Mould Prevention and Collection Recovery: Guidelines for Heritage Collections – Technical Bulletin 26
By Sherry Guild and Maureen MacDonald
Revised by Tom Strang and Sherry Guild
CCI Technical Bulletins
Technical Bulletins are published at intervals by the Canadian Conservation Institute (CCI) in Ottawa as a means of disseminating information on current techniques and principles of conservation of use to curators and conservators of Canada's cultural objects and collection care professionals worldwide. The authors welcome comments.
Abstract
Mould infestation in heritage collections can damage objects and may pose a health risk to individuals who work with these collections. This Technical Bulletin presents information on mould morphology, prevention of mould growth, actions to take should mould occur and health effects relating to mould exposure. It informs the reader on how to remove mould growth from objects and it describes the appropriate personal protective equipment to wear when working in a mould-contaminated environment or when working with mould-infested objects.
Authors
Sherry Guild graduated from the Art Conservation Techniques program at Sir Sandford Fleming College, now Fleming College (Peterborough, Ontario). In 1984, she started at CCI as a conservator in the paper laboratory, specializing in the conservation of works of art on paper. During her career, she assisted many clients on the recovery of mould-contaminated objects. She retired from CCI as a Senior Conservator, Works of Art on Paper, in 2015.
Maureen MacDonald worked in the Preventive Conservation Services Division at CCI from 1981 to 2010. Her areas of expertise included environmental monitoring equipment, ultraviolet filtering materials and biological materials. During her career, she worked on projects involving the microscopic characterization of frozen archaeological skins and preparation techniques for natural history specimens, and she studied mould and mouldy materials. She was named an Honourary Life Member of the Canadian Association for Conservation of Cultural Property (CAC) in 1998.
Tom Strang earned a Ph.D. in Conservation from the University of Gothenburg, a Master's of Art Conservation (Artifacts) from Queen's University and a B.Sc. (Hons) in Biology from Carleton University. Since joining CCI in 1988, Tom has generated solutions to problems presented by organisms that can damage cultural property. He is an expert in integrated pest management (IPM) for all types of cultural collections facilities and has conducted IPM reviews for leading museums, galleries and archives in North America. He has established the efficacy of thermal control methods against insect pests and investigated the risk of adverse effects of pest control treatments on cultural heritage objects.
Disclaimer: The information provided here is based on the current understanding of the issues presented. The guidelines given in this Technical Bulletin will not necessarily provide complete protection in all situations or protection against every possible adverse effect caused by the use of products in museum contexts.
Table of contents
- List of abbreviations
- Introduction
- 1. Mould prevention
- 2. Collection recovery
- 2.1 Personal protective equipment
- 2.2 Cleaning mould-contaminated objects
- 2.3 Cleaning the collection storage area
- 2.4 Cleaning methods and considerations for some objects
- 2.5 Considerations after cleaning
- Conclusion
- Acknowledgements
- Suppliers
- Websites and other sources of information
- Appendix: Removing mould
- Bibliography
- Endnotes
List of abbreviations
- µm
- micrometre
- aw
- water activity
- ASHRAE
- American Society of Heating, Refrigerating and Air-Conditioning Engineers
- CFU
- colony forming unit
- dB(A)
- decibel
- EMC
- equilibrium moisture content
- ERH
- equilibrium relative humidity
- HEPA
- high efficiency particulate air
- MVOC
- microbial volatile organic compounds
- PAPR
- powered air purification respiratory (system)
- PPE
- personal protective equipment
- PVC
- poly(vinyl chloride)
Introduction
This Technical Bulletin provides general information for the archive, library, museum and gallery community regarding fungalEnd Note 1 (or mould) infestation in heritage collections. It presents information on fungi morphology, prevention of fungal growth, actions to take should fungal growth occur and health effects connected to mould exposure. It informs the reader on how to remove mould growth from objects and describes the appropriate personal protective equipment (PPE) to wear when working in a mould-contaminated environment or when working with mould-infested objects.
The treatment of infested objects with chemicals (thymol, ethanol, ortho-phenylphenol, etc.) or by non-chemical methods (gamma radiation, ultraviolet light, microwaves, etc.) is not addressed in this Technical Bulletin. For information on these topics, consult the conservation literature.End Note 2 In general, using chemicals to treat mould is no longer recommended for heritage collections. Although chemical methods have been used in the past, their efficacy, their possible deleterious effects on the object and the considerations regarding the effects of these substances on humans have not, in some cases, been fully investigated. Also, it is important to note that killing fungal organisms usually does not destroy their antigenic or toxic properties. This means that dead fungal growth on the object remains a health concern and that no chemical treatment confers lasting or residual mould control. These factors have contributed to the shift away from using chemicals to treat mould infestation in heritage collections.End Note 3
Any treatment to combat mould infestation should focus mainly on measures that keep the level of moisture in the air and the moisture content of the object below the level conducive for mould growth. In the event of a mould infestation, the conscientious removal of visible mould growth and the reduction of spores from objects should be undertaken.
This Technical Bulletin adopts information regarding levels of mould contamination and appropriate PPE from guidelines for assessment and remediation of mould in buildings and applies it to heritage collections. It does not address mould remediation in buildings, building envelopes or heating, ventilation and air conditioning systems. For information on these subjects, consult the appropriate professionals.
Note: It is strongly recommended to read the entire Technical Bulletin and to consider the information carefully before proceeding to treat mould-infested collections. Mould can be a serious health concern and medical research into its effect on humans is a rapidly developing area. If in doubt, consult professionals.
1. Mould prevention
1.1 Mould
The nature of fungi, how they grow, under what conditions, the viability of mould spores and triggers of activation are discussed in this section. Although mould remediation in buildings is not addressed in this Technical Bulletin, mould, or conditions conducive to its growth in buildings, presents a risk to the collection. For this reason, the Bulletin presents techniques for the detection of mould spores and the level of mould spores likely to be found in indoor environments, as well as a chart identifying potential sources of mould in buildings and the factors contributing to its growth.
What is mould?
Moulds are members of the kingdom Fungi. There are thousands of fungi species, ranging from yeasts, which are single-celled organisms, to the more complex multi-cellular mushrooms and toadstools. The majority of fungi lie somewhere in between. Fungi play an important role in the cycle of nature. Unlike green plants, fungi lack chlorophyll and cannot photosynthesize their own nutrients from carbon dioxide and water. Most fungi are saprophytic: they are organisms that live on and derive their energy from dead or decayed organic matter, such as plants, food, leaves, etc. They feed solely by digesting the substrate on which they grow. A few are parasitic and some live symbiotically with a host. Their growth is often called mould. Mildew, a term often used to describe mould in the home, is actually a parasitic fungi that grows only on living plants.
Under a microscope, mould looks like a network of thread-like filaments, referred to as hyphae, woven into a network, called the mycelium. Moulds spread by and grow from various types of microscopic spores, of which some are referred to as conidia.End Note 4 The conidia are produced during the asexual reproductive phase from a specialized structure or fruiting body called a conidiophore. The shape of a spore is determined by the species. A spore can be round, elongated, oblong, cylindrical, sickle-shaped, single-celled, multi-cellular, etc. In general, spores and fruiting structures must be present before most moulds can be positively identified. Depending on the species, spores range from 1 to 200 μm in length (a micrometer [μm] is one-millionth of a metre or 1/25,000 of an inch). Even the largest spores are buoyant enough to be carried long distances by air currents.
In the temperate regions of Canada, the most common species of moulds are Cladosporium, Alternaria, Aspergillus and Penicillium. The latter two species are mostly found indoors. These species are known as filamentous or conidial types. The spores (conidia) form at the ends of branched (tree-like) structures called conidiophores (Figure 1). In general, the species of mould found inside a building are the same as found outdoors, but they are found in much lesser concentrations.
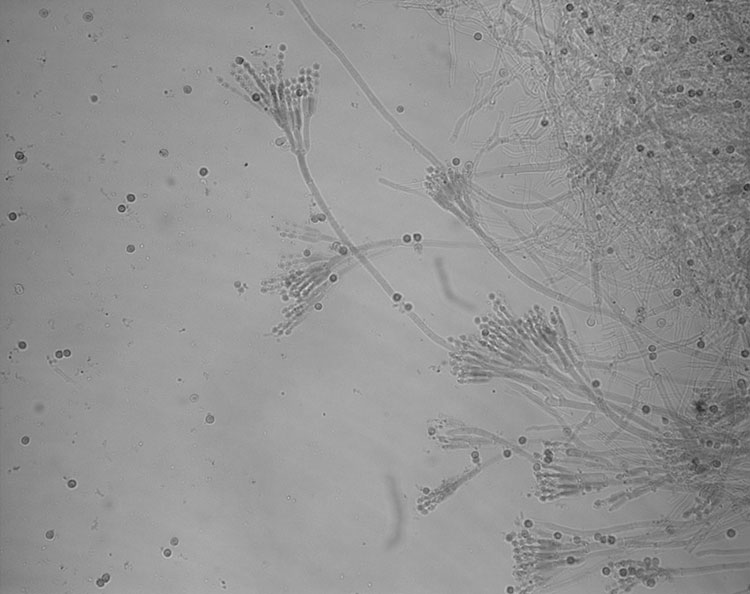
© Government of Canada, Canadian Conservation Institute. CCI 127508-0001
Figure 1. Conidia and conidiophores of a Penicillium species.
Mould growth
Mould can grow under a very wide range of conditions. When the environment is suitable for germination, the spore swells and a germ tube extends outward. For most mould, this action is triggered by a significant change in temperature or in the elevation of moisture. A germ tube is a vegetative cell that elongates as it assesses the amount of moisture and nutrients in the substrate. If adequate moisture exists, the germ tube begins to branch as it continues to elongate. These branches multiply and become the hyphae. The hyphae grow and form an interwoven colony of fibres called the mycelium. Depending on the texture or porosity of the material on which the mould is growing, the hyphae will penetrate, to some degree, into the substrate (Figure 2). Sporulation, the production of fruiting bodies and spores, can result from a change in the growing conditions of the mycelium (Griffin 1981). Specialized hyphae form into conidiophores that mature and release spores into the air, which starts the cycle again. Conditions that have the potential to trigger sporulation include:
- exhaustion of nutrients
- production of chemical by-products
- changes in light and temperature
Mould grows outwards from a central point in a circular pattern (Figure 3). The centre of the mycelium is the first to die out. It does so for two reasons:
- the nutrients become exhausted
- different chemicals appear during metabolism that inhibit mould from growing back on previously infested areas
These mechanisms ensure that the hyphae are always spreading outwards and seeking new nutrients. The newest growth, usually white, is on the leading edge. The centre of the mycelium can develop a colour, which results when spores are formed. Colour change can also indicate a pH change in the substrate. As the spores mature, the colour of the area can also change.
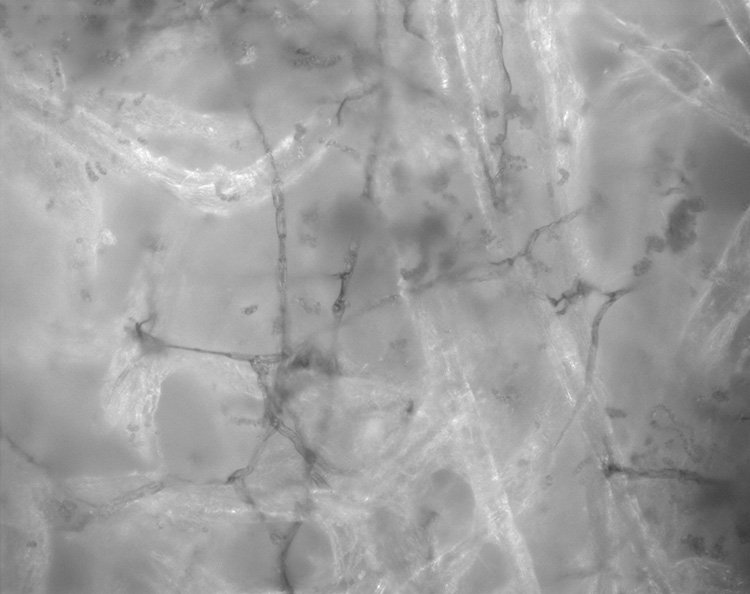
© Government of Canada, Canadian Conservation Institute. CCI 127508-0006
Figure 2. Mould hyphae and spores embedded into paper fibres.
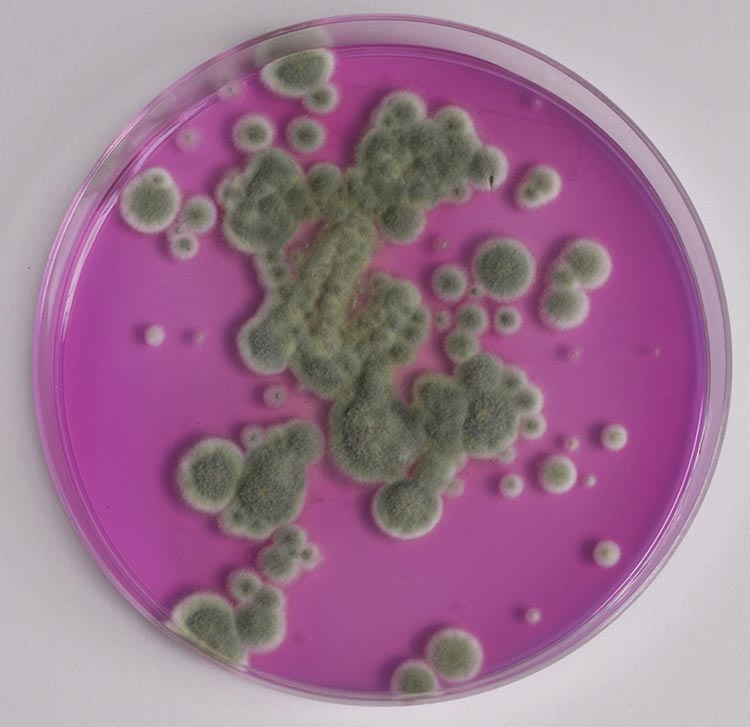
© Government of Canada, Canadian Conservation Institute. CCI 74903-0018
Figure 3. Agar plate of mould.
Requirements for growth
Nutrients
The nutrients required for mould growth are very basic and are supplied by organic materials. Enzymes break down the organic substrate into the necessary nutrients that are absorbed through the hyphae walls. Nutrients are derived from simple sugars, starches, small peptides and complex carbon-containing substances such as amino acids.
In fact, fungi are able to secrete a tremendous number of enzymes capable of digesting any organic material, either plant or animal. However, non-organic materials, such as glass or metals, can also support growth if there are residues of organic materials on their surfaces.
Moisture
In order for a mould spore to grow, several conditions must be present. The first and most important is an adequate amount and supply of moisture. Each mould species requires a minimum amount of water in order for the spore to swell and begin germination. This water comes from the substrate material. Water vapour in the air, measured as the relative humidity (RH), influences the moisture in the substrate material. This source in the substrate is the only moisture available to the mould.
Biologists describe the moisture available in a substrate with the term water activity (aw), expressed as a fraction of 1 (Ayerst 1969). Water activity is equivalent to the equilibrium RH (ERH) of air adjacent to the material or within its pores. ERH is measured by placing the material in a sealed container and then measuring the RH of the trapped air, after enough time for equilibration to have elapsed. Thus, a measurement of 90% RH indicates an ERH of 90% and, in turn, an aw of 0.9. The majority of moulds will not grow until the aw of the substrate material is 0.75 or higher (Onions et al. 1981). (This value is discussed in more detail in section 1.2 How to prevent mould growth in the collection.)
Temperature
Moulds are able to grow under a very wide range of temperatures, as exemplified by mouldy food in the refrigerator. Most mould spores will grow in temperatures from 4°C to 30°C (39°F to 86°F). The rate of mould growth can be regulated by temperature. Growth decreases with lower or higher than optimal temperatures. The majority of heritage collections are maintained between 15°C and 25°C (59°F and 77°F). These temperatures are ideal for mould growth. (It bears noting that Canadian collections kept in unheated winter conditions consistently below 0°C [32°F] are, therefore, not at risk of mould growth until spring.) Short-term exposure to temperatures slightly beyond those for optimal growth will bring about dormancy. A return to optimal conditions will activate growth. Freezing temperatures kill growth. However, some spores can also tolerate extended periods of extremely low or high temperatures. Spore viability is diminished by the alternating action of freezing and thawing.
pH
The pH (acidic, neutral or alkaline) of the substrate will affect the germination, colour and growth of mould. The pH range of the substrate for spore germination may extend from 2 to 9. The optimum is between pH 4 and 7. Many objects in heritage collections fall within this range. Actions, such as washing or deacidification of paper objects, to adjust pH do not deter mould. As with moisture, certain species of mould have different pH triggers. The pH of the substrate material is likely to be altered due to the chemicals released during metabolism, such as metabolites, enzymes and exudates.


Air circulation
Good air circulation will help maintain an even level of RH and eliminate areas where a microclimate of high or low RH might develop. If water damage occurs, a good flow of dry air facilitates rapid evaporation and drying of the substrate material, making it less conducive to fungal growth. In some circumstances (for example, air-drying wet or damp items), air circulation may be a factor in determining whether or not mould grows. An adequate airflow, one that feels “drafty,” will help prevent fungal growth.
Light
The role that light plays in mould growth is not well defined. Some studies of certain moulds have shown that light affects moulds in the following ways: it may inhibit growth, affect the direction and rate of growth, and affect the production of certain compounds (toxins and volatile organic compounds). Light can also affect the reproductive processes of mould. For some species, light is essential, for others, it is not required.
Objects stored in the dark to reduce the rate of deterioration are not more susceptible to mould growth. Light is not a critical factor for controlling indoor fungal growth: nutrients, moisture and temperature are the critical factors (Shaughnessy et al. 1999). However, mould growth may go undetected for long periods of time if routine inspections of storage areas or areas where human occupation is minimal are not regularly carried out. Also, if there is less air circulation in dark storage areas, it may contribute to mould growth.
Viability
Dormant (inactive) spores wait for the right amount of water and nutrients before beginning to grow. As spores wait for optimal growth conditions, they age and their viability decreases. Some species of mould spores are capable of remaining viable for many years, while others are able to survive for only a few hours. Within the particular range of a certain species, environmental conditions play an important role in the viability of spores. Fluctuations in temperature and RH are factors of viability, as is the presence of many chemical agents, such as fungicides. Spores of the species Aspergillus and Penicillium are known to be viable for up to 10 years (Sussman and Halvorson 1996).
It is also believed that certain species of mould spores are activated by chemicals, such as detergents or organic solvents (for example, acetone and ethanol/water mixtures). This action is not well understood. Perhaps some of the chemicals acting as wetting agents cause the activation (Griffin 1981).
Levels of mould spores
All mould spores originate from the outdoor air. The species of mould and the concentration of spores in the air vary with the season, temperature, RH and geographical location. Spores fall on objects, regardless of the location from which the objects originate, or where they travel or are stored or displayed. Thus, it is important to remember that objects are never completely spore free.
Every mould species has preferences with regards to the environment and nutrients for optimal growth. In the temperate regions of North America and similar climates, the highest concentrations of mould spores outdoors are in the spring and fall. In other climate regions, such as the tropics, high concentrations may be found throughout the year or mould concentrations may have higher and lower periods depending if it is the rainy or dry season. In tropical regions, Aspergillus and Penicillium species are more dominant than in the temperate regions of the world (Mullins 2001).
The species of mould found indoors should be similar to those found outdoors. In a normal healthy indoor environment, concentrations of mould spores are much less than those found outdoors. It has been reported that at peak times during the year, outdoor fungal particles can reach 104/m3 (Miller 2001). An indoor air-handling system filters out a portion of the spore material based on the filter's efficiency.
If indoor air quality tests identify a fungus that is not found outdoors at that time of the year, it may indicate an indoor source, referred to as a fungal amplifier, growing inside a building or collection. These higher levels may indicate a moisture problem or other conditions conducive to mould growth.
High concentrations of certain mould species pose a health risk for humans. Breathing air containing a normal array of mould spores does not usually affect healthy humans. However, if a mould problem exists in a collection (or a building), the concentration of spores can be many times the concentration normally found indoors. This could lead to adverse health issues in hypersensitive people. (Health issues are addressed further in section 1.5 Health effects.) Further investigation will be required to determine the source of the mould.
Health Canada does not have any numerical exposure limits for mould. Since people have different sensitivities, it is not possible to establish a “safe” limit for mould. Health Canada recommends removing any mould found growing indoors and fixing the underlying moisture problem (Health Canada 2014).
Detection
Mould growth on objects can be recognized by a fuzzy coloured appearance and sometimes, but not always, a mouldy or musty smell. The smell is the odour of microbial volatile organic compounds (MVOCs), such as alcohols, esters, terpenes and ketones, which are produced by the mould. The fuzzy growth is often black or white but can appear in other colours depending on the substrate it is growing on. The growth consists of thread-like filaments—the hyphae. When actively growing, the mould will be damp and smear if brushed.
Fungal growth on objects may also be encountered in a dormant state. When dry, it may appear as a coloured stain, smudge or dirt. It may smear when brushed. Objects that exhibit signs of previous contact with water may contain fungal growth. If this growth has had a recent contact with water and crucial growth factors are present, it may become active again. Given enough time without critical growth factors, the mould hyphae may die. Dormant fungal growth may not emit the characteristic mouldy smell. Although dormant mould does not pose an immediate risk to an object, it may be a health risk to humans. It is important to note that mould does not lose its allergenic or toxigenic properties when it is dormant or non-viable.
One of the most obvious places to encounter mould is in a basement or in a below-ground-level storage area. Basements are usually damp, dusty and dark locations with stagnant air. These four conditions can make a basement or any area a likely habitat for mould to grow. Most often these locations are below ground level and have earthen floors, crawl spaces and cement or masonry-type walls that transfer water vapour. Basements are also prone to lower temperatures, higher RH and poor air circulation. Crawl spaces are usually poorly ventilated, which creates areas with chronic high humidity. It is important that properly screened, adequate venting be installed and maintained throughout the year. Venting should be open in the warmer months and closed in the colder months. Calculations to determine venting requirements can be found in ASHRAE 2013.
Fungal growth can also be found where there has been a saturation of water. Accidents, such as a leak from a broken pipe or roof, backed-up sewers, the failure of a heating, ventilation or air conditioning (HVAC) system as well as fire and floods can all elevate the RH and may initiate mould growth. A significant temperature drop can also increase the RH. Outside corners of buildings at roof or floor lines are susceptible to cooler temperatures, generating higher humidity. If these conditions are not dealt with quickly (within 48 hours), mould growth can begin.
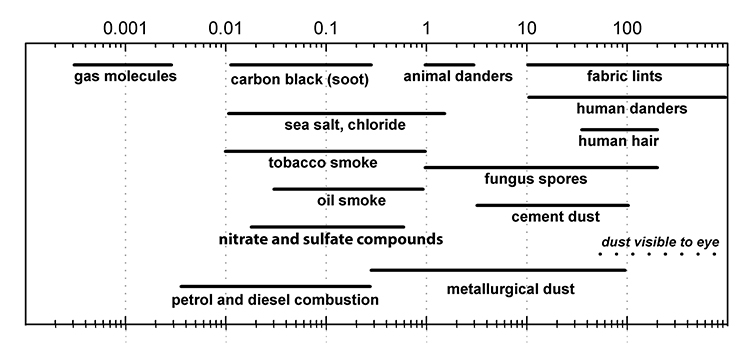
Dirt and dust are hygroscopic (water loving) and a source of nutrients for mould. Their presence in the collection may increase the potential for mould to grow. Mould spores and mould fragments, such as hyphae and mycelium, settle out of the air at a rate determined by the size of particles. Spores can fall at the rate of 0.5 cm/s to 2.8 cm/s onto surfaces and become part of the dust and dirt (Mullins 2001, Tétreault 2003). Figure 7 shows the comparative sizes of various airborne pollutants.
Some storage conditions may contribute to mould growth on objects. Cardboard, wood, adhesives and sizes provide a suitable nutritional base for mould. Boxes, especially cardboard or wood sitting on cold floors or against exterior cement walls, absorb moisture and may support mould growth. Mould growth on the container may contaminate the contents of the container as well as the surrounding environment, including any objects in the area. Other examples of susceptible items include wooden furniture or paintings stacked against an exterior cement wall. It is not recommended to store objects directly on floors or leaning against exterior masonry walls.
Objects stored in archival-quality matboard boxes and paper and plastic enclosures are somewhat more protected against fungal growth than those stored in the open. For short periods of time, a box or another container made from a hygroscopic material, such as paper, cardboard or wood, can buffer the object from increased ambient RH. However, a prolonged period of elevated RH is enough to raise the aw of the object inside the container to a point where it can support mould growth. Contact of the container (or the object inside) with water is a significant risk. If an object is in a well-sealed impermeable container (polyethylene bag or container), it is protected from sudden increases in ambient RH and from direct contact with water. It is crucial for the object to be dry when placed inside the container and not to have an equilibrium moisture content (EMC) capable of elevating the ERH in the container above 65%. If periods of elevated RH are prolonged, staff should regularly examine the items stored in enclosures (paper or plastic) for mould growth.
It is important to note that, eventually, those objects within polyethylene containers will reach equilibrium with the average outside ambient RH (Strang 2001), as polyethylene is not impermeable to water vapour, although it greatly delays its transfer.

Figure 6. Water-damaged building materials.
© Government of Canada, Canadian Conservation Institute. CCI 127508-0008
Figure 7. Graph showing the particle size in µm of various materials.
Description of Figure 7
The graph shows the comparative size of various airborne pollutants in particle sizes between 0.0001 and 1000 µm. The size for gas molecules is the smallest, between 0.0003 and 0.003 µm. Petrol and diesel combustion molecules range in size from 0.003 to 0.3 µm. Carbon black (soot) particulates range from 0.01 to 0.5 µm. Tobacco smoke particulates range from 0.01 to 1 µm. Sea salt, chloride particles range from 0.01 to 2 µm. Nitrate and sulfate compounds range from 0.02 to 0.7 µm. Oil smoke particulates range from 0.03 to 1 µm. Metallurgical dust ranges from 0.5 to 100 µm. Fungus spores range from 1 to 200 µm. Animal danders range from 1 to 3 µm. Cement dust ranges from 3 to 100 µm. Fabric lints and human danders have the same range for particle sizes: from 10 to 1000 µm. Human hair ranges in size from 30 to 200 µm. Dust visible to the eye ranges between 40 and 700 µm.
The first two columns of Table 1 follow Macher (1999) and illustrate potential sources of bioaerosol (mould, bacteria, viruses, etc.) contamination in buildings and the factors contributing to mould growth. The building engineer or maintenance person in charge of the building and its air-handling system should be aware of these potential trouble spots. The third column suggests programs or measures to prevent the problems from happening.
| Source | Contributing factors | Preventive measures |
|---|---|---|
| Building exterior |
|
|
| Outside air |
|
|
| HVAC systems, air intakes |
|
|
| Filters |
|
|
| Heat exchanger |
|
|
| Plenums and duct work |
|
|
| Air diffusers |
|
|
| Occupied space (water damage) |
|
|
| Constant condensation |
|
|
| Window air conditioners |
|
|
| Potted plants |
|
|
| Carpets |
|
|
| Fabric partitions, drapes and furniture |
|
|
| Portable humidifiers and dehumidifiers |
|
|
1.2 How to prevent mould growth in the collection
Because mould spores are everywhere in the air, it is impossible to completely eliminate them from a building, a collection and storage or exhibition areas. They circulate in the air, moving with the air currents, some falling on the surface of objects, floors, walls, ceilings and furnishings. They drift indoors through the air-handling system, open doors or windows and drop off people and materials coming into the building.
The most effective strategy to prevent damage to objects and to prevent adverse health effects for humans is to ensure that the environment and other conditions inhibit mould growth. This section focuses on ways to prevent fungal growth in a collection and a building.
Critical factors to control
Reducing indoor spores
Mould spores can only be reduced, not eliminated. The following actions can be taken to limit spores entering the indoor environment (Flannigan 2011):
- closing windows and doors reduces outdoor spores by 2%
- using central air-conditioning reduces outdoor spores by 5%
- using electrostatic filtration reduces outdoor spores by 3%
Nutrient sources
When they are broken down into simple sugars, amino acids and small peptides, many objects provide suitable food for mould growth. Objects made from organic material or that have organic components, such as paper (cellulose, sizes and coatings), some media, book cloth, leather, basketry, wood, upholstery, cotton, linen, wool and photographic materials, are definitely food sources. Some inorganic objects may also be susceptible to mould because of their age and surface accretions of dust, dirt, insect debris and oils from handling. Where possible, remove nutrient sources that encourage fungal growth by cleaning and maintaining the collection as well as storage and display areas. Dust is a major source of fungal spores and nutrients.
Moisture
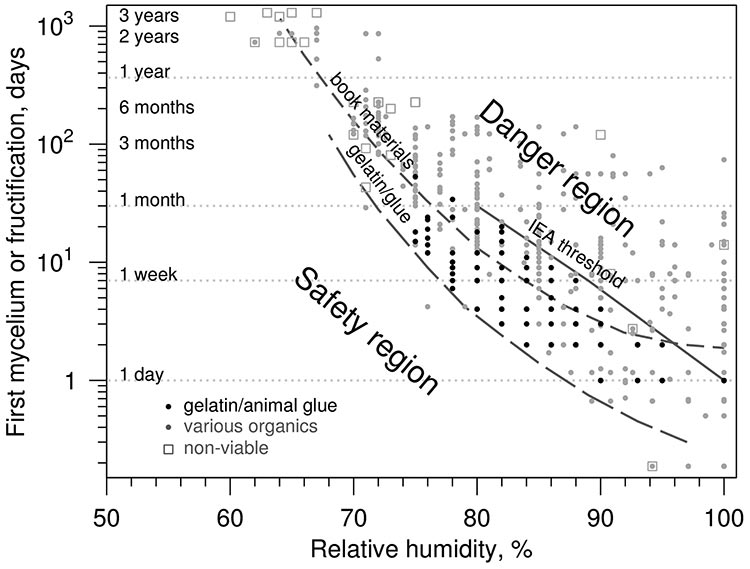
Moisture, as has already been stated, is essential for mould to germinate and grow. Because it is practically impossible to eliminate all nutrients, moisture becomes the easiest factor to control. It is essential to avoid two conditions conducive to mould growth: all peaks of very high RH that initiate spore germination, and prolonged periods of elevated RH that support mould growth. Figure 9 illustrates the relationship between RH/aw and mould growth (based on Strang 2012):
- at 65% RH, 0.65 aw, experiments lasting three years show no noticeable mould growth
- at 75% RH, 0.75 aw, growth is noticeable between two weeks and three months
- at 80–90% RH, 0.80–0.90 aw, growth is noticeable between one and five days
Based on this information, one can ignore short periods of “high” RH in each range given, as long as the duration is substantially less than the period stated above for germination or noticeable growth.
The risk of mould damage to objects obviously increases with extended exposure to RH and depends on the ability of the object to absorb water vapour. The risk of mould beginning to grow can be greatly reduced by storing objects in containers. The important issue is how long the particular container will protect the enclosed object from high humidity exposure. In uncontrolled environments, containers such as closed cardboard boxes protect items for only a very short period of time. For long-term protection, sealed plastic containers are more effective. Once the objects inside reach levels greater than 0.75 aw, containers or boxes work against maintaining mould-free objects. Objects that have already absorbed water vapour are slow to dry. Although the environment inside the container will eventually reach an equilibrium with the normal outside environment, the object will react more slowly. Therefore, for a period of time, the object may retain a level of water activity sufficient to support mould growth. Two properties govern the environment within containers: the rate at which water vapour can permeate the container and the ability of the object to buffer the internal humidity/water activity.

© Government of Canada, Canadian Conservation Institute. CCI 74903-0021
Figure 8. Water-damaged book showing mould growth.
© Government of Canada, Canadian Conservation Institute. CCI 127508-0010
Figure 9. Graph showing time required for visible mould growth.
Description of Figure 9
The x axis shows RH between 50 and 100%. The y axis shows the first mycelium or fructification in days, from less than one day to three years. The graph illustrates the relationship between RH, aw and mould growth:
- at 65% RH and 0.65 aw, experiments lasting three years show no noticeable mould growth
- at 75% RH and 0.75 aw, growth is noticeable between two weeks and three months
- at 80 to 90% RH and 0.80 to 0.90 aw, growth is noticeable between one and five days.
Gelatin and animal glue materials develop mould growth faster than book materials, and both can develop mould growth faster than the International Energy Agency (IEA) threshold, which runs from one month at 80% RH to one day at 100% RH. The IEA provides guidance on restricting time at elevated humidity to preserve a healthy indoor climate in modern buildings.
Points in the graph show the range of susceptibility for initial growth or fructification on various materials at room temperatures. Lines highlight gradation in risk from the most sensitive materials (animal glue) to the less sensitive ones (book materials) and the International Energy Agency (1991) threshold for operating healthy buildings based on avoiding Aspergillus versicolor (minimum aw 0.75). The spread of data points above the lines shows diversity in fungal response (species, substrate effects) and explains why giving a “hard number” is not straightforward.
Figure 10a shows the time it takes for moisture to permeate or leak through various barriers. It is assumed the conditions are at equilibrium. Note that by bagging an object, the response rate to moisture changes can be decreased by minutes and hours, up to a year.
Once the objects have been stored in polyethylene bags, it is still important to maintain the ambient temperature so as not to promote the growth of mould within the bags. Following are some suggestions for bagging objects (Strang 2001):
- bag objects with an ERH less than 65% and avoid elevating temperatures that would raise the ERH above 65%
- do not bag objects at an ERH above 65%
- do not store the bags on a cold shelf or countertop (this creates a temperature drop, which increases the RH in the part of the bag closest to the shelf)
- do not store the bag in a high RH environment because, eventually, water will permeate the bag (note that this permeation significantly slows down if the object is well wrapped in RH-buffering material and if the entrapped air volume is small when compared to the wrapped object)
Figure 10b graphically illustrates the above situations that may arise with bagged objects. A change in the ambient temperature will cause a change in the ERH. If the object was initially bagged at 55% RH (I) and there is a 15°C drop from the ambient temperature, the RH within the bag will climb to 80% (C). An increase of 15°C will lower the RH within the bag to 35% (W).
There is no single way or one instrument that can be used to measure the moisture content of all substrates. To determine the ERH, the following steps can be taken. Place the object into a clear, airtight container that allows one to read the hygrometer inside the container. Allow time for equilibrium to be reached. The reading on the hygrometer shows the RH value equivalent to the object's moisture content/water activity.
Ambient RH is often used to indicate mould growth potential. By keeping RH values below 60%, it is logical to assume that corresponding aw values in substrate materials would be limited to 0.6, which is the lowest aw level at which certain moulds can begin to grow.
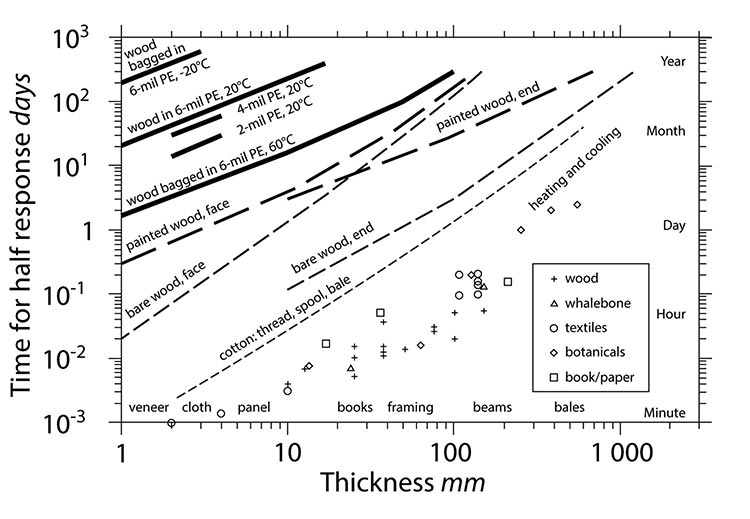
© Government of Canada, Canadian Conservation Institute. CCI 127508-0002
Figure 10a. Half-time response curve.
Description of Figure 10a
Figure 10a shows the time it takes for moisture to permeate or leak through various barriers, from the thin diffusion controlled layer of still air around everything to paint films and polythene bags. The x axis shows thickness of the barriers, from 1 to 1000 mm. The y axis shows the time for a half-time response in days, from one minute to a year. Wood bagged in 6-mil polyethylene at −20°C takes a year for a half-time response. Wood bagged in 6-mil polyethylene at 20°C takes between a month and a year for a half-time response. Wood bagged in 4-mil polyethylene at 20°C takes months for a half-time response. Wood bagged in 2-mil polyethylene at 20°C takes a month for a half-time response. Wood bagged in 6-mil polyethylene at 60°C takes between a couple of days and a year for a half-time response. The face of painted wood takes between less than a day and a year, while the end of painted wood takes between a couple of days and a year for a half-time response. The face of bare wood takes between an hour and a year for a half-time response, while the end of bare wood takes between half a day and a year. Cotton takes between a minute and a month for a half-time response for individual thread, a large industrial spool and a bale of raw cotton.
Points show that the temperature acclimation half-times for different sized pieces of wood, whalebone, paper, and textiles are more rapid by an order of magnitude than the thinnest of materials undergoing moisture change (cotton fibre). This means that the driving effect of changing temperature levels out long before any bulk moisture change.

© Government of Canada, Canadian Conservation Institute. CCI 127508-0003
Figure 10b. Changes to initial ERH caused by temperature differential within a vapour barrier enclosure.
Description of Figure 10b
The graph shows changes to initial ERH caused by a temperature differential within a vapour barrier enclosure of an object. The x axis shows initial ERH between 0 and 100%. The y axis shows the final ERH between 0 and 100%. The graph illustrates how a temperature gradient from one side to the other of the bagged object will cause a gradient in the ERH inside the bag. An object that was initially bagged at 55% RH (noted as “I” on the graph), if subjected to a 15°C (59°F) differential across the object will show an increase in RH within the bag to 80% on the cold side (“C” on the graph) and will lower the RH within the bag to 35% on the warm side (“W” on the graph). A consequence of prolonged exposure to a temperature gradient in storage is mould growth in bagged objects at the cooler, damp side. This is to be avoided by proper placement in storage that avoids temperature gradients.
Limiting the aw of materials in heritage collections to less than 0.75 (as described by Michalski 1993) will prevent mould problems from rapidly building up. However, maintaining a level less than 0.6 should be the primary goal.
In the event of water intrusion or elevated RH, remedial action must be taken as quickly and as efficiently as possible. Staff must be ready to respond to eliminate the moisture problem in order to avoid, or at least limit, a mould outbreak.

© Government of Canada, Canadian Conservation Institute. CCI 122867-0018
Figure 11. Improper storage limits air circulation.

© Government of Canada, Canadian Conservation Institute. CCI 127508-0012
Figure 12. Example of a mouldy box.
Air conditioning
It is crucial to control the moisture content of the air so that it does not exceed 60% RH when using air conditioning systems. This can be achieved if dehumidifiers are used in conjunction with large HVAC systems. Small, portable window air conditioners must be properly sized for the space. Window air conditioners tend to be cyclic. Warm, moist air passes over a refrigeration unit and is cooled. At the same time, moisture condenses and runs into a drip pan. As the refrigeration unit cycles, it shuts down and the frost/ice buildup melts into the drip pan. Because warm air is still being passed over this unit and the drip pan, it, therefore, picks up some of the moisture in the drip pan and carries it into the room. An effective way to ensure good humidity control is to also operate a dehumidifier. Domestic dehumidifiers with a capacity of 5.7 m3 (200 cu. ft.) per minute will handle a 339 m3 (12,000 cu. ft.) room (this equipment is capable of exchanging the room air once per hour). Be aware that the dehumidifier will not operate efficiently below 18°C (65°F) or 35% RH. Some dehumidifiers using desiccant wheels will work efficiently below 18°C (65°F). The dehumidifier must be properly sized for the space and the drip pans on both the humidifiers and air conditioners must be cleaned regularly.
Air circulation
Maintaining room RH below 60% may keep materials dry, but it does not eliminate the possibility of mould growing in local cold spots (exterior walls and corners) or of water leakage that may raise the RH near the substrate material over 70% (Flannigan and Miller 1993). For example, in a room with a temperature of 20°C (68°F) and 60% RH, the surface temperature of an exterior wall may be 15°C (60°F) and the resulting RH, 80%. The wall will absorb moisture, increasing the aw from 0.60 to 0.80 and enabling certain moulds to grow. If the surface temperature of the wall reaches dew point temperature,End Note 5 the resulting condensation may allow mould to grow despite the ambient RH being in the right range. Proper air circulation would help eliminate these concerns. It is very important not to store cardboard boxes next to a cold exterior wall because they will absorb moisture and be vulnerable to mould growth (Figure 12). In order to prevent this from happening, storage enclosures, objects or other materials should be located 35 to 45 cm (15 to 18 in.) away from outside walls. Maintaining this space will allow good housekeeping to be practised, some circulation, and access to the wall if water seeps through it.
Temperature
In many buildings, the temperature is determined by human comfort level, which ranges from 18°C to 24°C (65°F to 75°F). This temperature range is also within the optimal range for mould growth. In areas not normally occupied by humans (seasonal museums, long-term storage, etc.), a lower temperature will benefit the collection, provided the RH is also kept lower than 60%.
Preventive measures checklist
In summary, the following checklist of preventive measures can be used to maintain a stable environment and prevent mould growth in a collection:
- maintain RH below 60% (this includes preventing any temperature drops)
- maintain good air circulation
- conduct regular inspections of objects on display and in storage
- remove dust by instituting a program for regularly cleaning floors, shelves, cabinets, surfaces of materials, such as boxes, plastic coverings, etc.
- isolate incoming objects, examine them and, if necessary, clean off dirt and debris before housing the objects with a collection
- guard against water leaking into a building and a collection
- use ongoing programs to maintain environmental control systems, portable humidifiers and dehumidifiers
- always separate plants and food areas from object storage and display areas
After a water leak, immediate action is required to prevent the onset of mould growth:
- at the first sign of mould in a collection, isolate the contaminated object(s), locate the causative agent and correct the situation
- immediately begin—within 48 hours—cleaning up water spillage or leakage
- if RH is above 60%, circulate air and dehumidify to below 40% until materials are dry; once materials are dry, resume a rise to normal humidity
- the movement of dryer air into the problem area from another space or from the outdoors can quickly lower the RH and dry things out
- develop a disaster plan and, where appropriate, be prepared to either air-dry or freeze water-damaged objects within 48 hours
1.3 Mould outbreak — the first steps
The following information outlines a general course of action to deal with a mould infestation in a heritage collection—be it an archive, library, museum or gallery. It is intended as a guide to help with immediate decisions and first steps to control the infestation and to protect people and the collection. The information is appropriate for small (1 m2 or less of visible mould) to medium-sized (1 to 4 m2 of visible mould) outbreaks of mould. However, if the mould growth is large (4 to 10 m2 of visible mould) or extensive (>10 m2 of visible mould), professional remediation may be required.
Protect staff
Take preventive measures to protect staff working in the facility. People with allergies or those with asthma should not be in contact with affected material or find themselves where the mould infestation is located. For more information, refer to section 1.5 Health effects and section 2.1 Personal protective equipment.
Isolate objects
If only one or a few objects are contaminated, isolate them. If many objects are involved, isolate the collection area. This will prevent mould spores from dispersing into clean areas of a collection and the rest of the building. Objects can be isolated, and the contamination contained, by placing them in a sealed box or bag. If large, they can be wrapped in plastic sheeting. If the objects are wet or damp, this should be a temporary measure until they can be dried. If the objects are dry, they can stay in containers or wrapped in plastic until they are cleaned of visible mould growth. If the collection area must be isolated, seal the entrance and return air intake vents with 5 cm (2 in.) tape and heavy gauge plastic/polyethylene sheeting. This will prevent mould spores from entering clean areas of a building.

© Government of Canada, Canadian Conservation Institute. CCI 69081-0012
Figure 13. Covering a flag to prevent the spread of spores.
Isolate affected objects or contaminated areas and control access to them so as to expose people to mould as little as possible. People entering the isolated area or opening sealed objects should always wear the appropriate PPE. (Consult section 2.1 Personal protective equipment.) Determine the extent of the mould infestation, consider the available resources and determine whether the infestation can be handled in-house or whether outside help is required.
Identify and eliminate the causative agent
Determine the cause of the mould outbreak and take immediate action to correct it. This may include measures to lower the RH, to increase air circulation, to lower room temperature or to remove any standing water. If necessary, employ a company that specializes in desiccant drying to dry the affected facilities and furnishings, such as carpets and drapes.
Deactivate the mould
The mould is actively growing if it feels damp and smears when brushed or if a musty or mouldy smell is present. Deactivating the mould will stop its growth and prevent further damage to the objects. This can be done either by air-drying or by freezing. Once the object is dry, mould can be removed. Detailed information on cleaning procedures, techniques and equipment is presented in section 2. Collection recovery. If cleaning is delayed, keep the dry object isolated and well wrapped or in a sealed container until it can be cleaned. This will prevent any inactive, but still viable, spores from dispersing.
Air-drying
Objects can be air-dried by lowering the RH and increasing air circulation. Because the mould spores of most species are easily airborne, it is important to conduct air-drying in a way that does not disperse mould spores and to take measures to prevent the spores from dispersing throughout the building. Seal the return air vent and, if possible, open windows to vent air outdoors. Select an isolated room with a minimum of furniture that will be easy to clean afterwards. Allow objects to air-dry or use fans. If using fans, place them so that the airflow is directed away from the objects. This will limit spore dispersal and keep the drying process slow enough to reduce physical distortions, such as fine checks or cracks that can result from an abrupt decrease in the object's moisture content. Objects made of a thick layer of organic material or objects that are composed of different elements joined together (such as inlays or veneers) are the most likely to undergo high physical stresses leading to damage during air-drying. Once the object is dry, consult section 2. Collection recovery for detailed information on cleaning procedures for objects, furnishings and storage areas.
If air-drying indoors cannot be accomplished without dispersing fungal spores, it is preferable to either freeze the material, air-dry a few at a time in a fume hood or dry the material outdoors. Air-drying outdoors should be done on a clear day in a sheltered spot out of direct sunlight. Be sure to keep the objects away from people and building air-intake systems, and bring them in at night.
Freezing
Freezing is a quick method of killing actively growing mould. However, although a mould's vegetative growth will freeze and break down, the spores are able to withstand the cold temperatures and remain viable.
Freezing is a good option when there are numerous water-damaged or mouldy objects, as this method eliminates the urgency to safely dry all wet objects within a short time frame. But freezing is not appropriate for all objects. In general, it is safe for textiles, furs, feathers, leather, paper and wood. However, it is not recommended for glass plate negatives, oil paintings or acrylic paintings. If in doubt, check with a conservator first. Before freezing, seal the object in a clear polyethylene bag or wrap with polyethylene film and seal with tape.
Household horizontal chest freezers, which generally operate between -18°C (-64.4°F) and -28°C (-82.4°F), can be used for a small number of objects. Freezing on a large scale requires a larger freezer. Large walk-in freezers can be rented. Freezer trucks can also be rented and brought to a site. The labour-intensive drying process can then be postponed until the staff is fully organized and has secured the space, time, resources and people to deal with the objects.
1.4 Indoor fungal investigations
Begin an indoor fungal investigation if there are health concerns or symptoms from individuals, evidence of mould growth, a mouldy smell or if water has leaked into a collection or a building. An indoor fungal investigation may include visual observation, collection of surface and bulk samples and/or air sampling. The methods used are determined by established indoor air quality protocols for investigating indoor fungal contamination.
The investigation may also include reviewing the history of the building to find clues that might explain or help locate previous water damage that has supported mould growth which has not yet been seen. If people are experiencing negative health effects, a health evaluation or interview may be conducted. Adverse health effects may be related to a specific activity or place where mould is growing. By using this information, air-quality experts may be able to narrow down the source of the contamination more easily.
In general, identifying mould is not required in order to respond to an outbreak in a heritage collection, to remove visible mould from heritage material or to treat objects damaged by mould. Identifying mould is done for specific reasons; for example, if there is a health hazard concern or if some staff members have health problems.
The following sections provide basic information on techniques and equipment that can be used in an indoor air investigation. For more detailed information, consult the literature.
Visual observation
Visual observation consists of walking through a building or collection and using sight and smell to evaluate the indoor air. Observations may include noting the presence of humidifiers, standing water, water-damaged materials, poor housekeeping, poor ventilation and high RH. Often the first step in indoor air investigations, such observations may provide sufficient evidence to develop a remediation action plan for a building or collection.
Surface and bulk sampling
Surface samples may be taken with sterile swabs, or bulk samples (small pieces of contaminated material, such as wallboard or carpet) may be removed for testing. Both types of samples are analyzed by either direct contact with growth media or by diluting the sample and distributing a specific amount over a growth medium. After an incubation period, usually seven days, the mould colonies are identified and counted. The mould is either identified to a genus level,End Note 6 for example, Aspergillus, or to a species level, such as Aspergillus flavus, Aspergillus fumigatus or Aspergillus versicolor. If certain types of mould are found, it may be appropriate to identify the mould to the species level to determine if it is toxigenic. This level of identification may be necessary when health risks or concerns are an issue.
The results are reported as total colony forming units (CFU). Each species is identified as a percentage of the total. Generally, laboratories report surface samples as colony forming units per square centimetre of area sampled (CFU/cm2). Bulk samples are reported as colony forming units per gram of material sampled (CFU/g).
Bulk and surface samples may be useful to detect certain mould species. For example, the species Stachybotrys chartarum (previously known as Stachybotrys atra) is not easily measured using air samples because the spores, when growing, are wet and sticky and not easily airborne. Once they are dry and then disturbed, they can become airborne for short periods of time but soon fall because of their heaviness. The spores are normally found in the dust on the horizontal surfaces of furniture or floors. In many cases, Stachybotrys chartarum can be found only by bulk, surface or aggressiveEnd Note 7 air sampling.
Air sampling
Air sampling can be used to determine if higher-than-normal concentrations of mould spores are present in the environment. The technique may be used when mould is not clearly evident; for example, if mould growth is not visible, but individuals are experiencing unexplained health symptoms that might be related to exposure to bioaerosols.
When indoor air sampling is performed, a sample of outdoor air may be taken at the same time to use as a comparison. In general, when results indicate a higher level of fungal contamination in the indoor air and/or the type of mould species differ from those found outdoors, there is cause for further investigation.
Not all fungi or moulds can be found in air samples. It should be noted that an air sample indicates only the bioaerosol material present at that moment. Air sampling is effective when used before and after remediation. It can also be used during remediation, if part of the building is still occupied, to ensure that the air quality is fit for human habitation.
Sampling the air for mould involves using special sampling equipment. These instruments are used by industrial hygienists or by indoor air-quality experts, who follow a protocol determined by the manufacturer of the equipment.
Viable spore collection method
Collecting and culturing viable mould spores is a method that has been used extensively. The data derived from this method has been compiled into an accumulated database that is used to compare results. The method also helps distinguish one organism from another, although it also has disadvantages. The use of the growth media may create a culture bias. For example, some organisms may not grow well on certain growth media and others will have different competitive abilities against other mould species collected. These factors may influence the end result. The technique takes time—usually seven days to culture the initial sample, and as many as 15 to 20 days in frequent instances. If fast-growing cultures overpower slower ones, subsequent culturing may be necessary. Finally, the method does not detect non-viable or sterile spores in the indoor environment. Non-viable and sterile spores can still cause negative health effects.

© Government of Canada, Canadian Conservation Institute. CCI 74903-0029
Figure 14. Anderson single-stage air sampler with Petri dish.
This sampling technique uses an instrument that pumps a certain volume of air during a given time period through a specific pore-sized filter onto special plates or strips containing a nutrient medium that supports biological growth. The Anderson multi-hole impactor sampler (Figure 14) and the Reuter centrifugal sampler are two examples of indoor air-quality sampling equipment.
Once the samples have been collected, the plate or strip is removed and incubated at temperatures optimal for mould growth for a set period of time, usually seven days. The plate or strip is examined and the biological growth counted and identified. The results will be reported as colony forming units per cubic metre of air sampled (CFU/m3).
Non-viable spore collection methods
Non-viable methods, such as spore trap or tape methods, are used to collect particulate matter from the air, but the sample is not cultivated. As a result, one of the advantages of these collection methods is that the evaluation of the sample usually takes place within 48 hours. It is a useful sampling method because organisms that will not grow on growth media and/or do not compete well with other organisms may be detected. These methods, therefore, may yield a more complete representation of the fungal composition of total (viable and non-viable) spore levels. The disadvantage of these methods is the inability to distinguish between species that produce morphologically similar spores.
Spore traps, such as the “Air-O-Cell” from Zefon International and the MK3, use specialized equipment that collects the material from the air onto a sticky surface or a slide coated with a collection media. The slide can be immediately examined under a microscope and the spores counted. Because the physical attributes (size, shape, texture) of most spores are fairly specific, a general level of identification is possible. As well, certain moulds, such as Stachybotrys chartarum, are limited in species and easily identifiable by this sampling technique.
In some instances, further sampling by culturing may be recommended to identify the mould species and to determine if it is toxigenic.
Interpreting results
All sampling techniques require specific knowledge and expertise. This work is performed by a technically qualified professional who uses an accreditedEnd Note 8 laboratory for analysis. Identifying the fungi should be done by someone trained in mycology.
The indoor air-quality expert and the laboratory prepare a report and forward it to the client. The following points are typical of a good indoor air-quality report:
- description of the sampling methodology
- time and date when samples were taken and date when samples were processed
- location of each sample taken (room number, location in room)
- total number of particles
- number of particles per mould genus and species
The report should indicate if there are toxigenic mould species present. If so, a cautionary note should be included that underlines their potential as a possible health hazard. Recommendations about remediation or the appropriate level of PPE will be given. A list of references used in the identification process should also be included.
1.5 Health effects
Mould is ubiquitous. Normal background concentrations of mould do not usually affect healthy individuals. In contaminated environments, however, the risk of health effects from exposure to mould increases. Reactions are varied and depend on the nature of the species involved, the metabolic products being produced by these species, the amount and duration of exposure to mould and mould products, and the susceptibility of the individual. Roughly 8% of the population is predisposed to be affected by exposure to mould. As well, people with asthma or respiratory problems, those suffering from allergies or an allergy to mould, those with compromised immune systems and anyone taking steroids may be affected. Several studies have shown that the presence of fungi in the home can be equated to health effects on the occupants (Brunekreef et al. 1999, Dales et al. 1991a, b). Large areas contaminated with visible mould resulted in more symptoms. One study showed that those who had skin sensitivities to mould spores were at higher risk of severe asthma attacks (O'Hallaren et al. 1991).
Generally, health effects fall into the following categories: irritation, allergy, toxicity and infection (Ammann 2003), each outlined below.
Irritation
MVOCs, such as short-chained alcohols, aldehydes, esters and ketones, are produced by some fungi. They are responsible for the characteristic mouldy, musty, earthy smell associated with damp buildings. Although the health effects from exposure to MVOCs have not been well studied, they may be responsible for symptoms of headache, dizziness and eye and mucous membrane irritation among people living or working in fungal-contaminated buildings (Levetin 1995).
Allergy
Mould is a well-known source of allergens (any substance that causes an allergic reaction). Many, possibly all, fungal spores contain allergens. Mould spores do not have to be viable to retain allergenic properties. Exposure to dormant mould can trigger an allergenic response in individuals. People who are allergic may develop symptoms when their respiratory system or skin is exposed to mould or mould products.
People who suffer from asthma or allergies or who have become sensitized through prolonged association with mouldy materials or a mouldy environment may respond after inhaling spores with hay fever-like symptoms, such as runny nose, sneezing, red, itchy eyes and dermatitis (skin rash). A first-time exposure for a healthy person may result in a mild form of any of the above symptoms. Repeated exposure to mould may result in increased sensitization and a more intense response. Once the allergy develops, it may be a permanent, lifetime condition.
Although it is recognized that there is a cause and effect relationship between exposure to mould spores and allergic reaction, threshold levels either for sensitization or symptom development have not been clearly defined for any fungal allergen. People handling contaminated material should be advised of the risks that increase with the frequency and duration of exposure to mould. Exposure to mould may aggravate existing health conditions, such as asthma.
Allergy tests performed by physicians are for specific mould allergens. There are few mould allergens available to use for testing; therefore, while a positive result may indicate sensitization to a specific mould antigen, a negative test result does not rule out mould allergy for susceptible individuals (Ammann 2001).
Toxicity
The naturally occurring substances produced by some moulds that cause a toxic response are called mycotoxins.
Not all moulds produce mycotoxins, but many species do. The genera Aspergillus, Penicillium and Stachybotrys are usually found indoors and are known producers of mycotoxins. Sampling and species identification of mould by a mycologist can determine whether a mould is toxic. Viability of spores is not essential to toxicity. Non-viable spores can still be a source of toxins.
Mycotoxins produce a variety of health effects through ingestion, skin contact and inhalation. Depending on the kind of mycotoxins and the nature of exposure, effects may include mucous membrane irritation, skin rashes, dizziness, nausea, immunosuppression, birth defects and cancer. Nearly all of the mycotoxin literature focuses on ingestion exposure, although the role of inhaled mycotoxins in human disease is currently under study. In view of the potential severity of resulting disease, a conservative approach that limits exposure to mycotoxins is recommended (Burge and Otten 1999).
Infection
Infection from exposure to moulds that grow indoors is not common, but it can occur in susceptible individuals, such as those with a compromised immune system due to disease or drug treatment. For example, Aspergillus fumigatus is a weak pathogen capable of causing infections, called aspergilloses, in susceptible individuals. Infections can affect the skin, the eyes, the lung or other organs and systems (Summerbell 2001). Other fungi can also cause systemic infections, such as Coccidioidomycosis, Histoplasmosis and Blastomycosis. These fungi grow in soil or may be carried by bats and birds but do not generally grow in indoor environments (Dales and Miller 2001). Bird or bat droppings that have accumulated in air intakes, ducts and/or rooms frequently contain virulent pathogenic fungi. These organisms are not all reliably detected by sampling the air or droppings. Accumulated droppings should automatically be regarded as hazardous sources of pathogenic fungi. Appropriate action should be taken to safely remove any accumulations of bird or bat droppings (Health Canada 1995).
Symptoms
Symptoms that result from exposure to abnormal levels of indoor moulds, including toxigenic moulds, may include the following (Johanning and Landsbergis 1999):
- nasal irritation, burning, itchiness, stuffiness and congestion, bloody nasal discharge, throat irritation and soreness
- cough, shortness of breath, wheezing, chest congestion and tightness
- severe headaches, concentration problems, irritability, dizziness or lightheadedness, fatigue
- burning, irritation, blurry vision
- burning, skin rash
- low-grade fever, flu-like symptoms
People experiencing any of the above symptoms that may be related to exposure to abnormal levels of indoor moulds should consult their physician.
2. Collection recovery
This section informs the reader on how to remove mould growth from objects. It also describes the appropriate PPE to wear when working in a mould-contaminated environment or when working with mouldy objects.
Before beginning a collection recovery, follow the guidelines in section 1.3 Mould outbreak — the first steps, which outlines a general course of action to follow in response to a mould outbreak in a heritage collection, including identifying the causative agent and taking steps to correct it, how to protect staff, how to isolate affected objects and how to deactivate vegetative mould growth on objects.
2.1 Personal protective equipment
Mould is a serious health concern, so every effort should be made to limit human exposure to it. PPE that should be worn when handling mouldy objects or when working in mould-contaminated areas is described below. Under occupational health and safety legislation in Canada (federal, provincial or territorial regulations), an employer must take every reasonable precaution to protect the worker. Employers should provide training on the safe handling and cleaning of mould-contaminated objects and on the proper manner of wearing and maintaining PPE. Employees have a responsibility to follow these instructions.
Basing remediation decisions on the amount of visible mould growth is an approach that has been accepted in the field and is used by most guidelines. There is no conclusive research that specifies a level of personal protection that is appropriate at a certain number of square metres of contamination (Environmental Protection Agency 2001) and different guidelines will have some variation in size classification of visible mould. These guidelines are not derived from an examination of health effects. If in doubt regarding the appropriate level of PPE, consult health and safety experts. As well, consult a reputable supplier of safety equipment for information on specific products. Some companies have technical consultants trained to help select appropriate PPE.
The minimum level of personal protection suggested in this Technical Bulletin (Table 2) is based on the total surface area contaminated by visible mould growth: small (1 m2 or less), medium (1 to 4 m2), large (4 to 10 m2) or extensive (>10 m2). These levels and the general guidelines for PPE in Table 2 are adapted from guidelines found in the literature for mould remediation (National Collaborating Centre for Environmental Health 2014;End Note 9 New York City Department of Health and Mental Hygiene 2008).
The following sections describe the PPE appropriate for working with mould.
Respiratory protection
The American-based National Institute for Occupational Safety and Health (NIOSH) is the only North American organization to test and certify respirators. Accordingly, its standards are used as a benchmark by federal and provincial occupational health and safety bodies. Under a standard adopted in July 1998, there are nine classifications of air-purifying respirators based on filter efficiency and use limitations.
Generally, for mould infestations in heritage collections, particulate filters from the N series (N for not resistant to oil) are appropriate. Filters described as N95 filter at least 95% of airborne particles. Filters described as N100 filter at least 99.97% of airborne particulates and offer the greatest protection against particulate matter. These filters are also referred to as HEPAEnd Note 10 (high-efficiency particulate air) filters. When a mouldy smell is present, a filter that combines HEPA and organic vapour cartridges is recommended. For more information on organic vapour cartridges, refer to the section Respiratory protection and microbial volatile organic compounds.
Respirators, including disposable respirators, should be test fitted by a qualified individualEnd Note 11 to ensure a proper fit. It is useful to have several different styles and sizes of respirators on hand to find the right fit. Respirators should always be stored in a clean area or in a bag to avoid buildup of particulate matter on the respirator. At the end of each day of use, respirators should be cleaned according to the manufacturer's instructions. Used respirator filters will support mould growth. To prevent this, allow the respirator (and filters) to air out before storing them in a sealed bag.
Some individuals may be unable to wear disposable, half- or full-face respirators. Facial hair or unusual facial shapes may prevent the apparatus from establishing a proper seal against the face. Others may be unable to wear respirators for health or psychological reasons; for example, claustrophobics may experience extreme discomfort.
Disposable particulate respirators
Disposable respirators are inexpensive and maintenance free, but proper use requires careful reading and closely following the manufacturer's instructions. In order to get a good fit from a disposable respirator, follow the instructions on the package. For example, most models require that users fit the seal of the mask against the contour of their nose by pushing the nose area of the mask down against the bridge of their nose.
Disposable respirators are not appropriate for every individual. If improper facial contact is made with the respirator, it may actually increase the concentration of mould spores being inhaled. These devices, therefore, are not appropriate for people with facial hair. Although many brands of disposable respirators are available in one size only, some MSA models (Affinity Pro N95 and N100) are available in different sizes.
It may be difficult to find, and awkward to wear, protective goggles in combination with a disposable respirator. This is even more problematic when prescription eyeglasses are worn. Some combinations of respirator and goggles may work better than others.
If a disposable respirator is used more than once, be sure to air it out after use and store it in a clean environment. Stuff the inside of the respirator with clean tissue to ensure that it remains clean. If the respirator becomes damaged or soiled, or breathing becomes difficult while using it, discard it.
Half-face respirators
A half-face respirator consists of an assembled face piece worn over the mouth and nose. Cartridges selected to protect against a hazardous environment are attached to the face piece. Particulate and combination cartridges are available. Half-face respirators are more expensive than disposable respirators but are reusable because the cartridges can be replaced. Half-face respirators are available in different shapes, styles and sizes.
They are not appropriate for people with facial hair. It may be difficult, if not impossible, to accommodate protective goggles while wearing a half-face respirator. This may be even more difficult if a person wears prescription eyeglasses.

© Government of Canada, Canadian Conservation Institute. CCI 121761-0013
Figure 15. Conservator is wearing a N100 disposable particulate respirator and gloves while conducting a survey of a potentially mould-infested collection.
Full-face respirators
A full-face respirator consists of an assembled face piece worn over the mouth, nose and eyes. Cartridges selected to protect against a hazardous environment are attached to the face piece. Particulate and combination cartridges are available. Full-face respirators can be reused by replacing the cartridges, as required. Full-face respirators are available in different materials, styles and sizes.
A full-face respirator is not appropriate for people with facial hair. It is not necessary to wear protective goggles with a full-face respirator, but the seal of the respirator may be compromised if the person wears prescription eyeglasses. It may be necessary to consult with a reputable technical consultant of respiratory equipment to select the most appropriate full-face respirator. Full-face respirators or a powered air purification respiratory system are recommended when dealing with extensive mould growth.
Powered air purification respiratory systems
A powered air purification respiratory (PAPR) system is a positive-pressure airflow respirator that delivers a steady supply of filtered air. The user of a PAPR system wears a battery, fan, motor and cartridge on a belt. Filtered air passes through the breathing tube and into the headpiece. PAPR systems allow for up to eight hours of continuous use, after which the batteries must be recharged. PAPR systems protect individuals with facial hair and do not require test fitting. There are various headpieces, cartridges and combinations of cartridges available. Polyethylene non-woven (Tyvek) hoods and head covers are economical, disposable and offer maximum protection. The PAPR system is more expensive than other respiratory options and is recommended when dealing with extensive mould growth.
Two systems of note are the 3M Breathe-Easy PAPR system and the 3M Air-Mate HEPA PAPR system (HEPA 10 and HEPA 12). The two types of HEPA PAPR systems are less expensive but can be used only with a HEPA filter. The difference between the Air-Mate 10 and 12 is one of head covering. The Air-Mate 10 has a shoulder cape attached to the hood. The Air-Mate 12 has a hood. The Breathe-Easy PAPR system is more expensive, but it is also more versatile because a number of different cartridges or combinations of cartridges are available (such as a combination HEPA filter and organic vapour cartridges). If an adaptor breathing hose is also purchased, the Breathe-Easy PAPR system can be worn with either of the polyethylene non-woven (Tyvek) hoods.


Respiratory protection and microbial volatile organic compounds
Some fungi produce volatile metabolitesEnd Note 12 that cause unpleasant odours, including the characteristic mouldy smell associated with damp areas or materials. Although many different types of compounds have been identified, the MVOCs are primarily short-chained alcohols and aldehydes. The health effects of exposure to MVOCs have not been well studied. They may be responsible for headaches, dizziness and eye and mucous membrane irritation (Levetin 1995).
If a mouldy smell is present, a combination of HEPA and organic vapour cartridges is recommended. Organic vapour cartridges, not particulate respirators or cartridges, protect against MVOCs. Some particulate disposable respirators incorporate nuisance-level organic vapour relief and may be appropriate for some circumstances. Half-face and full-face respirators and some PAPR systems can be fitted with combination HEPA and organic vapour cartridges that protect against MVOCs.
Goggles
Protective goggles must also be worn when dealing with mould. The appropriate, recommended goggles are not ventilated and must accommodate a disposable or half-face respirator. If the user wears prescription eyeglasses, tight-fitting yet comfortable goggles may be difficult to locate. In this case, a full-face respirator or a PAPR system may be an option.
Protective splash goggles should be worn when working with wetting or cleaning agents (such as bleach).
Gloves
Protective gloves should be worn when handling mouldy material. Due to concerns about latex allergies, vinyl [poly(vinyl chloride) (PVC)] or nitrile gloves are recommended. Disposable gloves are replaced as required. Torn gloves should be replaced immediately. Disposable gloves are removed by grasping the cuff of one glove and peeling it off while at the same time turning it inside out. Once removed, this glove is balled up and placed in the palm of the hand still wearing a glove. As the remaining glove is peeled off and turned inside out, the balled up glove is covered up inside the worn glove (Figures 17a and b). This reduces the dispersion of mould spores by containing one glove within the other and by turning the gloves inside out, so the non-contaminated side of the glove is on the outside. Hands should be washed with soap and water after handling contaminated material, even when gloves have been worn.


Appropriate gloves must be worn when working with wetting or cleaning agents (heavy duty vinyl [PVC] or nitrile for high concentrations of bleach). Other suitable glove materials include natural rubber, neoprene and polyethylene.
Protective clothing
When dealing with mouldy material, protective clothing must be available for all personnel and properly worn and fastened. Coveralls and protective hair and shoe covers should be used when significant amounts of mould spores might be released into the air. Disposable clothing is recommended, but reusable protective clothing (lab coats or coveralls) may be appropriate for small and medium-sized levels of contamination. Protective clothing must not be worn outside the contaminated area. It should be removed on-site and washed in hot water and bleach.
Discarded disposable personal protective equipment
Always exercise caution when handling and discarding contaminated PPE. Place disposable clothing, gloves, etc., in thick (6 mil) plastic garbage bags or two layers of thin plastic garbage bags. Seal the bags and discard them in an outdoor garbage container.
Ear protection
When cleaning mould from objects involves using a vacuum cleaner extensively, noise levels may be a health and safety concern. Ear protection may be necessary when using machinery for long periods of time. Sound intensity is measured in decibels (dB[A]). Noise rated at 120 to 130 dB(A) causes pain, and sound above 140 dB(A) can harm ears permanently. The Canadian Centre for Occupational Health and Safety (CCOHS) reports that many regulatory agencies recommend a time-weighted sound level of 85–90 dB(A) as a noise exposure limit for an eight-hour work day.
Manufacturers of vacuum cleaners will provide the noise level rating for specific vacuum cleaners. For example, the Nilfisk GM-80 HEPA vacuum cleaner is rated as under 70 dB(A) at six feet. If earplugs are worn while working with mouldy material, they should be changed daily to reduce the possibility of microbial contamination.
Back safety
When mould remediation projects involve lifting heavy boxes, the risk of back injury may be a concern. The CCOHS has done research that proves back injury is the cause of one third of all lost work time and 40% of workers' compensation costs. The weight limit it recommends for workers is 20 kg. Lifting loads weighing over 20 kg creates too much stress on the back. An additional factor in back injury is working with heavy weight loads in confined spaces, which causes workers to twist and turn as they lift. The CCOHS also reports that employees required to manually lift or carry loads over 10 kg must be trained in safe lifting skills.
| Personal protective equipment | Level 1: small isolated areas (1 m2 of visible mould or less) |
Level 2: medium isolated areas (1 to 4 m2 of visible mould) |
Level 3: large isolated areas (4 to 10 m2 of visible mould) |
Level 4: extensive contamination (>10 m2 of visible mould) |
|---|---|---|---|---|
| Minimum respiratory protection |
|
|
|
|
| Other |
|
|
|
|
| Additional protection |
|
|
|
|
| Additional comments |
|
|
|
|
2.2 Cleaning mould-contaminated objects
This section addresses cleaning objects that have mould growth. This information can be used to design a collection recovery plan or to assess a plan submitted by outside professionals. Keep in mind that each institution will encounter a unique set of circumstances during a mould infestation; therefore, the information presented here may need to be modified or adapted. If possible, professional experience and judgment should be used to evaluate the level of mould contamination and to assess the proposed collection recovery plan.
If proper precautions are taken, small (1 m2 or less) or medium (1 to 4 m2) outbreaks of mould on objects can safely be cleaned. It is, however, possible to establish, after having evaluated the information presented here, that the situation cannot be resolved without professional help. In this case, consult professionals with experience regarding mould infestations to determine an appropriate course of action.
For large (4 to 10 m2) and extensive (>10 m2) mould contamination in a collection, it is advisable to seek professional help. Indoor air sampling, a site inspection and an evaluation of health risks by professionals may be necessary. A conservator with experience in the recovery of mould-contaminated objects can advise on the safest and most appropriate way to remove the mould growth or on which outside cleaning services to consider. Removing mould from objects on this scale will require a containment structure to minimize dispersion of mould spores as well as specialized cleaning equipment, procedures and PPE.
There is no research that proves that one cleaning technique over another is the appropriate one for a certain level of contamination. The following information is intended as a general guideline and is subject to the judgment of professionals with experience in cleaning mould-contaminated objects and working in contaminated environments.
Before cleaning objects
Before attempting any cleaning, consider the following:
- appropriate safety procedures to protect people and the work environment should be in place
- individuals with allergies or respiratory problems such as asthma and those with suppressed immune systems should not handle or clean mould-infested objects
- appropriate PPE must be worn
- respirators should be test fitted
- appropriate cleaning equipment and procedures should be in place
- objects should be examined for loose pieces and any detached piece should be either immediately secured prior to cleaning or else be bagged, and its location on the object, identified, so that it can be reattached later
- appropriate cleaning and disinfecting solutions should be used
- handwashing facilities should be located near the work area and must be used by staff before leaving the area
- all actions taken to remove mould should be recorded and retained as part of the documentation for the object; this should include details such as environmental conditions that may have led to the infestation, cleaning methodology and identification of the mould, if undertaken
Proceed with caution and, if in doubt, consult experts. Be prepared to stop and reassess the situation should mould levels be greater than originally anticipated. Be prepared to upgrade PPE, modify procedures or seek outside help, as required.
Can the mould be removed?
Take steps to deactivate vegetative mould growth before attempting to clean the object. Consult section 1.3 Mould outbreak — the first steps for information on how to do this. Test the mouldy area with a small brush to determine whether mould growth can be easily removed. In many cases, dormant mould can be removed by vacuuming and brushing. However, neither vacuuming nor brushing will remove mould stains from an object. These stains require treatment by a conservator and may prove difficult, if not impossible, to remove.
Prevent dispersion of mould spores
Mould spores are easily airborne on air currents or by HVAC systems, or they can travel on people to other areas of a collection and to clean areas of a building. Take the necessary measures to prevent mould spores from dispersing and to avoid contamination from infested objects spreading to non-infested or decontaminated objects (Florian 2000).
These measures include the following:
- Where possible, dry wet, infested material in a fume hood or class 1 biological safety enclosure.
- If this equipment is not available and it is necessary to dry wet, infested objects indoors, take precautions to prevent mould spores from dispersing by keeping objects covered with tissue paper during drying (this will capture any spores that become airborne as the object dries; if fans are used to dry mouldy objects, direct a gentle flow of air away from the object). If air-drying indoors cannot be accomplished without dispersing fungal spores, it is preferable to either freeze the material, air-dry a few objects at a time in a fume hood or dry the material outdoors.
- Isolate and store contaminated, dry objects in sealed containers or wrap and seal them in heavy plastic until they can be cleaned.
- Unwrap or open the container at the mould cleaning location (inside a fume hood, a class 1 biological safety enclosure or outdoors).
- Use a vacuum cleaner fitted with a HEPA filter.
- Vacuum objects in the room or immediate vicinity of the mould infestation because mould spores may be present.
- Clean or discard contaminated packing materials.
- Clean and disinfect the cleaning equipment, hand tools, storage area and furniture.
- Remove protective clothing by turning it inside out during disrobing.
- Discard mouldy materials (disposable PPE, packing materials, vacuum cleaner bags and filters) by placing them directly in thick (6 mil) plastic garbage bags or two layers of thin plastic garbage bags. Seal and discard the bags in an outdoor garbage container.
- Wash hands with soap and warm water immediately after removing protective gloves.
- Do not allow eating, drinking, chewing or smoking in the cleaning area.
Choosing a cleaning location
If at all possible, clean mould-infested objects inside a class 1 biological safety enclosure or fume hood. Using a brush or vacuum cleaner indoors without the appropriate mould containment enclosure is not recommended.
If a class 1 biological safety enclosure or fume hood is not available, cleaning mouldy objects outdoors may be an option. Choose a clear, dry day and set up away from people and the building's air-intake systems. Wear the appropriate PPE. After cleaning the objects or when finished for the day, clean and disinfect all work surfaces, tools and equipment before returning them to the building. Contaminated material, such as disposable PPE and any used packing or wrapping material, should be placed in thick (6 mil) plastic garbage bags or two layers of thin plastic garbage bags, sealed and discarded in an outdoor garbage container.
If inclement weather prevents cleaning outdoors, the dry objects can be wrapped in plastic or placed in a sealed container and, until suitable weather arrives, maintained in an environment that does not support mould growth. Under these conditions, the mould growth on the object and the deterioration of the object as a result of mould growth are stopped.
Equipment and tools
Equipment and tools used to clean objects include a vacuum cleaner fitted with a HEPA filter (and its accessories), brushes, a negative pressure fume hood and a class 1 biological safety enclosure.
Cleaning by vacuuming
Close vacuuming is an effective way to remove mould growth and reduce the number of mould spores on the object. Be systematic and thorough when removing the mould and carefully vacuum the object all over, not just where mould growth can be seen. Mould growth invisible to the naked eye may be present on unsuspected areas. If possible, go over the object at least twice. The second time, pass over the object in a perpendicular angle from the first vacuuming motion.
The following techniques should be considered for vacuuming:
- Apply the vacuum suction close to, but not directly on, the object.
- Dislodge tenacious mould with a soft brush, directing the mould toward the vacuum nozzle.
- Use a “light touch” with the brush to avoid embedding mould into the substrate, especially if it is porous.
- Use a mini-brush tool attachment or vacuum brush attachment for cracks or crevices.
- Vacuum flat, fragile objects through a cleaning screen.
Be vigilant and do not let the vacuum cleaner nozzle “get away from you” and accidentally touch the object. Vacuum suction applied directly to the object may cause damage. Devise a method to secure the vacuum cleaner hose when not in use during cleaning. This will keep the nozzle out of the way when the object is being repositioned. If working at a fume hood or class 1 biological safety enclosure, the contaminated vacuum cleaner hose and nozzle, as well as other cleaning tools, must stay inside the safety enclosure.
Vacuum cleaner fitted with a HEPA filter and accessories
A vacuum cleaner works well for cleaning visible mould growth from objects. To avoid dispersing mould spores into the environment, a vacuum cleaner fitted with a HEPA filter is strongly recommended. Vacuum cleaners with a variable speed control that can modify the vacuum suction are preferred.
Vacuum cleaner bags should be changed when they are half-full to keep the machine's suction at its maximum strength.
© Government of Canada, Canadian Conservation Institute. CCI 74903-0033
Figure 18. Conservator is working at a class 1 biological safety enclosure. The vacuum cleaner nozzle is secured inside the hood. This helps ensure the nozzle does not accidentally touch the object and that the contaminated nozzle remains in the enclosure.
Vacuum cleaner bags should be changed outdoors, in the fume hood or in the class 1 biological safety enclosure. Wear appropriate PPE when changing vacuum cleaning bags to avoid accidental exposure to mould spores. Discard contaminated vacuum cleaner bags (and filters) by putting them in thick (6 mil) plastic garbage bags or two layers of thin plastic garbage bags. Seal the bags and discard them in an outdoor garbage container.
Change the HEPA filter and pre-filter on the vacuum cleaner according to instructions provided by the manufacturer. Change the filters outdoors, in a fume hood or class 1 biological safety enclosure while wearing the appropriate PPE. Damp wipe the work surface and the exterior of the vacuum cleaner and vacuum hose to capture any mould spores.
Record the installation date of the new filters on the vacuum cleaner in a log book or in the manual. Depending on use and the level of contamination, the HEPA filter can remain effective for several years; pre-filters may need to be changed more frequently and will extend the life of the HEPA filter, if changed regularly.
A useful vacuum cleaner accessory is a mini tool kit (Figure 19). The kit contains small brushes and nozzles that come with an adaptor to fit onto any vacuum cleaner. Inexpensive mini tool kits are available at sewing, computer and vacuum cleaner stores.

© Government of Canada, Canadian Conservation Institute. CCI 74903-0013
Figure 19. Mini cleaning tools for the vacuum cleaner.

© Government of Canada, Canadian Conservation Institute. CCI 121761-0014
Figure 20. Conservator is using a cleaning screen, small brush attachment and low vacuum suction to clean fragile paper.
© Government of Canada, Canadian Conservation Institute. CCI 127508-0007
Figure 21. A protective screen over the vacuum cleaner hose.
Many objects can be cleaned through a cleaning screen (Figure 20). A cleaning screen is composed of fibreglass window screen held taut in a metal frame. When placed over the object during cleaning, it secures and protects the object while permitting the user to apply the vacuum nozzle close to it. This technique is suitable for flat, fragile objects. If the object is large, it is possible to vacuum through the screen, cleaning the object section by section. Cleaning screens can be made to order at window and glass suppliers. Alternatively, the screen can be loose, not held taut in a frame, providing that the sharp edges of the screening material are covered to protect the object. Twill tape sewn on the edges of the screen will work. A quicker method is to cover the edge of the screen with masking tape, providing the adhesive is not exposed.
The practice of covering a vacuum cleaner nozzle with window screen (Figure 21), to prevent small detached pieces of the object from being sucked into the vacuum cleaner, is often used when vacuuming objects. This technique prevents the loss of detached pieces and is suitable for vacuum cleaning most three-dimensional objects. When vacuuming flat, fragile objects, the cleaning screen described above offers additional protection.
A funnel or bell-shaped nozzle attachment for the vacuum cleaner can be useful for removing mould (Figure 22). Compared to a straight vacuum nozzle, it is designed to smooth the airflow. This increases the efficiency and capture rate and draws in more particulate matter toward the suction than does a straight vacuum cleaner nozzle. The attachment is most useful when using a brush to direct mould towards the vacuum nozzle.

© Government of Canada, Canadian Conservation Institute. CCI 74903-0016
Figure 22. Conservator is using a bell-shaped attachment on the vacuum nozzle and a soft brush to remove tenacious mould from a paper object.
Brushes
Brushes should have firm, but not rigid, bristles. It is useful to have several inexpensive brushes of different widths so that the best brush for the object to be cleaned can be selected. If necessary, cover the ferrule (the metal piece that holds the bristles onto the handle of the brush) with Teflon plumbers' tape so that the sharp metal edge cannot accidentally scratch the object. Brushes to remove mould should be labelled as such and not used on objects that are not mouldy.
Negative pressure fume hood
A negative pressure fume hood is designed to contain hazardous vapours and gases generated inside the hood and to exhaust them outside the building. Mould growth can be cleaned off objects by vacuuming them inside a negative pressure fume hood. After cleaning contaminated objects, the inside of the fume hood should be cleaned and disinfected so that other objects placed in the fume hood do not become contaminated.
Class 1 biological safety enclosure
A class 1 biological safety enclosure is a ventilated enclosure for personal protection in which the inward airflow is directed away from the operator. This type of enclosure meets the requirements of health and safety guidelines for working with mould (Health Canada 2004). The unit is fitted with a HEPA filter to protect the surrounding environment. During operation, the room air is drawn into the front of the enclosure, preventing aerosols from escaping into the room. Before leaving the enclosure, air is forced through an exhaust HEPA filter so that particulate-free air recirculates into the room. A class 1 biological safety enclosure is not a chemical fume hood and cannot be used for work with solvents.
If possible, mouldy objects should be cleaned inside a class 1 biological safety enclosure. Before using a class 1 biological safety enclosure, it must be certified by a qualified certification technician. An inflow velocity test and HEPA filter leak tests should be performed. Under normal operating conditions, the enclosure should be re-certified annually. If there is a pre-filter, it may need to be changed regularly to keep the unit working efficiently. The HEPA filter should be changed by the certifier. Usually, the filter requires changing every five years, depending on its use. If the class 1 biological safety enclosure is moved to a new location, it should be re-certified.
Few commercially available class 1 biological safety enclosures can accommodate oversized objects. If necessary, consider consulting the manufacturers of these products to custom-design a larger unit specific to the user's requirements.End Note 13 Otherwise, setting up outdoors to remove mould from oversized objects may be an option.
Working at a fume hood or class 1 biological safety enclosure
Follow the recommended practices for laboratory hoods; for example, keep all apparatus at least 15 cm back from the face of the hood (a strip on the bench is a good visual reminder) and do not put your head in the hood when contaminants are being generated. In order to ensure that the airflow operates at maximum efficiency, keep the slots in the hood free of obstruction and keep unnecessary items out.
In addition to safety practices for working at a fume hood or class 1 biological safety enclosure, it is important to implement a cleaning methodology that prevents dispersion of mould spores outside the laboratory hood. For example, do not unwrap or remove mouldy objects from the container until they are inside the fume hood or class 1 biological safety enclosure. Procedures may vary from one cleaning project to another and may depend on the number and type of objects to be cleaned. Providing precautions are taken, respiratory protection is not required while cleaning contaminated objects at a fume hood or class 1 biological safety enclosure. Protective clothing and gloves are still necessary.
Vacuuming or, if necessary, a combination of vacuuming and brushing towards the vacuum nozzle is recommended. This cleaning technique will keep the inside of the fume hood or class 1 biological safety enclosure cleaner.

© Government of Canada, Canadian Conservation Institute. CCI 84089-0001
Figure 23. A HEPA-filtered safety enclosure, custom-designed to accommodate large objects.

© Government of Canada, Canadian Conservation Institute. CCI 74903-0014
Figure 24. Microzone class 1 biological safety enclosure and vacuum cleaner fitted with a HEPA filter and variable suction.

© Government of Canada, Canadian Conservation Institute. CCI 84089-0002
Figure 25. Conservator is working at a HEPA-filtered safety enclosure.
How and when to clean and disinfect tools, equipment and work surfaces
To remove mould spores from tools, equipment and surfaces in the work area, fume hood or biological safety enclosure, start by using a vacuum cleaner fitted with a HEPA filter. Follow this by cleaning with a detergent solution, then, if necessary, with a disinfecting solution.
Cleaning solution
Work surfaces, tools and other equipment can be wiped down or washed with a water and detergent solution. Household detergent can be used.
Disinfecting solution
If necessary, work surfaces, tools and equipment can be disinfected with a dilute solution of household bleach and water. A solution of 0.5% sodium hypochlorite (household bleach) in water is recommended for disinfecting purposesEnd Note 14 (Maheux 2002). The bleach is diluted by adding 1 L of a 5% sodium hypochlorite solution (5% is generally the concentration of household bleach) to 9 L of water to make a final volume of 10 L. Alternatively, add 250 mL of bleach in 2.25 L of water, making a total of 2.5 L. In both examples, the bleach is diluted by a factor of 10.
Thoroughly wet the surface with the disinfecting solution. A contact time of between 10 and 15 minutes is required to ensure complete disinfection. Wear the appropriate PPE, respiratory protection, gloves and splash goggles when preparing and using a bleach solution. Once prepared, the bleach and water solution begins to lose its potency, so prepare only the amount that is required. Never mix bleach with ammonia or cleaning products that contain ammonia. The two products combine to create noxious fumes, which may be harmful.
Frequency of cleaning and labelling of equipment
During mould cleaning projects that extend over several days or weeks, clean the work surfaces and tools two or three times a week.
When mould removal is complete, all equipment and work surfaces (the vacuum cleaner, vacuum brush attachments, cleaning screens and brushes) should be cleaned, disinfected and thoroughly dried. Small hand tools, vacuum accessories and brushes can be stored in sealed containers. Label the container as containing equipment used to remove mould. Inexpensive or worn brushes can be discarded as contaminated garbage.
2.3 Cleaning the collection storage area
For small and medium levels of mould contamination, the affected storage area should remain isolated, its access should be restricted and the entrance should be draped with polyethylene until the area has been thoroughly cleaned. Large or extensive levels of contamination will require professional remediation.
Investigate the cause of the mould infestation and correct the situation before returning objects to the area. Clean and disinfect the storage area before returning any objects. This includes the walls, floor, ceiling and any furnishings, such as furniture, shelving units, books, papers, carpet, window coverings and computers. Exterior surfaces of a computer can be carefully damp-wiped with a cleaning solution. Typically, because of the airflow generated by the cooling fan, the inside of a computer is quite dusty. If a computer has been in a mould-contaminated environment, the interior may need to be cleaned. In such a case, consult a company that specializes in cleaning computers.
Use the cleaning and disinfecting solutions recommended above. Take the necessary precautions and wear the appropriate PPE.
Where possible, use cleaning methods that do not stir up dust. Damp-wipe hard surfaces, such as floors, walls and metal shelving units. Vacuum porous surfaces, such as window coverings, papers and fabric-covered furniture, with a vacuum cleaner fitted with a HEPA filter. If using a vacuum cleaner to clean the storage area, place the vacuum cleaner outside the room and use a long vacuum hose to vacuum. This will reduce air movement in the room and help prevent stirring up dust. Make several passes with the vacuum cleaner, in opposite directions on each pass, to pick up residual mould spores. Check ducts, filters and heat exchange coils in HVAC systems or other building/room features. If necessary, have them cleaned and disinfected.
Storage or packing materials with porous surfaces, such as cardboard boxes, that have mould growth should be discarded as contaminated material and replaced. If necessary, vacuum the exterior of the garbage bags before moving them, using the most direct route through the building to the outdoor garbage container.
2.4 Cleaning methods and considerations for some objects
Close overall vacuuming with a HEPA-filtered vacuum cleaner is an effective way to remove mould from objects. A conservator may suggest other cleaning techniques to further reduce the total spore amount.
Additional information and other cleaning techniques for specific types of objects are found in the Appendix: Removing Mould.
2.5 Considerations after cleaning
Objects cleaned for mould must be monitored and the health risk that these objects present must be managed.
Monitoring the collection
Objects infested with mould and then cleaned may be more susceptible to future mould growth than objects that have never been infested with mould. Objects themselves (or a selection of them if the numbers are large) should be visually inspected periodically (every six months). In the event of high RH or water intrusion, objects previously infested with mould should be inspected first.
Health concerns after cleaning
How clean is clean? Currently, it is not known to what extent infested but cleaned material poses a health concern. Thorough and conscientious cleaning will remove visible mould growth. Careful, overall vacuuming of the object will reduce the total amount of mould spores on the object. Unfortunately, other than a visual examination, no simple method, as yet, indicates how effectively a cleaning has been done, hence the importance of cleaning carefully and thoroughly, with the right equipment, according to recommendations.
However, even though an object has been carefully cleaned, some fungal spores and fragments, particularly on porous substrates such as paper, are likely to remain and may become a health concern for some people. Individual susceptibility will vary with genetic predisposition, age, health, and concurrent and repeated exposures.
There are no guidelines or regulations for handling objects after removing mould. It may be appropriate to adopt a cautious approach when handling and using these objects. For example, identify the object in a manner that allows users to take precautions before touching it. This should include wearing disposable gloves when handling the object and washing hands with soap and water afterwards.
There may be circumstances when information from mouldy books, papers and documents is transferred, under controlled conditions, to another format (photocopies). In some cases, minimal cleaning may be required before a good reproduction can be obtained. If the original material is retained, it should be carefully labelled to identify it as mould-infested and should be stored in an environment not conducive to mould growth.
Conclusion
Mould is a serious health concern and every effort should be made to limit human exposure to it. Actions to prevent mould infestation in heritage collections should primarily focus on measures to control the environment. In the event of a mould outbreak, the conscientious removal of visible mould growth and reduction of the total amount of spores from infested objects is necessary. If precautions are taken, small (1 m2 or less) and medium (1 to 4 m2) outbreaks of mould in a collection can safely be cleaned by following the guidelines in this Technical Bulletin. For large (4 to 10 m2) and extensive (>10 m2) mould contamination in the collection, it is advisable to seek professional help. When in doubt, always consult professionals with experience regarding mould infestations to determine an appropriate course of action.
Acknowledgements
The authors would like to thank the following individuals for generously giving their time to review the manuscript and offer advice:
- Dr. Cavaliere, Professor of Biology, Biology Department, Gettysburg College, Gettysburg, Pennsylvania;
- Diana Hobart Dicus, Objects Conservator, private practice, Boise, Idaho;
- Mary-Lou Florian, Research Associate, Royal British Columbia Museum, Victoria, British Columbia;
- Luc Maheux, Manager, Workplace Surveillance Unit, Workplace Health and Public Safety Programme, Health Canada; and
- Hanna Szczepananowska, Paper Conservator, Anthropology Conservation Lab, National Museum of Natural History, Washington, D.C., and Assistant Professor at the Anthropology Department, George Washington University, Washington, D.C.
Thanks to CCI colleagues for their comments on the manuscript. Special thanks to Tom Strang for his review and revisions in 2015.
Suppliers
Note: The following information is provided only to assist the reader. Inclusion of a company in this list does not in any way imply endorsement by CCI. Other companies may exist that offer similar products.
Personal protective equipment
PPE includes respiratory protection, goggles, gloves, protective clothing, head and shoe coverings and ear protection.
- Fisher Scientific
1-800-234-7437 - Acklands-Grainger
1-905-731-5516 or 1-866-248-8801 - Canadian Standards Association
(for more information regarding respirators, for example)
CSA Standard Z94.4-11 “Selection, Use, and Care of Respirators” (published 2011)
Vacuum cleaners fitted with a HEPA filter
Some brands of household vacuum cleaners are fitted with HEPA filters and may be appropriate for small cleaning projects; check local hardware, department and vacuum cleaner stores.
- Carr McLean
1-800-268-2123 (English)
1-800-268-2138 (French) - Nilfisk-Advance Canada Company
1-800-668-8400 - Nilfisk-Advance America, Inc.
1- 800-989-2235
Bell/funnel-shaped nozzle for vacuum cleaner
Can be custom made, available from:
- Safe-T-Air Inc.
1-877-692-0811
Mini tool attachments for vacuum cleaner
- sewing, computer and vacuum cleaner stores
Brushes
- local hardware or art stores
Window screen, cleaning cloths, garbage bags (heavy duty)
- local hardware, department and grocery stores
Sodium hypochlorite
- local grocery and hardware stores (household bleach in Canada is generally 5% sodium hypochlorite)
Class 1 biological safety enclosures
- Microzone Corporation
1-888-636-8609 - Labconco Corporation
1-800-821-5525
Custom-designed safety enclosure with HEPA filter exhaust hood
- Design Filtration Microzone Inc.
1-888-636-8609
Websites and other sources of information
Note: The following is provided only to assist the reader. Inclusion of a company in this list does not in any way imply endorsement by CCI. Other companies may exist that offer similar products.
General information
American Industrial Hygiene Association (AIHA)
ASHRAE
Position document on Limiting Indoor Mold and Dampness in Buildings
Canadian Centre for Occupational Health and Safety (CCOHS)
- CCOHS Publications
- CCOHS Course
- CCHOS Webinar
Health Canada
- Fungal Contamination in Public Buildings: Health Effects and Investigation Methods (PDF format) (2004)
- Mould
Institut national de santé publique du Québec (INSPQ)
Health Risks Associated With the Indoor Presence of Moulds: Summary Document (PDF format)
The National Collaborating Centre for Environmental Health (NCCEH)
The NCCEH website is an online resource for environmental health practitioners and policy-makers across Canada. It focuses on health risks associated with the physical environment and identifies evidence-based interventions to mitigate those risks.
- Health Effects from Mould Exposure or Dampness in Indoor Environments (PDF format)
- Mould Assessment Recommendations (PDF format)
- NCCEH Mould Investigation Toolkit (PDF format)
- Mould Remediation Recommendations (PDF format)
New York City Department of Health and Mental Hygiene
Guidelines on Assessment and Remediation of Fungi in Indoor Environments (PDF format)
Environmental Protection Agency
Laboratories
Hiring a conservator
- Canadian Association of Professional Conservators
- American Institute for Conservation of Historic and Artistic Works
Disaster recovery and remediation services
- Belfor (numerous branches in Canada, the United States and worldwide)
- On Side Restoration (branches across Canada)
1-888-663-6604
Appendix: Removing mould
The following table is intended as a general guideline only. Refer to the main text for specific information regarding the appropriate way to vacuum, PPE, and health and safety issues pertaining to cleaning objects with mould. Take steps to deactivate vegetative mould before cleaning the object. Consult section 1.3 Mould outbreak — the first steps for information on how to deactivate mould growth on objects. Before performing any treatment to remove mould, thoroughly examine the object to determine its condition and to find any loose or fragile components. If such components are found, secure them or be prepared to bag them and document their location if they become detached. Be prepared to document your actions. Any treatment to remove mould should be recorded and the documentation, retained. Neither vacuuming nor brushing will remove mould stains from an object. These stains require treatment by a conservator and may prove difficult, if not impossible, to remove. To determine if basic cleaning techniques (beyond vacuuming and brushing) are possible, consult the appropriate CCI Notes. Conservators should refer to the conservation literature for information about specialized treatments to remove mould growth or mould stains from objects.
| Object | Action |
|---|---|
| Water-damaged objects | Handle objects with care because they may be physically weak. A few objects can be dried by lowering the RH and increasing air circulation. Objects with mould growth should be air-dried in a manner that prevents mould spores from being dispersed near people or in a building. For example, if fans are used to dry a mouldy object, direct a gentle flow of air away from the object and cover it with tissue paper during drying to capture any spores that become airborne as the object dries. Alternatively, air-dry objects outdoors or in a fume hood or class 1 biological safety enclosure. Once the mould growth has been deactivated, the object can be cleaned, as described in this table and elsewhere in this Technical Bulletin. Freezing stops mould growth. Numerous water-damaged objects can be frozen and either freeze-dried or air-dried when time and circumstances permit. The hyphae (living filaments) of the mould will be killed by freezing, but the spores (reproductive bodies) will not. Mould spores can withstand freezing temperatures while in the dormant state, although they are less resistant to alternating freeze-thaw conditions. Active mould spores, however, will be killed by the freeze-thaw treatment. This method is suitable for numerous water-damaged or mouldy objects provided they are frozen and either freeze-dried or air-dried as soon as possible. For more information, refer to the conservation literature on disaster recovery for heritage collections. |
| Books | Inspect for mould on the inside and outside covers, the spine and throughout the text block. Mould may cause softening and weakening of the paper and cover boards. This softening will present handling and cleaning challenges. If books exhibit mould within the text block, each affected page should be examined and cleaned. If pages are not affected, keep the book firmly closed when cleaning the exterior. Use a vacuum cleaner fitted with a HEPA filter to vacuum the edges of the text block and cover boards. Mini tool attachments are useful for cleaning small crevices in the spine. Do overall vacuuming to reduce the total spore amount. If necessary, dry- or damp-wipe the cover boards after vacuuming. This will remove residual mould spores. Discard or wash cleaning cloths. For more information on the care and cleaning of books, specifically how to vacuum books, refer to CCI Note 11/7 Basic Care of Books. |
| Paper | Use a vacuum cleaner fitted with a HEPA filter. If necessary, use a brush to dislodge tenacious mould. If paper is fragile, carefully vacuum through a cleaning screen. Do overall vacuuming, on both sides, to reduce the total spore amount. After vacuuming, surface cleaning the paper with recommended erasing compounds may remove residual spores and mould fragments, as well as dirt that acts as a nutrient for future mould growth. The erasing compound will be contaminated with mould spores. Thoroughly remove it from the object and carefully discard it by sealing it in waste paper and placing it in the garbage. Consult CCI Technical Bulletin 11 Dry Methods for Surface Cleaning of Paper for more information on surface cleaning. Washing, solvent cleaning, enzyme treatments and bleaching may be further treatment options. However, they require the expertise of a paper conservator. Even so, stains caused by mould growth may not be completely removed. |
| Objects in containers (boxes, file folders, trunks) |
Remove object(s) from the container. Clean mouldy objects as described in this table and elsewhere in this Technical Bulletin. Examine the container for mould. Before discarding the container, make sure that it is not itself valuable. If it is, clean and retain it. If not, discard the mouldy container and rehouse the object in a new container. Transfer any information from the discarded container. Coroplast or Cor-X boxes can be cleaned, disinfected, dried and reused. |
| Parchment or vellum | Parchment and vellum are very susceptible to changes in RH. Documents may be folded, brittle and stuck together. Parchment and vellum documents should be unfolded only after careful humidification. Use a vacuum cleaner fitted with a HEPA filter. If necessary, use a brush to dislodge tenacious mould. If the document is fragile, vacuum through a cleaning screen. Do overall vacuuming, on both sides, to reduce the total spore amount. Because parchment and vellum are very sensitive to humidity, cleaning with moisture should be avoided. After vacuuming, it may be possible to surface clean the document with recommended erasing compounds to remove residual spores and mould fragments, as well as dirt that acts as a nutrient for future mould growth. The erasing compound will be contaminated with mould spores. Thoroughly remove it from the object and carefully discard it by sealing it in waste paper and placing it in the garbage. Consult CCI Technical Bulletin 11 Dry Methods for Surface Cleaning of Paper for more information on surface cleaning. Documents may have seals made of wax or shellac. These may be brittle and should be gently brushed rather than vacuumed. Vellum and parchment bindings in good condition can be cleaned in the same manner used to clean a book. Refer to CCI Note 11/7 Basic Care of Books for more information on caring for parchment and vellum bindings. |
| Photographs | Remove surface mould with gentle techniques, such as using a soft brush or mini brush attachments on a vacuum cleaner fitted with a HEPA filter. Do not use water or solutions containing water because mould may have made the photograph's emulsion water-soluble. Gelatin, the major component in the emulsion of films and prints, is an excellent nutrient for fungi. Fungal growth frequently concentrates around fingerprints on prints and film due to salts in the fingerprints, which create localized moist conditions. It is not recommended to treat photographs with fungicides; therefore, it is important to control RH in areas where photographs are stored. Alternatively, a suitable frost-free refrigerator will provide an excellent humidity controlled microenvironment for storing both colour and black and white photographs. Photographs must be packaged in envelopes or boxes and placed in polyethylene bags or wrapped with polyethylene, and the seams must be taped with freezer tape, before they are placed in a frost-free refrigerator (Wilhelm 1993). Consult a photographic conservator for more information before storing photographs in this manner. |
| Magnetic media | Compact discs: Isolate the affected disc from the rest of the collection. Place it in a low-humidity environment until cleaning can begin. A temperature of less than 23°C and a humidity less than 50% RH (down to 20% RH is acceptable) are recommended. Remove mould from the disc with a vacuum cleaner fitted with a HEPA filter. Brush tenacious mould off in a radial direction (perpendicular to the grooves) and direct the mould towards the vacuum cleaner. Once cleaned, the CD should be copied. Retain the original and store it in an environment that is not conducive to mould growth. Magnetic diskettes: Remove the mould growth from the diskette enclosure with a vacuum cleaner fitted with a HEPA filter. Remove the diskette from the enclosure. Refer to CCI Technical Bulletin 25 Disaster Recovery of Modern Information Carriers: Compact Discs, Magnetic Tapes and Magnetic Disks for instructions on how to do this. Follow the cleaning procedure described above for compact discs, except brush mould growth off in a circular motion. Magnetic tapes: Isolate the affected tapes from the rest of the collection. Place tapes in a low-humidity environment until cleaning can begin. A temperature of less than 23°C and a humidity less than 50% RH (down to 20% RH is acceptable) are recommended. To clean, use a vacuum cleaner fitted with a HEPA filter and remove the mould from the tape pack. Avoid any direct contact between the vacuum nozzle and the tape. Tenacious mould can be brushed from the tape pack using a soft brush. It is recommended to run the tape through a winder/cleaner because mould is very invasive and will grow between the strands of tape in a tape pack. The original tape should then be copied onto another tape. Retain the original and store it in an environment that is not conducive to mould growth. The recorder should be cleaned to avoid contaminating other tapes with mould spores (Iraci 2002). Additional cleaning procedures, such as those recommended for removing stubborn deposits from compact discs, magnetic diskettes and magnetic tapes, are possible. Consult CCI Technical Bulletin 25 Disaster Recovery of Modern Information Carriers: Compact Discs, Magnetic Tapes, and Magnetic Disks for more information. |
| Negatives, slides, microfilm and microfiche | Remove surface mould by gently brushing with a soft brush or with a mini brush attachment on a vacuum cleaner fitted with a HEPA filter. Remove slides from mounts (glass, cardboard or plastic), clean and return to new mounts. Wear clean cotton gloves to avoid putting fingerprints on the film. For more information, seek the expertise of a conservator. |
| Friable media (pastel, chalk, charcoal, powdery pigments on any object) |
Friable media is at risk of being dislodged by any cleaning method. Removal of mould, if at all possible, requires a high degree of manual skill and precision while working with fine tools. Seek the expertise of a conservator. The natural binders used in the manufacture of pastels make them particularly susceptible to mould growth. The surface of a pastel is very fragile. These artworks are among the most difficult to treat for mould growth. |
| Paintings | Deactivate mould growth. To avoid an abrupt change and extremely dry conditions, both of which can promote cracking in oil paintings, lower the RH conditions gradually, over one to two hours, and do not lower the conditions below 40% RH. Loosely wrap the painting to prevent dispersion of mould spores. Consult a paintings conservator for further advice. Due to the complex structure of paintings, permanent damage can result from even the most cautious attempts to clean a painting by untrained personnel. For more information, refer to CCI Notes 10/1 Cleaning Paintings: Precautions and 10/13 Basic Handling of Paintings. |
| Textiles | Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. If the textile is fragile, carefully vacuum through a cleaning screen. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming, on both sides, to reduce the total number of spores. Conservation treatments such as washing or dry cleaning may remove embedded spores and mould fragments, as well as stains and dirt that act as nutrients for future mould growth. Refer to CCI Notes 13/7 Washing Non-coloured Textiles, 13/9 Anionic Detergent, 13/13 Commercial Dry Cleaning of Museum Textiles and 13/15 Mould Growth on Textiles for more information. Stains resulting from mould growth may not be completely removed by washing or dry cleaning. |
| Basketry (items of woven plant material) |
Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. Examine the object for powdery pigment and evidence of use (such as seeds) before vacuuming. These elements are important and should not be altered or removed. If either are present, consult a conservator before cleaning. For more information on cleaning options, refer to CCI Note 6/2 Care of Basketry. |
| Leather | Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. For more information, refer to CCI Note 8/1 Removing Mould from Leather. |
| Wood | Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. When vacuuming furniture, remove all drawers and open all doors in order to completely clean the interior as well as the underside and other sides of the object. Water-damaged veneer surfaces of furniture may be poorly attached or folded away from the substrate and may also be brittle. To minimize the loss of veneer pieces, use a soft brush to gently dislodge mould, directing it toward the vacuum nozzle. Be sure the vacuum cleaner nozzle is covered with screening material. Additional cleaning techniques, such as surface cleaning and damp swabs for unfinished wood and damp cloth or solvent cleaning of painted finishes in good condition, may be possible. For more information, refer to CCI Notes 7/1 Care and Cleaning of Unfinished Wood and 7/2 Care of Furniture Finishes. |
| Ivory, bone, horn and antler | Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. Additional cleaning possibilities are found in CCI Note 6/1 Care of Ivory, Bone, Horn and Antler. |
| Glass and ceramics | Organic residues on non-porous surfaces will support mould growth. Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. Glass and ceramic objects in good condition may be further cleaned by washing. Refer to CCI Note 5/1 Care of Ceramics and Glass for information on how to proceed. |
| Metal | Organic residues on non-porous surfaces will support mould growth. Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. For additional cleaning possibilities, refer to the following CCI Notes: 9/3 The Cleaning, Polishing and Protective Waxing of Brass and Copper, 9/4 Basic Care of Coins, Medals and Medallic Art, 9/6 Care and Cleaning of Iron, 9/7 Silver – Care and Tarnish Removal and 9/9 Care of Objects Made of Zinc. |
| Rubber and plastic | Organic residues on non-porous surfaces will support mould growth. Use a vacuum cleaner fitted with a HEPA filter. If necessary, brush to dislodge tenacious mould. Mini tool attachments for the vacuum cleaner may be useful. Do overall vacuuming to reduce the total number of spores. For additional cleaning possibilities, refer to CCI Note 15/1 Care of Objects Made from Rubber and Plastic. |
Bibliography
- American Society of Heating, Refrigeration and Air-Conditioning Engineers (ASHRAE). 2013 ASHRAE Handbook – Fundamentals. Atlanta, GA: American Society of Heating, Refrigerating and Air-Conditioning Engineers, Inc., 2013.
- American Society of Heating, Refrigeration and Air-Conditioning Engineers (ASHRAE). 2011 ASHRAE Handbook: Heating, Ventilating, and Air-Conditioning Applications. Atlanta, GA: American Society of Heating, Refrigerating and Air-Conditioning Engineers, Inc., 2011.
- Ammann, H. “Is Indoor Mold Contamination a Threat to Health? (Technical Brief).” Journal of Environmental Health 64,6 (January/February 2002), pp. 43–44.
- Ayerst, G. “The Effects of Moisture and Temperature on Growth and Spore Germination in Some Fungi.” Journal of Stored Products Research 5,2 (1969), pp. 127–141.
- Brunekreef, B., D.W. Dockery, F.E. Speizer, J.H. Ware, J.D. Spengler and B.G. Ferris. “Home Dampness and Respiratory Morbidity in Children.” American Review of Respiratory Disease 140,5 (1989), pp. 1363–1367.
- Burge, H.A. Bioaerosols. Boca Raton, Florida: Center for Indoor Air Research, CRC Press Inc., 1995.
- Burge, H.A., and J. Otten. “Mycotoxins.” In J. Macher, ed., Bioaerosols: Assessment and Control. Cincinnati, OH: American Conference of Governmental Industrial Hygienists, 1999, p. 19-7.
- Cole, E.C., and K.K. Foarde. “Chapter 16: Biocides and Antimicrobial Agents.” In J. Macher, ed., Bioaerosols: Assessment and Control. Cincinnati, OH: American Conference of Governmental Industrial Hygienists, 1999, pp. 16-1–16-9.
- Dales, R.E., and J.D. Miller. “Building-related Illness: Epidemiological and Case-related Evidence.” In B. Flannigan, R.A. Samson and J.D. Miller, eds., Microorganisms in Home and Indoor Work Environments, 2nd ed. London, UK, and New York, NY: Taylor & Francis, 2011, pp. 217–228.
- Dales, R.E., R. Burnett and H. Zwanenburg. “Adverse Effects Among Adults Exposed to Home Dampness and Molds.” American Review of Respiratory Disease 143,3 (1991a), pp. 505–509.
- Dales, R.E., H. Zwanenburg, R. Burnett and C.A. Franklin. “Respiratory Health Effects of Home Dampness and Molds Among Canadian Children.” American Journal of Epidemiology 134,2 (1991b), pp. 196–203.
- Daniels, V., and B. Boyd. “The Yellowing of Thymol in the Display of Prints.” Studies in Conservation 31,4 (1986), pp. 156–158.
- Dicus, D.H. “One Response to a Collection-Wide Mold Outbreak: How Bad Can it Be - How Good Can it Get?” Journal of the American Institute of Conservation 39,1 (2000), pp. 85–105.
- Dillon, H.K., P.A. Heinsohn and J.D. Miller, eds. Field Guide for the Determination of Biological Contaminants in Environmental Samples. Fairfax, VA: American Industrial Hygiene Association, 1996.
- Environmental Protection Agency. “Remediation.” (PDF format) In Mold Remediation in Schools and Commercial Buildings. Washington, D.C.: Environmental Protection Agency/Office of Radiation and Indoor Air, Indoor Environments Division, 2008, pp. 9–26.
- Environmental Protection Agency. “Table 2: Guidelines for Remediating Building Materials with Mold Growth Caused by Clean Water.” (PDF format) In Mold Remediation in Schools and Commercial Buildings. Washington, D.C.: Environmental Protection Agency/Office of Radiation and Indoor Air, Indoor Environments Division, 2008, pp. 14–15.
- Federal-Provincial Committee on Environmental and Occupational Health. Fungal Contamination in Public Buildings: A Guide to Recognition and Management (PDF format).Ottawa, ON: Environmental Health Directorate, Health Canada, June 1995.
- Flannigan, B., and J.D. Miller. Humidity and Fungal Contaminants, Proceedings: Bugs, Mold & Rot II: A Workshop on Control of Humidity for Health, Artifacts and Buildings. Champaign-Urbana, IL: National Institute of Building Sciences, 1993.
- Flannigan, B., R.A. Samson, and J.D. Miller. Microorganisms in Home and Indoor Work Environments: Diversity, Health Impacts, Investigation and Control, 2nd ed. London, UK, and New York, NY: Taylor & Francis, 2011.
- Florian, M.-L. Heritage Eaters: Insects and Fungi in Heritage Collections. London, UK: James & James Ltd., 1997.
- Florian, M.E. “Aseptic Technique: A Goal to Strive for in Collection Recovery of Mouldy Archival Material and Artifacts.” Journal of the American Institute for Conservation 39,1 (2000), pp. 107–115.
- Florian, M.E. Fungal Facts: Solving Fungal Problems in Heritage Collections. London, UK: Archetype Publications, 2002.
- Griffin, D.H. Fungal Physiology. New York, NY: John Wiley & Sons, 1981.
- Gustafson, R. “Fungicidal Efficacy of Selected Chemicals in Thymol Cabinets.” Journal of the American Institute for Conservation 29,2 (1990), pp. 153–168.
- Health Canada. “Chapter 2: Biological Safety,” and “Chapter 4: Laboratory Design and Physical Requirements.” In Laboratory Biosafety Guidelines, 3rd ed. Ottawa, ON: Health Canada, 2004.
- Health Canada. Fungal Contamination in Public Buildings: Health Effects and Investigation Methods (PDF format). Ottawa, ON: Health Canada, 2004.
- Health Canada. Environment and Workplace Health: Mould in Indoor Air. Ottawa, ON: Health Canada, 2004.
- International Energy Agency. Annex XIV: Condensation and Energy, Vol. 2, Guidelines & Practice. Leuven, Belgium: International Energy Agency, 1990.
- Iraci, J. Disaster Recovery of Modern Information Carriers: Compact Discs, Magnetic Tapes, and Magnetic Disks. Technical Bulletin 25. Ottawa, ON: Canadian Conservation Institute, 2002.
- Johanning, E., and P. Landsbergis. “Clinical Findings Related to Indoor Fungal Exposure – Review of Clinical Data of Specialty Clinic.” In E. Johanning, ed., Bioaerosols, Fungi and Mycotoxins: Health Effects, Assessment, Prevention and Control. Albany, NY: Eastern New York Occupational & Environmental Health Center, 1999, pp. 70–78.
- Levetin, E. “Fungi.” In H.A. Burge, ed., Bioaerosols. Boca Raton, FL: Center for Indoor Air Research, CRC Press Inc., 1995, pp. 87–120.
- Macher, J., ed. Bioaerosols: Assessment and Control. Cincinnati, OH: American Conference of Governmental Industrial Hygienists, 1999.
- Maheux, L. Personal communication. Manager, Workplace Surveillance Unit, Workplace Health and Public Safety Programme, Health Canada, 2002.
- Manitoba Department of Labour and Immigration. Guidelines for the Investigation, Assessment and Remediation of Mould in Workplaces. Winnipeg, MB: Manitoba Department of Labour, Workplace Safety and Health Division, 2001.
- Michalski, S. “Relative Humidity: A Discussion of Correct/Incorrect Values.” In J. Bridgland, ed., ICOM Committee for Conservation 10th Triennial Meeting, Washington, D.C., 22–27 August 1993: Preprints. Paris, France: International Council of Museums Committee for Conservation, 1993, pp. 624–629.
- Michalski, S. Guidelines for Humidity and Temperature for Canadian Archives. Technical Bulletin 23. Ottawa, ON: Canadian Conservation Institute, 2000.
- Miller, J.D. “Mycological Investigations of Indoor Environments.” In B. Flannigan, R.A. Samson and J.D. Miller, eds., Microorganisms in Home and Indoor Work Environments. London, UK, and New York, NY: Taylor & Francis, 2001, pp. 231–246.
- Mullins, J. “Microorganisms in Outdoor Air.” In B. Flannigan, R.A. Samson and J.D. Miller, eds., Microorganisms in Home and Indoor Work Environments. London, UK, and New York, NY: Taylor & Francis, 2001, pp. 3–16.
- National Collaborating Centre for Environmental Health. Mould Remediation Recommendations (PDF format). Vancouver, B.C.: National Collaborating Centre for Environmental Health, 2014.
- New York City Department of Health and Mental Hygiene. Guidelines on Assessment and Remediation of Fungi in Indoor Environments (PDF format). New York, NY: New York City Department of Health and Mental Hygiene, 2008.
- Nittérus, M. “Ethanol as Fungal Sanitizer in Paper Conservation.” Restaurator 21,2 (2000), pp. 101–115.
- Nittérus, M. “Fungi in Archives and Libraries – A Literary Survey.” Restaurator 21,1 (2000), pp. 25–40.
- Ogden, S. Preservation of Library and Archival Materials: A Manual. Andover, MA: Northeast Document Conservation Center, 1994.
- O'Hallaren, M.T., J.W. Yunginger, K.P. Offord, et al. “Exposure to Aeroallergen as a Possible Precipitating Factor in Respiratory Arrest in Young Patients with Asthma.” New England Journal of Medicine 324,6 (1991), pp. 359–363.
- Onions, A.H.S., D. Allsopp and H.O.W. Eggins. Smith's Introduction to Industrial Mycology, 7th ed. London, UK: Edward Arnold, 1981.
- Price, L.O. Managing a Mold Invasion: Guidelines for Disaster Response (PDF format). Philadelphia, PA: Conservation Center for Art & Historic Artifacts, 1996, revisions 2009 and 2013.
- Samson, RA., E.S. Hoekstra, J.C. Frisvad and O. Filtenborg. Introduction to Food and Airborne Fungi. Utrecht, Netherlands: Centraalbureau voor Schimmelcultures, 2000.
- Shaughnessy, R., P.R. Morey and E.C. Cole. “Prevention and Control of Microbial Contamination.” In J. Macher, ed., Bioaerosols: Assessment and Control. Cincinnati, OH: American Conference of Governmental Industrial Hygienists, 1999, pp. 10-1–10-13.
- Strang, T.J.K., and J.E. Dawson. Controlling Museum Fungal Problems. Technical Bulletin 12. Ottawa, ON: Canadian Conservation Institute, 1991.
- Strang, T.J.K. “Principles of Heat Disinfection.” In H. Kingsley, et. al, eds., Integrated Pest Management for Collections: Proceedings of 2001: A Pest Odyssey. London, UK: James and James, 2001, pp. 114–129.
- Strang, T.J.K. Studies in Pest Control for Cultural Property. Gothenburg Studies in Conservation 30. Gothenburg, Sweden. University of Gothenburg, 2012.
- Summerbell, R. “Respiratory Tract Infections Caused by Indoor Fungi.” In B. Flannigan, R.A. Samson and J.D. Miller, eds., Microorganisms in Home and Indoor Work Environments. London, UK, and New York, NY: Taylor & Francis, 2001, pp. 195–215.
- Sussman, A.S., and H.O. Halvorson. Spores: Their Dormancy and Germination. New York, NY: Harper & Row, 1966.
- Szczepanowska, H. “Biodeterioration of Art Objects on Paper.” The Paper Conservator 10 (1986), pp. 31–39.
- Szczepanowska, H., and C.M. Lovett, Jr. “Fungal Stains on Paper: Their Removal and Prevention.” In J.S. Mills, P. Smith and K. Yamasaki, eds., The Conservation of Far Eastern Art: Preprints of the contributions to the Kyoto Congress, 19–23 September 1988. London, UK: International Institute for Conservation of Historic and Artistic Works, 1988, pp. 13–14.
- Szczepanowska, H., and C.M. Lovett, Jr. “A Study of the Removal and Prevention of Fungal Stains on Paper.” Journal of the American Institute for Conservation 31,2 (1992), pp. 147–160.
- Szczepanowska, H., and W. Momaw. “Laser Stain Removal of Fungus-induced Stains from Paper.” Journal of the American Institute for Conservation 33,1 (1994), pp. 25–32.
- Tétreault, J. Airborne Pollutants in Museums, Galleries, and Archives: Risk Assessment, Control Strategies, and Preservation Management. Ottawa, ON: Canadian Conservation Institute, 2003.
- Wellheiser, J.G. Nonchemical Treatment Processes for Disinfestation of Insects and Fungi in Library Collections. IFLA (International Federation of Library Associations and Institutions) Publication 60. Munich, Germany: K.G. Saur, 1992.
- Wilhelm, H. “Chapter 19. Frost-Free Refrigerators for Storing Color and Black-and-White Films and Prints: Recommendations.” The Permanence and Care of Color Photographs: Traditional and Digital Color Prints, Color Negatives, Slides, and Motion Pictures. Grinnell, IA: Preservation Publishing Company, 1993, pp. 658.
- Wilhelm, H. “Prevention of Fungus on Photographs.” The Permanence and Care of Color Photographs: Traditional and Digital Color Prints, Color Negatives, Slides, and Motion Pictures. Grinnell, IA: Preservation Publishing Company, 1993, pp. 560–561.
- World Health Organization. Laboratory Biosafety Manual, 3rd ed. Geneva, Switzerland: World Health Organization, 2004.
© Government of Canada, Canadian Conservation Institute, 2020
Published by:
Canadian Conservation Institute
Department of Canadian Heritage
1030 Innes Road
Ottawa ON K1B 4S7
Canada
Revised and corrected edition
First published in 2004
Cat. No.: CH57-3/1-26-2020E-PDF
ISSN 2562-0282
ISBN 978-0-660-32802-7