ARCHIVED - Point-of-Care HIV Testing Using Rapid HIV Test Kits: Guidance for Health-Care Professionals
Volume: 33S2 - November 2007
Supplement
For readers interested in the PDF version, the document is available for downloading or viewing:
Table of Contents
- Introduction
- Legal, Ethical, and Human Rights Considerations
- Standard versus Rapid HIV Testing
- Challenges in Providing Rapid HIV Testing in the POC Setting
- Rapid HIV Test Kits
- Rapid HIV Tests and Seroconversion
- HIV Epidemiology and Public Health Considerations
- Post-marketing Surveillance Activities and Reporting Problems with Rapid HIV Test Kits
- Acknowledgements
- References
- Sources
- Figures and Tables
- Figure 1. HIV Testing in the Laboratory (Standard) Setting versus POC Setting
- Table 1. Adapting HIV Counselling for POC HIV Testing
- Table 2. Checklist for POC HIV Testing Using Rapid HIV Test Kits
- Table 3. Implementing a Quality Assurance Program and Quality Control Practices
- Table 4. Sources for Laboratory Expert Advice and Consultation on HIV Testing
- Box 1: CMA on consent
- Box 2: Some provincial positions on consent
Key Messages
Point-of-Care (POC) HIV testing refers to the practice, undertaken by health care professionals (HCPs), of providing:
- Pre- and post-test HIV counselling, modified to suit POC settings*; and
- HIV testing using rapid HIV tests in the POC setting.
*Note that POC setting refers to settings where HIV testing is offered at the point of care but where the test is performed outside of a designated laboratory.
POC rapid HIV testing needs to be placed within a careful analysis of legal, ethical, and human rights issues.
In addition, those HCPs, approved to perform HIV testing as defined within each province or territory, who are contemplating POC HIV testing using rapid HIV test kits, should be aware of the following:
- They must adapt the administration of pre- and post-test counselling in the POC setting recognizing the benefits and limitations of this test.
- As with all HIV testing, the use of the rapid test requires specific, informed consent from the person being tested.
- In general, a negative result is accurate and immediately available.
- False negative results can occur when the sample is obtained during the 'window period' (seroconversion).
- False reactive results can be experienced with this type of test in a low-prevalence setting like Canada.
- It is recommended that the term positive should not be used when discussing results from this test with the patient. Instead, the term reactive or 'the test result is preliminary positive for HIV antibodies' should be used.
- All reactive results obtained by a rapid HIV test require confirmatory testing at an approved HIV testing laboratory.
1. Introduction
In October 2005, Health Canada licenced a rapid HIV test for use by HCPs in the POC setting. One requirement for the use of these test kits in the POC setting is that HIV counselling, such as that described in the Canadian Medical Association's 1995 publication Counselling Guidelines for HIV Testing (new guidelines to be published in 2008).
This document replaces the previous version published in 2000 and provides updated clinical and technical information, as well as links to additional information that will help Canadian HCPs implement POC HIV testing with the use of rapid HIV test kits. The HCPs are encouraged to seek additional information by consulting resources/websites found in Section 10 of this document.
2. Legal, Ethical, and Human Rights Considerations
POC rapid HIV testing should be carefully considered within the framework of legal, ethical, and human rights concerns. The Federal Initiative to Address HIV/AIDS in Canada and Leading Together: Canada Takes Action on HIV/AIDS (2005-2010) frame the response to HIV/AIDS in a rights-based approach. Further analysis of legal, ethical, and human rights considerations will be provided in a policy framework to address HIV testing in Canada to be published by the Public Health Agency of Canada (PHAC) in 2008.
3. Standard versus Rapid HIV Testing
Standard HIV testing is performed in designated laboratories and consists of the following three steps (Figure 1):
- A serum or plasma sample is tested on an enzyme immunosorbent assay (EIA);
- If the initial test is reactive, then the sample is retested in duplicate on the same or a different EIA; and
- Confirmatory HIV testing with a Western Blot or other approved confirmatory HIV testing protocol is performed on samples that have remained reactive in Step 2.
Rapid POC HIV testing is performed at a POC setting outside the laboratory. Rapid HIV test kits are designed for single use only to test for HIV antibodies (similar to step 1 in Standard HIV testing). Currently, licenced rapid HIV tests in Canada use either whole blood or serum as the specimen type. HCPs should be aware that rapid HIV test kits may be designed to detect either HIV-1 or HIV-2 antibodies only, or both. Any claims made by a manufacturer are verified by Health Canada through a licencing process. Confirmatory HIV testing at an approved HIV testing laboratory is required for all patients with an HIV reactive result using a rapid HIV test. Parallel testing for non-reactive HIV tests may also be appropriate as part of quality assurance procedures, when required by a specific provincial/territorial policy.
4. Challenges in Providing Rapid HIV Testing in the POC Setting
Challenges include, but are not limited to, the following:
-
POC HIV testing requires that pre- and post-test counselling be modified from the usual HIV counselling that accompanies standard HIV testing (Table 1). This results from the immediate availability of test results. Note that pre- and post-test HIV counselling by trained HCPs remain equally critical components in either the standard HIV or rapid HIV testing scenarios. It is noted, however, that certain clinical situations may make detailed pre-test counselling difficult, e.g. rapid testing for pregnant women in labour. In these situations, informed consent for testing is a minimum requirement.
Pre- and post-test counselling has implications beyond legal and human rights. It is a delicate point in clinical relations with the test subject, and presents ethical issues that the HCP must consider to fully discharge professional duties.
Informed consent is a process of communication that enables a person to make a reasonable and informed decision. Consent is always contextual. Capacity to give fully informed consent implies the ability to understand the nature, benefits and consequences of a proposed health service and potential diagnosis. To be truly informed, consent requires full awareness of all relevant information - and to be true counselling, the process requires substantively more than mere delivery of information, important as this may be. The Canadian Medical Association has taken a position on consent, and has provided directives to medical practitioners (see Box 1). Some provinces have directions on informed consent. Please refer to Table 4 to obtain further information.
Pre-test counselling is critical in preparing patients for the implications of the test, and in cases of reactive test results, ensuring that they return for confirmatory test results.
Post-test counselling has obvious implications whether or not the result is reactive. In the case of a non-reactive test result, the counselling session provides the opportunity for an exchange of information on the individual's perceived risk and co-factors, and on harm reduction and prevention.
With reactive test results, the implications are just as great in terms of reduction in risk of transmission, provision of health care information, and referral to community services.
However, the HCP, in a post-test counselling situation with a patient with reactive test results now also has the responsibility to provide information beyond the usual public health contexts on matters, such as harm reduction, prevention, and partner notification.
Following the Supreme Court of Canada's decision in R vs. Cuerrier requiring individuals who know they are HIV positive to disclose their HIV status to sexual partners before engaging in conduct that poses a significant risk of serious bodily harm, it is recommended that HCPs inform a newly diagnosed HIV+ individual of the possibility of criminal charges arising from failure to disclose and/or exposure of another to HIV, even in cases where actual transmission does not occur.
-
In contrast to the situation in standard HIV testing, the HCP in the POC setting assumes responsibility for both specimen collection and testing (Table 3). Adequate resources, appropriate training and the implementation of quality assurance practices will be critical in ensuring the proper administration of the test and the correct interpretation of the test result. Further information concerning quality assurance practices is available in the package insert accompanying the test kits.
5. Rapid HIV Test Kits
-
Rapid HIV test kits must be licenced by Health Canada before they can be offered for sale in Canada. Manufacturers are required to provide data to Health Canada demonstrating that the kits can be used safely and effectively by the intended users. Health Canada requires that these kits only be used in settings where pre- and post-test HIV counselling is available. Any use of the kit for other than the approved and intended use could be viewed as a breach of ethical conduct by the HCP, who may be subject to a complaint to the provincial or territorial licencing body and liable for legal action.
-
Performance Characteristics of Rapid HIV Test Kits
The rapid HIV test kit is designed for single use on one specimen. The package insert for the rapid test kit indicates the appropriate sample type (e.g., finger prick blood, saliva, etc.). A result is obtained rapidly, normally within minutes. Any rapid HIV test kit licenced by Health Canada for POC HIV testing will have similar sensitivity, specificity, and other performance characteristics to HIV diagnostic test kits licenced for laboratory use. Currently, available rapid HIV tests have a reported sensitivity and specificity of ≥ 99.0%. However, due to the possibility of both false negative and false positive results, it is recommended that all reactive tests are accompanied by confirmatory testing. Note that some jurisdictions may require standard/parallel testing to confirm some or all negative tests with standard/parallel testing.
-
Rapid HIV test kits from various manufacturers will differ in directions, contents, time-sensitive steps, read times, and configurations. HCPs will need to familiarize themselves with the directions, contents, specific processing steps, and configurations of the kit(s) they will be using.
In general, a specimen from the patient is collected as per appropriate sample type (e.g. fingerstick blood, saliva, etc. depending on the test kit). The sample may be mixed with specimen diluent prior to being added to the device, or alternatively, the sample may be added directly to the device. A short incubation may ensue and a detection reagent may be added.
When HIV antibodies are present in the patient's sample in sufficient quantity, a colour reaction will be observed along the test strip.
Rapid HIV test kits are designed to include an internal sample control to ensure the specimen was added and the assay procedure was followed properly. For a test result to be valid, a reaction must be observed with this control.
In addition, a manufacturer of a rapid HIV test kit must have external test controls (both positive and negative) available. These controls serve to ensure that the test has been performed properly and that the user is able to correctly interpret test results.
Patients with a negative rapid HIV test result
The rapid HIV test kit provides a reliable final test result that permits the HCP to complete HIV testing and counselling in a single visit. However, as for other HIV antibody tests, a small number of HIV-infected individuals will have a negative HIV rapid test result. Pre- and post-test counselling is critical to assess a person's risk of infection (Table 1).
Despite a negative rapid HIV test result, all patients who have had any HIV risk activity in the previous 3 months should be informed of the benefits of repeat HIV testing (refer to Section 6: Rapid HIV Tests and Seroconversion). In the case of other clinical circumstances, such as hepatitis C infection, symptoms or signs of an acute seroconversion illness, or any other unexplained clinical features in the context of HIV risk activity, the HCP may wish to consult with HIV experts (Table 4) regarding additional testing to detect early HIV infection and recommend repeat HIV testing for patients with an initial negative HIV test. In addition, in acute care settings (e.g., pregnant women in labour) with a non-reactive rapid HIV test, further management will be influenced by a risk assessment of the patient's situation to consider if she/he might be in the window period for HIV detection.
Patients with a (preliminary) positive test
A small number of people who are not HIV infected will produce a positive (reactive) result when tested with an HIV antibody test kit, including the rapid test. Because of this, all reactive test results must be confirmed using a laboratory-based confirmatory test. The importance for confirmatory testing at an approved HIV testing laboratory (see Figure 1) needs to be emphasized to rule out the possibility of a false-positive result in the rapid HIV test and to confirm a true positive result. In pre-test counselling, patients should be informed of, and consent to, a venous sample being sent to an approved HIV testing laboratory should their rapid HIV test result be positive, invalid or indeterminate. In addition, counselling should detail that immediate action will be taken in response to a preliminary positive test result in certain clinical situations (e.g., recipients in blood and body fluid exposures may be started on HIV post-exposure prophylaxis if the source tests reactive to HIV, or pregnant women in labour may be started on intravenous AZT, etc.).
Further Action Required by the HCP with a Preliminary Positive Result - The HCP should write the test result, the kit name, and lot number on the HIV testing requisition. The HIV testing laboratory can process the patient's sample accurately for HIV confirmatory testing when provided with this information. Similar detailed information on the kit and lot number should be written in the patient's medical record. HCPs may also wish to know the expected time of confirmed results so that they can provide appropriate information and schedule a follow-up appointment for their patient.
Patients with an invalid or indeterminate rapid HIV test result
In some cases, the rapid HIV test may produce an invalid test result. An invalid test result will occur if there was a problem with the sample or with the test device. The test can be repeated with a new sample and a new device. If the test result is again invalid, then a venous blood sample must be drawn and submitted to an approved laboratory for confirmatory testing (as per a preliminary positive result). Some rapid HIV test kits may also describe a situation where a test result is considered indeterminate. An indeterminate test result cannot be interpreted as positive (reactive) or negative. In the case of an indeterminate test result, a venous blood sample must be drawn and submitted to an approved laboratory for confirmatory testing (as per a preliminary positive result).
6. Rapid HIV Tests and Seroconversion
All HIV tests that rely on the presence of HIV-specific antibodies are less reliable for patients who are in the period of HIV seroconversion, often called the HIV window period. During the window period, the patient's immune response has not yet produced detectable levels of HIV-specific antibodies. HIV testing may not be reliable in patients who have had HIV-risk activity in the previous 3 months, consequently they should be counselled on the need for appropriate repeat HIV testing. Experts at HIV testing laboratories can provide guidance to the HCP, in addition to providing more comprehensive, specific HIV testing procedures for patients who may be in the HIV window period. HCPs may wish to provide specific clinical information on the HIV testing requisition that will alert laboratory experts to undertake a more complex panel of HIV tests for specimens from such patients. Details such as the date of HIV-risk activity or exposure and any clinical symptoms or signs suggestive of HIV seroconversion might be included.
7. HIV Epidemiology and Public Health Considerations
HIV infection is reportable under public health legislation in all provinces and territories. HCPs are required to report identified cases of HIV infection to the public health system; to assist in contact tracing and counselling of the patient; and to maintain accurate HIV epidemiologic information. Approved HIV testing laboratories ensure that cases of HIV infection are reported to the public health system.
In the rare event that a patient with a preliminary positive or invalid result using a rapid HIV test kit refuses to provide a venous blood sample for confirmatory HIV testing, the HCP must encourage and support the patient to undergo such testing.
If the patient still does not complete confirmatory HIV testing, the HCP should solicit advice from public health officials on provincial or territorial public health reporting requirements.
Data on negative test results can provide an indicator of testing volume and, to some extent, of testing patterns and behaviours. In addition, negative test results can be used to develop incidence and prevalence estimates for HIV. Reporting requirements for all types of positive and negative HIV tests differ from one province and territory to another.
8. Post-marketing Surveillance Activities and Reporting Problems with Rapid HIV Test Kits
In the event of kit failure, the HCP should report the problem to the manufacturer. The manufacturer is required to keep records of reported problems and is subject to mandatory problem reporting requirements under the Medical Devices Regulations. The HCP must also consider reporting the issue separately to the Health Products and Food Branch Inspectorate (HPFBI), Health Canada, at 1-800-267-9675.
9. Acknowledgements
PHAC acknowledges the assistance of the Federal/Provincial/Territorial Advisory Committee on HIV/AIDS; National Laboratory for HIV Reference Services, Surveillance and Risk Assessment Division, the Canadian Association for HIV Clinical Laboratory Specialists (CAHCLS) and the HIV/AIDS Policy, Coordination and Programs Division, Centre for Infectious Disease Prevention and Control, Public Health Agency of Canada; expert reviewers of this document; and the Medical Devices Bureau, Therapeutic Products Directorate, HPFB, Health Canada.
The CAHCLS has reviewed this document.
10. References
- http://www.cdc.gov/hiv/rapid_testing/ US Center for Disease Control website Specific guidelines found on this website include:
- Revised Guidelines for HIV Counselling, Testing and Referral (Nov. 2001)
- Rapid HIV Testing of Women in Labour and Delivery. The MIRIAD Study (June 2003)
- Rapid HIV Testing (Apr. 2005).
- http://www.bccdc.org/content.php?item=83: British Columbia Centre for Disease Control website guidelines for use of POC in BC. Guidelines for Use of the POC HIV Test Kit Information for the Health Care Professional in BC (August 2007) and Point-of-Care HIV Counselling Guidelines (May 2007).
- Ministère de la Santé et des Services sociaux. Guide québécois de dépistage - Infections transmissibles sexuellement et par le sang, Québec, ministère de la Santé et des Services sociaux, 2006, 149 p. http://www.msss.gouv.qc.ca/itss, menu « Documentation », section « Professionnels de la santé », onglet « Guides ».
- Rapid HIV-1 Antibody Testing during Labor and Delivery for Women of Unknown HIV Status: A Practical Guide and Model Protocol http://www.cdc.gov/hiv/topics/testing/resources/guidelines/rt-labor&delivery_ appendixA.htm
- http://www.health.state.ny.us/diseases/aids/testing/rapid/index.htm - General guidelines by the New York State Department of Health.
- http://www.who.int/diagnostics_laboratory/evaluations/hiv/en/ - World Health Organization, with several reports on the evaluation of many rapid HIV test kits, including HIV Simple/Rapid Assays: Operational Characteristics, Report 14, 2004.
- http://www.nastad.org/Docs/Public/Publication/2006216_RapidTestingPrimer.pdf - Authored the publication Implementing Rapid HIV Testing: A Primer for State Health Departments by NASTAD (Northern Alliance for State and Territorial AIDS Directors.
- http://www.aidsaction.org Authored the publication Safe, Fast and Reliable - A New Generation of HIV Testing, which serves as a general set of guidelines.
- http://www.aidslaw.ca - Canadian HIV/AIDS Legal Network authored the publication Rapid HIV Screening at the Point-of-Care: Legal and Ethical Questions, 2000. Also deals with several issues outside of the laboratory aspect of rapid HIV testing.
- Expert Working Group on HIV Testing: Counselling Guidelines. Counselling Guidelines for HIV Testing. Ottawa: Canadian Medical Association, 1995.
- R Elliott, R Jürgens. Rapid HIV screening at the point of care: Legal and ethical questions. URL: <www.aidslaw.ca>. Date of access: January 2000.
- Giles RE, Perry KR, Parry JV. Rapid test devices for anti-HIV screening: Do they come up to the mark? J Med Virol 1999;59:104-09.
- Kane B. Rapid testing for HIV: Why so fast? Ann Intern Med 1999;131:481-83.
- Ng KP, Saw TL, Baki A et al. Evalution of a rapid test for the detection of antibodies to human immunodeficiency virus type 1 and 2. Int J STD AIDS 1999;10:401-04.
- Shafran SD, Prasad E, Zaidi A et al. Evaluation of the Merlin immediate HIV-1 and -2 test for HIV antibody performed at point of care. In: Abstracts of the 7th Conference on Retroviruses and Opportunistic Infections. January 2000, San Francisco, California. Alexandria, Virginia: Foundation for Retrovirology and Human Health, 2000:49. Abstract 766.
11. Sources
This document is a revised version of the initial document published in 2000 by the following authors:
DL-P Kilby, MD, Ottawa, Ontario; CJ Major, BSc, MLT, Central Public Health Laboratory, Etobicoke, Ontario; MH Steben, MD, Montreal,Quebec; WD Sutherland, MD, Bureau of HIV/AIDS, STD and TB, Laboratory Centre for Disease Control, Health Protection Branch; GA Bally, MD, HIV/AIDS Policy, Coordination and Programs Division, Health Promotion and Programs Branch, Health Canada, Ottawa, Ontario.
12. Figures and Tables
Figure 1: HIV Testing in the Laboratory (Standard) Setting versus POC Setting
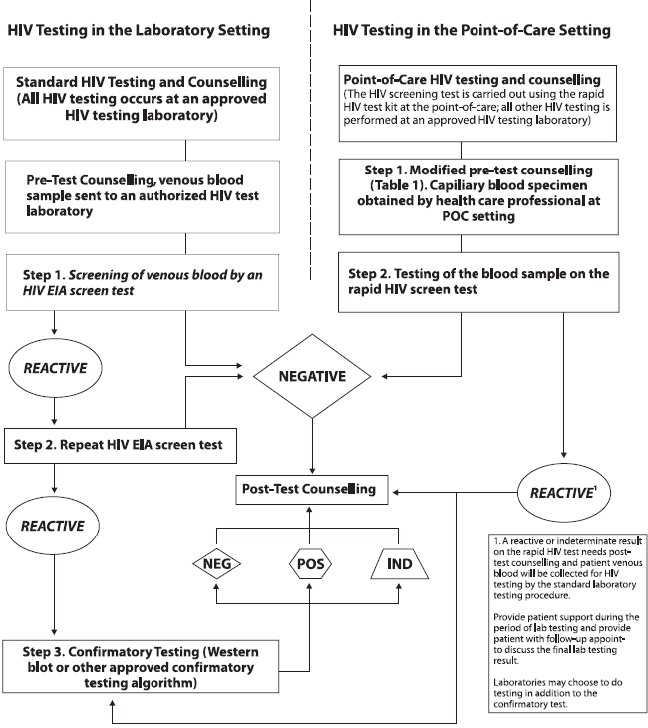
Figure 1, Text Equivalent
Figure 1: HIV Testing in the Laboratory (Standard) Setting versus POC Setting
HIV Testing - Standard: In the Laboratory Setting
STEP ONE:
Standard HIV testing is offered in healthcare venues such as clinics, physician offices, and hospitals. Pre-test counselling is provided to the patient and then a venous blood sample is collected and sent to an approved medical laboratory with a requisition ordering an HIV test.
STEP TWO:
A) The blood sample is screened using an enzyme immuno assay (EIA) (although some laboratories use a newer test known as chemiluminescent microparticle immuno assay). These tests will generate either a “reactive” or “non-reactive” (negative) finding. Test results can also be “indeterminate”, which are treated as “reactive” for the purpose of this algorithm. A reactive finding is not necessarily an HIV-positive diagnosis; a reactive finding requires that the blood sample be retested to rule out or confirm a positive HIV diagnosis.
B) Non-reactive findings are considered negative and test results are provided to the patient. No further action is required.
STEP THREE
Reactive and indeterminate samples from Step 2A are retested. Non-reactive results are treated the same as in Step 2B. A second reactive or indeterminate result requires the sample to go to Step 4.
STEP FOUR
Reactive and indeterminate samples undergo a more sensitive confirmatory test known as the Western Blot assay. As with previous steps, the western blot assay can result in a reactive, non-reactive or indeterminate result. Non-reactive results are addressed as described in Step 2B. Reactive or indeterminate results are addressed in Step Five.
STEP FIVE
Reactive (HIV-positive) and indeterminate test results are provided to the patient along with post-test counselling. Those testing indeterminate are encouraged to be re-tested.
HIV Testing - In the Point-of-Care Setting
STEP ONE:
Point of care testing is offered on-site in healthcare venues such as clinics, physician offices and hospitals. Test results are available in about one minute, so a combined pre- and post-test counselling session is provided. Most commonly, fingerstick blood or a saliva sample is used, depending on the test kit.
STEP TWO:
A) The blood or saliva sample is tested using the kit which will generate either a “reactive” or “non-reactive” (negative) finding. Test results can also be “indeterminate”, which are treated as “reactive” for the purpose of this algorithm. A reactive finding is not necessarily an HIV-positive diagnosis; a venous blood sample needs to be collected so that confirmatory testing can be done as described in Step Three.
B) Non-reactive findings are considered negative and test results are provided to the patient. No further action is required.
STEP THREE:
The reactive or indeterminate results are shared with the patient. A venous blood sample is obtained to rule out or confirm a positive HIV diagnosis.
STEP FOUR:
The venous blood sample undergoes a confirmatory test known as the Western Blot assay. Some labs may test the sample using the full standard protocol. As with previous steps, the western blot assay can result in a reactive, non-reactive or indeterminate result. Non-reactive results are addressed as described in Step 2B. Reactive or indeterminate results are addressed in Step Five.
STEP FIVE
Reactive (HIV-positive) and indeterminate test results are provided to the patient along with post-test counselling. Those testing indeterminate are encouraged to be re-tested.
HCPs using POC HIV testing will need to adapt their pre- and post-test counselling and their standard HIV testing and counselling procedures. Reference to the benefits of rapid HIV testing including quicker results, early access to health care, etc. are important factors that should be relayed to the client.
Pre-test counselling
- Inform patients of the possibility of false positive or false negative HIV test results, when using the rapid HIV tests (as with all HIV diagnostic tests).
- Stress the importance of laboratory HIV confirmatory testing of all preliminary positive, indeterminate or invalid results from a rapid HIV test. confirmatory HIV testing will require a venous blood sample, which will be sent to an approved HIV testing laboratory. Patients must understand that they are consenting to this further confirmatory HIV testing in the event of a preliminary positive rapid test result when they agree to HIV testing at the POC setting using a rapid test kit. In addition, in some jurisdictions, negative tests may be accompanied by standard parallel HIV testing.
- Inform patients of any costs related to POC HIV testing (if relevant), such as purchasing a rapid HIV test kit.
- Inform patients in situations where immediate medical care will be affected by test results, e.g. provide information on available antiretroviral medications.
Preliminary post-test counselling after a negative result from a rapid HIV test kit
Assess the risks that the patient undergoing testing may face, and provide supportive counselling and information, as appropriate. This will involve providing accurate information on risk behaviour, potential links to community resources, and rationale for behaviour changes needed to reduce risk.
Preliminary post-test counselling after a preliminary positive, indeterminate or invalid result from a rapid HIV test kit
- Provide continued counselling and support to the patient while a blood sample is being processed for confirmatory HIV testing at an approved HIV testing laboratory (often the provincial HIV laboratory).
- Arrange a specific follow-up appointment corresponding to the time required for the HIV testing laboratory to complete confirmatory HIV testing (ranging from several days to 2 weeks). Consult with experts at the HIV testing laboratory for the necessary information to include on the HIV requisition and the specific time required for results of confirmatory HIV testing.
- In clinical situations where immediate patient management will be influenced by positive test result (e.g. pregnant women in labour) inform/discuss with patient that actions(s) will be taken, e.g. starting antiretroviral medications, doing tests, etc. based on rapid/POC HIV test results.
- Inform patients about community resources or HIV/AIDS organizations that can provide psychosocial support.
Before Using Kits
- Contact experts at an approved HIV testing laboratory before carrying out the first rapid HIV test (Table 4).
- Find out the time for results of confirmatory HIV testing (usually varies between 3 and 14 days).
- Learn how to label and send a venous blood specimen from patients with a preliminary positive, indeterminate or invalid result from a rapid HIV test kit.
- Ask a public health or clinical HIV expert about the recommended repeat HIV testing protocol for patients reporting HIV-risk behaviour in the previous 3 months.
- Prepare yourself and your staff for a preliminary positive, indeterminate or invalid HIV test result.
- Ensure that the kit has not passed the expiration date and record the kit lot number.
Staff Education
- Use the package insert for directions. Each rapid HIV test kit will have different directions, contents, specific processing steps, and configurations.
- Establish a practice procedure for POC HIV testing that includes pre- and post-test counselling and ensures the patient's specific, informed consent to this HIV testing process.
- Work with staff to do practice runs when introducing POC HIV testing, and when changing the type of rapid HIV test kit.
- Establish quality control procedures, which include the use of positive and negative controls, recording and noting any changes outside of the normal range of the kit (e.g. temperature, humidity, etc.), kit lot#, new operator, etc.
- Ensure ongoing staff education, particularly for new staff members who are delegated to use rapid HIV test kits, and for all delegated staff when switching to a new rapid HIV test kit.
Counselling
- See Table 1.
Use of the Rapid HIV Test Kit
- Remember that each rapid HIV test kit is configured differently and has different processing instructions.
- Process the kit with care and do not leave it unattended. The kit is invalid if not read in a specified time (maximum read time ranges from minutes to 2 hours) after the sample and buffer have been added.
- Label the device of the kit with a non-nominal, unique patient identifier. Most devices need to be labeled using an indelible felt marker (laboratory marker).
- Ensure patient confidentiality, particularly when processing kits in multi-use areas.
- Match patient, patient specimen, and kit.
- Mark kit name and lot number in clinical record.
- In the event of kit failure, the HCP should report the problem to the manufacturer. The manufacturer is required to keep records of reported problems and is subject to mandatory problem reporting requirements in the Medical Devices Regulations. The HCP must also report the issue separately to the Health Products and Food Branch Inspectorate (HPFBI), Health Canada, at 1-800-267-9675.
Expiry Date, Storage, and Disposal
- Before use, verify that the rapid HIV kit has not exceeded its expiry date. All expired kits should be disposed of safely, and should not be discarded in regular office garbage. Lancets and other components in the kit can be dangerous.
- Use universal precautions during specimen collection, kit processing, and disposal.
- Remove or ink over patient information on the device or any other part of the kit.
Each POC setting will have unique challenges to address before rapid HIV testing can be offered. However there are several basic elements of quality assurance and quality control practices that can be implemented to ensure highquality HIV testing. These include the following:
- Identification of Responsibility (Who's in Charge?) - Identification of an individual who will be ultimately responsible for all issues surrounding the delivery of health services related to HIV rapid testing. This individual will be responsible for changes in the counselling procedures that are required, all personnel issues (see below), communications, etc.;
- Personnel issues - training, qualifications and competency.;
- Quality control procedures for pre-, actual and post-rapid HIV testing; and
- Use of internal and external controls.
There is no question that implementing these procedures will require the development of new documentation (forms) within each POC setting.
A. Sources for laboratory experts on HIV testing
Newfoundland Public Health Laboratory
The Leonard A. Miller Centre
St. John's, Newfoundland and Labrador
Dr. Sam Ratnam
(709) 777-6568
Department of Laboratory Medicine
Queen Elizabeth Hospital Inc.
Charlottetown, Prince Edward Island
Dr. Lou Abbott
(902) 894-2309
Department of Pathology and Laboratory Medicine
QEII Health Science Centre
Halifax, Nova Scotia
Dr. Todd Hatchette
(902) 473-6885
Microbiology Department
Dr. Everett Chalmers Hospital
Fredericton, New Brunswick
Dr. Jeanne Henry
(506) 452-5465
New Brunswick Provincial Laboratory for Viral Illnesses
Dr. Georges L. Dumont Regional Hospital
Moncton, New Brunswick
Dr. Richard Garceau
(506) 862-4820
Laboratoire de santé publique du Québec
Sainte-Anne-de-Bellevue (Québec)
Christiane Claessens
(514) 457-2070
Cadham Provincial Laboratory
Winnipeg, Manitoba
Dr. Magdy Dawood
(204) 945-7545
Saskatchewan Disease Control Laboratory
Regina, Saskatchewan
Dr. Paul Levett
(306) 787-3135
Provincial Laboratory for Public Health (Alberta)
Calgary, Alberta:
Dr. K. Fonseca, (403) 670-1203
Edmonton, Alberta:
Dr. Bonita Lee, (780) 407-3414
Provincial Laboratory, Virology Service
BC Centre for Disease Control
Vancouver, British Columbia
Dr. Mel Krajden
(604) 660-6044
University of British Columbia
Diagnostic Virology and Reference Laboratory
Vancouver, British Columbia
Dr. Chris Sherlock
(604) 806-8422
National Laboratory for HIV Reference Services
Public Health Agency of Canada
Ottawa, Ontario
Dr. John Kim
(613) 957-9666
HIV Laboratory, Central Public Health Laboratory
Etobicoke, Ontario
Carol Swantee
(416) 235-5727
For further current information and advice on counselling for HIV testing, please contact your respective provincial/territorial Health ministries.
Box 1: The Canadian Medical Association on consent
Extracted from CMA Code of Ethics (Update 2004).
Available online at http://www.cma.ca
Communication, Decision Making and Consent
21. Provide your patients with the information they need to make informed decisions about their medical care, and answer their questions to the best of your ability.
22. Make every reasonable effort to communicate with your patients in such a way that information exchanged is understood.
23. Recommend only those diagnostic and therapeutic services that you consider to be beneficial to your patient or to others. If a service is recommended for the benefit of others, as for example in matters of public health, inform your patient of this fact and proceed only with explicit informed consent or where required by law. (emphasis added)
24. Respect the right of a competent patient to accept or reject any medical care recommended. (emphasis added)
27. Ascertain wherever possible and recognize your patient's wishes about the initiation, continuation or cessation of life-sustaining treatment.
Box 2: Some provincial positions on consent
Provinces and territories, such as Ontario and Quebec, have enacted legislation governing consent.
1. Ontario's legislation, the Health Care Consent Act (HCCA) RSO 1990- ch 7, is an example.
Note: “Treatment” includes diagnostic testing for the purposes of interpretation of the HCCA. The HCCA provides, in part:
Elements of consent
1. (1) The following are the elements required for consent to treatment:
- The consent must relate to the treatment.
- The consent must be informed.
- The consent must be given voluntarily.
- The consent must not be obtained through misrepresentation or fraud. 1996, c. 2, Sched. A, s. 11 (1).
Informed consent
(2) A consent to treatment is informed if, before giving it,
- the person received the information about the matters set out in subsection (3) that a reasonable person in the same circumstances would require in order to make a decision about the treatment; and
- the person received responses to his or her requests for additional information about those matters. 1996, c. 2, Sched. A, s. 11 (2).
Same
(3) The matters referred to in subsection (2) are:
- The nature of the treatment.
- The expected benefits of the treatment.
- The material risks of the treatment.
- The material side effects of the treatment.
- Alternative courses of action.
- The likely consequences of not having the treatment. 1996, c. 2, Sched. A, s. 11 (3).
An electronic version of the HCCA
2. In Quebec: Civil code of Quebec, Law on the health services and the social services, codes of ethics of nurses and physicians ...
This information can be found in the "Guide québécois de depistage des ITSS pages 19-21, at "Documentation", section "Professionnels de la santé", /"Guides"
3. Other provincial and territorial jurisdictions may also have taken position on consent. Refer to table 4 to obtain specific provincial information.
Page details
- Date modified: