2019-20 Departmental Results Report
Patented Medicine Prices Review Board
Patty Hajdu
Minister of Health
ISSN: 2561-0732
Table of Contents
Chairperson’s message
I am pleased to present the 2019-20 Departmental Results Report for the Patented Medicine Prices Review Board (PMPRB).
The PMPRB is an independent quasi-judicial body established by Parliament in 1987 under the Patent Act (Act). The PMPRB protects and informs Canadians by ensuring that the prices of patented medicines sold in Canada are not excessive and by reporting on pharmaceutical trends.
This past year was a busy and important one for the PMPRB. In 2019-20, the organization focused on completing the final steps necessary for reforming and modernizing its regulatory framework. To that end, in November 2019, we published a draft set of new pricing GuidelinesFootnote i, followed by the most intensive and far reaching public consultation in our more than three decades long history. In addition to hosting several policy forums, webinars and bilateral meetings in Ottawa with key stakeholders, the PMPRB attended over 60 meetings across Canada, with more than 260 members of its stakeholder community. Over 120 written submissions were received at the close of this 85-day process. In June 2020, following the Government’s announcement of its decision to postpone the coming into force of the amended Patented Medicines Regulations by six months, to January 1, 2021, due to the COVID-19 pandemic, the PMPRB published a second draft set of new GuidelinesFootnote ii that reflected our understanding of the feedback we received on the first draft. This was followed by a 45-day written consultation period which ended August 4, 2020.
The final steps in our multiyear effort to reform and modernize our regulatory framework took place alongside our usual and customary work under our reporting mandate of identifying and analyzing key pharmaceutical trends in Canada. In addition to our Annual Report tabled yearly in Parliament, in 2019-20, the PMPRB published seven analytical reports, three chartbooks, and eight postersFootnote iii under its National Prescription Drug Utilization Information System (NPDUIS) banner. It also released updated Guidelines for Conducting Pharmaceutical Budget Impact Analyses for Submission to Public Drug Plans in Canada.Footnote iv These Guidelines provide a standardized approach and detailed recommendations for developing a Budget Impact Analysis for submission to the Canadian Agency for Drugs and Technologies in Health (CADTH) or to one of the participating federal/provincial/territorial drug plans. The final document is the result of a multi-year process that included extensive research and consultation with relevant stakeholders, including CADTH and participating public drug plans.
The PMPRB also underwent a major overhaul of its office space in 2019-20, with the implementation of Workplace 2.0 – a government-wide initiative to modernize the workplace. Despite major construction ongoing throughout the year, PMPRB staff experienced minimal disruption in their everyday working lives, thanks to the phased-in approach carefully conceived and meticulously carried out by its Corporate Services branch. The same can be said of the impact of the COVID-19 pandemic on the PMPRB’s day to day operations, thanks also to the foresight of its Corporate Services branch and the efforts it has made in recent years to develop a cutting edge and fully mobile IT system so that its employees can work seamlessly from home or wherever their lives take them.
Once the PMPRB’s new Guidelines are finalized and the amended regulations have come into force in January 2021, we look forward to working with all of our stakeholders on a comprehensive Guidelines Monitoring and Evaluation Plan (GMEP) that will enable us to assess the impact of the new regime and fine tune it in real time so that the PMPRB can continue to protect Canadians from excessive prices while avoiding any unintended consequences to other aspects of our health care system.
Dr. Mitchell Levine
Results at a glance
2019-20
Total actual spending: $13 million
Total actual full-time equivalents: 79
Priority 1 – Framework Modernization
- The Steering Committee on Modernization of Price Review Process Guidelines (Steering Committee) and the Technical Working Group published their final reports on the PMPRB’s proposed new risk-based framework for regulating prices of patented medicines.
- The amendments to the Patented Medicines Regulations received royal assent from the Governor-in-Council.
- The PMPRB published new draft Guidelines (November 2019 draft Guidelines) and held an 85-day public consultation on the proposed changes.
This priority supports the government’s agenda in the 2019 Speech from the Throne to keep Canadians safe and healthy.
Priority 2 – Capacity Development
- The PMPRB completed a refit of its workspace to Workplace 2.0 Fit-up Standards in order to accommodate the increased staff needed to administer the PMPRB’s new regulatory framework.
- The PMPRB increased its FTE count by approximately 20 percent over the course of the fiscal year, in an effort to hire new staff with the requisite skill set and expertise to apply the new excessive pricing factors under the amended regulations.
- The PMPRB hosted multiple training sessions for its staff on health technology assessment and organized an interchange arrangement with INESSS.
- PMPRB staff held mock “Red/Green” exercises simulating Board hearings under the new regulatory framework.
Priority 3 – Employee Engagement
- The PMPRB held a Town Hall where staff feedback was sought on new strategic priorities.
- A wellness room was added to the PMPRB’s office space as part of the 2.0 Workplace refit, with weekly yoga and meditation sessions.
- The Workplace Improvement Team spearheaded several initiatives to improve the work environment for staff.
- Junior staff shadowed senior officials throughout the consultations on the new Guidelines.
- An interbranch working group on Guideline reform was established to troubleshoot options for responding to stakeholder feedback on the draft Guidelines.
- PMPRB staff from various branches were engaged in the development of the Guidelines Monitoring and Evaluation Plan (GMEP)Footnote v.
For more information on the PMPRB’s plans, priorities and results achieved, see the “Results: what we achieved” section of this report.
Results: what we achieved
Core responsibility
Regulate Patented Medicine Prices
Description: The PMPRB regulates the prices of patented medicines by setting non-excessive price ceilings and taking enforcement action before the Board in the event of non-compliance.
Results
Since 1987, pharmaceutical costs in Canada have grown at an average annual rate of 7.2%Footnote vi, outpacing all other health care costs and growing at well over 3 times the pace of inflation. At 15.7% of total health care spending, pharmaceuticals now rank ahead of spending on physicians.Footnote vii About 1 in 5 Canadians reports having no prescription medicine coverage and many more are under-insured or face high deductibles or co-pays. Almost 1 in 10 Canadians have had to forego filling a prescription medicine in the past year for reasons related to cost.Footnote viii
The introduction, of new, extremely high-priced patented medicines has been a major driver of sales growth in recent years. High cost medicines now account for close to 40 percent of pharmaceutical expenditures in Canada yet cover less than two percent of claimants.
In 2019-20 the PMPRB once again, saw a decline in the rate of compliance with its Guidelines to 85.9% down from 90.5% in 2018-19 and well below the 95% target. In recent years, patentees have been more apt to challenge the price ceilings applied under the PMPRB’s pricing Guidelines to the latest generation of very high cost medicines that is coming to dominate the market. In 2019-20, the PMPRB recovered $3,568,299.329 in excess revenues through payments to the Government of Canada via Voluntary Compliance Undertakings and Board Orders.
The PMPRB is currently consulting on changes to its Guidelines to give effect to the Government’s recent amendments to the Patented Medicines Regulations (Regulations) and formalize its move to a more risk-based approach to regulating prices. Although changes to the PMPRB’s Regulations and Guidelines are expected to result in an initial uptick in litigation as patentees test the boundaries of the new regime, compliance levels should normalize over time. With the implementation of the new Guidelines, the PMPRB will be reviewing this indicator, its target, and the timeframe for achieving the target.
The amendments to the Regulations are being challenged by the pharmaceutical industry in both the Federal Court and the Quebec Superior Court. The PMPRB is working closely with the Minister of Health and the Attorney General to responsibly defend these challenges.
Gender-based analysis plus
The PMPRB recognizes that sex and gender differences, race, ethnicity, age and mental or physical disability are factors to consider in the accessibility, affordability and appropriate use of prescription medicines and medical devices. Differences in sex and gender+ roles, income and utilization of health care services can affect access to medicines and health insurance, prescribing patterns and medicine use and may have important repercussions for health and well-being.
Since the price of a medicine does not vary for the sex or gender of the user, the PMPRB’s price review process does not take explicit account of the diversity of user groups or their economic situation, lower medicine prices, and associated savings for all payers, will benefit all, both sex and gender+ populations directly through lower out of pocket costs and indirectly through health system reinvestments and improved access to better care. In addition, the very high-cost medicines, which will be the focus of the PMPRB’s new risk-based regulatory framework, often treat rare diseases that can impact certain minority ethnic groups disproportionately.
Results achieved
| Departmental results | Performance indicators | Target | Date to achieve target | 2017–18 Actual results | 2018–19 Actual results | 2019–20 Actual results |
|---|---|---|---|---|---|---|
| Affordable patented medicine prices | % of patented medicines prices in Canada are below the median of the PMPRB’s comparator countries | 50%(a) | March 31, 2020 | 56.4% | 57.1% | 56.9%(b) |
| % of patented medicine prices in Canada within the thresholds set out in the Guidelines | 95%(c) | March 31, 2020 | 91.0% | 90.5% | 85.9%(d) |
(a) Operating under the premise that the PMPRB would continue to conduct its price reviews without significant changes in its regulatory framework, the PMPRB established a target of 50% of patented medicine prices being below the median price. Analysis in the PMPRB’s 2015 Annual Report indicated that the percentage of patented medicines priced below the median price of the PMPRB’s comparator countries was 51.8%, a decline from the previous two years. Based on these factors, it was determined that 50% would be a reasonable target.
(b) The 56.9% of patented medicine prices in Canada reported as being below the median international price includes a significant number of patented medicines being sold in fewer than five countries and therefore are not being compared to the actual median international price. Of the 1,348 patented medicines sold in Canada in 2019, only 778 were sold in five or more countries. Of this 778, only 365 patented medicines (47%) had a Canadian price below the median price. This is a significant difference from the reported 56.9%.
(c) This percentage, based on the number of price reviews completed by March 31 of the fiscal year referred to, is calculated as follows: the sum of the number of price reviews found to be within the Guidelines, plus the number of price reviews which did not trigger an investigation, plus the number of Voluntary Compliance Undertakings; divided by the number of patented medicines for which the price review was completed at March 31 of the fiscal year.
(d) As of March 31, 2020, 60 patented medicines were still under review, and 128 were under investigation, two were the subject of a hearing and one was subject to a Stay Order.
Budgetary financial resources (dollars)
| 2019–20 Main Estimates | 2019–20 Planned spending | 2019–20 Total authorities available for use | 2019–20 Actual spending (authorities used)* | 2019–20 Difference (Actual spending minus Planned spending) |
|---|---|---|---|---|
| 13,370,895 | 13,370,895 | 13,115,485 | 9,336,597 | (4,034,298)(a) |
(a) ‘Planned spending’ and ‘Total authorities available for use’ include funding for a Special Purpose Allotment (SPA) to conduct Public Hearings, in Vote 1 (Program expenditures) of $4,276,566. The SPA can only be used to cover the costs of public hearings, such as external legal counsel and expert witnesses, etc. For purposes of forecasting Planned Spending, it is necessary to assume that the entire SPA funding will be spent because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. In 2019-20, the PMPRB spent $968,852 from the SPA. Any unspent amount is returned to the Consolidated Revenue Fund.
Human resources (full-time equivalents)
| 2019–20 Planned full-time equivalents | 2019–20 Actual full-time equivalents | 2019–20 Difference (Actual full-time equivalents minus Planned full-time equivalents) |
|---|---|---|
| 62 | 58 | (4)(a) |
(a) Delays in the regulatory reform process resulted in the postponement of some intended staffing actions.
Financial, human resources and performance information for the PMPRB’s Program Inventory is available in GC InfoBaseFootnote ix.
Internal services
Description
Internal Services are those groups of related activities and resources that the federal government considers to be services in support of programs and/or required to meet corporate obligations of an organization. Internal Services refers to the activities and resources of the 10 distinct service categories that support Program delivery in the organization, regardless of the Internal Services delivery model in a department. The 10 service categories are:
- Acquisition Management Services
- Communication Services
- Financial Management Services
- Human Resources Management Services
- Information Management Services
- Information Technology Services
- Legal Services
- Material Management Services
- Management and Oversight Services
- Real Property Management Services
Budgetary financial resources (dollars)
| 2019–20 Main Estimates | 2019–20 Planned spending | 2019–20 Total authorities available for use | 2019–20 Actual spending (authorities used) | 2019–20 Difference (Actual spending minus Planned spending) |
|---|---|---|---|---|
| 3,241,616 | 3,241,616 | 4,042,060 | 3,977,283 | 735,667(a) |
(a) The difference between ‘Actual spending’ and Planned spending’ is largely the result of the allocation of 2019-20 Carry Forward to the PMPRB’s Workplace 2.0 project. The ‘Carry Forward’ is not included in the Main Estimates nor Planned Spending amounts for 2019-20.
Human resources (full-time equivalents)
| 2019–20 Planned full-time equivalents | 2019–20 Actual full-time equivalents | 2019–20 Difference (Actual full-time equivalents minus Planned full-time equivalents) |
|---|---|---|
| 20 | 21 | 1 |
In 2019-20, the PMPRB made progress on reaching it full FTE complement, however, delays in the operationalization of the new excessive pricing factors meant the PMPRB could delay staffing some positions because those skill sets were not yet required. The PMPRB developed a flexible and pragmatic approach to staffing which has allowed it to hire individuals as they are needed.
The PMPRB completed the Workplace 2.0 initiative and all staff are now in their permanent office space. In addition, it has completed the design phase for its new hearing room space and work on its construction began in April 2020.
The PMPRB also completed the structure/framework of the Automatic Classification Metadata Enhancements (ACME) Project for all branches but, refinements were required which resulted in the Project taking longer than expected. This was in part because the PMPRB structure was more complex than was anticipated. The ACME is intended to reduce the manual information end-users are required to input into the Electronic Document and Records Management System (EDRMS). The ACME will automate processes such as: classification; metadata generation and tagging; and, information governance. Post implementation performance will be monitored, and user experience will be gauged to determine the project’s success and the need for modifications.
Analysis of trends in spending and human resources
Actual expenditures
Departmental spending trend graph
The following graph presents actual and planned (voted and statutory spending) over time.
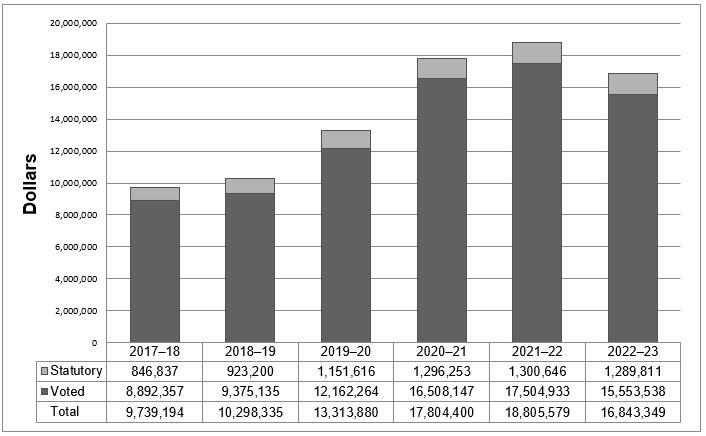
Voted spending in 2019-20 was higher than voted spending in 2018-19 due to increased funding received through Budget 2017, which was used to hire additional regulatory officers, health economists, data analysts, and legal counsel, as well as for work on the Workplace 2.0 fit-up and additional funding for the Special Purpose Allotment (SPA).
As announced in Budget 2017, the PMPRB received additional funding for future years; $3,849,215 in 2018-19, $5,694,677 in 2019-20, $6,671,853 in 2020-21, $7,668,725 in 2021-22 and $5,680,633 in 2022-23 and ongoing, including Employee Benefits Payments (EBP) and increased funding for the SPA.
For purposes of forecasting Planned Spending for 2020-21 and future years, it is necessary to assume that the entire SPA funding for hearings will be spent because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. The amount of the SPA for 2020-21 is $5,257,786; 2021-22 is $6,206,486; and, for 2022-23 and beyond the amount of the SPA is $4,463,361. This is the reason for declined planned spending in 2022-23.
Budgetary performance summary for Core Responsibilities and Internal Services* (dollars)
| Core responsibilities and Internal Services | 2019–20 Main Estimates | 2019–20 Planned spending | 2020–21 Planned spending | 2021–22 Planned spending | 2019–20 Total authorities available for use | 2019–20 Actual spending (authorities used) | 2018–19 Actual spending (authorities used) | 2017–18 Actual spending (authorities used) |
|---|---|---|---|---|---|---|---|---|
| Affordable patented medicine prices | 13,370,895 | 13,370,895 | 14,728,961 | 15,755,364 | 13,115,485 | 9,336,597 | 7,343,076 | 7,109,924 |
| Subtotal | 13,370,895 | 13,370,895 | 14,728,961 | 15,755,364 | 13,115,485 | 9,336,597 | 7,343,076 | 7,109,924 |
| Internal Services | 3,241,616 | 3,241,616 | 3,075,439 | 3,050,215 | 4,042,060 | 3,977,283 | 2,955,259 | 2,629,270 |
| Total | 16,612,511 | 16,612,511 | 17,804,400 | 18,805,579 | 17,157,545 | 13,313,880 | 10,298,335 | 9,739,194 |
Planned spending in 2019-20 was higher than Actual spending is largely due to a lapse of funding for the Special Purpose Allotment (SPA) to conduct Public Hearings. The SPA can only be used to cover the costs of public hearings, such as external legal counsel and expert witnesses, etc. For purposes of forecasting Planned Spending, it is necessary to assume that the entire SPA funding will be spent because these expenditures are dependent on the number of hearings, and the length and complexity of the hearings held, which are difficult to predict. In 2019-20, the SPA was $4,276,566 and the PMPRB only spent $968,852. Any unspent amount is returned to the Consolidated Revenue Fund.
Actual human resources
Human resources summary for core responsibilities and Internal Services
| Core responsibilities and Internal Services | 2017–18 Actual full-time equivalents | 2018–19 Actual full-time equivalents | 2019–20 Planned full-time equivalents | 2019–20 Actual full-time equivalents | 2020–21 Planned full-time equivalents | 2021–22 Planned full-time equivalents |
|---|---|---|---|---|---|---|
| Affordable patented medicine prices | 42 | 47 | 62 | 58 | 65 | 65 |
| Subtotal | 42 | 47 | 62 | 58 | 65 | 65 |
| Internal Services | 18 | 19 | 20 | 21 | 22 | 22 |
| Total | 60 | 65 | 82 | 79 | 87 | 87 |
The increase in planned FTEs for 2019-20 and beyond is a result of the additional funding received in the 2017 Budget and the need for additional staff and expertise to address the requirements of framework modernization. These additional resources will make the PMPRB a more relevant and effective price regulator, with more legal capacity to manage a greater number of hearings, more expertise in health economics, epidemiology and financial accounting and more modern and user-friendly IT infrastructure.
Expenditures by vote
For information on the PMPRB’s organizational voted and statutory expenditures, consult the Public Accounts of Canada 2019–2020.Footnote x
Government of Canada spending and activities
Information on the alignment of the PMPRB’s spending with the Government of Canada’s spending and activities is available in GC InfoBase.Footnote xi
Financial statements and financial statements highlights
Financial statements
The PMPRB’s financial statements (unaudited) for the year ended March 31, 2020, are available on the departmental website.
Financial statement highlights
Condensed Statement of Operations (unaudited) for the year ended March 31, 2020 (dollars)
| Financial information | 2019–20 Planned results* | 2019–20 Actual results | 2018–19 Actual results | Difference (2019–20 Actual results minus 2019–20 Planned results) | Difference (2019–20 Actual results minus 2018–19 Actual results) |
|---|---|---|---|---|---|
| Total expenses | 18,187,140 | 14,905,203 | 11,161,743 | (3,281,937) | 3,743,460 |
| Total revenues | - | 1,551 | 5,529 | 1,551 | (3,978) |
| Net cost of operations before government funding and transfers | 18,187,140 | 14,903,652 | 11,156,214 | (3,283,488) | 3,747,438 |
Condensed Statement of Financial Position (unaudited) as of March 31, 2020 (dollars)
| Financial information | 2019–20 | 2018–19 | Difference (2019–20 minus 2018–19) |
|---|---|---|---|
| Total net liabilities | 2,639,317 | 2,065,743 | 573,574 |
| Total net financial assets | 1,878,279 | 1,448,179 | 430,100 |
| Departmental net debt | 761,038 | 617,564 | 143,474 |
| Total non-financial assets | 144,385 | 209,562 | (65,177) |
| Departmental net financial position | (616,653) | (408,002) | (208,651) |
Additional information
Corporate information
Organizational profile
Appropriate minister: The Honourable Patty Hajdu
Institutional head: Dr. Mitchell Levine, Chairperson
Ministerial portfolio: Health
Enabling instruments: Patent ActFootnote xii and Patented Medicines RegulationsFootnote xiii
Year of incorporation / commencement: 1987
Other: The Minister of Health is responsible for the pharmaceutical provisions of the Patent Act set out in sections 79 to 103. Although the PMPRB is part of the Health Portfolio, because of its quasi-judicial responsibilities the PMPRB carries out its mandate at arm’s length from the Minister. It also operates independently of Health Canada, which approves drugs for safety, efficacy and quality; other Health Portfolio members, such as the Public Health Agency of Canada, the Canadian Institutes of Health Research and the Canadian Food Inspection Agency; and federal, provincial and territorial (F/P/T) public drug plans, which approve the listing of drugs for their respective formularies for reimbursement purposes; and the Common Drug Review, administered by the Canadian Agency for Drugs and Technologies in Health (CADTH), which recommends drugs that should qualify for reimbursement purposes by participating public drug plans.
Raison d’être, mandate and role: who we are and what we do
“Raison d’être, mandate and role: who we are and what we do” is available on the PMPRB’s websiteFootnote xiv.
For more information on the department’s organizational mandate letter commitments, see the Minister’s mandate letterFootnote xv .
Reporting framework
The PMPRB’s Departmental Results Framework and Program Inventory of record for 2019–20 are shown below.
| Departmental Results Framework | Core Responsibility: Regulate Patented Medicine Prices | Internal Services | |
| Departmental Result: Affordable patented drug prices | Indicator 1: % of patented drug prices in Canada are below the median price of the PMPRB’s comparator countries | ||
| Indicator 2: % of patented drug prices in Canada within the thresholds set out in the Guidelines | |||
| Program Inventory | Patented Medicine Price Regulation Program | ||
| Pharmaceutical Trends Program | |||
Supporting information on the program inventory
Financial, human resources and performance information for the PMPRB’s Program Inventory is available in GC InfoBase.Footnote xvi
Supplementary information tables
The following supplementary information tables are available on the PMPRB’s website:
- Departmental Sustainable Development Strategy
- Gender-based analysis plus
Federal tax expenditures
The tax system can be used to achieve public policy objectives through the application of special measures such as low tax rates, exemptions, deductions, deferrals and credits. The Department of Finance Canada publishes cost estimates and projections for these measures each year in the Report on Federal Tax Expenditures.Footnote xvii This report also provides detailed background information on tax expenditures, including descriptions, objectives, historical information and references to related federal spending programs. The tax measures presented in this report are the responsibility of the Minister of Finance.
Organizational contact information
The Patented Medicine Prices Review Board
Box L40
Standard Life Centre
333 Laurier Avenue West
Suite 1400
Ottawa, Ontario K1P 1C1
Toll-free no.: 1-877-861-2350
Facsimile: (613) 288-9643
TTY: (613) 288-9654
Email: PMPRB.Information-Renseignements.CEPMB@pmprb-cepmb.gc.ca
Appendix: definitions
appropriation (crédit)
Any authority of Parliament to pay money out of the Consolidated Revenue Fund.
budgetary expenditures (dépenses budgétaires)
Operating and capital expenditures; transfer payments to other levels of government, organizations or individuals; and payments to Crown corporations.
core responsibility (responsabilité essentielle)
An enduring function or role performed by a department. The intentions of the department with respect to a core responsibility are reflected in one or more related departmental results that the department seeks to contribute to or influence.
Departmental Plan (plan ministériel)
A report on the plans and expected performance of an appropriated department over a 3-year period. Departmental Plans are usually tabled in Parliament each spring.
departmental priority (priorité)
A plan or project that a department has chosen to focus and report on during the planning period. Priorities represent the things that are most important or what must be done first to support the achievement of the desired departmental results.
departmental result (résultat ministériel)
A consequence or outcome that a department seeks to achieve. A departmental result is often outside departments’ immediate control, but it should be influenced by program-level outcomes.
departmental result indicator (indicateur de résultat ministériel)
A quantitative measure of progress on a departmental result.
departmental results framework (cadre ministériel des résultats)
A framework that connects the department’s core responsibilities to its departmental results and departmental result indicators.
Departmental Results Report (rapport sur les résultats ministériels)
A report on a department’s actual accomplishments against the plans, priorities and expected results set out in the corresponding Departmental Plan.
experimentation (expérimentation)
The conducting of activities that seek to first explore, then test and compare the effects and impacts of policies and interventions in order to inform evidence-based decision-making, and improve outcomes for Canadians, by learning what works, for whom and in what circumstances. Experimentation is related to, but distinct from innovation (the trying of new things), because it involves a rigorous comparison of results. For example, using a new website to communicate with Canadians can be an innovation; systematically testing the new website against existing outreach tools or an old website to see which one leads to more engagement, is experimentation.
full-time equivalent (équivalent temps plein)
A measure of the extent to which an employee represents a full person-year charge against a departmental budget. For a particular position, the full-time equivalent figure is the ratio of number of hours the person actually works divided by the standard number of hours set out in the person’s collective agreement.
gender-based analysis plus (GBA+) (analyse comparative entre les sexes plus [ACS+])
An analytical process used to assess how diverse groups of women, men and gender-diverse people experience policies, programs and services based on multiple factors including race ethnicity, religion, age, and mental or physical disability.
government-wide priorities (priorités pangouvernementales)
For the purpose of the 2019–20 Departmental Results Report, those high-level themes outlining the government’s agenda in the 2019 Speech from the Throne, namely: Fighting climate change; Strengthening the Middle Class; Walking the road of reconciliation; Keeping Canadians safe and healthy; and Positioning Canada for success in an uncertain world.
horizontal initiative (initiative horizontale)
An initiative where two or more federal organizations are given funding to pursue a shared outcome, often linked to a government priority.
non-budgetary expenditures (dépenses non budgétaires)
Net outlays and receipts related to loans, investments and advances, which change the composition of the financial assets of the Government of Canada.
performance (rendement)
What an organization did with its resources to achieve its results, how well those results compare to what the organization intended to achieve, and how well lessons learned have been identified.
performance indicator (indicateur de rendement)
A qualitative or quantitative means of measuring an output or outcome, with the intention of gauging the performance of an organization, program, policy or initiative respecting expected results.
performance reporting (production de rapports sur le rendement)
The process of communicating evidence-based performance information. Performance reporting supports decision making, accountability and transparency.
plan (plan)
The articulation of strategic choices, which provides information on how an organization intends to achieve its priorities and associated results. Generally, a plan will explain the logic behind the strategies chosen and tend to focus on actions that lead to the expected result.
planned spending (dépenses prévues)
For Departmental Plans and Departmental Results Reports, planned spending refers to those amounts presented in Main Estimates.
A department is expected to be aware of the authorities that it has sought and received. The determination of planned spending is a departmental responsibility, and departments must be able to defend the expenditure and accrual numbers presented in their Departmental Plans and Departmental Results Reports.
program (programme)
Individual or groups of services, activities or combinations thereof that are managed together within the department and focus on a specific set of outputs, outcomes or service levels.
program inventory (répertoire des programmes)
Identifies all the department’s programs and describes how resources are organized to contribute to the department’s core responsibilities and results.
result (résultat)
A consequence attributed, in part, to an organization, policy, program or initiative. Results are not within the control of a single organization, policy, program or initiative; instead they are within the area of the organization’s influence.
statutory expenditures (dépenses législatives)
Expenditures that Parliament has approved through legislation other than appropriation acts. The legislation sets out the purpose of the expenditures and the terms and conditions under which they may be made.
target (cible)
A measurable performance or success level that an organization, program or initiative plans to achieve within a specified time period. Targets can be either quantitative or qualitative.
voted expenditures (dépenses votées)
Expenditures that Parliament approves annually through an appropriation act. The vote wording becomes the governing conditions under which these expenditures may be made.