Guidelines for Canadian Drinking Water Quality: Guideline Technical Document – Escherichia coli

Download the alternative format
(PDF format, 838 KB, 55 pages)
Organization: Health Canada
Date published: March 2020
Table of Contents
- Part I. Overview and Application
- Part II. Science and Technical Considerations
- 4.0 Significance of E. coli in drinking water
- 5.0 Analytical methods
- 6.0 Sampling for E. coli
- 7.0 Treatment technology and distribution system considerations
- 8.0 Risk assessment
- 9.0 Rationale
- 10.0 References
- Appendix A: Decision tree for routine microbiological testing of municipal-scale systems
- Appendix B: Decision tree for routine microbiological testing of residential-scale systems
- Appendix C: List of acronyms
Part I. Overview and Application
1.0 Guideline
The maximum acceptable concentration (MAC) for Escherichia coli in drinking water is none detectable per 100 mL.
2.0 Executive summary
This guideline technical document was prepared in collaboration with the Federal-Provincial-Territorial Committee on Drinking Water and assesses all available information on Escherichia coli.
Escherichia coli (E. coli) is a species of bacteria that is naturally found in the intestinal system of humans and animals. It is present in feces in high numbers and can be easily measured in water, which makes it a useful indicator of fecal contamination for drinking water providers. E. coli is the most widely used indicator for detecting fecal contamination in drinking water supplies worldwide. In drinking water monitoring programs, E. coli testing is used to provide information on the quality of the source water, the adequacy of treatment and the safety of the drinking water distributed to the consumer.
2.1 Significance of E. coli in drinking water systems and their sources
E. coli monitoring should be used, in conjunction with other indicators, as part of a multi-barrier approach to producing drinking water of an acceptable quality. Drinking water sources are commonly impacted by fecal contamination from either human or animal sources and, as a result, may contain E. coli. Its presence in a water sample is considered a good indicator of recent fecal contamination. The ability to detect fecal contamination in drinking water is a necessity, as pathogenic microorganisms from human and animal feces in drinking water pose the greatest danger to public health.
Under a risk management approach to drinking water systems such as a source-to-tap or water safety plan approach, monitoring for E. coli is used as part of the water quality verification process to show that the natural and treatment barriers in place are providing the necessary level of control needed. The detection of E. coli in drinking water indicates fecal contamination and therefore that fecal pathogens may be present which can pose a health risk to consumers. In a groundwater source, the presence of E. coli indicates that the groundwater has been affected by fecal contamination, while in treated drinking water the presence of E. coli can signal that treatment is inadequate or that the treated water has become contaminated during distribution. If testing confirms the presence of E. coli in drinking water, actions that can be taken include notifying the responsible authorities, using a boil water advisory and implementing corrective actions.
Using multiple parameters in drinking water verification monitoring as indicators of general microbiological water quality (such as total coliforms, heterotrophic plate counts) or additional indicators of fecal contamination (enterococci) is a good way for water utilities to enhance the potential to identify issues and thus trigger responses.
2.2 Treatment
Surface water or groundwater under the direct influence of surface waters (GUDI) systems that meet the guidelines for enteric protozoa and enteric viruses (minimum 3 log removal and/or inactivation and minimum 4 log removal and/or inactivation, respectively) and groundwater systems that meet the guidelines for enteric viruses (minimum 4 log removal and/or inactivation), will be capable of achieving the MAC of none detectable per 100 mL for E. coli. Detecting E. coli in drinking water indicates that there is a potential health risk from consuming the water; however, E. coli testing on its own is not able to confirm the presence or absence of drinking water pathogens.
For municipal-scale systems, it is important to apply a monitoring approach which includes the use of multiple operational and water quality verification parameters (e.g., turbidity, disinfection measurements, E. coli), in order to verify that the water has been adequately treated and is therefore of an acceptable microbiological quality. For residential-scale systems, regular E. coli testing combined with monitoring of critical processes, regular physical inspections and a source water assessment can be used to confirm the quality of the drinking water supply.
2.3 International considerations
The MAC for E. coli is consistent with drinking water guidelines established by other countries and international organizations. The World Health Organization (WHO), the European Union (EU), the United States Environmental Protection Agency (U.S. EPA) and the Australian National Health and Medical Research Council have all established a limit of zero E. coli per 100 mL.
3.0 Application of the guideline
Note: Specific guidance related to the implementation of drinking water guidelines should be obtained from the responsible drinking water authority in the affected jurisdiction.
E. coli is the most widely used fecal indicator organism in drinking water risk management worldwide. For municipal-scale and residential-scaleFootnote 1 systems, its primary role is as an indicator of fecal contamination during routine monitoring to verify the quality of the drinking water supply. The presence of E. coli indicates fecal contamination of the drinking water and as a result, there is an increased risk that enteric pathogens may be present. For treated, distributed drinking water, the detection of E. coli is a signal of inadequate control or of an operational failure in the drinking water treatment or distribution system. Consequently, the detection of E. coli in any drinking water system is unacceptable.
Fecal contamination is often intermittent and may not be revealed by the examination of a single sample. Therefore, if a vulnerability assessment or inspection of a drinking water system shows that an untreated supply or treated water (e.g., during distribution and storage) is subject to fecal contamination, or that treatment is inadequate, the water should be considered unsafe, irrespective of the results of E. coli analysis. Implementing a risk management approach to drinking water systems, such as the source-to-tap or water safety plan approach is the best method to reduce waterborne pathogens in drinking water. These approaches require a system assessment that involves: characterizing the water source; describing the treatment barriers that prevent or reduce contamination; highlighting the conditions that can result in contamination; and implementing control measures to mitigate those risks through the treatment and distribution systems to the consumer.
E. coli concentrations of none detectable per 100 mL of water leaving the treatment plant should be achieved for all treated water supplies. Treatment of surface water sources or GUDI should include adequate filtration (or technologies providing an equivalent log reduction credit) and disinfection. Treatment of groundwater sources should include a minimum 4 log (99.99%) removal and/or inactivation of enteric viruses. A jurisdiction may choose to allow a groundwater source to have less than the recommended minimum 4 log reduction if the assessment of the drinking water system meets the provincial or territorial requirements in place to ensure that the risk of enteric virus presence is minimal. Surface water and GUDI systems that meet the guidelines for enteric protozoa and enteric viruses (minimum 3 log removal and/or inactivation and minimum 4 log removal and/or inactivation, respectively), and groundwater systems that meet the guidelines for enteric viruses (minimum 4 log removal and/or inactivation), are capable of providing adequate removal and/or inactivation of E. coli. Source water assessments may determine that log reductions greater than the minimum requirements are necessary to produce water of an acceptable microbiological quality.
The appropriate type and level of treatment should take into account the potential fluctuations in water quality, including short-term water quality degradation, and variability in treatment performance. Pilot testing or optimization processes may be useful for determining treatment variability. In systems with a distribution system, a disinfectant residual should be maintained throughout the system at all times. The existence of an adequate disinfectant residual is an important measure for controlling microbial growth during drinking water distribution. Under some conditions (e.g., the intrusion of viruses or protozoa from outside of the distribution system), the disinfectant residual may not be sufficient to ensure effective pathogen inactivation. More information on how source water assessments and, treatment technologies and distribution system operations are used to manage risks from pathogens in drinking water can be found in Health Canada's guideline technical documents on enteric protozoa and on enteric viruses. When verifying the quality of treated drinking water, the results of E. coli tests should be considered together with information on treatment and distribution system performance to show that the water has been adequately treated and is therefore of acceptable microbiological quality. Water system owners should contact the appropriate drinking water authority in the affected jurisdiction to confirm the specific requirements that apply to their system.
3.1 Municipal-scale drinking water supply systems
3.1.1 Monitoring E. coli in water leaving the treatment plant
E. coli should be monitored at least weekly in water leaving a treatment plant. If E. coli is detected, this indicates a serious breach in treatment and is therefore unacceptable. E. coli tests should be used in conjunction with other operational indicators, such as residual disinfectant and turbidity monitoring as part of a source-to-tap or water safety plan approach.
The required frequency for all testing at the treatment plant is specified by the responsible drinking water authority. Best practice commonly involves a testing frequency beyond these minimum recommendations based upon the size of system, the number of consumers served, the history of the system, and other site-specific considerations, such as the results of source water assessments. Events that lead to changes in source water conditions (e.g., spring runoff, storms or wastewater spills) are associated with an increased risk of fecal contamination. Water utilities should consider additional sampling during these events.
3.1.2 Monitoring E. coli within water distribution and storage systems
In municipal-scale distribution and storage systems, the number of samples collected for E. coli testing should reflect the size of the population being served, with a minimum of four samples per month. The frequency and sampling points for E. coli testing within distribution and storage systems will be specified and/or approved by the responsible drinking water authority.
Changes to system conditions that result in an interruption of supply or cause low and negative transient pressures can be associated with an increased risk of fecal contamination. These changes can occur during routine distribution system operation/maintenance (e.g., pump start/stops, valve opening and closing) or unplanned events such as power outages or water main breaks. Operational indicators (e.g., disinfectant residual, pressure monitoring) should be used in conjunction with E. coli tests as part of a source-to-tap or water safety plan approach.
3.1.3 Notification
If E. coli is detected in a sample of drinking water from a municipal-scale drinking water system (i.e. water leaving a treatment plant, or in a distribution or storage system), the system owner/operator and the laboratory processing the samples should immediately notify the responsible authorities. The system owner/operator should also:
- immediately resample and test the E. coli-positive site(s) and adjacent sites;
- conduct an assessment to ensure treatment barriers are operating correctly (this may include gathering information on water treatment performance and other operational monitoring data); and
- carry out any corrective actions necessary (See Section 3.1.4) in order to resume control or normal system operations.
If resampling and testing confirm the presence of E. coli in drinking water, the system owner/operator should immediately issue a boil water advisoryFootnote 2 in consultation with the responsible authorities, and carry out the appropriate corrective actions (Section 3.1.4). The owner/operator should also cooperate with the responsible authorities in any surveillance for possible waterborne disease outbreaks.
In addition, where E. coli contamination is detected in the first sampling—for example, E. coli-positive sample results from a single site, or from more than one location in the distribution system—the owner/operator or the responsible authority may decide to notify consumers immediately to boil their drinking water or use an alternative supply known to be safe and initiate corrective actions without waiting for confirmation. A boil water advisory should be immediately issued where there is evidence of:
- a significant failure of a critical treatment barrier; or
- illness in the community that is suspected to be associated with drinking water.
A decision tree is provided in Appendix A to assist system owners/operators.
3.1.4 Corrective actions
If resampling and testing confirms the presence of E. coli in drinking water, the owner/operator of the waterworks system should carry out appropriate corrective actions, which could include the following measures:
- Verify the integrity and the optimal operation of the treatment process.
- Verify the integrity of the distribution system.
- Verify that the required disinfectant residual is present throughout the distribution system.
- Increase disinfectant dosage, flush water mains, clean treated-water storage tanks (municipal reservoirs and domestic cisterns), and check for the presence of cross-connections and pressure losses. The responsible authority should be consulted regarding the correct procedure for dechlorinating water being discharged into the environment.
- Sample and test the E. coli-positive site(s) and locations adjacent to the E. coli-positive site(s). At a minimum, one sample upstream and one downstream from the original sample site(s) plus the treated water from the treatment plant as it enters the distribution system should be tested. Other follow-up samples should be collected and tested according to an appropriate sampling plan for the distribution system. Tests performed should include those for E. coli, total coliforms (as a general indicator of microbiological quality and inadequate treatment) and operational monitoring parameters such as disinfectant residual and turbidity. Testing for enterococci as an additional fecal verification indicator may also be performed.
- Conduct an investigation to identify the problem and prevent its recurrence; this would include measuring raw water quality (e.g., bacteriology, turbidity, colour, natural organic matter, and conductivity) and variability.
- Continue selected sampling and testing (e.g., bacteriology, disinfectant residual, turbidity) of all identified sites during the investigative phase to confirm the extent of the problem and to verify the success of the corrective actions.
3.1.5 Rescinding a boil water advisory
Once the appropriate corrective actions have been taken and only after a minimum of two consecutive sets of bacteriological samples, collected 24 hours apart, produce negative results, an E. coli-related boil water advisory may be rescinded. Additional water quality monitoring and actions may be required by the responsible drinking water authority. Further information on boil water advisories can be found in Health Canada's Guidance for Issuing and Rescinding Boil Water Advisories in Canadian Drinking Water Supplies. Over the long term, only a history of bacteriological and operational monitoring data together with validation of the system's design, operation and maintenance can be used to confirm the quality of a drinking water supply.
3.2 Residential-scale drinking water systems
3.2.1 Monitoring E. coli in water from disinfected and undisinfected supplies
Testing frequencies for residential-scale systems are determined by the responsible drinking water authority in the affected jurisdiction, and should include times when the risk of contamination of the drinking water source is the greatest, for example, in early spring after the thaw, after an extended dry spell, or following heavy rains. Homeowners with private wells should regularly test (at a minimum two times per year) their well for E. coli, ideally during these same at-risk times. New or rehabilitated wells should also be tested before their first use to confirm microbiological safety. The responsible drinking water authority in the affected jurisdiction should be consulted regarding their specific requirements for well construction and maintenance.
3.2.2 Notification
Residential-scale systems that serve the public may be subject to regulatory or legislative requirements and should follow any actions specified by the responsible drinking water authority. If E. coli is detected in a sample of drinking water from a residential-scale system that serves the public, the system owner/operator and the laboratory processing the samples should immediately notify the responsible authorities. The system owner/operator should also immediately resample and test the drinking water to confirm the presence of E. coli. The responsible authority should advise the system owner/operator to boil the drinking water or to use an alternative supply that is known to be safe in the interim. If E. coli is detected in a private drinking water supply, homeowners should also be advised to boil their drinking water or to use an alternative supply that is known to be safe; and to resample and test their drinking water to confirm the presence of E. coli.
If resampling confirms that the source is contaminated with E. coli, the system owner/operator should immediately carry out the appropriate corrective actions (see Section 3.2.3 and 3.2.4). The owner/operator should also cooperate with the responsible authorities in any surveillance for possible waterborne disease outbreaks. As a precautionary measure, some jurisdictions may recommend immediate corrective actions without waiting for confirmatory results. A decision tree is provided in Appendix B to assist system owners/operators.
3.2.3 Corrective actions for disinfected supplies
The first step, if it has not already been taken, is to evaluate the physical condition of the drinking water system as applicable, including water intake, well, well head, pump, treatment system (including chemical feed equipment, if present), plumbing, barriers to animal access (e.g., birds, vermin), and the surrounding area.
Any identified faults should be corrected. If the physical conditions are acceptable, some or all of the following corrective actions may be necessary:
- In a chlorinated system, verify that a disinfectant residual is present throughout the system.
- Increase the disinfectant dosage; flush the system thoroughly and clean treated water storage tanks and domestic cisterns. The responsible authority should be consulted regarding the correct procedure for dechlorinating water that may be discharged into the environment.
- For systems where the disinfection technology does not leave a disinfectant residual, such as ultraviolet (UV), it may be necessary to shock chlorinate the well and plumbing system.
- Ensure that the disinfection system is working properly and maintained according to manufacturer's instructions.
After the necessary corrective actions have been taken, samples should be collected and tested for E. coli to confirm that the problem has been corrected. If the problem cannot be corrected, additional treatment or a new source of drinking water should be considered. In the interim, any initial precautionary measures should continue; for example, drinking water should continue to be boiled or an alternative supply of water known to be safe should continue to be used.
3.2.4 Corrective actions for undisinfected wells
The first step, if it has not already been taken, is to evaluate the condition of the well, well head, pump, plumbing, and surrounding area. Any identified faults should be corrected. If the physical conditions are acceptable, then the following corrective actions should be carried out:
- Shock-chlorinate the well and plumbing system.
- Flush the system thoroughly and retest to confirm the absence of E. coli. Confirmatory tests should be delayed until either 48 hours after tests indicate the absence of a chlorine residual or five days have elapsed since the well was treated. For residential-scale systems that serve the public, the responsible drinking water authority may determine acceptable practice. The responsible authority should also be consulted regarding the correct procedure for dechlorinating water that may be discharged to the environment.
If the water remains contaminated after shock-chlorination, further investigation into the factors likely contributing to the contamination should be carried out. If these factors cannot be identified or corrected, either an appropriate disinfection device or well reconstruction or replacement should be considered. Drinking water should be boiled or an alternative supply of water known to be safe should continue to be used in the interim.
3.2.5 Rescinding a boil water advisory
Once the appropriate corrective actions have been taken, an E. coli-related boil water advisory should be rescinded only after a minimum of two consecutive sets of samples, collected 24 hours apart, produce negative results. Additional water quality monitoring and actions may be required by the responsible drinking water authority. Further information on boil water advisories can be found in Health Canada's Guidance for Issuing and Rescinding Boil Water Advisories in Canadian Drinking Water Supplies. Additional tests should be taken after three to four months to ensure that the contamination has not recurred. Over the long term, only a history of bacteriological and operational monitoring data in conjunction with regular physical inspections and a source water assessment can be used to confirm the quality of a drinking water supply.
Part II. Science and Technical Considerations
4.0 Significance of E. coli in drinking water
4.1 Description
Escherichia coli (E. coli) is a member of the coliform group of bacteria, part of the family Enterobacteriaceae, and described as a facultative anaerobic, Gram‑negative, non‑spore-forming, rod‑shaped bacterium. The vast majority of E. coli are harmless bacteria that live in the intestinal system of humans and animals and which assist with digestion. There are also several pathogenic types of E. coli, like the well-known E. coli O157:H7 serotype, which have acquired traits that make them harmful to humans. These pathogenic E. coli can be important causes of waterborne enteric illness if they are introduced into drinking water supplies through contaminated human or animal feces. Pathogenic E. coli are discussed in detail in the Health Canada's Guidance on Waterborne Bacterial Pathogens. When testing for E. coli, it is important for water utilities to understand that the analytical methods are designed to detect the non-pathogenic E. coli as an indicator of the presence of fecal contamination. The detection of E. coli signifies that the drinking water may pose a health risk because fecal pathogens can also be present.
The complexity of the E. coli species has become better understood with the use of advanced molecular characterization methods and the accumulation of whole genome sequence data (Lukjancenko et al., 2010; Chaudhuri and Henderson, 2012, Gordon, 2013). Presently it is recognized that E. coli strains can be categorized into one of several phylogenetic groups (A, B1, B2, C, D, E, F) based on differences in their genotype. Strains in the different groups show some variation in their physical and biological properties (e.g., their ability to utilize different nutrients), the fecal and environmental habitats in which they have been encountered and their predisposition for causing disease (Clermont et al., 2000; Walk et al., 2007; Tenaillon et al., 2010; Chaudhuri and Henderson, 2012; Gordon, 2013; Jang et al., 2017). More research is needed to better understand the practical impacts these differences have on drinking water microbiology and the implications for human health (Van Elsas et al., 2011; Gordon, 2013).
4.2 Sources
E. coli is naturally found in the intestinal system and feces of humans and a wide variety of animals. It is most prevalent in warm-blooded animals (domestic and wild), but can also be found in numerous cold-blooded animal species (Gordon, 2013, Tenaillon et al., 2010; Gordon and Cowling, 2003; Frick et al., 2018). Within human feces, E. coli is present at a concentration of 107 ̶ 109 cells per gram (Edberg et al., 2000; Leclerc et al., 2001; Tenaillon et al., 2010; Ervin et al., 2013). Numbers in feces of domestic animals (e.g., farm animals and pets) can vary considerably, but typically fall within the range from 104 ̶ 109 cells per gram (Lefebvre et al., 2006; Duriez and Topp, 2007; Diarra et al., 2007; Tenaillon et al., 2010; Ervin et al., 2013). Information on the prevalence and abundance of E. coli in feces of wild animal species is comparatively limited (Tenaillon et al., 2010; Farnleitner et al., 2010). Studies have found that E. coli concentrations in feces of wild warm-blooded and cold-blooded animals can reach values of 104-109 cells per gram (Farnleitner et al. 2010; Ervin et al., 2013; Frick et al., 2018, The very high concentrations of E. coli in human and animal feces make it possible to detect the fecal contamination of water at sample volumes that are convenient to collect and transport to a laboratory.
Sources of fecal contamination that can impact surface water or ground water source supplies include point sources (e.g., sewage and industrial effluents, septic systems, leaking sanitary sewers) and non-point or diffuse sources (e.g., runoff from agricultural, urban and natural areas) (Gerba and Smith, 2005; Hynds et al., 2012, 2014; Wallender et al., 2014; Lalancette et al., 2014; Staley et al., 2016).
4.3 Survival
Once shed from animal hosts into secondary habitats, the survival of E. coli is determined by a combination of physical and biological factors including temperature, solar radiation, presence and types of other microflora, availability of nutrients and the ability to persist in biofilms (Foppen and Schijven, 2006; Van Elsas et al., 2011; Blaustein et al., 2013; Jang et al., 2017). The impact of specific factors varies depending on the type of water involved (e.g., groundwater, surface water, treated distribution water). In water environments, conditions for survival are considerably less favourable than in the intestinal system and populations of E. coli naturally decline (Winfield and Groisman, 2003; Van Elsas et al., 2011). Results from freshwater microcosm studies demonstrate E. coli populations die off rapidly, with declines from one to several logs observed in time frames on the order of a week to 10 days (Flint, 1987; Lim and Flint, 1989; Bogosian, 1996; Sampson et al., 2006). Population declines occur more slowly in groundwater, with data from microcosm studies indicating time frames for one log reduction on the order of one to two weeks (Keswick et al., 1982; Filip et al., 1986; John and Rose, 2005).The fate of E. coli populations in complex natural environments is not easy to predict (Van Elsas et al., 2011). Some studies demonstrated that the organism survived in lake water for several weeks at a temperature of 4°C (Sampson et al., 2006) and in filtered groundwater (0.45µm) for 14 weeks at 10°C (Filip et al., 1986).
In general, E. coli has a lifespan that is similar in scale to that of other enteric bacteria (Edberg et al., 2000; John and Rose, 2004; Sinton et al., 2007; Pachepsky et al., 2014). However, its lifespan is much shorter than those of more resistant microorganisms such as protozoan (oo)cysts and many enteric viruses (John and Rose, 2004; King and Monis, 2007; Kotwal and Cannon, 2014; Hamilton et al., 2018). As a result of its die-off rate in natural waters and relative sensitivity to environmental stresses, detection of E. coli in source waters is considered indicative of recent fecal contamination (Edberg et al., 2000; WHO and OECD, 2003).
In natural and engineered water environments (e.g., distribution systems), when E. coli encounter conditions that do not support growth, they can adapt to enter a viable but non-culturable (VBNC) state where they do not grow on laboratory media, but are otherwise alive and capable of resuscitation when conditions become favourable (Bjergbæk and Roslev, 2005, Li et al., 2014). This VBNC state may be induced by a wide range of stress factors including nutrient limitation, pH, temperature, oxygenation, osmotic pressure, and exposure to antimicrobial agents, such as drinking water disinfectants (Bjergbæk and Roslev, 2005; Li et al., 2014, Chen et al., 2018). The VBNC state is a primary survival strategy for bacteria that has been observed with numerous species (Lee et al., 2007; Li et al., 2014; van der Kooij and van der Wielen, 2014). A greater understanding of the VBNC state in bacteria relevant to drinking water is needed (Li et al., 2014; van der Kooij and van der Wielen, 2014).
4.3.1 Environmentally-adapted E. coli
It is now well-recognized by the scientific community that E. coli can survive long-term and grow in habitats outside of the lower intestinal tract of human and animals provided that the pressures exerted by certain factors (e.g., temperature, nutrient and water availability, pH, solar radiation, presence of other microorganisms) are within their tolerance limits (Ishii et al., 2010; Van Elsas et al., 2011; Byappanahalli et al., 2012b; Tymensen et al., 2015; Jang et al., 2017). It has also become evident that some strains of E. coli can adapt to live independently of fecal material and become naturalized members of the microbial community in environmental habitats (Ishii and Sadowsky, 2008; Ishii et al., 2010; Byappanahalli et al., 2012b). E. coli genotypes that are distinct from those found in human or animal feces have been discovered in sands, soils, sediments, aquatic vegetation, septic waste and raw sewage (Gordon et al., 2002; Byappanahalli et al., 2006; Ksoll et al., 2007; Ishii and Sadowsky, 2008; Ishii et al., 2010; Badgley et al., 2011; Zhi et al., 2016). Over time, research has shown that environmental habitats may serve as potential sources of most of the groups of bacteria that have been used for detecting fecal contamination of drinking water, including total coliforms, thermotolerant coliforms, E. coli and enterococci (Edberg et al., 2000; Whitman et al., 2003; Byappanahalli et al., 2012a). While these findings change the perception that E. coli is exclusively associated with fecal wastes, it is accepted that E. coli is predominantly of fecal origin andremains a valuable indicator of fecal contamination in drinking water (See Section 4.5). More research is needed to improve our understanding of the behaviour of E. coli in the environment.
4.4 Role of E. coli as an indicator of drinking water quality
Of the contaminants that can be found in drinking water, pathogenic microorganisms from human and animal feces pose the greatest danger to public health. Although modern microbiological techniques have made the detection of pathogenic bacteria, viruses and protozoa possible, it is not practical to attempt to routinely isolate these microbes from drinking water (Payment and Pintar, 2006; Allen et al., 2015). For this reason, indicator organisms are used to assess the microbiological safety of drinking water. These indicators are less difficult, less expensive, and less time consuming to monitor. This encourages testing of a higher number of samples which gives a better overall picture of the water quality and, therefore, better public health protection. Different indicator organisms can be used for specific purposes in drinking water risk management, in areas such as source water assessment, operational monitoring, validation of drinking water treatment processes and drinking water quality verification (WHO, 2005).
Worldwide, E. coli is the most widely used indicator of fecal contamination in drinking water supplies (Edberg et al., 2000; Payment et al., 2003). E. coli is predominantly associated with human and animal feces, and its detection is considered more specific to fecal contamination than other bacterial indicators such as thermotolerant coliforms or enterococci (Edberg et al., 2000; WHO and OECD, 2003; Standridge et al., 2008; Lin and Ganesh, 2013).
E. coli bacteria are excreted in human and animal feces in high numbers, they typically do not multiply in drinking water and they can be rapidly, easily and affordably detected. These features in particular make E. coli highly useful for detecting fecal contamination even when the contamination is greatly diluted.
The primary role for E. coli is as an indicator of fecal contamination during monitoring to verify the microbiological quality of drinking water. Drinking water quality verification is a fundamental aspect of a source–to-tap or water safety plan approach to drinking water systems that includes monitoring to confirm that the system as a whole is operating as intended (Health Canada, 2001; CCME, 2004; WHO, 2005; 2012). E. coli can also be used as a parameter in source water assessments and during drinking water system investigations in response to corrective actions or surveillance.
E. coli is not intended to be a surrogate organism for pathogens in water (Health Canada, 2019c, 2019d). Numerous studies have documented that the presence of E. coli does not reliably predict the presence of specific enteric or non-enteric waterborne pathogens (Wu et al., 2011; Payment and Locas, 2011; Edge et al., 2013; Hynds et al., 2014; Lalancette et al., 2014; Ashbolt, 2015; Falkinham et al., 2015; Krkosek et al., 2016; Fout et al., 2017). During drinking water treatment, the rates of physical removal for E. coli are different than those for enteric protozoa and enteric viruses; E. coli is also more easily inactivated by drinking water disinfectants than these two groups (See Section 7.0). The presence of E. coli in water is an indicator that the water has been subject to recent fecal contamination and thus, there is a strong potential for a health risk, regardless of whether specific pathogens are observed.
4.4.1 Role in groundwater sources
The presence of E. coli in a groundwater well indicates that the well has been affected by fecal contamination and serves as a trigger for further action (See Sections 3.1.3 and 3.2.2). E. coli monitoring is an essential component of public health protection for all drinking water supplies, disinfected and undisinfected. Small drinking water systems and in particular undisinfected groundwater systems are more vulnerable to the effects of fecal contamination and test positive more frequently for bacteriological indicators, including E. coli, than large systems (Cretikos et al.,2010; Invik et al., 2017; Messner et al., 2017; Health Canada, 2018d). Studies of the groundwater quality of Canadian municipal wells have demonstrated the importance of historical E. coli data for raw groundwater when evaluating a well's potential susceptibility to fecal contamination (Payment and Locas, 2005; Locas et al., 2007, 2008). Recurrent detection of E. coli in a groundwater source indicates a degradation of the source water quality and a greater likelihood of pathogen occurrence (Payment and Locas, 2005, 2011; Locas et al., 2007, 2008; Fout et al., 2017).
Investigations of outbreaks of waterborne illness from small drinking water supplies have also demonstrated the usefulness of E. coli monitoring in verifying fecal contamination and/or the inadequate treatment of a groundwater source (Laursen et al., 1994; Fogarty et al., 1995; Engberg et al., 1998; Novello, 2000; Olsen et al., 2002; O'Connor, 2002a; Hrudey and Hrudey, 2014; Government Inquiry into Havelock North Drinking Water, 2017; Kauppinen et al., 2017). Fatalities have been associated with a number of these outbreaks, including, the Washington County Fair, New York (1999); Walkerton, Ontario (2000), and Havelock North, New Zealand (2016) (Novello, 2000; O'Connor, 2002a; Hrudey and Hrudey, 2014; Government Inquiry into Havelock North Drinking Water, 2017).
Groundwater from private wells is generally perceived safe for drinking by consumers (Hynds et al., 2013; Murphy et al., 2017), however this is not always an accurate assumption. Studies have shown that private wells can test positive for E. coli more frequently than municipal-scale systems and residential-scale systems that provide drinking water to the public (Krolik et al., 2013; Invik et al., 2017; Saby et al., 2017). Further, researchers have estimated that the consumption of water from contaminated unregulated private wells may be responsible for a large proportion of the total burden of acute gastrointestinal illness associated with drinking water sources (DeFelice et al., 2016; Murphy et al., 2016b).
The above information emphasizes the importance of regular testing of untreated groundwater as well as treated groundwater to improve the ability of a monitoring program to detect wells affected by fecal contamination. Further guidance to help water utilities, owners and operators develop best practices for controlling and responding to microbiological hazards is available (CCME, 2004; WHO, 2012; AWWA, 2014).
4.4.2 Role in surface water sources
Although the relationships seems to be site-specific, monitoring for E. coli in raw water can provide data relative to the impact and timing of sources of fecal pollution which affect the drinking water source. Similarly, it can provide information on the effects of source water protection or hazard control measures implemented in the watershed. Source water E. coli data can also be used to provide supplementary information in assessing microbiological risks and treatment requirements for surface water sources (U.S. EPA, 2006b; Hamouda et al., 2016).
Correlations between indicator organisms and pathogens can sometimes be observed in heavily polluted waters, but these correlations quickly deteriorate due to dilution and the differences in the fate and transport of different microorganisms in various water environments (Payment and Locas, 2011). Lalancette et al. (2014) found that E. coli were potentially good indicators of Cryptosporidium concentrations at drinking water intakes when source waters are impacted by recent and nearby municipal sewage, but not at intakes where sources were dominated by agricultural or rural fecal pollution sources or more distant wastewater sources. Increased odds of detecting enteric pathogens (Campylobacter, Cryptosporidium, Salmonella and E. coli O157:H7) in surface water samples have been shown in some studies where densities of E. coli exceeded 100 colony-forming units (CFU) per 100 mL (Van Dyke et al., 2012, Banihashemi et al., 2015; Stea et al., 2015).
4.4.3 Role in treatment monitoring
Detection of E. coli in water immediately after treatment or leaving the treatment plant signifies inadequate treatment and is unacceptable. Cretikos et al. (2010) examined the factors associated with E. coli detection at public drinking water systems in New South Wales, Australia. Undisinfected systems and small water supply systems serving less than 500 people were most strongly associated with E. coli detection. E. coli detections were also significantly associated with systems disinfected with only UV or with higher post-treatment turbidity.
Drinking water outbreaks have been linked to municipal supplies where water quality parameters (including E. coli) were below the acceptable limits recognized at the time (Hayes et al., 1989; Maguire et al., 1995; Goldstein et al., 1996; Jack et al., 2013). E. coli has different removal rates through physical processes and is more sensitive to drinking water disinfectants than enteric viruses and protozoa. While testing for E. coli is useful in assessing the treatment efficacy, it is not sufficient as a parameter in isolation of other factors with respect to assessing the impact on these pathogens (Payment et al., 2003). E. coli can be used as part of the water quality verification process in conjunction with information on treatment performance to show that the water has been adequately treated and is therefore of acceptable microbiological quality (Payment et al., 2003; Stanfield et al., 2003). However, under a source-to-tap or water safety plan approach to drinking water systems, validation of treatment and disinfection processes are also important to show that the system can operate as required and achieve the required levels of hazard reduction (CCME, 2004; WHO, 2005).
4.4.4 Role in distribution system monitoring
Microorganisms can enter the distribution system by passing through treatment and disinfection barriers during inadequate treatment, or through post-treatment contamination via intrusions, cross-connections or during construction or repairs.
The presence of E. coli in a distribution system sample can indicate that treatment of the source water has been inadequate, or that the treated water has become contaminated with fecal material during distribution. Post-treatment contamination, for example, through cross-connections, back siphonage, low or negative transient pressure events, contamination of storage reservoirs, and contamination of mains from repairs, have been identified as causes of distribution system contamination linked to illness (Craun, 2002; Hunter et al., 2005; Breitenmoser et al., 2008; Falco and Williams, 2009; Laine et al., 2011; Hrudey and Hrudey, 2014; Puleston et al., 2014). These include fatal outbreaks at Nokia, Finland (2007), and Alamosa, Colorado (2008) (Falco and Williams, 2009; Laine et al., 2011).
The detection of E. coli is expected to be sporadic and rare in properly designed and well-operated treatment and distribution systems. Water quality reports provided by large municipal drinking water utilities in Canada have shown that the number of distribution system samples that test positive for E. coli is typically less than 1% annually (Health Canada, 2018d). Data demonstrating the quality of the drinking water in individual provinces and territories can be obtained from the responsible drinking water authority or the water utilities. The detection of E. coli in the distribution system can indicate an increased potential of exposure to enteric pathogens for consumers in affected areas. Miles et al. (2009) analyzed point-of-use (POU) filters found in drinking water vending machines in Arizona to evaluate the microbiological quality of large volumes (e.g., 1,000-17,000 L) of treated, distributed drinking water and observed that 60% (3/5) of the filters that tested positive for E. coli also tested positive for enteroviruses.
Results from studies of model, pilot-scale and full-scale systems have shown that E. coli can accumulate in low numbers in distribution system biofilms, predominantly in a viable-but-not-culturable state (Fass et al. 1996; Williams and Braun-Howland, 2003; Juhna et al., 2007; Lehtola et al., 2007; Abberton et al., 2016; Mezule and Juhna, 2016). However, once embedded within the biofilm matrix, E. coli concentrations are controlled by the natural microbial community through processes such as predation and competition for nutrients (Fass et al. 1996; Abberton et al., 2016; Mezule and Juhna, 2016). Consequently, the detection of E. coli in a water distribution system is a good indication of recent fecal contamination. The presence of E. coli in any distribution and/or storage system sample is unacceptable and should result in further action (see Section 3.1.3). Further guidance to help water utilities develop best practices for controlling microbial hazards is available (CCME, 2004; AWWA, 2017; Hill et al., 2018).
4.4.5 Role of E. coli in a decision to issue boil water advisories
Boil water advisories are public announcements advising consumers that they should boil their drinking water prior to consumption in order to eliminate any disease-causing microorganisms that are suspected or confirmed to be in the water. These announcements are used as part of drinking water oversight and public health protection across the country. Health Canada (2015) provides more information on issuing and rescinding drinking water advisories.
Drinking water data (primarily on boil water advisories) are collected on the Canadian Network for Public Health Intelligence (CNPHI) Drinking Water Advisories application, a secure, real-time web-based application, and by provincial and territorial regulators. Provincial, territorial and municipal drinking water data resides with and are provided by the responsible drinking water authority in the affected jurisdiction. Although the data in CNPHI does not provide a complete national picture, the trends within these data provide useful insight into the nature of boil water advisories and the challenges that exist in drinking water systems in Canada. A review of the available Canadian boil water advisory records (9,884 boil water advisory records issued between 1984 to the end of 2017) found that 594 (6%) of the boil water advisories noted “E. coli detected in drinking water system” as the reason for issuing the advisory (Health Canada, 2018c). The remaining boil water advisories were issued for other reasons, the most common of these being equipment and process-related (see Figure 1).
Figure 1. Overall proportions of reasons for issuing boil water advisories*
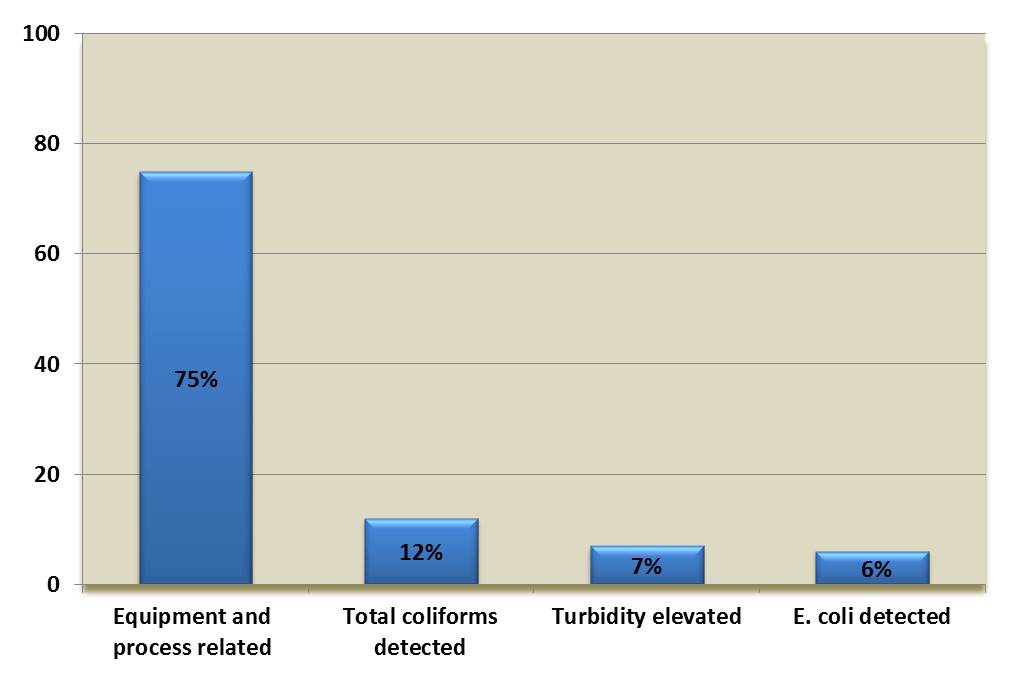
*Data from 1984 to 2017 (n=9884)
Text description
Figure 1 is a bar chart showing the overall proportions of reasons for boil water advisories as; 75% equipment and process related, 12% due to detection of total coliforms, 7% due to elevated turbidity and 6% due to detection of E. coli.
Over 99% of the 594 boil water advisories associated with the detection of E. coli occurred in small drinking water systems (see Figure 2), and were almost equally split between surface water and ground water sources (see Figure 3) (Health Canada, 2018c). More than half of these advisories were issued without any additional operational context recorded (see Figure 4), which may indicate that they were issued solely in response to a positive E. coli test during routine sampling. Overall, the data support the evidence that small drinking water systems face increased contamination risk. The data also highlight the importance of monitoring for operational parameters in addition to conducting regular E. coli testing when confirming the quality of the drinking water supply.
Figure 2. Population served by drinking water systems affected by E. coli-detection related boil water advisories*
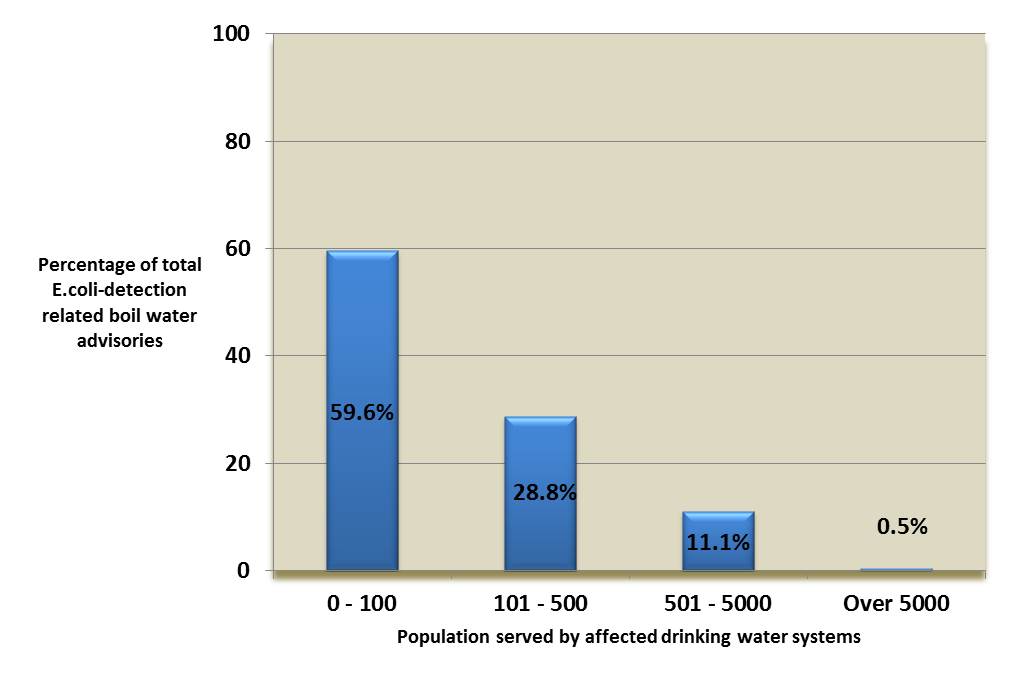
*Data from 1984 to 2017 (n=9884)
Text description
Figure 2 is a bar chart that shows the proportions of population served by drinking water systems affected by boil water advisories (BWAs) issued due to the detection of E. coli. Of the affected drinking water systems, 59.6% serve 0 – 100 people, 28.8% serve 101 – 500 people, 11.1% serve 501-5000 people and 0.5% serve over 5000 people.
Figure 3. Source water used by drinking water systems affected by E. coli-detection related boil water advisories*
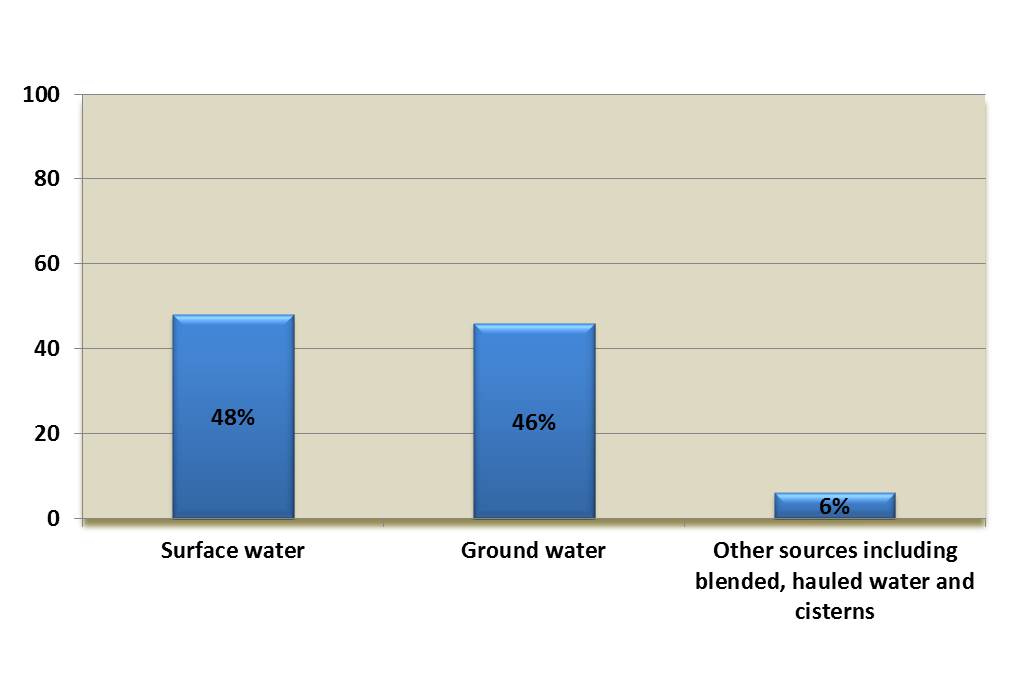
*Data from 1984 to 2017 (n=9884)
Text description
Figure 3 is a bar chart showing the source water used by drinking water systems affected by E. coli-detection related boil water advisories. The proportions show 48% use surface water, 46% use ground water and 6% use other sources including blended (surface and ground water), hauled water and cisterns.
Figure 4. Operational context associated with E. coli-detection related boil water advisories*
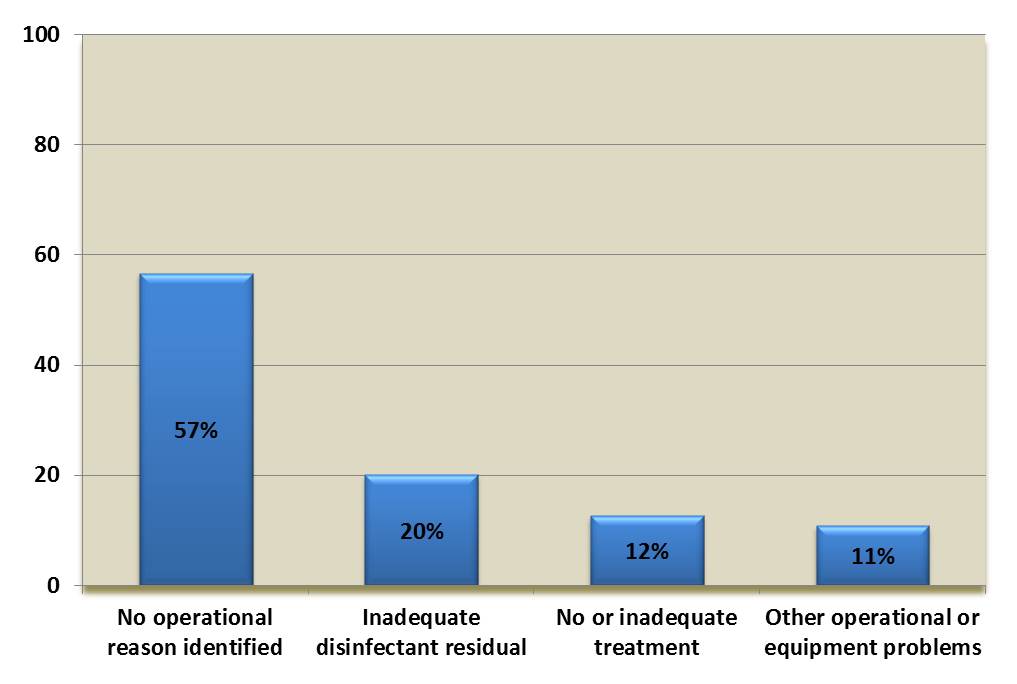
*Data from 1984 to 2017 (n=9884)
Text description
Figure 4 is a bar chart showing the operational context associated with E-coli-detection related boil water advisories. 57% do not identify any operational reason, 20% note inadequate disinfectant residual, 12% note no or inadequate treatment and 11% note other operational or equipment related problems.
5.0 Analytical methods
All analyses for E. coli should be carried out as directed by the responsible drinking water authority. In many cases, this authority will recommend or require the use of accredited laboratories. In some cases, it may be necessary to use other means to analyze samples in a timely manner, such as on-site testing using commercial test kits by trained operators. It is important to use validated or standardized methods to make correct and timely public health decisions. When purchasing laboratory services or selecting analytical methods for analysis to be performed in-house, water utilities should consult with the analytical laboratory or manufacturer on issues of method sensitivity, specificity and turnaround time. To ensure reliable results, a quality assurance program, which incorporates quality control practices, should be in place. Analyses conducted using test kits used should be performed according to the manufacturer's instructions.
5.1 Culture-based methods
Standardized methods available for the detection of E. coli in drinking water are summarized in Table 1. Methods that target E. coli are based on the presence of the β-D-glucuronidase enzyme. This is a distinguishing enzyme that is found in the vast majority of E. coli isolates. The uidA gene which encodes for the β-glucuronidase enzyme is present in > 97% of E. coli isolates (Feng et al., 1991; Martins et al., 1993; Maheux et al., 2009). The gene may also be found in a low proportion of Shigella and Salmonella strains and in some strains of other bacterial species; but is rarely present in other coliforms (Feng et al., 1991; Fricker et al., 2008, 2010; Maheux et al., 2008, 2017.). Although E. coli serotype O157:H7 and some Shigella strains do carry nucleotide sequences for the uidA gene, most isolates do not exhibit enzyme activity (Feng and Lampel, 1994, Maheux et al., 2011). Detection methods also take advantage of biochemical characteristics specific to E. coli and use media additives and incubation temperatures to inhibit the growth of background microorganisms. All of the methods listed in Table 1 are capable of detecting total coliforms and simultaneously differentiating E. coli.
When verification of a positive test result is required, there are numerous ways to identify E. coli from other coliforms and other bacteria species. Biochemical tests for differentiating members of the family Enterobacteriaceae, including E. coli, and commercial media and identification kits for verifying E. coli are available (APHA et al., 2017). E. coli confirmation can also be done by subjecting coliform-positive samples to media that tests for the β-D-glucuronidase enzyme (APHA et al., 2017, ISO, 2018). The use of multiple biochemical tests for confirmation will improve the accuracy of the identification (Maheux et al., 2008).
| Organization - Method | Media | Results format | Total coliforms detected (Y/N) | Turnaround time |
|---|---|---|---|---|
| Membrane Filtration | ||||
| SM 9222 JTable 1 Footnote a U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c |
m-ColiBlue24® broth | P-A, C | Y | 24 h |
| SM 9222 KTable 1 Footnote a U.S. EPA 1604Table 1 Footnote bTable 1 Footnote c |
MI agar or broth | P-A, C | Y | 24 h |
| ISO 9308-1:2014Table 1 Footnote d U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c |
Chromocult® Coliform Agar | P-A, C | Y | 21-24 h |
| Enzyme substrate | ||||
| SM 9223 BTable 1 Footnote a U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c |
Colilert® medium | P-A, C | Y | 24-28 h |
| SM 9223 BTable 1 Footnote a ISO 9308-2: 2012Table 1 Footnote d U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c |
Colilert-18® medium | P-A, C | Y | 18-22 h |
| SM 9223 BTable 1 Footnote a U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c |
Colisure® medium | P-A, C | Y | 24-28 h |
| U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c | E*Colite® medium | P-A | Y | 28-48 h |
| U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c | Readycult® Coliforms 100 broth | P-A | Y | 24 h |
| U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c | Modified Colitag™ medium | P-A | Y | 16-22 h |
| U.S. EPA – N/ATable 1 Footnote bTable 1 Footnote c | Tecta™ EC/TC medium | P-A | Y | 18 h |
N/A – not available; P-A – presence-absence; C – count |
||||
Other methods may be approved for use in other jurisdictions (CEAEQ, 2018, MECP, 2019). The results of E. coli test methods are presented as either presence-absence (P-A) or counts (C) of bacteria. P-A testing does not provide any information on the concentration of organisms in the sample. The quantitation of organisms is sometimes used to assess the extent of the contamination, and as such is considered a benefit of the more quantitative methods. For decision-making, the focus is the positive detection of E. coli, regardless of quantity; as the guideline for E. coli in drinking water is none per 100 mL, qualitative results are sufficient for protecting public health.
5.1.1 Performance of detection methods
There are limitations in the detection capabilities of culture-based methods which rely upon the expression of the β-glucuronidase enzyme for a positive identification of E. coli (Maheux et al., 2008; Zhang et al., 2015). There is also variability in the performance of commercialized E. coli methods observed during laboratory testing of isolates from different settings (e.g., clinical, environmental), water types and geographic locations (Bernasconi et al., 2006; Olstadt et al., 2007; Lange et al., 2008; Maheux et al., 2008; Maheux et al., 2015a; 2015b; Martin and Gentry, 2016; Maheux et al., 2017). Factors that can affect the ability of culture-based methods to detect E. coli include: the natural variability in the percentage of β-D-glucuronidase negative strains in the source population (Feng and Lampel, 1994; Maheux et al., 2008); the composition of the media (Hörman and Hänninen, 2006; Olstadt et al., 2007; Maheux et al., 2008, 2017; Fricker et al., 2010; Martin and Gentry, 2016); the concentration of the organisms and their physiological state (Ciebin et al., 1995; Maheux et al., 2008; Zhang et al., 2015); and, water quality characteristics (Olstadt et al., 2007).
Standardized methods have been validated against established reference methods to ensure that the method performs to an acceptable level (APHA et al., 2017). Nevertheless, there is a need to continually evaluate the efficacy of E. coli test methods, and to improve their sensitivity and specificity. The accuracy of future methods may be improved with advanced techniques combining biochemical characteristics with molecular tests (Maheux et al., 2008). Other useful strategies can include efforts by approval bodies to conduct regular reviews of screening criteria and method performances, and continued work by manufacturers towards optimizing their medium formulations (Zhang et al., 2015). Criteria for consideration when designing studies for the evaluation of microbiological methods are discussed in other publications (Boubetra et al., 2011; APHA et al., 2017; Duygu and Udoh, 2017).
5.2 Molecular methods
Given the limitations associated with culture-based methods for detecting E. coli (e.g., required time of analysis, lack of universality of the β-D-glucuronidase enzyme signal, their inability to detect VBNC organisms), molecular-based detection methods continue to be of interest (Martins et al., 1993; Heijnen and Medema, 2009; Mendes Silva and Domingues, 2015). No molecular methods for detecting E. coli in drinking water have been standardized or approved for drinking water compliance monitoring.
Polymerase chain reaction (PCR)-based detection methods are the most commonly described molecular methods for the detection of microorganisms in water (Maheux et al., 2011; Gensberger et al., 2014; Krapf et al., 2016). In recent years, the number of techniques available has increased considerably and the costs associated with their use have been significantly reduced (Mendes Silva and Domingues, 2015). However, the most significant challenge associated with PCR analysis of drinking water samples remains the need to provide a sufficiently low detection limit. Descriptions of the different types of molecular methods explored for the detection of E. coli in water sources are available elsewhere (Botes et al., 2013; Mendes Silva and Domingues, 2015). At present, the limits of detection reported for the vast majority of methods encountered in the literature are higher than the sensitivity limit of 1 E. coli per 100 mL required for drinking water analysis (Heijnen and Medema 2009; Maheux et al., 2011, Gensberger et al., 2014; Mendes Silva and Domingues, 2015; Krapf et al., 2016). More work is needed to further optimize the sensitivity of molecular detection methods for E. coli and to develop standardized methods for routine monitoring that are accurate, reliable, easy to use and affordable.
5.3 Rapid online monitoring methods
The need for more rapid and frequent monitoring of E. coli in drinking water distribution systems has led researchers to explore on-line water quality sensor technologies capable of detecting E. coli contamination in real-time. Some of the sensors investigated have been based on measurements of electrical impedance (Kim et al., 2015), immunological or biomarker linked fluorescence (Golberg et al., 2014, Gutiérrez-del-Río, 2018) or water quality parameters such as conductivity, particle counts, pH, turbidity, UV absorbance, total organic carbon, alone or in combination (Miles et al., 2011; Ikonen et al., 2017).The most significant challenge facing potential rapid online detection methods is the need for sensitivity at very low E. coli concentrations (Kim et al., 2015; Ikonen et al., 2017). Additional obstacles include requirements for equipment, user training and data interpretation (Golberg et al., 2014; Ikonen et al., 2017). As with the molecular methods of detection, more work is needed before rapid methods are suitable for widespread use.
6.0 Sampling for E. coli
6.1 Sample collection
Proper procedures for collecting samples must be observed to ensure that the samples are representative of the water being examined. Guidance materials to assist water utilities are available (WHO and OECD, 2003; APHA et al. 2017). Sampling for microbiological analysis requires good sampling technique to prevent the introduction of contamination from external surfaces (e.g., hands, the tap) during sampling (WHO and OECD, 2003; APHA et al., 2017). Sample collectors are also advised to carefully choose sample locations, avoiding (or alternatively disinfecting) taps subject to external contamination (e.g., close to a sink bottom or the ground) and taps of questionable cleanliness (WHO and OECD, 2003; APHA et al., 2017).
Generally, samples for microbiological testing should be packed with ice packs but protected from direct contact with them to prevent freezing. Packing the sample with loose ice is not recommended as it may contaminate the sample. During transport, samples should be kept cool but unfrozen at temperatures between 4 and 10°C (Payment et al., 2003; APHA et al., 2017). Commercial devices are available for verifying that the proper transport temperatures are being achieved. During the summer and winter months, additional steps may be required to maintain the optimal temperature of samples while in transport. These steps may include adding additional ice packs, or communicating with couriers to ensure that the cooler will not be stored in areas where freezing or excessive heating could occur.
To avoid unpredictable changes in the bacterial numbers of the sample, E. coli samples should always be analyzed as soon as possible after collection. Where on-site facilities are available or when an accredited laboratory is within an acceptable travel distance, analysis of samples within 6-8 hours is suggested (Payment et al., 2003; APHA et al., 2017). Ideally, for E. coli analysis of drinking water samples, the holding time between the collection of the sample and the beginning of its examination should not exceed 30 hours (APHA et al., 2017).
Studies of the effects of holding time on the detection of E. coli in water samples have been limited. Ahammed et al. (2003) reported that in spiked groundwater samples held at 4°C, E. coli counts (initial concentration: 21 MPN/100mL) declined by 3% and 16 % after storage for 6 and 24 hours, respectively. Other studies have observed that holding times longer than 8-24 hours did not result in significant changes in E. coli counts. In a surface water study, Pope et al. (2003) found that when maintained at 4°C, samples from three of four sites did not show significant decreases in counts (concentration range: 76-7949 E. coli/100mL) until samples had been held for 48 hours. Bushon et al. (2015) studied the effects of holding time on E. coli in groundwater (concentration range: 3-63 CFU/100mL) by comparing samples tested within 8 hours using presence/absence methods with those tested within 18–30 hours using quantitative methods. The data indicated increasing the holding time from 8 hours to within 18-30 hours did not reduce the number of E. coli detections. In another groundwater study, Maier et al., (2015) reported that declines in E. coli concentrations in spiked samples (range: 72-130 CFU, volume not provided) did not exceed 10% in samples held for 48 hours. This evidence suggests that E. coli samples may be analyzed using common detection methods and generate comparable detection data, even 8 hours or more after sample collection. However, study authors also noted that there were variations in E. coli survival rates at some locations or with different isolates (Pope et al., 2003; Maier et al., 2015). As well, studies by McDaniels et al., (1985) and Ferguson (1994) have indicated that holding times can be more critical for members of the coliform group when concentrations are low.
The implications of an extended holding time should be discussed with the responsible drinking water authority in the affected jurisdiction. Specifically, it is important to consider the likelihood and impact of reporting a false negative result as a result of declines in the bacterial indicator count during extended storage. This should be weighed against the impact of samples being rejected or not being submitted at all if a water utility is unable to have them delivered to the laboratory within the required holding time (Maier et al., 2015).
Water utilities should also discuss with the laboratory, the protocols for rejecting or analysing samples that exceed holding time specifications. For samples that are analysed despite not meeting holding time requirements, a positive result is sufficient to provide evidence of contamination, however a negative result is not able to confirm the absence of contamination. When a sample is rejected, it is recommended that utilities immediately collect and submit a resample from that test location. When long holding times are anticipated, onsite testing with commercialized test methods (see Table 1) in combination with appropriate training and quality control procedures offers a reliable, standardized analytical option for verification and compliance monitoring. Water utilities should first consult with the responsible drinking water authority about the acceptability of this practice and any other requirements that may apply. The use of a delayed incubation procedure is another option for water utilities encountering challenges in shipping samples within the recommended time frame. A delayed incubation procedure for total coliforms has been described and verification methods can be used to confirm the presence of E. coli from positive samples (APHA et al., 2017). This procedure permits shipping to the laboratory a membrane filter from a sample filtered on site by placing it on a medium designed to keep coliform organisms viable during transport (APHA et al., 2017). The test is completed at the laboratory where the filter is transferred to fresh growth media and is incubated (APHA et al., 2017).
Samples should be labelled according to the requirements specified by the responsible drinking water authority and the analytical laboratory. In most cases, much of the information and the sample bottle identification number are recorded on the accompanying submission forms and, in cases where samples are collected for legal purposes, chain-of-custody paperwork. When analysis will be delayed, it is particularly important to record the duration and temperature of storage, as this information should be taken into consideration when interpreting the results. Water utilities may wish to consult with the analytical laboratory for specific requirements regarding the submission of samples.
To obtain a reliable estimate of the number of E. coli in treated drinking water, a minimum volume of 100 mL of water should be analyzed. Smaller volumes or dilutions may be more appropriate for testing samples from waters that are high in particulates or where high numbers of bacteria might be expected. Analysis of larger drinking water volumes can increase both the sensitivity and the reliability of testing. Large volume (20 L) sample analysis using a capsule filter was useful in improving the detection of total coliforms (E. coli was not detected) in distribution system samples during field trials at three drinking water utilities (Hargy et al., 2010). More study of large volume sample testing is needed to assess the added value of results. The main challenges of large volume sampling are determining how results are to be interpreted against current drinking water regulations, and optimizing methodologies to be suitable for routine use by water utilities. Additional statistical and field work are needed that simultaneously consider the parameters of sample volume, monitoring frequency, detection method, false/true positives and negatives, and cost.
6.2 Sampling frequency considerations
When determining sampling frequency requirements for municipal-scale systems, the application of a universal sampling formula is not possible due to basic differences in factors such as source water quality, adequacy and capacity of treatment, and size and complexity of the distribution system (WHO, 2004). Instead, the sampling frequency should be determined by the responsible drinking water authority after due consideration of local conditions, such as variations in raw water quality and history of the treated water quality. As part of operational and verification monitoring in a drinking water quality management system using a source-to-tap or water safety plan approach, water leaving a treatment plant and within the distribution system should be tested at least weekly for E. coli and daily for disinfectant residual and turbidity.
A guide for the recommended minimum sampling frequency for E. coli testing of drinking water distribution systems is provided in Table 2. The number of samples for bacteriological testing should be increased in accordance with the size of the population served.
| Population served | Minimum number of samples per monthTable 2 Footnote a |
|---|---|
| Up to 5000 | 4 |
| 5000–90 000 | 1 per 1000 persons |
| 90 000+ | 90 + (1 per 10 000 persons) |
|
|
Sampling frequency in municipal and residential-scale systems may vary by jurisdiction but should include times when the risk of contamination of the source water is greatest, such as during spring thaw, heavy rains, or dry periods. Associations have been observed between climate factors (peak rainfall periods, warmer temperatures) and E. coli detections for small groundwater systems that are susceptible to fecal contamination (Valeo et al., 2016; Invik et al., 2017). Extreme weather events, such as intense rainfall, flash floods, hurricanes, droughts and wildfires can have significant water quality impacts and are expected to increase in frequency and severity with climate change (Thomas et al., 2006; Nichols et al., 2009; Wu et al., 2011; Wallender et al., 2014; Khan et al., 2015; Staben et al., 2015). Water utilities impacted by such events should consider conducting additional sampling during and/or following their occurrence.
New or rehabilitated wells should also be sampled before their first use to confirm acceptable bacteriological quality. In municipal systems, increased sampling may be considered when changes occur from the normal operations of the water treatment system.
It must be emphasized that the frequencies suggested in Table 2 are only general guides. In many systems, the water leaving the treatment plant and within the distribution system will be tested for E. coli well in excess of these minimum recommendations. The general practice of basing sampling requirements on the population served recognizes that smaller water supply systems serve a smaller population thus fewer people are at risk. However, small water supplies have more facility deficiencies and are responsible for more disease outbreaks than are large ones (Schuster et al., 2005, Wallender et al., 2014; Murphy et al., 2016a, 2016b). Emphasis on regular physical inspections of the water supply system and monitoring of critical processes and activities is important for all small drinking water supplies and particularly for those where testing at the required frequency may be impractical (Robertson et al., 2003; WHO, 2005).
Supplies with a history of high‑quality water may use greater process control and regular inspections as a means for reducing the number of samples taken for bacteriological analysis. Conversely, supplies with variable water quality may be required to sample on a more frequent basis.
Even at the recommended sampling frequencies for E. coli, there are limitations that should be considered when interpreting the sampling results. Simulation studies have shown that it is very difficult to detect a contamination event in a distribution system unless the contamination occurs in a water main, a reservoir, at the treatment plant, or for a long duration at a high concentration (Speight et al., 2004; van Lieverloo et al., 2007). Some improvement in detection capabilities were seen when sampling programs had more staggered sampling across several days including weekends and holidays, as opposed to clustering samples on a single day per week (van Lieverloo et al., 2007). Hrudey and Leiss (2003) point out that there are challenges in accurately interpreting evidence from monitoring programs for environmental hazards when the frequency of the hazard detection is rare. The authors stress the importance of acquiring evidence from more than one source when managing risks from low frequency hazards that can have serious consequences. This highlights the importance of operational monitoring of critical processes and use of multiple microbiological indicators for drinking water verification.
Disinfectant residual tests should be conducted when bacteriological samples are taken. Daily sampling recommendations for disinfectant residual and turbidity testing may not apply to supplies served by groundwater sources in which disinfection is practised to increase the safety margin. Further information on monitoring for turbidity can be found in the guideline technical document for turbidity (Health Canada, 2012a). Other parameters can be used alongside E. coli as part of the water quality water verification process. These include indicators of general microbiological water quality (total coliforms, heterotrophic plate counts) and additional indicators of fecal contamination (e.g., enterococci, coliphages) (WHO and OECD, 2003; WHO, 2005, 2014). More information can be obtained from the corresponding Health Canada documents (Health Canada, 2012b, 2018a, 2019a).
6.3 Location of sampling points
In municipal-scale systems, the location of sampling points must be selected or approved by the responsible drinking water authority. The sampling locations selected may vary depending on the monitoring objectives. For example, sampling at fixed points may be used to help establish a history of water quality within the distribution system, whereas sampling at random (e.g., variable) points throughout the distribution system may provide more coverage of the system. A random sampling strategy such as rotating among sampling sites throughout the distribution system may also improve the probability of detecting water quality issues (WHO, 2014).A combination of both fixed and random sample point locations is common (Narasimhan et al., 2004). Speight et al. (2004) have published a methodology for developing customized distribution system sampling designs that incorporate randomized sample point locations.
Sample sites should include the point of entry into the distribution system and points in the distribution systems that are representative of the quality of water supplied to the consumer. If the water supply is obtained from more than one source, the location of sampling sites should ensure that water from each source is periodically sampled. Distribution system drawings can provide an understanding of water flows and directions and can aid in the selection of appropriate sampling locations. Focus should be placed on potential problem areas, or areas where changes in operational conditions may be expected to occur. Areas with long water detention times (e.g., dead ends), areas of depressurization, reservoirs, locations downstream of storage tanks, areas farthest from the treatment plant, and areas with a poor previous record are suggested sampling sites. Source water and system assessments may identify additional areas that are appropriate sampling sites.
In residential-scale systems that provide drinking water to the public, samples are generally collected from the locations recommended by the responsible drinking water authority.
7.0 Treatment technology and distribution system considerations
The primary goal of treatment is to reduce the presence of disease-causing organisms and associated health risks to an acceptable or safe level. This can be achieved through one or more treatment barriers involving physical removal and/or inactivation. A source-to-tap approach, including watershed or wellhead protection, optimized treatment barriers and a well-maintained distribution system is a universally accepted approach to reduce the concentration of waterborne pathogens in drinking water (O'Connor, 2002b; Health Canada, 2001; CCME, 2004; WHO, 2012). Monitoring for E. coli as part of the verification of the quality of the treated and distributed water is an important part of this approach. The Health Canada Guideline Technical Documents: Enteric Protozoa in Drinking Water: Giardia and Cryptosporidium and Enteric Viruses in Drinking Water should be consulted for more information on the requirements for drinking water treatment and disinfection (Health Canada 2012b, 2019d)
7.1 Municipal-scale
An array of options is available for treating source waters to provide high-quality drinking water. The type and the quality of the source water will dictate the degree of treatment necessary. In general, minimum treatment of supplies derived from surface water sources or GUDI should include adequate filtration (or technologies providing an equivalent log reduction credit) and disinfection. As most surface waters and GUDI supplies are subject to fecal contamination, treatment technologies should be in place to achieve a minimum 3 log (99.9%) removal and/or inactivation of Giardia and Cryptosporidium,anda minimum 4-log (99.99%) removal and/or inactivation of enteric viruses. Subsurface sources should be evaluated to determine whether the supply is susceptible to contamination by enteric viruses and protozoa. Those sources determined to be susceptible to viruses should achieve a minimum 4-log removal and/or inactivation of viruses. A jurisdiction may consider it acceptable for a groundwater source not to be disinfected if the assessment of the drinking water system meets provincial or territorial requirements in place to ensure that the risk of enteric virus presence is minimal (Health Canada, 2019d).
In systems with a distribution system, a disinfectant residual should be maintained at all times. It is essential that the removal and inactivation targets are achieved before drinking water reaches the first consumer in the distribution system. Adequate process control measures and operator training are also required to ensure the effective operation of treatment barriers at all times (Smeets et al., 2009; AWWA, 2011).
Overall, the evidence shows that enteric bacterial pathogens are much more sensitive to chlorination than Giardia, Cryptosporidium, and numerous enteric viruses, and more sensitive to UV inactivation than numerous enteric viruses (Health Canada, 2019c, 2019d). Therefore, water that meets the requirements established in the guidelines for enteric viruses and enteric protozoa should have an acceptable bacteriological quality, including achieving E. coli concentrations of none detectable per 100 mL of water leaving the treatment plant.
7.1.1 Physical removal
Physical removal of indicator organisms (E. coli, total coliforms, enterococci) can be achieved using various technologies, including chemically-assisted, slow sand, diatomaceous earth and membrane filtration or an alternative proven filtration technology. Physical log removals for indicator organisms (E. coli, total coliforms, enterococci) reported for several filtration technologies are outlined in Table 3. Reverse osmosis (RO) membranes are expected to be as effective as ultrafiltration based on their molecular weight cut-off (LeChevallier and Au, 2004; Smeets et al., 2006). However, there is currently no method to validate the log removal for RO units (Alspach, 2018).
| TechnologyTable 3 Footnote a | Log removals | |||
|---|---|---|---|---|
| Minimum | Mean | Median | Maximum | |
| Conventional filtration | 1.0 | 2.1 | 2.1 | 3.4 |
| Direct filtration | 0.8 | 1.4 | 1.5 | 3.3 |
| Slow sand filtration | 1.2 | 2.7 | 2.4 | 4.8 |
| Microfiltration | Not given | Not given | Not given | 4.3 |
| Ultrafiltration | Not given | >7 | Not given | Not given |
| ||||
7.1.2 Disinfection
Primary disinfection is required to protect public health by killing or inactivating harmful protozoa, bacteria and viruses, whereas secondary disinfection is used to maintain a residual in the distribution system. A residual in the distribution helps control bacterial regrowth and provide an indication of system integrity (Health Canada, 2009). Primary disinfection is typically applied after treatment processes that remove particles and organic matter. This strategy helps to ensure efficient inactivation of pathogens and minimizes the formation of disinfection by-products. It is important to note that when describing microbial disinfection of drinking water, the term “inactivation” is used to indicate that the pathogen is non-infectious and unable to replicate in a suitable host, although it may still be present.
The five disinfectants commonly used in drinking water treatment are: free chlorine, monochloramine (chloramine), ozone, chlorine dioxide and UV light. Free chlorine is the most common chemical disinfectant used for primary disinfection because it is widely available, is relatively inexpensive and provides a residual that can also be used for secondary disinfection. Chloramine is much less reactive than free chlorine, has lower disinfection efficiency and is generally restricted to use in secondary disinfection. Ozone and chlorine dioxide are effective primary disinfectants against bacteria, viruses and protozoa, although they are typically more expensive and complicated to implement, particularly for small systems. Ozone decays rapidly after being applied, therefore cannot be used for secondary disinfection. Chlorine dioxide is also not recommended for secondary disinfection because of its relatively rapid decay (Health Canada, 2008a). Through a physical process, UV light provides effective inactivation of bacteria, protozoa and most enteric viruses with the exception of adenovirus, which requires a high dose for inactivation. Similar to ozone and chlorine dioxide, UV light is highly effective for primary disinfection, but an additional disinfectant (usually chlorine or chloramine) needs to be added to for secondary disinfection.
7.1.2.1 Chemical disinfection
The efficacy of chemical disinfectants can be predicted based on knowledge of the residual concentration of a specific disinfectant and factors that influence its performance, mainly temperature, pH, contact time and the level of disinfection required (AWWA, 2011). This relationship is commonly referred to as the CT concept, where CT is the product of “C” (the residual concentration of disinfectant, measured in mg/L) and “T” (the disinfectant contact time, measured in minutes) for a specific microorganism under defined conditions (e.g., temperature and pH). To account for disinfectant decay, the residual concentration is usually determined at the exit of the contact chamber rather than using the applied dose or initial concentration. Also, the contact time T is often calculated using a T10 value, which is defined as the detention time at which 90% of the water meets or exceeds the required contact time. The T10 value can be estimated by multiplying the theoretical hydraulic detention time (i.e., tank volume divided by flow rate) by the baffling factor of the contact chamber. The U.S. EPA (U.S. EPA, 1991) provides baffling factors for sample contact chambers. Alternatively, a hydraulic tracer test can be conducted to determine the actual contact time under plant flow conditions. Because the T value is dependent on the hydraulics related to the construction of the treatment installation, improving the hydraulics (i.e., increasing the baffling factor) is more effective to achieve CT requirements than increasing the disinfection dose.
CT values for 99% (2 log) inactivation of E. coli using chlorine, chlorine dioxide, chloramine, and ozone are provided in Table 4. For comparison, CT values for Giardia lamblia and for viruses have also been included. The CT values illustrate the fact that compared with most protozoans and viruses, E. coli are easier to inactivate using the common chemical disinfectants. Table 4 also highlights that chloramine is a much weaker disinfectant than free chlorine, chlorine dioxide or ozone, since much higher concentrations and/or contact times are required to achieve the same degree of inactivation. Consequently, chloramine is not recommended as a primary disinfectant.
In a well-operated treatment system, the CT provided for Giardia or viruses will result in a much greater inactivation than 99% for bacteria. The literature indicates that the enteric bacterial pathogens Salmonella, Campylobacter and E. coli O157:H7 are comparable to non-pathogenic E. coli in terms of their sensitivity to chemical disinfection (Lund, 1996; Rice et al., 1999; Wojcicka et al., 2007; Chauret et al., 2008; Rasheed et al., 2016; Jamil et al., 2017). Published CT values for these pathogens have been limited. Laboratory studies have demonstrated that a 2-4 log inactivation of E. coli O157:H7 can be achieved with CT values of < 0.3 mg·min/L for free chlorine and <30 mg·min/L for monochloramine (Chauret et al., 2008; Wojcicka et al., 2007).
| Disinfectant agent | pH | E. coliTable 4 Footnote a (mg·min/L) [5°C] |
Giardia lambliaTable 4 Footnote b (mg·min/L) [5°C] |
VirusesTable 4 Footnote c (mg·min/L) [5-15°C] |
|---|---|---|---|---|
| Free chlorine | 6–7 | 0.034–0.05 | 70–99 | 0.01–12 |
| Chloramines | 8–9 | 95–180 | 1470 | 360–6476 |
| Chlorine dioxide | 6–7 | 0.4–0.75 | 17 | 0.17–6.7 |
| Ozone | 6–7 | 0.02 | 1.3 | 0.006–0.5 |
|
||||
7.1.2.2 UV disinfection
For UV disinfection, the product of light intensity “I” (measured in mW/cm2 or W/m2) and time “T” (measured in seconds) results in a computed dose (fluence) in mJ/cm2 for a specific microorganism. This relationship is referred to as the IT concept.
Log inactivations using UV light disinfection are listed in Table 5. Due to the importance of E. coli as a public health indicator, it has been used as a representative bacterial species. For comparison, UV light doses for representative protozoa and viruses have also been included. A review of the data on inactivation using UV light shows that, of the representative organisms, bacteria (in this instance, E. coli) and protozoa require comparable doses of UV light to achieve the same level of inactivation, whereas certain viruses are much more resistant.
Non-pathogenic E. coli, Salmonella, Campylobacter and pathogenic E. coli (including E. coli O157:H7) have similar sensitivities to UV disinfection (Sommer et al., 2000; Smeets et al., 2006; Zimmer-Thomas et al., 2007). In studies involving pathogenic E. coli strains, 2-6 log inactivation has been achieved with UV doses ranging from 3-12.5 mJ/cm2 (Sommer et al., 2000; Zimmer-Thomas et al., 2007).
Bacteria have natural defense mechanisms for reversing photochemical damage caused by UV light (e.g., photoreactivation and dark repair) (Hijnen et al., 2006). Experimental studies using high density E. coli populations have found that low levels of dark repair are possible in drinking water with UV light applied at low doses (Zimmer-Thomas et al., 2007; Bohrerova et al., 2015). Present evidence suggests that for water treatment plant operations with well-maintained UV reactors delivering a minimum UV dose of 40 mJ/cm2 followed by the presence of a suitable disinfectant residual, E. coli repair of UV-induced damage should not be of concern (Zimmer-Thomas et al., 2007; Bohrerova et al., 2015).
| Log inactivation | E. coliTable 5 Footnote aTable 5 Footnote c | CryptosporidiumTable 5 Footnote a | AdenovirusTable 5 Footnote b | RotavirusTable 5 Footnote b | GiardiaTable 5 Footnote a |
|---|---|---|---|---|---|
| 1 | 1.5–5 | 2.5 | 10–76 | 7.1–10 | 2.1 |
| 2 | 2.8–9 | 5.8 | 26–137 | 14.8–26 | 5.2 |
| 3 | 4.1–14 | 12 | 39–199 | 23–44 | 11 |
| 4 | 5.0–18 | 22 | 51–261 | 36–61 | 22 |
| |||||
7.1.3 Distribution system
A well-maintained distribution system is a critical component of a source-to-tap or water safety plan approach to provide safe drinking water (Health Canada, 2001; CCME, 2004; WHO, 2014; AWWA 2017). Distribution system water quality is known to deteriorate due to a variety of issues, including long term biofilm regrowth and short term transients/intrusions that may result due to day-to-day operations, as well as accidental cross-contamination or intentional contamination. Large disruptions in distribution system water quality (e.g., resulting from backflows, cross-connections, construction or repairs) have been associated with outbreaks of waterborne illness (Risebro et al., 2007; Craun et al., 2010; Hrudey and Hrudey, 2014). Performance deficiencies during routine operations (e.g., loss of pipe integrity, pressure loss, lack of adequate residual) can also contribute to an increased risk of gastrointestinal illness for consumers (Ercumen et al., 2014). Water quality in the distribution system should be regularly monitored (e.g., microbial indicators, disinfectant residual, turbidity, pH), operations/maintenance programs should be in place (e.g., water main cleaning, cross-connection control, asset management) and strict hygiene should be practiced during all water main construction, repair or maintenance to ensure drinking water is transported to the consumer with minimum loss of quality (Kirmeyer et al., 2001, 2014).
Secondary disinfection may be applied to the treated water as it leaves the treatment plant or at rechlorination points throughout the distribution system. Free chlorine and chloramine are the chemicals commonly used to provide a disinfectant residual. Chloramine penetrates biofilms better than free chlorine, whereas free chlorine has decreased biofilm penetration but acts more effectively where it does penetrate (Lee et al., 2011, Pressman et al., 2012). When chloramine is used as a residual disinfectant in drinking water distribution systems, treatment processes should be optimized for chloramine stability (Cl2:NH3 weight ratio of 4.5:1 – 5:1, pH > 8.0) (Health Canada, 2019b).
The main function of the disinfectant residual is to protect against microbial regrowth (LeChevallier and Au, 2004). The residual can also serve as a sentinel for water quality changes. A drop in residual concentration can provide an indication of treatment process malfunction, inadequate treatment, system contamination or a break in the integrity of the distribution system (LeChevallier, 1998; Haas, 1999; O’Connor, 2002a; AWWA, 2017). The ability of a secondary disinfectant to maintain control of microbiological growth in the distribution system depends on the residual type (i.e., free chlorine or chloramine), concentration, contact time, the residual demand (generated by the water and materials present or entering into the distribution system), the protective effects of biofilms and the disinfectant resistance of the microorganisms present (LeChevallier and Au, 2004). Researchers have noted that a chlorine residual of “detectable” is not sufficient to effectively limit bacterial growth in the distribution system (Gagnon et al., 2008; Wahman and Pressman, 2015). Specific requirements for disinfectant residual concentrations are set by the responsible drinking water authority and may vary among jurisdictions.
Issues of deterioration in distribution system water quality can occur which would not be detected by E. coli monitoring on its own. Of concern in drinking water treatment is the development of biofilms and their potential to capture enteric and opportunistic waterborne pathogens that may show varying ability to survive, multiply and be further released into the distribution system (Ashbolt, 2015). Low and negative transient pressures can create the opportunity for contamination to enter the distribution system from intrusions from outside the pipes or cross connections and/or backflow from domestic, industrial or institutional facilities (Gullick et al., 2004). Secondary disinfectant residuals may not provide sufficient inactivation of pathogenic organisms introduced into the distribution system through intrusions (Payment et al., 1999; Betanzo et al., 2008). Flushing (e.g., conventional flushing, unidirectional flushing) and chlorination are important corrective actions in response to fecal contamination or microbiological water quality deterioration issues (Szabo and Minamyer, 2014). However, if not properly implemented, flushing techniques can mobilize and spread deposits and contamination within the distribution system instead of facilitating their controlled removal. It is therefore important that water utilities identify and implement the most appropriate flushing technique for addressing the particular microbiological water quality issue of concern. Guidance for water utilities on managing water quality in the drinking water distribution system can be found elsewhere (Health Canada, 2019a,).
An issue relevant to remote communities is the potential for contamination between the treated source water and the point of consumption for households and businesses that rely on trucked water and/or on-premise water storage systems. Studies conducted in First Nations and Inuit communities have observed that water samples collected from household water storage containers (Farenhorst et al., 2017) or from water delivery trucks or taps receiving trucked water (Daley et al., 2017; Farenhorst et al., 2017; Mi et al., 2019) had an increased likelihood of testing positive for E. coli compared to piped water supplies. Tap water samples from households receiving trucked water were also noted to have lower free chlorine levels than those recorded in piped drinking water supplies (Daley et al., 2017; Farenhorst et al., 2017). These studies highlight the importance of best management practices for drinking water transport and storage within those communities to reduce the risk of contamination. Specific guidance related to the operation, maintenance and monitoring requirements for trucked water systems and drinking water cisterns or storage tanks should be obtained from the responsible drinking water authority in the affected jurisdiction. Information can also be found in other publications (Agriculture and Agri-Food Canada, 2006; Indian and Northern Affairs Canada, 2006; Health Canada, 2013).
7.2 Residential-scale
Residential-scale treatment is also applicable to small drinking water systems. Evidence indicates that small private and community drinking water supplies are more vulnerable to drinking water contamination and at greater risk for outbreaks of waterborne illness than large municipal drinking water systems (Schuster et al. 2005; Murphy et al., 2016b, Messner et al., 2017). For small groundwater wells that are susceptible to fecal contamination and that provide drinking water to the public, disinfection is the best means for protecting public health (Payment and Locas, 2011). Further guidance on characterizing risks in small systems can be found elsewhere (WHO, 2012).
In cases where an individual household obtains its drinking water from a private well, the susceptibility of the source to fecal contamination should be assessed. Although it is difficult for homeowners to conduct a detailed assessment of the susceptibility of their well to fecal contamination, steps can be taken to minimize the likelihood of a well becoming contaminated. General guidance on well construction, maintenance, protection and testing is typically available from provincial/territorial jurisdictions. If insufficient information is available to determine if a well is susceptible to fecal contamination, treatment of the well is a way to reduce risk. In general, surface water is not recommended as a residential-scale water supply unless it is properly filtered, disinfected and monitored for water quality.
Various options are available for treating source waters to provide high-quality pathogen-free drinking water. These include filtration or disinfection with chlorine-based compounds or UV light. These technologies are similar to the municipal treatment barriers, but on a smaller scale. Many of these technologies have been incorporated into point-of-entry devices, which treat all water entering the system, or point-of-use devices, which treat water at only a single location—for example, at the kitchen tap. Because of the potential public health risks from the use of microbiologically-contaminated drinking water, if POU devices are used instead of a point-of-entry system, all points of water used for drinking, food and beverage preparation, hygiene or washing dishes should be equipped with point-of-use treatment devices.
Specific guidance on technologies that can be used in small systems should be obtained from the responsible drinking water authority in the affected jurisdiction. Health Canada does not recommend specific brands of drinking water treatment devices, but it strongly recommends that consumers use devices that have been certified by an accredited certification body as meeting the appropriate NSF International (NSF)/American National Standards Institute (ANSI) drinking water treatment unit standards. These standards have been designed to safeguard drinking water by helping to ensure the material safety and performance of products that come into contact with drinking water.
Certification organizations provide assurance that a product conforms to applicable standards and must be accredited by the Standards Council of Canada (SCC). In Canada, the following organizations have been accredited by the SCC to certify drinking water devices and materials as meeting NSF/ANSI standards (SCC, 2019):
- CSA Group;
- NSF International;
- Water Quality Association;
- Underwriters Laboratories Inc.;
- Truesdail Laboratories Inc.;
- Bureau de Normalisation du Québec; and
- International Association of Plumbing & Mechanical Officials.
An up-to-date list of accredited certification organizations can be obtained from the SCC (2019).
Residential-scale supplies that use liquid chlorine should use hypochlorite solutions that are certified as meeting NSF/ANSI/CAN Standard 60 (NSF/ANSI/CAN, 2018b) and follow the handling and storage recommendations for hypochlorite outlined in (Health Canada, 2018b).
For UV disinfection systems, NSF/ANSI Standard 55 provides performance criteria for two categories of certified systems, Class A and Class B (NSF/ANSI, 2019). UV systems certified to NSF/ANSI Standard 55 Class A are designed to deliver a UV dose that is at least equivalent to 40 mJ/cm2 in order to inactivate microorganisms, including bacteria, viruses, Cryptosporidium oocysts, and Giardia cysts, from contaminated water. However, they are not designed to treat wastewater or water contaminated with raw sewage and should be installed in visually clear water. Systems certified to NSF Standard 55 Class B are not intended for the disinfection of microbiologically unsafe water. Class B system are only certified for supplemental bactericidal treatment of disinfected public drinking water or other drinking water that has been tested and deemed acceptable for human consumption.
Some jurisdictions may require semi-public systems to provide a UV dose of 186 mJ/cm2 if adenovirus may be present (e.g., sewage influence). UV systems, designed in accordance with the U.S. EPA UV Disinfection Guidance Manual, are commercially available to deliver 186 mJ/cm2 for supplies up to 24 U.S. gpm and >68% UV transmittance (U.S. EPA, 2006c).
RO membranes have a pore size smaller than bacteria and viruses and could provide a physical barrier to remove them. However, NSF/ANSI Standard 58 (NSF/ANSI, 2018a) does not include claims for bacteria or virus reduction. It is important to note that RO systems are intended for POU installation only. This is because water treated by a RO system may be corrosive to internal plumbing components. These systems also require larger quantities of influent water to obtain the required volume of drinking water and are generally not practical for point-of-entry (POE) installation.
Ultrafiltration membranes have pore sizes ranging from 0.005 to 0.05 µm and could also provide a physical barrier to bacteria and viruses, although there is no NSF/ANSI standard for residential-scale ultrafiltration systems. For drinking water systems requiring larger capacity, some ultrafiltration units certified to NSF/ANSI Standard 419 (NSF/ANSI, 2018c) have been tested for virus reduction; these would also provide bacteria reduction. However, direct integrity testing does not have enough resolution to validate bacteria or virus log removals. The responsible drinking water authority in the affected jurisdiction should be contacted to confirm the acceptability and/or applicability of these units. To verify that the treatment device is effective, the system owner/operator or homeowner should periodically test both the water entering the treatment device and the treated water for E. coli and total coliforms using an accredited laboratory. Treatment devices lose their removal capacity through usage and time and need to be maintained and/or replaced. Consumers should verify the expected longevity of the components in their treatment device according to the manufacturer's recommendations and establish a clearly defined maintenance schedule. Treatment devices should be inspected and serviced in accordance with the maintenance schedule and manufacturer's recommendations.
8.0 Risk assessment
A health-based risk assessment for E. coli is not appropriate since E. coli is used only as an indicator organism. Risk assessments have been done for specific microbiological organisms that have health implications, such as the enteric protozoa and enteric viruses (Health Canada, 2019c, 2019d).
8.1 International considerations
Other countries and multi-national organizations use E. coli as an indicator organism in drinking water monitoring. The WHO Guidelines for drinking-water quality (WHO, 2017) recommend E. coli as the first indicator of choice in verification monitoring programs under a Water Safety Plan-based approach to drinking water quality management. E. coli or thermotolerant coliforms must not be detected in any sample of water directly intended for drinking or in any sample of treated water entering into or within the distribution system (100 mL sample volume). Immediate investigative action must be taken if E. coli is detected (WHO, 2017).
In the United States, the U.S. EPA's Revised Total Coliform Rule (U.S. EPA, 2013) applies to all public water systems and specifies a maximum contaminant level (MCL) and a maximum contaminant level goal (MCLG) of zero E. coli in any drinking water sample (standard volume of 100 mL). Detection of E. coli in any routine monitoring or repeat sample is a violation of the MCL.
The U.S. EPA's Groundwater Rule (U.S. EPA, 2006a), applies to public water systems that use ground water. Under the rule, systems not providing 4-log virus reduction must conduct triggered source water monitoring whereby systems notified of a positive total coliform must test for a fecal indicator (i.e., E. coli, enterococci or coliphage).
The EU Drinking Water Directive outlines the legislative requirements for all its Member States (EU, 1998). Under the Directive, E. coli is categorized as a Part A parameter to verify that the measures in place to control risks to human health throughout the water supply chain are working effectively and that water at the point of compliance is wholesome and clean. The EU standard for E. coli is a value of zero per 100 mL (EU, 1998).
The Australian Drinking Water Guidelines (NHMRC, NRMMC, 2017) specify that for verification of microbiological quality of drinking water a regular testing program should be instituted for the indicator E. coli wherever possible. According to the Guidelines, the performance measure for E. coli as an indicator of short term water quality within the distribution system is none detected in a minimum 100 mL sample of drinking water.
9.0 Rationale
E. coli is the most widely used indicator for detecting fecal contamination in drinking water supplies worldwide. As a fecal indicator, E. coli is predominantly associated with human and animal feces and is more fecal-specific than other indicator groups such as total coliforms, thermotolerant coliforms and enterococci. Its high numbers in feces and ability to be easily and affordably measured make it a useful indicator for detecting fecal contamination that has been reduced to low levels in drinking water environments. An additional advantage to using E. coli as a fecal indicator is that many test methods detect total coliformswhile simultaneously differentiating E. coli. Using multiple parameters in drinking water monitoring as indicators of general microbiological water quality (such as total coliforms, heterotrophic plate counts) or additional indicators of fecal contamination (enterococci) is a good way for water utilities to enhance the potential to identify issues and thus trigger responses. As an indicator, E. coli is not without its limitations. E. coli is more easily inactivated by environmental stresses and drinking water disinfectants than many waterborne pathogens, in particular, enteric viruses and enteric protozoa. Nevertheless, E. coli is an important indicator of the performance of treatment barriers in place to remove or inactivate these organisms.
Current drinking water guidelines encourage the adoption of a drinking water quality management system that uses a source-to-tap or water safety plan approach. Under a system of drinking water management where hazards in the drinking water system are being controlled and operational monitoring parameters are in place to show that the system is operating adequately, E. coli has an important role in drinking water quality verification. Monitoring for E. coli provides a check of the performance of the system controls in place to produce water that is microbiologically acceptable.
The detection of E. coli in drinking water indicates the inadequate function of one or more system controls and that a pathway exists for fecal contamination to potentially reach the consumer, which is unacceptable. As a result, the guideline for E. coli in drinking water systems is a maximum acceptable concentration of none detectable per 100 mL.
10.0 References
- Abberton, C.L., Bereschenko, L., van der Wielen, P.W.J.J. and Smith, C.J. (2016). Survival, biofilm formation, and growth potential of environmental and enteric Escherichia coli strains in drinking water microcosms. Appl. Environ. Microbiol., 82(17): 5320-5331.
- Agriculture and Agri-Food Canada (2006). Water quality matters: Maintaining safe domestic water quality with on-farm cisterns and water tanks. Agriculture and Agri-Food Canada, Ottawa, Ontario. TRE-120-2006-02.
- Ahammed, M.M. (2003). Effect of holding time and temperature on bacterial counts. Indian J. Environ. Health, 45(3): 209-212.
- Allen, M.J., Edberg, S.C., Clancy, J.L. and Hrudey, S.E. (2015). Drinking water microbial myths. Crit. Rev. Microbiol., 41(3): 366-373.
- Alspach, B.(2018). Pathogen Rejection in Potable Reuse: The Role of NF/RO and Importance of Integrity Testing: J. Am. Water Works Assoc., 110 (3):39-44.
- APHA, AWWA and WEF (2017). Standard methods for the examination of water and wastewater. 23rd edition. American Public Health Association, American Water Works Association and Water Environment Federation, Washington, DC.
- Ashbolt, N.J. (2015). Environmental (saprozoic) pathogens of engineered water systems: Understanding their ecology for risk assessment and management. Pathogens, 4(2): 390-405.
- AWWA (2011). Water quality and treatment: A handbook of community water supplies. 6th. Edzwald, J.K. (ed.). McGraw-Hill, New York.
- AWWA (2014). Small Systems Field Guide, Water and Wastewater. American Water Works Association Denver, CO.
- AWWA (2017). Manual of Water Supply Practices M68– Water Quality in Distribution Systems. American Water Works Association, Denver, CO.
- Badgley, B.D., Ferguson, J., Heuvel, A.V., Kleinheinz, G.T., McDermott, C.M., Sandrin, T.R., Kinzelman, J., Junion, E.A., Byappanahalli, M.N., Whitman, R.L. and Sadowsky, M.J. (2011). Multi-scale temporal and spatial variation in genotypic composition of cladophora-borne Escherichia coli populations in Lake Michigan. Water Res., 45(2): 721-731.
- Banihashemi, A., Van Dyke, M.I. and Huck, P.M. (2015). Detection of viable bacterial pathogens in a drinking water source using propidium monoazide-quantitative PCR. J. Water Supply Res. T., 64(2): 139-148.
- Bernasconi, C., Volponi, G. and Bonadonna, L. (2006). Comparison of three different media for the detection of E. coli and coliforms in water. pp. 141-145.
- Betanzo, E.W., Hofmann, R., Hu, Z., Baribeau, H. and Alam, Z. (2008). Modeling the impact of microbial intrusion on secondary disinfection in a drinking water distribution system. J. Environ. Eng., 134(4): 231–237.
- Bjergbæk, L.A., Roslev, P. (2005). Formation of nonculturable Escherichia coli in drinking water. J. Appl. Microbiol., 99 (5):1090-1098.
- Blaustein, R.A., Pachepsky, Y., Hill, R.L., Shelton, D.R. and Whelan, G. (2013). Escherichia coli survival in waters: Temperature dependence. Water Res., 47(2): 569-578.
- Bogosian, G., Sammons, L.E., Morris, P.J.L., O'Neil, J.P., Heitkamp, M.A. and Weber, D.B. (1996). Death of the Escherichia coli K-12 strain W3110 in soil and water. Appl. Environ. Microbiol., 62(11): 4114-4120.
- Bohrerova, Z., Rosenblum, J. and Linden, K.G. (2015). Importance of recovery of E. coli in water following ultraviolet light disinfection. J. Env. Eng. (United States), 141(6).
- Botes, M., De Kwaadsteniet, M. and Cloete, T.E. (2013). Application of quantitative PCR for the detection of microorganisms in water. Anal. Bioanal. Chem., 405(1): 91-108.
- Boubetra, A., Nestour, F.L., Allaert, C., Feinberg, M. (2011). Validation of alternative methods for the analysis of drinking water and their application to Escherichia coli. Appl. Environ. Microbiol., 77 (10): 3360-3367.
- Breitenmoser, A., R. Fretz, J. Schmid, A. Besl, and R. Etter. 2011. Outbreak of acute gastroenteritis due to a washwater-contaminated water supply, Switzerland, 2008. J. Water Health. 9(3): 569–576.
- Bushon, R.N., Brady, A.M.G. and Lindsey, B.D. (2015). Holding-time and method comparisons for the analysis of fecal-indicator bacteria in groundwater. Environ. Monit. Assess., 187(11).
- Byappanahalli, M.N., Whitman, R.L., Shively, D.A., Sadowsky, M.J. and Ishii, S. (2006). Population structure, persistence, and seasonality of autochthonous Escherichia coli in temperate, coastal forest soil from a Great Lakes watershed. Environ. Microbiol., 8 (3):504-513.
- Byappanahalli, M.N., Nevers, M.B., Korajkic, A., Staley, Z.R. and Harwood, V.J. (2012a). Enterococci in the environment. Microbiol. Mol. Biol. Rev., 76: 685–706.
- Byappanahalli, M.N., Yan, T., Hamilton, M.J., Ishii, S., Fujioka, R.S., Whitman, R.L. and Sadowsky, M.J. (2012b). The population structure of Escherichia coli isolated from subtropical and temperate soils. Sci. Total Environ., 417-418: 273-279.
- CCME (2004). From source to tap: Guidance on the multi-barrier approach to safe drinking water. Canadian Council of Ministers of the Environment. Winnipeg, Manitoba. Available at: www.ccme.ca/files/Resources/water/source_tap/mba_guidance_doc_e.pdf.
- CEAEQ (2018). Centre d'Expertise en Analyse Enivironmentale du Québec. Lignes directrices concernant les travaux analytiques en microbiologie, DR-12-SCA-02, Québec, Ministère du Développement durable, de l'Environnement et de la Lutte contre les changements climatiques, 2018, 38 p.
- Chaudhuri, R.R. and Henderson, I.R. (2012). The evolution of the Escherichia coli phylogeny. Infect. Genet. Evol., 12(2): 214-226.
- Chauret, C., Smith, C. and Baribeau, H. (2008). Inactivation of Nitrosomonas europaea and pathogenic Escherichia coli by chlorine and monochloramine. J. Water Health, 6(3): 315-322.
- Chen, S., Li, X., Wang, Y., Zeng, J., Ye, C., Li, X., Guo, L., Zhang, S., Yu, X. (2018). Induction of Escherichia coli into a VBNC state through chlorination/chloramination and differences in characteristics of the bacterium between states. Water Res., 142:279-288.
- Ciebin, B.W., Brodsky, M.H., Eddington, R., Horsnell, G., Choney, A., Palmateer, G., Ley, A., Joshi, R. and Shears, G. (1995). Comparative evaluation of modified m-FC and m-TEC media for membrane filter enumeration of Escherichia coli in water. Appl. Environ. Microbiol., 61(11): 3940-3942.
- Clermont, O., Bonacorsi, S. and Bingen, E. (2000). Rapid and simple determination of the Escherichia coli phylogenetic group. Appl. Environ. Microbiol., 66(10): 4555-4558.
- Craun, G.F., Nwachuku, N., Calderon, R.L., Craun, M.F. (2002). Outbreaks in drinking-water systems, 1991-1998. J. Environ. Health, 65 (1): 16-23.
- Craun, G.F., Brunkard, J.M., Yoder, J.S., Roberts, V.A., Carpenter, J., Wade, T., Calderon, R.L., Roberts, J.M., Beach, M.J. and Roy, S.L. (2010). Causes of outbreaks associated with drinking water in the united states from 1971 to 2006. Clin. Microbiol. Rev., 23(3): 507-528.
- Cretikos, M., Byleveld, P., Durrheim, D.N., Porigneaux, P., Merritt, T., Leask, S. (2010). Supply system factors associated with microbiological drinking water safety in regional New South Wales, Australia, 2001-2007. J Water Health, 8 (2):257-268.
- Daley, K., Truelstrup Hansen, L., Jamieson, R.C., Hayward, J.L., Piorkowski, G.S., Krkosek, W., Gagnon, G.A., Castleden, H., MacNeil, K., Poltarowicz, J., Corriveau, E., Jackson, A., Lywood, J. and Huang, Y. (2017). Chemical and microbial characteristics of municipal drinking water supply systems in the Canadian Arctic. Environ. Sci. Pollut. R., (Epub ahead of print) doi: 10.1007/s11356-017-9423-5.
- Defelice, N.B., Johnston, J.E., Gibson, J.M. (2016). Reducing emergency department visits for acute gastrointestinal illnesses in North Carolina (USA) by extending community water service. Environ. Health Perspect., 124 (10):1583-1591.
- Diarra, M.S., Silversides, F.G., Diarrassouba, F., Pritchard, J., Masson, L., Brousseau, R., Bonnet, C., Delaquis, P., Bach, S., Skura, B.J. and Topp, E. (2007). Impact of feed supplementation with antimicrobial agents on growth performance of broiler chickens, Clostridium perfringens and Enterococcus counts, and antibiotic resistance phenotypes and distribution of antimicrobial resistance determinants in Escherichia coli isolates. Appl. Environ. Microbiol., 73 (20): 6566-6576.
- Duriez, P. and Topp, E. (2007). Temporal dynamics and impact of manure storage on antibiotic resistance patterns and population structure of Escherichia coli isolates from a commercial swine farm. Appl. Environ. Microbiol., 73 (17): 5486-5493.
- Duygu, D.Y. and Udoh, A.U. (2017). Validation of microbiological testing methods. Trakya University Journal of Natural Sciences, 18(1): 65-69.
- Edberg, S.C., Rice, E.W., Karlin, R.J. and Allen, M.J. (2000). Escherichia coli: The best biological drinking water indicator for public health protection. J. Appl. Microbiol. Symposium Supplement, 88(29): 106s-116s.
- Edge, T.A., Khan, I.U.H., Bouchard, R., Guo, J., Hill, S., Locas, A., Moore, L., Neumann, N., Nowak, E., Payment, P., Yang, R., Yerubandi, R. and Watson, S. (2013). Occurrence of waterborne pathogens and Escherichia coli at offshore drinking water intakes in Lake Ontario. Appl. Environ. Microbiol., 79(19): 5799-5813.
- Engberg, J., Gerner-Smidt, P., Scheutz, F., Nielsen, E.M., On, S.L.W., Mølbak, K. (1998). Water-borne Campylobacter jejuni infection in a Danish town - A 6-week continuous source outbreak. Clinical Microbiology and Infection, 4 (11): 648-656.
- Ercumen, A., Gruber, J.S. and Colford Jr., J.M. (2014). Water distribution system deficiencies and gastrointestinal illness: A systematic review and meta-analysis. Environ. Health Perspect., 122(7): 651-660.
- Ervin, J.S., Russell, T.L., Layton, B.A., Yamahara, K.M., Wang, D., Sassoubre, L.M., Cao, Y., Kelty, C.A., Sivaganesan, M., Boehm, A.B., Holden, P.A., Weisberg, S.B. and Shanks, O.C. (2013). Characterization of fecal concentrations in human and other animal sources by physical, culture-based, and quantitative real-time PCR methods. Water Res., 47(18): 6873-6882.
- EU (1998). Council Directive of 3 November 1998 on the quality of water intended for human consumption. Official Journal of the European Communities. L 330/32 5.12.98.
- Falco, R., and S.I. Williams. 2009.Waterborne Salmonella Outbreak in Alamosa, Colorado. March and April 2008: Outbreak Identification, Response and Investigation. Denver, Colo.: Colorado Department of Public Health and Environment. Safe Drinking Water Program, Water Quality Division.
- Falkinham, J.O.,III, Pruden, A. and Edwards, M. (2015). Opportunistic premise plumbing pathogens: Increasingly important pathogens in drinking water. Pathogens, 4(2): 373-386.
- Farenhorst, A., Li, R., Jahan, M., Tun, H.M., Mi, R., Amarakoon, I., Kumar, A. and Khafipour, E. (2017). Bacteria in drinking water sources of a First Nation reserve in Canada. Sci. Total Environ., 575: 813-819.
- Farnleitner, A.H., Ryzinska-Paier, G., Reischer, G.H., Burtscher, M.M., Knetsch, S., Kirschner, A.K.T., Dirnböck, T., Kuschnig, G., Mach, R.L., Sommer, R. (2010). Escherichia coli and enterococci are sensitive and reliable indicators for human, livestock and wildlife faecal pollution in alpine mountainous water resources. J. Appl. Microbiol. 109 (5): 1599-1608.
- Fass, S., Dincher, M.L., Reasoner, D.J., Gatel, D. and Block, J.C. (1996). Fate of Escherichia coli experimentally injected in a drinking water distribution pilot system. Water Res., 30(9): 2215-2221.
- Feng, P., Lum, R. and Chang, G.W. (1991). Identification of uidA gene sequences in β-D-glucuronidase-negative Escherichia coli. Appl. Environ. Microbiol., 57(1): 320-323.
- Feng, P. and Lampel, K.A. (1994). Genetic analysis of uidA expression in enterohaemorrhagic Escherichia coli serotype O157:H7. Microbiology, 140(8): 2101-2107.
- Frick, C., Vierheilig, J., Linke, R., Savio, D., Zornig, H., Antensteiner, R., Baumgartner, C., Bucher, C., Blaschke, A.P., Derx, J., Kirschner, A.K.T., Ryzinska-Paier, G., Mayer, R., Seidl, D., Nadiotis-Tsaka, T., Sommer, R., Farnleitner, A.H. (2018). Poikilothermic animals as a previously unrecognized source of fecal indicator bacteria in a backwater ecosystem of a large river. Appl. Environ. Microbiol. 84 (16), art. no. e00715-18.
- Filip, Z., Kaddu-Mulindwa, D. and Milde, G. (1986). Survival and adhesion of some pathogenic and facultative pathogenic micro-organisms in groundwater. Water Sci. Technol., 19(7): 1189.
- Flint, K.P. (1987). The long-term survival of Escherichia coli in river water. J. Appl. Bacteriol., 63(3): 261-270.
- Fogarty, J., Thornton, L., Hayes, C., Laffoy, M., O'flanagan, D., Devlin, J., Corcoran, R. (1995). Illness in a community associated with an episode of water contamination with sewage. Epidemiol. Infect., 114 (2):289-295.
- Foppen, J.W.A. and Schijven, J.F. (2006). Evaluation of data from the literature on the transport and survival of Escherichia coli and thermotolerant coliforms in aquifers under saturated conditions. Water Res., 40(3): 401-426.
- Fout, G.S., Borchardt, M.A., Kieke, B.A., Jr. and Karim, M.R. (2017). Human virus and microbial indicator occurrence in public-supply groundwater systems: Meta-analysis of 12 international studies. Hydrogeol. J., 25(4): 903-919.
- Fricker, C.R., DeSarno, M., Warden, P.S. and Eldred, B.J. (2008). False-negative β-D-glucuronidase reactions in membrane lactose glucuronide agar medium used for the simultaneous detection of coliforms and Escherichia coli from water. Lett. Appl. Microbiol., 47(6): 539-542.
- Fricker, C.R., Warden, P.S. and Eldred, B.J. (2010). Understanding the cause of false negative β-D-glucuronidase reactions in culture media containing fermentable carbohydrate. Lett. Appl. Microbiol., 50(6): 547-551.
- Gagnon, G.A., Baribeau, H., Rutledge, S.O., Dumancic, R., Oehmen, A., Chauret, C. and Andrews, S. (2008). Disinfectant efficacy in distribution systems: A pilot-scale assessment. J. Water Supply Res. T., 57(7): 507-518.
- Gensberger, E.T., Polt, M., Konrad-Köszler, M., Kinner, P., Sessitsch, A. and Kostić, T. (2014). Evaluation of quantitative PCR combined with PMA treatment for molecular assessment of microbial water quality. Water Res., 67: 367-376.
- Gerba, C.P. and Smith Jr., J.E. (2005). Sources of pathogenic microorganisms and their fate during land application of wastes. J. Environ. Qual., 34(1): 42-48.
- Golberg, A., Linshiz, G., Kravets, I., Stawski, N., Hillson, N.J., Yarmush, M.L., Marks, R.S. and Konry, T. (2014). Cloud-enabled microscopy and droplet microfluidic platform for specific detection of Escherichia coli in water. PLoS ONE, 9 (1), art. no. e86341.
- Goldstein, S.T., Juranek, D.D., Ravenholt, O., Hightower, A.W., Martin, D.G., Mesnik, J.L., Griffiths, S.D., Bryant, A.J., Reich, R.R. and Herwaldt, B.L. (1996). Cryptosporidiosis: An outbreak associated with drinking water despite state-of-the-art water treatment. Ann. Intern. Med., 124(5): 459-468.
- Gordon, D.M., Bauer, S. and Johnson, J.R. (2002). The genetic structure of Escherichia coli populations in primary and secondary habitats. Microbiology, 148(5): 1513-1522.
- Gordon, D.M. (2013). The ecology of Escherichia coli. In: Escherichia coli: Pathotypes and Principles of Pathogenesis: Second Edition. Donnenberg, M.S. (ed.) Academic Press. London. pp. 3-20.
- Government Inquiry into Havelock North Drinking Water (2017). Report of the Havelock North drinking water inquiry: Stage 2. December 2017, Auckland, New Zealand. ISBN: 978-0-473-39743-2. Available at: https://www.dia.govt.nz/Government-Inquiry-into-Havelock-North-Drinking-Water
- Gutiérrez-del-Río, I., Marín, L., Fernández, J., Millán, M.Á.S., Ferrero, F.J., Valledor, M., Campo, J.C., Cobián, N., Méndez, I., Lombó, F. (2018). Development of a biosensor protein bullet as a fluorescent method for fast detection of Escherichia coli in drinking water. PLoS ONE, 13 (1), art. no. e0184277.
- Gullick, R.W., LeChevallier, M.W., Svindland, R.C. and Friedman, M.J. (2004). Occurrence of transient low and negative pressures in distribution systems. J. Am. Water Works Assoc., 96(11): 52–66.
- Haas, C.N. (1999). Benefits of using a disinfectant residual. J. Am. Water Works Assoc., 91(1): 65-69.
- Hamilton, K.A., Waso, M., Reyneke, B., Saeidi, N., Levine, A., Lalancette, C., Besner, M.-C., Khan, W., Ahmed, W. (2018). Cryptosporidium and Giardia in wastewater and surface water environments. J. Environ. Qual., 47 (5):1006-1023.
- Hamouda, M.A., Anderson, W.B., Van Dyke, M.I., Douglas, I.P., McFadyen, S.D., Huck, P.M. (2016). Scenario-based quantitative microbial risk assessment to evaluate the robustness of a drinking water treatment plant. Water Qual. Res. J. Can., 51 (2), pp. 81-96.
- Hargy, T.M., Rosen, J., Lechevallier, M., Friedman, M. and Clancy, J.L. (2010). A high-volume sampling method for total coliform and E. coli. J. Am. Water Works Assoc., 102(3): 79-86.
- Hayes, E.B., Matte, T.D., O'brien, T.R., Mckinley, T.W., Logsdon, G.S., Rose, J.B., Ungar, B.L.P., Word, D.M., Wilson, M.A., Long, E.G., Hurwitz, E.S., Juranek, D.D. and Fishman, A.P. (1989). Large community outbreak of cryptosporidiosis due to contamination of a filtered public water supply. N. Engl. J. Med., 320(21): 1372-1376.
- Health Canada (2001). Guidance for Safe Drinking Water in Canada: From Intake to Tap Prepared by the Federal-Provincial-Territorial Subcommittee on Drinking Water of the Federal-Provincial-Territorial Committee on Environmental and Occupational Health. December, 2001. Health Canada, Ottawa, Canada. Accessed October 2019 from: https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/water-quality/guidance-safe-drinking-water-canada-intake-health-canada-2001.html.
- Health Canada (2006). Guidelines for Canadian drinking water quality: Guideline technical document — Trihalomethanes. Water Quality and Health Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-trihalomethanes.html
- Health Canada (2008a). Guidelines for Canadian drinking water quality: Guideline technical document — Chlorite and chlorate. Water Quality and Health Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-chlorite-chlorate.html
- Health Canada (2008b). Guidelines for Canadian drinking water quality: Guideline technical document — Haloacetic acids. Water, Air and Climate Change Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-haloacetic-acids.html
- Health Canada (2009) Guidelines for Canadian drinking water quality: Guideline Technical Document—Chlorine. Water, Air and Climate Change Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-chlorine-guideline-technical-document.html
- Health Canada (2011). Guidelines for Canadian drinking water quality: Guideline technical document — N-nitrosodimethylamine. Water, Air and Climate Change Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-n-nitrosodimethylamine-ndma.html
- Health Canada (2012a). Guidelines for Canadian drinking water quality: Guideline technical document — Turbidity. Water, Air and Climate Change Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-turbidity.html
- Health Canada (2012b). Guidelines for Canadian drinking water quality: Guideline technical document — Total coliforms. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-total-coliforms.html
- Health Canada (2013). Guidance on providing safe drinking water in areas of federal jurisdiction, version 2, Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidance-providing-safe-drinking-water-areas-federal-jurisdiction-version-2.html
- Health Canada (2015). Guidance for issuing and rescinding boil water advisories in Canadian drinking water supplies. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidance-issuing-rescinding-boil-water-advisories-canadian-drinking-water-supplies.html
- Health Canada (2018a). Guidance on the use of enterococci as an indicator in Canadian drinking water supplies - in preparation. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario.
- Health Canada (2018b). Guidelines for Canadian drinking water quality: Guideline technical document — Bromate. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-bromate/guidance-document.html
- Health Canada (2018c). Personal communication with T. Beattie. Analysis based on data from the Drinking Water Advisories Application of the Canadian Network for Public Health Intelligence (CNPHI).
- Health Canada (2018d). Summary of E. coli monitoring data from annual drinking water quality reports published by Canadian drinking water utilities. Available upon request.
- Health Canada (2019a). Guidance on monitoring the biological stability of drinking water in distribution systems – in preparation. Document for public consultation. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario.
- Health Canada (2019b). Guidelines for Canadian drinking water quality: Guideline technical document — Chloramines. Water Quality and Health Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-chloramines.html
- Health Canada (2019c). Guidelines for Canadian drinking water quality: Guideline technical document — Enteric protozoa: Giardia and Cryptosporidium. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/water-quality/enteric-protozoa-giardia-cryptosporidium.html
- Health Canada (2019d). Guidelines for Canadian drinking water quality: Guideline technical document — Enteric viruses. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. Available at: https://www.canada.ca/en/health-canada/services/publications/healthy-living/guidelines-canadian-drinking-water-quality-guideline-technical-document-enteric-viruses.html
- Heijnen, L. and Medema, G. (2009). Method for rapid detection of viable Escherichia coli in water using real-time NASBA. Water Res., 43(12): 3124-3132.
- Hijnen, W.A.M., Beerendonk, E.F. and Medema, G.J. (2006). Inactivation credit of UV radiation for viruses, bacteria and protozoan (oo)cysts in water: Water Res., 40(1): 3-22.
- Hill, A.S., Friedman, M., Hallett, M., Salo-Zieman, V., Booth, S., Hanson, A., Gupta, K., Akagi, Y., Kochiss, C.,Koperski, L., Igoe, P., Kirby, L. and Harper, W. (2018). Use of flushing as a corrective action under the revised total coliform rule. Water Research Foundation. Denver, CO.
- Hoff, J.C. (1986). Inactivation of microbial agents by chemical disinfectants. U.S. Environmental Protection Agency, Cincinnati, OH (EPA/600/S2-86/067).
- Hörman, A. and Hänninen, M.-. (2006). Evaluation of the lactose Tergitol-7, m-Endo LES, Colilert 18, Readycult Coliforms 100, Water-Check-100, 3M Petrifilm EC and DryCult Coliform test methods for detection of total coliforms and Escherichia coli in water samples. Water Res., 40(17): 3249-3256.
- Hrudey, S.E. and Leiss, W. (2003). Risk management and precaution: Insights on the cautious use of evidence. Environ. Health Persp., 111 (13):1577-1581.
- Hrudey, S.E. and E.J. Hrudey. 2014 Ensuring Safe Drinking Water – Learning from Frontline Experience with Contamination. American Water Works Association. Denver, CO. 269pp.
- Hunter, P.R., Chalmers, R.M., Hughes, S. and Syed, Q. (2005). Self-reported diarrhea in a control group: a strong association with reporting of low-pressure events in tap water. Clin. Infect. Dis., 40: e32–e34.
- Hynds, P.D., Misstear, B.D. and Gill, L.W. (2012). Development of a microbial contamination susceptibility model for private domestic groundwater sources. Water Resour. Res., 48(12).
- Hynds, P.D., Misstear, B.D. and Gill, L.W. (2013). Unregulated private wells in the Republic of Ireland: Consumer awareness, source susceptibility and protective actions. J. Environ. Manage., 127: 278-288.
- Hynds, P.D., Thomas, M.K. and Pintar, K.D.M. (2014). Contamination of groundwater systems in the US and Canada by enteric pathogens, 1990-2013: A review and pooled-analysis. Plos One, 9(5).
- Ikonen, J., Pitkänen, T., Kosse, P., Ciszek, R., Kolehmainen, M. and Miettinen, I.T. (2017). On-line detection of Escherichia coli intrusion in a pilot-scale drinking water distribution system. J. Environ. Manage., 198: 384-392.
- Indian and Northern Affairs Canada (2006). Protocol for safe drinking water in First Nations communities. Indian and Northern Affairs Canada, Gatineau, Quebec. Available at: www.aadnc-aandc.gc.ca/eng/1100100034913/1100100034920
- Invik, J., Barkema, H.W., Massolo, A., Neumann, N.F. and Checkley, S. (2017). Total coliform and Escherichia coli contamination in rural well water: Analysis for passive surveillance. J. Water Health, 15(5): 729-740.
- Ishii, S. and Sadowsky, M.J. (2008). Escherichia coli in the environment: Implications for water quality and human health. Microbes Environ., 23(2): 101-108.
- Ishii, S., Yan, T., Vu, H., Hansen, D.L., Hicks, R.E. and Sadowsky, M.J. (2010). Factors controlling long-term survival and growth of naturalized Escherichia coli populations in temperate field soils. Microbes Environ., 25(1): 8-14.
- ISO (2018) ISO ICS 07.100.20 – Microbiology of water. International Organization for Standardization. Geneva, Switzerland. Available at: www.iso.org/ics/07.100.20/x/
- Jack, S., Bell, D. and Hewitt, J. (2013). Norovirus contamination of a drinking water supply at a hotel resort. N. Z. Med. J., 126(1387): 98-107.
- Jamil, A., Farooq, S. and Hashmi, I. (2017). Ozone disinfection efficiency for indicator microorganisms at different pH values and temperatures. Ozone: Science and Engineering, 39(6): 407-416.
- Jang, J., Hur, H.-., Sadowsky, M.J., Byappanahalli, M.N., Yan, T. and Ishii, S. (2017). Environmental Escherichia coli: Ecology and public health implications—a review. J. Appl. Microbiol., 123(3): 570-581.
- Juhna, T., Birzniece, D., Larsson, S., Zulenkovs, D., Sharipo, A., Azevedo, N.F., Ménard-Szczebara, F., Castagnet, S., Féliers, C. and Keevil, C.W. (2007). Detection of Escherichia coli in biofilms from pipe samples and coupons in drinking water distribution networks. Appl. Environ. Microbiol., 73(22): 7456-7464.
- Kauppinen, A., Pitkänen, T. and Miettinen, I.T. (2017). Persistent norovirus contamination of groundwater supplies in two waterborne outbreaks. Food Environ. Virol., 10(2): 1-12.
- Keswick, B.H., Secor, S.L., Gerba, C.P. and Cech, I. (1982). Survival of enteric viruses and indicator bacteria in groundwater. J. Environ. Sci. Heal. A., 17(6): 903-912.
- Khan, S.J., Deere, D., Leusch, F.D.L., Humpage, A., Jenkins, M., Cunliffe, D. (2015). Extreme weather events: Should drinking water quality management systems adapt to changing risk profiles? Water Res., 85: 124-136.
- Kim, M., Jung, T., Kim, Y., Lee, C., Woo, K., Seol, J.H. and Yang, S. (2015). A microfluidic device for label-free detection of Escherichia coli in drinking water using positive dielectrophoretic focusing, capturing, and impedance measurement. Biosens. Bioelectron., 74: 1011-1015.
- King, B.J., Monis, P.T. (2007). Critical processes affecting Cryptosporidium oocyst survival in the environment. Parasitology, 134 (3): 309-323.
- Kirmeyer, G.J., Friedman, M., Martel, K., Howie, D., LeChevallier, M., Abbaszadegan, M., Karim, M., Funk, J. and Harbour, J. (2001). Pathogen intrusion into the distribution system. AWWA Research Foundation and American Water Works Association, Denver, CO.
- Kirmeyer, G.J., Thomure, T.M., Rahman, R., Marie, J.L., LeChevallier, M.W., Yang, J., Hughes, D.M. and Schneider, O. (2014). Effective microbial control strategies for main breaks and depressurization. Water Research Foundation, Denver, CO.
- Kotwal, G., Cannon, J.L. (2014). Environmental persistence and transfer of enteric viruses. Curr. Opin. Virol., 4: 37-43.
- Krapf, T., Kuhn, R.M., Kauf, P., Corinne, H.G-D. and Fieseler, L. (2016). Quantitative real-time PCR does not reliably detect single fecal indicator bacteria in drinking water. Wat. Sci. Tech.-W. Sup., 16(6): 1674-1682.
- Krkosek, W., Reed, V. and Gagnon, G.A. (2016). Assessing protozoan risks for surface drinking water supplies in Nova Scotia, Canada. J. Water Health, 14(1): 155-166.
- Krolik, J., Maier, A., Evans, G., Belanger, P., Hall, G., Joyce, A. and Majury, A. (2013). A spatial analysis of private well water Escherichia coli contamination in Southern Ontario. Geospatial Health, 8(1): 65-75.
- Ksoll, W.B., Ishii, S., Sadowsky, M.J. and Hicks, R.E. (2007). Presence and sources of fecal coliform bacteria in epilithic periphyton communities of Lake Superior. Appl. Environ. Microbiol., 73(12): 3771-3778.
- Laine, J., E. Huovinen, M.J. Virtanen, M. Snellman, J. Luomio, P. Ruutu, E. Kujansuu, R. Vuento, T. Pitkanen, I. Miettinen, J. Herrala, O. Lepisto, J. Antonen, J. Helenius, M.-L. Hanninen, L. Maunula, J. Mustonen, M. Kuusi, and the Pirkanmaa Outbreak Study Group. (2011). An extensive gastroenteritis outbreak after drinking-water contamination by sewage effluent, Finland. Epidemiol. Infect. 139(7): 1105–1113.
- Lalancette, C., Papineau, I., Payment, P., Dorner, S., Servais, P., Barbeau, B., Di Giovanni, G.D. and Prévost, M. (2014). Changes in Escherichia coli to Cryptosporidium ratios for various fecal pollution sources and drinking water intakes. Water Res., 55: 150-161.
- Lange, B., Strathmann, M. and Oßmer, R.(2013).Performance validation of chromogenic coliform agar for the enumeration of Escherichia coli and coliform bacteria. Lett. in Appl. Microbiol., 57 (6):547-553.
- Laursen, E., Mygind, O., Rasmussen, B., Ronne, T. (1994). Gastroenteritis: A waterborne outbreak affecting 1600 people in a small Danish town. J. Epidemiol. Commun. H., 48 (5): 453-458.
- LeChevallier, M.W. and Au, K.K. (2004). Water treatment and pathogen control: Process efficiency in achieving safe drinking water. IWA Publishing, London, UK, on behalf of the World Health Organization, Geneva.
- LeChevallier, M.W. (1998). Benefits of employing a disinfectant residual in distribution systems. Water Supp., 16(3-4): 61-73.
- Leclerc, H., Mossel, D.A.A., Edberg, S.C. and Struijk, C.B. (2001). Advances in the bacteriology of the coliform group: Their suitability as markers of microbial water safety. Annu. Rev. Microbiol., 55: 201-234.
- Lee, D.-G., Park, S. and Kim, S.-J. (2007). Influence of Pipe Materials and VBNC Cells on Culturable Bacteria in a Chlorinated Drinking Water Model System. J. Microbiol. Biotechnol., 17(9): 1558-1562.
- Lee, W.H., Wahman, D.G., Bishop, P.L. and Pressman, J.G. (2011). Free chlorine and monochloramine application to nitrifying biofilm: Comparison of biofilm penetration, activity, and viability. Environ. Sci. Technol., 45(4): 1412-1419.
- Lefebvre, B., Malouin, F., Roy, G., Giguère, K. and Diarra, M.S. (2006). Growth performance and shedding of some pathogenic bacteria in feedlot cattle treated with different growth-promoting agents. J. Food Prot., 69 (6):1256-1264.
- Lehtola, M.J., Torvinen, E., Kusnetsov, J., Pitkänen, T., Maunula, L., Von Bonsdorff, C.-., Martikainen, P.J., Wilks, S.A., Keevil, C.W. and Miettinen, I.T. (2007). Survival of Mycobacterium avium, Legionella pneumophila, Escherichia coli, and calicivirases in drinking water-associated biofilms grown under high-shear turbulent flow. Appl. Environ. Microbiol., 73(9): 2854-2859.
- Li, L., Mendis, N., Trigui, H., Oliver, J.D. and Faucher, S.P. (2014). The importance of the viable but non-culturable state in human bacterial pathogens. Front Microbiol. 5(258) 1-20.
- Lim, C.-. and Flint, K.P. (1989). The effects of nutrients on the survival of Escherichia coli in lake water. J. Appl. Bacteriol., 66(6): 559-569.
- Lin, J. and Ganesh, A. Water quality indicators: Bacteria, coliphages, enteric viruses(2013). Int. J. Environ. Heal. R., 23 (6): 484-506.
- Locas, A., Barthe, C., Barbeau, B., Carrière, A. and Payment, P. (2007). Virus occurrence in municipal groundwater sources in Quebec, Canada. Can. J. Microbiol., 53(6): 688-694.
- Locas, A., Barthe, C., Margolin, A.B. and Payment, P. (2008). Groundwater microbiological quality in Canadian drinking water municipal wells. Can. J. Microbiol., 54(6): 472-478.
- Lukjancenko, O., Wassenaar, T.M. and Ussery, D.W. (2010). Comparison of 61 sequenced Escherichia coli genomes. Microb. Ecol., 60(4): 708-720.
- Lund, V. (1996). Evaluation of E. coli as an indicator for the presence of Campylobacter jejuni and Yersinia enterocolitica in chlorinated and untreated oligotrophic lake water. Water Res., 30(6): 1528-1534.
- Maguire, H.C., Holmes, E., Hollyer, J., Strangeways, J.E.M., Foster, P., Holliman, R.E. and Stanwell-Smith, R. (1995). An outbreak of cryptosporidiosis in South London: What value the p values ? Epidemiol. Infect., 115(2): 279-287.
- Maheux, A.F., Huppé, V., Boissinot, M., Picard, F.J., Bissonnette, L., Bernier, J.-.T. and Bergeron, M.G. (2008). Analytical limits of four β-glucuronidase and β-galactosidase-based commercial culture methods used to detect Escherichia coli and total coliforms. J. Microbiol. Methods, 75(3): 506-514.
- Maheux, A.F., Picard, F.J., Boissinot, M., Bissonnette, L., Paradis, S. and Bergeron, M.G. (2009). Analytical comparison of nine PCR primer sets designed to detect the presence of Escherichia coli/Shigella in water samples. Water Res., 43(12): 3019-3028.
- Maheux, A.F., Bissonnette, L., Boissinot, M., Bernier, J.-.T., Huppé, V., Picard, F.J., Bérubé, E. and Bergeron, M.G. (2011). Rapid concentration and molecular enrichment approach for sensitive detection of Escherichia coli and Shigella species in potable water samples. Appl. Environ. Microbiol., 77(17): 6199-6207.
- Maheux, A.F., Dion-Dupont, V., Bisson, M.-A., Bouchard, S., Jubinville, É., Nkuranga, M., Rodrigue, L., Bergeron, M.G., Rodriguez, M.J. (2015a). Multiparametric comparison of chromogenic-based culture methods used to assess the microbiological quality of drinking water and the mFC method combined with a molecular confirmation procedure. J. Water Health, 13 (1):67-72.
- Maheux, A.F., Dion-Dupont, V., Bouchard, S., Bisson, M.-A., Bergeron, M.G. and Rodriguez, M.J. (2015b). Comparison of four ß-glucuronidase and ß-galactosidasebased commercial culture methods used to detect Escherichia coli and total coliforms in water. J. Water Health, 13 (2): 340-352.
- Maheux, A.F., Bouchard, S., Bérubé, E. and Bergeron, M.G. (2017). Comparison of MI, Chromocult® coliform, and Compass CC chromogenic culture-based methods to detect Escherichia coli and total coliforms in water using 16S rRNA sequencing for colony identification. J. Water Health, 15(3): 353-359.
- Maier, A., Krolik, J., Fan, S., Quintin, P., McGolrick, D., Joyce, A. and Majury, A. (2015). Evaluating appropriate maximum holding times for private well water samples. Environmental Health Review (Journal of the Canadian Institute of Public Health Inspectors (CIPHI)). 58:35-40.
- Martin, E.C. and Gentry, T.J. (2016). Impact of enumeration method on diversity of Escherichia coli genotypes isolated from surface water. Lett. Appl. Microbiol., 63 (5): 369-375.
- Martins, M.T., Rivera, I.G., Clark, D.L., Stewart, M.H., Wolfe, R.L. and Olson, B.H. (1993). Distribution of uidA gene sequences in Escherichia coli isolates in water sources and comparison with the expression of β-glucuronidase activity in 4- methylumbelliferyl-β-D-glucuronide media. Appl. Environ. Microbiol., 59(7): 2271-2276.
- McDaniels, A.E., Bordner, R.H., Gartside, P.S., Haines, J.R., Brenner, K.P. and Rankin, C.C. (1985). Holding effects on coliform enumeration in drinking water samples. Appl. Environ. Microbiol., 50(4): 755-762.
- Mendes Silva, D. and Domingues, L. (2015). On the track for an efficient detection of Escherichia coli in water: A review on PCR-based methods. Ecotoxicol. Environ. Saf., 113: 400-411.
- Messner, M.J., Berger, P. and Javier, J. (2017). Total coliform and E. coli in public water systems using undisinfected ground water in the United States. Int. J. Hyg. Environ. Health, 220(4): 736-743.
- Mezule, L. and Juhna, T. (2016). Effect of labile organic carbon on growth of indigenous Escherichia coli in drinking water biofilm. Chemical Engineering Transactions. 49: 619-624.
- Mi, R., Patidar, R., Farenhorst, A., Cai, Z., Sepehri, S., Khafipour, E., Kumar, A. (2019). Detection of fecal bacteria and antibiotic resistance genes in drinking water collected from three First Nations communities in Manitoba, Canada FEMS Microbiol. Lett., 366 (6).
- Miles, S.L., Gerba, C.P., Pepper, I.L., Reynolds, K.A. (2009). Point-of-use drinking water devices for assessing microbial contamination in finished water and distribution systems. Environ. Sci. Technol., 43 (5): 1425-1429.
- Miles, S.L., Sinclair, R.G., Riley, M.R. and Pepper, I.L. (2011). Evaluation of select sensors for real-time monitoring of Escherichia coli in water distribution systems. Appl. Environ. Microbiol., 77(8): 2813-2816.
- MECP (2019). Protocol of Accepted Drinking Water Testing Methods. Version 2.0. Laboratory Services Branch 2002. Ontario Ministry of the Environment, Conservation and Parks. Accessed September 2019 from: https://www.ontario.ca/page/accepted-drinking-water-testing-methods.
- Murphy, H.M., Thomas, M.K., Medeiros, D.T., McFadyen, S. and Pintar, K.D.M. (2016a). Estimating the number of cases of acute gastrointestinal illness (AGI) associated with Canadian municipal drinking water systems. Epidemiol. Infect., 144(7): 1371-1385.
- Murphy, H.M., Thomas, M.K., Schmidt, P.J., Medeiros, D.T., McFadyen, S. and Pintar, K.D.M. (2016b). Estimating the burden of acute gastrointestinal illness due to Giardia, Cryptosporidium, Campylobacter, E. coli O157 and norovirus associated with private wells and small water systems in Canada. Epidemiol. Infect., 144(7): 1355-1370.
- Murphy, H.M., Prioleau, M.D., Borchardt, M.A. and Hynds, P.D. (2017). Review: Epidemiological evidence of groundwater contribution to global enteric disease, 1948–2015. Hydrogeol. J., 25 (4): 981-1001.
- Narasimhan, R., Brereton, J., Abbaszadegan, M., Alum, A. and Ghatpande, P. (2004). Sample collection procedures and locations for bacterial compliance monitoring. AWWA Research Foundation, Denver, CO.
- NHMRC, NRMMC (2017) Australian Drinking Water Guidelines Paper 6 National Water Quality Management Strategy (Version 3.4 Updated October 2017). National Health and Medical Research Council, National Resource Management Ministerial Council, Commonwealth of Australia, Canberra.
- Nichols, G., Lane, C., Asgari, N., Verlander, N.Q. and Charlett, A. (2009). Rainfall and outbreaks of drinking water related disease and in England and Wales. J. Water Health, 7(1): 1-8.
- Novello, A. (2000). The Washington County Fair Outbreak Report. Albany, New York State Department of Health: 108 pp.
- NSF/ANSI (2018a). Standard 58 - Reverse osmosis drinking water treatment systems. NSF International, Ann Arbor, Michigan.
- NSF/ANSI/CAN (2018b). Standard 60- Drinking water treatment chemicals - Health effects. NSF International, Ann Arbor, Michigan.
- NSF/ANSI (2018c). Standard 419 - Public drinking water equipment performance - Filtration. NSF International, Ann Arbor, Michigan.
- NSF/ANSI (2019). Standard 55—Ultraviolet microbiological water treatment systems. NSF International, Ann Arbor, Michigan.
- O'Connor, D.R. (2002a). Report of the Walkerton inquiry, Part one: The events of May 2000 and related issues. Ontario Ministry of the Attorney General (ISBN: 0-7794-2559-6).
- O'Connor, D.R. (2002b). Report of the Walkerton inquiry, Part two report: A strategy for safe drinking water. Ontario Ministry of the Attorney General (ISBN: 0-7794-2621-5).
- Olsen, S.J., Miller, G., Breuer, T., Kennedy, M., Higgins, C., Walford, J., McKee, G., Fox, K., Bibb, W., Mead, P. (2002). A waterborne outbreak of Escherichia coli O157:H7 infections and hemolytic uremic syndrome: Implications for rural water systems. Emerg. Infect. Dis., 8(4): 370-375.
- Olstadt, J., Schauer, J.J., Standridge, J. and Kluender, S. (2007). A comparison of ten USEPA approved total coliform/E. coli tests. J. Water Health, 5(2): 267-282.
- Pachepsky, Y.A., Blaustein, R.A., Whelan, G., Shelton, D.R. (2014). Comparing temperature effects on Escherichia coli, Salmonella, and Enterococcus survival in surface waters. Lett. Appl Microbiol., 59 (3), pp. 278-283.
- Payment, P. (1999). Poor efficacy of residual chlorine disinfectant in drinking water to inactivate waterborne pathogens in distribution systems. Can. J. Microbiol., 45(8): 709–715.
- Payment, P. and Locas, A. (2005). Évaluation et contrôle de la qualité virologique des eaux souterraines. Projet: 3331-24-02-01. Institut Armand-Frappier, Institut national de la recherche scientifique. Laval, Québec.
- Payment, P. and Locas, A. (2011). Pathogens in water: Value and limits of correlation with microbial indicators. Ground Water, 49(1): 4-11.
- Payment, P. and Pintar, K. (2006). Waterborne pathogens: a critical assessment of methods, results and data analysis. Rev. Sci. Eau, 19: 233–245.
- Payment, P., Waite, M. and Dufour, A. (2003). Chapter 2: Introducing parameters for the assessment of drinking water quality. In: Assessing microbial safety of drinking water. Dufour, A. et al. eds. World Health Organization and The Organization for Economic Co-operation and Development. IWA Publishing, London, UK. Pp. 47-77.
- Pope, M.L., Bussen, M., Feige, M.A., Shadix, L., Gonder, S., Rodgers, C., Chambers, Y., Pulz, J., Miller, K., Connell, K. and Standridge, J. (2003). Assessment of the effects of holding time and temperature on Escherichia coli densities in surface water samples. Appl. Environ. Microbiol., 69(10): 6201-6207.
- Pressman, J.G., Lee, W.H., Bishop, P.L. and Wahman, D.G. (2012). Effect of free ammonia concentration on monochloramine penetration within a nitrifying biofilm and its effect on activity, viability, and recovery. Water Res., 46(3): 882-894.
- Puleston, R.L., Mallaghan, C.M., Modha, D.E., Hunter, P.R., Nguyen-Van-Tam, J.S., Regan, C.M., Nichols, G.L., Chalmers, R.M.(2014).The first recorded outbreak of cryptosporidiosis due to Cryptosporidium cuniculus (formerly rabbit genotype), following a water quality incident (2014) J. Water Health, 12 (1), pp. 41-50.
- Rasheed, S., Hashmi, I. and Campos, L. (2016). Inactivation of Escherichia coli and Salmonella with chlorine in drinking waters at various pH and temperature levels. Proceedings of the Pakistan Academy of Sciences, 53(2B): 83-92.
- Rice, E.W., Clark, R.M. and Johnson, C.H. (1999). Chlorine inactivation of Escherichia coli O157:H7. Emerg. Infect. Dis., 5(3): 461-463.
- Risebro, H.L., Doria, M.F., Andersson, Y., Medema, G., Osborn, K., Schlosser, O. and Hunter, P.R. (2007). Fault tree analysis of the causes of waterborne outbreaks. J. Water Health, 5(Suppl. 1): 1-18.
- Robertson, W., Stanfield, G., Howard, G. and Bartram, J. (2003). Chapter 6 - Monitoring the quality of drinking water during storage and distribution. In: Assessing microbial safety of drinking water. Dufour, A. et al. eds. World Health Organization and The Organization for Economic Co-operation and Development. IWA Publishing, London, UK. Pp. 179-204.
- Saby, M., Larocque, M., Pinti, D.L., Barbecot, F., Gagné, S., Barnetche, D. and Cabana, H. (2017). Regional assessment of concentrations and sources of pharmaceutically active compounds, pesticides, nitrate, and E. coli in post-glacial aquifer environments (Canada). Sci. Total Environ., 579: 557-568.
- Sampson, R.W., Swiatnicki, S.A., Osinga, V.L., Supita, J.L., McDermott, C.M. and Kleinheinz, G.T. (2006). Effects of temperature and sand on E. coli survival in a Northern lake water microcosm. J Water Health, 4(3): 389-393.
- SCC (2019). Directory of accredited product, process and service certification bodies. Standards Council of Canada, Ottawa, Ontario. Accessed December 2019 from: www.scc.ca/en/accreditation/product-process-and-service-certification/directory-of-accredited-clients
- Schuster, C.J., Ellis, A.G., Robertson, W.J., Charron, D.F., Aramini, J.J., Marshall, B.J. and Medeiros, D.T. (2005). Infectious disease outbreaks related to drinking water in Canada, 1974-2001. Can. J. Public Health, 96(4): 254-255.
- Sinton, L., Hall, C., Braithwaite, R. (2007). Sunlight inactivation of Campylobacter jejuni and Salmonella enterica, compared with Escherichia coli, in seawater and river water. J. Water Health, 5 (3): 357-365.
- Smeets, P., Rietveld, L., Hijnen, W., Medema, G. and Stenstrom, T-A. (2006). Efficacy of water treatment processes. in: MicroRisk - microbiological risk assessment: A scientific basis for managing drinking water safety from source to tap. April 2006. (available from: www.kwrwater.nl).
- Smeets, P.W.M.H., Medema, G.J. and Van Dijk, J.C. (2009). The Dutch secret: How to provide safe drinking water without chlorine in the Netherlands. Drink. Water Eng. Sci., 2(1): 1–14.
- Sommer, R., Lhotsky, M., Haider, T. and Cabaj, A. (2000). UV inactivation, liquid-holding recovery, and photoreactivation of Escherichia coli O157 and other pathogenic Escherichia coli strains in water. J. Food Prot., 63(8): 1015-1020.
- Speight, V.L., Kalsbeek, W.D. and DiGiano, F.A. (2004). Randomized stratified sampling methodology for water quality in distribution systems. J. Water Resour. Plann. Manage., 130(4): 330-338.
- St Laurent, J. and Mazumder, A. (2012). The influence of land-use composition on fecal contamination of riverine source water in Southern British Columbia. Water Resour. Res., 48(12).
- Staben, N., Nahrstedt, A., Merkel, W. (2015). Securing safe drinking water supply under climate change conditions. Water Sci. Technol.-W. Sup., 15 (6): 1334-1342.
- Staley, Z.R., Grabuski, J., Sverko, E., Edge, T.A. (2016). Comparison of microbial and chemical source tracking markers to identify fecal contamination sources in the Humber River (Toronto, Ontario, Canada) and associated storm water outfalls. Appl. Environ. Microbiol., 82 (21):6357-6366.
- Standridge, J. (2008). E. coli as a public health indicator of drinking water quality. J. Am. Water Works Assoc., 100(2): 65-75.
- Statistics Canada (2013a). Households and the environment survey (HES) 2011.
- Stanfield, G., LeChevallier, M. and Snozzi, M. (2003). Chapter 5: Treatment Efficiency In: Assessing microbial safety of drinking water. Dufour, A. et al. eds. World Health Organization and The Organization for Economic Co-operation and Development. IWA Publishing, London, UK. pp. 159-178.
- Stea, E.C., Truelstrup Hansen, L., Jamieson, R.C. and Yost, C.K. (2015). Fecal contamination in the surface waters of a rural and an urban-source watershed. J. Environ. Qual., 44(5): 1556-1567.
- Szabo, J. and Minamyer, S. (2014). Decontamination of chemical agents from drinking water infrastructure: A literature review and summary. Environ. Int., 72: 119-123.
- Tenaillon, O., Skurnik, D., Picard, B. and Denamur, E. (2010). The population genetics of commensal Escherichia coli. Nat. Rev. Microbiol., 8(3): 207-217.
- Thomas, M.K., Charron, D.F., Waltner-Toews, D., Schuster, C., Maarouf, A.R. and Holt, J.D. (2006). A role of high impact weather events in waterborne disease outbreaks in Canada, 1975-2001. Int. J. Environ. Health Res., 16(3): 167-180.
- Tymensen, L.D., Pyrdok, F., Coles, D., Koning, W., Mcallister, T.A., Jokinen, C.C., Dowd, S.E. and Neumann, N.F. (2015). Comparative accessory gene fingerprinting of surface water Escherichia coli reveals genetically diverse naturalized population. J. Appl. Microbiol., 119(1): 263-277.
- U.S. EPA (1991). Guidance manual for compliance with the filtration and disinfection requirements for public water systems using surface water sources. U.S. Environmental Protection Agency, Washington, DC.
- U.S. EPA (1999). EPA guidance manual: Disinfection profiling and benchmarking. U.S. Environmental Protection Agency, Washington, DC. (EPA-815-R-99-013).
- U.S. EPA (2006a). 40 CFR parts 9, 141 and 142. National primary drinking water regulations: Ground water rule. final rule. U.S. Environmental Protection Agency, Washington, DC. Fed. Regist., 71(216): 65573–65660.
- U.S. EPA (2006b). National Primary Drinking Water Regulations: Long Term 2 Enhanced Surface Water Treatment; final rule. U.S. Environmental Protection Agency, Washington, DC. Fed. Regist., 71(3):678–671; 782–783.
- U.S. EPA (2006c). Ultraviolet disinfection guidance manual. Office of Water, U.S. Environmental Protection Agency, Washington, DC (Report No. EPA/815/R-06/007).
- U.S. EPA (2013). National primary drinking water standards: Revisions to the total coliform rule. final rule. U.S. Environmental Protection Agency, Washington, DC. Fed. Regist., 78(30): 10269.
- U.S. EPA (2017a). Analytical methods approved for compliance monitoring under the groundwater rule. Office of Water, U.S. Environmental Protection Agency, Washington, DC. EPA 821-F-17-004.
- U.S. EPA (2017b). Analytical methods approved for compliance monitoring under the revised total coliform rule. Office of Water, U.S. Environmental Protection Agency, Washington, DC. EPA 821-F-17-004.
- Valeo, C., He, J., Checkley, S. and Neumann, N. (2016). Rainfall and microbial contamination in Alberta well water. J. Environ. Eng. Sci., 11(1): 18-28.
- van der Kooij, D. and van der Wielen, P.W.J.J. (2014). Chapter 15 – Research needs. In: Microbial growth in drinking-water supplies: Problems, causes, control and research needs. IWA Publishing, London, UK. Pp. 423-443
- Van Dyke, M.I., Ong, C.S.L., Prystajecky, N.A., Isaac-Renton, J.L. and Huck, P.M. (2012). Identifying host sources, human health risk and indicators of Cryptosporidium and Giardia in a Canadian watershed influenced by urban and rural activities. J. Water Health, 10(2): 311-323.
- Van Elsas, J.D., Semenov, A.V., Costa, R. and Trevors, J.T. (2011). Survival of Escherichia coli in the environment: Fundamental and public health aspects. ISME J., 5(2): 173-183.
- van Lieverloo, J.H.M., Mesman, G.A.M., Bakker, G.L., Baggelaar, P.K., Hamed, A. and Medema, G. (2007). Probability of detecting and quantifying faecal contaminations of drinking water by periodically sampling for E. coli: A simulation model study. Water Res., 41(19): 4299-4308.
- Wahman, D.G. and Pressman, J.G. (2015). Distribution system residuals-is "detectable" still acceptable for chloramines? J. Am. Water Works Assoc., 107(8): 53-63.
- Walk, S.T., Alm, E.W., Calhoun, L.M., Mladonicky, J.M. and Whittam, T.S. (2007). Genetic diversity and population structure of Escherichia coli isolated from freshwater beaches. Environ. Microbiol., 9(9): 2274-2288.
- Wallender, E.K., Ailes, E.C., Yoder, J.S., Roberts, V.A., Brunkard, J.M. (2014). Contributing Factors to Disease Outbreaks Associated with Untreated Groundwater. Groundwater, 52 (6): 886-897.
- Whitman, R.L., Shively, D.A., Pawlik, H., Nevers, M.B. and Byappanahalli, M.N. (2003). Occurrence of Escherichia coli and enterococci in Cladophora (Chlorophyta) in nearshore water and beach sand of Lake Michigan. Appl. Environ. Microbiol., 69: 4714-4719.
- WHO (2004). Guidelines for drinking water quality. 3rd Edition. World Health Organization, Geneva, Switzerland.
- WHO (2005). Managing drinking-water quality from catchment to consumer. World Health Organization, Geneva, Switzerland.
- WHO (2012). Water safety planning for small community water supplies. World Health Organization, Geneva, Switzerland. Available at: www.who.int/water_sanitation_health/publications/2012/water_supplies/en/.
- WHO (2014). Water safety in distribution systems. WHO Document Production Services, Geneva, Switzerland.
- WHO (2017). Guidelines for drinking-water quality: fourth edition incorporating the first addendum. World Health Organization, Geneva.
- WHO and OECD (2003). Assessing Microbial Safety of Drinking Water – Improving approaches and methods. World Health Organization and Organisation for Economic Co-operation and Development. IWA Publishing, London UK. pp 1-295.
- Williams, M.M. and Braun-Howland, E.B. (2003). Growth of Escherichia coli in model distribution system biofilms exposed to hypochlorous acid or monochloramine. Appl. Environ. Microbiol., 69(9): 5463-5471.
- Winfield, M.D., Groisman, E.A. (2003). Role of nonhost environments in the lifestyles of Salmonella and Escherichia coli. Appl. Environ. Microbiol., 69 (7): 3687-3694.
- Wojcicka, L., Hofmann, R., Baxter, C., Andrews, R.C., Auvray, I., Lière, J., Miller, T., Chauret, C. and Baribeau, H. (2007). Inactivation of environmental and reference strains of heterotrophic bacteria and Escherichia coli O157:H7 by free chlorine and monochloramine. J. Water Supply Res. T., 56(2): 137-150.
- Wu, J., Long, S.C., Das, D. and Dorner, S.M. (2011). Are microbial indicators and pathogens correlated? A statistical analysis of 40 years of research. J. Water Health, 9(2): 265-278.
- Wu, J., Rees, P., Dorner, S. (2011).Variability of E. coli density and sources in an urban watershed. J. Water Health, 9: 94-106.
- Zhang, Y., Hong, P.-., LeChevallier, M.W. and Liu, W.-. (2015). Phenotypic and phylogenetic identification of coliform bacteria obtained using 12 coliform methods approved by the U.S. Environmental Protection Agency. Appl. Environ. Microbiol., 81(17): 6012-6023.
- Zhi, S., Banting, G., Li, Q., Edge, T.A., Topp, E., Sokurenko, M., Scott, C., Braithwaite, S., Ruecker, N.J., Yasui, Y., McAllister, T., Chui, L. and Neumann, N.F. (2016). Evidence of naturalized stress-tolerant strains of Escherichia coli in municipal wastewater treatment plants. Appl. Environ. Microbiol., 82(18): 5505-5518.
- Zimmer-Thomas, J.L., Slawson, R.M. and Huck, P.M. (2007). A comparison of DNA repair and survival of Escherichia coli O157:H7 following exposure to both low- and medium- pressure UV irradiation. J. Water Health, 5(3): 407-415.
Appendix A: Decision tree for routine microbiological testing of municipal-scale systems
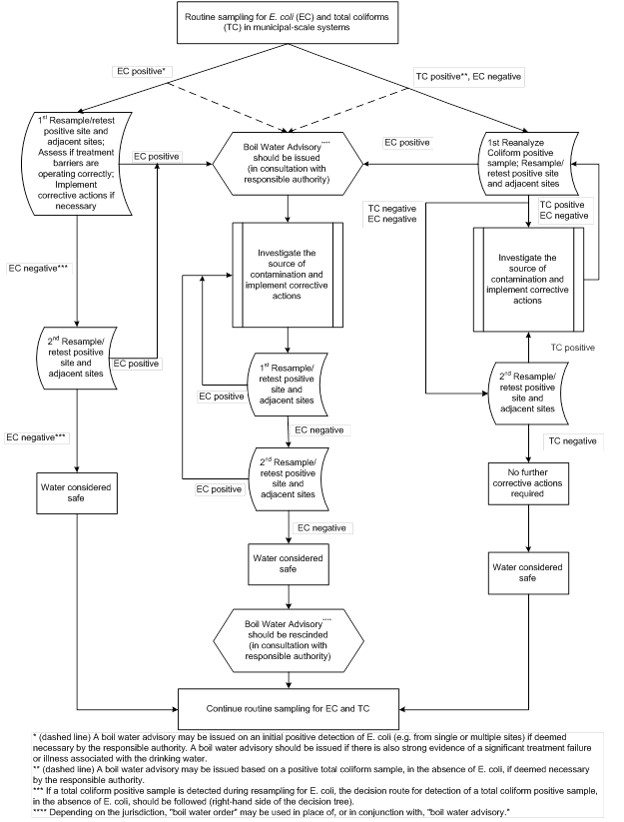
Text description
The recommended actions for responding to routine drinking water samples in public systems that are positive for E. coli and/or total coliforms are presented as a decision tree. The decision tree is made up of boxes that are connected to each other using single direction arrows. Each box in the decision tree contains a recommended action. The path taken through the decision tree, and therefore the recommended actions, are dependent on the results from the microbiological sampling. The left hand side of the decision tree presents the actions to be taken when samples are positive for E. coli. The right hand side of the decision tree presents the actions to be taken when samples are positive for total coliforms but negative for E. coli. The middle of the decision tree presents the actions to be taken if a boil water advisory has been issued.
On the left hand side of the decision tree, if E. coli is detected during routine sampling, the recommended action is to issue a boil water advisory in consultation with the responsible authorities. If only a single site is positive for E. coli, the recommended action is the positive site and adjacent sites should undergo a first re-sampling for E. coli and total coliforms (*). If any re-sampled site is positive for E. coli, the recommended action is to issue a boil water advisory in consultation with the responsible authorities. Depending on the jurisdiction, a boil water advisory may be issued on a single site contamination. If the re-sampled site was negative for E. coli and total coliforms, the recommended action is to carryout a second re-sampling at the positive site and at the adjacent sites. If any of the second re-sampled sites are positive for E. coli, the recommended action is to issue a boil water advisory in consultation with the responsible authorities. If the second re-sampled sites are negative for E. coli and total coliforms, the water is considered safe and the recommended action is to continue routine sampling.
The right hand side of the decision tree should be followed if a total coliform sample is positive in the absence of E. coli. Therefore, if total coliforms are detected during the first or second re-sampling for E. coli, or, if a routine sample is positive for total coliforms, in the absence of E. coli, the recommended action is to re-sample the positive site and adjacent sites for E. coli and total coliforms (**). If E. coli is detected, the recommended action is to issue a boil water advisory in consultation with the responsible authorities. A boil water advisory may be issued based on a positive total coliform, in the absence of E. coli, if deemed necessary by the responsible authority. If the re-sampled sites are negative for both total coliforms and E. coli, the recommended action is the positive sites and the adjacent sites should undergo a second re-sampling for E. coli and total coliforms. If these sites are negative for E. coli and total coliforms, then no further action is required, the water is considered safe and the recommended action is to continue with routine sampling. If total coliforms are detected in any of the re-sampled sites, in the absence of E. coli, the recommended action is to investigate the source of the contamination, implement corrective actions, and then return to the top of the decision tree, to the box recommending the first re-sampling of the positive sites and adjacent sites (***).
In all instances where the decision route leads to issuing a boil water advisory, the centre of the decision tree should be followed (****). After issuing the boil water advisory, the recommended actions are to investigate the source of the contamination, implement corrective actions and undergo a first re-sampling of the positive sites and adjacent sites. If E. coli is detected, the arrow directs the decision route back to investigating and implementing corrective actions. If the first re-sampling is negative for E. coli, a second re-sampling at the positive sites and adjacent sites is recommended. If the second re-sampling is positive for E. coli, the decision route is directed back to investigating and implementing corrective actions. If the second re-sampling is negative for E. coli, the water is considered safe and the recommended action is to rescind the boil water advisory in consultation with the responsible authorities, and to then continue with routine sampling.
Appendix B: Decision tree for routine microbiological testing of residential-scale systems
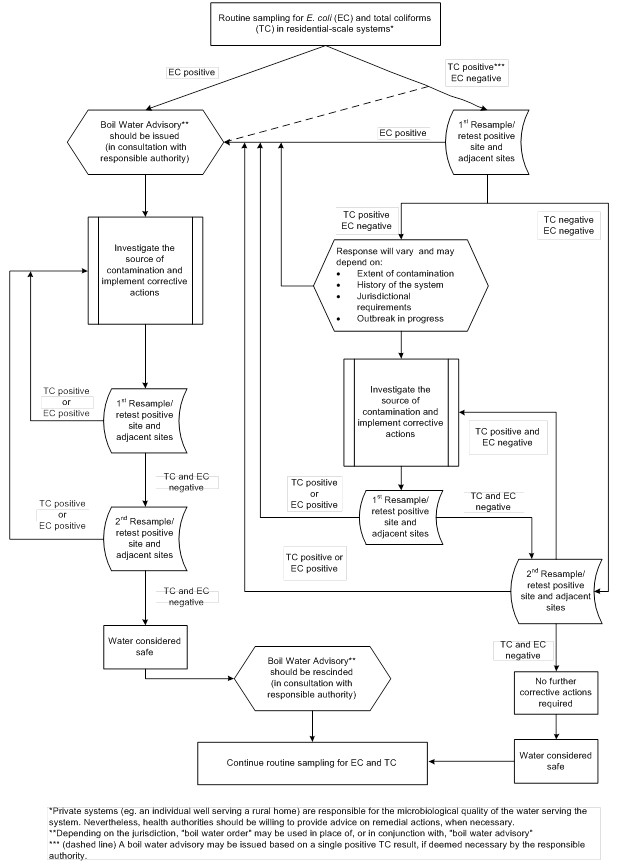
Text description
The recommended actions for responding to routine drinking water samples in semi-public systems that are positive for E. coli and/or total coliforms are presented as a decision tree (*). The decision tree is made up of boxes that are connected to each other using single direction arrows. Each box in the decision tree contains a recommended action. The path taken through the decision tree, and therefore the recommended actions that should be taken, are dependent on the results from the microbiological sampling. The left hand side of the decision tree presents the actions to be taken when samples are positive for E. coli and a boil water advisory has been issued (**). The right hand side of the decision tree presents the actions to be taken when samples are positive for total coliforms but negative for E. coli.
On the left hand side of the decision tree, if E. coli is detected during routine sampling the recommended action is to issue a boil water advisory in consultation with the responsible authorities.
The right hand side of the decision tree should be followed if a total coliform sample is positive in the absence of E. coli. For this decision route, the recommended action is to undergo a first re-sampling of the positive sites and at adjacent sites for E. coli and total coliforms (***). If E. coli is detected, the recommended action is to issue a boil water advisory in consultation with the responsible authorities. A boil water advisory may also be issued based on a positive total coliform result in the absence of E. coli, if deemed necessary by the responsible authority. If the first re-sampled sites are negative for both total coliforms and E. coli, the recommended action is the positive sites and the adjacent sites should undergo a second re-sampling for E. coli and total coliforms. If E. coli is detected in the second re-sampling, the recommended action is to issue a boil water advisory in consultation with the responsible authorities. If the second re-sampling sites are negative for E. coli and total coliforms, then no further corrective actions are required, the water is considered safe and the recommended action is to continue with routine sampling. If total coliforms are detected in any of the re-sampled sites, in the absence of E. coli, the recommended action depends on the extent of the contamination, the history of the system, the jurisdictional requirements and if there is an outbreak in progress (***). The recommendation can be to either investigate the source of the contamination, implement corrective actions, and carryout a first re-sampling of the positive sites and adjacent sites, or to issue a boil water advisory in consultation with the responsible authority. If the decision route taken was to carryout the first re-sampling, and the sites are negative for both total coliforms and E. coli, the recommended action is to undergo a second re-sampling for E. coli and total coliforms. If these sites are negative for E. coli and total coliforms, than the water is considered safe and the recommended action is to continue with routine sampling. If either the first or the second re-sampling results in a total coliform or an E. coli positive sample, the recommended action is to issue a boil water advisory in consultation with the responsible authority.
In all instances where the decision route leads to issuing a boil water advisory, the recommended action is to then investigate the source of the contamination, implement corrective actions and undergo a first re-sampling of the positive sites and adjacent sites. If E. coli or total coliforms are detected, the arrow directs the decision route back to investigating and implementing corrective actions. If the first re-sampling is negative for E. coli and total coliforms, a second re-sampling at the positive sites and adjacent sites is recommended. If the second re-sampling is positive for E. coli or total coliforms, the decision route is directed back to investigating and implementing corrective actions. If the second re-sampling is negative for E. coli and total coliforms, the water is considered safe and the recommended action is to rescind the boil water advisory in consultation with the responsible authorities, and to then continue with routine sampling.
Appendix C: List of acronyms
- ANSI
- American National Standards Institute
- CFU
- colony-forming unit
- CNPHI
- Canadian Network for Public Health Intelligence
- CT
- concentration of disinfectant × contact time
- E. coli
- Escherichia coli
- EU
- European Union
- GUDI
- groundwater under the direct influence of surface water
- MAC
- maximum acceptable concentration
- MCL
- maximum contaminant level (U.S.)
- MCLG
- maximum contaminant level goal (U.S.)
- NSF
- NSF International
- P-A
- presence–absence
- PCR
- polymerase chain reaction
- POE
- point-of-entry
- POU
- point-of-use
- RO
- reverse osmosis
- SCC
- Standards Council of Canada
- U.S. EPA
- United States Environmental Protection Agency
- UV
- ultraviolet
- VBNC
- viable but non-culturable
- WHO
- World Health Organization
- Footnote 1
-
For the purposes of this document, a residential-scale water supply system is defined as a system with a minimal or no distribution system that provides water to the public from a facility not connected to a municipal supply. Examples of such facilities include private drinking water supplies, schools, personal care homes, day care centres, hospitals, community wells, hotels, and restaurants. The definition of a residential-scale supply may vary between jurisdictions.
- Footnote 2
-
For the purpose of this document, the use of the term “boil water advisory” is taken to mean advice given to the public by the responsible authority in the affected jurisdiction to boil their water, regardless of whether this advice is precautionary or in response to an outbreak. Depending on the jurisdiction, the use of this term may vary. As well, the term “boil water order” may be used in place of, or in conjunction with, a “boil water advisory.”