2018-19 Departmental Results Report
Patented Medicine Prices Review Board
The Honourable Patty Hajdu
Minister of Health
ISSN: 2561-0732
Table of Contents
Chairperson’s message
I am pleased to present the 2018-19 Departmental Results Report for the Patented Medicine Prices Review Board (PMPRB).
The PMPRB is an independent quasi-judicial body established by Parliament in 1987 under the Patent Act (Act). The PMPRB protects and informs Canadians by ensuring that the prices of patented medicines sold in Canada are not excessive and by reporting on pharmaceutical trends.
Over the past three years, the PMPRB has evolved into a purpose-driven agency at the front lines of the federal government’s efforts to make prescription medicines more affordable for Canadians. This transformation is a realization of the Board and staff’s shared vision and strongly held belief in the potential of the PMPRB to make a unique and valued contribution to improving the pharmaceutical system in Canada. It was made possible through the very deliberate and methodical implementation of a strategy to revitalize the organization and reaffirm the relevance of its mandate, as set out in its 2015-2018 Strategic Plan, which the PMPRB has faithfully executed in the three years since its release.
No stone has been left unturned as part of that process. From the regulatory framework governing how the PMPRB sets ceiling prices for patented medicines, which is in the final stage of reform, to its information management (IM) and information technology (IT) infrastructure, which is increasingly paperless, wireless and interconnected, virtually every facet of the organization has been improved and modernized in the past three years. With the injection of new funding from Budget 2017, the PMPRB has also hired additional staff with the requisite knowledge and expertise to operationalize important amendments to its regulations that will come into force in July 2020.
In 2018-19, the PMPRB focussed its efforts on completing the one strategic priority from its 2015-2018 Strategic Plan that remains unrealized, namely framework modernization. In anticipation of the eventual passage of the aforementioned regulatory amendments, the PMPRB launched a two-track consultation process on a proposed new pricing framework that sought high level feedback from a steering committee composed of select stakeholders, and more targeted feedback from an expert working group. The outcome of the deliberations of these two groups has since been presented to the Board for its consideration prior to the release of new draft Guidelines for broader public consultation in the fall of 2019.
As it enters the homestretch in its efforts to modernize its regulatory framework and readies itself for the coming into force of the amended regulations in July 2020, the PMPRB has also been renovating its office space to Workplace 2.0 Fit-up standards and acquired additional space which will be converted into its own hearing room for excessive pricing and failure-to-file cases. That work is expected to be completed in early 2020-21.
Of the PMPRB’s many successes in the past three years, perhaps the most meaningful is how employees have responded to the transformation in the organization and its impact on their daily working lives. Even positive change can be disruptive for the people immediately affected and the PMPRB has made a sustained effort to make its employees feel empowered and personally vested in the modernization process. Its efforts in this regard have been rewarded in the form of consistent, across the board improvement in public service survey results since 2014, and strikingly so in the all important areas of employee engagement, leadership and workplace well being, where the PMPRB consistently scores among the top five organizations in the public service. Like any government organization, our greatest asset is our people. As we turn our attention to a new round of strategic priorities for the coming three years, our goal of being a healthy and engaging workplace and an employer of choice within the public service is one thing we have no intention of changing.
Dr. Mitchell Levine
Results at a glance
Priority 1 – Framework Modernization
In an effort to respond to concerns from certain stakeholders that Health Canada’s proposed amendments to the Patented Medicines Regulations did not provide enough information for them to properly evaluate the impact of the new regime, the PMPRB issued a scoping paper and additional guidance documents to explain how it intends to operationalize the regulatory amendments through changes to its non-binding Guidelines. The PMPRB sought feedback on these changes from a steering committee of key stakeholders and a working group of technical experts. Reports on the deliberations of the steering committee and working group were submitted to the Board for its consideration prior to the publication of new draft Guidelines for broader public consultation, which is taking place in the fall of 2019.
Priority 2 – Consumer-focused Regulation
In 2018-19, the PMPRB negotiated Voluntary Compliance Undertakings (VCU) totalling $6,996,128.49 in excess revenue payments by patentees to the Government of Canada.
In January 2019, the PMPRB issued a Notice of Hearing in the matter of Horizon Pharma and the medicine Cysteamine Bitartrate sold under the trade name “Procysbi”, which is set to be heard by the Board in February 2020. The investigation into the price of Procysbi, was commenced as the result of a complaint to the PMPRB by the pan-Canadian Pharmaceutical Alliance (pCPA).
In its 2016 Discussion Paper on Guidelines Modernization, the PMPRB signalled its intent to move to more of a risk-based approach to price regulation, which recognizes that patented medicines have differing potential to exercise market power and charge excessive prices. Although this new approach won’t be formalized until the PMPRB’s new Guidelines are finalized, the way in which the PMPRB prioritizes investigations into suspected instances of excessive pricing already reflects this line of thinking. Each investigation is subject to a decision matrix that includes consideration of such things as direct and indirect indicators of market power.
Priority 3 – Strategic Partnerships and Public Awareness
Domestically, in 2018-19, the PMPRB continued to conduct research and provide analytical support to public payers through its NPDUIS initiative. In addition, the latest edition of the PMPRB’s new Market Intelligence Report series, which provide detailed information on specific therapeutic market segments of importance to Canadians, analyzed the rapidly growing market for biologic anti-vascular endothelial growth factor (anti-VEGF) medicines. Anti-VEGF medicines are used in the treatment of wet (neovascular) age-related macular degeneration (AMD) and other retinal conditions. AMD is the leading cause of visual impairment in adults over 50, with the more serious "wet" form affecting over 100,000 Canadians. Despite a higher than average usage of these medicines in Canada, the Report found that their list prices here are substantially higher than international norms.
Internationally, heightened interest in Canada’s groundbreaking efforts to reform how it regulates patented medicines prices prompted a number of invitations to speak at various relevant international fora, including the World Organization’s (WHO) Fair Pricing summit in Johannesburg South Africa. The PMPRB availed itself of multiple such opportunities to share best practices with international health partners and continued to strengthen multilateral relations through active participation in the Pharmaceutical Pricing and Reimbursement Information (PPRI) networkFootnote i and engagement with representatives of the BeNeLuxA initiativeFootnote ii (Belgium, the Netherlands, Luxembourg, Austria and Ireland).
For more information on the Patented Medicine Prices Review Board’s (PMPRB) plans, priorities and results achieved, see the “Results: what we achieved” section of this report.
Results: what we achieved
Core Responsibilities
Regulate patented medicine prices
Description
The PMPRB regulates the prices of patented medicines by setting non-excessive price ceilings and taking enforcement action before the Board in the event of non-compliance.
Results
Results achieved
| Departmental results | Performance indicators | Target | Date to achieve target | 2018–19 Actual results | 2017–18 Actual results | 2016–17 Actual results |
|---|---|---|---|---|---|---|
| Affordable patented medicine prices | % of patented medicines prices in Canada below the median price of the PMPRB’s comparator countries | 50%(a) | March 31, 2019 | 57.1%(b) | 56.4% | 58% |
| % of patented medicine prices in Canada are within the thresholds set out in the PMPRB’s Excessive Price Guidelines | 95%(c) | March 31, 2019 | 90.5%(d) | 91.0% | 92.3% |
(a) Operating under the premise that the PMPRB would continue to conduct its price reviews without significant changes in its regulatory framework the PMPRB established a target of 50% of patented medicine prices being below the median price. Analysis in the PMPRB’s 2015 Annual Report indicated that the percentage of patented medicines priced below the median price of the PMPRB’s comparator countries was 51.8%, a decline from the previous two years. Based on these factors, it was determined that 50% would be a reasonable target.
(b) The 57.1% of patented medicine prices in Canada reported as being below the median international price includes a significant number of patented medicines being sold in fewer than five countries and therefore are not being compared to the actual median international price. Of the 1,399 patented medicines sold in Canada in 2018, only 830 were sold in five or more countries. Of this 830, only 395 patented medicines (48%) had a Canadian price below the median price. This is a significant difference from the reported 57.1%.
(c) This percentage, based on the number of price reviews completed by March 31 of the fiscal year referred to, is calculated as follows: the sum of the number of price reviews found to be within the Guidelines, plus the number of price reviews which did not trigger an investigation, plus the number of Voluntary Compliance Undertakings; divided by the number of patented medicines for which the price review was completed at March 31 of the fiscal year.
(d) As of March 31, 2019, 51 patented medicines were still under review, 128 were under investigation, two were the subject of a hearing and one was subject to a Stay Order.
Budgetary financial resources (dollars)
| 2018–19 Main Estimates | 2018–19 Planned spending | 2018–19 Total authorities available for use | 2018–19 Actual spending (authorities used) | 2018–19 Difference (Actual spending minus Planned spending) |
|---|---|---|---|---|
| 11,227,006 | 11,227,006 | 11,315,126 | 7,343,076 | (3,883,930)a |
a ‘Planned spending’ and ‘Total authorities available for use’ include funding for a Special Purpose Allotment (SPA) to conduct Public Hearings, in Vote 1 (Program expenditures) of $3,419,481. The SPA can only be used to cover the costs of public hearings, such as external legal counsel and expert witnesses, etc. For purposes of forecasting Planned Spending, it is necessary to assume that the entire SPA funding will be spent because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. In 2018-19, the PMPRB spent $358,978 from the SPA. Any unspent amount is returned to the Consolidated Revenue Fund.
Human resources (full-time equivalents)
| 2018–19 Planned full-time equivalents | 2018–19 Actual full-time equivalents | 2018–19 Difference (Actual full-time equivalents minus Planned full-time equivalents) |
|---|---|---|
| 51.0 | 47.0 | (4.0) |
The PMPRB’s Guidelines were conceived from the notion that the most effective and efficient way to protect the public from excessive prices is to provide patentees with the information and tools needed to facilitate voluntary compliance with its price ceilings. In 2018-19, the PMPRB recovered $6,996,128.49 in excess revenues through Voluntary Compliance Undertaking and Board Order payments to the Government of Canada.
Since the PMPRB’s inception, prices of patented medicines in Canada have generally remained below both inflation and the international median, based on the seven comparator countries (PMPRB7)Footnote iii named in the Regulations. More recently, however, Canadian patented medicine prices have been steadily rising relative to prices in the PMPRB7 and are on average 17% above median OECD prices. Canadian prices are third highest among the 31 OECD countries, behind only the United States, Germany and Switzerland.
While in the past the PMPRB enjoyed a relatively high rate of compliance with its Guidelines, that trend is coming under increasing strain owing to an influx of very high cost medicines which have increasingly come to dominate the market for new pharmaceuticals, not only in Canada but globally. It is expected that the amendments to the Regulations, which will come into force in July 2020, may place further strain on the degree to which patentees comply voluntarily with the pricing Guidelines, particularly with respect to very high cost, so-called “Category 1” medicines. The PMPRB will revisit the appropriateness of this indicator and its expected result at a later time once the new regime is fully operational.
In January 2019, the PMPRB issued a Notice of Hearing in the matter of Horizon Pharma and the medicine Cysteamine Bitartrate sold under the trade name “Procysbi”. The purpose of the hearing is to determine whether Horizon Pharma is selling or has sold Procysbi in any market in Canada at a price that, in the Board’s opinion, is or was excessive and if so, what order, if any, should be made.
Financial, human resources and performance information for the PMPRB’s Program Inventory is available in the GC InfoBase.Footnote iv
Internal Services
Description
Internal Services are those groups of related activities and resources that the federal government considers to be services in support of programs and/or required to meet corporate obligations of an organization. Internal Services refers to the activities and resources of the 10 distinct service categories that support Program delivery in the organization, regardless of the Internal Services delivery model in a department. The 10 service categories are:
- Acquisition Management Services
- Communications Services
- Financial Management Services
- Human Resources Management Services
- Information Management Services
- Information Technology Services
- Legal Services
- Materiel Management Services
- Management and Oversight Services
- Real Property Management Services
Results
Budgetary financial resources (dollars)
| 2018–19 Main Estimates | 2018–19 Planned spending | 2018–19 Total authorities available for use | 2018–19 Actual spending (authorities used) | 2018–19 Difference (Actual spending minus Planned spending) |
|---|---|---|---|---|
| 3,644,866 | 3,644,866 | 3,830,160 | 2,955,259 | (689,607) |
Human resources (full-time equivalents)
| 2018–19 Planned full-time equivalents | 2018–19 Actual full-time equivalents | 2018–19 Difference (Actual full-time equivalents minus Planned full-time equivalents) |
|---|---|---|
| 21.0 | 19.1 | (1.9) |
In 2018-19, the PMPRB took a number of steps to create an agile, empowered and engaged workforce with a new approach to its office technology:
- All desktop computers were replaced with tablets, enabling employee mobility;
- MobiKeys (remote connection to desktop computers) were replaced with a Virtual Private Network (VPN), allowing employees access to the PMPRB network from their tablets from any location; and
- Wi-Fi access was installed throughout the PMPRB office space.
In addition, the PMPRB has made significant process in the Workplace 2.0 initiative. It has acquired additional space for Hearing rooms and work on the retrofit of its current office space is progressing well. The initiative is on target to meet its March 2020 completion.
Analysis of trends in spending and human resources
Actual expenditures
Departmental spending trend graph
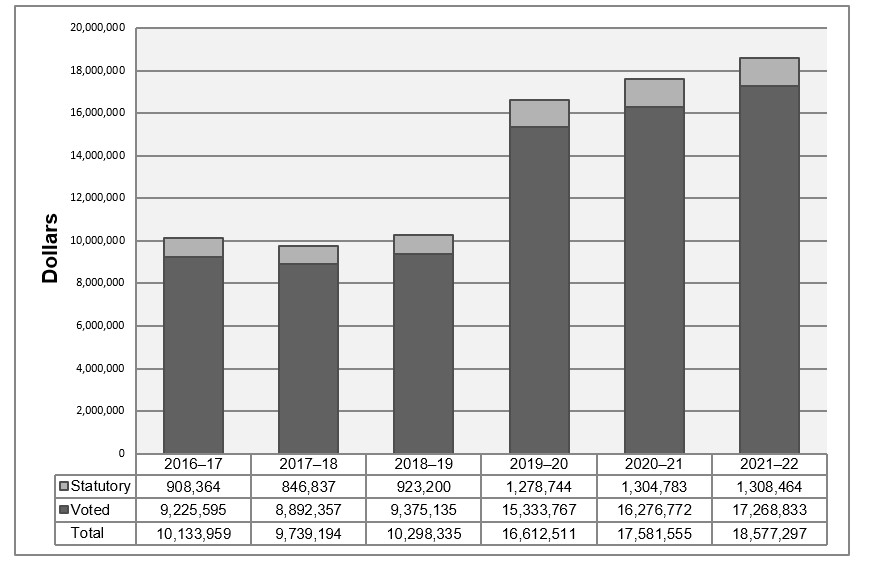
Figure description
The following graph shows the PMPRB's spending trend over time. It illustrates on a bar graph the actual statutory and voted spending for 2016-17 to 2018-19, and planned statutory and voted spending for 2019-20 and 2021-22.
| 2016-17 | 2017-18 | 2018-19 | 2019-20 | 2020-21 | 2021-22 | |
|---|---|---|---|---|---|---|
| Statutory | 908,364 | 846,837 | 923,200 | 1,278,744 | 1,304,783 | 1,308,464 |
| Voted | 9,225,595 | 8,892,357 | 9,375,135 | 15,333,767 | 16,276,772 | 17,268,833 |
| Total | 10,133,959 | 9,739,194 | 10,298,335 | 16,612,511 | 17,581,555 | 18,577,297 |
Voted spending in 2018-19 was higher than voted spending in 2017-18 due to increased funding received through Budget 2017, which was used to hire additional regulatory officers, health economists, data analysts, and legal counsel, as well as to spend on the Workplace 2.0 fit-up.
Voted spending in 2017-18 was lower than voted spending in 2016-17 due in large part to decreased spending for hearings, offset by retroactive payments to employees pertaining to collective bargaining. In 2017-18, the PMPRB spent $893,209 from the SPA, as compared to $1,883,121 in 2016-17, a difference of $989,912.
As announced in Budget 2017, the PMPRB received additional funding for future years; $3,849,215 in 2018-19, $5,694,677 in 2019-20, $6,671,853 in 2020-21, $7,668,725 in 2021-22 and $5,680,633 in 2022-23 and ongoing, including Employee Benefits Payments (EBP) and increased funding for the SPA.
For purposes of forecasting Planned Spending for 2019-20 and future years, it is necessary to assume that the entire SPA funding for hearings will be spent because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict.
Budgetary performance summary for Core Responsibilities and Internal Services (dollars)
| Core Responsibilities and Internal Services | 2018–19 Main Estimates | 2018–19 Planned spending | 2019–20 Planned spending | 2020–21 Planned spending | 2018–19 Total authorities available for use | 2018–19 Actual spending (authorities used) | 2017–18 Actual spending (authorities used) | 2016–17 Actual spending (authorities used) |
|---|---|---|---|---|---|---|---|---|
| Regulate Patented Medicine Prices | 11,227,006 | (a) 11,227,006 | 13,370,895 | 14,564,842 | 11,315,126 | 7,343,076 | 7,109,924 | 7,714,937 |
| Subtotal | 11,227,006 | 11,227,006 | 13,445,565 | 14,564,842 | 11,315,126 | 7,343,076 | 7,109,924 | 7,714,937 |
| Internal Services | 3,644,866 | 3,644,866 | 3,241,616 | 3,016,713 | 3,830,160 | 2,955,259 | 2,629,270 | 2,419,022 |
| Total(b) | 14,871,872 | 14,871,872 | 16,612,511 | 17,581,555 | 15,145,286 | (c) 10,298,335 | 9,739,194 | 10,133,959 |
(a) For purposes of forecasting Planned Spending for 2018-19 and future years it is assumed the entire SPA funding for hearings will be spent. This is done because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict.
(b) As announced in Budget 2017, the PMPRB received additional funding for future years: $3,849,215 in 2018-19, $5,694,677 in
2019-20, $6,671,853 in 2020-21, $7,668,725 in 2021-22 and $5,680,633 in 2022-23 and ongoing, including Employee Benefits Payments (EBP) and increased funding for the SPA.
(c) Actual spending in 2018-19 was $4,846,951 lower than the Total authorities available for use. This was largely due to delays experienced in the regulatory reform process. These delays resulted in the PMPRB postponing some staffing actions that created a $583,035 lapse in salary spending. In addition, the PMPRB only used $362,358 of the SPA funding (compared to $893,209 in 2017-18), contributing $3,057,123 to the funding lapse. Finally, there were delays in the Workplace 2,0 refit project which, combined with lower general goods and services spending, resulted in a funding lapse of $1,206,793.
Actual human resources
Human resources summary for Core Responsibilities and Internal Services (full-time equivalents)
| Core Responsibilities and Internal Services | 2016–17 Actual full-time equivalents | 2017–18 Actual full-time equivalents | 2018–19 Planned full-time equivalents | 2018–19 Actual full-time equivalents | 2019–20 Planned full-time equivalents | 2020–21 Planned full-time equivalents |
|---|---|---|---|---|---|---|
| Regulate Patented Medicine Prices | 44.4 | 42.3 | 51.0 | 47.0 | 60.5 | 63.0 |
| Subtotal | 44.4 | 42.3 | 51.0 | 47.0 | 60.5 | 63.0 |
| Internal Services | 19.3 | 18.0 | 21.0 | 19.1 | 21.5 | 20.0 |
| Total | 63.7 | 60.3 | (a) 72.0 | (b) 66.1 | 82.0 | 83.0 |
(a) As announced in Budget 2017, the PMPRB received additional funding for future years; some of that funding will be used to staff additional FTEs.
(b) Delays in the regulatory reform process resulted in the postponement of some intended staffing actions.
Expenditures by vote
For information on the PMPRB’s organizational voted and statutory expenditures, consult the Public Accounts of Canada 2018–2019.Footnote v
Government of Canada spending and activities
Information on the alignment of the PMPRB’s spending with the Government of Canada’s spending and activities is available in the GC InfoBaseFootnote vi.
Financial statements and financial statements highlights
Financial statements
The PMPRB’s financial statements (unaudited) for the year ended March 31, 2019, are available on the departmental websiteFootnote vii.
Financial statements highlights
Condensed Statement of Operations (unaudited) for the year ended March 31, 2019 (dollars)
| Financial information | 2018–19 Planned results | 2018–19 Actual results | 2017–18 Actual results | Difference (2018–19 Actual results minus 2018–19 Planned results) | Difference (2018–19 Actual results minus 2017–18 Actual results) |
|---|---|---|---|---|---|
| Total expenses | 16,107,128 | 11,161,743 | 11,175,045 | (4,945,385) | (13,302) |
| Total revenues | 0 | 5,529 | 686 | 5,529 | 4,843 |
| Net cost of operations before government funding and transfers | 16,107,128 | 11,156,214 | 11,174,359 | (4,950,914) | (18,145) |
Condensed Statement of Financial Position (unaudited) as of March 31, 2019 (dollars)
| Financial Information | 2018–19 | 2017–18 | Difference (2018–19 minus 2017–18) |
|---|---|---|---|
| Total net liabilities | 2,065,743 | 2,087,459 | (21,716) |
| Total net financial assets | 1,448,179 | 1,307,889 | 140,290 |
| Departmental net debt | 617,564 | 779,570 | (162,006) |
| Total non-financial assets | 209,562 | 120,272 | 89,290 |
| Departmental net financial position | (408,002) | (659,298) | 251,296 |
Supplementary information
Corporate information
Organizational profile
Appropriate minister: The Honourable Ginette Petitpas Taylor
Institutional head: Dr, Mitchell Levine, Chairperson
Ministerial portfolio: Health
Enabling instrument: Patent ActFootnote viii and Patented Medicines RegulationsFootnote ix
Year of incorporation / commencement: 1987
Other:
The Minister of Health is responsible for the pharmaceutical provisions of the Act set out in sections 79 to 103. Although the PMPRB is part of the Health Portfolio, because of its quasi-judicial responsibilities the PMPRB carries out its mandate at arm’s length from the Minister. It also operates independently of Health Canada, which approves drugs for safety, efficacy and quality; other Health Portfolio members, such as the Public Health Agency of Canada, the Canadian Institutes of Health Research and the Canadian Food Inspection Agency; and federal, provincial and territorial (F/P/T) public drug plans, which approve the listing of drugs for their respective formularies for reimbursement purposes; and the Common Drug Review, administered by the Canadian Agency for Drugs and Technologies in Health (CADTH), which recommends drugs that should qualify for reimbursement purposes by participating public drug plans.
Raison d’être, mandate and role: who we are and what we do
“Raison d’être, mandate and role: who we are and what we do” is available on the PMPRB’s websiteFootnote x.
For more general information about the department, see the “Supplementary information” section of this report.
For more information on the department’s organizational mandate letter commitments, see the Minister’s mandate letterFootnote xi.
Operating context and key risks
Information on operating context and key risks is available on the PMPRB’s website.Footnote xii
The PMPRB’s Departmental Results Framework and Program Inventory of record for 2018–19 are shown below.
Graphical presentation of Departmental Results Framework and Program Inventory
| Departmental Results Framework | Core Responsibility: Regulate Patented Medicine Prices | Internal Services | |
| Departmental Result: Affordable patented drug prices | Indicator 1: % of patented drug prices in Canada are below the median price of the PMPRB’s comparator countries | ||
| Indicator 2: % of patented drug prices in Canada within the thresholds set out in the Guidelines | |||
| Program Inventory | Patented Medicine Price Regulation Program | ||
| Pharmaceutical Trends Program | |||
Supporting information on the Program Inventory
Financial, human resources and performance information for the PMPRB’s Program Inventory is available in the GC InfoBaseFootnote xiii.
Supplementary information tables
The following supplementary information tables are available on the PMPRB’s website:
Federal tax expenditures
The tax system can be used to achieve public policy objectives through the application of special measures such as low tax rates, exemptions, deductions, deferrals and credits. The Department of Finance Canada publishes cost estimates and projections for these measures each year in the Report on Federal Tax Expenditures.Footnote xvi This report also provides detailed background information on tax expenditures, including descriptions, objectives, historical information and references to related federal spending programs. The tax measures presented in this report are the responsibility of the Minister of Finance.
Organizational contact information
The Patented Medicine Prices Review Board
Box L40
Standard Life Centre
333 Laurier Avenue West
Suite 1400
Ottawa, Ontario K1P 1C1
Toll-free no.: 1-877-861-2350
Facsimile: (613) 288-9643
TTY: (613) 288-9654
Email: PMPRB.Information-Renseignements.CEPMB@pmprb-cepmb.gc.ca
Website: www.pmprb-cepmb.gc.ca
Appendix: definitions
appropriation (crédit)
Any authority of Parliament to pay money out of the Consolidated Revenue Fund.
budgetary expenditures (dépenses budgétaires)
Operating and capital expenditures; transfer payments to other levels of government, organizations or individuals; and payments to Crown corporations.
Core Responsibility (responsabilité essentielle)
An enduring function or role performed by a department. The intentions of the department with respect to a Core Responsibility are reflected in one or more related Departmental Results that the department seeks to contribute to or influence.
Departmental Plan (plan ministériel)
A report on the plans and expected performance of an appropriated department over a three-year period. Departmental Plans are tabled in Parliament each spring.
Departmental Result (résultat ministériel)
A Departmental Result represents the change or changes that the department seeks to influence. A Departmental Result is often outside departments’ immediate control, but it should be influenced by program-level outcomes.
Departmental Result Indicator (indicateur de résultat ministériel)
A factor or variable that provides a valid and reliable means to measure or describe progress on a Departmental Result.
Departmental Results Framework (cadre ministériel des résultats)
Consists of the department’s Core Responsibilities, Departmental Results and Departmental Result Indicators.
Departmental Results Report (rapport sur les résultats ministériels)
A report on an appropriated department’s actual accomplishments against the plans, priorities and expected results set out in the corresponding Departmental Plan.
experimentation (expérimentation)
Activities that seek to explore, test and compare the effects and impacts of policies, interventions and approaches, to inform evidence-based decision-making, by learning what works and what does not.
full-time equivalent (équivalent temps plein)
A measure of the extent to which an employee represents a full person-year charge against a departmental budget. Full-time equivalents are calculated as a ratio of assigned hours of work to scheduled hours of work. Scheduled hours of work are set out in collective agreements.
gender-based analysis plus (GBA+) (analyse comparative entre les sexes plus [ACS+])
An analytical process used to help identify the potential impacts of policies, Programs and services on diverse groups of women, men and gender differences. We all have multiple identity factors that intersect to make us who we are; GBA+ considers many other identity factors, such as race, ethnicity, religion, age, and mental or physical disability.
government-wide priorities (priorités pangouvernementales)
For the purpose of the 2018–19 Departmental Results Report, those high-level themes outlining the government’s agenda in the 2015 Speech from the Throne, namely: Growth for the Middle Class; Open and Transparent Government; A Clean Environment and a Strong Economy; Diversity is Canada’s Strength; and Security and Opportunity.
horizontal initiative (initiative horizontale)
An initiative where two or more departments are given funding to pursue a shared outcome, often linked to a government priority.
non-budgetary expenditures (dépenses non budgétaires)
Net outlays and receipts related to loans, investments and advances, which change the composition of the financial assets of the Government of Canada.
performance (rendement)
What an organization did with its resources to achieve its results, how well those results compare to what the organization intended to achieve, and how well lessons learned have been identified.
performance indicator (indicateur de rendement)
A qualitative or quantitative means of measuring an output or outcome, with the intention of gauging the performance of an organization, program, policy or initiative respecting expected results.
performance reporting (production de rapports sur le rendement)
The process of communicating evidence-based performance information. Performance reporting supports decision making, accountability and transparency.
plan (plan)
The articulation of strategic choices, which provides information on how an organization intends to achieve its priorities and associated results. Generally a plan will explain the logic behind the strategies chosen and tend to focus on actions that lead up to the expected result.
planned spending (dépenses prévues)
For Departmental Plans and Departmental Results Reports, planned spending refers to those amounts presented in Main Estimates.
A department is expected to be aware of the authorities that it has sought and received. The determination of planned spending is a departmental responsibility, and departments must be able to defend the expenditure and accrual numbers presented in their Departmental Plans and Departmental Results Reports.
priority (priorité)
A plan or project that an organization has chosen to focus and report on during the planning period. Priorities represent the things that are most important or what must be done first to support the achievement of the desired Strategic Outcome(s) or Departmental Results.
program (programme)
Individual or groups of services, activities or combinations thereof that are managed together within the department and focus on a specific set of outputs, outcomes or service levels.
result (résultat)
An external consequence attributed, in part, to an organization, policy, program or initiative. Results are not within the control of a single organization, policy, program or initiative; instead they are within the area of the organization’s influence.
statutory expenditures (dépenses législatives)
Expenditures that Parliament has approved through legislation other than appropriation acts. The legislation sets out the purpose of the expenditures and the terms and conditions under which they may be made.
Strategic Outcome (résultat stratégique)
A long-term and enduring benefit to Canadians that is linked to the organization’s mandate, vision and core functions.
target (cible)
A measurable performance or success level that an organization, program or initiative plans to achieve within a specified time period. Targets can be either quantitative or qualitative.
voted expenditures (dépenses votées)
Expenditures that Parliament approves annually through an Appropriation Act. The Vote wording becomes the governing conditions under which these expenditures may be made.