Research Ethics Board (REB) Annual Report 2020-21
Table of contents
- Executive summary
- Message from the Chair
- About the Health Canada-PHAC REB
- REB meetings and reviews
- Profile of applications received in 2020-21
- Application review process and outcomes
- Secretariat activities
- Membership
- References
Executive summary
The Health Canada (HC) and Public Health Agency of Canada (PHAC) Research Ethics Board (REB) is a joint board for both the Department and the Agency.
The REB provides support and independent ethics review for all proposed and ongoing research involving humans carried out under the auspices of Health Canada and PHAC, to help ensure that the highest ethical standards are being met and the greatest protection is provided to participants.
The 2020-21 fiscal year was defined by the COVID-19 pandemic, which impacted the REB's work in a number of ways. First, there were no in-person REB meetings in 2020-21; instead, all meetings (12 full board and 58 delegated review) were held by teleconference or videoconference. Second, many applications were considered urgent and required that the REB be available on short notice to conduct expedited reviews. This resulted in a significant number of ad hoc meetings outside of the regularly-scheduled monthly full board meetings and weekly delegated review meetings. And finally, the REB saw an increase in amendment requests as many researchers had to modify their protocols in response to public health restrictions and laboratory closures.
The REB received 44 new applications in 2020-21, of which 19 were from Health Canada and 25 from PHAC. Reflecting the impact of the COVID-19 pandemic on research, 19 applications were related to COVID-19 and 11 applications were from the PHAC National Microbiology Laboratory. The majority of initial applications (34) were deemed to be minimal risk and therefore reviewed at delegated review meetings, which represented a significant change from the previous year in which only 13 out of 43 applications were considered minimal risk. The time from application submission to approval was an average of 32 calendar days for non-expedited protocols (21 days with the REB and 11 days with the applicant), while for expedited reviews the average was only 15 days (6 days with the REB and 9 days with the applicant). The REB also reviewed 112 annual progress reports, 73 amendment requests and 2 adverse event reports in 2020-21, and 25 protocols were closed.
In addition to conducting ethics reviews, the REB received a number of education and information sessions in 2020-21. While there were fewer sessions than in previous years because of the pandemic, the shift to videoconference meetings increased participation by making the sessions more easily accessible to the entire membership. In terms of membership, the REB said farewell to four long-time members whose terms ended in 2020-21 and welcomed six new members to the board.
The REB Secretariat continued its outreach efforts among Health Canada and PHAC staff, including training sessions, targeted consultations and participation in the Health Canada Science Forum. The Secretariat implemented additional operational improvements in 2020-21, including a redesigned REB website, new fillable PDF forms for continuing ethics review, and a Microsoft Access database to better track protocols and workflow. While the COVID-19 pandemic clearly created significant challenges, the REB and the Secretariat were able to quickly adapt and successfully deliver on their mandate. The learnings and operational improvements from 2020-21 have ensured that the REB is well-positioned for the coming year.
Message from the Chair

On behalf of the Health Canada (HC) and Public Health Agency of Canada (PHAC) Research Ethics Board (REB), I am pleased to present this report on our REB's activities for the 2020-21 fiscal year.
This report supports the Department's and Agency's commitment to transparency and accountability, with respect to the REB's work.
The REB helps ensure that human participants in research receive the protections they deserve, and that decision-makers have access to ethically sound research when developing policies and regulations. The REB's work in this regard was particularly important over the past year due to the challenges associated with the COVID-19 global pandemic.
It goes without saying that 2020-21 was an exceptional year as Health Canada and PHAC scientists and researchers, and Canadians nationwide, stepped up to support Canada's efforts to curb the pandemic.
The National Microbiology Laboratory played a key role, reorienting its considerable resources and expertise, almost overnight, to focus on pandemic response.
At the same time, staff carried out other essential research to deliver on Department and Agency mandates and to help inform policy and regulatory decisions, all while dealing with limited access to labs and offices, remote working and other disruptions.
The urgency created by the pandemic meant many research protocols required expedited ethics reviews and the REB responded in kind.
I would like to express my gratitude to our REB members for delivering on short notice, adapting quickly to working virtually, and providing expert reviews of urgent studies in the pandemic context. In addition, the REB Secretariat provided exceptional support to the Board in administering the ethics review process.
This year, we also welcomed new members of the REB, and said farewell to valued members whose terms ended. I am grateful to the past and current members who have ensured we have maintained our momentum.
On a personal note, I was honoured that my term as Chair was extended for an additional year, to provide continuity during the pandemic. It is a great privilege to serve in this capacity.
During my time at the REB, it is clear that Health Canada and PHAC are committed to the highest standards of research ethics. This is especially evident through ongoing Department and Agency support, and the REB's wide-ranging collaborative relationships that continue to expand and flourish.
Thank you to past and new members, and our partners for your commitment to supporting and delivering on the REB's mandate.
Barbara McGillivray, MD, FRCP, FCCMG
Chair, Health Canada-PHAC Research Ethics Board
About the Health Canada-PHAC REB
The Health Canada (HC) and Public Health Agency of Canada (PHAC) Research Ethics Board (REB) provides support and independent ethics review for all proposed and ongoing research involving humans conducted under the auspices of the Department or Agency, to ensure the highest ethical standards are being met, and that the greatest protection is provided to participants. The REB reviews applications in accordance with the considerations set out in the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (TCPS 2) as the minimum standard. The REB is also guided by relevant federal laws and regulations, such as the Privacy Act and clinical trial regulations, where applicable.
The REB reports to the Deputy Minister of Health Canada and the President of PHAC, who jointly appoint REB members, approve REB procedures and authorize research to be initiated or terminated. The REB makes recommendations to the Deputy Minister and President (through their delegated Decisional Authorities) as to whether research projects should be approved, rejected, modified or terminated. The REB also provides ethical oversight of all approved research throughout the duration of the project. Finally, the REB serves various educational functions for Department and Agency managers and researchers.
The REB is responsible for the ethics review of all research involving humans that is:
- Carried out by Health Canada or PHAC;
- Performed by the Department or Agency in collaboration with external researchers;
- Carried out on Department or Agency premises; or
- Conducted under contract to the Department or Agency.
The REB may also review research involving humans that is funded through the Department or Agency grants and contributions.
The REB is supported by a Secretariat that manages its operations and administers the REB review process. The Secretariat works closely with REB members and applicants to ensure a quality and timely review of all research proposals received for ethics review. The Secretariat is based at Health Canada within the Strategic Policy Branch (Science Policy Directorate).
In the case of applications from PHAC researchers, the REB Secretariat collaborates with the PHAC Office of the Chief Science Officer which administers its approval process and provides additional advice to applicants. The REB Secretariat and the PHAC Office of the Chief Science Officer also work closely together on policies, procedures and guidance related to the REB.
REB meetings and reviews
The full Research Ethics Board (REB) typically meets monthly, either remotely or face-to-face in Ottawa. A minimum of five members is required for quorum (one member each with expertise in ethics and in law, one community member, and two researcher members). All REB members also attend delegated review meetings on a rotating basis. Delegated review meetings are held weekly by videoconference (bi-weekly in July and August) and are attended by the Chair (or Deputy Chair) and one other REB member.
Applications for initial review of research involving humans are generally reviewed at the monthly meetings of the full REB, while delegated review meetings typically evaluate submissions for continuing ethics review (i.e., annual progress reports, amendment requests, adverse event reports and completion/termination reports). Initial applications that represent minimal risk to participants may also be sent to delegated review. For the review of initial applications, the applicant is invited to the REB meeting to give a brief presentation of their proposal and to answer any questions from the REB members.
Types and number of REB meetings in 2020-21:
- 12 full board meetings:
- 7 regular monthly meetings (includes an extra full board meeting in December to handle the volume of submissions)
- 5 ad hoc meetings, 3 of which were for expedited review of urgent COVID-19 studies
- all meetings held by teleconference or videoconference due to the COVID-19 pandemic
- 58 delegated review meetings:
- 40 regular weekly meetings
- 18 ad hoc meetings, 16 of which were to review minimal risk initial applications (many which required expedited review)
The number of open protocols varies throughout the year as the REB approves new applications and closes old protocols. There were 153 open protocols on March 31, 2021, and 25 protocols were closed in 2020-21.
Figure 1. Submissions reviewed by the REB in 2020-21Footnote 1
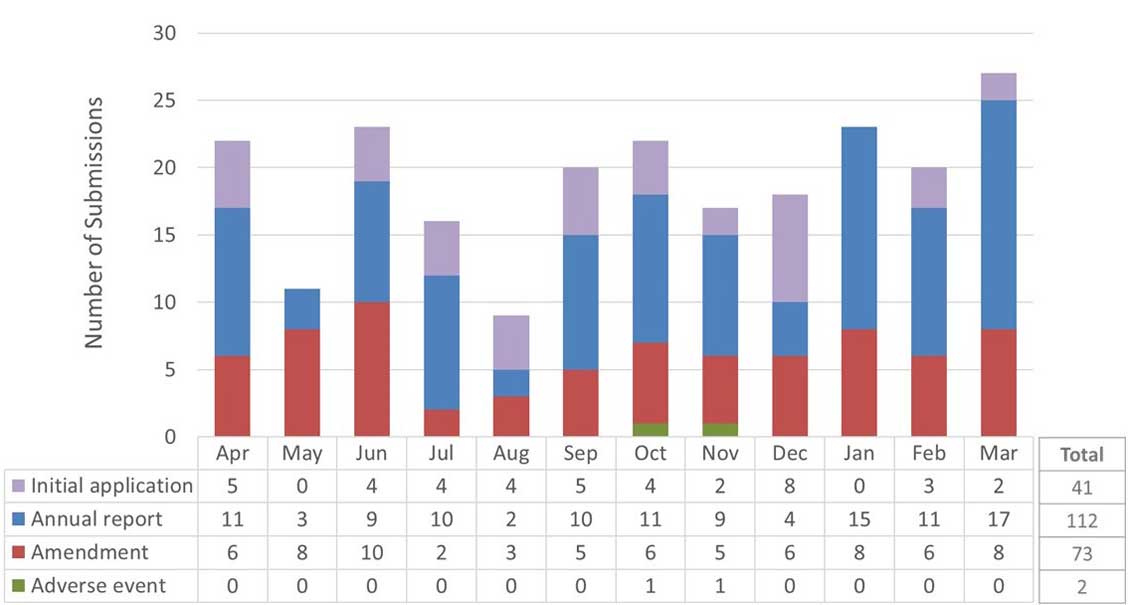
Figure 1: Text description
This figure is a bar chart with 12 bars showing the number of submissions reviewed by the REB in the 2020-2021 fiscal year. Each bar corresponds to a month of the year, starting with April and ending with March. Each bar is further subdivided into 4 sections corresponding to the type of submission that was reviewed (initial application, annual progress report, amendment request and adverse event report). The data are as follows:
| Month | Initial applications | Annual progress reports | Amendments | Adverse events |
|---|---|---|---|---|
| April | 5 | 11 | 6 | 0 |
| May | 0 | 3 | 8 | 0 |
| June | 4 | 9 | 10 | 0 |
| July | 4 | 10 | 2 | 0 |
| August | 4 | 2 | 3 | 0 |
| September | 5 | 10 | 5 | 0 |
| October | 4 | 11 | 6 | 1 |
| November | 2 | 9 | 5 | 1 |
| December | 8 | 4 | 6 | 0 |
| January | 0 | 15 | 8 | 0 |
| February | 3 | 11 | 6 | 0 |
| March | 2 | 17 | 8 | 0 |
| Total | 41 | 112 | 73 | 2 |
Profile of applications received in 2020-21
The Research Ethic Board (REB) received 44 applications in 2020-21, 19 from Health Canada (HC) and 25 from the Public Health Agency of Canada (PHAC). By comparison, 43 applications were received in 2019-20, and 31 in 2018-19. External protocols were led by researchers outside of the Department and Agency, either in collaboration with Health Canada or PHAC researchers or funded by the Department or Agency.
Figure 2. Affiliation of the principal investigator for REB applications
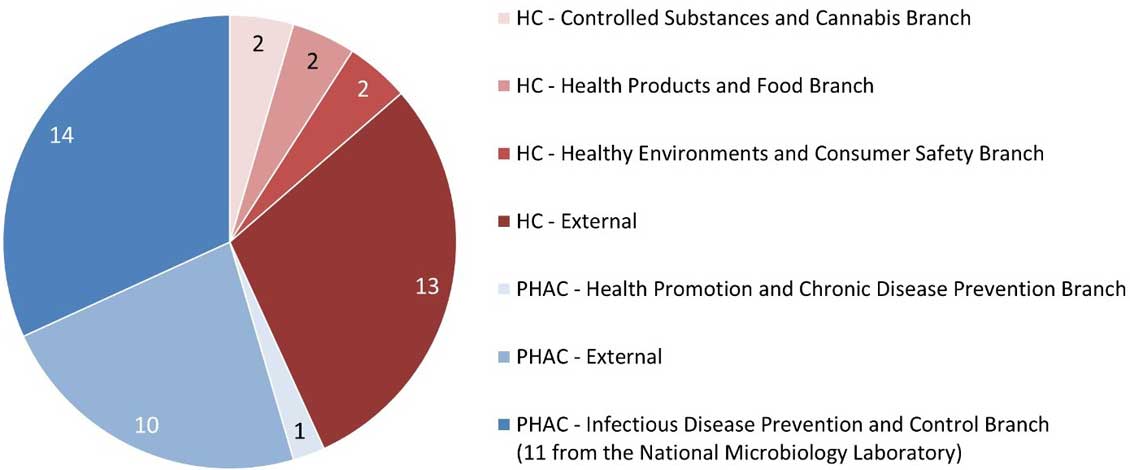
Figure 2: Text description
This pie chart illustrates the affiliation of the lead investigator for the REB applications received during the 2020-2021 fiscal year. The data are as follows:
- Among the 19 applications received from Health Canada:
- 2 were from the Controlled Substances and Cannabis branch
- 2 were from the Health Products and Food Branch
- 2 were from the Healthy Environments and Consumer Safety Branch
- 13 were external.
- Among the 25 applications received from PHAC:
- 1 was from the Health Promotion and Chronic Disease Prevention Branch
- 10 were external
- 14 were from the Infectious Disease Prevention and Control Branch (11 of which were from the National Microbiology Laboratory).
The protocols received spanned a broad range of research areas. One study focused on Indigenous populations, and 19 were related to COVID-19.
Figure 3. Subject areas of the protocols received by the REB
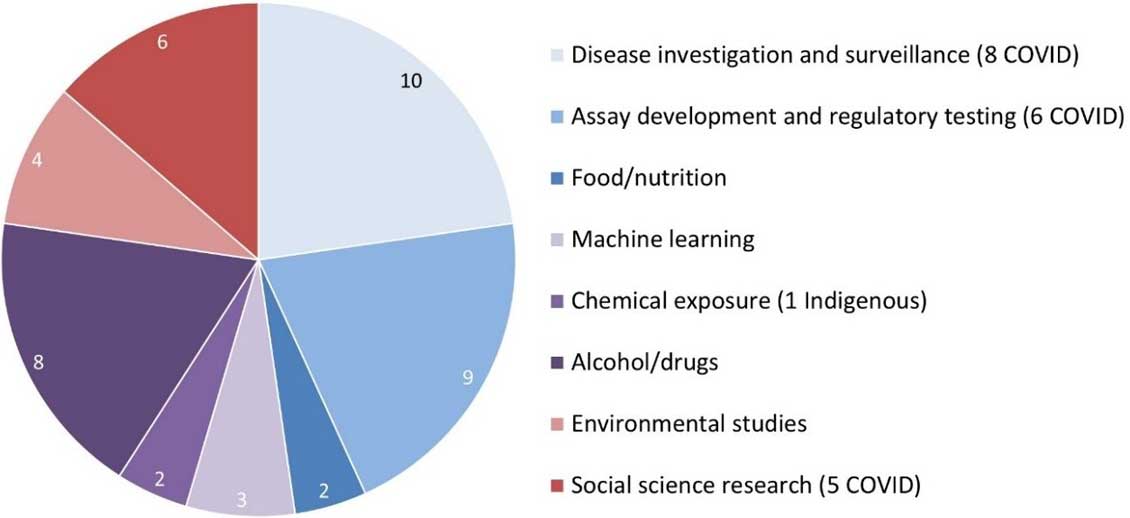
Figure 3: Text description
This pie chart illustrates the subject areas of the protocols received by the REB for the 2020-2021 fiscal year. The data are as follows:
- 10 were about disease investigation and surveillance (8 of which were related to COVID)
- 9 were about assay development and regulatory testing (6 of which were related to COVID)
- 2 were about food and nutrition
- 3 were about machine learning
- 2 were about chemical exposure (1 of which was focused on Indigenous populations)
- 8 were about alcohol and drugs
- 4 were environmental studies
- 6 were social science research (5 of which were about COVID)
Application review process and outcomes
The Research Ethics Board (REB) applies a proportionate approach to research ethics review; that is, the level of risk to participants determines the level of scrutiny by the REB. Thus, at the discretion of the REB chair, applications deemed to be minimal risk can be sent to delegated review (and if the risk is determined to be greater than initially thought, subsequently referred to the full board for further review).
Following the review of an application, the REB makes one of the following recommendations:
- A: Approved as submitted: The application meets ethics requirements for research involving humans and is recommended for approval.
- B: Approved with modifications/clarifications: The application meets ethics requirements for research involving humans, but the REB members require modification of the application or clarification or further information to secure approval. The revised material may be reviewed via delegated review or a subset of the members as directed by the REB Chair.
- C: Deferral: The application does not meet ethics requirements for research involving humans and is not approved as conditions need to be met. The revised material must be reviewed by the full board via teleconference or in person, or by email circulation to all REB members.
- D: Disapproval: The application fails to meet the ethical standards for approval and revision is unlikely to enable the REB to reach a positive determination.
Figure 4. Level of review for initial applications
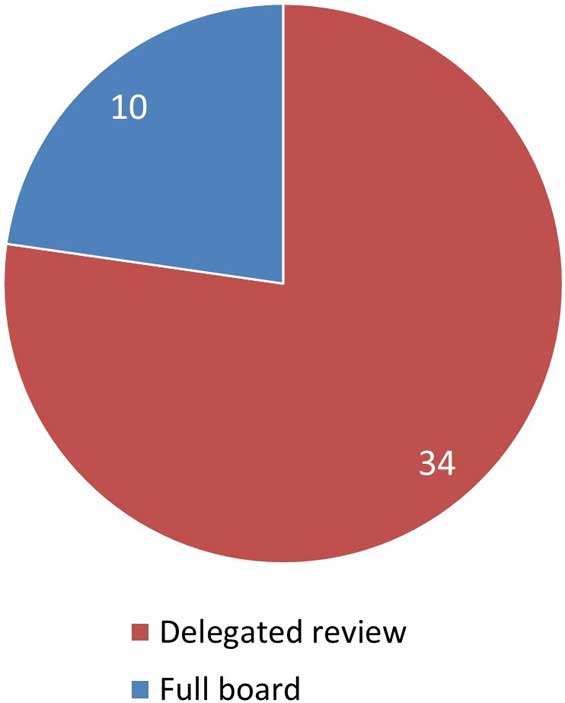
Figure 4: Text description
This pie chart illustrates the level of review that initial applications received by the REB. 34 applications were sent to delegated review and 10 applications received full board review.
Figure 5. Outcome of REB review for initial applications
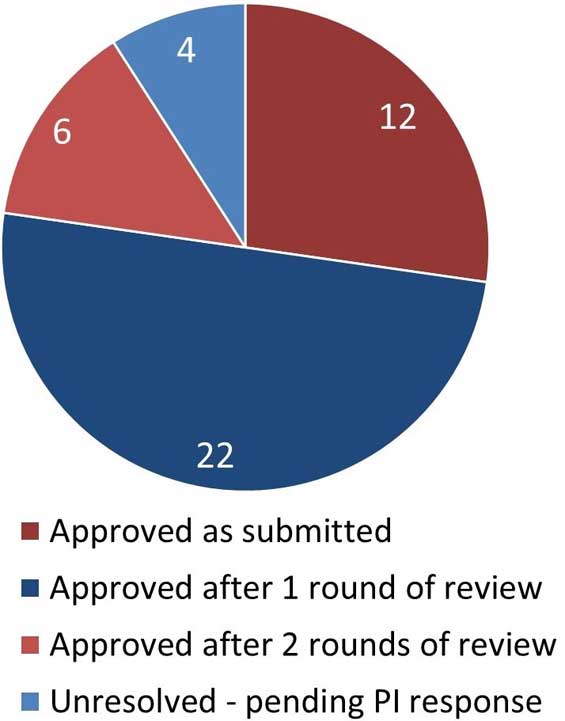
Figure 5: Text description
This pie chart illustrates the outcome of the REB review for initial applications. The data are as follows:
- 12 applications were approved as submitted
- 22 applications were approved after 1 round of review
- 6 applications were approved after 2 rounds of review
- 4 applications were unresolved, pending a response from the Principal Investigator
Approval times: The time from application submission to approval is a function of the REB Secretariat, the REB and the applicant. The time taken by the applicant to respond to the REB's recommendations and other requests from the Secretariat is beyond the REB's control; thus, the approval time is broken down into days with the REB and days with the applicant. In terms of time with the REB:
- The application deadline is 3 weeks before each monthly REB meeting. If an application is deemed to represent minimal risk to participants and is sent to delegated review rather than the full board, the time between submission and review is often reduced.
- The REB Secretariat's service standard is to communicate the REB's recommendations to the applicant within one week of the REB meeting. In fact, in many cases this was done within 24 hours.
- The REB will conduct expedited reviews for time-sensitive research projects where the urgency is due to public health emergencies or other circumstances beyond the researchers' control. In 2020-21, the REB conducted a record 15 expedited reviews, the vast majority of which were for COVID-19 related studies.
| Non-expedited (n = 25) | Expedited (n = 15) | |||
|---|---|---|---|---|
| Average (± SD) | Median | Average (± SD) | Median | |
| Days with REB | 21 (± 11) | 22 | 6 (± 5) | 5 |
| Days with applicant | 11 (± 17) | 5 | 9 (± 7) | 8 |
| Total | 32 (± 22) | 26 | 15 (± 10) | 13 |
Figure 6. Time distribution for each of the 40 applications that were approved by March 31, 2021
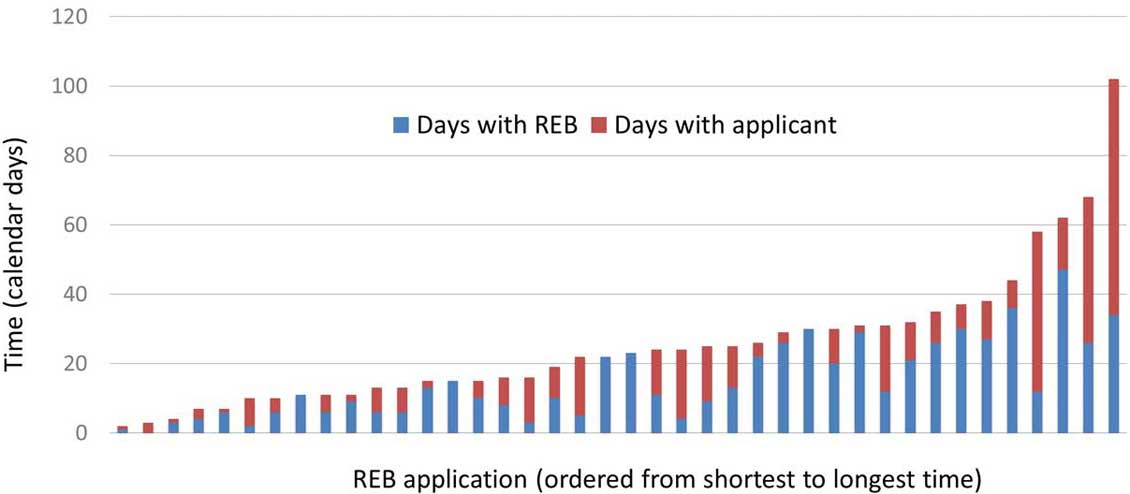
Figure 6: Text description
This bar graph contains 40 bars, 1 bar for each application that was approved by March 31, 2021. The bars are further divided into 2 sections: number of days with the REB and number of days with the applicant. The 40 bars are ordered from the shortest to longest amount of time by calendar days.
| Application number | Days with REB | Days with applicant | Total |
|---|---|---|---|
| 1 | 1 | 1 | 2 |
| 2 | 0 | 3 | 3 |
| 3 | 3 | 1 | 4 |
| 4 | 4 | 3 | 7 |
| 5 | 6 | 1 | 7 |
| 6 | 2 | 8 | 10 |
| 7 | 6 | 4 | 10 |
| 8 | 11 | 0 | 11 |
| 9 | 6 | 5 | 11 |
| 10 | 9 | 2 | 11 |
| 11 | 6 | 7 | 13 |
| 12 | 6 | 7 | 13 |
| 13 | 13 | 2 | 15 |
| 14 | 15 | 0 | 15 |
| 15 | 10 | 5 | 15 |
| 16 | 8 | 8 | 16 |
| 17 | 3 | 13 | 16 |
| 18 | 10 | 9 | 19 |
| 19 | 5 | 17 | 22 |
| 20 | 22 | 0 | 22 |
| 21 | 23 | 0 | 23 |
| 22 | 11 | 13 | 24 |
| 23 | 4 | 20 | 24 |
| 24 | 9 | 16 | 25 |
| 25 | 13 | 12 | 25 |
| 26 | 22 | 4 | 26 |
| 27 | 26 | 3 | 29 |
| 28 | 30 | 0 | 30 |
| 29 | 20 | 10 | 30 |
| 30 | 29 | 2 | 31 |
| 31 | 12 | 19 | 31 |
| 32 | 21 | 11 | 32 |
| 33 | 26 | 9 | 35 |
| 34 | 30 | 7 | 37 |
| 35 | 27 | 11 | 38 |
| 36 | 36 | 8 | 44 |
| 37 | 12 | 46 | 58 |
| 38 | 47 | 15 | 62 |
| 39 | 26 | 42 | 68 |
| 40 | 34 | 68 | 102 |
REB education and information sessions
As noted in TCPS 2, an important component of Research Ethics Board (REB) meetings is the inclusion of educational opportunities that may benefit the overall operation of the REB. To this end, a broad range of educational sessions were organized over the course of the year, as described below. The list also includes presentations from researchers about their programs of research. These interactions served not only to educate Board members about emerging research areas, but also to strengthen the relationships between the REB and researchers. The REB would like to thank all the presenters for generously giving their time to enlighten the Board.
- Overview of National Microbiology Laboratory (NML) COVID-19 activities – Dr. Guillaume Poliquin, National Microbiology Laboratory (June 2020)
- The Canadian Health Measures Survey – Ron Gravel, Statistics Canada (June 2020)
- Research overview: Effects of cannabis and alcohol use on health and behaviour – Dr. Bruna Brands, Healthy Environments and Consumer Safety Branch (June 2020)
- Proposed exemption from REB review for research involving cell lines – Wendy Burgess, Secretariat on Responsible Conduct of Research, and Dr. Lehana Thabane, REB member (November 2020)
- TCPS 2: 2018 updates, Q&A – Wendy Burgess, Secretariat on Responsible Conduct of Research (November 2020)
- Re-identification Risk in Health Research – Dr. Khaled El Emam, Electronic Health Information Laboratory, University of Ottawa (February 2021)
- Privacy, Ethics, Data & Artificial Intelligence (AI) – Madelaine Saginur and Andréa Rousseau, Health Canada-PHAC Privacy Management Division (February 2021).
Secretariat activities
The Research Ethics Board (REB) Secretariat administers the REB review process and supports the Board in fulfilling its mandate. Important elements of the Secretariat's work are to build partnerships and increase awareness about the REB, and to continually improve the Board's processes. Key Secretariat activities for 2020-21 included the following:
Outreach
- The REB Secretariat provided an overview of the REB process to staff from Health Canada's Substance Use and Addictions Program, with an emphasis on the requirements associated with research funded through grants and contributions. Plans for other outreach activities, including a return to the National Microbiology Laboratory, were unfortunately placed on hold due to the pandemic.
- The 2019-20 REB Annual Report was presented at Health Canada's DG Science committee and shared broadly with executive management at both Health Canada and the Public Health Agency of Canada (PHAC). The report was also published on the REB website.
- Dr. Barbara McGillivray (REB Chair) participated in a panel presentation and discussion on Indigenous research considerations at the annual Health Canada Science Forum in January, 2021.
- Dr. McGillivray and the REB Secretariat provided advice to Health Canada's Office of Clinical Trials regarding the Ministerial Interim Order respecting clinical trials for medical devices and drugs relating to COVID-19.
- The REB Secretariat worked with other federal departments (in particular, the First Nations and Inuit Health Branch of Indigenous Services Canada) and other REBs (such as the Community Research Ethics Office in Waterloo, ON) to discuss research ethics issues, explore avenues for collaboration and share best practices.
- Dr. McGillivray and the REB Secretariat began a dialogue with the Nunavut Research Institute (NRI) to explore opportunities to better support the NRI's research licensing process. Discussions also focused on the unique ethical considerations associated with research in the North and how to better support researchers at Health Canada, PHAC and beyond in responding to these issues.
Operations
- The REB website redesign was completed, which included reorganizing the information into a simplified structure, correcting outdated information and publishing additional guidance on issues like the consent process.
- New fillable PDF forms and instructions were created for continuing ethics review (annual progress report, amendment request, adverse event report and completion/termination report), as well as for scientific peer review.
- The Microsoft Access database was fully implemented to support better tracking of REB protocols and Secretariat workflow, leading to increased efficiency and accuracy in the Secretariat's work.
- The REB Secretariat worked closely with PHAC's Office of the Chief Science Officer and the Health Canada-PHAC Privacy Management Division to further strengthen existing processes for information sharing and coordination. These efforts were particularly important because of the COVID-19 pandemic and the need for expedited reviews of urgent studies.
Membership
The REB consists of nine regular and nine alternate members with expertise in the following areas:
- Two members with knowledge/expertise in research ethics;
- One member with knowledge/expertise in law;
- One member from Health Canada with knowledge/expertise in Health Canada research;
- One member from the Public Health Agency of Canada (PHAC) with knowledge/expertise in PHAC research;
- One member external to Health Canada and PHAC with broad knowledge/expertise in both Health Canada and PHAC research;
- One member with broad expertise in public health;
- One member recruited from the community (general population) served by Health Canada and PHAC; and
- One member from the Indigenous community.
Members are appointed by the Deputy Minister of Health and the President of PHAC for a term of three years, renewable once. The terms for four long-time REB members ended in 2020-21:
- Carla Moore, M.A. – Community Member, Indigenous Population
- Hanan Abramovici, Ph.D. – Health Canada Researcher
- Ghislaine Cleret de Langavant, Ph.D. – Ethics
- Aïssata Sako, B.A. – Community Member, General Population
Health Canada and PHAC are especially grateful for their expert contributions to the REB over the years, and they are very much missed by the REB members and Secretariat.
The REB welcomed the following individuals who joined the REB in 2020-21 to fill various vacancies:
- Kathleen Makela, B.A., LL.B. – Community Member, Indigenous Population (alternate): Kathleen Makela is a Métis born in Alberta and now living in Saskatoon. She is a descent of Old Man Beaulieu from Fort Resolution (Deninu), NWT, on her mother's side, and Finnish on her father's side (he is a first generation Canadian). She is a program manager with The Gordon Foundation and worked previously at University of Saskatchewan's Native Law Centre of Canada and Aboriginal Students' Centre.
- Glenn G. Griener, Ph.D. – Ethics (regular): Dr. Glenn Griener is a philosopher whose primary areas of scholarly activity are applied ethics and the philosophy of science, with a particular focus on the issues arising in health care and the ethics of biomedical research. He has also participated in or led several bodies developing national policy on research ethics. He retired from the Department of Philosophy at the University of Alberta in 2019.
- Janaki Jayanthan, MPH – Community Member, General Population (alternate): Janaki Jayanthan is a Senior Primary Care Program Developer (Vancouver Island) with the Primary Care Division of the British Columbia Ministry of Health. In her current role she supports the implementation of Urgent and Primary Care Centres and Community Health Centres as part of provincial efforts towards team-based primary and community care. She has contributed to public health research and community-based projects in Canada and abroad and has a long-standing personal and professional interest in research ethics.
- Stéphane P. Ahern, M.D., Ph.D. – Ethics (alternate): Dr. Stéphane Ahern is an Associate Professor of Medicine at the Université de Montréal in the Faculty of Medicine. He is a specialist in general internal medicine and adult critical care. Since 2007, he has served as Chair or Vice-Chair of the Research Ethics Board (REB) at Maisonneuve-Rosemont Hospital and now at CIUSSS de l'Est-de-l'Île-de-Montréal – REBs operating in fields at the forefront of stem cell transplantation, psychiatry, advanced ophthalmologic therapy and oncology, among others.
- Lehana Thabane, Ph.D. – Researcher External to Health Canada and the Public Health Agency of Canada (alternate): Dr. Lehana Thabane is a Professor of Biostatistics in the Department of Health Research Methods, Evidence and Impact, and Associate member of the Departments of Pediatrics and Anesthesia, School of Nursing, and School of Rehabilitation Science, in the Faculty of Health Sciences at McMaster University (Hamilton, ON). He is also the Director of Biostatistics at St Joseph's Healthcare Hamilton. His research interests include clinical trials, pragmatic trials, pilot and feasibility trials; reporting of trials; systematic reviews; and mentorship. He is a former member of the Interagency Advisory Panel on Research Ethics.
- Madzouka Kokolo, M.A. – Health Canada Researcher (alternate): Madzouka Kokolo is academically trained both in Epidemiology (Master of Science, University of Ottawa) and in Public Ethics (Master of Arts, Saint Paul University). She has been involved in health science projects in academic, governmental and non-profit contexts, locally, nationally, and internationally (e.g., Canadian Society for International Health, United Nations Population Fund). She has extensive knowledge and experience in clinical research designs, research conduct and methodological assessments and is currently working as an Epidemiologist in Health Canada's Health Products and Food Branch, routinely conducting critical appraisals of scientific evidence to support regulatory decision-making.
Finally, Charles Dupras, Ph.D., an Academic Associate with the McGill Centre of Genomics and Policy, joined the REB in July 2020 as an alternate member with expertise in ethics. Dr. Dupras was a valued contributor to the REB who brought a wealth of ethics knowledge and research experience. Unfortunately for the REB, Dr. Dupras was unable to continue on the board following a change in employment and resigned in January 2021. He is certainly missed by the REB and we wish him all the best in his new endeavours.
REB membership as of March 31, 2021
- Stéphane Ahern, M.D., Ph.D. – Ethics
- Stephanie Booth, D.Phil. – Public Health Agency of Canada Researcher
- Michael Wray Clarke, Ph.D. – Public Health Community Member
- Marie-Ève Couture Ménard, D.C.L. – Law
- Meredith Curren, Ph.D. – Health Canada Researcher
- Glenn Griener, Ph.D. – Ethics
- Janaki Jayanthan, MPH – Community Member, General Population
- Madzouka Kokolo, M.A. – Health Canada Researcher
- Kathleen Makela, B.A., LL.B. – Community Member, Indigenous Population
- Barbara McGillivray, M.D. – Chair and Researcher External to the Health Canada and the Public Health Agency of Canada
- Melanie McPhail, J.D. – Law
- Jean-Frédéric Ménard, LL.B., B.C.L. – Law
- Stuart Nicholls, Ph.D. – Researcher External to Health Canada and the Public Health Agency of Canada
- Meeka Otway – Community Member, Indigenous Population
- Guillaume Poliquin, M.D., Ph.D. – Public Health Agency of Canada Researcher (on temporary leave from the REB since June, 2020)
- Sandra Romain, Ph.D. – Community Member, General Population
- Lehana Thabane, Ph.D. – Researcher External to Health Canada and the Public Health Agency of Canada
- Nancy Walton, Ph.D. – Ethics
- Kue Young, M.D., D.Phil. – Public Health Community Member
REB Secretariat
- Gregory Huyer, Ph.D. – Manager
- Melinda Lee Choon – Senior Research Ethics Coordinator
- Suzi Vivolo – Administrator
- Gabriella Hilkes – Junior Analyst (co-op student)
PHAC liaison to the REB Secretariat
- Erica Bernier – Policy Analyst, Office of the Chief Science Officer (April-Dec., 2020)
- Trista Takacs – Ph.D. – Policy Analyst, Officer of the Chief Science Officer (effective Jan., 2021)
References
- Footnote 1
-
Note that this chart summarizes the submissions reviewed in 2020-21, and not submissions received. For example, 41 applications were reviewed in 2020-21, which includes 40 of the 44 applications received between April 1, 2020 and March 31, 2021, plus one application received at the end of the 2019-20 fiscal year.
- Footnote 2
-
"Days with REB" includes time with both the REB Secretariat and the REB. All times are measured in calendar days. 44 applications were received in 2020-21, but four remained outstanding as of March 31, 2021 and were therefore excluded from this analysis. In certain cases, additional time was required following REB review for the Privacy Management Division to complete its assessment; this additional time was excluded from these calculations.