Caring for ceramic and glass objects
Judith A. Logan and Tara Grant
Caring for ceramic and glass objects is part of CCI 's Preventive conservation guidelines for collections online resource. This section presents key aspects of managing the care of ceramic and glass objects in heritage collections based on the principles of preventive conservation and risk management.
Table of contents
List of abbreviations
- CCI
- Canadian Conservation Institute
- portable document format
- RH
- relative humidity
- UV
- ultraviolet
Understanding ceramic and glass objects
Ceramics and glass are often thought of as being inert. Both are made from materials that have been altered by heat, but there are variations in their composition that affect their robustness and stability. Also, objects may have been modified through certain historic uses or due to their burial environment.
Ceramics
Composition of ceramics
Ceramics are as variable as the cultures and individuals who created them. In general, ceramics are made of fired clay with inclusions of other non-plastic materials, called “temper,” that reduce shrinkage and allow gases to escape when the ceramic is fired. The most common inclusions are crushed, previously fired ceramic (grog), chert, volcanic ash and quartz sand. The type of clay will determine the firing temperature of the ceramic, which affects its physical properties, such as porosity and hardness. The term “fabric” is used to describe the clay body of a ceramic and any inclusions. The fabric is sometimes sealed or decorated with glazes or slips. A combination of surface finishes may also be used.
The more highly fired a ceramic is (i.e. the higher the temperature used), the more impervious it is to its environment. It will still be brittle but more resistant to water. The porosity of the fabric, the type of surface finish and, to some extent, its colour are indications of a ceramic's firing temperature. Ceramics with porous, coloured fabrics—typically buff, red or red-grey—are fired at a lower temperature than ceramics with a white fabric.
Ceramic finishes
Glazes
Not all ceramics are glazed; glazes are added as decoration and to seal the surface of a ceramic. A glaze is a layer of glass formed over a ceramic fabric. It is formulated to match the thermal expansion and contraction rates of the fabric to which it is applied. Glaze is composed of silica with fluxes that lower its melting point, allowing it to fuse to the clay body during firing. Typical fluxes are oxides of lead, sodium and potassium. The temperature at which glazes form must be the same as, or below, the optimum firing temperature of the clay.
Glazes are the most common form of finish on European and Asian ceramics. The highest fired ceramics are the true porcelains, which have a white, non-porous fabric and a glaze that is very similar in composition to the fabric (Figures 1a and 1b). The glaze on Chinese porcelain, termed "feldspathic," appears to be an integral part of the porcelain body. The two fuse together so completely in the firing process that it is very difficult to see a boundary between the glaze and fabric.

© Government of Canada, Canadian Conservation Institute. CCI 79511-0002
Figure 1a. This doll's head is made of glazed porcelain. The ceramic material is fired at a high temperature to form a hard, glassy, non-porous material.

© Government of Canada, Canadian Conservation Institute. CCI 79511-0001
Figure 1b. Porcelain is very brittle and will break easily, as can be seen from the shattered head of this porcelain figurine.
Almost as highly fired as true porcelain is stoneware. Stoneware fabric is non-porous and is often greyish in colour. Stoneware glazes are often formed from the clay itself. Such is the case for salt-glazed and ash-glazed stonewares. Both salt and wood ash contain fluxes that melt the silica in the clay minerals on exposed clay surfaces, thus creating a very thin yet resistant layer. Salt glazes are described as having an "orange peel" or dimpled texture. Stoneware vessels have been used as storage containers due to their resistance to damage from water and chemicals.
The most common type of European ceramics in Canadian museum collections will have a glaze based on a mixture of lead oxides and silica. These glazes are clear or coloured and will be found on coarse earthenware as well as on more refined wares such as creamware, pearlware and transfer-printed earthenware (Figures 2a and 2b). Lead glazes are low to moderately high fired and their stability will depend on the ratio of lead to silica. Lead glazes can be damaged by acidic food and beverages and, because these glazes are softer than those on porcelain or stoneware, they are usually scratched if the object was used or handled a lot. Lead glazes may also have fine crazing (a pattern of small cracks) over their surface (Figure 3).

© Government of Canada, Canadian Conservation Institute. CCI 2005275-0001
Figure 2a. A 16th-century Hafner stove made of brightly coloured lead-glazed earthenware tiles. This object belongs to the Museum of Anthropology at the University of British Columbia (identification number: Ch267).

© Government of Canada, Canadian Conservation Institute. CCI 2005275-0003
Figure 2b. Detail of a tile from the 16th-century Hafner stove in Figure 2a made of a molded red earthenware body with lead glaze.
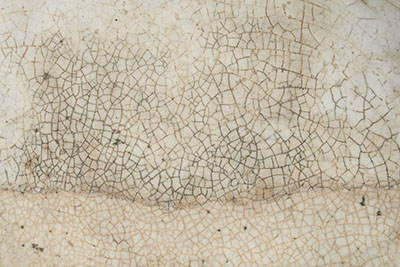
© Government of Canada, Canadian Conservation Institute. CCI 126393-0016
Figure 3. Crazing and staining typical of deteriorated lead glaze.
At the other end of the firing spectrum from porcelain is tin-glazed earthenware, which is composed of a soft, porous fabric and a soft, opaque white glaze that is sometimes poorly fused to the underlying ceramic. The opacity of the glaze is caused by the addition of tin oxide to a clear lead glaze. Objects made with this type of glaze are referred to as "faience," "Delftware," "majolica" or "tin-glazed." The white glaze is often painted with bright, fired-on patterns (Figure 4).

© Government of Canada, Canadian Conservation Institute. CCI 129979-0017
Figure 4. A majolica jug from a 16th-century Basque site. The coloured body is decorated with an opaque tin glaze to mimic more expensive white porcelain.
Glazed wares can be further decorated by applying designs under or over the glaze. Any design applied over a glaze is more susceptible to damage than a design under the glaze. A good example is gilding on dinnerware, which can be abraded away by repeated use.
Slips
Slips are coatings composed of liquid clay applied to an unfired ceramic. Slips are usually quite distinct from the underlying fabric but must match the firing properties of the clay body. Slips can be applied by dipping the unfired vessel into liquid clay or trailing and drizzling liquid clay on the fabric. Designs are often created by combining different colours of liquid slips into each other or by inscribing designs through the slip. Slips may be intrinsically glossy, or they may be covered with a glaze.
Burnishing
Burnishing, or smoothing a damp clay surface, aligns the clay particles and results in a thin, semi-lustrous finish when the object is fired. Burnished surfaces are usually red or black, with the colour coming from the iron oxides in the clay (Figure 5).

© Government of Canada, Canadian Conservation Institute. CCI 126209-0481
Figure 5. This pre-Columbian vessel was decorated with coloured slips and burnished to a lustrous surface. This object, which was a gift from Paul Maillot in 1986, belongs to the Leonard & Bina Ellen Art Gallery, Concordia University.
Paint
Sometimes decoration was painted onto fired ceramics, using coloured pigments or clays in a binding medium. These types of finishes are easily damaged. The term "paint" is also used to describe decoration that was hand-painted then fired onto ceramics, either under or over a glaze.
Forming a ceramic
There are many methods of forming a vessel or object from clay: casting liquid clay into a porous mold, throwing on a wheel, coiling and paddling, pinching, building in slabs and sections, pressing slabs into molds and sculpting. When handling ceramic objects, it is important to keep in mind that not all such objects were made in one piece. Handles, spouts and molded decorations are usually added to a previously formed vessel. These are held in place using liquid clay and are then fired on (Figure 6).
Firing ceramics
When clay is fired, the minerals in it undergo changes in their crystal structure. Each type of clay has an optimum firing temperature, which is the temperature at which it will become hard, or sinter, without sagging. In porcelains and stoneware, the altered clay minerals actually fuse together into a glassy matrix that gives them their characteristic vitreous quality. All clays shrink when fired.
The reds, greys and buff tones of earthenware are caused by iron oxides that change colours depending on the amount of oxygen in the air around the clay. Oxygen content will also influence the colour of glazes, since metallic oxides are the colouring agents in glazes.
Most glazed ceramics are high-fired in kilns in which the temperature and amount of oxygen can be controlled. Low-fired wares might have been fired in poorly regulated kilns, or in firepits, with little control over temperature and oxygen. It is possible to find a piece of ceramic with some sections underfired and some overfired. Low-fired pottery can be damaged by water.

© Government of Canada, Canadian Conservation Institute. CCI 128599-0152
Figure 6. The base of this tea cup has separated from the rest of the object due to extreme heat from a fire. The base was fabricated separately from the cup and attached with liquid clay before firing. The joints between the cup and the base are inherently weak and can separate when stressed by heat or cold.
Glass
Glass is an amorphous (non-crystalline) solid, consisting of a silica network in which other elements, such as fluxes and stabilizers, are bound (Figures 7a and 7b). Silica is difficult to melt since it has an extremely high melting point; as such, fluxes are added to lower the melting point. Common fluxes are soda (sodium oxide) and potash (potassium oxide) or a mixture of the two. A stabilizer, lime (calcium oxide), used to be added to make glass stronger and insoluble in water.

© Government of Canada, Canadian Conservation Institute. CCI 2002275-0001
Figure 7a. Glass bottles in a liquor chest.

© Government of Canada, Canadian Conservation Institute. CCI 2002275-0002
Figure 7b. Broken glass objects with mild staining of the glass due to the liquor contained in the bottles.
Common types of glass include:
- Soda-lime glass: made of silica melted with sodium and lime (calcium oxide) in varying proportions. Soda-lime glass is light and has been the most common type of glass manufactured from antiquity until the present.
- Potash glass: made of silica melted with potassium and lime in varying proportions. This glass is slightly denser but less durable than soda-lime glass. Potash glass was more common in Europe from the medieval period to the 19th century due to the lack of local sources of soda.
- Mixed alkali glass: a combination of sodium and potassium with lime in varying proportions. This glass is also less durable than soda-lime glass.
- Lead crystal: contains lead oxides (lead[II] oxide) instead of lime, often in a concentration as high as 20–30%. While not a crystalline structure, the material was named after rock crystals due to its high refractive index, which results in a brilliant clarity. It is considerably heavier than soda-lime glass. The lead lowers the melting temperature, making it easy to melt, shape, cut and engrave. However, it has poor durability and is easily scratched (Figure 8).

© Government of Canada, Canadian Conservation Institute. CCI 129979-0008
Figure 8. Regency-period (1844) lead crystal glass chandelier from the West Block, Parliament Hill, Ottawa, being carefully dismantled prior to renovations to the building in order to prevent it from being scratched or damaged.
Iron, copper, manganese and cobalt, either as impurities or additions, give glass colour. Some glass objects are further decorated by gilding, etching and grinding.
In the manufacture of glass objects, the glass is manipulated while it is in a semi-molten, plastic state. The rate of cooling of the object determines the amorphous quality and must be carefully controlled by the glassmaker. Glass objects are annealed, or reheated, shortly after they are formed in order to control their final physical properties.
Causes of damage to ceramic and glass objects and preventive conservation strategies
The primary danger to ceramic and glass objects is physical force. These materials are easily broken, chipped or scratched. If the ceramic or glass objects are stable, then other agents of deterioration such as relative humidity (RH) or light have minimal effect on them.
A small proportion of ceramic and glass objects are more susceptible to the agents of deterioration. While RH is normally not an issue for ceramics, absorption of chemicals such as salts may cause the object to be unstable in fluctuating RH. Ceramics and glass may have manufacturing faults that can become apparent long after the piece has been produced. Archaeological material may be more fragile due to the leaching of components during burial. All archaeological pieces are weaker, more prone to certain types of damage and may require specific care to prevent deterioration.
The following agents of deterioration should be considered for ceramic and glass objects.
Ceramics
The main agents of deterioration of ceramics are:
- physical forces
- water
- pollutants
- incorrect relative humidity
- manufacturing defects
Physical forces
The chief agent of deterioration for ceramics is physical forces—the brittleness and fragility of ceramics is well known (Figure 9). Handles, spouts and molded decorations are particularly prone to damage. These pieces are held in place using liquid clay before firing. Often, the joints are not strong, and sometimes an improperly secured handle may be attached only by a layer of glaze.
Ceramics can have traces of use, such as scratches or cracks in the glaze (Figure 10), worn overglaze decoration and possibly spalls in the glaze. Pieces created as artworks will not generally carry wear marks of this kind. Some decorative works of ceramic art may contain non-ceramic elements, such as paper, paints and lacquers. These are susceptible to damage through incorrect handling and cleaning.

© Government of Canada, Canadian Conservation Institute. CCI 126393-0019
Figure 9. A broken china plate showing the characteristic fragility of ceramics.

© Government of Canada, Canadian Conservation Institute. CCI 126209-0097
Figure 10. Ceramics, particularly those fired at lower temperatures, are easily scratched. This object, which was a gift from Paul Maillot in 1986, belongs to the Leonard & Bina Ellen Art Gallery, Concordia University.
Designs applied over a glaze are more susceptible to damage than designs underneath. Gilding on dinnerware, for example, can be abraded easily by repeated use and incautious handling or cleaning. Decorations painted onto fired ceramics are easily damaged.
Ceramic objects might have been broken and repaired and will be weaker at these points. Signs of repair are adhesive residues, glue lines, metallic staples or rivets (Figure 11) and infills or overpainting. Repairs are an important part of an object's history and should be documented.
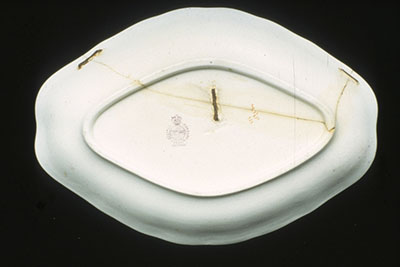
© Government of Canada, Canadian Conservation Institute. CCI 129979-0009
Figure 11. Old repair to a plate using metal staples. The metal staples have corroded, causing staining and discolouration of the plate. The adhesive has degraded and discoloured as well.
Water
Usually, water entering a museum accidentally is dirty and contains dissolved residues that can be absorbed by porous ceramics and can stain glazes by seeping into cracks. This also happens to archaeological ceramics buried in humid or wet soil (Figure 12). Low-fired ceramics, those fired below 600°C, can deform or disintegrate if soaked in water. Water-soluble tempers, such as gypsum in porous ceramics, can dissolve. Water can also damage materials that were used to repair ceramics, such as plaster or adhesives. Old repairs could discolour, swell or fall apart.

© Government of Canada, Canadian Conservation Institute. CCI 128599-0156
Figure 12. Iron staining under the glaze of a glazed plate. Excavated from a land site, it has absorbed iron from the burial environment through the cracks in the glaze into its porous ceramic body.
Pollutants
In a dusty or polluted environment, ceramics will become dirty if not protected from particulates. If dirt is left on the surfaces of objects, particularly porous, low-fired or unglazed ceramics, staining can develop. Dirt can also lodge in cracks and areas of repair, resulting in unsightly discolouration. Preventing the deposition of dust or dirt is important because dusting or washing ceramics increases the risk of damaging the object during handling. Staining may occur in both the fabric and glaze from materials that have been absorbed during use or during burial for archaeological material. Salt, iron and oil are common contaminants. Iron will form unsightly rust-coloured or blue-black stains, which are difficult to remove, while oil will form dark grey patches. Salty ceramics, if kept in fluctuating RH, can be physically damaged by repeated crystallization of the salt (for more information, consult Agent of deterioration: incorrect relative humidity). In the case of archaeological and ethnographic ceramics, remnants of food or black fire marks may be indications of historical use and should be preserved.
Incorrect relative humidity
RH is not a concern for most ceramic collections, as the constituent materials of their fabric and glazes are not affected by ambient RH. However, at high RH (above 65%), mould can occur on ceramic surfaces if there are even small amounts of soiling, oils and food residues. Earthenware objects are more likely to be affected due to their porosity. Consult Agent of deterioration: incorrect relative humidity, which provides a chart indicating the rate of mould growth by humidity level.
High RH can also cause water-sensitive adhesives used in repairs to become tacky and, above 75% RH, weak enough to risk releasing fragments.
RH is, however, a greater concern for ceramics contaminated with salts. When the RH rises above a certain critical RH, the salts deliquesce (they absorb enough moisture to form a solution) and later will crystallize when the RH falls below this critical RH. During the drying and crystallizing stage, the salt solution moves towards the zone of evaporation and crystals grow to the point of exploding the pore structure. The resultant flaking of the ceramic is called spalling (Figures 13a, 13b, 13c and 14).

© Government of Canada, Canadian Conservation Institute. CCI 129979-0010
Figure 13a. Progression of salt spalling shown in an experimental ceramic vessel that was filled with a salt solution. The salts are absorbed into the vessel but, as moisture evaporates, the salts crystallize out on the surface.

© Government of Canada, Canadian Conservation Institute. CCI 129979-0011
Figure 13b. Heavy salt contamination can result in the entire surface being covered with salt crystals.

© Government of Canada, Canadian Conservation Institute. CCI 129979-0012
Figure 13c. When salts are removed, there is evidence that the original surface has spalled off or been lost.

© Government of Canada, Canadian Conservation Institute. CCI 0000927-0001
Figure 14. Detail of detached and incipient salt spalls inside a bowl. This glazed ceramic, recovered from a saltwater environment, shows the round, shallow divots characteristic of salt damage.
The most common soluble salts are chlorides found in seawater, urine, blood, acids, bleaches, leather tanning products and salt used in food preparation and preservation; nitrates found in decaying organic materials, urine and gunpowder; and phosphates found in ash from kiln sites. Pure salts of chlorides and nitrates deliquesce (absorb moisture from the air to form a liquid compound) at around 70–76% RH; however, mixtures of soluble salts deliquesce at an RH significantly lower than that of pure salts (consult Rörig-Dalgaard 2014), so RH should be maintained well below this level. Since, experimentally, salt mixtures have been shown to deliquesce at an RH as low as 53%, it would be best for these items, to be safe, to remain below 50% RH. Fluctuations in RH above and below a critical RH which cause salts to deliquesce and recrystallize are a major cause of damage and should be avoided.
Spalling is well known in archaeological ceramics from salty environments, but this can also happen to non-archaeological objects that once contained salty solutions, particularly those with unglazed, poorly glazed or crazed surfaces. Chamber pots are a good example: spalling of the glaze might be observed in the lower portion.
Manufacturing defects
As part of the manufacturing process, some pieces may be damaged by mistakes made during forming, glazing and firing. If these defects are severe, the piece may be discarded by the manufacturer. Ceramics are graded by their quality: the best being the most expensive, and the seconds, with minor imperfections, being cheaper. Canada, as an English colony, was a market for wares produced by British potteries, and most of the ceramic tableware exported to colonial markets was not of top quality. Mistakes or sloppiness during manufacture can result in poorly attached handles and spouts, microfissures within the fabric, poorly fused overglaze decoration, unstable glazes, kiln marks in the glaze, unglazed areas and misshapen pottery. This should be kept in mind when handling pieces in museum collections, as these objects may be more susceptible to physical forces and damage from water or pollutants.
Glass
In a museum setting, well-made glass is fairly inert. Water in the atmosphere is the greatest enemy. Excess humidity can cause the dissolution of silica, particularly for poorly formulated glass. A small proportion of glass can be classified as chemically unstable. Experimentation to produce crystal clear glass that was easier to work with resulted in the reduction of lime (calcium oxide), a critical stabilizer for glass. This glass is referred to as crizzled or weeping glass. Unstable glass also includes some archaeological glasses, as burial conditions have often caused some degree of leaching. These objects will be more sensitive to the agents of deterioration.
The main agents of deterioration of glass are:
- physical forces
- water
- pollutants
- light and ultraviolet
- incorrect relative humidity
Physical forces
Like ceramics, glass is brittle and the most common form of damage is breakage. Since glass is amorphous, once a crack has been propagated, it tends to continue unimpeded due to the lack of crystal boundaries. Glass undergoes a certain amount of elastic deformation when it is broken; for this reason, it is difficult to align broken shards of glass.
Breakage is particularly noticeable in glass due to its transparency. Even after a professional repair, breakage will be visible (Figure 15). Stable adhesives that minimally discolour with time should be used. Adhesives that match the glass's refractive index can be used to create a less noticeable glue line under normal lighting. Consult Koob (2001, p. 48) and Down (2015) for more information on adhesives used for glass objects and on matching the refractive index between an adhesive and glass.

© Government of Canada, Canadian Conservation Institute. CCI 0000608-0001
Figure 15. A 16th-century broken archaeological glass vase. Breakage is particularly noticeable in clear glass even after professional repair.
Glass objects might have been repaired, and if so, they will be weaker at these points. Signs of adhesive residues, glue lines and infills are all indications of repair. Repairs are an important part of an object's history and should be documented.
Water
Historic glass, being a non-porous material, is not greatly affected by water over the short term. However, in the case of archaeological glass excavated from wet burial sites (pH 9 or lower), the alkali component of the glass will have been leached from the surface, forming iridescence. Multiple layers of remaining silica interfere with the transmission of light to create the iridescence. Extremely degraded archaeological glass will have many layers of fragile iridescent glass, referred to as "onion skin" degradation, which will flake or crumble when touched. This glass is too fragile to wash (Figure 16). Glass from burial sites where the soil's pH is very alkaline (greater than pH 9) will lose its silica component and the surface will appear etched.

© Government of Canada, Canadian Conservation Institute. CCI 128599-0162
Figure 16. Iridescence on glass is due to loss of the alkali component, which creates multiple layers that interfere with light transmission. The condition is sometimes called "onion skin." The surface of such glass is very fragile.
Very deteriorated glass, such as archaeological specimens, is porous and can absorb not only water (from leaks, floods, etc.) but also any dirt, minerals or organic extracts dissolved in the water, resulting in staining or physical damage (Figure 15). Further information on the effects of water on glass can be found under Incorrect relative humidity.
Lead crystal containers, such as decanters, that were used to store liquids often become cloudy on the inside. This is usually a sign of deterioration due to use. The lead has been leached from the glass and the surface will appear cloudy. Fluids left in lead crystal containers for long periods may become contaminated with lead and become toxic.
Pollutants
Like ceramics, in a dusty or polluted environment, glass will become dirty if not protected from particulates. Even slight amounts of dust will be visible on the transparent surfaces of glass, resulting in an unaesthetic appearance. Preventing dust deposition is far safer than having to deal later with dusting and cleaning—which may not always be safe or recommended, and which, at the very least, involves handling and therefore unnecessarily increases the risk of physical damage.
Because of the potential for deteriorated glass to attract moisture, dust and dirt can stick to the fragile surface and can be extremely difficult to remove without damaging it. Conversely, dust and dirt are hygroscopic and aid in attracting atmospheric moisture to the surface, where it can contribute to deterioration (consult Incorrect relative humidity for more details).
Light and ultraviolet
Some types of glass were tinted with manganese oxide to make them appear colourless. If exposed to ultraviolet (UV) light, this type of glass can turn purple. If glass contains a small amount of arsenic and selenium, it will develop a brown tint. This shift in colour is called "solarization." In some cases, this is considered aesthetically pleasing, but it is not the object's original colour. Solarization only occurs after long periods of high-intensity UV exposure. There is a low risk of this occurring under the low to moderate light levels and filtered UV typically found in a museum environment. However, adhesives used to repair glass may become discoloured from exposure to light and UV.
Incorrect relative humidity
Most glass in collections is stable and can be safely stored at an RH between 40–50% (Koob 2006, p. 133). Fluctuations in RH should be avoided. Unstable glass, however, requires extremely tight control of the atmospheric environment to slow the deterioration from glass disease. The proportion of stabilizers within the glass determines its chemical stability. Glass that has less than 4% lime (calcium oxide) will be extremely sensitive to moisture. In soda-lime glass, the excess sodium in the glass absorbs moisture directly from the air to form sodium hydroxide, a strong base, which then dissolves the silica in the glass. It is a cyclic reaction, as the removal of silica frees up more sodium for reaction. The glass appears foggy and has a soapy feel (Figures 17a and b). At a higher RH, the result is weeping, a condition in which the glass surface becomes damp and forms droplets. Initially, washing will remove the hazy layer and return the transparency of the glass; however, over time, as more damage occurs, the result is permanent and the cloudiness cannot be removed. Leaching of the silica produces very fine cracks in the glass. Strict control of the RH is essential for unstable glass. Sodium deliquesces at 42% RH; therefore, to prevent severely deteriorated glass from deteriorating even more, the RH must be lower than 42%. However, at an RH below 30%, severely deteriorated glass will desiccate and turn opaque as a network of fine cracks is exposed. This condition is called "crizzling" and is irreversible.
Potash-based glass is more sensitive to leaching. Placing an unstable glass object in direct sunlight will cause the RH to drop drastically due to localized heating, which will desiccate the glass and cause crizzling. As well, heat from lights in display cases should be avoided as this may cause local desiccation and crizzling. If not kept in stable RH, crizzled glass will eventually disintegrate.

© Government of Canada, Canadian Conservation Institute. CCI 126827-0007
Figure 17a. Due to a lack of stabilizing lime flux when the glass was first made, this wineglass is unstable. The foggy appearance is a classic sign of unstable glass and is due to the sodium component of the glass absorbing moisture from the air to form sodium hydroxide. At a pH of 10, the sodium hydroxide will dissolve the silica component. At a higher RH, the sodium hydroxide will form droplets, referred to as weeping glass. Unstable glass will continue to deteriorate if not stored below 42% RH.

© Government of Canada, Canadian Conservation Institute. CCI 126827-0011
Figure 17b. Detail of unstable glass from Figure 17a.
Even stable glass, if stored in extremely high RH, can develop glass disease. Microclimates, created by enclosed spaces such as the inside of decanters, clock faces, daguerreotype covers or framed artwork, often produce moist environments that can lead to crizzling. Dust and other pollutants attract moisture and can contribute to the problem. The alkaline deposits of weeping glass beads, with a pH of 10 or more, can damage the textile or leather substrates to which they are sewn, as well as the thread holding the beads onto the object. Beads may become detached or lost. Consult Deteriorating glass beads for a case study of this type of deterioration.
General preventive conservation strategies for ceramic and glass objects
Ceramics and glass objects have aesthetic appeal, whether they are utilitarian or made primarily as works of art. Damage, such as breaks, chips, cracks and scratches, can detract from the aesthetic appeal. These forms of damage can be reduced by proper handling, storage and display techniques.
Unstable glass objects and ceramics that are suspected of containing salts should be segregated and treated separately to provide them with enhanced environmental and storage protection against potential risks of physical and chemical damage.
Handling and storage
Ceramics and glass should be handled carefully. Damage commonly occurs when the objects are dropped or knocked against a hard surface, or when they fall off a shelf. Handling, moving and cleaning are times of highest risk.
Recommendations
Handling ceramics and glass requires care and common sense:
- Do not handle unless necessary.
- Handle extremely carefully and plan all moves to prevent breaking the object.
- Support the object with one hand directly under its centre of gravity, and use the other hand to stabilize it and prevent any movement.
- Handle with clean hands to prevent transferring oils or salts to the surface. Do not use hand lotion.
- Do not wear gloves when handling as gloves can be slippery, especially cotton ones. For unstable glass where wearing gloves is necessary, use nitrile gloves as the glass should not be exposed to moisture or salts from the hands.
- Never pick up objects by handles or spouts since these could break off (Figure 18).
- Beware of loose pieces, such as lids, as these may fall and break when handled.
Consult Handling heritage objects for more guidance.

© Government of Canada, Canadian Conservation Institute. CCI 129979-0013
Figure 18. A poor joint on a porcelain teacup. The person handling the cup should not be picking it up by its handle as the joint is at risk of failing. The adhesive was applied too thickly and has become discoloured.
Padded storage containers and enclosures, including closed storage and display cabinets, will protect these objects from physical damage as well as dirt and dust. Safe storage measures should include the following:
- Protect all objects from dust, thus reducing the need to clean them.
- Enclose water-sensitive objects in waterproof containers.
- Do not stack objects, as the excess weight can cause abrasions and stress.
- Pad out items to prevent rolling or touching of other objects, which may cause abrasion (Figures 19, 20 and 21).
- Provide tall or unstable items with form-fitting mounts to prevent them from tipping and breaking.
- When moving fragile ceramic or glass objects, place them in padded boxes to prevent them from touching each other or rolling.

© Government of Canada, Canadian Conservation Institute. CCI 129979-0014
Figure 19. Large, coarse earthenware vessels held securely on a storage shelf. The fabric of this type of pottery is relatively soft and can be abraded if the pots are allowed to rub against each other.

© Government of Canada, Canadian Conservation Institute. CCI 129979-0015
Figure 20. Pots with small bases can be stored upside down, which is a more secure position, to prevent tipping and breakage. The foam pads protect the painted, burnished surfaces.

© Government of Canada, Canadian Conservation Institute. CCI 129979-0016
Figure 21. Archaeological glass objects, some with degraded surfaces. Each object has a custom-made spot on the storage tray that not only pads the object and prevents it from rolling around, but it also makes it easy for custodians to spot missing or displaced pieces.
The environment
Ceramics and glass in good condition are usually robust and do well in general museum environmental conditions. Unstable ceramics and glass, such as salt-contaminated ceramics or glass showing glass disease, require special environments.
Recommendations
- Keep objects out of direct sunlight and expose UV-sensitive items to low levels of UV only (e.g. filtered museum lighting or dark storage).
- Keep objects away from any sources of heat or cold, as these conditions will cause considerable, and potentially harmful, changes in RH; avoid fluctuating temperatures for the same reason.
- Glass should be kept between 40–50% RH (Koob 2006, p. 133). Store unstable glass, particularly if it is prone to weeping or crizzling, in an environment of 40% RH with minimal fluctuations. Higher ranges of RH cause increasing risk of weeping. RH below 30% should be avoided as it will result in permanent cracking (Koob 2006, p. 130).
- Store ceramics suspected of containing salts in a stable environment with few fluctuations. A set point between 40–50% RH should be selected with only minor fluctuations, especially toward the higher RH ranges.
- For unstable objects, control of RH in a space with less stringently controlled conditions can be done by creating a microclimate using a separate, inner enclosure with stringent RH controls (e.g. using silica gel conditioned to maintain unstable glass at 30 to 40% RH); examples of a microclimate are a controlled display case within a larger room, a wrapped object in a box or a sealed bag kept in a closed storage cabinet.
Examples of preventive conservation practices
This section provides practical examples in the care of ceramic and glass objects.
Low-fired Thule ceramics
Thule ceramics from the Arctic are extremely low fired due to the lack of firewood in the region. These pots are coarse and crumbly. High amounts of organic temper were also added to make the clay more workable, less prone to cracking during drying and able to withstand the heating and cooling cycles of a cooking pot. Due to these factors, low-fired ceramics were extremely permeable to water and were difficult to heat since water seeped into the ceramic and cooled off the surface. The Thule solved this problem by coating the interior of their ceramics with seal blood and seal oil. Thule ceramics should never be washed as the clay may dissolve and any remnants of seal blood and oil may be removed. Consolidation should only be done by a conservator and only after consulting with an archaeologist, as this treatment may interfere with future scientific analysis. Due to their fragility, Thule ceramics require well-designed storage mounts. The mount should have a solid base for lifting and moving, and it should support the object on all sides. The support should reduce the amount of handling needed (Figures 22a to 22g).

© Government of Canada, Canadian Conservation Institute. CCI 98507-0074
Figure 22a. This unusual partial Thule lamp has a natural stone base with low-fired clay sides. More clay was added at a later date to make the vessel higher.

© Government of Canada, Canadian Conservation Institute. CCI 98507-0086
Figure 22b. A solid base with padded polyethylene non-woven (Tyvek) cushion to support the lamp.

© Government of Canada, Canadian Conservation Institute. CCI 98507-0087
Figure 22c. The heavy, uneven base of the lamp is supported on a polyethylene non-woven (Tyvek) cushion.

© Government of Canada, Canadian Conservation Institute. CCI 98507-0088
Figure 22d. Velcro dots allow multiple Ethafoam pieces, cut to conform to the lamp's shape, to be attached to stop the lamp from moving.
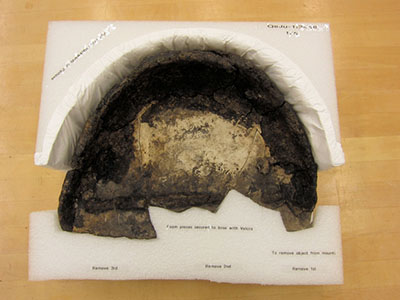
© Government of Canada, Canadian Conservation Institute. CCI 98507-0089
Figure 22e. A higher back piece of Ethafoam is covered with polyethylene non-woven sheet (Tyvek) to prevent abrasion of the fragile clay.

© Government of Canada, Canadian Conservation Institute. CCI 98507-0090
Figure 22f. A strong wood box is padded to prevent the object from shifting during shipping. Foam inserts are placed in the hollow of the lamp to keep it from lifting up during shipping.

© Government of Canada, Canadian Conservation Institute. CCI 98507-0091
Figure 22g. Two outer rings of absorbent foam protect the boxed object from shocks and vibration during transportation.
Deteriorating glass beads
A Mi'kmaq chief's coat dating back to 1841 is made from silk-lined wool decorated with multicoloured glass seed beads. The light blue beads display the typical symptoms of weeping and crizzling seen in poorly made glass beads (Figure 23a and 23b). These unstable glass beads are the result of inaccurate mixing in the preparation of the glass. Too little lime flux (calcium oxide) relative to the amount of alkali (sodium or potassium) resulted in unstable glass that is sensitive to moisture in the air. The excess alkali begins to absorb moisture when the RH is 42% or above and, at higher RH, leaches out of the glass and forms highly alkaline droplets (pH 10+) which dissolve the silica in the glass (Koob 2006, p. 118). The alkaline solution also attacks the associated organic materials, dissolving the thread and discolouring the backing fabric (Figure 23c). The hygroscopic nature of the wool fabric and cotton thread contributed to the deterioration, the worst of which being around the thread hole (Figure 23d).

© Government of Canada, Canadian Conservation Institute. CCI 2003656-0007
Figure 23a. Due to a mixing error in the ratio of flux when the glass was first made, the pale blue glass beads are disintegrating. The dark blue beads are stable, thus making it evident that the deterioration is specific to the production of the individual beads and not to the environment. Many of the beads have broken and been lost. The stringing and the fabric have deteriorated.

© Government of Canada, Canadian Conservation Institute. CCI 2003656-0006
Figure 23b. Detail of crizzled glass beads. A colour change in some beads is noticeable due to cracking, which makes them less transparent.

© Government of Canada, Canadian Conservation Institute. CCI 2003656-0008
Figure 23c. Detail of the deterioration of the silk lining upon which the beads were stitched. The alkali used in the glass-making process has leached out of the beads, causing bleaching and weakening of the support fabric.
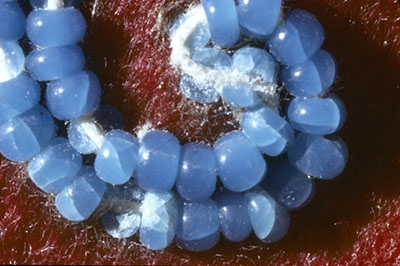
© Government of Canada, Canadian Conservation Institute. CCI 2003656-0004
Figure 23d. Detail of unstable glass beads. Due to the hygroscopic nature of the cotton thread, the interior of the beads deteriorated quicker, causing them to crack from side to side. The thread and surrounding fabric were degraded by the highly alkaline by-product.
Glass that has been poorly made will have inherent problems that are virtually impossible to correct. The main recourse is control of RH, preferably by enclosing the affected item in a container in which a microclimate can be established. RH should be tightly controlled. An RH of 42% or above will allow the deliquescence of sodium or potassium to occur (Davison 2003, p. 315). Below 30%, desiccation of the glass occurs, revealing a fine network of permanent cracks (Koob 2006, p. 127). For severely deteriorated beads, tighter controls may be necessary. The beads on the Mi’kmaq coat continued to deteriorate, albeit much more slowly, even at 37% RH. Cleaning to remove the alkaline material is recommended for early stages of crizzling; however, without tight control of the RH, the glass will continue to deteriorate. The use of water and alkaline materials for cleaning, such as soap or detergents, may cause further deterioration if not completely removed.
Bibliography and further reading
Bibliography
Brill, R. "Crizzling: A Problem in Glass Conservation." In Conservation in Archaeology and the Applied Arts. London, UK: International Institute for Conservation, 1975, pp. 121–34.
Davison, S. Conservation and Restoration of Glass, 2nd ed. Oxford, UK: Butterworth-Heinemann, 2003.
Down, J., ed. Adhesive Compendium for Conservation. Ottawa, ON: Canadian Conservation Institute, 2015.
Hodges, H. Artifacts: An Introduction to Early Materials and Technology. London, UK: J. Baker, 1971.
Institute of Conservation (ICON). "Care and Conservation of Ceramic and Glass." Conservation Register. London, UK: ICON, 2011.
National Park Service. Preservation of Low-Fired Ceramic Objects (PDF format). Conserv O Gram 8/3. Washington, D.C.: National Park Service, 2002.
National Park Service. Soluble Salts and Deterioration of Archaeological Materials (PDF format). Conserv O Gram 6/5. Washington, D.C.: National Park Service, 1993.
National Park Service. Removing Dust from Ceramic and Glass Objects (PDF format). Conserv O Gram 8/1. Washington, D.C.: National Park Service, 1993.
Newton, C., and J. Logan. Care of Ceramics and Glass, revised. CCI Notes 5/1. Ottawa, ON: Canadian Conservation Institute, 2007.
Newton, R., and S. Davison. The Conservation of Glass. London, UK: Butterworths, 1989.
Pearson, C. "Conservation of Ceramics, Glass and Stone." In Conservation of Marine Archaeological Objects. London, UK: Butterworths, 1987, pp. 253–267.
Rhodes, D. Clay and Glazes for the Potter. Radnor, PA: Chilton Book Company, 1973.
Rörig-Dalgaard, I. “Determination of the Deliquesce Point in Double Salts and in In-situ Multicomponent Salts with DVS Equipment.” (PDF format) In Proceedings of the 3rd International Conference on Salt Weathering of Buildings and Stone Sculptures. KIK-IRPA, 2014, pp. 223–236.
Tennent, N.H., ed. The Conservation of Glass and Ceramics: Research, Practice and Training. London, UK: James & James, 1999.
Williams, N. Porcelain Repair and Restoration: A Handbook, 2nd ed. Revised by L. Hogan and M. Bruce-Mitford. Philadelphia, PA: University of Pennsylvania Press, 1993.
Further reading
Buys, S., and V. Oakley. Conservation and Restoration of Ceramics. London, UK: Butterworths, 1993.
Koob, S. Conservation and Care of Glass Objects. New York, NY: Archetype, 2006.
National Park Service. "Appendix P: Curatorial Care of Ceramic, Glass, and Stone Objects." (PDF format) NPS Museum Handbook, Part 1. Washington, D.C.: National Park Service, 2000.
© Government of Canada, Canadian Conservation Institute, 2018
Published by:
Canadian Conservation Institute
Department of Canadian Heritage
1030 Innes Road
Ottawa, ON K1B 4S7
Canada
Cat. No.: CH57-4/6-8-2018E-PDF
ISBN 978-0-660-27981-7