Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS) 2018: Integrated Findings
Download the PDF version, 3123 KB
To promote and protect the health of Canadians through leadership, partnership, innovation and action in public health, Public Health Agency of Canada.
Working towards the preservation of effective antimicrobials for humans and animals, Canadian Integrated Program for Antimicrobial Resistance Surveillance.
Également disponible en français sous le titre :
Programme intégré canadien de surveillance de la résistance aux antimicrobiens (PICRA) 2018 : Résultats intégrés
To obtain additional information, please contact:
Dolly Kambo
Executive assistant
Public Health Agency of Canada
370 Speedvale Avenue West, Guelph, ON N1H 7M7
Telephone: 519-826-2174
Fax: 519-826-2255
E-mail: phac.cipars-picra.aspc@canada.ca
This publication can be made available in alternative formats upon request.
©Her Majesty the Queen in Right of Canada, as represented by the Minister of Health, 2020
Publication date: December 2020
This publication may be reproduced for personal or internal use only without permission provided the source is fully acknowledged.
Cat.: HP2-4/2018E-2-1-PDF
ISSN: 978-0-660-36795-8
Pub.: 200321
Suggested Citation:
Government of Canada. Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS) 2018: Integrated Findings. Public Health Agency of Canada, Guelph, Ontario, 2020.
On this page
- Overview of CIPARS activities
- 2018 Key findings
- Integrated findings and discussion
- What's new for CIPARS in 2018
- Glossary
Overview of CIPARS activities
Figure 1. CIPARS brings together diverse sources of data in a robust and sound manner
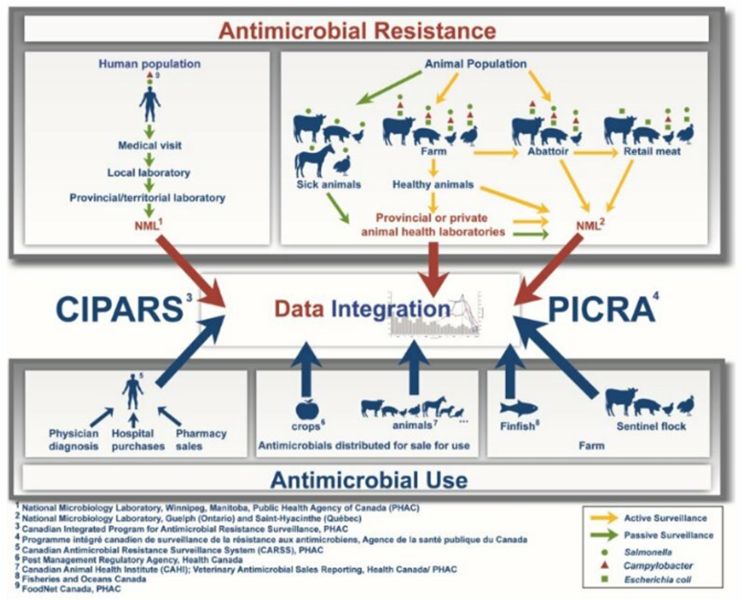
Figure 1 - Text Description
CIPARS brings together diverse sources of data in a robust and sound manner
This diagram represents all of the surveillance components that contribute to the Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS). The top half of the figure depicts all of the surveillance components related to antimicrobial resistance. On the left side, a component of passive surveillance (green arrows) associated with clinical samples of Salmonella (green circle) and Campylobacter (red triangle) of human origin is presented. These human samples are taken during the medical visit and sent to a local laboratory, then to a provincial/territorial laboratory, and finally to the National Microbiology Laboratory of Public Health Agency of Canada (PHAC) in Winnipeg. On the right side, there is a passive surveillance component (green arrows) and three active surveillance components (yellow arrows) from the Agri-Food sector. The passive surveillance component (green arrows) relies on clinical samples from sick animals (cattle, pigs, chickens, horses, and turkeys) that are submitted to provincial or private animal health laboratories for the isolation of Salmonella (green circle). Further on the right are presented the active surveillance components (yellow arrows) based on animal samples from the farm (cattle, pigs, chickens and turkeys), the abattoir (beef cattle, chickens, pigs) and also on retail meat samples (beef, pork, chicken and turkey). In general, samples from diseased animals are sent to the PHAC National Microbiology Laboratory in Guelph through provincial or private animal health laboratories. For other Agri-Food samples, most will be sent directly to the Saint-Hyacinthe National Microbiology Laboratory of the PHAC for the isolation of Salmonella (green circle), Campylobacter (red triangle), and Escherichia coli (green square).
The bottom of the diagram lists the surveillance components for antimicrobial use. On the left, three components related to the use of antimicrobials in humans and based on physician diagnostic data, hospital purchases and pharmacy sales data are shown. The data from humans are analyzed by the Canadian Antimicrobial Resistance Surveillance System of the PHAC and will be integrated with other CIPARS data. In the center, there is a component that monitors antimicrobials distributed for sale for use in crops. These data are processed by Health Canada's Pest Management Regulatory Agency and are also incorporated into some CIPARS data. There is also a component that monitors antimicrobials distributed to animals. This includes antimicrobials distributed for sale for use in production animals (cattle, pigs, chickens, turkey, etc.) and companion animals (dogs, cats, horses, etc.). The latter are processed by the Canadian Animal Health Institute and are sent to CIPARS for further integration. On the right, there are two surveillance components related to antimicrobial use on farm, the finfish antimicrobial use data (processed by Fisheries and Oceans Canada) and the sentinel flock antimicrobial use data (cattle, pigs, chickens and turkeys).
The central section of this diagram illustrates the collection of all data related to antimicrobial resistance and antimicrobial use within the CIPARS program. All data collected are integrated to figure out trend evolution and status of antimicrobial resistance and use in humans and the agri-food sector in Canada.
2018 key findings
Antimicrobial use
- Antimicrobial sales increased between 2017 and 2018.
- For broiler chickens, farm surveillance showed a reduction of use in 2018 compared to 2017 data, with some provincial variations.
- For pigs, AMU has significantly decreased overall and substantially in some provinces.
- For turkeys, AMU has increased overall with some provincial variations.
Antimicrobial resistance
Quinolone-resistant Salmonella Enteritidis
Quinolone (nalidixic acid) resistance in S. Enteritidis from agri-food sources is extremely rare, especially from chicken(s).
In 2018, nalidixic acid resistance in S. Enteritidis from chickens and chicken meat was detected at levels never observed before by CIPARS.
Highly drug resistant Salmonella
We observed the highest number of highly-resistant Salmonella isolates to-date across all human, animal, and food sources in 2018.
Additionally, we isolated highly-resistant Salmonella (serovar Infantis) from a chicken source for the first time.
Integrated antimicrobial use and resistance data
Chicken and people
The poultry industry initiative to eliminate the use of Category I antimicrobials (including the 3rd generation cephalosporins and fluoroquinolones) for disease prevention appears to reduce antimicrobial resistance in most scenarios.
Ceftiofur use and ceftriaxone resistance
- There has been no reported ceftiofur use in broiler chickens since 2015.
- In most scenarios, there has been a reduction in ceftriaxone (a 3rd generation cephalosporin) resistance in both E. coli and Salmonella recovered from chickens, and Salmonella isolates recovered from people.
- However, there was an increase in ceftriaxone-resistant Salmonella from chickens on farm.
Fluoroquinolone-resistant Campylobacter
- In 2018, fluoroquinolones were reported for treatment of sick chickens in a single flock in British Columbia.
- Similar to previous years, there were regional differences in the prevalence of fluoroquinolone-resistant Campylobacter from chicken and chicken meat.
- Resistance to ciprofloxacin (a fluoroquinolone) was more commonly identified in human Campylobacter isolates and retail chicken from British Columbia compared to Alberta and Ontario.
Integrated findings and discussion
Integrated antimicrobial use data
Antimicrobials are grouped into categories based on their importance to human medicine and the potential consequences of resistance to these drugs:
- Category I: Very high importance
Examples: cephalosporins (3rd and 4th generation), carbapenems, fluoroquinolones - Category II: High importance
Examples: macrolides, penicillins - Category III: Medium importance
Examples: tetracyclines - Category IV: Low importance
Examples: ionophores, flavophospholipids.
Medically important antimicrobials include Category I to III. Antimicrobials of low importance (Category IV, with the exception of flavophospholipids) were removed from the integrated AMU reporting. Data will be available in other CIPARS products.
Note: chemical coccidiostats are considered uncategorized antimicrobials.
For reporting data on antimicrobials used in animals, we use different metrics or ways of reporting the information.
Why do we use different metrics?
- There are several different ways to collect, analyze, and report antimicrobial use data.
- No single approach is appropriate for all purposes. Certain metrics are better suited to looking at trends over time, while others may be more appropriate for comparing different regions or different host species, and others may be better for understanding relationships between use and resistance.
Comparing humans, animals, and crops
Figure 2. Human and animal population estimates with total kilograms of antimicrobials distributed and/or sold in 2018
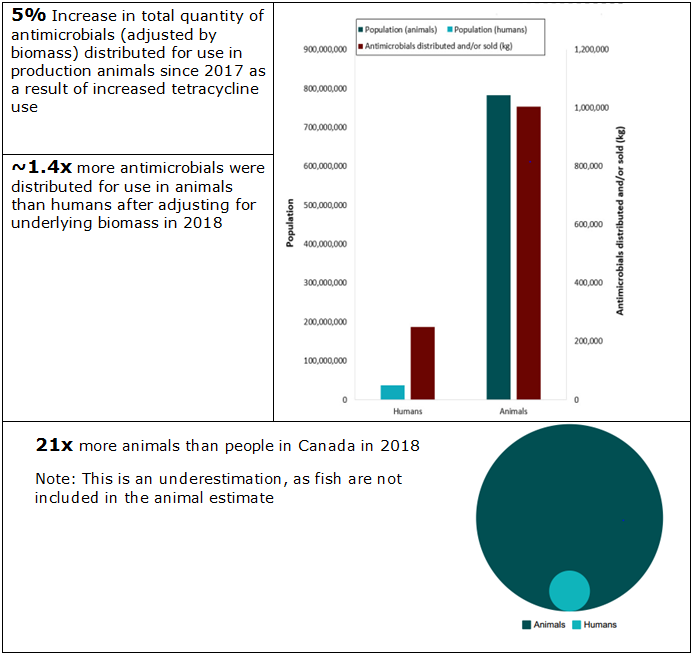
Figure 2. Human and animal population estimates with total kilograms of antimicrobials distributed and/or sold in 2018 - Text Description
| Population | Estimated number | Antimicrobials distributed and/or sold (kg) | Ratio |
|---|---|---|---|
| Humans | 37,058,856 | 1 (4.55 %) | 270,635 |
| Animals | 782,985,707 | 21 (95.45 %) | 1,004,392 |
Of the antimicrobials distributed or sold* in 2018:
- 78% were intended for production animals
- 21% were intended for humans
- 1% were intended for companion animals
- <1% were intended for crops
* Animal distribution data currently do not account for quantities imported for own use, or as active pharmaceutical ingredients intended for further compounding; hence, these are underestimates of total quantities used.
For both humans and animals, the β-lactams (penicillins) were one of the main antimicrobial classes distributed/sold on a per kg of antimicrobial basis.
Similar antimicrobials were licensed for use in humans and animals; however, some antimicrobial classes were sold or distributed more for use in humans than animals and vice-versa.
The relative quantity of cephalosporins and fluoroquinolones (Category I) intended for use in people is higher compared to animals (about 7 times and 25 times higher for people, respectively).
Tetracyclines (Category III) are used predominantly in production animals.
Notes:
- Cephalosporins are β-lactam antimicrobials, but we are displaying them separately for visualization purposes.
- The percentages are based on total kilograms of active ingredients intended for use in that host species.
- Other antimicrobials for animals: avilamycin, bacitracins, bambermycin, chloramphenicol, chlorhexidine gluconate, florfenicol, fusidic acid, novobiocin, polymixin B, tiamulin, and virginiamycin.
- Other antimicrobials for humans: bacitracin, chloramphenicol, colistimethate, colistin, daptomycin, fidaxomicin, fosfomycin, fusidic acid, linezolid, methenamine hippurate, methenamine mandelate, metronidazole, nitrofurantoin, polymyxin b, quinupristin/dalfopristin, and vancomycin.
Figure 3. The proportions of total kilograms of antimicrobial classes distributed or sold in 2018 in humans, production animals, and companion animals
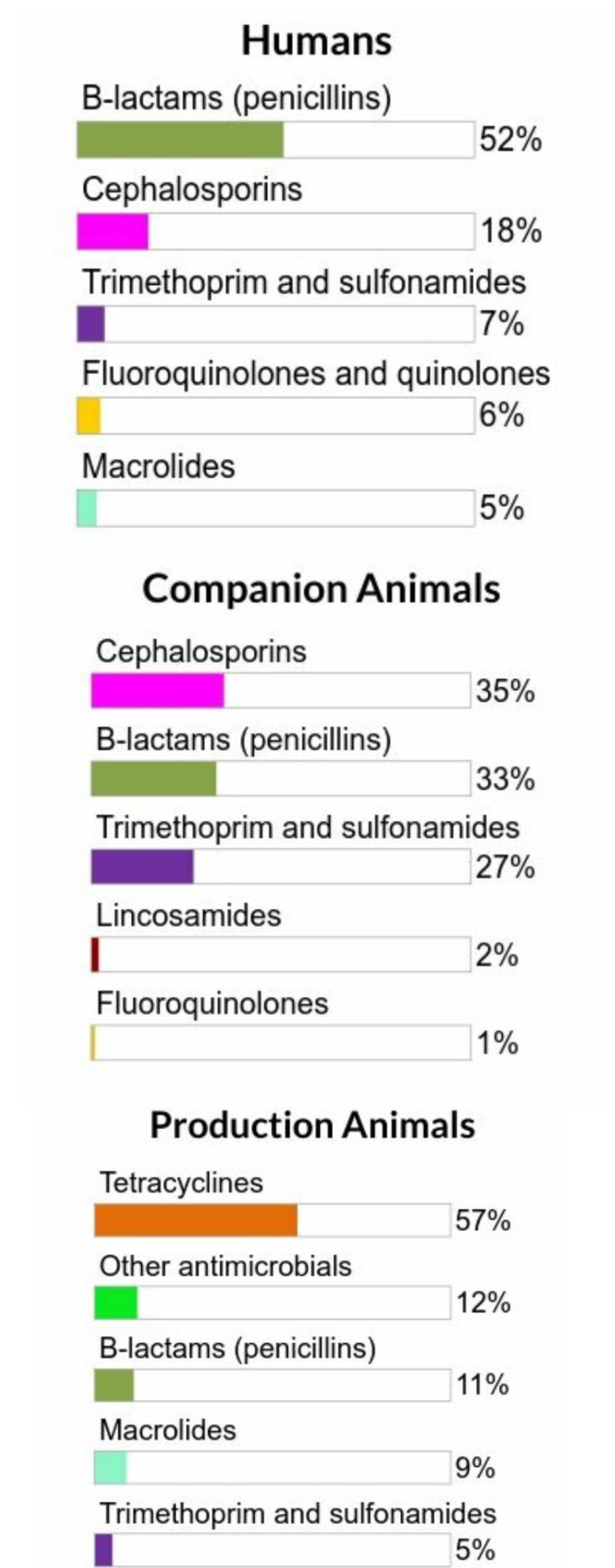
Figure 3 - Text Description
| Humans | Companion Animals | Production Animals | |||
|---|---|---|---|---|---|
| Antimicrobial Class | % | Antimicrobial Class | % | Antimicrobial Class | % |
| B-lactams (penicillins) | 52% | Cephalosporins | 35% | Tetracyclines | 57% |
| Cephalosporins | 18% | B-lactams (penicillins) | 33% | Other antimicrobials | 12% |
| Trimethoprim and sulfonamides | 7% | Timethoprim and sulfonamides | 27% | B-lactams (penicillins) | 11% |
| Fluoroquinolones and quinolones | 6% | Lincosamides | 2% | Macrolides | 9% |
| Macrolides | 5% | Fluoroquinolones | 1% | Trimethoprim and sulfonamides | 5% |
The total quantities of antimicrobials distributed for sale for use in production animals increased. When measured in kilograms, the total quantities distributed increased by 6% compared to 2017. When total quantities were adjusted for biomass (mg/PCU), the increase was 5% compared to 2017.
Figure 4. Quantities of antimicrobials distributed for use in animals
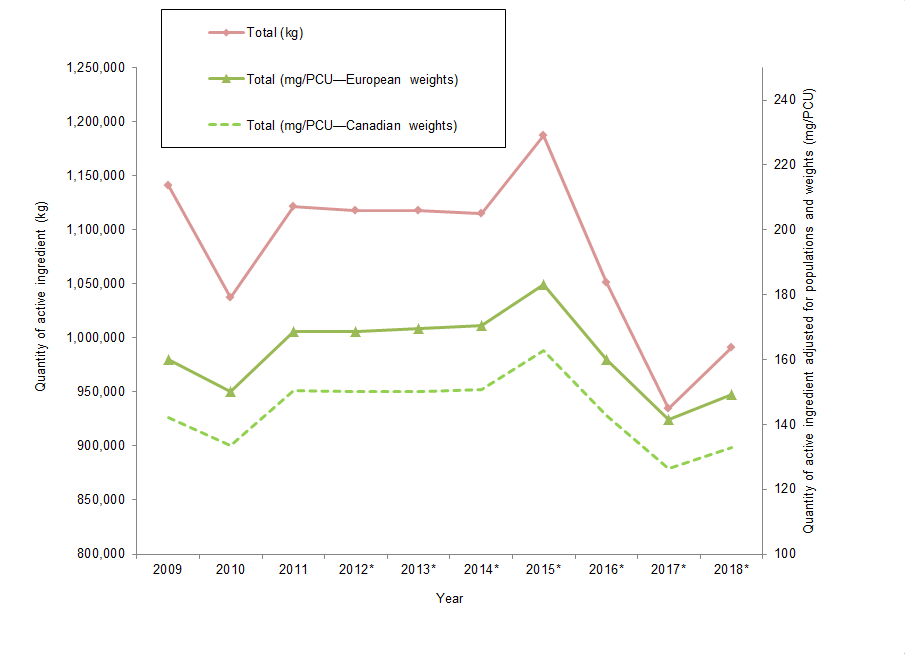
Figure 4 - Text Description
| Year | Total (kg) | Total (mg/PCU European weights) | Total (mg/PCU Canadian weights) |
|---|---|---|---|
| 2009 | 1,141,213 | 160 | 142 |
| 2010 | 1,037,313 | 150 | 133 |
| 2011 | 1,121,715 | 168 | 150 |
| 2012Figure 4 footnote * | 1,117,457 | 169 | 150 |
| 2013Figure 4 footnote * | 1,118,097 | 170 | 150 |
| 2014Figure 4 footnote * | 1,114,837 | 170 | 151 |
| 2015Figure 4 footnote * | 1,187,136 | 183 | 163 |
| 2016Figure 4 footnote * | 1,051,010 | 160 | 143 |
| 2017Figure 4 footnote * | 934,873 | 142 | 126 |
| 2018Figure 4 footnote * | 991,150 | 149 | 133 |
|
|||
Canada is the 6th highest country (in comparison to Europe) for quantities of antimicrobials sold (mg/PCU).
Figure 5. Quantities of antimicrobials used (mg/PCU) by Canada (2018) and countries participating in the European Surveillance of Veterinary Antimicrobial Consumption (ESVAC) network (2017)
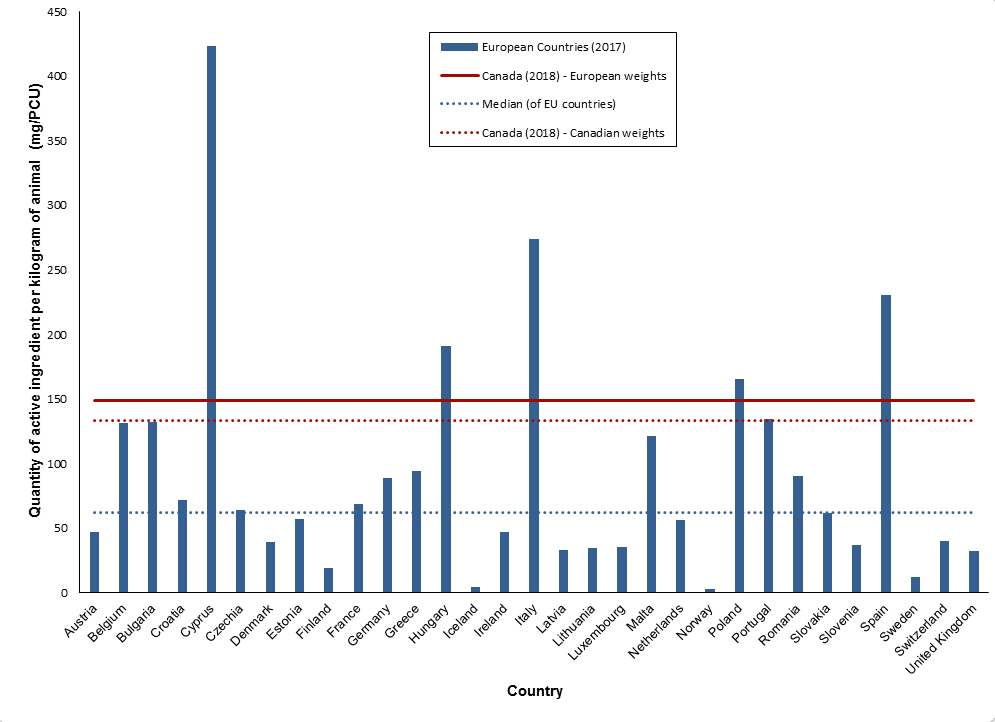
Figure 5 - Text Description
| European Countries (2017) | mg/PCU | Canada (2018) European weights | Median (of EU countries) | Canada (2018) Canadian weights |
|---|---|---|---|---|
| Austria | 47 | 149 | 62 | 133 |
| Belgium | 131 | 149 | 62 | 133 |
| Bulgaria | 132 | 149 | 62 | 133 |
| Croatia | 72 | 149 | 62 | 133 |
| Cyprus | 423 | 149 | 62 | 133 |
| Czech Republic | 64 | 149 | 62 | 133 |
| Denmark | 39 | 149 | 62 | 133 |
| Estonia | 57 | 149 | 62 | 133 |
| Finland | 19 | 149 | 62 | 133 |
| France | 69 | 149 | 62 | 133 |
| Germany | 89 | 149 | 62 | 133 |
| Greece | 94 | 149 | 62 | 133 |
| Hungary | 191 | 149 | 62 | 133 |
| Iceland | 5 | 149 | 62 | 133 |
| Ireland | 47 | 149 | 62 | 133 |
| Italy | 274 | 149 | 62 | 133 |
| Latvia | 33 | 149 | 62 | 133 |
| Lithuania | 35 | 149 | 62 | 133 |
| Luxembourg | 35 | 149 | 62 | 133 |
| Malta | 121 | 149 | 62 | 133 |
| Netherlands | 56 | 149 | 62 | 133 |
| Norway | 3 | 149 | 62 | 133 |
| Poland | 165 | 149 | 62 | 133 |
| Portugal | 135 | 149 | 62 | 133 |
| Romania | 90 | 149 | 62 | 133 |
| Slovakia | 62 | 149 | 62 | 133 |
| Slovenia | 37 | 149 | 62 | 133 |
| Spain | 230 | 149 | 62 | 133 |
| Sweden | 12 | 149 | 62 | 133 |
| Switzerland | 40 | 149 | 62 | 133 |
| United Kingdom | 33 | 149 | 62 | 133 |
*Weights used in the biomass calculation are European standard weights at treatment.
**This figure makes the assumption that the data are comparable between countries.
Data Sources: Canadian Animal Health Institute (CAHI), ESVAC, Fisheries and Oceans Canada, Health Canada's Pest Management Regulatory Agency, human pharmacy and hospital data from IQVIA via the Canadian Antimicrobial Resistance Surveillance System, Statistics Canada, Agriculture and Agri-Food Canada, and Equine Canada.
Comparing farm antimicrobial use data
Comparison of antimicrobial classes*
There are important differences in the types and relative quantities of antimicrobials reported for use between food animal species, which is why we need ongoing surveillance across the food animal species.
*The percentages are based on total kilograms of active ingredients intended for use in that host species.
The relative quantities of antimicrobial classes reported for use (mg/PCU) in broiler chickens, grower-finisher pigs, and turkeys in 2018
Figure 6.1 The relative quantities of antimicrobial classes reported for use (mg/PCU) in animals in 2018: broiler chickens
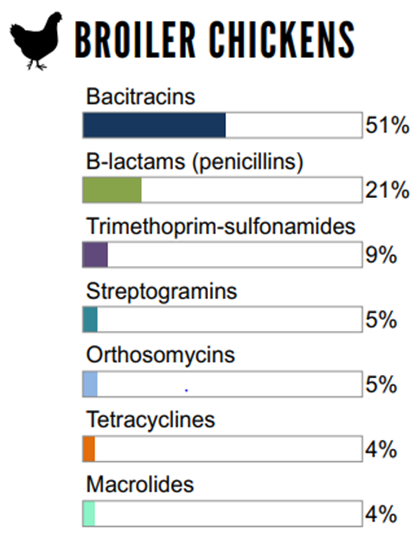
Not shown: aminoglycosides (1%), lincosamides-aminocyclitols (<1%).
Figure 6.2 The relative quantities of antimicrobial classes reported for use (mg/PCU) in animals in 2018: grower-finisher pigs
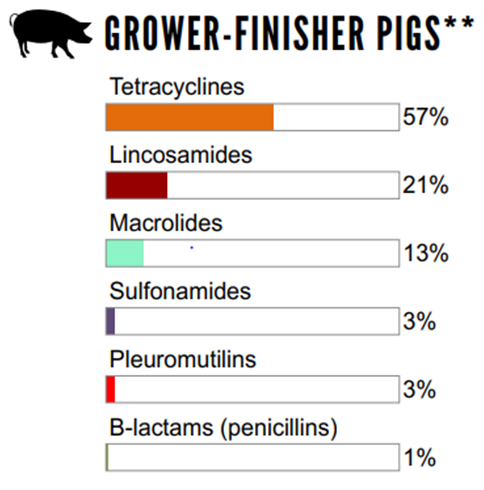
Not shown: streptogramins (1%)
**used in feed only.
Figure 6.3 The relative quantities of antimicrobial classes reported for use (mg/PCU) in animals in 2018: turkeys
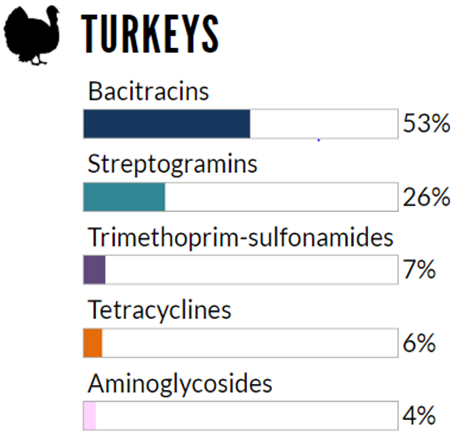
Not shown: B-lactams (penicillins) (3%), orthomycins (1%), fluoroquinolones (<1%).
Figure 6.1, 6.2, 6.3 - Text Description
| Broiler Chickens | Grower-Finisher Pigs | Turkeys | |||
|---|---|---|---|---|---|
| Antimicrobial Class | % | Antimicrobial Class | % | Antimicrobial Class | % |
| Bacitracins | 51% | Tetracyclines | 57% | Bacitracins | 53% |
| B-lactams (penicillins) | 21% | Lincosamides | 21% | Streptogramins | 26% |
| Trimethoprim and sulfonamides | 9% | Macrolides | 13% | Trimethoprim and sulfonamides | 7% |
| Streptogramins | 5% | Sulfonamides | 3% | Tetracyclines | 6% |
| Orthosomycins | 5% | Pleuromutilins | 3% | Aminoglycosides | 4% |
| Tetracyclines | 4% | B-lactams (penicillins) | 1% | no data | no data |
| Macrolides | 4% | no data | no data | no data | no data |
Temporal trends in antimicrobial use
Broiler chickens
Overall (nationally), farm surveillance showed a reduction in antimicrobial use in 2018 compared with 2017 data in broiler chickens. The two most commonly reported classes of antimicrobials used in broilers were bacitracins (Category III) and penicillins (Category II).
There were regional differences in the number of doses of antimicrobials administered. Compared to 2017:
- The Prairies and Ontario decreased their overall antimicrobial use (AMU).
- There was an increase in overall AMU in flocks from British Columbia and Québec.
Figure 7. Temporal trends in nDDDvetCA/1000 chicken-days at risk, 2013 to 2018
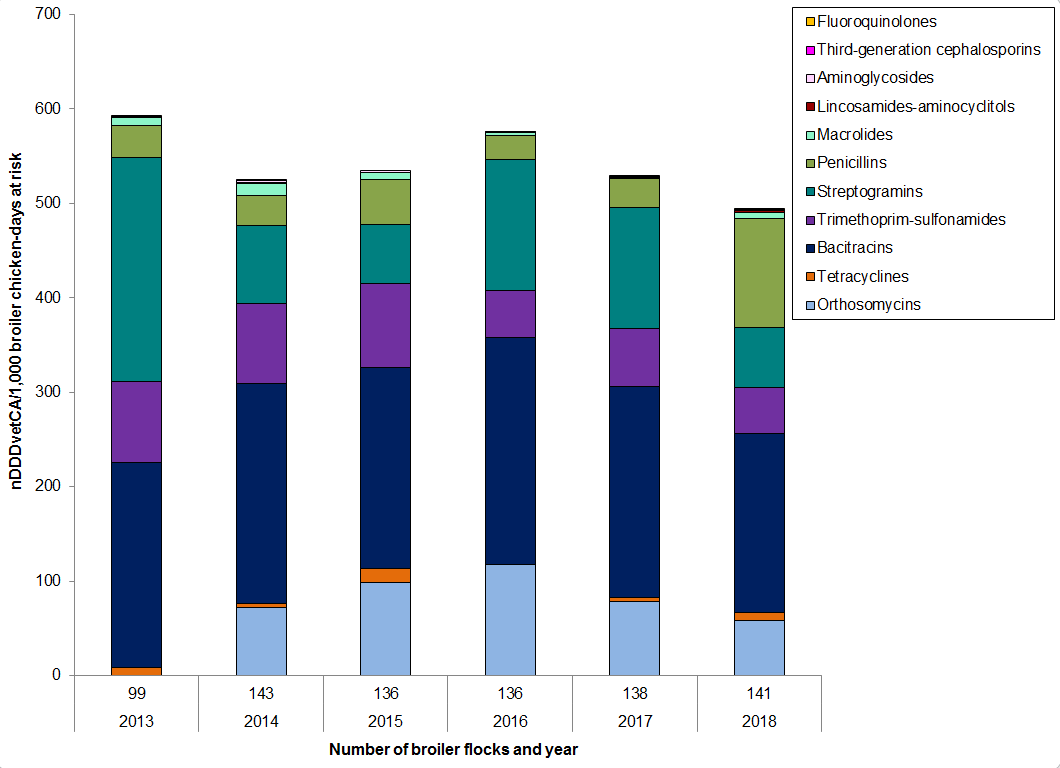
Figure 7 - Text Description
| Year | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | |
|---|---|---|---|---|---|---|---|
| Number of flocks | 99 | 143 | 136 | 136 | 138 | 141 | |
| Antimicrobial class | |||||||
| I | Fluoroquinolones | < 0.1 | 0 | 0 | 0 | 0 | < 0.1 |
| Third-generation cephalosporins | 1 | 0.1 | 0 | 0 | 0 | 0 | |
| II | Aminoglycosides | < 0.1 | 2 | 2 | 1 | 1 | 1 |
| Lincosamides-aminocyclitols | 1 | 1 | 1 | 0.5 | 0.5 | 2 | |
| Macrolides | 8 | 12 | 7 | 3 | 1 | 6 | |
| Penicillins | 34 | 33 | 47 | 25 | 31 | 116 | |
| Streptogramins | 237 | 83 | 63 | 139 | 128 | 63 | |
| Trimethoprim and sulfonamides | 85 | 85 | 89 | 50 | 61 | 49 | |
| III | Bacitracins | 217 | 232 | 213 | 239 | 224 | 190 |
| Tetracyclines | 9 | 4 | 15 | 1 | 4 | 8 | |
| N/A | Orthosomycins | 0 | 72 | 98 | 117 | 79 | 58 |
| Total | 591 | 524 | 535 | 576 | 529 | 493 | |
Figure 8. Temporal trends in nDDDvetCA/1000 chicken-days at risk, by province/region, 2014 to 2018
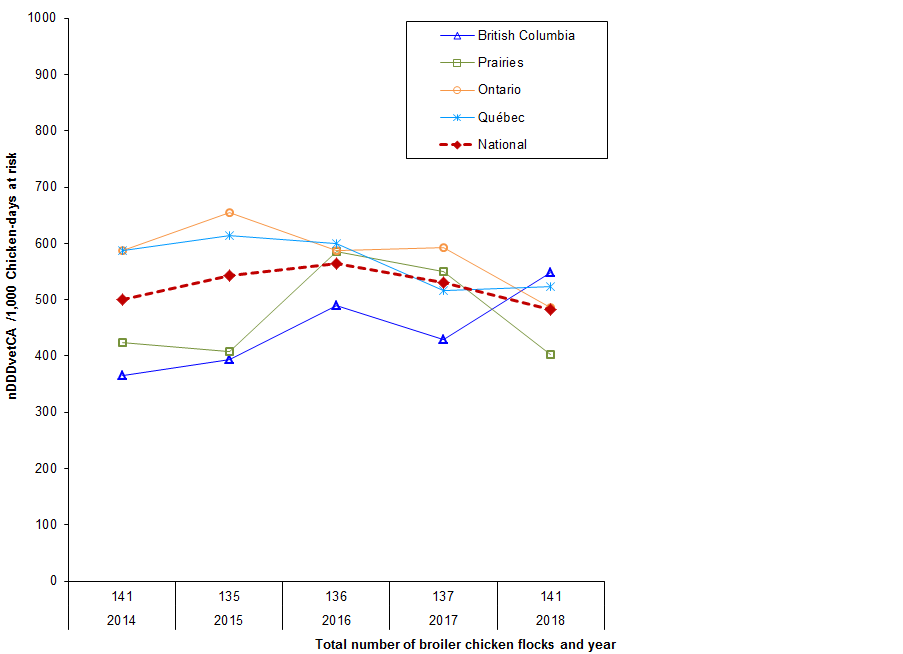
Figure 8 - Text Description
| Year | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|
| Number of broiler chicken flocks | 141 | 135 | 136 | 137 | 141 |
| Province/region | |||||
| British Columbia | 380 | 405 | 491 | 439 | 558 |
| Prairies | 450 | 419 | 595 | 568 | 410 |
| Ontario | 616 | 679 | 601 | 601 | 504 |
| Québec | 624 | 665 | 606 | 521 | 544 |
| National | 528 | 566 | 573 | 541 | 495 |
Grower-finisher pigs
Farm surveillance showed a decrease in antimicrobials used in feed (nDDDvetCA/1000 grower-finisher pig-days at risk) between 2014 and 2018. The most commonly reported classes of antimicrobials used in pigs included tetracyclines (Category III), lincosamides (Category II), and macrolides (Category II).
There were regional differences in the quantity and the number of doses of antimicrobials administered through feed over a grower-finisher feeding period. Compared to 2017:
- Québec and Ontario herds decreased their overall AMU in feed.
- There was an increase in overall AMU in feed in herds from the Prairies.
Figure 9. Temporal trends in nDDDvetCA/1000 grower-finisher pig-days at risk, 2009 to 2018
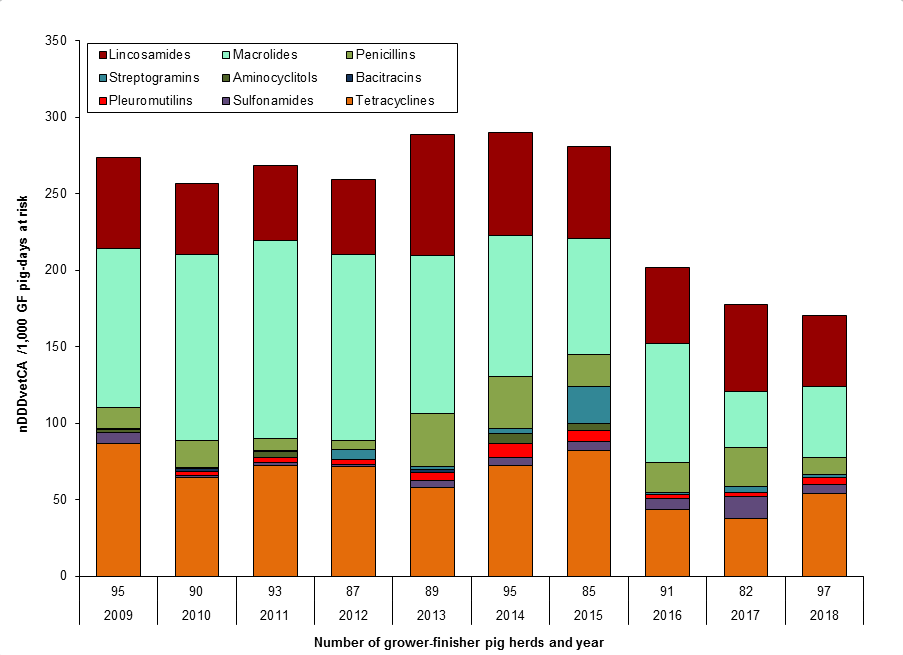
Figure 9 - Text Description
| Year | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Number of grower-finisher pig herds | 95 | 90 | 93 | 87 | 89 | 95 | 85 | 91 | 82 | 97 | |
| Antimicrobial class | |||||||||||
| II | Lincosamides | 59 | 47 | 49 | 49 | 79 | 68 | 60 | 49 | 57 | 46 |
| Macrolides | 104 | 122 | 129 | 122 | 103 | 92 | 76 | 78 | 37 | 47 | |
| Penicillins | 14 | 17 | 8 | 5 | 35 | 34 | 21 | 20 | 25 | 11 | |
| Streptogramins | 0 | 0 | 0 | 7 | 2 | 4 | 24 | 1 | 4 | 2 | |
| III | Aminocyclitols | 2 | 1 | 4 | 0 | 0 | 6 | 4 | 0 | 0 | 0 |
| Bacitracins | 0 | 2 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | |
| Pleuromutilins | 0 | 3 | 3 | 3 | 5 | 9 | 7 | 3 | 2 | 4 | |
| Sulfonamides | 7 | 2 | 2 | 1 | 5 | 5 | 5 | 7 | 15 | 6 | |
| Tetracyclines | 87 | 64 | 73 | 72 | 58 | 72 | 82 | 44 | 38 | 54 | |
| Total | 274 | 257 | 269 | 259 | 289 | 290 | 281 | 202 | 178 | 171 | |
Figure 10. Temporal trends in nDDDvetCA/1000 grower-finisher pig-days at risk for antimicrobials administered in feed, 2014 to 2018
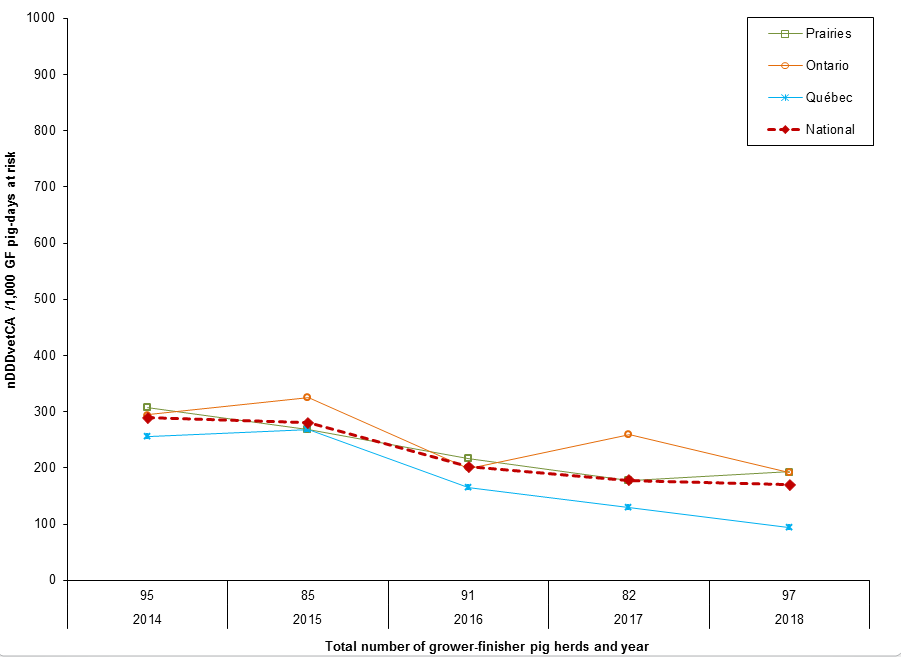
Figure 10 - Text Description
| Year | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|
| Number of grower-finisher pig herds | 95 | 85 | 91 | 82 | 97 |
| Province/region | |||||
| Prairies | 308 | 268 | 217 | 177 | 193 |
| Ontario | 294 | 325 | 200 | 259 | 192 |
| Québec | 255 | 268 | 164 | 130 | 94 |
| National | 290 | 281 | 202 | 178 | 171 |
Turkeys
In 2018, the overall reported antimicrobial use in turkeys increased. The top reported classes of antimicrobials used in turkeys included streptogramins (Category II) and bacitracins (Category III). Compared to 2017:
- Ontario and Québec flocks decreased their overall antimicrobial use (AMU).
- There was an increase in overall AMU in flocks from the British Columbia.
Figure 11. Temporal trends in DDDvet per 1000 turkey-days at risk in Canada, 2016 to 2018
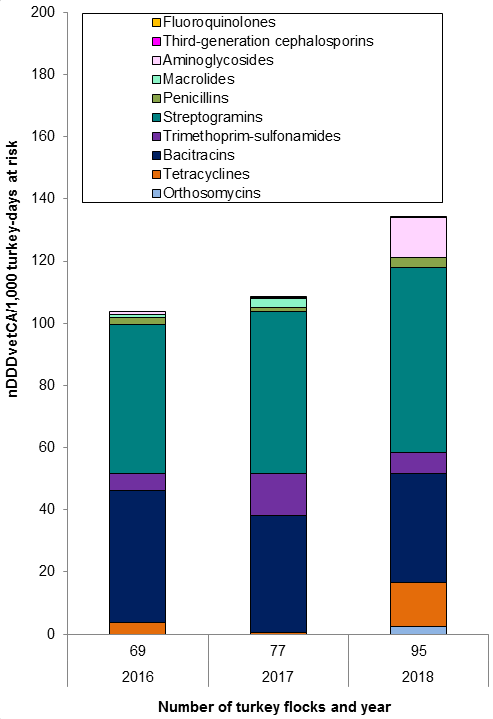
Figure 11 - Text Description
| Year | 2016 | 2017 | 2018 | |
|---|---|---|---|---|
| Number of turkey flocks | 69 | 77 | 95 | |
| Antimicrobial class | ||||
| I | Fluoroquinolones | 0 | < 0.1 | 0.1 |
| Third-generation cephalosporins | 0 | 0 | 0 | |
| II | Aminoglycosides | 1 | 0.3 | 13 |
| Macrolides | 1 | 3 | 0 | |
| Penicillins | 2 | 1 | 3 | |
| Streptogramins | 48 | 52 | 60 | |
| Trimethoprim and sulfonamides | 5 | 13 | 7 | |
| III | Bacitracins | 43 | 38 | 35 |
| Tetracyclines | 4 | 0.4 | 14 | |
| Not applicable | Orthosomycins | 0 | 0 | 2 |
| Total | 104 | 108 | 134 | |
Figure 12. Temporal trends in DDDvet per 1000 turkey-days at risk in Canada, by province/region, 2016 to 2018
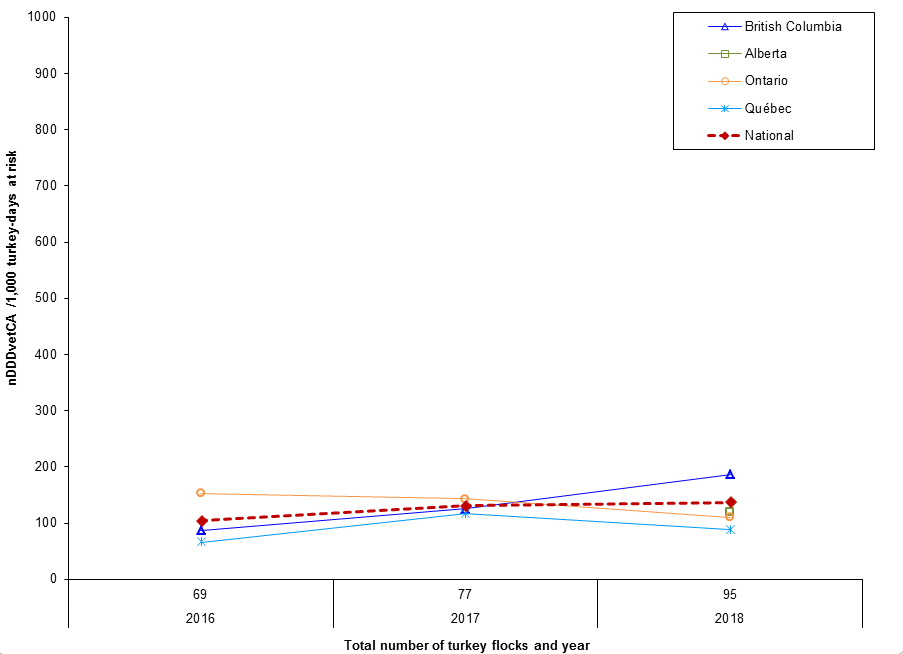
Figure 12 - Text Description
| Year | 2016 | 2017 | 2018 |
|---|---|---|---|
| Number of turkey flocks | 69 | 77 | 95 |
| Province | |||
| British Columbia | 86 | 125 | 186 |
| Alberta | Not applicable | Not applicable | 119 |
| Ontario | 152 | 143 | 110 |
| Québec | 66 | 117 | 89 |
| National | 104 | 131 | 137 |
Reasons for antimicrobial use
- In broiler chickens (Br. Chicken), turkeys, and grower-finisher pigs (G-F Pig), the predominant reason for administering antimicrobials was for disease prevention.
- In grower-finisher pigs, there continues to be reported use of antimicrobials for growth promotion.
Figure 13. Quantity of antimicrobials used (mg/PCU) by reason for use; CIPARS Farm 2014 to 2018
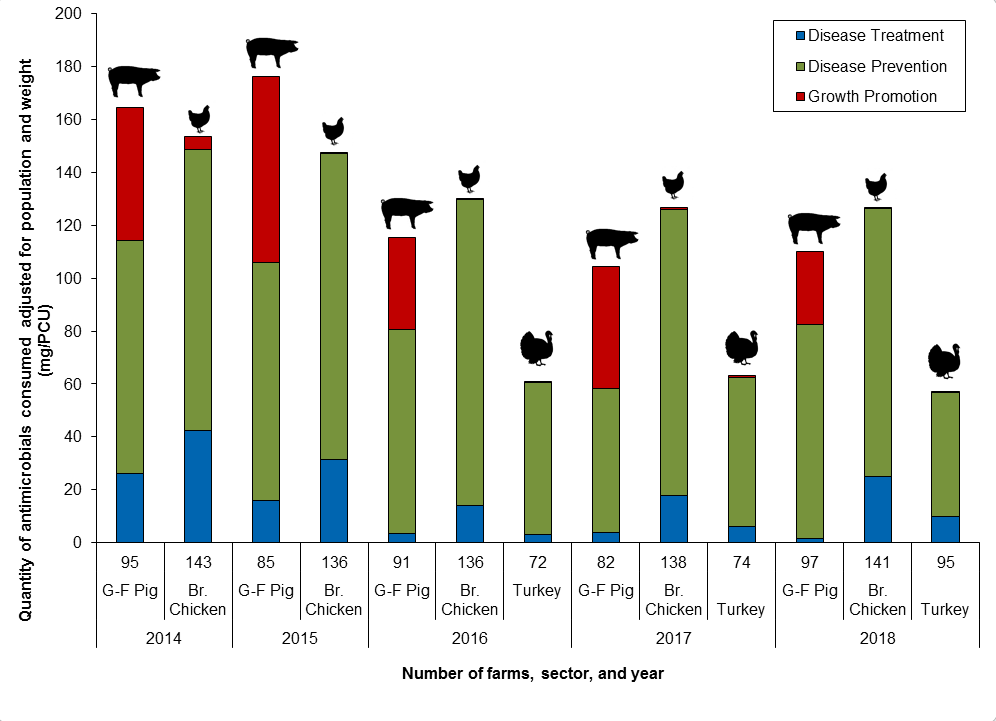
Figure 13 - Text Description
| Year | 2014 | 2015 | 2016 | 2017 | 2018 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sector | G-F Pig | Br. Chicken | G-F Pig | Br. Chicken | G-F Pig | Br. Chicken | Turkey | G-F Pig | Br. Chicken | Turkey | G-F Pig | Br. Chicken | Turkey |
| Number of Farms | 95 | 143 | 85 | 136 | 91 | 136 | 72 | 82 | 138 | 74 | 97 | 141 | 95 |
| Disease Treatment | 26 | 43 | 16 | 31 | 4 | 14 | 3 | 4 | 18 | 6 | 2 | 25 | 10 |
| Disease Prevention | 88 | 106 | 90 | 116 | 77 | 115 | 58 | 54 | 108 | 56 | 81 | 101 | 47 |
| Growth Promotion | 50 | 5 | 70 | 1 | 35 | 0 | 0 | 46 | 1 | 1 | 28 | 0 | 0 |
| Total | 164 | 153 | 176 | 148 | 116 | 130 | 61 | 104 | 127 | 63 | 110 | 126 | 57 |
Note: Swine data are for antimicrobial use in feed only; chicken and turkey data include all routes of administration.
Integrated antimicrobial resistance data
In this section, we highlight two resistance stories for 2018:
- The detection of quinolone-resistant Salmonella Enteritidis from chicken.
- The increase in highly drug-resistant Salmonella from human and agri-food sources.
In 2018, CIPARS tested for resistance to 7 classes of antimicrobials.
Although there is no international standard for defining highly resistant isolates, CIPARS considers isolates which have resistance to 6 or more classes of antimicrobials to be highly drug resistant.
Detection of quinolone resistance in Salmonella Enteritidis from chicken
In 2018, a clear increase in nalidixic acid (a quinolone) resistance among S. Enteritidis from chickens occurred across several surveillance components from multiple provinces.
Retail
- 1 isolate from a chicken burger in British Columbia (FoodNet Canada)
Retail
- 1 isolate from Alberta
- Not previously observed
Abattoir
- 2 isolates from Ontario and Québec
- Not previously observed
Clinical cases
- 2 isolates from Ontario (sick chicken)
- Previously observed in a single isolate in Manitoba (2010)
The majority of S. Enteritidis from animal and food sources were susceptible to all antimicrobials tested.
From 2003 to 2018, nalidixic acid resistance was only observed in a single isolate from a sick chicken (clinical isolate) from Manitoba in 2010.
Most S. Enteritidis from people were susceptible to all antimicrobials tested. When resistance did occur, it was most commonly to nalidixic acid and these human cases may be related to travel outside of Canada.
This may be a new source of human exposure to nalidixic acid-resistant S. Enteritidis in Canada. This unprecedented and sudden increase in chickens and chicken meat will be monitored closely by CIPARS.
Highly drug-resistant Salmonella
- The number of highly resistant Salmonella isolates have increased substantially since 2008 in both human and animal sources.
- 2018 was the first time where highly resistant Salmonella were isolated from a chicken source.
- Despite a slight decrease in 2017, a substantial increase in the number of highly resistant Salmonella occurred in 2018 to levels not previously observed by CIPARS.
In 2018, 132 Salmonella isolates were identified as highly drug resistant from the following sources:
Chicken
- All highly resistant isolates from chicken were from retail samples (from chicken burgers) from British Columbia, the Prairies, and Ontario.
- All isolates were S. Infantis.
Cattle
- Sick cattle (clinical isolates).
- Most isolates were S. Dublin.
Swine
- Sick pigs (clinical isolates).
- Some isolates demonstrate resistance to all 7 classes of antimicrobials tested.
- Four serovars were detected.
Human
- Only clinical isolates tested.
- Most isolates were S. Infantis, S. Newport, and S. Typhimurium.
Figure 14. Number of Salmonella isolates resistant to 6 or more antimicrobial classes from 2008 to 2018
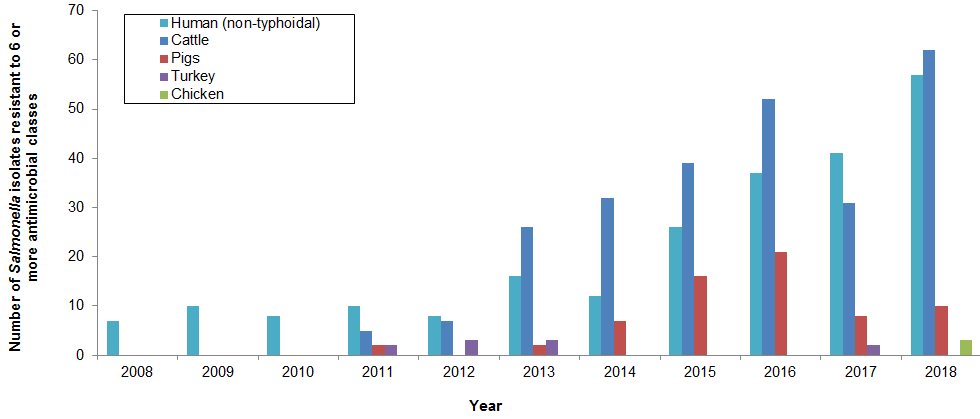
Figure 14 - Text Description
| Year | Cattle | Pigs | Chicken | Turkey | Human (non-typhoidal) |
|---|---|---|---|---|---|
| 2008 | 0 | 0 | 0 | 0 | 7 |
| 2009 | 0 | 0 | 0 | 0 | 10 |
| 2010 | 0 | 0 | 0 | 0 | 8 |
| 2011 | 5 | 2 | 0 | 2 | 10 |
| 2012 | 7 | 0 | 0 | 3 | 8 |
| 2013 | 26 | 2 | 0 | 3 | 16 |
| 2014 | 32 | 7 | 0 | 0 | 12 |
| 2015 | 39 | 16 | 0 | 0 | 26 |
| 2016 | 52 | 21 | 0 | 0 | 37 |
| 2017 | 31 | 8 | 0 | 1 | 41 |
| 2018 | 62 | 10 | 3 | 0 | 57 |
Integrated antimicrobial use and resistance data
In this section, we highlight two integrated antimicrobial use and resistance stories for 2018:
- Fluoroquinolone-resistant Campylobacter
- Ceftriaxone resistance in non-typhoidal Salmonella and generic E. coli
Fluoroquinolone resistance in Campylobacter
- In 2018, one flock from British Columbia reported using a fluoroquinolone for disease treatment. Since 2014, there was no reported fluoroquinolone use on sentinel broiler chicken farms.
- In general, the highest proportion of ciprofloxacin-resistant Campylobacter continued to be from British Columbia. However, resistance from chicken(s) continued to vary over time and across regions.
- In British Columbia, ciprofloxacin-resistant Campylobacter from chickens on farm decreased to 17% (8 out of 46 isolates were resistant).
- In Québec, ciprofloxacin-resistant Campylobacter from chickens on farm increased to 50% (8 out of 16 isolates were resistant).
Figure 15. Ciprofloxacin resistance in Campylobacter isolates over time and between regions; CIPARS 2011 to 2018
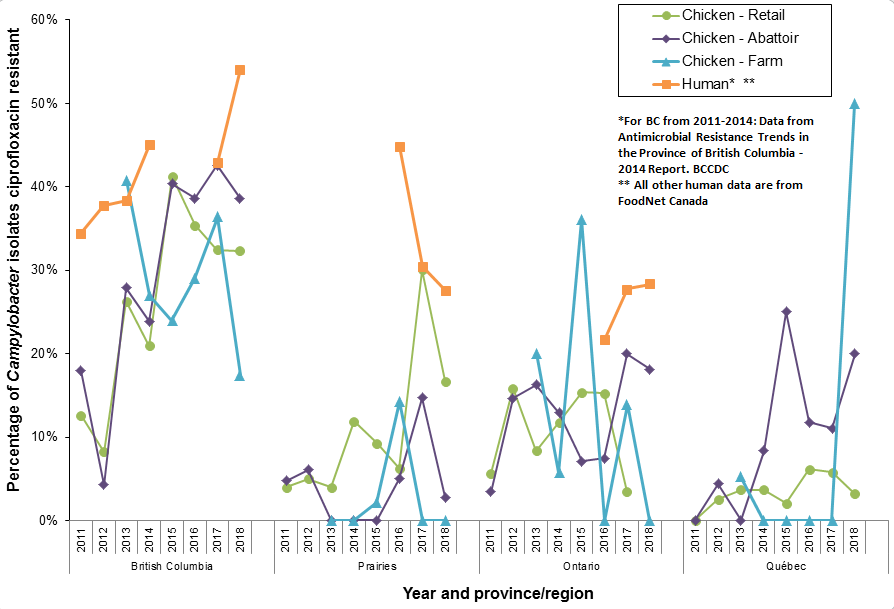
Figure 15 - Text Description
Ciprofloxacin resistance in Campylobacter isolates over time and between regions; CIPARS 2011 to 2018
The temporal trends of the percentage of Campylobacter isolates being resistant to ciprofloxacin are depicted in this figure by year (2011 to 2018) and by province (British Columbia, Ontario, and Québec) or region (Prairies). For each province or region, the temporal trends are represented by 4 series corresponding to 4 Campylobacter data sources as listed here:
- chicken from retail (green line and round marker)
- chicken from abattoir (purple line and diamond marker)
- chicken from farm (blue line and triangle marker)
- human origin*/** (orange line and square marker).
* For British Columbia from 2011 to 2014: Data from antimicrobial resistance Trends in the province of British Columbia - 2014 Report, British Columbia Centre for Disease Control (BCCDC).
** All other human data are from Foodnet Canada.
Integrated antimicrobial use and resistance data
Ceftriaxone resistance in non-typhoidal Salmonella and generic E. coli
Since 2015, there has been no reported ceftiofur use in sentinel broiler chicken flocks, as well as reduced ceftriaxone resistance in both E. coli and Salmonella from chickens and chicken meat in most scenarios.
Previously, ceftriaxone-resistant Salmonella in humans were primarily serovar Heidelberg isolates. However, in 2018, the majority of resistant isolates were serovar Infantis, followed by serovar Heidelberg.
In 2018, ceftriaxone resistance in S. Infantis decreased to 15%, compared to 17% in 2017. Ceftriaxone resistance in S. Heidelberg also decreased to 7% compared to 12% in 2017.
Overall, ceftriaxone resistance in Salmonella and E. coli isolates from chicken sources remained relatively stable or decreased after the 2014 initiative to eliminate the use of Category I antimicrobials for disease prevention. Most Salmonella isolates were S. Kentucky, followed by S. Heidelberg and S. Infantis.
However, there were some increases in ceftriaxone resistance in 2018 compared to 2017 data. This is most notable in Salmonella isolated from chickens at the farm level.
Figure 16. Reduction in reported use of ceftiofur on sentinel farms and changing resistance to ceftriaxone in non-typhoidal Salmonella from humans and chicken sources between 2013 and 2018
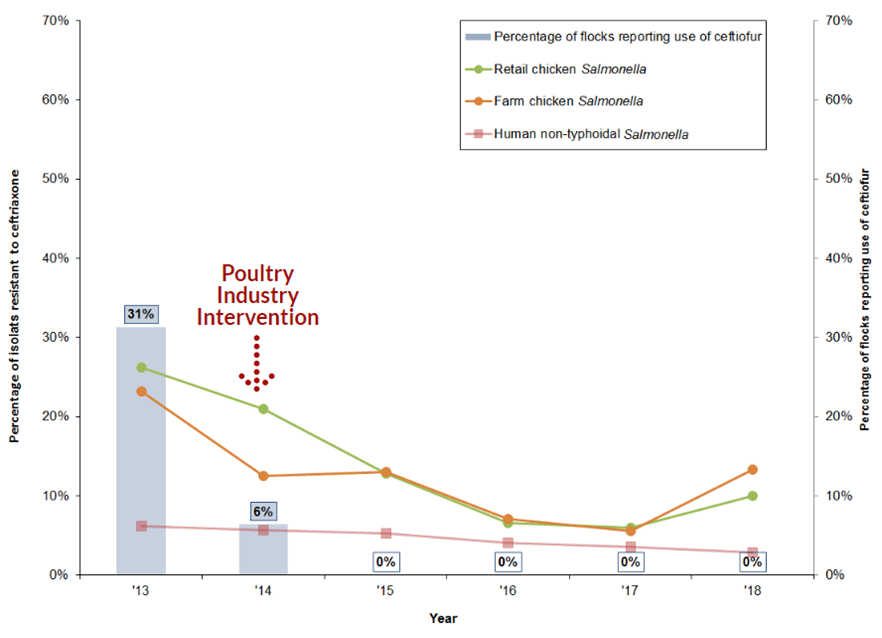
Figure 16 - Text Description
| Year | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|---|
| Component | ||||||
| Percentage of isolates resistant to ceftriaxone | ||||||
| Farm chicken Salmonella | 23% | 12% | 13% | 7% | 6% | 13% |
| Retail chicken Salmonella | 26% | 21% | 13% | 7% | 6% | 10% |
| Human non-typhoidal Salmonella | 6% | 6% | 5% | 4% | 4% | 3% |
| Percentage of flocks reporting the use of ceftiofur | ||||||
| Broiler chicken flocks on farm | 31% | 6% | 0% | 0% | 0% | 0% |
The reduction in ceftiofur use and associated decrease in ceftriaxone resistance compared to pre-2014 data in chickens and humans is a good example of a successful intervention to limit antimicrobial resistance.
What's New for CIPARS in 2018
We are modernizing how we share our information with different audiences and are transitioning to new communication tools and formats. In the meantime, CIPARS will continue to deliver the same information, but in a modified manner.
For the 2018 data, we will be releasing 4 documents:
- Executive Summary
- Figures and Tables (summarized information with little accompanying text)
- Design and Methods
- Integrated Findings
Antimicrobial Use
- Data collected under legislation by Health Canada from veterinary pharmaceutical manufacturers, importers, and compounders for 2018 is currently being analyzed. Results will be released as a separate report in Spring of 2020.
- In 2018, grower-finisher pig quantitative antimicrobial use metrics were reported for the first time for antimicrobial use in water and injectables.
- As of 2018, the antimicrobial use metric nDDDvet/PCU was no longer be reported.
Antimicrobial Resistance
- There was no placement broiler chicken sampling conducted in 2018.
- For farm surveillance, sampling in turkeys was initiated in Alberta.
- Only a partial year of retail sampling was conducted in Ontario and the Prairies, and no sampling occurred in the Atlantic region; therefore, no temporal retail data from these regions are presented in 2018.
In addition to the changes described above, we launched two sentinel farm surveillance activities in feedlot and dairy cattle with our stakeholders.
Glossary
- Antimicrobial class
- Antimicrobials are grouped into the same class if they have a common chemical structure and method to kill or stop the growth of bacteria. CIPARS uses the Clinical and Laboratory Standards Institute to define antimicrobial class.
- Biomass and Population Correction Unit (PCU)
- The PCU accounts for the size of the population. It includes both the number and weight (biomass) of animals or people in the population. CIPARS uses the PCU to interpret the antimicrobial use and consumption data, using the same approach as the European Surveillance of Veterinary Antimicrobial Consumption Project.
- DDDvet
- This is an acronym for the "Defined Daily Dose for animals". The amount of antimicrobials given during a treatment (dose) will vary depending on the antimicrobial, how the antimicrobial is given (e.g. by injection, through water or feed) and the population treated (cattle, chickens, pigs). CIPARS uses this metric to adjust for this variation and help interpret antimicrobial use data.
- Grower-finisher pig
- A pig that is approximately 25 kilograms to market weight.
- Medically important antimicrobials
- Antimicrobials deemed to be of very high importance (Category I), high importance (Category II), or medium importance (Category III) in human medicine.
- mg/PCU
- An antimicrobial use metric that adjusts the quantity (milligram/mg) of antimicrobial used, consumed or distributed by the size of the population.
- nDDDvet/1000 animal-days
- An antimicrobial use metric that adjusts for both variation in the amount of antimicrobial given during a treatment (DDDvet), and the length of time that an animal or group of animals are treated to help interpret antimicrobial use data.
- Non-typhoidal Salmonella
- All Salmonella serovars, excluding S. Typhi, and Paratyphi A and B.
CIPARS analysts are working to develop new ways of identifying emerging issues and integrating data across various host species, bacterial species, and across regions.
CIPARS will continue to monitor and communicate the impact of changing antimicrobial use practices on the occurrence of antimicrobial resistance to preserve the effectiveness of antimicrobials in animals and humans.