 5 to 11 demonstrated that adverse neurological effects, measured by poor performance in two maze tests, worsened into adulthood (on postnatal days 70 and 75 in comparison with postnatal days 35 and 40 for the elevated plus maze and the Morris maze, respectively) (Chen et al., 2012). These effects were significant at BaP doses as low as 0.02 mg/kg bw per day in female rats for the elevated plus maze test and at doses of 0.2 mg/kg bw per day and higher in male and female rats for the Morris maze test.
As these effects were significant at BaP doses of 0.2 mg/kg bw per day and higher, a NOAEL of 0.02 mg/kg bw per day (20 µg/kg bw per day) can be used as a point of departure in the calculation of the tolerable daily intake (TDI). The TDI for neurodevelopmental toxicity was calculated as follows:
![The equation used to calculate the tolerable daily intake of benzo[a]pyrene under the non-cancer risk assessment](/content/dam/phac-aspc/images/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-benzo-alpha-pyrene/fig02-eng.jpg)
Text Description
The tolerable daily intake (TDI) of benzo[a]pyrene under the non-cancer risk assessment is 0.0667 µg/kg bw per day. This is calculated by dividing the NOAEL of 20 µg/kg bw per day by the uncertainty factor 300.
where:
- 20 µg/kg bw per day is the NOAEL (Chen et al., 2012); and
- 300 is the uncertainty factor (× 10 for intraspecies variability; × 10 for interspecies variability; and (× 3 for database deficiencies).
An uncertainty factor of 3 was added to account for deficiencies in the database for non-cancer effects for BaP given studies on neurological and neurodevelopmental effects, as well as data on low-dose effects is limited, and there exists limited data to support any non-cancer MOAs for BaP. Using the TDI calculated above, an HBV can be derived as follows:
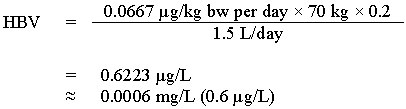
Text Description
The health based value under the non-cancer risk assessment is 0.6 µg/L (rounded). This is calculated by multiplying the TDI of 0.0667 µg/kg bw per day by the average body weight of a human adult (70 kg), then by the allocation factor for drinking water, 0.2. This product is then divided by the average daily intake of water by an adult, 1.5 L/day.
where:
- 0.0667 µg /kg bw per day is the TDI derived above;
- 70 kg is the default average body weight of a human adult;
- 0.2 is the default allocation factor for drinking water; it is used as a "floor value," since drinking water is not a major source of exposure to BaP, and there is evidence of its widespread presence in at least one of the other media (Krishnan and Carrier, 2013); and
- 1.5 L/day is the average daily intake of water by an adult; dermal and inhalation exposures from bathing and showering are not considered to be significant routes of exposure (as described in Section 5.6).
10.3 Comparison of cancer and non-cancer risk assessments
The concentration of BaP in drinking water associated with a lifetime excess risk of the most sensitive tumour endpoint (forestomach tumours) in female mice of 10-6 is determined as 0.04 µg/L. The non-cancer HBV, based on the neurological effects in offspring of exposed rats, is 0.6 µg/L. As the cancer risk assessment resulted in a more conservative value for BaP in drinking water compared with that generated by the non-cancer approach, the cancer risk assessment was determined to be the most appropriate driver of the MAC in drinking water. Moreover, there is much greater confidence in the known carcinogenic effects of BaP than in non-cancer effects. Thus, the HBV of 0.04 µg/L is considered to be protective of all cancers as well as the developmental effects of BaP.
10.4 International considerations
This section presents the various drinking water guidelines and standards from other international organizations. Variations in these limits can be attributed to the age of the assessments or to differing policies and approaches, including the choice of key study and the use of different consumption rates, body weights and allocation factors.
The U.S. EPA (1992) established a maximum contaminant level for BaP in drinking water of 0.2 µg/L, based on the data of Neal and Rigdon (1967). A CSF of 7.3 (mg/kg bw per day)−1 was derived using the geometric mean of four slope factors calculated with different assumptions, from Neal and Rigdon (1967) and Brune et al. (1981).
The World Health Organization (WHO, 2011) established a guideline value of 0.7 µg/L for BaP in 1996, which corresponds to an excess lifetime cancer risk of 10−5. This is also based on the oral carcinogenicity study of Neal and Rigdon (1967). The value was calculated using a two-stage birth-death mutation model.
The California Environmental Protection Agency (OEHHA, 2010) established a public health goal of 0.007 µg/L and a maximum contaminant level of 0.2 µg/L for BaP in drinking water based on carcinogenic effects in experimental animals. Studies in mice (Culp et al., 1998) and rats (Kroese et al., 2001) were considered instead of the study by Neal and Rigdon (1967) because of the substantive limitations of the latter study. The data in Kroese et al. (2001) are the same as those in Wester et al. (2012). A time-to-tumour model was employed to obtain lower-bound estimates of the dose associated with a 10% increased incidence of combined oral cavity tumours in female mice and rats of both sexes as well as liver tumours in rats. Tumours in the oral cavity and forestomach of female mice yielded the lowest lower-bound dose associated with a 10% increased incidence of tumours. The public health goal was calculated from the cancer potency, which was derived assuming a linear relationship between dose and response at low doses and corrected for age and early-life exposure. A health protective level of 4 µg/L was also derived from rat renal toxicity data in Knuckles et al. (2001) to account for non-cancer effects, using an uncertainty factor of 3000, a relative source contribution of 0.1 and the ingestion of 0.044 L/kg bw per day.
11.0 Rationale
Although BaP is not manufactured or imported, its presence in the environment is ubiquitous, as it is formed through natural and anthropogenic combustion processes. It may also enter drinking water through the distribution system, by leaching from coal tar (bituminous) and asphaltic linings in water storage tanks and distribution system pipes. However, drinking water is not a major source of exposure to BaP.
BaP has been classified as a human carcinogen (Group 1), with sufficient evidence in animals and strong evidence that the mechanisms of carcinogenesis observed in animals also operate in humans. There is evidence of non-cancer health effects, including neurological and neurodevelopmental effects, related to low levels of exposure to BaP in rodents. Both cancer and non-cancer risk assessments were considered. The cancer risk assessment was used to establish the MAC since it resulted in a more conservative value and there is greater confidence in the known carcinogenic effects of BaP than in non-cancer effects.
The estimated lifetime cancer risk from exposure to BaP in drinking water at levels ranging from 0.04 to 0.4 µg/L is considered to be "essentially negligible." The guideline for a carcinogen is normally established at a level at which the increased cancer risk is "essentially negligible" when a person is exposed at that level in drinking water over a lifetime. In the context of drinking water guidelines, Health Canada has defined this term as a range from one new cancer above background levels per 100 000 people to one new cancer above background levels per 1 million people (i.e., 10−5-10−6). An increased cancer risk of 10−6 is targeted in cases where intake from sources other than drinking water are significant. In the case of BaP, where food is the main source of exposure, the MAC is established at a concentration that would present an "essentially negligible" risk of one new cancer above background per million people (i.e., 10−6) exposed to BaP in drinking water at the MAC over a lifetime, which takes into consideration treatment.
In summary, a MAC of 0.000 04 mg/L (0.04 µg/L) is established for BaP based on the following considerations:
- It falls within the range considered to present an "essentially negligible" risk.
- It is detectable and measurable, with a method detection limit well below the MAC. The U.S. EPA has established a PQL of 0.0002 mg/L, based on the ability of laboratories to measure BaP within reasonable limits of precision and accuracy using approved methods. Although there is no similar process in place to establish a PQL specific to Canada, a limited of survey of commercial laboratories indicates that they can reliably measure concentrations of BaP equal to or less than 0.01 µg/L.
- The MAC must be achievable at reasonable cost. Municipal scale treatment technologies can consistently remove BaP from drinking water to below the MAC of 0.04 µg/L. At the residential level, although there are no treatment units currently certified to remove BaP, it is expected that the use of treatment devices using technologies such as activated carbon would capable of removing BaP to below the MAC.
As part of its ongoing guideline review process, Health Canada will continue to monitor new research in this area and recommend any change to the guideline that is deemed necessary.
12.0 References
- Aboutabl, M.E., Zordoky, B.N. and El-Kadi, A.O. (2009). 3-Methylcholanthrene and benzo[a]pyrene modulate cardiac cytochrome P450 gene expression and arachidonic acid metabolism in male Sprague Dawley rats. Br. J. Pharmacol., 158(7): 1808-1819.
- Aboutabl, M.E., Zordoky, B.N., Hammock, B.D. and El-Kadi, A.O. (2011). Inhibition of soluble epoxide hydrolase confers cardioprotection and prevents cardiac cytochrome P450 induction by benzo[a]pyrene. J. Cardiovasc. Pharmacol., 57(3): 273-281.
- Anderson, K.E., Kadlubar, F.F., Kulldorff, M., Harnack, L., Gross, M., Lang, N.P., Barber, C., Rothman, N. and Sinha, R. (2005). Dietary intake of heterocyclic amines and benzo[a]pyrene: associations with pancreatic cancer. Cancer Epidemiol. Biomarkers Prev., 14(9): 2261-2265.
- APHA, AWWA and WEF (2012). Standard methods for the examination of water and wastewater. 22nd edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
- Arafa, H.M., Aly, H.A., Abd-Ellah, M.F. and El-Refaey, H.M. (2009). Hesperidin attenuates benzo[α]pyrene-induced testicular toxicity in rats via regulation of oxidant/antioxidant balance. Toxicol. Ind. Health, 25(6): 417-427.
- Armstrong, B.G. and Gibbs, G. (2009). Exposure-response relationship between lung cancer and polycyclic aromatic hydrocarbons (PAHs). Occup. Environ. Med., 66(11): 740-746.
- Armstrong, B.G., Tremblay, C.G., Cyr, D. and Thériault, G.P. (1986). Estimating the relationship between exposure to tar volatiles and the incidence of bladder cancer in aluminum smelter workers. Scand. J. Work Environ. Health, 12(5): 486-493.
- Armstrong, B., Tremblay, C., Baris, D. and Thériault, G. (1994). Lung cancer mortality and polynuclear aromatic hydrocarbons: a case-cohort study of aluminum production workers in Arvida, Quebec, Canada. Am. J. Epidemiol., 139(3): 250-262.
- ATSDR (1995). Toxicological profile for polycyclic aromatic hydrocarbons. Agency for Toxic Substances and Disease Registry, Public Health Service, U.S. Department of Health and Human Services, Atlanta, Georgia. Available at: www.atsdr.cdc.gov/toxprofiles/tp69.pdf
- Awogi, T. and Sato, T. (1989). Micronucleus test with benzo[a]pyrene using a single peroral administration and intraperitoneal injection in males of the MS/Ae and CD-1 mouse strains. Mutat. Res., 223(4): 353-356.
- Basu, D.K. and Saxena, J. (1978). Polynuclear aromatic hydrocarbons in selected US drinking water and their raw water sources. Environ. Sci. Technol., 12: 795-798.
- Basu, D.K., Saxena, J., Stoss, F.W., Santodonato, J., Neal, M.W. and Kopfler, F.C. (1987). Comparison of drinking water mutagenicity with leaching of polycyclic aromatic hydrocarbons from water distribution pipes. Chemosphere, 16(10-12): 2595-2612.
- Blokker, E.J.M., van de Ven, B.M., de Jongh, C.M. and Slaats, P.G.G. (2013). Health implications of PAH release from coated cast iron drinking water distribution systems in the Netherlands. Environ. Health Perspect., 121(5): 600-606.
- Bock, K.W. and Bock-Hennig, B.S. (2010). UDP-glucuronosyltransferases (UGTs): from purification of Ah-receptor inducible UGT1A6 to coordinate regulation of subsets of CYPs, UGTs, and ABC transporters by nuclear receptors. Drug Metab. Rev., 42(1): 5-12.
- Bouayed, J., Desor, F., Rammal, H., Kiemer, A.K., Tybl, E., Schroder, H., Rychen, G. and Soulimani, R. (2009). Effects of lactational exposure to benzo[α]pyrene (B[α]P) on postnatal neurodevelopment, neuronal receptor gene expression and behaviour in mice. Toxicology, 259: 97-106.
- Bouayed, J., Bohn, T., Tybl, E., Kiemer, A.K. and Soulimani, R. (2012). Benzo[α]pyrene-induced anti-depressive-like behaviour in adult female mice: role of monoaminergic systems. Basic Clin. Pharmacol. Toxicol., 110(6): 544-550.
- Bradley, L.J., Magee, B.H. and Allen, S.L. (1994). Background levels of polycyclic aromatic hydrocarbons (PAH) and selected metals in New England urban soils. J. Soil Contam., 3(4): 349-361.
- British Columbia Ministry of Health (2013). Personal communication from B. Boettger.
- Brown, R.P., Delp, M.D., Lindstedt, S.L., Rhomberg, L.R. and Beliles, R.P. (1997). Physiological parameter values for physiologically based pharmacokinetic models. Toxicol. Ind. Health, 13: 407-484.
- Brune, H., Deutsch-Wenzel, R.P., Habs, M., Ivankovic, S. and Schmähl, D. (1981). Investigation of the tumorigenic response to benzo[a]pyrene in aqueous caffeine solution applied orally to Sprague-Dawley rats. J. Cancer Res. Clin. Oncol., 102(2): 153-157.
- Butković, V., Klasinc, L., Orhanović, M. and Turk, J. (1983). Reaction rates of polynuclear aromatic hydrocarbons with ozone in water. Environ. Sci. Technol., 17: 546-548.
- Butler, L.M., Sinha, R., Millikan, R.C., Martin, C.F., Newman, B., Gammon, M.D., Ammerman, A.S. and Sandler, R.S. (2003). Heterocyclic amines, meat intake, and association with colon cancer in a population-based study. Am. J. Epidemiol., 157(5): 434-445.
- Carlson, G.P., Fossa, A.A., Morse, M.A. and Weaver, P.M. (1986). Binding and distribution studies in the SENCAR mouse of compounds demonstrating a route-dependent tumorigenic effect. Environ. Health Perspect., 68: 53-60.
- Cavalieri, E.L. and Rogan, E.G. (1992). The approach to understanding aromatic hydrocarbon carcinogenesis. The central role of radical cations in metabolic activation. Pharmacol. Ther., 55(2): 183-199.
- Cavalieri, E.L. and Rogan, E.G. (1995). Central role of radical cations in metabolic activation of polycyclic aromatic hydrocarbons. Xenobiotica, 25(7): 677-688.
- Cavalieri, E., Rogan, E., Toth, B. and Munhall, A. (1981). Carcinogenicity of the environmental pollutants cyclopenteno-[cd]pyrene and cyclopentano[cd]pyrene in mouse skin. Carcinogenesis, 2: 277-281.
- Cavalieri, E.L., Higginbotham, S., Ramakrishna, N.V., Devanesan, P.D., Todorovic, R., Rogan, E.G. and Salmasi, S. (1991). Comparative dose-response tumorigenicity studies of benzo[alpha,I]pyrene versus 7,12-dimethylbenz[alpha]anthracene, benzo[alpha]pyrene and two dibenzo[alpha,I]pyrene dihydrodiols in mouse skin and rat mammary gland. Carcinogenesis, 12: 1939-1944.
- CCME (2010). Canadian soil quality guidelines: carcinogenic and other polycyclic aromatic hydrocarbons (PAHs) (environmental and human health effects). Scientific criteria document (revised). Canadian Council of Ministers of the Environment (PN 1401). Available at: www.ccme.ca/files/Resources/supporting_scientific_documents/pah_soqg_ssd_1401.pdf
- Chakravarti, D., Pelling, J.C., Cavalieri, E.L. and Rogan, E.G. (1995). Relating aromatic hydrocarbon-induced DNA adducts and c-H-ras mutations in mouse skin papillomas: the role of apurinic sites. Proc. Natl. Acad. Sci. U.S.A., 92(22): 10422-10426.
- Chang, L.H. (1943). The fecal excretion of polycyclic hydrocarbons following their administration to the rat. J. Biol. Chem., 151(1): 93-99.
- CHEMFATE (2013). CHEMFATE entry form for "Benzo[a]pyrene". SRC, Inc., Syracuse, New York. Available at: http://esc.syrres.com/scripts/CASCFcgi.exe?CASNUM=50328
- Chen, C., Tang, Y., Jiang, X., Qi, Y., Cheng, S., Qiu, C., Peng, B. and Tu, B. (2012). Early postnatal benzo[a]pyrene exposure in Sprague-Dawley rats causes persistent neurobehavioral impairments that emerge postnatally and continue into adolescence and adulthood. Toxicol. Sci., 125(1): 248-261.
- Chen, X., An, H., Ao, L., Sun, L., Liu, W., Zhou, Z., Wang, Y. and Cao, J. (2011). The combined toxicity of dibutyl phthalate and benzo[a]pyrene on the reproductive system of male Sprague Dawley rats in vivo. J. Hazard. Mater., 186(1): 835-841.
- Creasia, D.A., Poggenburg, J.K., Jr. and Nettesheim, P. (1976). Elution of benzo[α]pyrene from carbon particles in the respiratory tract of mice. J. Toxicol. Environ. Health, 1(6): 967-975.
- Crittenden, J.C., Trussell, R.R., Hand, D.W., Howe, K.J. and Tchobanoglous, G. (2005). Water treatment: principles and design. 2nd edition. John Wiley & Sons on behalf of MWH.
- Cross, A.J., Peters, U., Kirsh, V.A., Andriole, G.L., Reding, D., Hayes, R.B. and Sinha, R. (2005). A prospective study of meat and meat mutagens and prostate cancer risk. Cancer Res., 65(24): 11779-11784.
- Cross, A.J., Ward, M.H., Schenk, M.J., Kulldorff, M., Cozen, W., Davis, S., Colt, J.S., Hartge, P., Cerhan, J.R. and Sinha, R. (2006). Meat and meat-mutagen intake and risk of non-Hodgkin lymphoma: results from a NCI-SEER case-control study. Carcinogenesis, 27(2): 293-297.
- Crowell, S.R., Amin, S.G., Anderson, K.A., Krishnegowda, G., Sharma, A.K., Soelberg, J.J., Williams, D.E. and Corley, R.A. (2011). Preliminary physiologically based pharmacokinetic models for benzo[a]pyrene and dibenzo[def,p]chrysene in rodents. Toxicol. Appl. Pharmacol., 257(3): 365-376.
- Culp, S.J., Gaylor, D.W., Sheldon, W.G., Goldstein, L.S. and Beland, F.A. (1998). A comparison of the tumors induced by coal tar and benzo[a]pyrene in a 2-year bioassay. Carcinogenesis, 19(1): 117-124.
- Dean, J.H., Luster, M.I., Boorman, G.A., Lauer, L.D., Leubke, R.W. and Lawson, L. (1983). Selective immunosuppression resulting from exposure to the carcinogenic congener of benzopyrene in B6C3F1 mice. Clin. Exp. Immunol., 52(1): 199-206.
- De Flora, S., Izzotti, A., D'Agostini, F., Rossi, G.A. and Balansky, R.M. (1993). Pulmonary alveolar macrophages in molecular epidemiology and chemoprevention of cancer. Environ. Health Perspect., 99: 249-252.
- De Jong, W.H., Kroese, E.D., Vos, J.G. and Van Loveren, H. (1999). Detection of immunotoxicity of benzo[a]pyrene in a subacute toxicity study after oral exposure in rats. Toxicol. Sci., 50(2): 214-220.
- DeMarini, D.M., Landi, S., Tian, D., Hanley, N.M., Li, X., Hu, F., Roop, B.C., Mass, M.J., Keohavong, P., Gao, W., Olivier, M., Hainaut, P. and Mumford, J.L. (2001). Lung tumor KRAS and TP53 mutations in nonsmokers reflect exposure to PAH-rich coal combustion emissions. Cancer Res., 61(18): 6679-6681.
- De Stefani, E., Boffetta, P., Deneo-Pellegrini, H., Ronco, A.L., Aune, D., Acosta, G., Brennan, P., Mendilaharsu, M. and Ferro, G. (2009). Meat intake, meat mutagens and risk of lung cancer in Uruguayan men. Cancer Causes Control, 20(9): 1635-1643.
- de Waard, W.J., Aarts, J.M.M.J.G., Peijnenburg, A.A.C.M., Baykus, H., Talsma, E., Punt, A., de Kok, T.M.C.M., van Schooten, F.J. and Hoogenboom, L.A.P. (2008). Gene expression profiling in Caco-2 human colon cells exposed to TCDD, benzo[a]pyrene, and natural Ah receptor agonists from cruciferous vegetables and citrus fruits. Toxicol. In Vitro, 22(2): 396-410.
- DiGiovanni, J., Beltran, L., Rupp, A., Harvey, R.G. and Gill, R.D. (1993). Further analysis of c-Ha-ras mutations in papillomas initiated by several polycyclic aromatic hydrocarbons and papillomas from uninitiated, promoter-treated skin in SENCAR mice. Mol. Carcinog., 8(4): 272-279.
- Duarte-Salles, T., Mendez, M.A., Morales, E., Bustamante, M., Rodríguez-Vicente, A., Kogevinas, M. and Sunyer, J. (2012). Dietary benzo[a]pyrene and fetal growth: effect modification by vitamin C intake and glutathione S-transferase P1 polymorphism. Environ. Int., 45(1): 1-8.
- Ekwall, P., Ermala, P. and Setala, K. (1951). Gastric absorption of 3,4-benzpyrene. II. The significance of the solvent for the penetration of 3,4-benzpyrene into the stomach wall. Cancer Res., 11(10): 758-763.
- El-Bayoumy, K., Hecht, S.S. and Hoffmann, D. (1982). Comparative tumor initiating activity on mouse skin of 6-nitrobenzo[a]pyrene, 6-nitrochrysene, 3-nitroperylene, 1-nitropyrene and their parent hydrocarbons. Cancer Lett., 16: 333-337.
- El-Bayoumy, K., Chae, Y.H., Upadhyaya, P., Rivenson, A., Kurtzke, C., Reddy, B. and Hecht, S.S. (1995). Comparative tumorigenicity of benzo[a]pyrene, 1-nitropyrene and 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine administered by gavage to female CD rats. Carcinogenesis, 16(2): 431-434.
- Ermala, P., Setala, K. and Ekwall, P. (1951). Gastric absorption of 3,4-benzpyrene. I. The effect of physiological processes on absorption. Cancer Res., 11(10): 753-757.
- Fang, A.H., Smith, W.A., Vouros, P. and Gupta, R.C. (2001). Identification and characterization of a novel benzo[a]pyrene-derived DNA adduct. Biochem. Biophys. Res. Commun., 281(2): 383-389.
- Ferrucci, L.M., Sinha, R., Huang, W.Y., Berndt, S.I., Katki, H.A., Schoen, R.E., Hayes, R.B. and Cross, A.J. (2012). Meat consumption and the risk of incident distal colon and rectal adenoma. Br. J. Cancer, 106(3): 608-616.
- Fischer, A., Koeper, L.M. and Vohr, H.W. (2011). Specific antibody responses of primary cells from different cell sources are able to predict immunotoxicity in vitro. Toxicol. In Vitro, 25(8): 1966-1973.
- Forster, K., Preuss, R., Rossbach, B., Bruning, T., Angerer, J. and Simon, P. (2008). 3-Hydroxybenzo[a]pyrene in the urine of workers with occupational exposure to polycyclic aromatic hydrocarbons in different industries. Occup. Environ. Med., 65(4): 224-229.
- Foth, H., Kahl, R. and Kahl, G.F. (1988). Pharmacokinetics of low doses of benzo[a]pyrene in the rat. Food Chem. Toxicol., 26(1): 45-51.
- Friesen, M.C., Demers, P.A., Spinelli, J.J., Lorenzi, M.F. and Le, N.D. (2007). Comparison of two indices of exposure to polycyclic aromatic hydrocarbons in a retrospective aluminium smelter cohort. Occup. Environ. Med., 64(4): 273-278.
- Fronk, C.A. (1987). Destruction of volatile organic contaminants in drinking water by ozone treatment. Ozone Sci. Eng., 9: 265-288.
- Fu, Z., Deming, S.L., Fair, A.M., Shrubsole, M.J., Wujcik, D.M., Shu, X.O., Kelley, M. and Zheng, W. (2011). Well-done meat intake and meat-derived mutagen exposures in relation to breast cancer risk: the Nashville Breast Health Study. Breast Cancer Res. Treat., 129(3): 919-928.
- Garg, R., Gupta, S. and Maru, G.B. (2008). Dietary curcumin modulates transcriptional regulators of phase I and phase II enzymes in benzo[a]pyrene-treated mice: mechanism of its anti-initiating action. Carcinogenesis, 29(5): 1022-1032.
- Garner, R.C., Stanton, C.A., Martin, C.N., Harris, C.C. and Grafstrom, R.C. (1985). Rat and human explant metabolism, binding studies, and DNA adduct analysis of benzo[a]pyrene and its 6-nitro derivative. Cancer Res., 45(12): 6225-6231.
- Godschalk, R.W., Ostertag, J.U., Moonen, E.J., Neumann, H.A., Kleinjans, J.C. and van Schooten, F.J. (1998). Aromatic DNA adducts in human white blood cells and skin after dermal application of coal tar. Cancer Epidemiol. Biomarkers Prev., 7(9): 767-773.
- Government of Canada (1991). Toxic chemicals in the Great Lakes and associated effects. Vol. I. Contaminant levels and trends. Vol. II. Effects. Environment Canada, Department of Fisheries and Oceans and Health Canada, Ottawa, Ontario (En 37-95/1990-1E).
- Grova, N., Schroeder, H., Farinelle, S., Prodhomme, E., Valley, A. and Muller, C.P. (2008). Sub-acute administration of benzo[a]pyrene (B[a]P) reduces anxiety-related behaviour in adult mice and modulates regional expression of N-methyl-d-aspartate (NMDA) receptors genes in relevant brain regions. Chemosphere, 73(1 Suppl.): S295-S302.
- Gunter, M.J., Probst-Hensch, N.M., Cortessis, V.K., Kulldorff, M., Haile, R.W. and Sinha, R. (2005). Meat intake, cooking-related mutagens and risk of colorectal adenoma in a sigmoidoscopy-based case-control study. Carcinogenesis, 26(3): 637-642.
- Gwinn, M.R. and Weston, A. (2008). Application of oligonucleotide microarray technology to toxic occupational exposures. J. Toxicol. Environ. Health A, 71(5): 315-324.
- Gwinn, M.R., Keshava, C., Olivero, O.A., Humsi, J.A., Poirier, M.C. and Weston, A. (2005). Transcriptional signatures of normal human mammary epithelial cells in response to benzo[a]pyrene exposure: a comparison of three microarray platforms. OMICS, 9(4): 334-350.
- Habs, M., Schmahl, D. and Misfeld, J. (1980). Local carcinogenicity of some environmentally relevant polycyclic aromatic hydrocarbons after lifelong topical application to mouse skin. Arch. Geschwulstforsch., 50: 266-274.
- Habs, M., Jahn, S.A.A. and Schmahl, D. (1984). Carcinogenic activity of condensate from coloquint seeds (Citrullus colocynthis) after chronic epicutaneous administration to mice. J. Cancer Res. Clin. Oncol., 108(1): 154-156.
- Haddad, S., Withey, J., Laparé, S., Law, F. and Krishnan, K. (1998). Physiologically-based pharmacokinetic modeling of pyrene in the rat. Environ. Toxicol. Pharmacol., 5(4): 245-255.
- Hakami, R., Mohtadinia, J., Etemadi, A., Kamangar, F., Nemati, M., Pourshams, A., Islami, F., Nasrollahzadeh, D., Saberi-Firoozi, M., Birkett, N., Boffetta, P. and Malekzadeh, R. (2008). Dietary intake of benzo[a]pyrene and risk of esophageal cancer in north of Iran. Nutr. Cancer, 60(2): 216-221.
- Halappanavar, S., Wu, D., Williams, A., Kuo, B., Godschalk, R.W., Van Schooten, F.J. and Yauk, C.L. (2011). Pulmonary gene and microRNA expression changes in mice exposed to benzo[a]pyrene by oral gavage. Toxicology, 285(3): 133-141.
- Hammond, D. and O'Connor, R.J. (2008). Constituents in tobacco and smoke emissions from Canadian cigarettes. Tobacco Control, 17: 24-31.
- Harper, P.A., Wong, J.M.Y., Lam, M.S.M. and Okey, A.B. (2002). Polymorphisms in the human AH receptor. Chem. Biol. Interact., 141(1-2): 161-187.
- Hattemer-Frey, H.A. and Travis, C.C. (1991). Benzo-a-pyrene: environmental partitioning and human exposure. Toxicol. Ind. Health, 7(3): 141-157.
- Haugen, A., Becher, G., Benestad, C., Vahakangas, K., Trivers, G.E., Newman, M.J. and Harris, C.C. (1986). Determination of polycyclic aromatic hydrocarbons in the urine, benzo[a]pyrene diol epoxide-DNA adducts in lymphocyte DNA, and antibodies to the adducts in sera from coke oven workers exposed to measured amounts of polycyclic aromatic hydrocarbons in the work atmosphere. Cancer Res., 46(8): 4178-4183.
- He, S. and Baker, R.S.U. (1991). Micronuclei in mouse skin cells following in vivo exposure to benzo[a]pyrene, 7,12-dimethylbenz[a]anthracene, chrysene, pyrene and urethane. Environ. Mol. Mutagen., 17(3): 163-168.
- Hemminki, K., Grzybowska, E., Chorazy, M., Twardowska-Saucha, K., Sroczynski, J.W., Putman, K.L., Randerath, K., Phillips, D.H. and Hewer, A. (1990). Aromatic DNA adducts in white blood cells of coke workers. Int. Arch. Occup. Environ. Health, 62(6): 467-470.
- Heredia-Ortiz, R., Bouchard, M., Marie-Desvergne, C., Viau, C. and Maitre, A. (2011). Modeling of the internal kinetics of benzo[a]pyrene and 3-hydroxybenzo[a]pyrene biomarker from rat data. Toxicol. Sci., 122(2): 275-287.
- Hernandez, L.G., Slob, W., van Steeg, H. and van Benthem, J. (2011). Can carcinogenic potency be predicted from in vivo genotoxicity data?: a meta-analysis of historical data. Environ. Mol. Mutagen., 52(7): 518-528.
- Hirano, M., Tanaka, S. and Asami, O. (2013). Classification of polycyclic aromatic hydrocarbons based on mutagenicity in lung tissue through DNA microarray. Environ. Toxicol., 28(11): 652-659.
- Hockley, S.L., Arlt, V.M., Brewer, D., Giddings, I. and Phillips, D.H. (2006). Time- and concentration-dependent changes in gene expression induced by benzo[a]pyrene in two human cell lines, MCF-7 and HepG2. BMC Genomics, 7: 260 (www.biomedcentral.com/1471-2164/7/260).
- Hockley, S.L., Arlt, V.M., Jahnke, G., Hartwig, A., Giddings, I. and Phillips, D.H. (2008). Identification through microarray gene expression analysis of cellular responses to benzo[a]pyrene and its diol-epoxide that are dependent or independent of p53. Carcinogenesis, 29(1): 202-210.
- Hockley, S.L., Mathijs, K., Staal, Y.C.M., Brewer, D., Giddings, I., Van Delft, J.H.M. and Phillips, D.H. (2009). Interlaboratory and interplatform comparison of microarray gene expression analysis of HepG2 cells exposed to benzo[a]pyrene. OMICS, 13(2): 115-125.
- Hrubá, E., Vondrácek, J., Líbalová, H., Topinka, J., Bryja, V., Soucek, K. and Machala, M. (2011). Gene expression changes in human prostate carcinoma cells exposed to genotoxic and nongenotoxic aryl hydrocarbon receptor ligands. Toxicol. Lett., 206(2): 178-188.
- HSDB (2011). Benzo[a]pyrene. Hazardous Substances Data Bank, U.S. National Library of Medicine, Bethesda, Maryland. Available at: http://toxnet.nlm.nih.gov
- IARC (2010). Some non-heterocyclic polycyclic aromatic hydrocarbons and some related exposures. IARC Monogr. Eval. Carcinog. Risks Hum., 92: 1-853. Available at: http://monographs.iarc.fr/ENG/Monographs/vol92/mono92-6.pdf
- IARC (2012). A review of human carcinogens: chemical agents and related occupations. IARC Monogr. Eval. Carcinog. Risks Hum., 100F: 111-144. Available at: http://monographs.iarc.fr/ENG/Monographs/vol100F/mono100F-14.pdf
- Irwin, R.J. (1997). Benzo[a]pyrene (BaP). In: Environmental contaminants encyclopedia. Water Operations Branch, Water Resources Division, U.S. National Park Service, Fort Collins, Colorado.
- Izzotti, A., Balansky, R., Scatolini, L. and Coscia, N. (1991). Research on adducts of benzo[a]pyrene and DNA in different cells using synchronous fluorescence spectrophotometry. Boll. Soc. Ital. Biol. Sper., 67(9): 869-874.
- Jin, X., Peldszus, S. and Huck, P.M. (2012). Reaction kinetics of selected micropollutants in ozonation and advanced oxidation processes. Water Res., 46(19): 6519-6530.
- Jules, G.E., Pratap, S., Ramesh, A. and Hood, D.B. (2012). In utero exposure to benzo[a]pyrene predisposes offspring to cardiovascular dysfunction in later-life. Toxicology, 295(1-3): 56-67.
- Kao, J., Patterson, F.K. and Hall, J. (1985). Skin penetration and metabolism of topically applied chemicals in six mammalian species, including man: an in vitro study with benzo[a]pyrene and testosterone. Toxicol. Appl. Pharmacol., 81(3): 502-516.
- Kazerouni, N., Sinha, R., Hsu, C.H., Greenberg, A. and Rothman, N. (2001). Analysis of 200 food items for benzo[a]pyrene and estimation of its intake in an epidemiologic study. Food Chem. Toxicol., 39(5): 423-436.
- Keshava, C., Whipkey, D. and Weston, A. (2005). Transcriptional signatures of environmentally relevant exposures in normal human mammary epithelial cells: benzo[a]pyrene. Cancer Lett., 221(2): 201-211.
- Knaapen, A.M., Curfs, D.M., Pachen, D.M., Gottschalk, R.W., de Winther, M.P., Daemen, M.J. and Van Schooten, F.J. (2007). The environmental carcinogen benzo[a]pyrene induces expression of monocyte-chemoattractant protein-1 in vascular tissue: a possible role in atherogenesis. Mutat. Res., 621(1-2): 31-41.
- Knuckles, M.E., Inyang, F. and Ramesh, A. (2001). Acute and subchronic oral toxicities of benzo[a]pyrene in F-344 rats. Toxicol. Sci., 61(2): 382-388.
- Kolpin, D.W., Furlong, E.T., Meyer, M.T., Thurman, E.M., Zaugg, S.D., Barber, L.B., Buxton, H.T. (2002). Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999-2000: a national reconnaissance. Environ. Sci. Technol., 36(6): 1202-1211.
- Krishnan, K. and Carrier, R. (2008). Approaches for evaluating the relevance of multiroute exposures in establishing guideline values for drinking water contaminants. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev., 26(3): 300-316.
- Krishnan, K. and Carrier, R. (2013). The use of exposure source allocation factor in the risk assessment of drinking-water contaminants. J. Toxicol. Environ. Health B Crit. Rev., 16(1): 39-51.
- Kristensen, P., Eilertsen, E., Einarsdottir, E., Haugen, A., Skaug, V. and Ovrebo, S. (1995). Fertility in mice after prenatal exposure to benzo[a]pyrene and inorganic lead. Environ. Health Perspect., 103(6): 588-590.
- Kroese, E., Muller, J., Mohn, G. and Dortant, P.W. (2001). Tumorigenic effects in Wistar rats orally administered benzo[a]pyrene for two years (gavage studies). Implications for human cancer risks associated with oral exposure to polycyclic aromatic hydrocarbons. National Institute of Public Health and the Environment (RIVM), Bilthoven, Netherlands.
- Labib, S., Yauk, C., Williams, A., Arlt, V., Phillips, D.H., White, P. and Halappanavar, S. (2012). Sub-chronic oral exposure to benzo[a]pyrene leads to distinct transcriptomic changes in the lungs that are related to carcinogenesis. Toxicol. Sci., 129(1): 213-224.
- Lan, Q., Mumford, J.L., Shen, M., DeMarini, D.M., Bonner, M.R., He, X., Yeager, M., Welch, R., Chanock, S., Tian, L., Chapman, R.S., Zheng, T., Keohavong, P., Caporaso, N. and Rothman, N. (2004). Oxidative damage-related genes AKR1C3 and OGG1 modulate risks for lung cancer due to exposure to PAH-rich coal combustion emissions. Carcinogenesis, 25(11): 2177-2181.
- LaVoie, E.J., Amin, S., Hecht, S.S., Furuya, K. and Hoffmann, D. (1982). Tumour initiating activity of dihydrodiols of benzo[b]fluoranthene, benzo[j]fluoranthene, and benzo[k]fluoranthene. Carcinogenesis, 3(1): 49-52.
- Lemieux, C.L., Douglas, G.R., Gingerich, J., Phonethepswath, S., Torous, D.K., Dertinger, S.D., Phillips, D.H., Arlt, V.M. and White, P.A. (2011). Simultaneous measurement of benzo[a]pyrene-induced Pig-a and lacZ mutations, micronuclei and DNA adducts in Muta Mouse. Environ. Mol. Mutagen., 52(9): 756-765.
- Li, D., Day, R.S., Bondy, M.L., Sinha, R., Nguyen, N.T., Evans, D.B., Abbruzzese, J.L. and Hassan, M.M. (2007). Dietary mutagen exposure and risk of pancreatic cancer. Cancer Epidemiol. Biomarkers Prev., 16(4): 655-661.
- Li, J.Z., Pan, H.Y., Zheng, J.W., Zhou, X.J., Zhang, P., Chen, W.T. and Zhang, Z.Y. (2008). Benzo (a) pyrene induced tumorigenesity [sic] of human immortalized oral epithelial cells: transcription profiling. Chin. Med. J. (Engl.), 121(19): 1882-1890.
- Liu, Z., Muehlbauer, K.R., Schmeiser, H.H., Hergenhahn, M., Belharazem, D. and Hollstein, M.C. (2005). P53 mutations in benzo[a]pyrene-exposed human P53 knock-in murine fibroblasts correlate with P53 mutations in human lung tumors. Cancer Res., 65(7): 2583-2587.
- Lizarraga, D., Gaj, S., Brauers, K.J., Timmermans, L., Kleinjans, J.C. and Van Delft, J.H.M. (2012). Benzo[a]pyrene-induced changes in microRNA-mRNA networks. Chem. Res. Toxicol., 25(4): 838-849.
- Lu, X., Shao, J., Li, H. and Yu, Y. (2009). Early whole-genome transcriptional response induced by benzo[a]pyrene diol epoxide in a normal human cell line. Genomics, 93(4): 332-342.
- Lu, X., Shao, J., Li, H. and Yu, Y. (2010). Temporal gene expression changes induced by a low concentration of benzo[a]pyrene diol epoxide in a normal human cell line. Mutat. Res., 684(1-2): 74-80.
- Mackay, D. and Paterson, S. (1991). Evaluating the multimedia fate of organic chemicals: a level III fugacity model. Environ. Sci. Technol., 25(3): 427-436.
- Mackenzie, K.M. and Angevine, D.M. (1981). Infertility in mice exposed in utero to benzo[a]pyrene. Biol. Reprod., 24(1): 183-191.
- Maier, M., Maier, D. and Lloyd, B.J. (2000). Factors influencing the mobilisation of polycyclic aromatic hydrocarbons (PAHs) from the coal-tar lining of water mains. Water Res., 34(3): 773-786.
- Malik, A.I., Williams, A., Lemieux, C.L., White, P.A. and Yauk, C.L. (2012). Hepatic mRNA, microRNA, and miR-34a-target responses in mice after 28 days exposure to doses of benzo[a]pyrene that elicit DNA damage and mutation. Environ. Mol. Mutagen., 53(1): 10-21.
- Marie, C., Bouchard, M., Heredia-Ortiz, R., Viau, C. and Maître, A. (2010). A toxicokinetic study to elucidate 3-hydroxybenzo[a]pyrene atypical urinary excretion profile following intravenous injection of benzo[a]pyrene in rats. J. Appl. Toxicol., 30(5): 402-410.
- Mass, M.J., Jeffers, A.J., Ross, J.A., Nelson, G., Galati, A.J., Stoner, G.D. and Nesnow, S. (1993). Ki-ras oncogene mutations in tumors and DNA adducts formed by benz[j]aceanthrylene and benzo[a]pyrene in the lungs of strain A/J mice. Mol. Carcinog., 8(3): 186-192.
- Meinl, W., Ebert, B., Glatt, H. and Lampen, A. (2008). Sulfotransferase forms expressed in human intestinal Caco-2 and TC7 cells at varying stages of differentiation and role in benzo[a]pyrene metabolism. Drug Metab. Dispos., 36(2): 276-283.
- Michaelson, J.J., Trump, S., Rudzok, S., Gräbsch, C., Madureira, D.J., Dautel, F., Mai, J., Attinger, S., Schirmer, K., von Bergen, M., Lehmann, I. and Beyer, A. (2011). Transcriptional signatures of regulatory and toxic responses to benzo-[a]-pyrene exposure. BMC Genomics, 12: 502 (doi: 10.1186/1471-2164-12-502).
- Ministère du Développement durable, de l'Environnement, de la Faune et des Parcs du Québec (2013). Personal communication from C. Robert.
- Moffat, I., Chepelev, N.L., Labib, S., Bourdon-Lacombe, J., Kuo, B., Buick, J.K., Lemieux, F., Williams, A., Halappanavar, S., Malik, A.I., Luijten, M., Aubrecht, J., Hyduke, D.R., Fornace, A.J. Jr, Swartz, C.D., Recio, L. and Yauk, C.L. (2015). Comparison of toxicogenomics and traditional approaches to inform mode of action and points of departure in human health risk assessment of benzo[a]pyrene in drinking water. Crit. Rev. Toxicol., 45(1):1-43.
- Moir, D., Viau, A., Chu, I., Withey, J. and McMullen, E. (1998). Pharmacokinetics of benzo[a]pyrene in the rat. J. Toxicol. Environ. Health, 53(7): 507-530.
- Mullaart, E., Buytenhek, M., Brouwer, A., Lohman, P.H.M. and Vijg, J. (1989). Genotoxic effects of intragastrically administered benzo[a]pyrene in rat liver and intestinal cells. Carcinogenesis, 10(2): 393-395.
- Mumford, J.L., Lee, X., Lewtas, J., Young, T.L. and Santella, R.M. (1993). DNA adducts as biomarkers for assessing exposure to polycyclic aromatic hydrocarbons in tissues from Xuan Wei women with high exposure to coal combustion emissions and high lung cancer mortality. Environ. Health Perspect., 99: 83-87.
- Nakamura, B.N., Mohar, I., Lawson, G.W., Cortes, M.M., Hoang, Y.D., Ortiz, L., Patel, R., Rau, B.A., McConnachie, L.A., Kavanagh, T.J. and Luderer, U. (2012). Increased sensitivity to testicular toxicity of transplacental benzo[a]pyrene exposure in male glutamate cysteine ligase modifier subunit knockout (Gclm−/−) mice. Toxicol. Sci., 126(1): 227-241.
- Neal, J. and Rigdon, R.H. (1967). Gastric tumors in mice fed benzo[a]pyrene: a quantitative study. Tex. Rep. Biol. Med., 25(4): 553-557.
- New Brunswick Department of Health and Wellness (2013). Personal communication with K. Gould.
- Nova Scotia Department of Environment and Labour (2013). Personal communication with C. Mosher.
- NSF/ANSI (2013). NSF International/American National Standards Institute Standard 61: Drinking water system components-health effects. NSF International, Ann Arbor, Michigan.
- OEHHA (2010). Public health goals for chemicals in drinking water: benzo(a)pyrene. Office of Environmental Health Hazard Assessment, California Environmental Protection Agency, Sacramento, California. Available at: www.oehha.ca.gov/water/phg/pdf/091610Benzopyrene.pdf
- Ontario Ministry of the Environment (2013). Personal communication from S. Deshpande.
- Pan, G., Hanaoka, T., Yamano, Y., Hara, K., Ichiba, M., Wang, Y., Zhang, J., Feng, Y., Shujuan, Z., Guan, D., Gao, G., Liu, N. and Takahashi, K. (1998). A study of multiple biomarkers in coke oven workers-A cross-sectional study in China. Carcinogenesis, 19(11): 1963-1968.
- Perera, F.P., Hemminki, K., Young, T.L., Brenner, D., Kelly, G. and Santella, R.M. (1988). Detection of polycyclic aromatic hydrocarbon-DNA adducts in white blood cells of foundry workers. Cancer Res., 48(8): 2288-2291.
- Perera, F.P., Rauh, V., Whyatt, R.M., Tsai, W.Y., Bernert, J.T., Tu, Y.H., Andrews, H., Ramirez, J., Qu, L. and Tang, D. (2004). Molecular evidence of an interaction between prenatal environmental exposure and birth outcomes in a multiethnic population. Environ. Health Perspect., 112: 626-630.
- Perera, F.P., Tang, D., Rauh, V., Lester, K., Tsai, W.Y., Tu, Y.H., Weiss, L., Hoepner, L., Del Priore, G. and Lederman, S.A. (2005). Relationship among polycyclic aromatic hydrocarbon-DNA adducts, proximity to the World Trade Center, and effects on fetal growth. Environ. Health Perspect., 113: 1062-1067.
- Phillips, D.H. (1999). Polycyclic aromatic hydrocarbons in the diet. Mutat. Res., 443: 139-147.
- Poel, W.E. (1959). Effect of carcinogenic dosage and duration of exposure on skin-tumor induction in mice. J. Natl. Cancer Inst., 22: 19-43.
- Poel, W.E. (1963). Skin as a test site for the bioassay of carcinogens and carcinogen precursors. Natl. Cancer Inst. Monogr., 10: 611-625.
- Proctor, D.M., Gatto, N.M., Hong, S.J. and Allamneni, K.P. (2007). Mode-of-action framework for evaluating the relevance of rodent forestomach tumors in cancer risk assessment. Toxicol. Sci., 98(2): 313-326.
- Pucknat, A.W. (ed.) (1981). Health impacts of polynuclear aromatic hydrocarbons. Noyes Data Corp., Park Ridge, NJ. 271 pp. (Environmental Health Review No. 5).
- Ramesh, A., Greenwood, M., Inyang, F. and Hood, D.B. (2001a). Toxicokinetics of inhaled benzo[a]pyrene: plasma and lung bioavailability. Inhal. Toxicol., 13(6): 533-555.
- Ramesh, A., Inyang, F., Hood, D.B., Archibong, A.E., Knuckles, M.E. and Nyanda, A.M. (2001b). Metabolism, bioavailability, and toxicokinetics of benzo(α)pyrene in F-344 rats following oral administration. Exp. Toxicol. Pathol., 53(4): 275-290.
- Ramesh, A., Hood, D.B., Inyang, F., Greenwood, M., Nyanda, A.M., Archibong, A.E. and Knuckles, M.E. (2002). Comparative metabolism, bioavailability, and toxicokinetics of benzo[a]pyrene in rats after acute oral, inhalation, and intravenous administration. Polycycl. Aromat. Compd., 22(3-4): 969-980.
- Ringuette, S., Germain, A., Gonthier, C. and Perron, F. (1993). Presence of PAHs in the Canadian environment: an overview. Prepared for Environment Canada, Conservation and Protection, Quebec Region, Montréal, Quebec (Priority Substances List Supporting Document No. 2).
- Roe, F.J., Peto, R., Kearns, F. and Bishop, D. (1970). The mechanism of carcinogenesis by the neutral fraction of cigarette smoke condensate. Br. J. Cancer, 24: 788-806.
- Roszinsky-Kocher, G., Basler, A. and Roehrborn, G. (1979). Mutagenicity of polycyclic hydrocarbons. V. Induction of sister-chromatid exchanges in vivo. Mutat. Res., 66(1): 65-67.
- RTECS (1994). Registry of Toxic Effects of Chemical Substances, MEDLARS Online Information Retrieval System, U.S. National Library of Medicine, Bethesda, Maryland.
- Salamone, M.F. (1981). Toxicity of 41 carcinogens and noncarcinogenic analogs. In: de Serres, F.J. and Ashby, J. (eds.). Progress in mutation research. Vol. 1. Evaluation of short-term tests for carcinogens. Report of the international corroborative program. Elsevier-North Holland, Amsterdam, Netherlands. pp. 682-685.
- Sanches, S., Leitão, C., Penetra, A., Cardoso, V.V., Ferreira, E., Benoliel, M.J., Crespo, M.T. and Pereira, V.J. (2011). Direct photolysis of polycyclic aromatic hydrocarbons in drinking water sources. J. Hazard. Mater., 192(3): 1458-1465.
- Santodonato, J., Howard, P. and Basu, D. (1981). Health and ecological assessment of polynuclear aromatic hydrocarbons. J. Environ. Pathol. Toxicol., 5(1): 1-364.
- Saskatchewan Water Security Agency (2013). Personal communication from S. Ferris.
- Saunders, C.R., Das, S.K., Ramesh, A., Shockley, D.C. and Mukherjee, S. (2006). Benzo[a]pyrene-induced acute neurotoxicity in the F-344 rat: role of oxidative stress. J. Appl. Toxicol., 26(5): 427-438.
- SCC (2013). Directory of accredited product, process and service certification bodies. Standards Council of Canada, Ottawa, Ontario. Available at: www.scc.ca/en/accreditation/product-process-and-service-certification/directory-of-accredited-clients
- Shendrikova, I.A. and Aleksandrov, V.A. (1974). Comparative penetration of polycyclic hydrocarbons through the rat placenta into the fetus. Bull. Exp. Biol. Med., 77(2): 169-171.
- Shertzer, H.G., Clay, C.D., Genter, M.B., Chames, M.C., Schneider, S.N., Oakley, G.G., Nebert, D.W. and Dalton, T.P. (2004). Uncoupling-mediated generation of reactive oxygen by halogenated aromatic hydrocarbons in mouse liver microsomes. Free Radic. Biol. Med., 36(5): 618-631.
- Shi, Z., Dragin, N., Gálvez-Peralta, M., Jorge-Nebert, L.F., Miller, M.L., Wang, B. and Nebert, D.W. (2010). Organ-specific roles of CYP1A1 during detoxication of dietary benzo[a]pyrene. Mol. Pharmacol., 78(1): 46-57.
- Shimada, T., Martin, M.V., Pruess-Schwartz, D., Marnett, L.J. and Guengerich, F.P. (1989). Roles of individual human cytochrome P-450 enzymes in the bioactivation of benzo[a]pyrene, 7,8-dihydroxy-7,8-dihydrobenzo[a]pyrene, and other dihydrodiol derivatives of polycyclic aromatic hydrocarbons. Cancer Res., 49(22): 6304-6312.
- Shimizu, Y., Nakatsuru, Y., Ichinose, M., Takahashi, Y., Kume, H., Mimura, J., Fujii-Kuriyama, Y. and Ishikawa, T. (2000). Benzo[a]pyrene carcinogenicity is lost in mice lacking the aryl hydrocarbon receptor. Proc. Natl. Acad. Sci. U.S.A., 97(2): 779-782.
- Shum, S., Jensen, N.M. and Nebert, D.W. (1979). The murine Ah locus: in utero toxicity and teratogenesis associated with genetic differences in benzo[a]pyrene metabolism. Teratology, 20(3): 365-376.
- Sinha, R., Kulldorff, M., Gunter, M.J., Strickland, P. and Rothman, N. (2005a). Dietary benzo[a]pyrene intake and risk of colorectal adenoma. Cancer Epidemiol. Biomarkers Prev., 14(8): 2030-2034.
- Sinha, R., Peters, U., Cross, A.J., Kulldorff, M., Weissfeld, J.L., Pinsky, P.F., Rothman, N. and Hayes, R.B. (2005b). Meat, meat cooking methods and preservation, and risk for colorectal adenoma. Cancer Res., 65(17): 8034-8041.
- Sivak, A., Niemeier, R., Lynch, D., Beltis, K., Simon, S., Salomon, R., Latta, R., Belinky, B., Menzies, K., Lunsford, A., Cooper, C, Ross, A. and Bruner, R. (1997). Skin carcinogenicity of condensed asphalt roofing fumes and their fractions following dermal application to mice. Cancer Lett., 117: 113-123.
- Slaga, T.J., Huberman, E., Selkirk, J.K., Harvey, R.G. and Bracken, W.M. (1978). Carcinogenicity and mutagenicity of benz(a)anthracene diols and diol-epoxides. Cancer Res., 38: 1699-1704.
- Slaga, T.J., Gleason, G.L., Mills, G., Ewald, L., Fu, P.P., Lee, H.M. and Harvey, R.G. (1980). Comparison of the skin tumor-initiating activities of dihydrodiols and diol-epoxides of various polycyclic aromatic hydrocarbons. Cancer Res., 40: 1981-1984.
- Snoeyink, V.L. (1990). Adsorption of organic compounds. In: Water quality and treatment. 4th edition. American Water Works Association, Denver, Colorado.
- Snyder, S.A., Wert, E.C., Lei, H., Westerhoff, P. and Yoon, Y. (2007). Removal of EDCs and pharmaceuticals in drinking and reuse treatment processes. AWWA Research Foundation, American Water Works Association, Denver, Colorado (Report No. 91188).
- Sorrell, R.K., Brass, H.J. and Reding, R. (1980). A review of occurrences and treatment of polynuclear aromatic hydrocarbons in water. Environ. Int., 4(3): 245-254.
- Speth, T.F. (1990). Removal of volatile organic compounds from drinking water by adsorption. In: Ram, N.M., Christman, R.F. and Cantor, K.P. (eds.). Significance and treatment of volatile organic compounds in water supplies. Lewis Publishers, Chelsea, Michigan.
- Srinivasan, K.R. and Folger, H.S. (1990). Use of inorgano-organo-clays in the removal of priority pollutants from industrial wastewaters: adsorption of benzo[a]pyrene and chlorophenols from aqueous solutions. Clay Clay Miner., 38(3): 287-293.
- Stackelberg, P.E., Furlong, E.T., Meyer, M.T., Zaugg, S.D., Henderson, A.K. and Reissman, D.B. (2004). Persistence of pharmaceutical compounds and other organic wastewater contaminants in a conventional drinking-water-treatment plant. Sci. Total Environ., 329: 99-113.
- Stackelberg, P.E., Gibs, J., Furlong, E.T., Meyer, M.T., Zaugg, S.D. and Lippincott, R.L. (2007). Efficiency of conventional drinking-water-treatment processes in removal of pharmaceuticals and other organic compounds. Sci. Total Environ., 377: 255-272.
- Straif, K., Baan, R., Grosse, Y., Secretan, B., El Ghissassi, F., Cogliano, V., Drewski, D., Partanen, T., Vähäkangas, K., Stücker, I., Borlak, J., Feron, V.J., Marques, M.M., Gustavsson, P., Fletcher, T., Arey, J., Beland, F.A., Burchiel, S., Flowers, L., Herbert, R.A., Mukhtar, H., Nesnow, S., Penning, T.M., Sinha, R. and Shimada, T. (2005). Carcinogenicity of polycyclic aromatic hydrocarbons. Lancet Oncol., 6(12): 931-932.
- Sun, J.D., Wolff, R.K. and Kanapilly, G.M. (1982). Deposition, retention, and biological fate of inhaled benzo[a]pyrene adsorbed onto ultrafine particles and as a pure aerosol. Toxicol. Appl. Pharmacol., 65(2): 231-244.
- Tang, D., Li, T.Y, Liu, J.J., Chen, Y.H., Qu, L. and Perera, F. (2006). PAH-DNA adducts in cord blood and fetal and child development in a Chinese cohort. Environ. Health Perspect., 114: 1297-1300.
- Thyssen, J., Althoff, J., Kimmerle, G. and Mohr, U. (1981). Inhalation studies with benzo[a]pyrene in Syrian golden hamsters. J. Natl. Cancer Inst., 66(3): 575-577.
- Tram, K.L., Cross, A.J., Consonni, D., Randi, G., Bagnardi, V., Bertazzi, P.A., Caporaso, N.E., Sinha, R., Subar, A.F. and Landi, M.T. (2009). Intakes of red meat, processed meat, and meat mutagens increase lung cancer risk. Cancer Res., 69(3): 932-939.
- Trenholm, R.A., Vanderford, B.J., Holady, J.C., Rexing, D.J. and Snyder, S.A. (2006). Broad range analysis of endocrine disruptors and pharmaceuticals using gas chromatography and liquid chromatography tandem mass spectrometry. Chemosphere, 65: 1990-1998.
- Trush, M.A., Mimnaugh, E.G. and Gram, T.E. (1982). Activation of pharmacologic agents to radical intermediates. Implications for the role of free radicals in drug action and toxicity. Biochem. Pharmacol., 31(21): 3335-3346.
- Turusov, V.S., Nikonova, T.V. and Parfenov, Y.D. (1990). Increased multiplicity of lung adenomas in five generations of mice treated with benz(a)pyrene when pregnant. Cancer Lett., 55(3): 227-231.
- Uno, S., Dalton, T.P., Dragin, N., Curran, C.P., Derkenne, S., Miller, M.L., Shertzer, H.G., Gonzalez, F.J. and Nebert, D.W. (2006). Oral benzo[a]pyrene in Cyp1 knockout mouse lines: CYP1A1 important in detoxication, CYP1B1 metabolism required for immune damage independent of total-body burden and clearance rate. Mol. Pharmacol., 69(4): 1103-1114.
- Urso, P. and Gengozian, N. (1982). Alterations in the humoral immune response and tumor frequencies in mice exposed to benzo[a]pyrene and X-rays before or after birth. J. Toxicol. Environ. Health, 10(4-5): 817-835.
- Urso, P. and Gengozian, N. (1984). Subnormal expression of cell-mediated and humoral immune responses in progeny disposed toward a high incidence of tumors after in utero exposure to benzo[a]pyrene. J. Toxicol. Environ. Health, 14(4): 569-584.
- U.S. EPA (1990). Methods for determinations of organic compounds in drinking water-Supplement I. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC (EPA/600/4-90-020).
- U.S. EPA (1992). Drinking water; National Primary Drinking Water Regulations-synthetic organic chemicals and inorganic chemicals; National Primary Drinking Water Regulations implementation; final rule. Fed. Regist., 57(138): 31776.
- U.S. EPA (1995). Methods for determinations of organic compounds in drinking water-Supplement III. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC (EPA/600/R-95/131).
- U.S. EPA (1998). Small system compliance technology list for the non-microbial contaminants regulated before 1996. Office of Water, U.S. Environmental Protection Agency, Washington, DC (EPA 815-R-98-002).
- U.S. EPA (2003). Analytical feasibility support document for the six-year review of existing National Primary Drinking Water Regulations (reassessment of feasibility for chemical contaminants). Targeting and Analysis Branch, Standards and Risk Management Division, Office of Ground Water and Drinking Water, Office of Water, U.S. Environmental Protection Agency, Washington, DC (EPA 815-R-03-003). Available at: www.epa.gov/safewater/standard/review/pdfs/support_6yr_analytical_final.pdf
- U.S. EPA (2007). Benzo[a]pyrene TEACH chemical summary. Toxicity and Exposure Assessment for Children's Health, U.S. Environmental Protection Agency, Washington, DC. Available at: www.epa.gov/teach/chem_summ/BaP_summary.pdf
- U.S. EPA (2009a). Analytical feasibility support document for the second six-year review of existing National Primary Drinking Water Regulations. Office of Water, U.S. Environmental Protection Agency, Cincinnati, Ohio (EPA 815-B-09-003).
- U.S. EPA (2009b). Development of estimated quantitation levels for the second six-year review of National Primary Drinking Water Regulations. Office of Water, U.S. Environmental Protection Agency, Cincinnati, OH (EPA 815-B-09-005).
- U.S. EPA (2011). Benchmark Dose Software (BMDS) version 2.2. Software available for download from the U.S. Environmental Protection Agency. Available at: www.epa.gov/NCEA/bmds/index.html
- U.S. EPA (2013). Estimation Program Interface Suite™ for Microsoft®Windows, v. 4.11. U.S. Environmental Protection Agency, Washington, DC.
- Van de Wiel, J.A.G., Fijneman, P.H.S., Duijf, C.M.P., Anzion, R.B.M., Theuws, J.L.G. and Bos, R.P. (1993). Excretion of benzo[a]pyrene and metabolites in urine and feces of rats: influence of route of administration, sex and long-term treatment. Toxicology, 80(2-3): 103-115.
- Vreeburg, J. (2010). Discolouration in drinking water systems: the role of particles clarified. IWA Publishing, London, UK.
- Vu Duc, T. and Huynh, C.K. (1981). Les micropolluants organiques dans l'eau. Résultats préliminaires sur les haloformes et les hydrocarbures aromatiques polycycliques. Med. Soc. Prev., 26: 315-316.
- Wester, P.W., Muller, J.J.A., Slob, W., Mohn, G.R., Dortant, P.M. and Kroese, E.D. (2012). Carcinogenic activity of benzo[a]pyrene in a 2 year oral study in Wistar rats. Food Chem. Toxicol., 50(3-4): 927-935.
- Wester, R.C., Maibach, H.I., Bucks, D.A.W., Sedik, L., Melendres, J., Liao, C. and DiZio, S. (1990). Percutaneous absorption of [14C]DDT and [14C]benzo[a]pyrene from soil. Fundam. Appl. Toxicol., 15(3): 510-516.
- Westerhoff, P., Yoon, Y., Snyder, S. and Wert, E. (2005). Fate of endocrine-disruptor, pharmaceutical, and personal care product chemicals during simulated drinking water treatment processes. Environ. Sci. Technol., 39: 6649-6663.
- Weyand, E.H. and Bevan, D.R. (1986). Benzo[a]pyrene disposition and metabolism in rats following intratracheal instillation. Cancer Res., 46(11): 5655-5661.
- Weyand, E.H., He, Z.M., Ghodrati, F., Wu, Y., Marshall, M.V. and Lavoie, E.J. (1992). Effect of fluorine substitution on benzo[j]fluoranthene genotoxicity. Chem. Biol. Interact., 84: 37-53.
- Weyand, E.H., Chen, Y.C., Wu, Y., Koganti, A., Dunsford, H.A. and Rodriguez, L.V. (1995). Differences in the tumorigenic activity of a pure hydrocarbon and a complex mixture following ingestion: benzo[a]pyrene vs manufactured gas plant residue. Chem. Res. Toxicol., 8(7): 949-954.
- White, K.L., Jr. and Holsapple, M.P. (1984). Direct suppression of in vitro antibody production by mouse spleen cells by the carcinogen benzo[a]pyrene but not by the noncarcinogenic congener benzo(e)pyrene. Cancer Res., 44(8): 3388-3393.
- WHO (1998). Selected non-heterocyclic polycyclic aromatic hydrocarbons. International Programme on Chemical Safety, World Health Organization, Geneva, Switzerland (Environmental Health Criteria 202). Available at: www.inchem.org/documents/ehc/ehc/ehc202.htm
- WHO (2003). Polynuclear aromatic hydrocarbons in drinking-water: background document for development of WHO Guidelines for Drinking-water quality. World Health Organization, Geneva, Switzerland. Available at: WHO/SDE/WSH/03.04/59; www.who.int/water_sanitation_health/dwq/chemicals/polyaromahydrocarbons.pdf
- WHO (2011). Guidelines for drinking-water quality. 4th edition. World Health Organization, Geneva, Switzerland. Available at: www.who.int/water_sanitation_health/publications/2011/dwq_guidelines/en/index.html
- Wielgosz, S.M., Brauze, D. and Pawlak, A.L. (1991). Ah locus-associated differences in induction of sister-chromatid exchanges and in DNA adducts by benzo[a]pyrene in mice. Mutat. Res., 246(1): 129-137.
- WMS, Intera and NWRI (1989). Investigation of suspected PAH contamination of groundwater: Newcastle, New Brunswick. WMS Associates Ltd., Intera Technologies and National Water Research Institute, Fredericton, New Brunswick. 29 pp. (WMS 2988).
- Wojdani, A. and Alfred, L.J. (1984). Alterations in cell-mediated immune functions induced in mouse splenic lymphocytes by polycyclic aromatic hydrocarbons. Cancer Res., 44(3): 942-945.
- Wolff, M.S., Herbert, R., Marcus, M., Rivera, M., Landrigan, P.J. and Andrews, L.R. (1989). Polycyclic aromatic hydrocarbon (PAH) residues on skin in relation to air levels among roofers. Arch. Environ. Health, 44(3): 157-163.
- Wood, A.W., Levin, W., Chang, R.L., Huang, M.T., Ryan, D.E., Thomas, P.E., Lehr, R.E., Kumar, S., Koreeda, M., Akagi, H., Ittah, Y., Dansette, P., Yagi, H., Jerina, D.M. and Conney, A.H. (1980). Mutagenicity and tumor-initiation activity of cyclopenta(c,d)pyrene and structurally related compounds. Cancer Res., 40: 642-649.
- Wu, J., Ramesh, A., Nayyar, T. and Hood, D.B. (2003). Assessment of metabolites and AhR and CYP1A1 mRNA expression subsequent to prenatal exposure to inhaled benzo[a]pyrene. Int. J. Dev. Neurosci., 21(6): 333-346.
- Xia, Y., Cheng, S., He, J., Liu, X., Tang, Y., Yuan, H., He, L., Lu, T., Tu, B. and Wang, Y. (2011). Effects of subchronic exposure to benzo[a]pyrene (B[a]P) on learning and memory, and neurotransmitters in male Sprague-Dawley rat. Neurotoxicology, 32(2): 188-198.
- Xu, C., Chen, J.A., Qiu, Z., Zhao, Q., Luo, J., Yang, L., Zeng, H., Huang, Y., Zhang, L., Cao, J. and Shu, W. (2010). Ovotoxicity and PPAR-mediated aromatase downregulation in female Sprague-Dawley rats following combined oral exposure to benzo[a]pyrene and di-(2-ethylhexyl) phthalate. Toxicol. Lett., 199(3): 323-332.
- Xu, Z., Brown, L.M., Pan, G.W., Liu, T.F., Gao, G.S., Stone, B.J., Cao, R.M., Guan, D.X., Sheng, J.H., Yan, Z.S., Dosemeci, M., Fraumeni, J.F., Jr. and Blot, W.J. (1996). Cancer risks among iron and steel workers in Anshan, China, Part II: Case-control studies of lung and stomach cancer. Am. J. Ind. Med., 30(1): 7-15.
- Yang, H., Zhou, L., Wang, Z., Roberts, L.J., II, Lin, X., Zhao, Y. and Guo, Z. (2009). Overexpression of antioxidant enzymes in ApoE-deficient mice suppresses benzo[a]pyrene-accelerated atherosclerosis. Atherosclerosis, 207(1): 51-58.
- Yauk, C.L., Jackson, K., Malowany, M. and Williams, A. (2011). Lack of change in microRNA expression in adult mouse liver following treatment with benzo[a]pyrene despite robust mRNA transcriptional response. Mutat. Res., 722(2): 131-139.
- Yerushalmi, L., Nefil, S., Hausler, R. and Guiot, S.R. (2006). Removal of pyrene and benzo[a]pyrene from contaminated water by sequential and simultaneous ozonation and biotreatment. Water Environ. Res., 78(11): 2286-2292.
- Zheng, S.J., Tian, H.J., Cao, J. and Gao, Y.Q. (2010). Exposure to di(n-butyl)phthalate and benzo[a]pyrene alters IL-1β secretion and subset expression of testicular macrophages, resulting in decreased testosterone production in rats. Toxicol. Appl. Pharmacol., 248(1): 28-37.
Appendix A: List of acronyms
- AhR
- aryl hydrocarbon receptor
- ANSI
- American National Standards Institute
- ApoE
- apoliprotein E
- AOP
- advanced oxidation process
- BaP
- benzo[a]pyrene
- BMD
- benchmark dose
- BMD10
- benchmark dose for a 10% change in effect
- BMDL10
- 95% lower confidence limit on the benchmark dose for a 10% change in effect
- BPDE
- benzo[a]pyrene-7,8-diol-9,10-epoxide
- bw
- body weight
- CI
- confidence interval
- CSF
- cancer slope factor
- CYP
- cytochrome P450
- DNA
- deoxyribonucleic acid
- EPA
- Environmental Protection Agency (U.S.)
- EQL
- estimated quantitation level
- F
- filial generation (e.g., F1)
- FC
- fold change
- FDR
- false discovery rate
- GAC
- granular activated carbon
- GC/MS
- gas chromatography/mass spectrometry
- GC/MS/MS
- gas chromatography with tandem mass spectrometry
- HBV
- health-based value
- HPLC
- high-performance liquid chromatography
- IARC
- International Agency for Research on Cancer
- IOC
- inorgano-organo-clay
- Kow
- octanol-water partition coefficient
- LD50
- median lethal dose
- LLE
- liquid-liquid extraction
- LOAEL
- lowest-observed-adverse-effect level
- LSE
- liquid-solid extraction
- MAC
- maximum acceptable concentration
- MDL
- method detection limit
- MOA
- mode of action
- MRL
- minimum reporting level
- NF
- nanofiltration
- NOM
- natural organic matter
- NSF
- NSF International
- PAC
- powder activated carbon
- PAH
- polycyclic aromatic hydrocarbon
- PBPK
- physiologically based pharmacokinetic
- PQL
- practical quantitation level
- SCC
- Standards Council of Canada
- UF
- ultrafiltration
- UV
- ultraviolet
- WHO
- World Health Organization

Download the alternative format
(PDF format, 1.41 MB, 2 pages)
Organization: Health Canada
Type: Guidelines
Published: 2016-01