Assessment - Ketones group
Chemical Abstracts Service Registry Numbers:
- 78-93-3
- 107-87-9
- 108-10-1
- 110-12-3
- 123-42-2
- 513-86-0
- 123-54-6
- 431-03-8
- 600-14-6
- 141-79-7
Environment and Climate Change Canada
Health Canada
May 2026
Cat No.: En84-398/2025E-PDF
ISBN: 978-0-660-79377-1
Synopsis
Pursuant to section 68 of the Canadian Environmental Protection Act, 1999 (CEPA), the Minister of the Environment and the Minister of Health have conducted an assessment of 10 substances referred to collectively under the Chemicals Management Plan as the Ketones Group. The Chemical Abstracts Service Registry Numbers (CAS RNFootnote 1), their Domestic Substances List (DSL) names, and their common names and acronyms are listed in the table below.
| CAS RN | Subgroup | Domestic Substances List name | Common name (acronym) |
|---|---|---|---|
| 78-93-3 | 1 | 2-Butanone | Methyl ethyl ketone (MEK) |
| 107-87-9 | 1 | 2-Pentanone | Methyl propyl ketone (MPK) |
| 108-10-1 | 2 | 2-Pentanone, 4-methyl- | Methyl isobutyl ketone (MIBK) |
| 110-12-3 | 2 | 2-Hexanone, 5-methyl | Methyl isoamyl ketone (MIAK) |
| 123-42-2 | 2 | 2-Pentanone, 4-hydroxy-4-methyl- | Diacetone alcohol (DAA) |
| 431-03-8 | 3 | 2,3-Butanedione | Diacetyl |
| 513-86-0 | 3 | 2-Butanone, 3-hydroxy | Acetoin |
| 600-14-6 | 3 | 2,3-Pentanedione | 2,3-Pentanedione (2,3-PD) |
| 123-54-6 | Individual | 2,4-Pentanedione | 2,4-Pentanedione (2,4-PD) |
| 141-79-7 | Individual | 3-Penten-2-one, 4-methyl- | Mesityl oxide (MO) |
All 10 substances in the Ketones Group are commercially produced and are naturally present in the environment in various plants and/or food items or as substances produced by microbes and other organisms. Several of the ketones are also produced endogenously in humans, including MEK, diacetyl and acetoin. According to information reported in response to surveys under section 71 of CEPA, only DAA (23,000 kg) and 2,3-PD (1,200 kg) were reported to be manufactured in Canada in 2011. Reported imports in Canada for the substances in the Ketones Group ranged between 100 kg (for acetoin) and 6,000,000 kg (for MEK) in 2011. In the same year, no Canadian manufacturing or importing activities above the reporting threshold of 100 kg were reported for MO.
In general, ketones are primarily used as solvents in various products, including products available to consumers such as paints, coatings and adhesives, and in numerous industrial applications such as chemical intermediates and solvents, among others. They may also be used as food flavouring agents, in cosmetics, in natural health products, and as formulants in pest control products.
The ecological risks of the substances in the Ketones Group were characterized using the ecological risk classification of organic substances (ERC), which is a risk-based approach that employs multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. Hazard profiles are established based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Metrics considered in the exposure profiles include potential emission rate, overall persistence, and long-range transport potential. A risk matrix is used to assign a low, moderate or high level of potential concern for substances on the basis of their hazard and exposure profiles Based on the outcome of the ERC analysis, the substances in the Ketones Group are considered unlikely to be causing ecological harm.
Considering all available lines of evidence presented in this assessment, there is low risk of harm to the environment from MEK, MPK, MIBK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO. It is concluded that MEK, MPK, MIBK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
Several of these ketones have been previously reviewed internationally; these reviews and assessments were used to inform the health effects characterization in this assessment.
For the human health risk assessment, 8 of the substances in the Ketones Group have been addressed under 3 subgroups, with the remaining 2 substances having been addressed individually. For subgroup 1 (MEK and MPK), the critical health effects include developmental effects for MEK and decreased body weight gain for both MEK and MPK. The general population in Canada is exposed to MEK and MPK from air, food (from their natural occurrence and possible use as food flavouring agents), and products available to consumers, including cosmetics, paints, and do-it-yourself (DIY) products. MEK was also reported to be released to air as a result of industrial activities. A comparison of the levels of MEK and MPK found in environmental media and in food (from their possible use as food flavouring agents) to which people in Canada may be exposed against the levels associated with adverse effects in laboratory studies results in margins that are considered adequate to address uncertainties in the available exposure and health effects data used to characterize risk. However, the margins between critical health effect levels and exposures to MEK in some products available to consumers—namely, paint or coating removers or strippers (for example, lacquer removers), adhesive removers, degreasers, paint or coating thinners, liquid paints, and various spray products including spray paints—are considered inadequate to account for uncertainties in the exposure and health effects data used to characterize risk. Given the low acute toxicity of MPK and the absence of developmental effects via inhalation, there are no concerns related to the presence of MPK in products available to consumers.
For subgroup 2 (MIBK, MIAK, and DAA), the International Agency for Research on Cancer (IARC) considers MIBK to be in group 2B (“possibly carcinogenic to humans”), with “sufficient evidence” of carcinogenicity in laboratory animals. For non-cancer effects, effects on the liver (MIBK, MIAK) and kidney (MIBK, DAA) as well as developmental effects (MIBK, DAA) were observed in laboratory studies. In Canada, the general population may be exposed to MIBK, MIAK, and DAA from environmental media and food (from their natural occurrence, and for MIBK, from its potential use as a food flavouring agent), and from the use of products available to consumers, including rubbing alcohol, cosmetics, markers, paints, and DIY products. MIBK was also reported to be released to air as a result of industrial activities. A comparison of estimated levels of exposure to MIBK, MIAK, and DAA from environmental media and of exposure to MIBK from food (from its potential use as a food flavouring agent) against critical effect levels results in margins that are considered to be adequate to address uncertainties in the available exposure and health effects data used to characterize risk. Exposure to MIBK, MIAK, and DAA from their natural occurrence in food was not identified as a concern for human health. However, for MIBK, the resulting margins associated with the use of wood lacquers, liquid paints, and various spray products including spray paints are considered to be inadequate to address uncertainties in the available exposure and health effects data used to characterize risk.
For subgroup 3 (diacetyl, 2,3-PD, and acetoin), diacetyl was shown to be carcinogenic in laboratory studies. For diacetyl and 2,3-PD, non-cancer effects have also been observed, such as on the respiratory tract. For acetoin, only general systemic toxicity at high doses has been observed. The general population of Canada is primarily exposed to diacetyl, 2,3-PD, and acetoin through food (due to their natural occurrence and possible use as food flavouring agents) and to diacetyl and 2,3-PD through the use of a limited number of products available to consumers, including cosmetics and air fresheners, respectively. A comparison of estimated levels of exposure to diacetyl, 2,3-PD, and acetoin against critical effect levels results in margins that are considered to be adequate to address uncertainties in the available exposure and health effects data used to characterize risk.
The available information on the health effects of 2,4-PD indicates general systemic toxicity and developmental effects. 2,4-PD has shown some potential to be genotoxic but is not expected to be carcinogenic. The general population of Canada may be exposed to 2,4-PD from environmental media (air and water) and through its natural occurrence in food. A comparison of estimated levels of exposure to 2,4-PD and critical effect levels results in margins that are considered adequate to address uncertainties in the available exposure and health effects data used to characterize risk. In the draft assessment, 2,4-PD was identified as a concern for human health on the basis of its presence in a limited number of products available to consumers. Further investigation confirmed that 2,4-PD is not currently present in products available to consumers; therefore, 2,4-PD is no longer considered to be a concern for human health at current levels of exposure. While exposure of the general population to 2,4-PD is not of concern at current levels, there is the potential for exposures to increase in the future. Given the potential health effects of concern associated with this substance there may be a concern for human health if exposures were to increase.
People in Canada may be exposed to MO from its presence in air and food (due to its natural occurrence and potential use as a food flavouring agent). MO is not expected to be carcinogenic or genotoxic. General systemic toxicity has been associated with exposure to MO in laboratory studies. A comparison of estimated levels of exposure to MO in environmental media and food (from its potential use as a food flavouring agent) against critical effect levels results in margins that are considered to be adequate to address uncertainties in the available exposure and health effects data used to characterize risk.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects. In addition, exposures of people living near industrial releases were also considered.
Considering all of the information presented in this assessment, it is concluded that MEK and MIBK meet the criteria under paragraph 64(c) of CEPA as they are entering or may enter the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health. However, it is concluded that MPK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
It is therefore concluded that MEK and MIBK meet one or more of the criteria set out in section 64 of CEPA and that MPK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO do not meet any of the criteria set out in section 64 of CEPA.
1. Introduction
Pursuant to section 68 of the Canadian Environmental Protection Act, 1999 (CEPA) (Canada 1999), the Minister of the Environment and the Minister of Health have conducted an assessment of 10 substances referred to collectively as the Ketones Group to determine whether these substances present or may present a risk to the environment or to human health. The substances in this group were identified as priorities for assessment as they met categorization criteria or were prioritized through other mechanisms (ECCC, HC [modified 2017]).
The ecological risks of the substances in the Ketones Group were characterized using the ecological risk classification of organic substances (ERC) approach (ECCC 2016a). The ERC describes the hazard of a substance using key metrics including mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity, and considers the possible exposure of organisms in the aquatic and terrestrial environments on the basis of such factors as potential emission rates, overall persistence, and long-range transport potential in air. The various lines of evidence are combined to identify substances that warrant further evaluation of their potential to cause harm to the environment or that have a low likelihood of causing harm to the environment.
Some substances are assessed in subgroups because of similarities in chemical structure, properties, and/or toxicity. Given that these substances may be used in similar ways and applications, the potential for risk to human health is assessed using similar exposure assumptions across the group.
Some substances in the Ketones Group currently being evaluated have been reviewed internationally through the Organisation for Economic Cooperation and Development (OECD) Cooperative Chemicals Assessment Programme, and both the Screening Information Dataset (SIDS) and SIDS Initial Assessment Reports (SIARs) are available for these substances. These assessments undergo rigorous review (including peer review) and endorsement by international governmental authorities. Health Canada and Environment and Climate Change Canada are active participants in this process and consider these assessments to be reliable. Some of the substances have also been reviewed by the International Programme on Chemical Safety (IPCS), United States Environmental Protection Agency (US EPA), International Agency for Research on Cancer (IARC), Agency for Toxic Substances and Disease Registry (ATSDR), and the United States (US) National Toxicology Program (NTP). Reviews conducted by these institutions were used to inform the health effects characterization in this assessment.
Diacetyl, 2,3-PD, and acetoin have been identified in vaping products (Farsalinos et al. 2015; Klager et al. 2017) which may represent an additional source of exposure to diacetyl, 2,3-PD, and acetoin. Vaping products (such as electronic cigarettes and vaping devices containing cannabis) are being addressed through separate legislative frameworks. Health Canada has taken steps to inform industry that substances with known inhalation risks (for example, diacetyl and 2,3-pentanedione) should never be added to vaping products (Health Canada [modified 2020a,b]). In addition, Health Canada is conducting ongoing testing (under the open characterization project) that indicates that diketones are only infrequently detected in vaping products sold in Canada (personal communication, email from the Consumer and Hazardous Product Safety Directorate, [CHPSD], Health Canada, to the Existing Substances Risk Assessment Bureau, [ESRAB], Health Canada, dated December 3, 2020; unreferenced).
This assessment includes the consideration of information on chemical properties, environmental fate, hazards, uses and exposures, including additional information submitted by stakeholders. Relevant data were identified up to April 2017. Additional data were submitted up to October 2019. Targeted literature searches were conducted up to June 2021. Empirical data from key studies as well as some results from models were used to reach conclusions. Where available and relevant, information presented in assessments from other jurisdictions was considered.
This assessment was prepared by staff in the CEPA Risk Assessment Program at Health Canada and Environment and Climate Change Canada and incorporates input from other programs within these departments. The human health portions of this assessment have undergone external review and/or consultation. Comments on the technical portions relevant to human health were received from Theresa Lopez, Jennifer Flippin and Joan Garey (TetraTech Inc.), and from D.L. Morgan (National Toxicology Program, National Institute of Environmental Health Sciences, US). The ecological portion of this assessment is based on the ERC document (published July 30, 2016), which was peer-reviewed and subject to a 60-day public comment period. Additionally, the draft of this assessment (published January 19, 2019) was subject to a 60-day public comment period. While external comments were taken into consideration, the final content and outcome of the assessment remain the responsibility of Health Canada and Environment and Climate Change Canada.
Assessments focus on information critical to determining whether substances meet the criteria as set out in section 64 of CEPA by considering scientific information, including information, if available, on subpopulations who may have greater susceptibility or greater exposure, vulnerable environments and cumulative effects,Footnote 2 and by incorporating a weight of evidence approach and precaution.Footnote 3 This assessment presents the critical information and considerations on which the conclusions are based.
2. Identity of substances
The 10 substances evaluated in this assessment are ketones with the general formula shown in Figure 2-1. The ketones in this assessment have been divided into 3 subgroups and 2 individual assessments according to their chemical structure, properties, and/or toxicity.
Figure 2-1. General formula for ketones
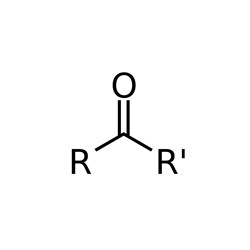
The Chemical Abstracts Service Registry Numbers (CAS RNsFootnote 1), Domestic Substances List (DSL) names, common names and/or acronyms for the individual substances in the Ketones Group are presented in Table 2‑1. A list of additional chemical names (for example, trade names) is available from the National Chemical Inventories (NCI 2015).
| Subgroup | CAS RN | DSL name (common name or acronym) | Chemical structure and molecular formula | Molecular weight (g/mol) |
|---|---|---|---|---|
| 1 | 78-93-3 | 2-Butanone (methyl ethyl ketone; MEK) | 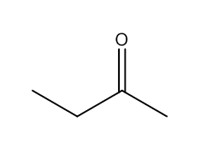 C4H8O |
72.11 |
| 1 | 107-87-9 | 2-Pentanone (methyl propyl ketone; MPK) | 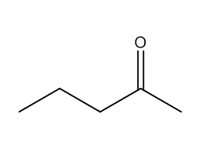 C5H10O |
86.13 |
| 2 | 108-10-1 | 2-Pentanone, 4-methyl- (methyl isobutyl ketone; MIBK) | 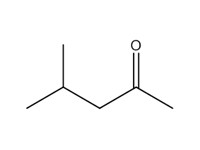 C6H12O |
100.16 |
| 2 | 110-12-3 | 2-Hexanone, 5-methyl- (methyl isoamyl ketone; MIAK) | 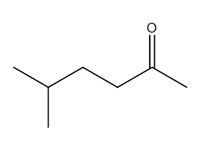 C7H14O |
114.19 |
| 2 | 123-42-2 | 2-Pentanone, 4-hydroxy-4-methyl- (diacetone alcohol; DAA) | 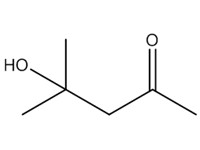 C6H12O2 |
116.16 |
| 3 | 431-03-8 | 2,3-Butanedione (diacetyl) | 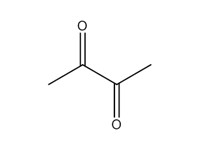 C4H6O2 |
86.09 |
| 3 | 600-14-6 | 2,3-Pentanedione (2,3-PD) | 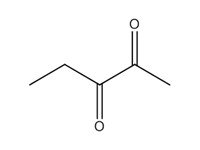 C5H8O2 |
100.12 |
| 3 | 513-86-0 | 2-Butanone, 3-hydroxy- (acetoin) | 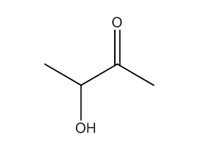 C4H8O2 |
88.11 |
| Individual | 123-54-6 | 2,4-Pentanedione (2,4-PD) | 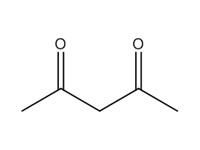 C5H8O2 |
100.12 |
| Individual | 141-79-7 | 3-Penten-2-one, 4-methyl- (mesityl oxide; MO) | 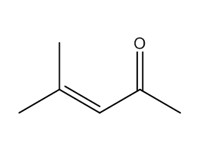 C6H10O |
98.14 |
2.1 Selection of analogues and use of (Q)SAR models
A read-across approach using data from analogues and the results of (quantitative) structure-activity relationship ([Q]SAR) models have been used, where appropriate, to inform the human health assessments. Analogues were selected that were structurally similar to and/or a metabolite for substances within this group (for example, similar physical-chemical properties, toxicokinetics) and that had relevant empirical data that could be used to read across to substances with limited empirical health effects data. The applicability of (Q)SAR models was determined on a case-by-case basis. Details of the read-across data selected to inform the human health assessments of each subgroup and MO are further discussed in the relevant sections of this report. Information on the identities and chemical structures of the analogues used to inform this assessment is presented in Table 2‑2. In some cases, substances within a subgroup were used as analogues to inform the human health assessments. Information on the physical-chemical properties and health effects data for the analogues as well as for the target substances in this group assessment are presented in Appendix A.
| Subgroup or substance being assessed | CAS RN | DSL name (common name or acronym) | Chemical structure and molecular formula | Molecular weight (g/mol) |
|---|---|---|---|---|
| Subgroup 1 | 78-92-2 | 2-Butanol | 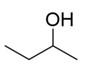 C4H10O |
74.1 |
| MO | 110-93-0 | 5-Hepten-2-one, 6-methyl- (6-methyl-5-heptene-2-one; MHE) | 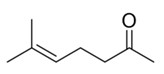 C8H14O |
126.2 |
3. Physical and chemical properties
A summary of physical and chemical property data for the substances in the Ketones Group is presented in Table 3‑1 to Table 3‑4. Additional physical and chemical properties are presented in ECCC (2016b).
| Property | MEK | MPK | Reference |
|---|---|---|---|
| Physical state | Colourless liquid | Colourless liquid | HSDB 1983- |
| Melting point (°C) | -85.9 | -76.9 | OECD 1997a, ChemIDplus 1993- |
| Vapour pressure (Pa) | 10,332 | 4,720 | OECD 1997a, ChemIDplus 1993- |
| Henry’s law constant (Pa·m3/mol) | 5.85 | 8.47 | ATSDR 2020, ChemIDplus 1993- |
| Water solubility (mg/L) | 276,000 | 43,000 | OECD 1997a, ChemIDplus 1993- |
| Log Kow (dimensionless) | 0.29 | 0.91 | OECD 1997a, ChemIDplus 1993- |
| Log Koc (dimensionless) | 0.55 | 1.87 [estimated] | ATSDR 2020, HSDB 1983- |
Abbreviations: Kow, octanol–water partition coefficient; Koc, organic carbon–water partition coefficient
| Property | MIBK | MIAK | DAA | Reference |
|---|---|---|---|---|
| Physical state | Colourless liquid | Colourless, clear liquid | Colourless liquid | OECD 1996, HSDB 1983- |
| Melting point (°C) | -84.7 | -74 | -44 | OECD 1996, ChemIDplus 1993- |
| Vapour pressure (Pa) | 2,653 | 769 | 228 | ChemIDplus 1993- |
| Henry’s law constant (Pa·m3/mol) | 14.0 [estimated] | 16.2 [estimated] | 0.026 [estimated] | ChemIDplus 1993- |
| Water solubility (mg/L) | 19,000 | 5,400 | 1,000,000 | ChemIDplus 1993- |
| Log Kow (dimensionless) | 1.31 | 1.88 | -0.34 [estimated] | ChemIDplus 1993- |
| Log Koc (dimensionless) | 2.08 [estimated] | 2.40 [estimated] | 1.32 [estimated] | HSDB 1983- |
Abbreviations: Kow, octanol–water partition coefficient; Koc, organic carbon–water partition coefficient
| Property | Diacetyl | 2,3-PD | Acetoin | Reference |
|---|---|---|---|---|
| Physical state | Greenish-yellow liquid | Dark yellow to green-yellow liquid | Slightly yellow liquid or crystals | HSDB 1983-, CDC 2016 |
| Melting point (°C) | -2.40 | -29.38 [estimated] | 15.0 | ChemIDplus 1993-, EPI Suite c2000-2012 |
| Vapour pressure (Pa) | 7,572 | 4 146 [estimated] | 359 [estimated] | ChemIDplus 1993-, EPI Suite c2000-2012 |
| Henry’s law constant (Pa·m3/mol) | 1.35 | 0.674 [estimated] | 1.04 [estimated] | ChemIDplus 1993-, EPI Suite c2000-2012 |
| Water solubility (mg/L) | 200,000 | 66,700 (at 15 °C) | 1,000,000 | ChemIDplus 1993- |
| Log Kow (dimensionless) | -1.34 | -0.85 [estimated] | -0.36 [estimated] | ChemIDplus 1993- |
| Log Koc (dimensionless) | -0.28 [estimated] | -0.004 [estimated] | 0.3 [estimated] | EPI Suite c2000-2012, HSDB 1983- |
Abbreviations: Kow, octanol–water partition coefficient; Koc, organic carbon–water partition coefficient
| Property | 2,4-PD | MO | Reference |
|---|---|---|---|
| Physical state | Colourless or slightly yellow liquid | Oily, colourless to light-yellow liquid | HSDB 1983- |
| Melting point (°C) | -23.0 | -59.0 | ChemIDplus 1993- |
| Vapour pressure (Pa) | 395 | 1,095 | ChemIDplus 1993- |
| Henry’s law constant (Pa·m3/mol) | 0.238 [estimated] | 3.72 [estimated] | ChemIDplus 1993- |
| Water solubility (mg/L) | 166,000 | 28,900 | ChemIDplus 1993- |
| Log Kow (dimensionless) | 0.34–0.4 | 1.2–1.7 | ChemIDplus 1993-, OECD 1997b, OECD 2001 |
| Log Koc (dimensionless) | 1.54 [estimated] | 1.04 | EPI Suite c2000-2012, OECD 1997b |
Abbreviations: Kow, octanol–water partition coefficient; Koc, organic carbon–water partition coefficient
4. Sources and uses
All 10 substances in the Ketones Group are naturally present in the environment in various plants and/or food items or as substances produced by microbes and other organisms, but may also be synthetically produced (VCCEP 2003; Burdock 2010; O’Donoghue 2012a,b). MEK, diacetyl, and acetoin are also produced endogenously in humans (WHO 1999a; VCCEP 2003; NTP 2007a).
All of the substances in the Ketones Group, except for MEK, have been included in a 2011 survey issued pursuant to section 71 of CEPA (Environment Canada 2012). MEK was included in a 2001 survey issued pursuant to section 71 of CEPA. Reported manufactured quantities for MEK ranged between 1 million and 10 million kg in the year 2000; however, manufacturing of this substance in Canada ceased in 2002 (Environment Canada 2001). Reported quantities of MEK imported into Canada in the year 2000 were greater than 10 million kg. According to the Canadian International Merchandise Trade Database (CIMT), annual average imports of MEK into Canada between 2011 and 2016 were approximately 4.9 million kg (CIMT 2017). Table 4‑1 presents a summary of the reported total manufacture and total import quantities for the Ketones Group.
| Common name | Total manufacturea (kg) | Total importsa (kg) |
|---|---|---|
| MEK | NR | 6,042,865(data for 2011; CIMT 2017) |
| MPK | NR | 1,097,844 |
| MIBK | NR | 1,241,783 |
| MIAK | NR | 35,906 |
| DAA | 23,000 | 265,529 |
| Diacetyl | NR | 1,430 |
| 2,3-PD | 1,200 | NR |
| Acetoin | NR | 100 to 1,000 |
| 2,4-PD | NR | 100,000 to 1,000000 |
| MO | NR | NR |
Abbreviations: NR, not reported above the reporting threshold of 100 kg
a Values reflect quantities reported in response to surveys conducted under section 71 of CEPA (Environment Canada 2012), except for MEK. See survey (Schedules 2 and 3) for specific inclusions and exclusions (Environment Canada 2012).
Table 4‑2 presents a summary of the major uses of the Ketones Group, according to information submitted in response to CEPA section 71 surveys (Environment Canada 2001, 2013) and Table 4‑3 presents additional uses identified in Canada.
| Major uses | Subgroup 1a | Subgroup 2 | Subgroup 3 | Individual |
|---|---|---|---|---|
| Paints and coatings | MEK, MPK | MIBK, MIAK, DAA | N/A | 2,4-PD |
| Food and beverage | N/A | N/A | 2,3-PD, Acetoin | N/A |
| Agricultural products, mixtures or manufactured items (non-pesticidal) | MEK | N/A | Diacetyl | N/A |
| Adhesives and sealants | MEK, MPK | MIBK, DAA | N/A | 2,4-PD |
| Ink, toner and colourants | MEK, MPK | MIBK, DAA | N/A | N/A |
| Automotive, aircraft and transportation | MEK, MPK | MIBK, DAA | N/A | N/A |
| Plastic and rubber materials not otherwise covered | N/A | MIBK | N/A | 2,4-PD |
| Electrical and electronics | N/A | MIBK, DAA | N/A | N/A |
| Floor coverings | MEK | MIBK | N/A | N/A |
| Cleaning and furnishing care | MEK | DAA | N/A | N/A |
| Personal care | N/A | DAA | 2,3-PD, Acetoin | N/A |
| Toys, playground and sporting equipment | N/A | DAA | N/A | N/A |
| Otherb | MEK, MPK | MIBK, DAA | Diacetyl | N/A |
Abbreviations: N/A, not applicable
a Non-confidential uses reported in response to a survey conducted under section 71 of CEPA (Environment Canada 2012). See survey (Schedules 2 and 3) for specific inclusions and exclusions. Results for MEK are from uses in 2000 and may no longer be relevant.
b “Other” refers to minor uses and/or uses that cannot be disclosed as a result of confidentiality claims.
| Use | Subgroup 1 | Subgroup 2 | Subgroup 3 | Individual |
|---|---|---|---|---|
| Food additivea | MEK | N | N | N |
| Food flavouring agenta | MEK, MPK | MIBK | Diacetyl, Acetoin, 2,3-PD | MO |
| Food packaging materialsa | MEK, MPK | MIBK, DAA | N | 2,4-PD |
| Incidental additivesa | MEK | N | N | N |
| Natural Health Products Ingredients Databaseb | MEK, MPK | MIBK, DAA | Diacetyl, Acetoin, 2,3-PD | MO |
| Licensed Natural Health Products Database as non-medicinal ingredients in natural health products in Canadac | MEK | MIBK | N | N |
| Notified to be present in cosmetics, based on notifications submitted under the Cosmetic Regulations to Health Canadad | MEK, MPK | MIBK, DAA | Diacteyl | N |
| Formulant in pest control products registered in Canadae | MPK | MIBK, MIAK, DAA | Diacetyl, Acetoin, 2,3-PD | N |
Abbreviations: N, no—this use was not reported as a possible use for this substance
a Personal communication, email from the Food Directorate (FD), Health Canada (HC), to the ESRAB, HC, dated August 18, 2016; unreferenced
b NHPID [modified 2022]
c LNHPD [modified 2021]
d Personal communication, email from the CHPSD, HC, to the ESRAB, HC, dated August, 2016, and November 2019; unreferenced
e Personal communication, email from the Pest Management Regulatory Agency, HC, to the ESRAB, HC, dated July 28, 2016; unreferenced
In general, ketones are primarily used as solvents in various products including products available to consumers, and in numerous industrial applications as chemical intermediates and solvents, among others (Braithwaite 2000; O’Donoghue 2012a,b). MEK is listed as a permitted food additive in natural extractives and in spice extracts, as prescribed in Health Canada’s List of Permitted Carrier or Extraction Solvents, incorporated by reference in its respective Marketing Authorization issued under the Food and Drugs Act. MEK, MPK, MIBK, DAA, and 2,4-PD may be used as components in the manufacture of food packaging materials that do not come into direct contact with food in Canada. MEK may also be used as a component in incidental additivesFootnote 4 (cleaners) in food processing establishments with no direct contact with food. No definitive information is available concerning the potential use of MEK, MPK, MIBK, diacetyl, acetoin, 2,3-PD, and MO as food flavouring agents in Canada; however, since these substances are known to be used as food flavouring agents in the United States or Europe, it is possible that these substances are present as flavouring agents in foods sold in Canada (personal communication, email from the FD, HC, to the ESRAB, HC, dated August 18, 2016; unreferenced).
MEK is listed in the International Council for Harmonization (ICH)’s Guideline for Residual Solvents (Q3C(R8)) as a Class 3 solvent, which is a solvent with low toxic potential that should be limited using good manufacturing practices or other quality-based requirements. It is considered acceptable without justification at amounts of no more than 50 mg/day (corresponding to 5,000 ppm or 0.5% when assuming a product mass of 10 g administered daily). MIBK is listed in ICH’s guideline as a Class 2 solvent, which is a solvent that should be limited and is further associated with a permitted daily exposure (PDE) of up to 45 mg/day (corresponding to 4,500 ppm or 0.45% when assuming a product mass of 10 g administered daily) (personal communication, email from the Biologics and Genetic Therapies Directorate, HC, to the ESRAB, HC, dated July 15, 2012; unreferenced).
MEK is listed in the Natural Health Products Ingredients Database (NHPID) with a medicinal role and classified as a natural health product (NHP) substance falling under Schedule 1, item 2 (an isolate) of the Natural Health Products Regulations, as well as with a non-medicinal role for topical use as a denaturant (up to 1%) or oral use as a flavour enhancer (up to 28.76 ppm). The NHPID also notes that MEK is subject to additional regulatory requirements in accordance with the Precursor Control Regulations since it is listed on Schedule VI of the Controlled Drugs and Substances Act. MPK, 2,3-PD, and MO are listed in the NHPID with a non-medicinal role for oral use as flavour enhancers; MIBK is listed with a non-medicinal role for oral use as a flavour enhancer (up to 25 ppm) or topical use as a denaturant (up to 4%); DAA is listed with a non-medicinal role for topical use only as a fragrance ingredient or solvent; and acetoin is listed with a non-medicinal role for oral use as a flavour enhancer or topical use as a fragrance ingredient. MIBK is also associated in the NHPID with a permitted daily exposure as residual solvent of up to 45 mg/day, equivalent to a concentration limit of 4,500 ppm when assuming a product mass of 10 g administered daily, further indicating that this limit applies to all dosage forms and routes of administration. Only MEK and MIBK are listed in the Licensed Natural Health Products Database as being present as non-medicinal ingredients in a limited number of currently licensed topical NHPs in Canada (NHPID [modified 2022]; LNHPD [modified 2021]; personal communication, email from the Natural and Non-prescription Health Products Directorate (NNHPD), HC, to the ESRAB, HC, dated August 16, 2016; unreferenced).
On the basis of notifications submitted under the Cosmetic Regulations to Health Canada, MEK, MPK, MIBK, DAA, and diacetyl are used in certain cosmetics in Canada, primarily in nail care products (personal communication, email from the CHPSD, HC, to the ESRAB, HC, dated August 2016, November 2019, and April 2020; unreferenced). According to publicly available sources, diacetyl was identified in additional cosmetics in Canada on the basis of information from retailer websites. MEK, MPK, MIBK, MIAK, and DAA are used in products available to consumers, including liquid and spray paints and coatings, automotive care products, do-it-yourself (DIY) products such as paint removers, adhesives, and pipe sealants (Environment Canada 2012; CPID [modified 2019]; SDS 2008, 2011, 2012a,b,c, 2013, 2014a,b, 2015a,b,c,d, 2016a,b,c,d, 2017a,b, 2018a,b,c, 2019a,b,c,d,e,f,g). MEK, MPK, and MIBK have been identified in emissions from various building materials (for example, wood, carpet, insulation) and products available to consumers (for example, paint, automotive cleaners, caulking) in Canada by the National Research Council of Canada (Won and Lusztyk 2011; Won and Yang 2012; Won et al. 2013; Won et al. 2014; Won et al. 2015).
MEK was identified in several different children’s products, including tents/tunnels (Hansen et al. 2004), slimy toys (Svendsen et al. 2005), rubber figures, and speed markers (Glensvig and Ports 2006) in Denmark. Under the Washington State Department of Ecology’s Children’s Product Safety Act (WSDE 2020), MEK was detected in various products intended for children 12 years and younger, including kids’ crafts, baby furniture, baby bibs, pacifiers/teething rings, children’s toys and games, and baby and children’s bedding and clothing, as well as footwear and camping gear (WSDE 2020). MEK was present in these products primarily as a contaminant but also as an adhesive, binding agent, colouration/pigment/dye/ink, component of plastic resin or polymer process, hardening agent, manufacturing additive, preservative, protective coating, reinforcement/strength agent, and solvent (WSDE 2020). MEK has also been measured in animal care products (Nylén et al. 2004) and adult toys (Nilsson et al. 2006) in Denmark.
Other sources of these ketones include vehicle exhaust (MEK, diacetyl) (IPCS 1993), tire crumb rubber and synthetic turf made with tire crumb rubber (MIBK) (US EPA & CDC/ATSDR 2019), cigarette smoke (MEK, diacetyl, 2,3-PD), and flavoured e-cigarette liquids (diacetyl, 2,3-PD, acetoin) (personal communication, emails from the Tobacco Control Directorate, HC, to the ESRAB, HC, dated August 15 to18, 2016 and November 16, 2017; unreferenced). Information received as part of the public comment period on the draft screen assessment presented additional sources of these ketones, including laboratory uses (MEK, DAA) and use during the assembly of optical fibers (MIBK), as well as in ink used to mark internal components of electronic equipment (for example, power supplies, inductors, drives) (MEK).
Two of the 10 substances, MEK and MIBK, are reportable under the National Pollutant Release Inventory (NPRI). Table 4‑4 summarizes the release and disposal data from 2011 to 2019 (NPRI 2011-2017a,b, 2021).
| Substance | On-site releases to aira,b | On-site releases to watera,b | On-site releases to landa,b | Disposal on-sitea,b | Disposal off-sitea,b | Off-site recyclinga,b |
|---|---|---|---|---|---|---|
| MEK | 720 to 1,380 | 5.4 to 20 | 0 to 2 | 0.802 to 67 | 90 to 1,563 | 1,441 to 3,074 |
| MIBK | 164 to 242 | 0.02 to 1.9 | 0 to 0.049 | 1.3 to 32 | 23 to 318 | 243 to 323 |
a Data used for this table is current as of June 22, 2021. Facilities can and do update their information reported to the NPRI at any time. Consequently, similar analysis done with different versions of the data may produce different results (NPRI 2021).
b Ranges from facilities that meet NPRI reporting threshold requirements. Values are rounded to the nearest tonne.
5. Environmental fate and behaviour
5.1 Environmental persistence
According to models used in the ERC approach (ECCC 2016b), MEK, DAA, diacetyl, 2,3-PD 2-4-PD, and MO are expected to persist in air, but are not expected to persist in water, sediment, or soil. However, for 2-4-PD, Information on the tautomerization of 2,4-PD indicates that the enol form of 2,4-PD is expected to be dominant over the keto form in the air (Spencer et al. 1982; Folkendt et al. 1985). In addition, calculations of the half-lives using multiple published empirical rate constants (Holloway et al. 2005; Zhou et al. 2008; Messaadia et al. 2015) and the AOPWIN model results (Atmospheric Oxidation Program for Microsoft Windows, 2010), suggest that 2,4-PD is unlikely to be persistent in air.
MPK, MIBK, MIAK, and acetoin are not expected to persist in air, water, sediment, or soil according to models used in ERC (ECCC 2016b).
5.2 Potential for bioaccumulation
Given their low log Kow and low bioconcentration factors (ECCC 2016b), MEK, MIAK, DAA, diacetyl, acetoin, 2,3-PD, 2,4-PD, and MO are not expected to significantly bioaccumulate in organisms. Although the log Kow value(s) for MPK and MIBK are high, the bioconcentration factors for these substances are low (1.4 and 2 L/kg, respectively). As a result, these substances are not expected to significantly bioaccumulate in organisms (ECCC 2016b).
6. Potential to cause ecological harm
6.1 Characterization of ecological risk
The ecological risks of the substances in the Ketones Group were characterized using the ERC approach (ECCC 2016a). The ERC is a risk-based approach that considers multiple metrics for both hazard and exposure, with weighted consideration of multiple lines of evidence for determining risk classification. The various lines of evidence are combined to discriminate between substances of lower or higher potency and lower or higher potential for exposure in various media. This approach reduces the overall uncertainty with risk characterization compared to an approach that relies on a single metric in a single medium (for example, median lethal concentration) for characterization. The following summarizes the approach, which is described in detail in ECCC (2016a).
Data on physical-chemical properties, fate (chemical half-lives in various media and biota, partition coefficients, and fish bioconcentration), acute fish ecotoxicity, and chemical import or manufacture volumes in Canada were collected from scientific literature, from available empirical databases (for example, OECD QSAR Toolbox 2014), and responses to surveys issued pursuant to section 71 of CEPA, or they were generated using selected (Q)SAR or mass-balance fate and bioaccumulation models. These data were used as inputs to other mass-balance models or to complete the substance hazard and exposure profiles.
Hazard profiles were based principally on metrics regarding mode of toxic action, chemical reactivity, food web-derived internal toxicity thresholds, bioavailability, and chemical and biological activity. Exposure profiles were also based on multiple metrics including potential emission rate, overall persistence, and long-range transport potential. Hazard and exposure profiles were compared to decision criteria in order to classify the hazard and exposure potentials for each organic substance as low, moderate, or high. Additional rules were applied (for example, classification consistency, margin of exposure) to refine the preliminary classifications of hazard or exposure.
A risk matrix was used to assign a low, moderate, or high classification of potential risk for each substance on the basis of its hazard and exposure classifications. ERC classifications of potential risk were verified using a two-step approach. The first step adjusted the risk classification outcomes from moderate or high to low for substances that had a low estimated rate of emission to water after wastewater treatment, representing a low potential for exposure. The second step reviewed low risk potential classification outcomes using relatively conservative, local-scale (that is, in the area immediately surrounding a point-source of discharge) risk scenarios, designed to be protective of the environment, to determine whether the classification of potential risk should be increased.
ERC uses a weighted approach to minimize the potential for both over- and under classification of hazard and exposure, and of subsequent risk. The balanced approaches for dealing with uncertainties are described in greater detail in ECCC (2016a). The following describes 2 of the more substantial areas of uncertainty. Error with empirical or modelled acute toxicity values could result in changes in classification of hazard, particularly metrics relying on tissue residue values (that is, mode of toxic action), many of which are predicted values from (Q)SAR models (OECD QSAR Toolbox 2014). However, the impact of this error is mitigated by the fact that overestimation of median lethality will result in a conservative (protective) tissue residue used for critical body residue analysis. Error with underestimation of acute toxicity will be mitigated through the use of other hazard metrics such as structural profiling of mode of action, reactivity, and/or estrogen binding affinity. Changes or errors in chemical quantity could result in differences in the classification of exposure as exposure and risk classifications are highly sensitive to emission rate and use quantity. The ERC classifications thus reflect exposure and risk in Canada on the basis of what is estimated to be the current use quantity and may not reflect future trends.
In addition, it should be noted that in this assessment, evaluation of the potential to cause ecological harm considered each substance individually. If exposure to multiple substances occurs simultaneously, this could result in cumulative effects on organisms and potentially present a higher risk. The potential for cumulative effects and how they may manifest in the environment were not further investigated due to the low ecological risk classification of these substances, considering both ecological exposure and hazard under the ERC approach..
The critical data and considerations used to develop the substance-specific profiles for the substances in the Ketones Group, and the hazard, exposure, and risk classification results are presented in ECCC (2016b).
The hazard and exposure classifications for the substances in the Ketones Group are summarized in Table 6‑1.
| Substance | ERC hazard classification | ERC exposure classification | ERC risk classification |
|---|---|---|---|
| MEKa | low | high | low |
| MPK | low | low | low |
| MIBKa | low | low | low |
| MIAK | low | low | low |
| DAA | low | high | low |
| Diacetyl | low | low | low |
| Acetoin | low | low | low |
| 2,3-PD | low | low | low |
| 2,4-PD | low | low | low |
| MO | low | low | low |
a Air concentration data from the National Air Pollution Surveillance (NAPS) program exist for these substances and were considered, but did not have any impact on the ecological classification (refer to sections 6.1.1 and 6.2.1 for NAPS data on MEK and MIBK, respectively).
On the basis of low hazard and low exposure classifications according to information considered under ERC, MPK, MIBK, MIAK, diacetyl, acetoin, 2,3-PD, 2,4-PD, and MO were classified as having a low potential for ecological risk. It is therefore unlikely that these substances are resulting in concerns for the environment in Canada.
According to information considered under ERC, MEK and DAA were classified as having a high exposure potential on the basis of high reported use quantities according to information submitted in response to a CEPA section 71 survey (Environment Canada 2013), in combination with a critically long half-life in air. MEK and DAA were classified as having a low hazard potential and a low potential for ecological risk. It is therefore unlikely that these substances are resulting in concerns for the environment in Canada.
7. Potential to cause harm to human health
7.1 Assessment of subgroup 1 (MEK, MPK)
7.1.1 Exposure assessment of subgroup 1 (MEK, MPK)
Environmental media
MEK was monitored by the National Air Pollution Surveillance (NAPS) program (ECCC 2017a). Measured concentrations of MEK in ambient air were compiled from 56 NAPS monitoring stations across Canada from 2006 to 2010. During this period, the mean and 95th percentile of 24-hour concentrations of MEK from the various stations ranged from 0.20 µg/m3 to 5.7 µg/m3 and 0.53 µg/m3 to 19.3 µg/m3, respectively (ECCC 2017a). Information available from the Hamilton Air Monitoring Network (HAMN) in Ontario indicates that MEK concentrations measured over various 12-day periods between 2015 and 2019 in ambient air at 3 locations within the Hamilton industrial sector ranged from below the reliable detection limit (RDL, 0.295 µg/m3) to a maximum of 31.5 µg/m3. The average concentrations from these sites ranged from 1.19 µg/m3 to 6.11 µg/m3 (HAMN 2020).
Outdoor air concentrations for MEK and MPK were also measured in 5 Canadian air studies conducted in Windsor, Regina, Halifax, Edmonton, and Ottawa (Zhu et al. 2005; Health Canada 2010a,b, 2012, 2013). Outdoor air samples from these studies were measured near homes (for example, backyards) that were included in the residential indoor air component of the studies described in further detail below.
MEK concentrations in ambient air from these Canadian studies ranged from <0.03 µg/m3 to 39.06 µg/m3, with geometric mean and 95th percentile concentrations ranging from 0.41 µg/m3 to 1.77 µg/m3 and from 0.78 µg/m3 to 5.53 µg/m3, respectively (Zhu et al. 2005; Health Canada 2010a,b, 2012, 2013). Ambient air concentrations for MPK from the 5 Canadian studies ranged from 0.002 µg/m3 to 14.38 µg/m3, with geometric mean and 95th percentile concentrations ranging from 0.055 µg/m3 to 0.49 µg/m3 and from 0.22 µg/m3 to 1.25 µg/m3 (Health Canada 2010a,b, 2012, 2013). The maximum concentration of 31.5 µg/m3 for MEK from the HAMN and the 95th percentile concentration of 1.25 µg/m3 for MPK from the Regina study were conservatively used to estimate outdoor air exposures to MEK and MPK for the general population in Canada.
As MEK can be used by facilities and is reported by the NPRI to be released to air in Canada (see section 4), the US EPA model SCREEN3 was used to estimate potential exposures to those living near facilities that release MEK to ambient air. Results of these modelled concentrations are presented in Table 7‑1, and input parameters for the model can be found in Appendix B. The highest estimated annual concentration of MEK in ambient air (406 µg/m3) was used to estimate potential high-end exposures of residents living near facilities releasing MEK to air.
| Exposure parameter | Concentration of MEK (µg/m3) |
|---|---|
| 1-hour maximum concentration | 142 to 2,029 |
| 24-hour maximum concentrationa | 57 to 812 |
| Annual concentrationb | 28 to 406 |
a Assuming a continuous release occurring over a 24-hour period and considering the changing wind direction over this period, a maximum 24-hour average concentration is estimated by multiplying the maximum 1-hour concentration by a factor of 0.4 (US EPA 1992).
b For exposures over the span of a year, it can be expected that, with changing wind directions, the substance air concentrations within an area release source may not vary to the same extent as those of point release sources. The meteorological conditions giving rise to a maximum 1-hour exposure can persist for a longer duration; thus, the maximum concentration for 1 year is determined by multiplying the maximum 1-hour concentration by a factor of 0.2 (US EPA 1992).
MEK and MPK were measured in the national Canadian indoor air study conducted from 2009 to 2011 as part of cycle 2 of the Canadian Health Measures Survey (CHMS). MEK was detected in 99% of the samples, with a geometric mean concentration of 1.14 µg/m3 (weighted data at the household level) and a 95th percentile concentration of 9.76 µg/m3. MPK was detected in 97% of the samples, with a geometric mean concentration (weighted data at the household level) of 0.36 µg/m3 and a 95th percentile concentration of 1.58 µg/m3 (Zhu et al. 2013). Similar indoor air concentrations for both MEK and MPK were observed in cycle 3 of the CHMS (2012–2013) (Li et al. 2019).
Indoor air concentrations for MEK and MPK were also measured across the same 5 Canadian studies referred to above. Indoor measurements were taken in the family or living rooms of selected residential homes. Between 45 and 48 non-smoking participant homes in Windsor were monitored from January 2005 to August 2006, with samples collected every 24 hours for 5 consecutive days (Health Canada 2010a). In the Regina study, a total of 146 homes, of which 34 had at least 1 smoking participant, were monitored in both the winter and summer of 2007, with one 24-hour sample and one 5-day sample collected from each household (Health Canada 2010b). In the Halifax study, 50 non-smoking homes were monitored in both the winter and summer of 2009, with samples collected every 24 hours for 7 consecutive days (Health Canada 2012). In the Edmonton study, 50 non-smoking homes were monitored in both the winter and summer of 2010, with samples collected every 24 hours for 7 consecutive days (Health Canada 2013). In the Ottawa study, 75 residential homes, of which 10 had smoking indoors, were monitored during the winter of 2002/2003, with samples of 10 L of air at a rate of 100 mL/min for 100 minutes (Zhu et al. 2005). Geometric mean concentrations of MEK in indoor air ranged from 1.14 µg/m3 to 9.81 µg/m3, with 95th percentile values ranging from 9.9 µg/m3 to 51.63 µg/m3 (Zhu et al. 2005; Health Canada 2010a,b, 2012, 2013). Geometric mean MPK concentrations in indoor air ranged from 0.089 µg/m3 to 2.77 µg/m3, with 95th percentile values ranging from 1.34 µg/m3 to 12.77 µg/m3 (Health Canada 2010a,b, 2012, 2013).
MEK was also measured in all 36 indoor air samples from an Ottawa field study, with a maximum concentration of 92.6 µg/m3 (Won et al. 2019). MEK was also measured in 81% of the 54 indoor air samples from a Nunavik field study, with a maximum concentration of 14.9 µg/m3 (Won et al. 2019).
As a conservative approach, the highest concentrations of MEK and MPK across all of the indoor air studies were used to estimate general population exposures (92.6 µg/m3 for MEK from the Ottawa field study and 12.77 µg/m3 for MPK from the Regina study).
MEK and MPK were also measured in personal air in the Windsor, Ontario air study (Health Canada 2010a). Personal air samples were taken every 24 hours for 5 consecutive days in the 2005 study. Personal air samples take into account exposures to MEK from both indoor and outdoor air at various locations including the home, office, and during transit. The geometric mean and 95th percentile concentrations for MEK in the summer were 8.79 µg/m3 and 28.22 µg/m3, respectively, which were higher than the values for the samples collected during the winter (geometric mean of 4.24 µg/m3 and 95th percentile of 11.44 µg/m3). The geometric mean personal air concentrations of MPK in the summer and winter were 2.91 µg/m3 and 1.44 µg/m3, respectively. The 95th percentile concentrations for MPK in the summer and winter were 12.65 µg/m3 and 6.30 µg/m3, respectively (Health Canada 2010a).
MEK and MPK have very high water solubilities but, on the basis of their high vapour pressures and moderate Henry’s law constants, they are expected to volatilize rapidly from water (HSDB 1983-). In addition, MEK and MPK are expected to biodegrade in water, making it unlikely for water to be a major source of human exposure (VCCEP 2003; HSDB 1983-). In 2000, MEK was not detected at above the limit of detection of 0.99 µg/L in potable water (sample size unknown) from Montreal, Quebec (Bernier 2000). No other Canadian data on the presence of MEK in water were identified. MEK was detected in a limited number of drinking water studies in the U.S with low detection frequencies (0.5% to 0.8% of samples) and concentrations ranging from 0.6 µg/L to 340 µg/L (Grady and Casey 2001; Delzer and Ivahnenko 2003; Grady 2003). No data were identified on the presence of MPK in water in Canada. MPK was detected in drinking water in Ottumwa, Iowa at a concentration of 0.1 µg/L (0.1 ppb) and was identified, but not quantified, in drinking water from 5 other US cities, as well as in England (HSDB 1983-). As a conservative approach and in order to estimate potential drinking water exposures to MEK and MPK, the detection limit of 0.99 µg/L from the Montreal study and the data from Iowa (0.1 µg/L) for MEK and MPK, respectively, were used.
For soils, one Canadian study was identified in which Golder Associates (1987) surveyed levels of MEK in soil in 2 parkland areas in the vicinity of southern Ontario petroleum refineries. MEK was detected in 19 of 30 soil samples, with a mean concentration of 5.78 µg/g (maximum: 25 µg/g). Using the maximum concentration of MEK in soil resulted in general population exposures of below 1 ng/kg bw/day for all age groups; therefore, exposure to MEK from soil is considered to be negligible. No information on the presence of MPK in soil or sediment was identified for Canada or elsewhere. ChemCAN v6.00 (ChemCAN 2003) was used to derive potential soil concentrations of MPK using the quantity data from Table 4‑1 (that is, 1,097,844 kg) and assuming that the total import quantity was released into a single region of Canada (that is, the Ontario Mixed-Wood Plain region). The estimated concentration of MPK in soil was 0.0682 µg/kg and resulted in intakes of less than 1 ng/kg bw/day for the general population of Canada, which are considered negligible.
Estimates of MEK and M[K exposure from environmental media (outdoor air, indoor air, and drinking water) ranged from 16.9 µg/kg bw/day for people 60 years of age and older to 51.0 µg/kg bw/day for people 6 months to 4 years of age and from 2.3 µg/kg bw/day for people 60 years of age and older to 6.8 µg/kg bw/day for people 6 months to 4 years of age, respectively (Health Canada 2024). More detail is available in Health Canada (2024).
Food
MEK and MPK in food or as volatiles derived from food have been measured in most food groups primarily as a result of their natural occurrence in plants or from production by microbes (that is, fermentation). In addition, it has been noted that MEK and MPK are used as flavouring agents in food, including baked goods, fats/oils, frozen dairy, gelatins/puddings, non-alcoholic beverages, and soft candy (Burdock 2010). In Canada, MEK is used as a food additive in natural extractives and in spice extracts; however, it is expected to be a minor contributor of MEK exposure compared to natural occurrence in foods. MEK (5 out of 12 samples) and MPK (4 out of 12 samples) were detected but not quantified in human milk (Pellizzari et al. 1982).
The Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Expert Committee on Food Additives (JECFA) evaluated a group of 39 saturated aliphatic acyclic secondary alcohols, ketones, and related saturated and unsaturated esters used as food flavouring agents, including MEK and MPK (WHO 1999b). As part of that evaluation, the Committee estimated the per capita intake of MEK and MPK from their use as a food flavouring agent to be 0.6 µg/kg bw/day and 0.7 µg/kg bw/day, respectively, for the US population and 2 µg/kg bw/day for both MEK and MPK for the European population (see Appendix C for more details). In the absence of data on actual use in Canada, these estimates of per capita intake are acceptable estimates of possible dietary exposure to MEK and MPK from their use as food flavoring agents for the general population (1 year of age and older).
Exposure is not expected when MEK and MPK are used as components in the manufacture of food packaging materials and when MEK is used as a component in incidental additives (cleaners) used in food processing establishments.
Estimates of exposure for MEK and MPK based on their natural occurrence in foodFootnote 5 ranged from 66 µg/kg bw/day for 14- to 18-year-olds to 185 µg/kg bw/day for 1-year-olds and from 68 µg/kg bw/day for 14- to 18-year-olds to 216 µg/kg bw/day for 1-year-olds, respectively (see Appendix C for more details).
Products available to consumers
MEK
Based on notifications submitted under the Cosmetic Regulations to Health Canada, MEK is used in certain cosmetic products in Canada such as face moisturizer and in various nail care products including base coats, top coats, nail polish, nail polish remover, nail adhesives, nail brush cleaners, nail hardeners, nail creams, products to reduce drying time, and nail repair products (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, dated August 2016, April 2017, and November 2019; unreferenced). The use of MEK in cosmetics functions as a solvent or a perfuming agent (European Commission 2017).
Concentrations of inhalation exposure were derived for certain sentinel products (top coat, nail polish, and nail polish remover) which represent the highest exposures when compared to values for similar products using ConsExpo Web (2016). Table 7‑2 summarizes the range of MEK concentrations for these various products, along with the associated estimates of inhalation exposure. Only exposure estimates for people 6 months to 4 years of age and 12 years of age and older are shown; however, these values represent the range of potential exposures for all age groups. Details on the method and parameters used to estimate inhalation exposures to MEK from cosmetics are available in Appendix D.
| Product scenario | Max concentrationa | Mean event concentration (mg/m3) | 7-hr TWAb (mg/m3) |
|---|---|---|---|
| Top coat (12 years and older) | 55.7% | 140 | 6.0 |
| Nail polish (12 years and older) | 94.7% | 520 | 43.3 |
| Nail polish (6 months to 4 years) | 94.7% | 180 | 15 |
| Nail polish remover (12 years and older) | 84%c | 280 | 5.3 |
| Nail polish remover (6 months to 4 years) | 79.6% | 230 | 4.4 |
Abbreviation: TWA, time-weighted average
a Personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017, and November 2019; unreferenced.
b 7-hour time-weighted average (TWA) concentrations were derived for all product scenarios to match up with the exposure durations in the critical effects study used to characterize risk. 7-hr TWA = mean event concentration (mg/m3) × exposure duration (min) / (7 × 60 min)
c Product not anticipated to be used by young children (personal communication, emails from the CHPSD, HC to the ESRAB, HC, dated April 2017 and November 2019; unreferenced).
Although dermal exposure would be expected to contribute to the overall exposure from use of products available to consumers, the primary route is considered to be inhalation. Wilkinson and Williams (2001) measured a dermal absorption of less than 1% for MEK in a non-occluded human in vitro study. Given the high volatility and low dermal absorption of MEK, dermal exposure is considered to be minimal in comparison to inhalation exposure; therefore, only inhalation estimates are presented.
Children’s products
MEK was identified in several different children’s products in the United States and Denmark, including pacifiers and teething rings (WSDE 2020) as well as slimy toys (Svendsen et al. 2005). It is considered that these MEK-containing products may also be present on the Canadian market. Oral (mouthing) and inhalation exposures are expected to be the main routes of exposure when these children’s products are used.
The potential oral exposure from mouthing toys or children’s objects containing MEK was estimated using a pacifier and teether as sentinel exposure scenarios. MEK was identified as a contaminant in pacifiers/teethers by the Washington State Department of Ecology (WSDE 2020), with concentrations ranging from equal to or greater than 100 ppm to less than 500 ppm. Oral exposures estimated using the approach outlined in Appendix D ranged from 110 µg/kg bw/day to 232 µg/kg bw/day for people 6 months to 4 years old and from 91 µg/kg bw/day to 300 µg/kg bw/day for people 0 to 6 months old.
Limited data were available regarding the migration of MEK from products available to consumers. According to Svendsen et al. (2005), MEK did not migrate into artificial sweat and saliva from slimy toys with concentrations of 2.3% and 9%. The breathing zone concentration for slimy toys was estimated to be 0.079 µg/m3 and 0.098 µg/m3 (Svendsen et al. 2005), which are lower than indoor air concentrations presented in the environmental media section. Nilsson et al. (2006) analyzed MEK in adult toys using headspace analyses and artificial sweat (pH of 4.5 and 6.5). Concentrations of MEK were 174 ng to 13,016 ng/180 minutes in headspace analyses, 12 µg/dm2 to 49 µg/dm2 in artificial sweat with a pH of 4.5, and 17 µg/dm2 in artificial sweat with a pH of 6.5.
Other products available to consumers
MEK is reported as being found as a non-medicinal ingredient in a limited number of currently licensed topical NHPs in Canada, including an antiseptic skin cleanser and an acne therapy product (LNHPD [modified 2021]). The sentinel scenarios for cosmetics are considered to address any exposures to MEK from the use of NHPs.
MEK is also used in numerous products available to consumers (CPID [modified 2019]). Only product scenarios that result in the highest levels of potential exposure to MEK by the inhalation route are presented in Table 7‑3. Potential inhalation exposures were estimated using ConsExpo Web (ConsExpo 2016). Appendix D summarizes the details of the parameters used in each model.
| Product scenario | MEK concentration | Mean event concentration (mg/m3) | 7-hr TWAa (mg/m3) |
|---|---|---|---|
| Paint or coating remover or stripper (for example, lacquer remover) | 20 to 40%b | 1,800 to 3,500 | 257 to 500 |
| Adhesive remover, degreaser | 100%c | 1,800 | 921 |
| Paint or coating thinners) | 100% diluted to 3% in coatingc | 840 | 120 |
| Liquid paint (solvent-rich) for truck bed | 10 to 20%d | 110 to 210 | 35 to 66 |
| Spray products (for example, spray paint) | 1 to 75%e,f | 57 to 3,900 | 3.4 to 232 |
| PVC primer only | 15 to 40%g | 7.7 to 18 | 4.4 to 10 |
| PVC cement only | 5 to 70%h | 2.6 to 18 | 1.5 to 10 |
| PVC cement and primeri | 10 to 55%j | 12 to 48 | 7 to 27 |
| Multi-purpose adhesive | 3 to 100%k,l | 8.8 to 280 | 1.7 to 53 |
| Automotive choke and throttle cleaner | 1 to 5%m | 0.099 to 0.23 | 0.0035 to 0.0082 |
| Water repellent/protectant (for example, windshield cleaner/protectant spray) | 10 to 25%n | 44 to 110 | 6.28 to 15.7 |
Abbreviation: TWA, time-weighted average
a 7-hour time-weighted average (TWA) concentrations were derived for all product scenarios to correspond with the exposure durations in the critical effects study used to characterize risk. 7-hr TWA = mean event concentration (mg/m3) × exposure duration (min) / (7 × 60 min)
b SDS 2018a
c SDS 2019a
d SDS 2021
e SDS 2016a
f SDS 2019b
g SDS 2019e
h SDS 2018b
i This scenario describes the exposure from application of PVC cement primer, followed by PVC cement, at the highest reported concentration for each product available to consumers in Canada, and post-application exposure to potential residue remaining from the application event.
j Average from the use of both primer and cement.
k SDS 2019c
l SDS 2014a
m SDS 2019d
n SDS 2016b
MPK
On the basis of notifications submitted under the Cosmetic Regulations to Health Canada, MPK is used in certain nail care products, including nail polish and nail adhesives (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, dated November 2019 and December 2019; unreferenced). MPK was also identified in several paint products (ECCC 2017b; CPID [modified 2019]).
Table 7‑4 summarizes inhalation exposure estimates, obtained using ConsExpo Web (ConsExpo 2016), for products available to consumers containing MPK. Similar to the case of MEK, although dermal exposure may contribute to overall exposure during use of products available to consumers, the primary route is considered to be inhalation; therefore, only inhalation estimates are presented.
| Product scenario | MPK concentration | Mean event concentration (mg/m3) | 6-hr TWAa (mg/m3) |
|---|---|---|---|
| Liquid paint for steel (high-solid) (20 years and older) | 1 to 10%b | 87 to 870 | 32 to 319 |
| Nail adhesive/nail polish (12 years and older) | 1 to 3%c | 4.9 to 15 | 0.48 to 1.46 |
| Nail polish (6 months to 4 years) | 0.3 to 1%c | 0.61 to 2 | 0.059 to 0.19 |
Abbreviation: TWA, time-weighted average
a 6-hour time-weighted average (TWA) concentrations were derived for all product scenarios to match up with the exposure durations in the critical effects study used to characterize risk. 6-Hr TWA = Mean event conc. × exposure duration / 6 × 60 min.
b ECCC 2017b
c Personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017, and November 2019; unreferenced.
7.1.2 Health effects assessment of subgroup 1 (MEK and MPK)
MEK and MPK are structurally similar monoketones that differ in chain length by only one carbon. Owing to the similarity of the exposure effects of MEK and 2-butanol, as well as the finding that 2-butanol is rapidly metabolized to MEK in rats, 2-butanol is used as an analogue to inform the health effects assessment of this group. Toxicity data on 2-butanol have been used to read across to MEK or MPK where required (see in Appendix A).
7.1.2.1 MEK
MEK has been reviewed by the OECD (1997a), the US EPA (2003a), and the ATSDR (2020), and these reviews provide a basis for the health effects characterization in this assessment. Literature searches were conducted beginning a year prior to the US EPA (2003a) Integrated Risk Information System report up until October 2019. Targeted literature searches were conducted up to March 2021. No health effect studies that would impact the risk characterization (that is, result in different critical endpoints or lower points of departure [POD] than those stated in US EPA 2003a) were identified.
Toxicokinetics
Orally administered MEK has been shown to be extensively absorbed from the gastrointestinal tract of rodents and rapidly eliminated (Dietz et al. 1981, reviewed in US EPA 2003a). Due to its high blood/air solubility ratio, it is also well absorbed in both humans and rats upon inhalation exposure. Similarly, it was found to be rapidly absorbed upon dermal exposure (Munies and Wurster 1965, reviewed in US EPA 2003a). The available information indicates that the metabolism of MEK is similar in humans and laboratory animals, with 2-butanol and 2,3-butanediol being the major metabolites (Perbellini et al. 1984; Liira et al. 1988, 1990a, reviewed in US EPA 2003a). In rats and guinea pigs, a small portion of absorbed MEK is reduced to 2-butanol, which is rapidly oxidized back to MEK (US EPA 2003a). In humans, MEK appears to form endogenously (since it has been identified as a minor but normal constituent of urine) as a constituent in the serum and urine of diabetics, and in expired air (WHO 1992, reviewed in US EPA 2003a).
Carcinogenicity and genotoxicity
The US EPA (2003a) concluded that the available “data are considered inadequate for an assessment of human carcinogenic potential,” given that studies of humans chronically exposed to MEK are inconclusive and that MEK has not been tested for carcinogenicity in animals by the oral or inhalation routes. ATSDR (2020) has also supported this conclusion and indicated that the preliminary epidemiological studies suggest that occupational exposure to MEK does not increase the development of neoplasms. Although the genotoxic effects of MEK have been examined using several different test systems, they have been consistently negative in genotoxicity studies, both in vitro and in vivo (US EPA 2003a).
Repeated-dose toxicity
No chronic-duration studies examining effects in animals following inhalation exposure were identified. However, several shorter-term, repeat exposure inhalation studies of MEK have been reported.
The reported health effects of MEK were primarily related to developmental effects (discussed in the following section) as well as reversible neurological effects. Absolute and/or relative organ weight increases, most frequently for liver, were also observed (US EPA 2003; ATSDR 2020).
Reproductive and developmental toxicity
There are no studies evaluating the reproductive toxicity potential of MEK by any route of exposure. Several studies investigating the potential for developmental toxicity after inhalation to MEK were identified.
MEK is described in ATSDR (2020) as slightly fetotoxic, and the effects described were reduced fetal weight, skeletal variations, and delayed or incomplete ossification.
The developmental toxicity study conducted by Deacon et al. (1981) exposed pregnant rats to average MEK concentrations of 1,215 mg/m3, 2,955 mg/m3, and 8,865 mg/m3 (in whole body dynamic exposure chambers) for 7 hours/day on gestation days (GD) 6 to 15. Decreased body weight gain and increased water consumption were observed in the dams at 8,865 mg/m3, indicating maternal toxicity. At this dose, skeletal abnormalities, including delayed ossification of the cervical centra and extra ribs, were also observed in the pups.
In a subsequent inhalation developmental toxicity study supporting the previous findings of skeletal anomalies, pregnant mice were exposed to mean concentrations of 0 ppm, 398 ppm, 1,010 ppm, and 3,020 ppm (0 mg/m3, 1,174 mg/m3, 2,980 mg/m3, and 8,909 mg/m3) MEK by inhalation in whole body exposure chambers for 7 hours/day on GD 6 to 15 (Schwetz et al. 1991, reviewed in US EPA 2003a). A slight but statistically significant dose-related increase in absolute liver weight was observed in dams at 8909 mg/m3 (an increase of approximately 7% when compared with the control). There was a statistically significant decrease in mean fetal weight (5%, per litter) in males and a 4% decrease for all fetuses combined at the highest dose tested. As exposure levels increased, the incidence of fetuses with misaligned sternebrae also increased, showing a positive trend. Other non-significant developmental effects (cleft palate, fused ribs, missing vertebrae, and syndactyly) were observed in exposed groups but not in controls. The no observed adverse effect concentration (NOAEC) for both maternal and developmental adverse effects was considered to be 2,980 mg/m3, while the developmental and maternal lowest observed adverse effect concentrations (LOAECs) were established at 8,909 mg/m3 on the basis of the decreased fetal weight among males, the increased incidence of misaligned sternebrae, and an increased relative liver weight in dams. On the basis of the data for misaligned sternebrae (considered the most protective endpoint), the US EPA (2003a) derived a lower limit on an effective concentration using a 10 percent response level (LEC10)Footnote 6 of 5,202 mg/m3 (95% lower confidence on the concentration associated with a 10% extra risk) for intermittent exposure (7 hours per day) and a LECHECFootnote 7 (human equivalent concentration adjusted for continuous exposure) of 1,517 mg/m3.
A inhalation developmental toxicity study in pregnant rats exposed to 0 mg/m3, 2,950 mg/m3, 5,900 mg/m3, 11 800 mg/m3, or 17,700 mg/m3 (as whole body exposures) for 6 hours/day on GD 6 to 20 also indicated developmental and maternal effects (Saillenfait et al. 2006). A NOAEC of 2,949 mg/m3 was considered on the basis of decreased fetal body weight observed at 5,899 mg/m3, while decreased maternal body weight gain (52%), food consumption (12%), and increases in the incidence of incomplete sternebrae ossification were also noted at 11,797 mg/m3 and above (Saillenfait et al. 2006). In another developmental study, developmental effects were reported in the absence of maternal toxicity at 7,723 mg/m3, with a NOAEC of 3,322 mg/m3 (Schwetz et al. 1974 as cited in US EPA 2003a).
In the absence of any reproductive studies for MEK nor any oral developmental toxicity studies reported for MEK, a read-across from the oral reproductive and developmental toxicity study with 2-butanol (which is rapidly metabolized to MEK in rats) was used. In a multigenerational drinking water study, male and female Wistar rats were exposed to 2-butanol concentrations of 0%, 0.3%, 1.0%, or 3.0% (equivalent to doses of 0 mg/kg bw/day, 538 mg/kg bw/day, 1,644 mg/kg bw/day, and 5,089 mg/kg bw/day [male rats] and 0 mg/kg bw/day, 594 mg/kg bw/day, 1,771 mg/kg bw/day, and 4,571 mg/kg bw/day [female rats]) for 8 weeks before mating (Cox et al. 1975, reviewed in US EPA 2003a). Because increased mortality and decreased body weight were observed in the F1A litters at the 3% dose level, the high-dose was reduced to 2% (average daily intake of 3,384 mg/kg bw/day in males and 3,122 mg/kg bw/day in females) for the remainder of the study. F0 females were mated again and F1A pups mated to produce litters F2. Concentrations as high as 3% did not affect reproductive performance in rats but did produce maternal toxicity (decreased body weight gain in dams). As noted by the US EPA (2003a), this study did not include evaluation of certain parameters routinely measured in studies of more current design. Deficiencies included the lack of measurements of estrous cyclicity, sperm parameters, and weights of uterus, epididymides, seminal vesicles, and brain; and less than complete clinical chemistry/hematology and histopathology. A no observed adverse effect level (NOAEL) of 1,771 mg/kg bw/day for 2-butanol for both maternal and developmental effects was established. For decreased pup body weights at postnatal day 21 in the F1A generation (representing the most sensitive effects observed), the US EPA (2003a) estimated a lowest effective dose (LED)05 (95% lower confidence limit on the effective dose [ED]) of 639 mg/kg bw/day for MEK (adjustment based on the molecular weights from the LED05 of 657 mg/kg bw/day for 2-butanol).
No dermal developmental toxicity studies were identified for MEK or the analogue 2-butanol.
Short-term and acute toxicity
Primates (4 juvenile baboons) exposed continually for 7 days to approximately 300 mg/m3 MEK were reported to show early signs of narcosis, incoordination, and delayed reaction times in neurobehavioural tests (Geller et al. 1979). As discussed in ATSDR (2020), the tests evaluated responses to a complex discriminant behavioural task, which was able to detect subtle neurobehavioural effects. However, it was also noted that only one exposure level was tested, that the observed effect decreased somewhat near the end of the experiment, and that no statistical tests were performed (IPCS 1993; ATSDR 2020).
Effects in humans
Neurological and respiratory effects are the most commonly examined effects of MEK exposure in human volunteers and other occupationally exposed individuals (ATSDR 2020). Chronic occupational exposure to MEK has been examined in several epidemiological studies reviewed by the US EPA (2003a). However, the available data were considered to be inadequate to demonstrate an association between exposure to MEK and chronic neurological, pulmonary, or other effects due to several limitations, which include limited characterization of exposure or methodological issues.
Mild central nervous system (CNS) effects (for example, headache, fatigue, and feelings of intoxication) were observed in volunteer subjects exposed to approximately 300 mg/m3 for 4 hours, with females scoring higher on symptom questionnaires compared with men (Tomicic et al. 2011). Similar effects were also observed in other studies at higher doses (Muttray et al. 2002; Dick et al. 1984, 1988, 1989, 1992 as reviewed in US EPA 2003a; ATSDR 2020).
7.1.2.2 MPK
MPK has not been assessed by other agencies. The following health effects assessment is based on the information identified from ECHA registration dossiers (ECHA c2007-2019a) and the US EPA High Production Volume Information System (HPVIS; US EPA 2010).
Toxicokinetics
No quantitative information on the absorption of MPK via any route of exposure was identified. MPK is structurally similar to MEK and its metabolite 2-butanol; therefore, it is expected that MPK will undergo similarly rapid and extensive absorption.
Carcinogenicity and genotoxicity
No oral, dermal, or inhalational carcinogenicity studies were reported for MPK. No positive (Q)SAR model predictions or presence of structural alerts for genotoxicity or carcinogenicity were found. In addition, MPK was not genotoxic in several in vitro assays (that is, Ames tests, chromosomal aberration test, and mouse lymphoma assay; ECHA c2007-2019a).
Repeated-dose toxicity
In an oral repeat-dose toxicity study, CD male rats were administered MPK in drinking water at concentrations of 0%, 0.25%, 0.5%, and 1.0% MPK (equivalent to doses of 0 mg/kg bw/day, 144 mg/kg bw/day, 250 mg/kg bw/day, and 454 mg/kg bw/day, respectively) for a period of 13 months. The only effect reported was decreased body weight gain (9%) at the highest dose tested (454 mg/kg bw/day). There were no observed clinical effects, nor were there any changes to organ weights or histology. Also, no pathological changes were observed in the central or peripheral nervous systems (Bingham et al. 2001 as cited in HSDB 1983; US EPA 2010).
In a sub-chronic inhalation study, rats were exposed to MPK for 6 hours per day on a 5-day per week basis for 13 weeks at concentrations of 250 ppm, 750 ppm, or 1,500 ppm (880 mg/m3, 2,640 mg/m3, or 5,300 mg/m3, respectively). Test substance-related observations included increased incidences of red material around the nose and facial areas in females and microscopic findings in the nasal area, larynx, and thyroid in males from all exposure groups. However, these effects were not considered adverse by the study authors. No effects on arousal-response were observed. The NOAEC was reported in the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) Registration Dossier as being 5300 mg/m3 for males and females (ECHA c2007-2019a).
For the dermal route, no suitable study was identified.
Reproductive and developmental toxicity
In a combined inhalation reproductive/developmental toxicity study, SD rats were exposed to up to 5,000 mg/m3 MPK 6 hours/day for 35 to 48 days (females) until GD19 and for 51 days (males) (ECHA c2007-2019a; US EPA AcToR 2015). At the highest dose, both males and females were observed to have decreased alertness, reduced activity level, and slower response to tapping on the chamber wall. No other significant effects were reported. The NOAEC for reproductive and developmental toxicity was reported in the REACH Registration Dossier as being 5,000 mg/m3 (ECHA c2007-2019a).
In a prenatal inhalation developmental toxicity study, female SD rats were exposed to concentrations of 0 ppm, 250 ppm, 750 ppm, and 1500 ppm (equivalent to 0 mg/m3, 880 mg/m3, 2,650 mg/m3, and 5,300 mg/m3, respectively) of MPK from GD 6 to 19 (ECHA c2007-2019a). No effects on mean body weights, body weight gains, net body weights, net body weight gains, gravid uterine weights, or food consumption in any test substance-exposed group were observed. No adverse effects on maternal animals or effects on intrauterine growth, survival, and fetal morphology were observed at any exposure level, including effects to arousal response. On the basis of these observations, the study authors identified a NOAEC of 1,500 ppm (5,300 mg/m3) (the highest exposure level evaluated) for maternal toxicity and embryo/fetal development.
The results of these studies indicate that MPK is not a developmental toxicant. In addition, based on the results of OECD toolbox in silico prediction tools, there is no structural alert indicating that MPK has any potential for inducing developmental toxicity.
No oral or dermal reproductive or developmental toxicity data are available for MPK.
7.1.3 Risk characterization of subgroup 1 (MEK and MPK)
MEK
Within the range of adverse effects on animals resulting from repeated inhalation exposure to MEK, there is consistent evidence from studies in rats and mice that developmental effects are the critical health effects, which occurred mainly in the presence of slight maternal toxicity (ATSDR 2020). From among the available studies, the US EPA (2003a) selected the study in mice by Schwetz et al. (1991), which has a LEC10 for intermittent exposure (7 hours/day) of 5,202 mg/m3 and a LECHEC for continuous exposure of 1,517 mg/m3, based on the incidence of misaligned sternebrae (US EPA 2003a). Therefore, these values were considered to be the most appropriate PODs to use for the risk characterization of MEK.
In the absence of oral repeat-dose toxicity studies for MEK, the reproductive and developmental drinking water toxicity study of 2-butanol in rats (Cox et al. 1975 as cited in US EPA 2003a) was selected to characterize risk by the oral route. The US EPA (2003a) estimated a LED05 (95% lower confidence limit on the effective dose, ED) of 639 mg/kg bw/day for MEK (adjustment based on the molecular weights from the LED05 of 657 mg/kg bw/day for 2-butanol) on the basis of decreased pup survival and decreased neonatal body weight in the presence of maternal toxicity.
The predominant source of exposure to MEK from environmental media (outdoor air, indoor air, and drinking water) and food for the general population is through the diet and, to a lesser extent, indoor air. On the basis of the available data, it is expected that the majority of dietary exposure to MEK results from its natural occurrence in foods. Intake of MEK from its natural occurrence in food was not identified as a concern for human health.
The general population of Canada may also be exposed to MEK when using various products available to consumers that contain the substance, including nail products, paints, and DIY products, primarily through inhalation. Table 7‑5 provides relevant exposure values and effect levels for critical health effects as well as the resultant margins of exposure (MOEs) for the risk characterization of MEK.
| Exposure scenario | Exposure | Critical effect level | Critical health effect | MOE |
|---|---|---|---|---|
| Environmental mediaa (all age groups) | 16.9 to 51.0 µg/kg bw/day | LED05 = 639b mg/kg bw/day | Developmental study for 2-butanol; decreased fetal and pup weights, and body weight gain in dams | > 12,529 |
| Environmental media for residents near facilities releasing MEKc (all age groups) | 80.6 to 243.6 µg/kg bw/day | LED05 = 639b mg/kg bw/day | Developmental study for 2-butanol; decreased fetal and pup weights, and body weight gain in dams | 2,623 to 7,922 |
| Food flavouring agent (oral) (1 year and older) | 0.6 to 2.0 µg/kg bw/day | LED05 = 639b mg/kg bw/day | Developmental study for 2-butanol; decreased fetal and pup weights, and body weight gain in dams | > 319,500 |
| Nail products (inhalation) (6 months and older) | 4.4 to 43.3 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 120 to 1,182 |
| Pacifiers and teethers (oral) (0 to 4 years) | 0.09 to 0.30 mg/kg bw/day |
LED05 = 639b mg/kg bw/day | Developmental study for 2-butanol; decreased fetal and pup weights, and body weight gain in dams | 2,130 to 7,100 |
| Paint or coating remover or stripper (for example, lacquer remover) (inhalation) (20 years and older) | 257 to 500 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 10 to 20 |
| Adhesive remover, degreaser (inhalation) (20 years and older) | 921 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 6 |
| Paint or coating thinners (inhalation) (20 years and older) | 120 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 43 |
| Liquid paint (solvent-rich) for truck bed (inhalation) (20 years and older) | 35 to 66 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 79 to 150 |
| Spray products (for example, spray paint) (inhalation) (20 years and older) | 3.4 to 232 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 22 to 1,530 |
| Acute inhalation exposure from PVC cement or PVC cement primer (5 to 70%) (20 years and older) | 1.5 to 10 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 520 to 3,468 |
| Acute inhalation exposure from use of PVC cement and primer (10 to 55%) (20 years and older) | 7 to 27 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 193 to 743 |
| Multi-purpose adhesives (inhalation) (20 years and older) | 1.7 to 53 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 98 to 3,060 |
| Automotive choke and throttle cleaner (inhalation) (20 years and older) | 0.0035 to 0.0082 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 634,390 to 1,486,286 |
| Water repellent/protectant (for example, windshield cleaner/protectant spray) (inhalation) (20 years and older) | 6.28 to 15.7 mg/m3 d | LEC10 = 5,202 mg/m3 e (intermittent exposure) | Increased incidence of misaligned sternebrae in fetus | 331 to 828 |
Abbreviations: LEC10, a lower limit on an effective concentration using a 10 percent response level; LED05, lowest effective dose (95% lower confidence limit on the effective dose [ED]); MOE, margin of exposure.
a This includes exposure from outdoor air, indoor air, and drinking water (Health Canada 2024).
b Molar adjustment calculated from read-across value (Cox et al. 1975 as cited in US EPA 2003a). Molecular weights are 74.12 g/mol for 2-butanol, 72.11 g/mol for MEK, and 86.13 g/mol for MPK. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation).
c The estimated intakes incorporate the highest estimated annual concentration of MEK in air (see Table 7‑1) associated with facilities that report their substance releases under the NPRI (2021) as well as from other environmental media (Health Canada 2024).
d 7-hour time-weighted average (TWA)
e Schwetz et al. 1991 as cited in US EPA 2003a. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation)
MOEs for exposure to MEK in environmental media, including for populations living near facilities that report releases to air, in food (from potential use as a food flavouring agent), and in pacifiers and teethers, are considered adequate to address any uncertainties in the available health effects and exposure data used to characterize risk. The JECFA (WHO 1999b) concluded there was “no safety concern with the estimated levels of intake as flavouring substances” on the basis of the dietary exposure estimates for MEK.
The calculated MOEs for inhalation exposure to nail products, multi-purpose adhesives, automotive choke and throttle cleaners, PVC cement and primers, and water repellent/protectants (for example, windshield cleaner/protectant sprays) are considered adequate. However, inhalation exposure to MEK from other products available to consumers (that is, paint or coating removers or strippers [for example, lacquer removers], adhesive removers, degreasers and paint or coating thinners, liquid paints, and spray products including spray paints) are considered inadequate to account for uncertainties in the available health effects and exposure data.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects. In addition, the potential for elevated exposure of people living near facilities that may release MEK to air was considered in the assessment.
MPK
No carcinogenicity studies were identified for MPK. However, there were no positive (Q)SAR model predictions or structural alerts for carcinogenicity. In addition, MPK was not genotoxic in several in vitro assays. In the only available oral toxicity study, minimal effects (slightly decreased body weight gain) were reported in rats at the highest dose of MPK administered in drinking water for 13 months (Bingham et al. 2001 as cited in HSDB 1983-); the NOAEL is considered to be 454 mg/kg bw/day. With respect to inhalation exposure, no adverse effects, other than slight transient neurotoxicity in rats during exposure, were observed in studies in which NOAECs of 5,000 mg/m3 and 5,300 mg/m3 were identified (ECHA c2007-2019a).
The predominant source of exposure to MPK from environmental media and food for the general population is through diet. Based on the available data, it is expected that the majority of dietary exposure to MPK results from its natural occurrence in food.
Comparing the exposure estimates for MPK from environmental media (2.3 µg/kg bw/day to 6.8 µg/kg bw/day) and from its potential use as a flavouring agent in food (0.7 µg/kg bw/day to 2.0 µg/kg bw/day) with the NOAEL of 454 mg/kg bw/day resulted in MOEs greater than 66,765, which are considered adequate to address any uncertainties in the available health effects and exposure data used to characterize risk. Additional intake of MPK from its natural occurrence in food was not identified as a concern for human health. Furthermore, the JECFA (WHO 1999b) concluded that there is “no safety concern with the estimated levels of intake as flavouring substances” on the basis of the dietary exposure estimates for MPK.
The general population of Canada may also be exposed to MPK when using products available to consumers that contain the substance, including paints and nail products, primarily through inhalation. Given the low hazard potential of MPK via inhalation, the risk to human health related to the presence of MPK in products available to consumers is considered to be low.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects.
7.1.4 Uncertainties in the evaluation of risk to human health for MEK and MPK
The key sources of uncertainty are presented in Table 7‑6.
| Key sources of uncertainty | Impact |
|---|---|
| Assumption that dermal exposures are minimal in comparison to inhalation exposures, given the high volatility of the substances. | - |
| Assumption that all of the MEK present in teethers or pacifiers can leach out of the product. | + |
| Lack of chronic oral or inhalation studies for MEK or MPK. | +/- |
| The use of 2-butanol as a read-across to assess repeated-dose toxicity and reproductive/developmental toxicity for MEK. | +/- |
| No data are available regarding the toxicity of MEK following long-term oral exposure; however, the use of 2-butanol, a major metabolite of MEK, was considered an appropriate analogue for use in risk characterization. | +/- |
+ = uncertainty with potential to cause overestimation of exposure/risk; - = uncertainty with potential to cause underestimation of exposure risk; +/- = unknown potential to cause over- or underestimation of risk.
7.2 Assessment of subgroup 2 (MIBK, MIAK, and DAA)
7.2.1 Exposure assessment of subgroup 2 (MIBK, MIAK, and DAA)
Environmental media
MIBK was monitored by the NAPS program (ECCC 2017a). Measured concentrations of MIBK in ambient air were compiled from 64 NAPS monitoring stations across Canada from 2006 to 2010. During that period, the mean and 95th percentile 24-hour concentrations of MIBK from the various stations ranged from 0.014 µg/m3 to 0.29 µg/m3 and 0.08 µg/m3 to 1.05 µg/m3, respectively (ECCC 2017a).
Information available from the HAMN in Ontario indicates that MIBK concentrations measured over various 24-hour periods between 2015 and 2019 in ambient air at 3 locations within the Hamilton industrial sector ranged from below the RDL (0.41 µg/m3) to a maximum of 6.55 µg/m3. The average concentrations from these sites ranged from levels below the RDL to 0.42 µg/m3 (HAMN 2020).
Geometric mean ambient air concentrations of MIBK from the 5 Canadian air studies referred to in section 6.1.1 ranged from less than 0.016 µg/m3 to 0.185 µg/m3, while 95th percentile concentrations ranged from 0.086 µg/m3 to 0.470 µg/m3 (Zhu et al. 2005; Health Canada 2010a,b, 2012, 2013). The highest concentration (6.55 µg/m3 from the HAMN) was used to estimate general population exposures. No information or data on MIAK and DAA in ambient air were identified in Canada or elsewhere.
As MIBK can be used by facilities and is reported to be released to air in Canada (see section 4), the US EPA model SCREEN3 (1996) was used to estimate levels of MIBK in ambient air attributable to on-site releases to air from facilities using the substance, as reported from the NPRI. The results of these modelled concentrations are presented in Table 7‑7, and input parameters for the model can be found in Appendix B. The highest estimated annual concentration of MIBK in ambient air (30 µg/m3) was used to estimate potential high-end exposures to residents living near facilities releasing MIBK to air.
| Exposure parameter | Concentration of MIBK (µg/m3) |
|---|---|
| 1-hour maximum concentration | 1.62 to 150 |
| 24-hour maximum concentrationa | 0.65 to 60 |
| Annual concentrationb | 0.325 to 30 |
a Assuming a continuous release occurring over a 24-hour period and considering the changing wind direction over this period, a maximum 24-hour average concentration is estimated by multiplying the maximum 1-hour concentration by a factor of 0.4 (US EPA 1992).
b For exposures over the span of a year, it can be expected that, with changing wind directions, the air concentrations of a substance within an area release source may not vary to the same extent as those of point release sources. The meteorological conditions giving rise to a maximum 1-hour exposure can persist for a longer duration; thus, the maximum concentration for 1 year is determined by multiplying the maximum 1-hour concentration by a factor of 0.2 (US EPA 1992).
MIBK was measured in the CHMS Cycle 2 Indoor Air Study and was detected in approximately 96% of the samples. The geometric mean concentration of MIBK from this study was 0.235 µg/m3, with a 95th percentile concentration of 1.62 µg/m3 (weighted data at the household level) (Zhu et al. 2013). MIAK was also measured in the CHMS Cycle 2 Indoor Air study but was only detected in 1.45% of samples (limit of detection of 0.19 µg/m3). Similar indoor air concentrations for MIBK and detection rates for MIAK were observed in cycle 3 of the CHMS (2012–2013) (Li et al. 2019).
Geometric mean MIBK concentrations from the 5 Canadian indoor air studies ranged from 0.14 µg/m3 to 1.33 µg/m3, with 95th percentile concentrations ranging 0.33 µg/m3 to 13.82 µg/m3 (Zhu et al. 2005; Health Canada 2010a,b, 2012, 2013). MIBK was also measured in 94% of the 36 indoor air samples from an Ottawa field study (maximum concentration of 5.5 µg/m3) and in 6% of the 54 indoor air samples from a Nunavik field study (maximum concentration of 0.6 µg/m3) (Won et al. 2019).
MIAK was measured in 24 indoor air samples from a Quebec field study, with a geometric mean concentration of 0.23 µg/m3 and a maximum concentration of 1.51 µg/m3 (Won and Lustyk 2011). DAA was measured in 15% of indoor air samples from the Nunavik field study, with a maximum concentration of 2.4 µg/m3 (Won et al. 2019).
The following indoor air concentrations were conservatively used to estimate general population exposures: the 95th percentile concentration of 13.82 µg/m3 for MIBK from the Windsor study, the maximum indoor air concentration of 1.51 µg/m3 for MIAK from the Quebec study, and the maximum concentration of 2.4 µg/m3 for DAA from the Nunavik study.
Personal air samples were also measured for MIBK in the Windsor study (Health Canada 2010a). The geometric mean personal air concentration of MIBK in the summer and winter were 1.04 µg/m3 and 0.23 µg/m3, respectively. The 95th percentile concentrations for MIBK were 7.96 µg/m3 and 0.99 µg/m3 in the summer and winter, respectively (Health Canada 2010a).
No studies measuring MIBK, MIAK, and DAA in drinking water in Canada were identified. No further information on the presence of MIAK in water elsewhere was identified. In a study conducted in the US, MIBK was detected in 3 out of 646 drinking water samples with concentrations ranging from 16 µg/L to 20 µg/L (Grady and Casey 2001). MIBK was also measured in several surface water studies (Sheldon and Hites 1978; Hall et al. 1987; Benfenati et al. 1992; Bianchi and Varney 1998). In addition, elevated concentrations of MIBK were found in groundwater, effluent, and leachate in the vicinity of landfill sites, oil reprocessing facilities, and sewage treatment plants (Mutch et al. 1983; Sabel and Clark 1984; Sawhney and Kozloski 1984; US EPA 1988; Canter and Sabatini 1994; Paxéus 1996). The maximum value of 20 µg/L reported in US drinking water was used to estimate general population exposures to MIBK. ChemCan v6.00 (ChemCAN 2003) was used to derive concentrations of MIAK and DAA in surface water using quantity data from Table 4‑1 and assuming that total import quantity was released into a single region of Canada (that is, the Ontario Mixed-Wood Plain region). Estimated surface water concentrations for MIAK and DAA were 0.15 ng/L and 46.6 ng/L, respectively, and were used to estimate drinking water intakes for the general population of Canada.
No information or data on MIBK, MIAK, and DAA in soil were identified in Canada. DAA was qualitatively detected in sediment in a lake in Saskatchewan, Canada (HSDB 1983-). Using an approach similar to that used to derive the surface water concentrations noted above, ChemCan (2003) was used to derive concentrations of MIBK, MIAK, and DAA in soil. Soil concentrations for MIBK, MIAK, and DAA were 0.13 µg/kg, 0.006 µg/kg, and 3.4 ng/kg, respectively; therefore, exposure to these substances from soil is considered to be negligible (intakes of less than 1 ng/kg bw/day).
Estimates of exposure from environmental media ranged from 2.7 µg/kg bw/day for people 60 years of age and older to 8 µg/kg bw/day for people 6 months to 4 years of age for MIBK; from 0.3 µg/kg bw/day for people 60 years of age and older to 0.9 µg/kg bw/day for people 6 months to 4 years of age for MIAK; and from 0.48 µg/kg bw/day for people 60 years of age and older to 1.44 µg/kg bw/day for people 6 months to 4 years of age for DAA (Health Canada 2024). More details are available in Health Canada (2024).
Food
MIBK was detected in various food items owing to its natural occurrence, including in beer, brandy, chicken, fruit, olive oil, eggs, coffee, and cow’s milk (Nijssen et al. 1963-2018; Burdock 2010). MIBK is also cited as being used as a flavouring ingredient in baked goods, frozen dairy, gelatins/puddings, meat products, non-alcoholic beverages, and soft candy (Burdock 2010). MIBK was detected (two out of 12 samples) but not quantified in human milk (Pellizzari et al. 1982).
The JECFA also evaluated a group of 39 saturated aliphatic acyclic secondary alcohols, ketones, and related saturated and unsaturated esters, including MIBK, used as food flavouring agents (WHO 1999b). As part of that evaluation, the Committee estimated the per capita intake of MIBK from its use as a food flavouring agent to be 0.03 µg/kg bw/day for the US population and 0.12 µg/kg bw/day for the European population (see Appendix C for more details). In the absence of data on actual usage in Canada, the per capita intake estimates are acceptable estimates of possible dietary exposure to MIBK from its use as food flavoring agent for the general population (1 year of age and older).
MIAK was identified as naturally occurring in a few food items, including coffee (0.5 µg/g) and papaya (0.001 µg/g) (Nijssen et al. 1963-2018), roasted filberts, fried bacon, cooked beef, and cooked pork (HSDB 1983-). DAA was detected as a natural component in various food items including fruit, vegetables, dairy, honey, nuts, eggs, chicken, and alcoholic beverages (Nijssen et al. 1963-2018).
Exposure is not expected when MIAK and DAA are used as components in the manufacture of food packaging materials.
Quantitative exposure estimates for MIBK, MIAK, and DAA from their natural occurrence in foodFootnote 5 ranged from 0.4 µg/kg bw/day for 4- to 8-year-olds to 32 µg/kg bw/day for adults 19 years and older for MIBK, from 0.001 µg/kg bw/day for 1-year-olds to 3 µg/kg bw/day for adults 19 years and older for MIAK, and from 0.04 µg/kg bw/day for 6- to 12-month-olds to 4 µg/kg bw/day for 2- to 3-year-olds for DAA (see Appendix C for details).
Biomonitoring
As part of the fifth cycle of the Canadian CHMS (2016 to 2017), MIBK was measured in 73.4% of whole blood samples from 2363 individuals representing the Canadian population aged 12 to 79 years (Health Canada 2019). The CHMS is an ongoing, nationally representative survey that collects important health and wellness data as well as biological samples from individuals across the country. The geometric mean and 95th percentiles of MIBK were 0.04 μg/L and 0.12 µg/L, respectively. The limit of detection was 0.029 µg/L. These data support the conclusion that there are potential chronic exposures to MIBK among the general Canadian population.
Products available to consumers
MIBK
MIBK was identified as being present as a non-medicinal ingredient in rubbing alcohol, licensed as an NHP, meant for topical use with a concentration of 0.98% (personal communication, email from the NNHPD, HC, to the ESRAB, HC, dated August 16, 2016; unreferenced). ConsExpo Web (2016) was used to estimate inhalation exposures to MIBK from use of this product for children 6 months to 4 years of age and adults 20 years and older. Parameters used in the model are outlined in Appendix D. The mean event concentration, regardless of age group for this exposure scenario, was estimated to be 0.19 mg/m3, the mean concentration on the day of exposure was estimated to be 2.7E-4 mg/m3, and the 6-hour time weighted average was estimated to be 0.003 mg/m3. Although dermal exposure could contribute to the overall exposure during use of products available to consumers, the primary route is considered to be inhalation in light of the high volatility of MIBK; therefore, only inhalation exposure estimates are presented.
On the basis of notifications submitted under the Cosmetic Regulations to Health Canada, MIBK is used in certain cosmetic products in Canada, such as temporary tattoos and in nail care products including nail polish, nail polish removers, and nail adhesives (personal communication, email from the CHPSD, HC, to the ESRAB, HC, dated November 2019; unreferenced).
MIBK is used in a variety of other products available to consumers that may result in exposure to the general population of Canada (CPID [modified 2019]). Only product scenarios that result in the highest levels of potential inhalation exposure to MIBK are presented in Table 7‑8. Incidental oral exposures for people 6 months to 4 years of age and 5 to 11 years of age (representing the highest exposed age groups) from the use of dry erase markers were also estimated, with per event exposure estimates ranging from 0.16 mg/kg bw to 0.48 mg/kg bw and from 0.32 mg/kg bw to 0.97 mg/kg bw for people 5 to 11 years of age and 6 months to 4 years of age, respectively. Potential exposures were estimated using ConsExpo Web (ConsExpo 2016) or relevant algorithms; see Appendix D for details.
| Product scenario | MIBK concentration | Mean event concentration (mg/m3) | 6-hr TWAa (mg/m3) |
|---|---|---|---|
| Nail adhesive (12 years and older)b | 1 to 3%c | 4.1 to 12 | 0.4 to 1.2 |
| Nail polish (6 months to 4 years) | 0.3 to 1%c | 0.42 to 1.4 | 0.04 to 0.14 |
| Nail polish remover (12 years and older) | 10 to 30%c | 18 to 56 | 0.4 to 1.2 |
| Nail polish remover (6 months to 4 years) | 10 to 30%c | 11 to 35 | 0.2 to 0.8 |
| Temporary tattoos (6 months and older) | 1 to 3%c | 0.035 to 0.1 | 0.0005 to 0.001 |
| Wood lacquer (20 years and older) | 2.5 to 10%d | 670 to 2,600 | 112 to 433 |
| Liquid paint (solvent-rich) for truck bed liners, appliances (20 years and older) | 10 to 15%e | 100 to 150 | 37 to 55 |
| Spray products (for example, spray paint) (20 years and older) | 0.4 to 25%f,g | 21 to 1,100 | 1.5 to 76 |
| Filler/putty from tube (automotive) (20 years and older) | 1 to 5%h | 6.7 to 28 | 1 to 5 |
| Dry erase marker (inhalation) (all age groups) | 10 to 30%i | 1.2 to 3.6 | 0.1 to 0.45 |
Abbreviation: TWA, time-weighted average
a 6-hour time-weighted average (TWA) concentrations were derived for all product scenarios to match up with the exposure durations in the critical effects study used to characterize risk. 6-Hr TWA = Mean event conc. × exposure duration / 6 × 60 min.
b Scenario covers the use of regular nail polishes.
c Personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017, and November 2019; unreferenced.
d SDS 2016c
e SDS 2021
f SDS 2020
g SDS 2017a
h SDS 2018c
i SDS 2015b
MIBK has been measured in recycled tire crumb rubber as well as in the air above synthetic turf fields (US EPA 2009a; US EPA & CDC/ATSDR 2019; Schneider et al. 2020a,b). Recycled tire crumb rubber may be used as infill or top dressing in sports fields as well as in sports centre and playground surfaces in Canada (Cantin 2009; AR 2017). The studies that measured MIBK in air samples collected at synthetic turf fields or playgrounds were considered most relevant to the general population of Canada. The US EPA (2009a) conducted a scoping study that sampled 3 synthetic turf fields and one playground, with air concentrations ranging from not detected to 0.13 ppbV (0.53 µg/m3). Schneider et al. (2020a,b) conducted a number of studies on recycled tire crumb, which involved analyzing infill samples and collecting air samples from 17 sports fields in Europe, including one indoor facility. The authors noted that the highest MIBK concentration came from an outdoor facility (Schneider et al. 2020b). The arithmetic mean concentration of MIBK from all sports facilities was 3.32 µg/m3 (median of 0.15 µg/m3) and the 95th percentile air concentration was 17.85 µg/m3. The 95th percentile concentration is lower than the highest estimated annual concentration of MIBK in ambient air (30 µg/m3), which was used to estimate potential high-end exposures to residents living near facilities releasing MIBK to air. Consequently, estimates of exposure to MIBK for individuals playing sports on synthetic turf or playing in playgrounds with synthetic turf would be lower than those estimated for individuals living near facilities reporting the release of MIBK to air.
MIAK
MIAK was identified in an automotive coating repair pen at 3% to 7% (SDS 2015c; CPID [modified 2019]). The mean event concentration derived from ConsExpo Web (ConsExpo 2016) for an adult using an automotive coating repair pen containing MIAK ranged from 1.5 mg/m3 to 3.5 mg/m3, while the 6-hour TWA concentrations ranged from 0.125 mg/m3 to 0.29 mg/m3 (see Appendix D for more details). Although dermal exposure could contribute to the overall exposure during use of products available to consumers, the primary route is considered to be inhalation in light of the high volatility of MIAK; therefore, only inhalation estimates are presented.
DAA
On the basis of notifications submitted under the Cosmetic Regulations to Health Canada, DAA is used as a solvent or as an odour masking agent in certain cosmetic products in Canada, such as eyeliner stickers and in various nail care products including base-coats, top-coats, nail polish, nail polish removers, and nail hardeners (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, dated August 2016, April 2017, and November 2019; unreferenced).
Inhalation exposure estimates were derived for certain sentinel products (nail polish and eyeliner stickers), which represent the highest exposures when compared to similar products. Table 7‑9 summarizes the concentration range for the various product scenarios, along with the associated inhalation exposure estimates. Details on the method and parameters used to estimate inhalation exposures to cosmetics are available in Appendix D.
Dermal exposure to DAA from use of these products is also possible and, unlike the other ketones described thus far, is more likely given its lower vapour pressure and high water solubility. A human in vitro dermal absorption study was identified, which found that the amount of DAA that penetrated the skin after 10 minutes, 60 minutes, and 24 hours was 0.04%, 0.15%, and 5.71% of a 25 mg/cm2 dose, respectively (Fasano and McDougal 2008; ECHA c2007-2019b). Table 7‑9 includes the estimated systemic dermal exposures, assuming 6% dermal absorption through the skin.
| Product scenario | Maximum concentrationa | Mean event concentration (mg/m3) | 6-Hr TWAb (mg/m3) | Systemic dermal exposurec (mg/kg -bw/event) |
|---|---|---|---|---|
| Nail polish (12 years and older) | 10% | 3.5 | 0.34 | 0.014 to 0.016 |
| Nail polish (6 months to 4 years) | 10% | 3.1 | 0.30 | 0.023 |
| Eyeliner sticker (12 years and older) | 30% | 0.67 | 1.3 | 0.007 to 0.008 |
Abbreviation: TWA, time-weighted average
a Personal communication, emails from the CHPSD, HC, to the ESRAB, HC, dated August 2016, April 2017, and November 2019; unreferenced.
b 6-hour time-weighted average (TWA) concentrations were derived for all product scenarios to correspond with the exposure durations in the critical effects study used to characterize risk. 6-Hr TWA = Mean event conc. × exposure duration / 6 × 60 min.
c A dermal absorption factor of 6% was incorporated.
DAA was identified in pipe thread sealants, spray products including paints, cleaners, automotive products, and paint thinners (used to dilute lacquers and clean brushes). Table 7‑10 summarizes the inhalation and dermal exposure scenarios for paint and DIY products available to consumers containing DAA, obtained using ConsExpo Web (ConsExpo 2016) (details in Appendix D).
| Product scenario | DAA concentration | Mean event concentration (mg/m3) | Internal inhalation dose on day of exposure (mg/kg bw/day) | Systemic dermal exposurea (mg/kg bw/event) |
|---|---|---|---|---|
| Pipe thread sealant (20 years and older) | 20 to 30%b | 35 to 51 | 1.3 to 2 | 0.014 to 0.02 |
| Automotive choke cleaner (spray) (20 years and older) | 1 to 30%c,d | 13 to 110 | 0.032 to 0.26 | 0.013 to 0.38 |
| Paint/marker remover (spray) (20 years and older) | 7%e | 71 | 0.22 | 0.089 |
| Spray paint (20 years and older) | 1 to 5%f | 21 to 72 | 0.085 to 0.28 | 0.017 to 0.085 |
| Paint thinner (for epoxy paint) (20 years and older) | 5 to 10%g | 55 to 71 | 0.26 to 0.34 | 0.09 to 0.18 |
a A dermal absorption factor of 6% was incorporated.
b SDS 2013
c SDS 2011
d SDS 2012b
e SDS 2018d
f SDS 2012a
g SDS 2017b
DAA was also identified in permanent markers (CPID [modified 2019]; SDS 2008, 2012c, 2014b). These products may be used by young children; therefore, incidental oral and dermal exposures were estimated. A concentration of 100% was assumed on the basis of limited data available on the quantity of DAA in markers. Per event incidental oral exposure estimates from the use of permanent markers were 1.6 mg/kg bw and 3.2 mg/kg bw for children 5 to 11 years of age and 6 months to 4 years of age, respectively. The daily systemic dermal exposure estimates, assuming a dermal absorption of 6%, ranged from 0.002 mg/kg bw/day for adults 20 years of age and older to 0.005 mg/kg bw/day for children 5 to 11 years of age (children 6 months to 4 years of age were assumed not to use permanent markers on a daily basis). Any inhalation exposures to DAA from use of permanent markers are considered to be included in the conservative oral or dermal exposure estimates (details in Appendix D).
7.2.2 Health effects assessment of subgroup 2 (MIBK, MIAK, and DAA)
MIBK and MIAK are structurally similar compounds, differing in chain length by only one methyl group. Biologically, they are likely to have similar interactions and metabolites in the body. DAA has been included in this subgroup as it is a primary metabolite of MIBK. It is likely to have similar metabolic clearance and is possibly responsible for some of the biological effects seen following MIBK exposure. Effects common to all 3 chemicals include decreased body weight, increased kidney and liver weights, and CNS depression. While no data on the metabolism of MIAK was identified, the structural similarities to MIBK suggest it would have analogous metabolites and metabolic rates. A summary of the toxicity data for this subgroup can be found in Table A-2 in Appendix A.
7.2.2.1 MIBK
MIBK has been reviewed by OECD (1996), IPCS (1997), US EPA (2003b), IARC (2013), NICNAS (2017), and ECHA (2019). These reviews provide a basis for the health effects characterization in this assessment. A literature search was conducted from a year prior to the OECD SIDS SIAR (1996) up to January 2019. Based on a two-year inhalation carcinogenicity study by the NTP (2007b) in 2 mammalian species (rat and mouse), IARC classified MIBK as group 2B (“possibly carcinogenic to humans”) with sufficient evidence of carcinogenicity in experimental animals (IARC 2013). MIBK is classified in the European Union (EU) as “suspected of causing cancer” (Carc. 2B) by ECHA (2019). Whereas the IARC (2013) report was the basis for characterization of cancer effects, the US EPA (2003b) was the basis for characterization of non-cancer effects of MIBK.
Toxicokinetics and metabolism
MIBK is rapidly absorbed following oral, inhalation, and dermal exposure (Hjelm et al. 1990; Hjelm et al. 1991; Duguay and Plaa 1995 as cited in US EPA 2003b and NTP 2007b). The major metabolite detected in the plasma of rats administered MIBK by gavage was DAA, with somewhat lesser amounts of 4-methyl-2-pentanol (DiVincenzo et al. 1976; Duguay and Plaa 1995 as cited in NTP 2007b and US EPA 2003b). However, 4-methyl-2-pentanol was the major metabolite (about twice as much as DAA) detected in the lung of rats following inhalation exposure. In humans, inhalation exposure to concentrations of 10 mg/m3, 100 mg/m3, or 200 mg/m3 MIBK for 2 hours resulted in a pulmonary retention of about 60%, with mean blood clearance of 1.6 L/h/kg, and about 0.04% of the total dose was excreted unchanged in the urine within 3 hours post-exposure (Hjelm et al. 1990 as cited in NTP 2007b). With respect to dermal exposure, the percutaneous uptake rate of MIBK in exposed guinea pigs ranged from 0.11 μmol/min/cm to 2.0 μmol/min/cm and averaged 1.1 μmol/min/cm (Hjelm et al. 1991 as cited in US EPA 2003b).
Carcinogenicity and genotoxicity
In a two-year inhalation study in male and female mice and rats, with exposures of 0, 450, 900, or 1,800 ppm (corresponding to approximately 0, 1840, 3,681 and 7,362 mg/m3) for 6 hours per day, 5 days per week, MIBK increased the incidences of hepatocellular adenoma as well as hepatocellular adenoma and carcinoma combined in male and female mice; it also increased incidences of renal tubule adenoma as well as renal tubule adenoma and carcinoma combined in male rats. Additionally, 2 rare renal malignant mesenchymal tumours were observed in high-dose female rats at a concentration of 7,362 mg/m3 (NTP 2007b; IARC 2013). 2 rare renal malignant mesenchymal tumours and an increased incidence of mononuclear cell leukemia were also observed at the highest dose in rats. The incidence of renal effects in rats, including chronic nephropathy, was significantly increased, with an increasing trend in severity. Although the pathological changes in male rats were consistent with the spectrum of α2u-globulin-induced nephropathy, and hyaline droplet formation has been reported in MIBK-exposed male rats in shorter-term studies (for example, Phillips et al. 1987; Nemec et al. 2004 as cited in NTP 2007b), in light of the observation of nephropathy in female rats also, the NTP (2007b) stated that renal tumours may arise independently of a α2u-globulin mechanism. Further, IARC (2013) determined that the relevance of kidney tumours in rats to humans could not be excluded.
Subsequent to the publication of the NTP (2007b) report and the IARC (2013) monograph, recent studies have suggested that the MIBK-induced kidney and liver tumours in rodents were induced by mechanisms such as α2u-globulin nephropathy and constitutive androstane receptor (CAR)-mediated mode of action, respectively, which are not relevant to humans (Borghoff et al. 2015; Hughes et al. 2016). In a review of these data, NICNAS (2017) concluded that, while the evidence supports the conclusion that liver tumours in mice arise from activation of the CAR, a mechanism other than that involving α2u-globulin may be responsible for the renal tumours in rats. This conclusion was also supported in the review by ECHA (2019). This information, along with the observation of mononuclear leukemia in male rats and the renal mesenchymal tumours in female rats, supports the conclusion that tumours in rats are relevant to humans and are sufficient to classify MIBK as a Category 2 carcinogen according to the Globally Harmonized System (GHS) of Classification and Labelling of Chemicals.
On the basis of numerous in vivo and in vitro studies, MIBK is not considered genotoxic (NTP 2007b; IARC 2013; ECHA 2019).
Repeated-dose toxicity
Repeated inhalation and oral exposure to MIBK has been associated with effects on liver and kidney weights, biochemical parameters, or CNS in rats and/or mice at concentrations starting at 410 mg/m3 (MacEwen et al. 1971; Phillips et al 1987; David et al. 1999, all as cited in US EPA 2003b).
In the two-year inhalation study in male and female mice and rats described above, the LOAEC corresponding to chronic non-cancer effects for MIBK was established at the lowest dose tested (1843 mg/m3) on the basis of minimal to mild nephropathy (NTP 2007b).
In an oral sub-chronic toxicity study, male and female rats were administered a dose of 0, 50, 250, or 1,000 mg/kg bw/day MIBK by gavage for 13 weeks (MAI 1986 as cited in US EPA 2003b and IPCS 1990). Alterations in a range of clinical chemistry parameters suggestive of hepatic and renal effects were observed in males and/or females at the highest dose; increased absolute and relative kidney weights were also observed in both sexes at 250 mg/kg bw/day and above. There was an increased incidence of mild nephropathy in males at 1000 mg/kg bw/day. No effects were noted at 50 mg/kg bw/day, which the IPCS (1990) and IARC (2013) considered to be the no observed effect level (NOEL). However, the US EPA (2003b) considered 1000 mg/kg bw/day to be a NOAEL, suggesting that the effects observed were difficult to interpret and may not be biologically adverse in light of the absence of clear histopathological changes. The NOAEL for an oral drinking water study was also considered by the US EPA to be around 1,000 mg/kg bw/day, on the basis of uncertainty regarding biological adversity of the observed renal effects (Carnegie-Mellon Institute of Research 1977a,b as cited in US EPA 2003b).
In light of the uncertainties regarding the adversity of the effects observed in the sub-chronic oral studies, the LOAEC of 1843 mg/m3 from the more recent, comprehensive inhalation study by the NTP (2007b) was extrapolated to an oral dose of 102 mg/kg bw/day for the characterization of risk following longer-term oral exposure; route-to-route extrapolation is considered appropriate for the critical internal effects in the kidneys of male and female rats.
In a dermal study, MIBK was applied to the tails (lower two-thirds) of an unspecified number of male white rats daily (4 hours/day) in doses of 300 or 600 mg/kg for 4 months. These doses induced morphological changes to the skin, brain, liver, adrenal gland, spleen, and testes as well as a reduction in the number of spermatocytes, spermatids, and spermatozoa (Malysheva 1988 as cited in NTP 2007b).
Reproductive and developmental toxicity
In an inhalation reproductive and developmental toxicity study, groups of 35 pregnant F344 rats and 30 CD-1 mice were exposed during GD 6 to 15 to airborne MIBK concentrations of 0, 300, 1,000, or 3,000 ppm (0, 1,229, 4,106, and 12,292 mg/m3) during GD 6 to 15 (Tyl et al. 1987 as cited in US EPA 2003b and IPCS 1990). In rats, signs of maternal toxicity were observed at 12,292 mg/m3, including decreased body weight gain and food consumption (which returned to normal upon cessation of exposure) and increased relative kidney weight. Maternal body weight was not affected in mice, although increased mortality and increased absolute and relative liver weights were observed in dams exposed to 12,292 mg/m3 MIBK. Other maternal effects, including hypoactivity, ataxia, and lacrimation, were observed at 12,292 mg/m3. In rats, fetal body weights were decreased at 1,229 and 12,292 mg/m3, but not at 4,106 mg/m3; the study authors attributed this observation to differential litter size and not exposure. Fetal body weights were also reduced in mice at the highest concentration. At 12,292 mg/m3, delays in some skeletal ossification parameters (considered reversible as the pups grow) were noted in both rats and mice. As mentioned previously with MEK, CNS depression observed in the pregnant dams during exposure may have impacted the observed effects in the offspring. On the basis of this study, the US EPA (2003b) established a NOAEC of 4,106 mg/m3 and LOAEC of 12,292 mg/m3 for maternal effects and delayed fetal skeletal ossification in rats and mice. A NOAECHEC of 1,026 mg/m3 was also derived for continuous exposure (LOAECHEC = 3073 mg/m3).
In a two-generation study, SD rats were exposed to MIBK concentrations of 0, 500, 1,000, and 2,000 ppm (0, 2,050, 4,100, and 8,200 mg/m3, respectively) prior and during mating, through gestation and weaning (Nemec et al. 2004). At 41,00 and 8,200 mg/m3, absent or decreased response to a sound stimulus in the F0 groups was observed. At the highest dose, pups exhibited clinical signs of neuro- or neuromuscular toxicity (rocking, lurching, swaying, prostration, half-closed eyelids). Other effects (mostly observed at 8,200 mg/m3) included increased weights of the liver, seminal vesicles, kidneys, ovaries, and adrenal glands. At 4,100 and 8,200 mg/m3, nephropathy was observed in males. The NOAEL for parental systemic toxicity was considered to be 2,050 mg/m3 (acute sedative effect). The NOAEL for neonatal toxicity (based on clinical signs of neuro- or neuromuscular toxicity) was also considered to be 4,100 mg/m3. The NOAEL for reproductive toxicity was considered to be 8,200 mg/m3 (Nemec et al. 2004 as cited in ECHA 2019).
Short-term and acute toxicity
MIBK has been shown to be of low toxicity following acute oral, dermal, and inhalation exposure (Smyth et al. 1951, 1956; Batyrova 1973; Zakhari et al. 1977; RTECS 1987). Neurological effects (primarily behavioural changes attributed to CNS depression) were observed in several studies in laboratory animals at concentrations as low as 200 mg/m3 (Geller et al. 1979; De Ceaurriz et al. 1981; David et al. 1999; ECHA 2019); however, some neurological effects were generally attenuated with prolonged exposure. Similarly, studies with human volunteers reported reversible irritation and CNS symptoms (headache, nausea, vertigo) at concentrations as low as 200 mg/m3 (Hjelm et al. 1990; Iregren et al. 1993).
7.2.2.2 MIAK
MIAK has not been assessed by other agencies. The following hazard assessment is based on the information identified from an ECHA registration dossier (ECHA c2007-2019c). A literature review was also conducted up to June 2017, with targeted literature searches up to January 2020. Additional information was included in this report where relevant.
Toxicokinetics
It has been reported that clearance of MIAK following oral administration is slower than that following inhalation exposure. Results of an in vitro dermal absorption study (OECD TG 428) indicate that dermal absorption is moderate (ECHA c2007-2019c).
Carcinogenicity and genotoxicity
No chronic studies were identified for MIAK. MIAK was not genotoxic in a variety of in vitro studies (ECHA c2007-2019c).
Repeated-dose toxicity
In a sub-chronic inhalation study, SD rats were exposed to MIAK vapour at concentrations of 0, 200, 1,000, and 2,000 ppm (equivalent to 0, 934, 4,670, and 9,340 mg/m3), for a total of 69 exposures spanning 96 days. There were no significant changes in body weight, hematology, serum clinic chemistry, or gross pathology. Dose-dependent, statistically significant increases of absolute and relative liver weights were seen in males and females at concentrations of 4,670 and 9,340 mg/m3. Absolute and relative kidney weights were also increased in males at both these concentrations, and relative kidney weights were elevated in females at the highest dose. Histopathology revealed hyaline droplet formation in the kidneys of male rats, as well as hepatocytic hypertrophy in livers and tubular epithelium regeneration in kidneys in both males and females exposed to MIAK at the 2 highest concentrations. Overall, the no-observed-effect concentration (NOEC) was determined to be 200 ppm (934 mg/m3) on the basis of organ weight changes and histological changes observed in the livers and kidneys of both males and females at 4,670 mg/m3 or higher (Katz et al. 1986; ECHA c2007-2019c).
With respect to oral exposure, in the sole repeated-dose oral study identified, only a very high dose was tested (2,000 mg/kg bw/day) and associated with a wide range of effects (mainly kidney and liver effects) (ECHA c2007-2019c).
In light of the limitations of the only repeated-dose oral study, an oral NOEL of 52 mg/kg bw/dayFootnote 8 was derived using route-to-route extrapolation from the inhalation study, on the basis of similar effects on the liver and kidney by both routes of exposure.
No dermal repeat-dose toxicity study was reported for MIAK.
Short-term and acute toxicity
Exposure of male and female SD rats to 1,000 or 2,000 ppm (4,670 or 9,340 mg/m3, respectively) MIAK via the inhalation route for a total of 12 exposures over 16 days resulted in a slight, dose-dependent increase in absolute and relative liver weights in rats, with no corresponding effects on serum clinical chemistries or histopathology. At 9,340 mg/m3, renal hyaline droplet formation was noted in males as was mineralization involving the heart in females (Anonymous 1983; Katz et al. 1986 as cited in ECHA c2007-2019c). On the basis of these observations, the authors determined a NOAEC of 1,000 ppm (4,670 mg/m3).
Maternal toxicity was also observed in the developmental study described below at lower doses. Dams exposed to MIAK for 14 days at 3,503 mg/m3 and above showed significant body weight changes and neurological effects (that is, reduced reactivity to stimuli). At the highest dose (7,005 mg/m3), significant decreases in mean body weights, mean body weight gains, and corresponding mean food consumption were also observed. The NOAEC for maternal toxicity was considered to be 3,505 mg/m3, and the NOAEC for neurological effects (CNS depression) was considered by the study authors to be 380 ppm (1,775 mg/m3) (ECHA c2007-2019c).
Exposure of 6 Swiss OF1 mice to concentrations of 1,942, 3,190, 4,871, or 7,075 mg/m3 for 15 minutes resulted in dose-dependent respiratory depression (De Ceaurriz et al. 1984). At the lowest dose tested, a 27% decrease in respiration was observed, which may represent a marker for general CNS depression.
Reproductive and developmental toxicity
In an inhalation reproductive/developmental toxicity screening test in rats, MIAK did not affect a range of parameters investigated in parents or offspring; the substance was therefore not considered to be a reproductive toxicant at up to the highest tested concentration of 5,000 mg/m3 (ECHA c2007-2019c). In an extended one-generation reproductive toxicity study, male and female rats were exposed to MIAK concentrations of 0, 380, 750, or 1,500 ppm (equivalent to 0, 1,775, 3,503, or 7,005 mg/m3) by whole body inhalation exposure for 6 hours/day. No effects on reproductive performance were observed. The only effect noted was lower mean offspring body weights and body weight gains at 7,005 mg/m3. On the basis of this effect, the study authors determined the NOAEC for neonatal toxicity to be 3,503 mg/m3 (ECHA c2007-2019c).
In a prenatal developmental toxicity test on MIAK, groups of bred female SD rats were exposed to MIAK via whole body inhalation at concentrations of 0, 380, 750, or 1,500 ppm (equivalent to 0, 1,775, 3,503, or 7,005 mg/m3) from GD 6 through 19. In the 7,005 mg/m3 group, significant decreases in mean body weights, mean body weight gains, and corresponding mean food consumption were observed in dams throughout the exposure period, while reduced reactivity to a noise stimulus was noted at concentrations of 3,503 or 7,005 mg/m3. The NOAEC for maternal toxicity was considered to be 1,775 mg/m3. No exposure-related external, visceral, skeletal malformations or developmental variations were seen in fetuses at doses of up to 7,005 mg/m3. However, significantly reduced fetal weights were seen in the highest tested group compared with the control group. Therefore, the NOAEC for developmental toxicity was considered to be 3,503 mg/m3 (ECHA c2007-2019c). In an additional developmental study, pregnant rabbits exposed to MIAK showed similar maternal toxicity effects as well as cardiac malformations in 3 pups from separate litters (not considered by the authors to be a secondary non-specific consequence of maternal toxicity). The NOAEC for maternal and developmental toxicity was considered to be 5,837 mg/m3 (ECHA c2007-2019c).
7.2.2.3 DAA
DAA has been reviewed by the OECD (2000). This review provides the basis for the health effects characterization in this assessment. A literature search was conducted from a year prior to the OECD SIDS SIAR (2000) until January 2020.
Toxicokinetics and metabolism
The low molecular weight, log Kow value, and physical state of DAA favour its absorption via various routes of exposure (oral, dermal, and inhalation); the available data suggest that absorption by the oral and inhalation routes is extensive. In an in vitro dermal absorption study, the skin penetration was found to be 0.04%, 0.15%, and 5.71% of the dose (25 mg/cm2) after 10 minutes, 60 minutes, and 24 hours, respectively (Fasano and McDougal 2008). As mentioned above, DAA is a direct metabolite of MIBK.
Carcinogenicity and genotoxicity
No carcinogenicity studies on DAA have been identified. DAA was reported to be non-genotoxic in various in vitro Ames tests, mammalian gene mutation assays, and mammalian chromosome aberration tests (OECD 2000; ECHA c2007-2019b).
Repeated-dose toxicity
In a combined repeat-dose reproductive/developmental toxicity screening test, DAA was administered daily for 45 days by gavage to male and female SD rats at doses of 0, 30, 100, 300, or 1,000 mg/kg bw/day (MHW 1997 as cited in OECD 2000). Decreased locomotor activity and stimulation response were observed at 300 and 1,000 mg/kg bw/day in both sexes, while altered hematological and blood chemistry parameters were noted in males at 1,000 mg/kg bw/day. In males, renal effects were observed, including hyaline droplet formation at 100 mg/kg bw/day or greater, basophilic tubules at 300 mg/kg bw/day or greater, and dilatation of distal tubules at 1000 mg/kg bw/day. At 300 and 1000 mg/kg bw/day, females showed dilatation of the distal tubules and fatty degeneration of the proximal tubular epithelium in the kidneys. The OECD considered the NOAELs for repeat-dose toxicity to be 30 mg/kg bw/day in males and 100 mg/kg bw/day in females. However, given that the hyaline droplet formation is only considered to be specific to male rats (and not biologically relevant to humans), the NOAEL was considered to be 100 mg/kg bw/day.
In an inhalation study, Wistar rats were exposed daily for 6 weeks to concentrations of 0, 233, 1,041, and 4,685 mg/m3 DAA (Butterworth et al. 1980 as cited in ECHA c2007-2019b). The authors identified a NOAEC of 4685 mg/m3 and a NOEC of 1,041 mg/m3. Only liver weight changes, not associated with histological alterations, were observed at 1,041 mg/m3. At 4,685 mg/m3, increased liver and kidney weights, as well as rat-specific eosinophilic hyaline droplets in the proximal tubular cells, were observed in males. OECD considered the middle concentration to be a NOAEC (reported as 1,035 mg/m3) (SHELL Research Ltd. 1979 as cited in OECD 2000).
No repeated-dose dermal studies were available for DAA. Therefore, the oral NOAEL of 100 mg/kg bw/day will be used to inform the risk from dermal exposure.
Reproductive and developmental toxicity
In the combined repeat-dose toxicity study with the reproduction/developmental toxicity screening test described above, there was a decrease in reproductive parameters (fertility index, number of implantations, and implantation index) and developmental parameters (number of pups born, delivery index, live birth index, number of pups alive, and viability index) at the highest dose of 1,000 mg/kg bw/day. Although none of these changes were statistically significant, DAA was considered to potentially cause reproductive/developmental effects at 1,000 mg/kg bw/day. Basing their conclusion on this, the authors considered a NOAEL of 300 mg/kg bw/day for reproductive and developmental effects, with a parental NOAEL of 100 mg/kg bw/day as described above (MHW 1997 as cited in OECD 2000). In another developmental rat study, no effects were observed at up to a dose of 1,000 mg/kg bw/day in rats (ECHA c2007-2019b).
In the absence of inhalation and dermal reproductive/developmental studies on DAA, the oral NOAEL of 300 mg/kg bw/day for reproductive/developmental effects was used for risk characterization.
Effects in humans
Human volunteers (~12, both male and female) exposed within a chamber to 475 mg/m3 of DAA for 15 minutes reported symptoms of headache, nausea, or vomiting (Silverman et al. 1946 as cited in OECD 2000).
7.2.3 Risk characterization of subgroup 2 (MIBK, MIAK, and DAA)
MIBK
MIBK is classified as a Category 2B carcinogen (“possibly carcinogenic to humans”) by IARC (2013) on the basis of increased incidences of tumours in two-year studies in rats and mice. It is also classified as Carc. 2 H351 (“Suspected of causing cancer”) by ECHA (2019). Although some evidence has suggested that these liver and kidney tumours may not be relevant to humans, recent analyses by NICNAS (2017) concluded that, while the evidence supports the conclusion that liver tumours in mice arise from activation of the CAR, a mechanism other than that involving α2u-globulin may be responsible for the renal tumours in rats. This information, along with the observation of mononuclear leukemia in male rats and renal mesenchymal tumours in female rats, supports the conclusion by NICNAS that these tumours in rats are relevant to humans and are sufficient to classify MIBK as a Category 2 carcinogen according to the GHS (NICNAS 2017). This conclusion was also supported in a recent review by ECHA (2019). However, MIBK is not expected to be genotoxic (NTP 2007b; IARC 2013; ECHA 2019).
The chronic inhalation LOAEC for non-cancer effects was established at 1,843 mg/m3 on the basis of minimal to mild nephropathy in the two-year bioassay (NTP 2007b). Renal effects were used as the POD to characterize risk to human health from chronic exposures and are considered protective of potential cancer effects in humans.
The available data from short-term MIBK inhalation studies in animals indicate that developmental effects were the critical health effects. A NOAEC of 4,106 mg/m3 was identified for developmental effects in mice and rats (Tyl et al. 1987 as cited in US EPA 2003b). The corresponding calculated NOAECHEC for developmental effects was 1026 mg/m3 for continuous exposure.
For the oral route, in light of the uncertainties regarding the adversity of the effects observed in the sub-chronic oral studies, the LOAEC from the more recent, comprehensive inhalation study by the NTP (2007b) was converted to a dose of 102 mg/kg bw/day (adjusted for continuous exposure) for the characterization of risk following longer-term oral exposure; route-to-route extrapolation is considered appropriate for the critical effects in the kidneys of male and female rats. In addition, this is considered protective of potential cancer effects at higher doses.
In the only dermal study available, MIBK induced morphological changes to the skin and multiple organs of rats administered doses of 300 mg/kg bw/day and higher for 4 months (Malysheva 1988 as cited in NTP 2007b). Although only limited details were available for this study, the critical effect level of 300 mg/kg bw/day is supported by the effects observed in rats administered 250 mg/kg bw/day orally for 13 weeks.
The predominant sources of exposure to MIBK from environmental media (outdoor air, indoor air, and drinking water) and food for the general population is through air and diet. On the basis of the available data, it is expected that the majority of dietary exposure to MIBK results from its natural occurrence in foods. Intake of MIBK from its natural occurrence in food was not identified as a concern for human health.
MIBK is used in a number of products available to consumers, including rubbing alcohol, dry erase markers, and a range of paint and DIY products. Table 7‑11 provides all of the relevant exposure values and the critical health effects as well as the resultant MOEs for the characterization of risk for MIBK.
| Exposure scenario | Exposure | Critical effect level | Critical health effect endpoint | MOE |
|---|---|---|---|---|
| Environmental mediaa (all age groups) | 2.7 to 8 µg/kg bw/day | LOAEL = 102 mg/kg bw/dayb | Renal effects in rats (hyperplasia, nephropathy, mineralization) in 2-year study (RtR from inhalation study, adjusted for continuous exposure) | 12,750 to 37,778 |
| Environmental media for residents near facilities releasing MIBKc (all age groups) | 6.07 to 18.26 µg/kg bw/day | LOAEL = 102 mg/kg/dayb | Renal effects in rats (hyperplasia, nephropathy, mineralization) in 2-year study (RtR from inhalation study, adjusted for continuous exposure) | 5,586 to 16,804 |
| Food flavouring agent (1 year and older) | 0.03 to 0.12 µg/kg bw/day | LOAEL = 102 mg/kg/day b | Renal effects in rats (hyperplasia, nephropathy, mineralization) in 2-year study (RtR from inhalation study, adjusted for continuous exposure) | ≥ 850,000 |
| Rubbing alcohol (inhalation) (6 months and older) | 0.003 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 1,369,000 |
| Nail products (inhalation) (6 months and older) | 0.04 to 1.2 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 3,422 to 102,650 |
| Temporary tattoos (inhalation) (6 months and older) | 0.0005 to 0.001 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 4,106,000 to 8,212,000 |
| Wood lacquer (inhalation) (20 years and older) | 112 to 433 mg/m3d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 9 to 37 |
| Liquid paint (solvent-rich) for trucks (inhalation) (20 years and older) | 37 to 55 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 75 to 111 |
| Spray products (for example, spray paint) (inhalation) (20 years and older) | 1.5 to 76 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 54 to 2,737 |
| Filler/putty from tube (inhalation) (20 years and older) | 1 to 5 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 821 to 4,106 |
| Dry erase markers (per event) [oral] | 0.16 to 0.48 mg/kg bw (5 to 11 years) | LOEL = 300 mg/kg bw/dayf | Morphological changes in several tissues at lowest dose tested in a 4-month dermal study. | 625 to 1,875 |
| Dry erase markers (per event) [oral] | 0.32 to 0.97 mg/kg bw (6 months to 4 years) | LOEL/NOAEL = 250 mg/kg bw/day g | Hepatic and renal effects at 1 000 mg/kg bw/day in a 13-week oral study. | 258 to 782 |
| Dry erase markers [inhalation] (all age groups) | 0.1 to 0.45 mg/m3 d | NOAEC = 4,106 mg/m3 e | Skeletal variations in mice and rats and reduced fetal body weight and increased fetal death in mice | 9,124 to 41,060 |
Abbreviations: RtR, route to route; LOAEL, lowest-observed-adverse-effect level; NOAEL, no-observed-adverse-effect level; LOEL, lowest-observed-effect level; NOAEC, no-observed-adverse-effect concentration; MOE, margin of exposure
a This includes exposure from outdoor air, indoor air, and drinking water (Health Canada 2024).
b NTP 2007b. Target MOE = 1000 (x10 for interspecies extrapolation; x10 for intraspecies variation; x10 for use of a LOAEL, considering severity of effect).
c The estimated intakes incorporate the highest estimated annual concentration of MIBK in air (see Table 7‑7) associated with facilities that report their substance releases under the NPRI (2021), as well as from other environmental media (Health Canada 2024).
d 6-hour time-weighted average (TWA) concentrations were derived for all product scenarios to match up with the exposure durations of the critical effects study used to characterize risk.
e Tyl et al. 1987 as cited in US EPA 2003b. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation)
f Malysheva 1988 as cited in NTP 2007b
g MAI 1986 as cited in US EPA 2003b and IPCS 1990. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation)
Calculated MOEs for exposure to MIBK in environmental media and food (from potential use as a food flavouring agent) for the general population of Canada as well as for those living near facilities reporting the release of MIBK to air are considered adequate to address any uncertainties in the available health effects and exposure data used to characterize risk. The JECFA (WHO 1999b) concluded that there is “no safety concern with the estimated levels of intake as flavouring substances” for MIBK used as a flavouring agent primarily on the basis of it being “metabolized to innocuous products.”
The MOEs for inhalation exposure to MIBK from the use of some products available to consumers, namely rubbing alcohol, dry erase markers, nail products, and temporary tattoos, are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk. The MOEs for inhalation exposure to MIBK from the use of wood lacquers, liquid paints, and spray products including spray paints are considered inadequate in light of the severity of the observed effects (that is, developmental toxicity) and uncertainty regarding the adversity of effects observed at lower concentrations.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects. In addition, the potential for elevated exposure of people living near facilities that may release MIBK to air was considered in the assessment.
MIAK
No chronic studies were identified for MIAK. MIAK was not genotoxic in a variety of studies.
Sub-chronic exposure to MIAK induced liver and kidney effects (organ weight and histological changes) in rats exposed to concentrations of 4,670 mg/m3 or higher. The NOEC was determined to be 934 mg/m3 (Katz et al. 1986; ECHA c2007-2019c). With respect to oral exposure, in the one repeated-dose oral study identified, a wide range of effects were observed at the only tested dose of 2,000 mg/kg bw/day (ECHA c2007-2019c). Therefore, an oral NOEL of 52 mg/kg bw/day was derived using route-to-route extrapolation from the inhalation study, given the similarity of the effects on liver and kidney by both routes of exposure.
With respect to short-term inhalation exposure to MIAK, the critical effect level was considered to be a NOAEC of 3,503 mg/m3 based on decreased body weight and body weight gains as well as reduced food consumption observed in pregnant rats exposed to 7,005 mg/m3 (ECHA c2007-2019c).
The general population of Canada may be exposed to MIAK from indoor air as well as from its natural presence in certain foods. MIAK is also used in an automotive coating repair pen available to consumers. Table 7‑12 provides all of the relevant exposure estimates and critical effect values, as well as the resulting MOEs.
| Exposure scenario | Exposure | Critical effect levela | Critical health effect endpoint | MOE |
|---|---|---|---|---|
| Environmental mediab | 0.3 to 0.9 µg/kg bw/day | NOEL = 52 mg/kg bw/day | Route-to-route extrapolation from NOEC = 934 mg/m3 | ≥ 57,778 |
| Automotive coating repair pen (20 years and older) | 0.125 to 0.29 mg/m3c | NOAEC = 3 503 mg/m3 | Significant decreases in mean body weights, mean body weight gains, and corresponding mean food consumption in dams at 7,005 mg/m3 | 12,079 to 28,024 |
Abbreviations: NOEL, no-observed-effect level; NOAEC; no-observed-adverse-effect concentration; MOE, margin of exposure; TWA, time-weighted average
a ECHA c2007-2019c. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation) for both critical effect levels
b This includes exposure from indoor and outdoor air. The concentration used to derive intakes for indoor air was also used to derive intakes from outdoor air (Health Canda 2022).
c 6-hour TWA concentrations were derived for the automotive coating repair pen scenario to match up with the exposure durations of the critical effect study used to characterize risk.
Calculated MOEs for exposure to MIAK in environmental media are considered adequate to address any uncertainties in the available health effects and exposure data used to characterize risk. Additional intake of MIAK from its natural occurrence in food was not identified as a concern for human health. The MOEs for inhalation exposure to MIAK in automotive coating repair pens available to consumers is considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects.
DAA
No chronic studies were identified for DAA. An oral NOAEL of 100 mg/kg bw/day for systemic toxicity (kidney changes and altered hematological and blood chemistry in rats) from the reproductive/developmental study is used to address the risk of chronic exposures to DAA (MHW 1997 as cited in OECD 2000; ECHA c2007-2019b). In the absence of dermal studies, route-to-route extrapolation using the oral NOAEL is used to characterize the risk of dermal exposure to DAA.
In the sole inhalation study in animals identified, only liver weight changes not associated with histological alterations were observed at 1,041 mg/m3 (reported as 1,035 mg/m3 by OECD 2000) in a 6-week inhalation study (Butterworth et al. 1980 as cited in ECHA c2007-2019b).
The reproductive and developmental NOAEL was established at 300 mg/kg bw/day on the basis of effects on numerous reproductive and developmental parameters at 1,000 mg/kg bw/day. In order to be protective of the developing fetus and children, a critical effect level of 300 mg/kg bw/day is used to characterize the risk for short-term use of products available to consumers. In the absence of dermal or inhalation reproductive/developmental studies on DAA, the oral NOAEL of 300 mg/kg bw/day was used.
The primary source of exposure to DAA from environmental media and food is from its presence in indoor air and its natural occurrence in various food items.
DAA is used in a range of products available to consumers, including nail care products and permanent markers, pipe thread sealant, floor coatings, thinner, and spray products including paint removers and automotive cleaners.
Table 7‑13 provides all of the relevant exposure estimates and critical effect values, as well as the resulting MOEs. Although limited, available dermal absorption data suggest that DAA is not extensively absorbed through the skin (less than 1% to approximately 6% in humans); therefore, the dermal estimates below incorporate a 6% dermal absorption value. Given that the same oral health effects study is being used to characterize the risks from both potential dermal and inhalation exposures to DAA from the use of products available to consumers, these exposure estimates were combined.
| Exposure scenario | Exposure | Critical effect level | Critical health effect endpoint | MOE |
|---|---|---|---|---|
| Environmental mediaa | 0.48 to 1.44 µg/kg bw /day | NOAEL = 100 mg/kg bw/dayb | Kidney changes and altered hematological and blood chemistry | >69,000 |
| Cosmetics (inhalation)c (all age groups) | 6-hr TWA = 0.3 to 1.3 mg/m3 | NOAEC = 1,035 mg/m3 d | Liver weight changes without histopathological alterations at 4,685 mg/m3 | 796 to 3,450 |
| Cosmetics (dermal)a (all age groups) | 0.007 to 0.023 mg/kg bw/event | NOAEL = 100 mg/kg bw/dayb | Kidney changes and altered hematological and blood chemistry | 4,348 to 14,286 |
| Paint/DIY products (inhalation and dermal) (20 years and older) | 0.013 to 2 mg/kg bw/event | NOAEL = 300 mg/kg bw/daye | Reproductive and developmental effects | 150 to 23,077 |
| Permanent markers (per event) [oral] | 1.6 mg/kg bw/day (5 to 11 years) to 3.2 mg/kg bw/day (6 months to 4 years) | NOAEL = 300 mg/kg bw/daye | Reproductive and developmental effects at 1,000 mg/kg bw/day | 94 to 188 |
| Permanent markers (daily) [dermal] | 0.002 (20 years and older) to 0.005 (5 to 11 years) mg/kg bw/day | NOAEL = 100 mg/kg bw/dayb | Kidney changes and altered hematological and blood chemistry | 20,000 to 50,000 |
Abbreviations: NOAEL, no-observed-adverse-effect level; NOAEC; no-observed-adverse-effect concentration; MOE, margin of exposure; TWA, time-weighted average
a This includes exposure from indoor and outdoor air. The concentration used to derive intakes for indoor air was also used to derive intakes from outdoor air (Health Canda 2023).
b MHW 1997 as cited in OECD 2000; ECHA c2007-2019b. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation)
c Range includes use of nail polishes and eyeliner stickers.
d SHELL Research Ltd. 1979 as cited in OECD 2000. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation)
e MHW 1997 as cited in OECD 2000. Target MOE = 100 (x10 for interspecies extrapolation; x10 for intraspecies variation)
Calculated MOEs for exposure to DAA in environmental media are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk. Additional intake of DAA from its natural occurrence in food was not identified as a concern for human health. In addition, the calculated MOEs from the use of products available to consumers (including the assumption of 100% DAA in permanent markers and the non-statistically significant changes in reproductive and developmental parameters used to characterize the risk associated with the use of markers and paint/DIY products) are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were routinely estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects.
7.2.4 Uncertainties in the evaluation of risk to human health for MIBK, MIAK and DAA
The key sources of uncertainty are presented in Table 7‑14.
| Key sources of uncertainty | Impact |
|---|---|
| Assumption that dermal exposures are minimal for MIBK and MIAK in comparison to inhalation exposures, given the high volatility of the substances; however, confidence is high that exposure is overestimated, owing to the conservative nature of the exposure models. | + |
| Assumption that DAA is present in permanent markers at a concentration of 100%. | + |
| Lack of chronic studies for MIAK or DAA. | +/- |
| Use of route-to-route extrapolation for long-term exposure to MIBK. | - |
| Use of route-to-route extrapolation of a 96-day inhalation study for both oral and dermal exposure scenarios for MIAK. | +/- |
| No short-term inhalation study was available for DAA. Route-to-route extrapolation from oral study was applied. | +/- |
| In terms of the risk posed by the use of products available to consumers for DAA, confidence in the adequacy of the margins is high, owing to the conservativeness associated with the dermal absorption value (based on the amount of DAA that penetrated the skin after a 24-hour exposure) and the non-statistically significant effects on reproductive and developmental parameters at the critical effect level. | + |
+ = uncertainty with potential to cause overestimation of exposure/risk; - = uncertainty with potential to cause underestimation of exposure risk; +/- = unknown potential to cause over- or underestimation of risk.
7.3 Assessment of subgroup 3 (diacetyl, 2,3-PD, and acetoin)
7.3.1 Exposure assessment of subgroup 3 (diacetyl, 2,3-PD, and acetoin)
Environmental media
Limited to no empirical data were identified for diacetyl, 2,3-PD, or acetoin in air (2,3-PD only), water, and soil in Canada.
Diacetyl has been identified as a by-product of ozone disinfection (US) and was detected in both drinking water (US) and surface water (France) (HSDB 1983-). No data were identified regarding the presence of diacetyl in soil or sediment; however, based on its low log Koc, diacetyl is not expected to be present in this media.
Only one ambient air study that detected 2,3-PD in air in Roseville, California, was identified; the study recorded concentrations of 11.1 and 8.1 ng/cm3 2,3-PD in the summer and winter, respectively (HSDB 1983-). In a Nunavik field study that measured 54 indoor air samples, 2,3-PD was detected in 4% of the samples, with a maximum concentration of 2.8 µg/m3 (Won et al. 2019). No other environmental concentration data were identified.
Given its physical and chemical properties, acetoin may be present in air and water but is not likely to be present in soil and sediment. Given its moderate Henry’s law constant, acetoin is expected to volatilize from water. The half-life in air is approximately 37 hours (HSDB 1983-).
ChemCAN v6.00 (ChemCAN 2003) was used to derive air, water, and soil concentrations of diacetyl, 2,3-PD (water and soil only), and acetoin for Canada using the upper-end quantity data from Table 4‑1 and assuming that the total import quantity was released into a single region of Canada (that is, the Ontario Mixed-Wood Plain region). These concentrations were then used to estimate exposure to diacetyl, 2,3-PD (water and soil), and acetoin from environmental media for the general population of Canada. All resulting exposures were less than 2.5 ng/kg bw/day. On the basis of the information presented above, exposure to these substances from environmental media is negligible.
Food
Diacetyl, 2,3-PD, and acetoin are naturally present in various food items and may also be used as food flavouring agents (Nijssen et al. 1963-2018; Burdock 2010). Diacetyl is being replaced in some products by 2,3-PD, acetoin, and other diketones with a similar “butter” aroma and taste (Doepker et al. 2012; Gaffney et al. 2015; CDC 2016).
The JECFA evaluated a group of aliphatic acyclic and alicyclic alpha-diketones and related alpha-hydroxyketones, including diacetyl, 2,3-PD, and acetoin (WHO 1999a). As part of that evaluation, the Committee estimated the per capita intake for diacetyl, 2,3-PD, and acetoin from their use as food flavouring agents. For the US population, per capita intake estimates were 133 µg/kg bw/day, 1 µg/kg bw/day, and 30 µg/kg bw/day, respectively. For the European population, per capita intake estimates were 55 µg/kg bw/day, 4 µg/kg bw/day, and 47 µg/kg bw/day, respectively (WHO 1999a) (see Appendix C for more details). In the absence of data on actual use in Canada, the per capita intake estimates are acceptable estimates of possible dietary exposure to diacetyl, 2,3-PD, and acetoin from their use as food flavouring agents for the general population (1 year of age and older).
Quantitative exposure estimates for diacetyl, 2,3-PD, and acetoin from their natural occurrence in foodFootnote 5 ranged from 281 µg/kg bw/day for 14- to 18-year-olds to 1625 µg/kg bw/day for 1-year-olds for diacetyl, from 6.5 µg/kg bw/day for 6- to-12-month-olds to 208 µg/kg bw/day for adults 19 years and older for 2,3-PD, and from 161 µg/kg bw/day for 14- to 18-year-olds to 369 µg/kg bw/day for 2- to 3-year-olds for acetoin (see Appendix C for details).
The estimates of daily intake for diacetyl from its natural occurrence in food are based primarily on the level in pasteurized milk identified in the literature (approximately 30 µg/g) (De Leonardis et al. 2013). Higher levels of diacetyl have been reported in raw milk and ultra-high temperature (UHT) milk (Macciola et al. 2008; De Leonardis et al. 2013). However, other studies reported much lower (by several orders of magnitude) diacetyl concentrations in milk ranging from 0.0002 µg/g to 0.024 µg/g (Nijssen et al. 1963-2018; Imhof et al. 1995; Shimoda et al. 2000; Valero et al. 2001; Toso et al. 2002). Assuming that levels of diacetyl in milk in Canada are similar to those in Europe, the highest value in pasteurized milk (semi-skim) from the De Leonardis et al. (2013) study was selected. The authors explain that the analytical method used in this study extracts the diacetyl that is linked primarily to the proteins and lactose found in the milk. The other aforementioned studies used headspace techniques, which only capture the volatile portion of the substance in the food (De Leonardis et al. 2013).
In addition to oral exposures to these substances, inhalation exposures may also be significant for certain food items such as microwaved popcorn. Diacetyl, acetoin, and 2,3-PD were measured in popcorn (Buttery et al. 1997; Rosati et al. 2007). According to Rosati et al. (2007), diacetyl and acetoin were measured in a chamber air study, with concentrations in the chamber air ranging from 0.02 mg/m3 to 5.8 mg/m3 and from 0.01 mg/m3 to 4.2 mg/m3 for diacetyl and acetoin, respectively. The average amount of diacetyl emitted from a bag of microwaved popcorn was 778.9 µg/bag (Rosati et al. 2007). Using information from Rosati et al. (2007) and an approach outlined in Zhu et al. (2001), this converts to approximately 0.03 mg/m3 of diacetyl in a standard room 1 hour after the popcorn is popped (maximum of 0.04 mg/m3 during the first hour after popping) (see Appendix E). There was insufficient information to quantify the air concentration of 2,3-PD from popcorn. Diacetyl and 2,3-PD have also been measured in the air of coffee roasting plants (McCoy et al. 2017), and diacetyl was measured in air in a study designed to simulate exposures that could occur in a small coffee shop (Pierce et al. 2015).
Products available to consumers
According to notifications submitted to Health Canada under the Cosmetic Regulations, diacetyl is used in body soaps and face moisturizers in Canada (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, dated November 2019 and April 2020; unreferenced). In addition, several cosmetics listing diacetyl as an ingredient were identified on retailer websites, including body moisturizers, body oils, body washes, shampoos, conditioners, hand and nail creams, and body soaps. Body moisturizers and face moisturizers are presented as sentinel scenarios. The function of diacetyl in cosmetics is as a perfuming agent (European Commission 2017).
2,3-PD was identified in certain air fresheners for home care as well as in fragrant oils, which can be used as air fresheners (SDS 2015d,e, 2019f; CPID [modified 2019]). Table 7‑15 summarizes the estimated inhalation exposures for products available to consumers that contain diacetyl or 2,3-PD. Although dermal exposure could contribute to the overall exposure during use of products available to consumers (European Commission 2014), the primary route of concern on the basis of the critical effects is considered to be inhalation; therefore, only inhalation estimates are presented.
Acetoin was not identified in any other products available to consumers in Canada.
| Product scenario | Concentration | Mean event concentration (mg/m3) | 6-hr TWAa (mg/m3) |
|---|---|---|---|
| Face moisturizerb (20 years and older) (diacetyl) | 0.1% | 0.0099 | 0.0198 |
| Face moisturizerb (5 to 11 years old) (diacetyl) | 0.1% | 0.0035 | 0.014 |
| Body moisturizer (12 years and older) (diacetyl)c | 0.1% | 0.72 | 0. 0.04 |
| Air freshenerd (2,3-PD) | 0.1 to 5% | 0.0031 to 0.064 | 0.004 to 0.14 |
Abbreviation: TWA, time-weighted average
a 6-hour time-weighted average (TWA) concentrations were derived for all product scenarios to match up with the exposure durations of the critical effects study used to characterize risk. 6-Hr TWA = Mean event conc. × exposure duration / 6 × 60 min.
b Personal communication, email from the CHPSD, HC, to the ESRAB, HC, April 2020; unreferenced.
c Given that no concentration data were available for body moisturizers, it is presumed that diacetyl is present at the same levels as in face moisturizers.
d Includes use of essential oils as air fresheners, as well as plug-in and gel type air fresheners.
7.3.2 Health effects assessment of subgroup 3 (diacetyl, 2,3-PD, and acetoin)
Diacetyl and 2,3-PD are alpha-diketones, and their structures are identical except for an additional methyl group on 2,3-PD. Their similarity in structure and functional groups is reflected in their comparable physicochemical and organoleptic properties.
It is expected that 2,3-PD is readily bioavailable via the oral, dermal, and inhalation routes, similar to diacetyl, and the impact of the additional methyl group in 2,3-PD is expected to be minimal. Both chemicals exhibit portal-of-entry effects on the respiratory tract following inhalation exposure. Following oral exposure, diacetyl is rapidly metabolized in liver to acetoin and 2,3-butanediol. For 2,3-PD, the OECD QSAR Toolbox (2017) in vivo and in vitro rat liver metabolism simulator predicted metabolites 2-hydroxy-3-pentanone, 3-hydroxy-2-pentanone, and 2,3-pentanediol (among others) that were similar in structure to metabolites of diacetyl. On the basis of this information, diacetyl, acetoin, and 2,3-PD were treated as a group, and toxicity data for these substances were used to read-across within the group (see Appendix A).
7.3.2.1 Diacetyl
Diacetyl has been reviewed by the IPCS (1999), EFSA (2004), SCOEL (2014), and CDC (2016). These reviews provide a basis for the health effect characterization in this assessment. A literature search was conducted from a year prior to the CDC report (2016) up to January 2020, and significant new information was included to support risk characterization.
Toxicokinetics
Diacetyl can be generated endogenously and is a metabolite of acetaldehyde in mammals (IPCS 1999; SCOEL 2014). It is anticipated that methyl ketones are principally metabolized by oxidation of the terminal methyl group at low concentrations. At higher levels, diacetyl is reduced to acetoin and 2,3-butanediol, and then conjugated with glucuronic acid and excreted. The regional dosimetry patterns from inhalation in humans differ greatly from those in rats, with much greater penetration of diacetyl to the human bronchiolar airways compared to rats. As a result, diacetyl toxicity is observed primarily in the upper respiratory tract of rodents and the lower respiratory tract of humans (Cichocki and Morris 2017).
To address this, a computational fluid dynamics-physiologically based pharmacokinetic (PBPK) model was developed to compare diacetyl absorption and tissue concentrations in the rat and human respiratory tracts (Gloede et al. 2011). The model estimated that the bronchiolar tissue concentrations of diacetyl in humans during light exercise exceeded those in rats by 20- to 40-fold. Further dosimetry modelling indicated that rat inhalation toxicity studies under-predict the risk of bronchiolar injury in humans (Cichocki and Morris 2017).
Carcinogenicity and genotoxicity
Diacetyl has not been classified by any agency on the basis of carcinogenicity or other health effects. However, recent NTP studies (NTP 2018) demonstrated some evidence of carcinogenic activity in two-year inhalation studies. Groups of Wistar Han rats and B6C3F1/N mice were exposed to diacetyl vapour by whole body inhalation at concentrations of 0, 12.5, 25, or 50 ppm (0, 45, 90, and 179 mg/m3, respectively) for 6 hours per day, 5 days per week for 105 weeks. In rats, increases were observed in the incidences of squamous cell carcinomas of the nasal cavity in males and females and in the combined incidence of squamous cell carcinomas and papilloma in the nasal cavity of males at 179 mg/m3. In mice, the incidence of adenocarcinoma of the nose was increased in females at this concentration. The NTP concluded that “there was some evidence of carcinogenic activity of diacetyl in male and female rats. There was no evidence of carcinogenic activity of diacetyl in male B6C3F1/N mice exposed to 12.5, 25, or 50 ppm. There was equivocal evidence of carcinogenic activity of diacetyl in female B6C3F1/N mice based on the occurrences of adenocarcinoma of the nose” (NTP 2018).
Available information indicated that diacetyl is mutagenic in a variety of in vitro tests (Bjeldanes and Chew 1979; Marnett et al. 1985; Shane et al. 1988; Kato et al. 1989; Dorado et al. 1992; Aeschbacher et al. 1989 reviewed in EFSA 2004 and NTP 2018; More et al. 2012).
In in vivo studies, increased unscheduled DNA synthesis (UDS) was seen in the stomach cells of rats exposed to diacetyl at a dose of 500 mg/kg bw/day by gavage (Furihata et al. 1985; Furihata and Matsushima 1987 reviewed in SCOEL 2014). However, diacetyl did not induce micronuclei in mice or rats exposed to up to 500 mg/kg bw/day by intraperitoneal injection for 3 days or to up to 100 ppm via inhalational exposure to diacetyl for 3 months, suggesting that diacetyl had no effect on erythropoiesis (NTP 2018).
Repeated-dose toxicity
In the NTP (2018) two-year inhalation studies referred to above, exposure to diacetyl also resulted in increased incidences of non-cancer effects in the nose, larynx, trachea, and lung of mice at all concentrations tested and in rats at 90 mg/m3 and 179 mg/m3, with more severe effects on the respiratory system occurring at higher concentrations in mice and rats. In rats, the nasal lesions for which incidences increased significantly included suppurative inflammation, respiratory epithelium hyperplasia and squamous metaplasia, olfactory epithelium atrophy, respiratory metaplasia, and necrosis (males), turbinate hyperostosis, and fibrosis of the lamina propria. Significantly increased incidences of chronic active or suppurative inflammation and epithelium hyperplasia were also observed in the larynx, trachea, lung, and eye at 179 mg/m3. At 90 mg/m3, significantly increased incidences were observed for squamous epithelium hyperplasia in larynx (male and female), epithelium regeneration in trachea (male), and epithelium hyperplasia in lung (female). In mice, similar effects were observed at 45 mg/m3 and included metaplasia of the respiratory epithelium, atrophy of olfactory epithelium and turbinate atrophy in the nose, and squamous epithelium hyperplasia in the larynx. Survival rate was moderately decreased in females at 90 mg/m3 and significantly decreased in males at 179 mg/m3. At the end of the study, mean body weights were decreased for both males and females at 179 mg/m3 compared with controls. Thus, on the basis of these observations of non-cancer effects observed in mice, a chronic inhalation LOAEC value of 45 mg/m3 is considered in this health effects assessment (NTP 2018).
A profile of effects similar to that seen in the chronic bioassay was also observed in sub-chronic inhalation studies in Wistar Han rats and B6C3F1/N mice (NTP 2018). Doses were 0, 6.25, 12.5, 25, 50, or 100 ppm (corresponding to 0, 22, 45, 90, 179, or 350 mg/m3) by whole body exposure, 6 hours per day, 5 days per week for 14 weeks. A NOAEC of 45 mg/m3 was derived based on a significant increase in the incidence of non-neoplastic lesions in the respiratory tract (nose, larynx, trachea, and lung) of mice and rats, primarily in the 179 mg/m3 and 350 mg/m3 groups after 14 weeks. At 90 mg/m3, increased incidences of squamous metaplasia of the respiratory epithelium were observed in male and female rats, along with increased degeneration of the olfactory epithelium in female rats. In mice, increased incidences of necrosis in respiratory epithelium were observed in males and females at 90 mg/m3; increased hyperplasia and chronic active inflammation were also observed in the larynx in females (NTP 2018). Similar effects were reported in an earlier study in mice exposed to diacetyl for 6 or 12 weeks at 90 mg/m3or more; the lowest concentration tested (25 ppm or 90 mg/m3) was considered to be the LOAEC on the basis of significant epithelial injury and peribronchial lymphocytic inflammation (Morgan et al. 2008).
In an oral repeat-dose toxicity study, male and female CFE (Crl:OFA(SD)) rats were administered 0, 10, 30, 90, or 540 mg/kg bw/day of diacetyl in water by oral intubation for 90 days. At the highest dose of 540 mg/kg bw/day, rats showed decreased body weight gain and increased water consumption. Effects observed included anemia, increased leucocyte counts, and an increase in relative weights of brain, liver, kidney, adrenals, and pituitary glands (organ weight increases were greater than the changes likely to be associated with decreased body weight). Ulcers were observed in both squamous and glandular parts of the stomach mucosa. No adverse effects were noted in lower dose groups. However, slight, but not statistically significant, anemia and increased relative weights of some organs were noted at lower doses in the absence of histopathological changes in the stomach. The authors concluded that the NOAEL of diacetyl in this study was 90 mg/kg bw/day, although it is not clear whether the effects observed at 540 mg/kg bw/day may have been secondary to the ulcer formation (Colley et al. 1969 reviewed in IPCS 1999; SCOEL 2014).
The results of another repeated-dose oral study suggest that diacetyl may induce neurological and reproductive effects (Bawazir 2016). Daily administration of 25 mg/kg bw/day diacetyl to rats via oral tube during a 4-week study resulted in changes in the levels of several neurotransmitters in different areas of the brain as well as a decrease in serum testosterone levels. Histological changes in the testes, which were associated with a significant decrease in mature sperm and tubular deficits, were observed. However, the published account of this study was very limited as only one dose was tested and the study lacked a proper control.
Short-term and acute toxicity
Acute and short-term exposures to diacetyl vapour caused a wide range of nasal and olfactory lesions in rodents (Hubbs et al. 2002; Morgan et al. 2008; Larsen et al. 2009; Morris and Hubbs 2009), affected sensory neurons (Goravanahally et al. 2014), and led to airway hyporeactivity (Zaccone et al. 2013).
Hubbs et al. (2008) exposed male Hla:(SD)CVF rats (6/exposure group) to diacetyl at concentrations over a 6-hour period through continuous exposure at time-weighted average (TWA) concentrations of 0, 99.3, 198.4, or 294.6 ppm (0, 355, 710, or 1,055 mg/m3, respectively); through 4 15-minute pulse exposures over a 6-hour period at TWA concentrations of 0, 122, 225, or 365 ppm (0, 437, 806, or 1,307 mg/m3, respectively); or through a single 15-minute pulse exposure to a TWA concentration of 92.9 ppm (333 mg/m3). Both pulsed and continuous exposure patterns caused epithelial injuries, including epithelial necrosis and suppurative/fibrinosuppurative inflammation in the nose, larynx, trachea, and bronchi. The most severe effect was observed in the nose. The larynx and trachea were affected at diacetyl concentrations of 225 ppm or higher, and bronchi were affected at concentrations of 294.6 ppm or higher. In rats exposed to the single 15-minute pulse exposure (TWA of 92.9 ppm), significant histological changes (that is, necrosis) occurred in the nose. The authors concluded that the NOAEC for acute inhaled diacetyl was less than 93 ppm (333 mg/m3), which is considered here to be the LOAEC.
Reproductive and developmental toxicity
In groups of CD‐1 mice and albino Wistar rats administered diacetyl by gavage on days 6 to 15 of gestation at doses of up to 1,600 mg/kg bw/day, no effects were seen on maternal survival, weight, and reproductive parameters or on fetal survival or microscopic appearance of external, skeletal, or soft tissues. Similarly, no maternal or developmental effects were observed in hamsters administered doses of up to 1,600 mg/kg bw/day on gestation days 6 to 10 (US FDA 1973 reviewed in WHO 1999a; EFSA 2004).
Effects in humans
In several epidemiological studies, inhalation of vapours containing diacetyl and other substances by workers at microwave popcorn or flavouring manufacturing facilities has been consistently associated with the development of diverse respiratory impairment conditions such as obliterative bronchiolitis (Kreiss et al. 2002; Parmet and Von Essen 2002; van Rooy et al. 2007; CDC 2016). Bronchiolitis obliterans (BO) is a rare, irreversible lung disease characterized by chronic scarring and severe narrowing of the bronchial airways (Hubbs et al. 2008). During the formation and development of BO, a number of other pathological conditions have been observed, including symptoms of allergic pulmonary inflammation, asthma, diffuse interstitial fibrosis, and granuloma, chronic obstructive lung disease with and without emphysema, exogenous allergic alveolitis, and pneumonia (Hubbs et al. 2008; European Commission 2014). Several case reports, case series, and cross-sectional studies have been reviewed by the EU (SCOEL 2014) and CDC (2016). The term “popcorn lung” is often used to describe the respiratory symptoms observed in these employees. Similar effects have been reported in case studies of repeated consumer exposure to microwave popcorn (that is, daily consumption) over 10 years (Egilman and Schilling 2012).
7.3.2.2 2,3-PD
2,3-PD has been reviewed by IPCS (1999), EFSA (2004), and CDC (2016). These reviews provide a basis for the health effect characterization in this assessment. A literature search was conducted from a year prior to the CDC (2016) up to January 2020, and significant new information was included to support risk characterization.
Toxicokinetics
Zaccone et al. (2015) showed that 2,3-PD could be metabolized to 2,3-hydroxy-3-pentanone by human airway epithelium.
Carcinogenicity and genotoxicity
No carcinogenicity data were identified for 2,3-PD. 2 studies showed that 2,3-PD was not mutagenic in Salmonella (Kim et al. 1987; Aeschbacher et al. 1989 reviewed in WHO 1999a and EFSA 2004, 2016). 2,3-PD did not cause formation of micronuclei in either rats or mice of both sexes (NTP 2017).
Repeated-dose toxicity
In a repeated-dose inhalation study, male and female Wistar Han rats and B6C3F1 mice were exposed to 2,3-PD by inhalation at concentrations of 0, 6.25, 12.5, 25, 50, or 100 ppm (0, 26, 51, 102, 205, or 409 mg/m3, respectively) 6 hours/day, 5 days/week for 14 weeks. Results similar to those reported in the sub-chronic study with diacetyl were observed. At 205 and 409 mg/m3, clinical observations were noted, including abnormal breathing, sneezing, and eye abnormality. Significantly increased incidences of non-neoplastic lesions occurred in the respiratory tract of male and female rats, including epithelium hyperplasia in the nose and metaplasia in the larynx. At 102 mg/m3 and above, incidences of respiratory epithelium hyperplasia in the nose were significantly increased in males, while incidences of respiratory epithelium metaplasia in the larynx were significantly increased in females. In mice, significantly decreased body weight gain and changes in internal organ weights of both sexes (in the absence of histopathological alterations) were observed at 205 mg/m3 and higher, along with increased incidences of non-neoplastic lesions of the respiratory tract. At 102 mg/m3, incidences of respiratory epithelium metaplasia and regeneration were significantly increased in male mice. The NTP considered the NOAEC for respiratory tract effects in both rats and mice to be 51 mg/m3 and the NOAEC for non-respiratory tract effects in mice to be 102 mg/m3 (NTP 2017).
No oral repeat-dose toxicity study was reported for 2,3-PD. Therefore, route-to-route extrapolation of the NOAEC for non-respiratory tract effects was carried out from the sub-chronic inhalation study on 2,3-PD to obtain a NOAEL of 24 mg/kg bw/day (see Appendix F). The NOAEL from the oral sub-chronic study with diacetyl was also used as a read-across to estimate a NOAEL of 105 mg/kg bw/day for 2,3-PD to be used as support for hazard characterization (see Appendix F).
Acute and short-term repeated-dose toxicity
Acute and short-term exposure to 2,3-PD for up to 2 weeks induced a range of histopathological lesions resembling bronchiolitis obliterans-like lesions similar to those occurring in humans (including necrotizing rhinitis, tracheitis, mucosal inflammation, squamous metaplasia and regenerative changes, epithelial atrophy, and exudate and lymphoid deposits) in the nose, larynx, trachea, and/or lungs of mice and rats at all concentrations tested (Morgan et al. 2012; Hubbs et al. 2012; Zaccone et al. 2013; NTP 2018).
Effects in humans
Similar to diacetyl, 2,3-PD is an additional α-dicarbonyl butter flavouring used in the microwave popcorn industry and other food manufacturing settings and has been implicated as a potential cause of occupational lung disease (Hubbs et al. 2019).
7.3.2.3 Acetoin
Acetoin has been reviewed by IPCS (1999) and EFSA (2004, 2016). These reviews provide a basis for the health effect characterization in this assessment. A literature search was conducted from a year prior to EFSA (2016) up to January 2020, and some information was identified in an NTP technical report on the carcinogenicity of diacetyl (NTP 2018).
Toxicokinetics
Acetoin is metabolized primarily via oxidation at low concentration in vivo and by reduction to diacetyl at high concentration (EFSA 2004; NTP 2007a).
Carcinogenicity and genotoxicity
In a screening test for pulmonary carcinogenicity of food additives, in which a limited number of tissues were evaluated, no increase in lung tumours was observed in strain A mice administered acetoin by intraperitoneal injection 3 times per week for 7 weeks (Stoner et al. 1973).
Several studies indicated that acetoin was not mutagenic in Salmonella or Escherichia coli (WHO 1999a; EFSA 2004; NTP 2017).
Repeated-dose toxicity
In an oral study, male and female CFE rats (15/group) were administered acetoin in drinking water at concentrations of 0, 750, 3,000, or 12,000 ppm (equivalent to 0, 85, 330, or 1,300 mg/kg bw/day) for 13 weeks (Gaunt et al. 1972). At concentrations of 85 and 330 mg/kg bw/day, no significant effects were observed with respect to body weight gain, hematological findings, serum chemistry, renal cell excretion, urinary concentration tests, organ weights, or histopathology. At the highest concentration, body weights were significantly decreased in males from week 5 onwards, and relative liver weight was significantly increased at weeks 2, 6, and 13; females showed the same effects after 13 weeks. These effects were not accompanied by any histopathological changes and were probably due to metabolic load from acetoin intake. Small but statistically significant decreases in hemoglobin concentration and erythrocyte counts were also observed at the highest dose in both sexes. The authors concluded that the NOAEL was 3,000 ppm in the drinking water (equivalent to 330 mg/kg bw/day) based on the effects of decreased body weight gain and hematological effects (Gaunt et al. 1972 reviewed in IPCS 1999).
Male and female Wistar Han rats and B6C3F1/N mice were exposed to acetoin by whole body inhalation at concentrations of up to 800 ppm (2,883 mg/m3) for 6 hours per day, 5 days/week for 14 weeks. No exposure-related effects on survival, body weights, organ weights, clinical pathology, or histopathology were observed in either species following exposure to acetoin concentrations up to the highest concentration tested (that is, 2,883 mg/m3) (NTP 2017).
7.3.3 Risk characterization of subgroup 3 (diacetyl, 2,3-PD, and acetoin)
Diacetyl
Although diacetyl has not been classified by any agency on the basis of carcinogenicity or other health effects, recent NTP studies (2018) demonstrated some evidence of carcinogenic activity in two-year inhalation studies in mice and rats, with significant increases in incidences of nasal tumours at 179 mg/m3. Evidence also indicates that diacetyl may be genotoxic. More recent information suggests that toxicity is largely due to diacetyl existing as a reactive short-chain α-dicarbonyl compound, which elicits cytotoxicity through modification of essential proteins, interactions with DNA, and cell injury by reactive oxygen species (Hubbs et al. 2019).
Chronic exposure to diacetyl also resulted in increased incidences of non-cancer effects in the nose, larynx, trachea, and lung of mice at all concentrations tested (LOAEC of 45 mg/m3), with more severe effects on the respiratory system occurring at higher concentrations in mice and rats. A similar spectrum of respiratory tract effects was also observed in sub-chronic, short-term, and acute studies in rodents. Both pulsed and continuous exposure to diacetyl for as short a duration as 15 minutes caused epithelial injuries, with the NOAEC for acute inhaled diacetyl considered to be less than 333 mg/m3 (Hubbs et al. 2008).
The data set available in regards to the toxicity of oral exposure to diacetyl is more limited. In a sub-chronic study in rats, effects on hematological parameters and several organ weights were noted at 540 mg/kg bw/day (NOAEL of 90 mg/kg bw/day), which may have been secondary to the observed stomach ulceration (Colley et al. 1969 reviewed in IPCS 1999; EFSA 2004; SCOEL 2014). In light of the limitations of the other oral studies identified (Bawazir 2016; US FDA 1973 reviewed in IPCS 1999; EFSA 2004) and the systemic oral NOAEL of 330 mg/kg bw/day reported for acetoin (a metabolite of diacetyl that has been demonstrated to be less potent than diacetyl in studies involving similar protocols) in a drinking water study (Gaunt et al. 1972), it is suggested that the true NOAEL for diacetyl could potentially be between 90 mg/kg bw/day and 330 mg/kg bw/day.
Several lines of evidence demonstrate that the effects observed in inhalation studies in laboratory animals are relevant to humans and support the conclusion of the epidemiologically based quantitative risk assessment for diacetyl (CDC 2016) on the basis of reports on the development of diverse respiratory impairment in workers at a microwave popcorn or flavouring manufacturing plants (Kreiss et al. 2002; Parmet and Von Essen 2002; van Rooy et al. 2007). However, dosimetry modelling estimates suggest that tissue concentrations of diacetyl in the respiratory tract of humans might be 20- to 40-fold greater than those in exposed rodents (Gloede et al. 2011) and that bioassay data in rodents might underestimate the risk of bronchiolar injury in humans (Cichocki and Morris 2017).
The predominant source of exposure to diacetyl for the general population is through the diet. The estimated oral intake from the use of diacetyl as a possible flavouring agent in foods for the general population 1 year of age and older (55 µg/kg bw/day to 133 µg/kg bw/day) is less than intakes that have been estimated from natural occurrence (281 µg/kg bw/day to 1,625 µg/kg bw/day) for this substance. Considering that the true NOAEL for diacetyl is likely to be between 90 mg/kg bw/day and 330 mg/kg bw/day (sub-chronic oral toxicity study in rats) and that effects in rats are likely linked to ulcer formations in the stomach, the intake of diacetyl from its natural occurrence in food was not identified as a concern for human health (MOEs fall between ≥55 and ≤203). MOEs for diacetyl from its use as a possible flavouring agent in food are ≥677 and are considered adequate to account for uncertainties in the available health effects and exposure data used to characterize risk. In addition, the JECFA (WHO 1999a) concluded that there is “no safety concern at current levels of intake when used as a flavouring agent; secondary components do not raise a safety concern” on the basis of the dietary exposure estimates for substances in this flavouring grouping.
Table 7‑16 provides all of the relevant exposure values and the critical health effects for diacetyl as well as the resulting MOEs for the characterization of risk.
| Exposure scenario | Exposure | Critical effect level | Critical health effect endpoint | MOE |
|---|---|---|---|---|
| Microwave popcorn (inhalation) | Concentration after 15 min. = 0.04 mg/m3 | LOAEC = 333 mg/m3 a | Nasal and laryngeal effects (necrosis/inflammation in epithelium) | 8,325 |
| Face moisturizer (inhalation) | 6-hr TWA = 0.0198 (20 years and older) to 0.014 (5 to 11 years) mg/m3 | LOAEC = 45 mg/m3 b | Nasal effects (exudate and atrophy of the olfactory epithelium) | 3,214 to 2,273 |
| Body moisturizer (inhalation) | 6-hr TWA = 0.04 mg/m3 (12 years and older) | LOAEC = 45 mg/m3 b | Nasal effects (exudate and atrophy of the olfactory epithelium) | 1,125 |
Abbreviations: LOAEC, lowest observed-adverse-effect concentration; MOE, margin of exposure; TWA, time-weighted average
a Hubbs et al. 2008. Target MOE = 1000 (x10 for interspecies extrapolation; x10 for intraspecies variation; x10 for use of a LOAEC, considering severity of effect).
b NTP 2018. Target MOE = 1000 (x10 for interspecies extrapolation; x10 for intraspecies variation; x10 for use of a LOAEC, considering severity of effect).
The MOEs for inhalation exposure to diacetyl from microwave popcorn and from its use in cosmetics are considered adequate to account for uncertainties in the available health effects and exposure data.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated.
2,3-PD
No carcinogenicity data were identified for 2,3-PD.
Based on the preliminary results of a sub-chronic inhalation study in rats and mice (NTP 2017), the respiratory tract (the nasal passage and larynx in particular) is the target of 2,3-PD-induced toxicity, similar to observations made with diacetyl. Incidences of non-neoplastic lesions were significantly increased in rats and mice at concentrations of 102 mg/m3 and above (NOAEC = 51 mg/m3). Similar results were reported in shorter-term studies with a LOAEC of 205 mg/m3, the lowest concentration tested (Morgan et al. 2012).
Although effects observed in mice and rats exposed to 2,3-PD were located mainly in the respiratory tract, systemic effects (decreased mean body weight gain and organ weights) were also observed at 205 mg/m3 and higher in the absence of histopathological alterations. Therefore, with respect to oral exposures, route-to-route extrapolation from the sub-chronic inhalation study, resulting in a NOAEL of 24 mg/kg bw/day for non-respiratory tract effects, and read-across from the sub-chronic oral study with diacetyl, resulting in a NOAEL of 105 mg/kg bw/day, were used to characterize the risk of ingested 2,3-PD.
The predominant source of exposure to 2,3-PD for the general population is through the diet, primarily from its natural occurrence in foods. The estimated oral intake from the use of 2,3-PD as a possible flavouring agent in foods for the general population 1 year of age and older (1 µg/kg bw/day to 4 µg/kg bw/day) is less than intakes that have been estimated from natural occurrence for this substance (6.5 µg/kg bw/day to 208 µg/kg bw/day). Although the MOE associated with an oral NOAEL of 24 mg/kg bw/day based on route-to-route extrapolation is low, in light of the absence of histopathological changes in internal organs in laboratory animals and the conservative approaches used to estimate exposure to 2,3-PD from its natural occurrence in food, intake of 2,3-PD from its natural occurrence in food was not identified as a concern for human health (MOEs range from 115 to 16,154, target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]). The MOEs for 2,3-PD from its use as a possible flavouring agent in food are ≥6,000 (target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]) and are considered adequate to account for uncertainties in the available health effects and exposure data used to characterize risk. Furthermore, the JECFA (WHO 1999a) concluded that there is “no safety concern at current levels of intake when used as a flavouring agent; secondary components do not raise a safety concern” on the basis of the dietary exposure estimates for 2,3-PD. Comparison of measured indoor air concentrations of 2,3-PD (2.8 µg/m3) and the NOAEC of 9.1 mg/m3 (adjusted for continuous exposureFootnote 9 from a NOAEC of 51 mg/m3) results in MOEs (3,250, target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]) that are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk.
2,3-PD was also identified in products available to consumers, including certain continuous air fresheners for home care and in fragrant oils, which can be used as an air freshener. Comparison of the 6-hour TWA estimated exposures from the use of air fresheners containing 2,3-PD (0.004 mg/m3 to 0.14 mg/m3) with the NOAEC of 51 mg/m3 resulted in MOEs ranging from 364 to 12,750 (target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]), which are considered adequate to address uncertainties in the available health effects and exposure data.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated.
Acetoin
A limited number of toxicity studies were available for acetoin. Acetoin did not promote lung tumours in a limited screening test for carcinogenicity of food additives in mice (Stoner et al. 1973). Acetoin is not expected to be genotoxic.
To characterize the risk associated with oral exposure to acetoin, a NOAEL of 330 mg/kg bw/day was used and was based on changes in body weight and hematological parameters in rats in a 13-week study (Gaunt et al. 1972 reviewed in WHO 1999a).
On the basis of a preliminary account of the results of an inhalation study, acetoin did not induce any effects in mice exposed to concentrations of up to 2,883 mg/m3 for 14 weeks (NTP 2017), which is considered to be the NOAEC.
The predominant source of exposure to acetoin for the general population is through the diet, primarily from its natural occurrence in foods. The estimated oral intake from the use of acetoin as a possible flavouring agent in foods for the general population 1 year of age and older (30 µg/kg bw/day to 47 µg/kg bw/day) is less than intakes that have been estimated from natural occurrence for this substance (161 µg/kg bw/day to 369 µg/kg bw/day). Additional intake of acetoin from its natural occurrence in food was not identified as a concern for human health. On the basis of the conservative approaches used to estimate exposure to acetoin from its natural occurrence in food, intake of acetoin from its natural occurrence in food was not identified as a concern for human health (MOEs range from 894 to 2050, target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]). The MOEs for acetoin from its use as a possible flavouring agent in food are ≥7,021 and are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk. Furthermore, the JECFA (WHO 1999a) concluded that there is “no safety concern at current levels of intake when used as a flavouring agent; secondary components do not raise a safety concern” considering the dietary exposure estimates for substances in this flavouring grouping. Potential inhalation exposures to acetoin in microwave popcorn were also considered. Comparison of the conservative estimates of air concentrations of acetoin from popping microwave popcorn (10 µg/m3 to 4,200 µg/m3) with the NOAEC of 2,883 mg/m3 resulted in MOEs ranging from 686 to 288,300 (target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]), which are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated.
7.3.4 Uncertainties in the evaluation of risk to human health for diacetyl, 2,3-PD, and acetoin
The key sources of uncertainty are presented in the table below. For diacetyl and 2,3-PD, while there is confidence that they induce adverse health effects following inhalation exposure, there is greater uncertainty regarding the toxicity of these substances following long-term oral exposure; this is because ingestion in food is an important source of population exposure owing to the natural occurrence of these diketones in food and their use as food flavouring agents. For acetoin, although the hazard data set is limited, there is sufficient evidence to indicate that it is of lower toxicity than diacetyl.
There is uncertainty on the natural presence of diacetyl in milk consumed by people in Canada and the estimated daily oral intake derived for diacetyl on the basis of a higher-end concentration, given the wide range in diacetyl concentrations in milk reported in the literature.
| Key sources of uncertainty | Impact |
|---|---|
| Absence of or limited Canadian monitoring data in environmental media and foods. | +/- |
| For the natural occurrence of diacetyl, 2,3-PD, and acetoin in food, where a range of concentrations were available from the literature, maximum values were chosen except in the case of outliers. | + |
| The potential inhalation exposures to diacetyl, 2,3-PD, and acetoin when cooking with foods containing these substances, both from natural sources and as a food flavouring agent. | - |
| Insufficient information to quantify inhalation exposures to 2,3-PD from microwave popcorn. | - |
| Potential co-exposure to diacetyl, 2,3-PD, and acetoin from microwave popcorn. | - |
| Assumption that dermal exposures are minimal for diacetyl in comparison to inhalation exposures, given the high volatility of the substance. | - |
| There are no chronic toxicity studies for acetoin and 2,3-PD, and the overall data set on acetoin is limited. | - |
| The data set on the oral toxicity of diacetyl, 2,3-PD, and acetoin is limited. | +/- |
| Humans are considered more sensitive to the respiratory effects of diacetyl (and potentially 2,3-PD) relative to rats and mice. | - |
| There are no dermal absorption data identified for diacetyl, 2,3-PD, and acetoin. | +/- |
| The use of diacetyl as a read-across to assess the data gap for 2,3-PD. | +/- |
+ = uncertainty with potential to cause overestimation of exposure/risk; - = uncertainty with potential to cause underestimation of exposure risk; +/- = unknown potential to cause over- or underestimation of risk.
7.4 Assessment of individual substance 2,4-PD
7.4.1 Exposure assessment
Environmental media
No data were identified on the presence of 2,4-PD in indoor or outdoor air, drinking water, or soil, in Canada or elsewhere. Considering its physical and chemical properties, if released into the environment, 2,4-PD is expected to be distributed mainly into water (~90%) and, to a lesser extent, into air (~10%) (HSDB 1983-; OECD 2001). 2,4-PD is likely to have high mobility in soil and is not likely to adhere to sediment or suspended particles in water (HSDB 1983-).
The level III fugacity model known as ChemCAN (2003) was run to derive potential environmental concentrations of 2,4-PD for Canada using the upper-end quantity data from Table 4‑1 (that is, 1,000,000 kg) and assuming that total import quantity was released into a single region of Canada (that is, the Ontario Mixed-Wood Plain region). The estimated concentration from air, water, and soil was 6.5 ng/m3, 2.7 ng/L, and 0.025 ng/g, respectively. These concentrations were used to estimate exposure to 2,4-PD from environmental media (air, water, and soil) for the general population of Canada. Concentrations of 2,4-PD derived from ChemCAN for soil were all in the nanogram range; with resulting exposures of less than 1 ng/kg bw/day, they are therefore considered to be negligible.
Estimates of exposure for 2,4-PD from environmental media ranged from 0.002 µg/kg bw/day for people 60 years of age and older to 0.004 µg/kg bw/day for people 6 months to 4 years of age (Health Canada 2024). More detail is available in Health Canada (2024).
Food
2,4-PD was identified as a naturally occurring volatile component in a few food items including beef, chicken, coffee, mushrooms, papaya, and passion fruit (Nijssen et al. 1963-2018). Quantitative data were only available for roasted chicken (0.004 ppm) and passion fruit juice (<0.01 ppm) and were used to estimate potential exposures to 2,4-PD from food for the general population of Canada. Quantitative exposure estimates for 2,4-PD from its natural occurrence in foodFootnote 5 ranged from 0.02 µg/kg bw/day for adults 19 years and older to 0.1 µg/kg bw/day for 1- to 3-year-olds (see Appendix C for details).
Exposure is not expected when 2,4-PD is used as a component in the manufacture of food packaging materials.
Products used by consumers
2,4-PD was not identified in any products currently available to consumers in Canada.
7.4.2 Health effects assessment
2,4-PD has been reviewed by OECD (2001) and EFSA (2004). These reviews provide a basis for the health effect characterization in this assessment. A literature search was conducted from a year prior to the OECD SIDS SIAR (2001) up to June 2017, in addition to targeted searches up to January 2020. Significant new information was included to support risk characterization.
Toxicokinetics
EFSA (2004) noted that the substance was expected to be readily absorbed from the gastrointestinal tract and widely distributed and metabolized, considering its structure and physicochemical characteristics. No toxicokinetic information was identified for dermal exposure of 2,4-PD.
Carcinogenicity and genotoxicity
In a 2-year inhalation carcinogenicity study, F344 rats and B6D2F1 mice were exposed to 2,4-PD vapours at target concentrations of 0, 100, 200, or 400 ppm (0, 417, 834, or 1,668 mg/m3, respectively) for 6 hours/day, 5 days/week for 2 years (JBRC 2010a,b). No significantly increased incidences of neoplastic lesions were observed in the extensive range of tissues examined from exposed animals.
The potential genotoxicity of 2,4-PD has been reviewed in OECD (2001) and EFSA (2004). Overall, 2,4-PD was not consistently mutagenic in vitro but showed a clastogenic potential (chromosomal aberrations and sister chromatid exchanges), notably in the absence of metabolic activation (Ballantyne and Cawley 2001; OECD 2001; NTP 2019). In vivo, a clastogenic potential (micronuclei) was observed in mice exposed via oral and intraperitoneal administration, but not in rats or mice exposed via inhalation, suggesting a route dependence (Ballantyne and Cawley 2001; OECD 2001; NTP 2019). Oral administration of 2,4-PD induced DNA damage in mice (NTP 2019), but not in rats (ECHA c2007-2019d). Negative results were obtained for chromosomal aberrations in spermatogonia of mice (OECD 2001), although inhalation exposure resulted in a slight transient dominant lethal effect in rats (Tyl et al. 1989). EFSA (2004) concluded that the use of 2,4-PD as a flavouring substance is not acceptable due to its genotoxic (clastogenic) potential.
Repeated-dose toxicity
In a dermal study, New Zealand White rabbits were exposed to 2,4-PD at doses of 0, 244, 975, and 1,463 mg/kg bw/day by occluded contact daily for 9 days (Ballantyne 2001 as cited in OECD 2001). In the high dose group (1,463 mg/kg bw/day), approximately 50% of animals of either sex died. At the middle and high doses, 2,4-PD caused signs of hypoactivity, prostration, salivation, tremors, gasping, convulsions, cyanosis (as derived from blue cutis of the nasal area), and reduced body weight gain and food consumption, as well as hemorrhages and neuronal degeneration in the brain. On the basis of systemic effects, the author established a NOAEL of 244 mg/kg bw/day and a LOAEL of 975 mg/kg bw/day.
With respect to oral exposure, according to limited data, administration by gavage of doses of 500 mg/kg bw/day to rats for 2 weeks resulted in a range of systemic effects in the bladder, lungs, eyes, thymus, liver, kidneys, heart, and lymph nodes, and all animals administered 1,000 mg/kg bw/day died; the NOAEL was considered to be 100 mg/kg bw/day (Eastman Kodak 1979 as cited in OECD 2001). Other gavage studies by these investigators cited in EFSA (2004) include a 126-day study in rats (NOAEL <200 mg/kg bw/day) and a 14-day study in rabbits (NOAEL = 250 mg/kg bw/day); however, details provided are insufficient for evaluation.
Rats and mice were exposed to 2,4-PD concentrations of up to 1,668 mg/m3 in a 2-year inhalation carcinogenicity study, as described above (JBRC 2010a,b). In rats, non-neoplastic lesions were located mainly in the nasal cavity in both sexes at 834 mg/m3 and above, including squamous metaplasia of the respiratory epithelium, inflammation hyperplasia of the transitional epithelium, and atrophy of the olfactory epithelium. Similarly, nasal cavity effects observed in mice included squamous metaplasia, eosinophilic change, ulcer, necrosis, and transitional cell hyperplasia in the respiratory epithelium; atrophy, respiratory metaplasia, eosinophilic change, and necrosis in the olfactory epithelium; and respiratory metaplasia in the sub-mucosal gland at 834 mg/m3 and 1,668 mg/m3, while exudate and atrophy in the olfactory epithelium occurred at all concentrations. Alterations in biochemical parameters were also noted in males and females at 834 mg/m3 and 1,668 mg/m3. Considering the nasal lesions, the study authors identified 417 mg/m3 as the NOAEC in rats and the LOAEC in mice (JBRC 2010a,b).
Inhalation exposure of rats to 0, 100, 300, or 650 ppm (0, 417, 1217, or 2,711 mg/m3, respectively) 2,4-PD for 14 weeks induced a wide range of systemic effects at 1,217 mg/m3 and above and high mortality at 2,711 mg/m3. On the basis of hematological, clinical, and urinary chemical effects in the 1,217 mg/m3 group and severe effects at 2,711 mg/m3, including histopathological effects in the brain and thymus, the authors established a NOAEC of 417 mg/m3 for this study (Dodd et al. 1986 as cited in OECD 2001). In a 2-week study by the same authors, the NOAEC and LOAEC were determined to be 834 mg/m3 and 1,668 mg/m3, respectively, on the basis of reduced organ weights and nasal necrosis or inflammation at higher concentrations (OECD 2001).
Since no long-term oral studies were available, a route-to-route extrapolation from the inhalation NOAEC of 417 mg/m3 from the14-week study above was carried out to obtain an oral NOAEL of 23 mg/kg bw/day (see Appendix F).
Reproductive and developmental toxicity
In the sub-chronic inhalation study in male and female F344 rats exposed to 2,4-PD at concentrations of up to 2,711 mg/m3, no significant pathological findings were noted in the testes and epididymis of males or in the uterus, cervix, and ovaries of females (Dodd et al. 1986 as cited in OECD 2001). No studies were identified in which the potential effects of exposure to 2,4-PD on reproductive performance were investigated.
In an inhalation developmental toxicity study, pregnant F344 rats were exposed to concentrations of 0, 53, 202, or 398 ppm (0, 217, 827, or 1,629 mg/m3, respectively) of 2,4-PD for 6 hours/day through GD 6 to 15 (Tyl et al. 1990). No effects were observed on the number of corpora lutea, viable implants per litter, pre- or post-implantation losses, or fetal sex ratio. Significantly reduced fetal body weights per litter (approximately 10%) were reported for males, females, and all fetuses at 1,629 mg/m3. At this concentration, a consistent pattern of reduced fetal ossification, skeletal variations and reduced maternal weight was also reported. At 827 mg/m3, there was a slight but statistically significant reduction (approximately 3%) in fetal body weights per litter in males only. The authors concluded the NOEC to be 217 mg/m3 for both maternal and developmental toxicity (Tyl et al. 1990). Likewise, the OECD (2001) considered 217 mg/m3 to be the developmental NOAEC. However, in light of the minimal decrease in body weight in male fetuses only, which is unlikely to be considered adverse, the NOAEC for developmental and maternal toxicity is considered in this health effects assessment to be 827 mg/m3.
7.4.3 Risk characterization of 2,4-PD
2,4-PD was not carcinogenic in rats and mice exposed via inhalation. Non-cancer lesions were mainly located in the nasal cavity (JBRC 2010a,b). However, clastogenic effects were reported for 2,4-PD, leading EFSA (2004) to conclude that the use of 2,4-PD as a food flavouring substance is not acceptable.
Systemic and developmental toxicities are identified as critical health effects for characterizing the risk of shorter-term inhalation exposure to 2,4-PD. The NOAEC for systemic and developmental toxicity in rats was established at 827 mg/m3. A NOAEC of 834 mg/m3 from a 14-day study in rats (Dodd et al. 1986 as cited in OECD 2001) supports the selection of this critical effect concentration for risk characterization. With respect to longer-term exposure, various systemic effects were observed in the 14-week study in rats, and the NOAEC was established at 417 mg/m3 (Dodd et al. 1986 as cited in OECD 2001).
With respect to chronic exposure, according to limited data, the short-term gavage study with a NOAEL of 100 mg/kg bw/day (Eastman Kodak 1979 as cited in OECD 2001) and a route-to-route extrapolation from the developmental inhalation study for longer-term exposure (resulting in a NOAEL of 23 mg/kg bw/day) were considered.
Dermal exposure to 2,4-PD also produced a range of effects in rabbits administered 500 mg/kg bw/day; therefore, a NOAEL of 244 mg/kg bw/day (Ballantyne 2001 as cited in OECD 2001) was used to characterize the risk associated with dermal exposure to 2,4-PD.
The general population of Canada is exposed to 2,4-PD in small amounts through environmental media and the diet as a result of its natural occurrence in foods. Comparison of the estimates of exposure for 2,4-PD from environmental media (0.002 µg/kg bw/day to 0.004 µg/kg bw/day) and the NOAELs of 100 mg/kg bw/day (systemic toxicity in a 2-week oral study) and 23 mg/kg bw/day (route-to-route extrapolation from a 14-week inhalation study) resulted in MOEs (>3 million, target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]) that are considered adequate to address any uncertainties in the available health effects and exposure data used to characterize risk. Additional intake of 2,4-PD from its natural occurrence in food was not identified as a concern for human health.
The calculated MOEs for exposure to 2,4-PD from environmental media are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk. Additional intake of 2,4-PD from its natural occurrence in food was not identified as a concern for human health.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures, including for infants and children, were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects.
While exposure of the general population to 2,4-PD is not of concern at current levels, there is the potential for exposures to increase in the future. Given the potential health effects of concern associated with this substance there may be a concern for human health if exposures were to increase.
7.4.4 Uncertainties in the evaluation of risk to human health
The key sources of uncertainty are presented in the table below. In light of the evidence of the genotoxicity of 2,4-PD, it would be helpful to have additional data from longer-term studies to increase confidence that potential risks from prolonged exposures would be low.
| Key source of uncertainty | Impact |
|---|---|
| Absence of or limited Canadian monitoring data in environmental media and foods. | +/- |
| There are no chronic oral or dermal studies or sub-chronic oral studies for 2,4-PD. | +/- |
+ = uncertainty with potential to cause overestimation of exposure/risk; - = uncertainty with potential to cause underestimation of exposure risk; +/- = unknown potential to cause over- or underestimation of risk.
7.5 Assessment of individual substance MO
7.5.1 Exposure assessment
Environmental media
MO was measured in the CHMS Cycle 2 indoor air study and in a smaller indoor air study from Quebec (personal communication, email from the Environmental Health Science and Research Bureau [EHSRB], HC, to the ESRAB, HC, dated December 2012, unreferenced; Won and Lusztyk 2011). MO was detected in less than 12% of samples (limit of detection 0.16 µg/m3) in the CHMS study, with a geometric mean concentration below the limit of detection and a 95th percentile concentration of 0.26 µg/m3 (weighted data at the household level) (Zhu et al. 2013). The geometric mean indoor air concentration of MO from 12 samples from the Quebec field study was 0.30 µg/m3, with a maximum of 4.15 µg/m3 (Won and Lusztyk 2011).
No Canadian data on the presence of MO in water were identified. MO was qualitatively detected in drinking water from Cincinnati, Ohio, as well as in a river in Switzerland (HSDB 1983-). Because of its moderate Henry’s law constant and high vapour pressure, MO is likely to volatilize from water (HSDB 1983-); therefore, drinking water is not expected to be a significant source of population exposure.
No data were identified on the presence of MO in soil in Canada or elsewhere. Owing to its low Log Koc, MO is likely to have high mobility in soil and is not likely to bind to sediment or suspended particles in water (HSDB 1983-); soil is therefore not expected to be an important source of exposure.
Estimates of exposure for MO from air (assuming indoor and ambient air concentrations are the same) ranged from 0.8 µg/kg bw/day for people 60 years of age and older to 2.5 µg/kg bw/day for people 6 months to 4 years of age (Health Canada 2024). More detail is available in Health Canada (2024).
Food
MO is naturally occurring in various food items, including nectarines, bell pepper, tomatoes, crisp bread, parmesan cheese, milk, coffee, tea, peanuts, basil, and shrimp, to name a few (Nijssen et al. 1963-2018; Burdock 2010). It can also be used as a food flavouring agent in baked goods, frozen dairy, gelatins/puddings, milk products, and soft candy (Burdock 2010). Quantitative exposure estimates for MO from its natural occurrence in foodFootnote 5 ranged from 0.3 µg/kg bw/day for people 6 to 12 months old to 2 µg/kg bw/day for people 2 to 3 years old (see Appendix C for details). In the absence of data on its actual use in Canada, the per capita intake estimate is an acceptable estimate of possible dietary exposure to MO from its use as food flavoring agent for the general population (1 year of age and older).
The JECFA evaluated a group of aliphatic secondary alcohols, ketones, and related esters used as flavouring substances, including MO (WHO 2003). As part of that evaluation, the Committee estimated the per capita intake of MO from its use as a food flavouring agent to be 0.0067 µg/kg bw/day for the European population (no intake data were reported for the US) (see Appendix C for details).
Products available to consumers
MO was not identified in any products available to consumers in Canada.
7.5.2 Health effects assessment of MO
MO has been reviewed by the OECD (1997b) and EFSA (2012). These reviews provide a basis for the health effects characterization in this assessment. A literature search was conducted from one year prior to the OECD publication (1997b) until October 2019. No new information was identified on the health effects associated with exposure to MO.
Toxicological information available for MO is limited; therefore, route-to-route extrapolation for MO is supported by read-across from the analogue 6-methyl—5-heptene-2-one (MHE, CAS RN 110-93-0). MHE was the only analogue identified for the read-across of MO on the basis of structural similarity and data availability (TERA 2004). Both MO and MHE are unsaturated ketones; MO contains an alkene group and a ketone group on adjacent carbon atoms, while MHE contains an additional 2 carbons in the chain between the alkene and ketone group. This structural difference suggests that MO may be more reactive (and therefore more toxic) than MHE due to the presence of an α, β-unsaturated carbonyl. Toxicity data on MHE have been used to read across to MO where required.
Carcinogenicity and genotoxicity
No carcinogenicity data were identified for MO.
MO was not mutagenic in Salmonella, Escherichia coli or the mouse lymphoma assay, nor did it induce micronuclei in cultured human lymphocytes or in mice administered MO by intraperitoneal injection (ECHA c2007-2019e).
Repeated-dose toxicity
Two studies are available for repeated-dose inhalation toxicity for MO, and no data are available by either the oral or dermal route.
A combined repeated-dose and reproductive/developmental toxicity study was conducted in groups of SD rats exposed through whole-body inhalation to MO at concentrations of 0, 31, 103, and 302 ppm (0, 124, 413, and 1,211 mg/m3, respectively) for 6 hours/day, 7 days/week for a total of 49 exposures for male rats and 36 to 49 exposures for female rats. There were no effects on mortality, hematological or urinalysis parameters, or in gross pathology for the parental animals. However, significant reductions in feed consumption, body weight, and body weight gain, along with clinical abnormalities and nasal passage pathology, were observed in all test groups in a concentration-dependent matter. The clinical abnormalities included an increased incidence of post-exposure porphyrin nasal discharge and sialorrhea. Histopathological findings included the presence of sero-cellular exudates, chronic focal inflammation, and focal metaplasia of the respiratory and olfactory epithelium of the nasal passage. These changes are considered a common response to an irritating vapour (Bernard and Faber 1992 as cited in OECD 1997b). An inhalation LOAEC of 124 mg/m3 was determined from the effects on feed consumption, body weights, body weight gain, and nasal passage histopathology (OECD 1997b; US EPA 2006; ECHA c2007-2019e). No inhalation NOAEC for systemic toxicity was identified in this study.
Groups of male Wistar rats and guinea pigs of both sexes were exposed through whole-body inhalation to MO at concentrations of 0, 50, 100, 250, or 500 ppm (0, 200, 400, 1,000, or 2,000 mg/m3, respectively) for 8 hours/day, 5 days/week for 6 weeks. Published results were not separated by species. An increased incidence of mortality (65%) was noted at 2,000 mg/m3, after which the authors limited the duration at this level to 10 exposures. Chronic conjunctivitis, nasal irritation, and mild albuminuria were observed at 1,000 mg/m3 and 2,000 mg/m3. Concentration-related increases of pathological effects were observed in the kidney, lung, and/or liver at levels of 400 mg/m3 and higher. The pathological changes included poor growth, congestion of the liver, dilated Bowman’s capsules, swollen convoluted tubular epithelium in the kidney, and congestion of the lungs. No effects were seen at 200 mg/m3 (Smyth et al. 1942). A NOAEC of 200 mg/m3 was presented in the ECHA REACH Registration Dossier on the basis of pathological changes in lung, liver, and/or kidney (ECHA c2007-2019e).
Reproductive and developmental toxicity
In the combined repeated-dose and reproductive/developmental toxicity study described above (Bernard and Faber 1992 as cited in OECD 1997b), in which groups of SD rats were exposed to MO for a total of 49 days for male rats and a total of 36 to 49 days for female rats (through day 20 of gestation), no significant effects on reproductive organs, performance, or gestation length were observed at any concentration tested. However, a reduction in the number of litters produced by the mating pairs was observed at 1211 mg/m3. No external malformations were noted; no histopathological examination was conducted in pups. The NOAEC for reproductive toxicity was 413 mg/m3 based on the decrease in litters (Bernard and Faber 1992 as cited in OECD 1997b; US EPA 2006; ECHA c2007-2019e).
No information has been identified on the potential reproductive and developmental toxicity from oral or dermal exposure to MO.
Route-to-route extrapolation and read-across for hazard characterization
Owing to the lack of data on the toxicity of orally administered MO, observations from inhalation studies are considered in a route-to-route extrapolation approach. In the combined repeated-dose and reproductive/developmental toxicity study in rats, the critical effects mainly occurred in the nasal passage (that is, the site of contact), which the OECD (1997b) attributed to the irritative property of MO. Therefore, it is considered more appropriate to use the NOAEC of 2200 mg/m3 from the 6-week inhalation study in rats, in which systemic effects in the liver and kidney were observed in order to derive an oral NOAEL for MO. The adjusted oral NOAEL for repeated-dose toxicity is 15.0 mg/kg bw/day (see Appendix F).
MHE (CAS RN 110-93-0) was identified as an analogue that could be used for read-across of MO because of its structural similarity and data availability (TERA 2017). A NOAEL of 39 mg/kg bw/day for MO can be derived by read-across from the oral NOAEL of 50 mg/kg bw/day from MHE (based on alterations in hematological and clinical chemistry parameters in a 90-day study with rats [OECD 2003] by adjusting for molecular weight [Appendix A, Table A-4]). This read-across-based NOAEL for MO is similar to the route-to-route extrapolation-based NOAEL (that is, it is only about 2.5-fold greater, as may be expected because of the longer chain).
7.5.3 Risk characterization of MO
No carcinogenic studies on MO have been identified.
The LOAEC of 124 mg/m3 for systemic toxicity in rats was used to characterize the risk associated with inhalation exposure to MO (OECD 1997b; ECHA c2007-2019e). Although no oral toxicity studies on MO have been identified, route-to-route extrapolation of the NOAEC from a 6-week inhalation study (Smyth et al. 1942; ECHA c2007-2019e) is considered appropriate to characterize potential hazards associated with ingestion of the substance. The equivalent oral NOAEL was determined to be 15 mg/kg bw/day (see Appendix F). This value is supported by reading across from toxicity data for an analogous substance, MHE.
The predominant source of exposure to MO for the general population is through air and the diet as a result of its natural occurrence in foods.
MO was not identified in any products available to consumers in Canada.
Comparison of the estimates of exposure to MO from air (assuming indoor and ambient air concentrations are the same,1.2 µg/kg bw/day to 2.5 µg/kg bw/day) and from its potential use as a flavouring agent in food for the general population 1 year of age and older (0.0067 µg/kg bw/day) with the NOAEL of 15 mg/kg bw/day resulted in MOEs greater than 6,000 (target MOE = 100 [x10 for interspecies extrapolation; x10 for intraspecies variation]), which are considered adequate to address uncertainties in the available health effects and exposure data used to characterize risk. Additional intake of MO from its natural occurrence in food was not identified as a concern for human health. Furthermore, the JECFA (WHO 2003) concluded that there is “no safety concern at current levels of intake when used as a flavouring agent,” considering the dietary exposure estimates for MO.
The human health assessment took into consideration those groups of individuals within the Canadian population who, due to greater susceptibility or greater exposure, may be more vulnerable to experiencing adverse health effects. For instance, age-specific exposures were estimated, and developmental and reproductive toxicity studies were evaluated for potential adverse health effects.
7.5.4 Uncertainties in the evaluation of risk to human health
The key sources of uncertainty are presented in the table below. Although the data set for MO is limited, in light of the large MOEs, confidence is high that the risk to the Canadian population from this substance is low.
| Key source of uncertainty | Impact |
|---|---|
| Limited environmental monitoring data for MO. | + |
| Limited hazard data set for MO (especially long-term data). | +/- |
8. Conclusion
Considering all available lines of evidence presented in this assessment, there is low risk of harm to the environment from MEK, MPK, MIBK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO. It is concluded that these 10 substances do not meet the criteria under paragraphs 64(a) or (b) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that have or may have an immediate or long-term harmful effect on the environment or its biological diversity or that constitute or may constitute a danger to the environment on which life depends.
On the basis of the information presented in this assessment, it is concluded that MEK and MIBK meet the criteria under paragraph 64(c) of CEPA as they are entering or may enter the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
On the basis of the information presented in this assessment, it is concluded that MPK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO do not meet the criteria under paragraph 64(c) of CEPA as they are not entering the environment in a quantity or concentration or under conditions that constitute or may constitute a danger in Canada to human life or health.
Therefore, it is concluded that MEK and MIBK meet one or more of the criteria set out in section 64 of CEPA and that MPK, MIAK, DAA, diacetyl, 2,3-PD, acetoin, 2,4-PD, and MO do not meet any of the criteria set out in section 64 of CEPA.
References
Aeschbacher HU, Wolleb U, Löliger J, Spadone JC, Liardon R. 1989. Contribution of coffee aroma constituents to the mutagenicity of coffee. Food Chem Toxicol. 27(4):227-232.
Annan NT, Plahar WA, Poll L, Jakobsen M. 2005. Effect of soybean fortification on Ghanaian fermented maize dough aroma. Int J Food Sci Nutr. 56(5):315-26.
[AOPWIN] Atmospheric Oxidation Program for Microsoft Windows [estimation model]. 2010. Ver. 1.92a. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
[AR] Alberta Recycling. 2017. Progress report 2017. Progress report. Edmonton (AB): AR.
[ATSDR] Agency for Toxic Substances & Disease Registry. 2020. Toxicological profile for 2-butanone [PDF]. Atlanta (GA): US Department of Health and Human Services, Public Health Service.
Ballantyne B. 2001. Systemic toxicity from repeated cutaneous contact with 2,4-pentanedione. Vet Hum Toxicol. 43(1):14-18.
Ballantyne B, Cawley TJ. 2001. Toxicology update: 2,4-pentanedione. J Appl Toxicol. 21(2):165-171.
Batyrova TF. 1973. Substantiation of the maximum permissible concentration of methylisobutyl ketone in air or workrooms. Gig Tr Prof Zabol. 17(11):52-53.
Bawazir AE. 2016. Evaluation of neurotoxicity and testicular toxicity of artificial Butter flavorings. Int J Pharm Res Allied Sci. 5(1):248-258.
Benfenati E, Di Toro N, Fanelli R, Lualdi G, Tridico R, Stella G, Buscaini P, Stimilli L. 1992. Characterization of organic and inorganic pollutants in the Adige river (Italy). Chemosphere. 25(11):1665-1674.
Bernier AM. 2000. Qualité de l’eau potable produite par la Ville de Montréal. Rapport annuel 2000.
Bianchi AP, Varney MS. 1998. Volatile organic compounds in the surface waters of a British estuary. Part 1: occurrence, distribution and variation. Water Res. 32(2):352-370.
Birch AN, Petersen MA, Arneborg N, Hansen ÅS. 2013. Influence of commercial baker's yeasts on bread aroma profiles. Food Res Int. 52(1):160-166.
Bjeldanes LF, Chew H. 1979. Mutagenicity of 1,2-dicarbonyl compounds: maltol, kojic acid, diacetyl and related substances. Mutat Res. 67(4):367-371.
Borghoff SJ, Poet TS, Green S, Davis J, Hughes B, Mensing T, Sarang SS, Lynch AM, Hard GC. 2015. Methyl isobutyl ketone exposure-related increases in specific measures of a2u-globulin (a2u) nephropathy in male rats along with in vitro evidence of reversible protein binding. Toxicology. 333:1-13.
Borkowski L. 2007. Another microwave popcorn problem. The Pump Handle: A water cooler for the public health crowd. [accessed 2017 September].
Braithwaite J. 2000. Ketones. In: Kirk-Othmer encyclopedia of chemical technology. Online version. New York (NY): John Wiley & Sons, Inc. [accessed 2016 July]. [restricted access].
Burdock GA. 2010. Fenaroli’s handbook of flavor ingredients. 6th ed. Orlando (FL): Burdock Group.
Buttery RG, Ling LC, Stern DJ. 1997. Studies on popcorn aroma and flavor volatiles. J Agric Food Chem. 45(3):837-843.
Canada. 1999. Canadian Environmental Protection Act, 1999. S.C. 1999, c.33. Canada Gazette Part III, vol. 22, no. 3.
Canter LW, Sabatini DA. 1994. Contamination of public ground water supplies by Superfund sites. Int J Environ Stud. 46(1):35-57.
Cantin S. 2009. Les pneus hors d’usage [PDF]. Information Sheet. Recyc-Quebec. [accessed 2018 Aug 13].
Cardoso DR, Bettin SM, Reche RV, Lima-Neto BS, Franco DW. 2003. HPLC-DAD analysis of ketones as their 2,4-dinitrophenylhydrazones in Brazilian sugar-cane spirits and rum. Journal of Food Composition and Analysis. 16:563-573.
[CDC] Centers for Disease Control and Prevention (US). 2016. Criteria for a recommended standard: occupational exposure to diacetyl and 2,3-pentanedione. Cincinnati (OH): US Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH). [accessed 2017 Oct 13]. Publication No. 2016-111.
ChemCAN [level III fugacity model of 24 regions of Canada]. 2003. Version 6.00. Peterborough (ON): Trent University, Canadian Centre for Environmental Modelling and Chemistry. [cited 2016 Oct 19].
ChemIDplus [database]. 1993- . Bethesda (MD): US National Library of Medicine. [updated 2012 Nov 26; accessed 2017 Mar].
Cichocki JA, Morris JB. 2017. Inhalation dosimetry modeling provides insights into regional respiratory tract toxicity of inhaled diacetyl. Toxicology. 388:30-39.
[CIMT] Canadian International Merchandise Trade [database]. 2017. 291412 Butanone (methyl ethyl ketone), 2011-2016. Ottawa (ON): Statistics Canada. [cited 2017 May].
Colley J, Gaunt IF, Lansdown AB, Grasso P, Gangolli SD. 1969. Acute and short-term toxicity of diacetyl in rats. Food Cosmet Toxicol. 7(6):571-580.
[ConsExpo Web] Consumer Exposure Web Model. 2016. Bilthoven (NL): Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment].
[CPID] Consumer Product Information Database [database]. [modified 2019]. DeLima Associates. [accessed 2019 December].
Curry P, Kramer G, Newhook R, Sitwell J, Somers D, Tracy B, Oostdam JV. 1993. Reference values for Canadian populations. Prepared by the Environmental Health Directorate Working Group on Reference Values. Health Canada. Unpublished. 1988 [updated in 1993].
David RM, Bernard LG, Banton MI, Tyler TR, Topping DC, Gill MW, O’Donoghue JL. 1999. The effect of repeated methyl iso-butyl ketone vapor exposure on schedule-controlled operant behavior in rats. Neurotoxicology. 20(4):583-593.
De Ceaurriz J, Micillino JC, Bonnet P, Guenier JP. 1981. Sensory irritation caused by various industrial airborne chemicals. Toxicol Lett. 9(2):137-143.
De Ceaurriz J, Micillino JC, Marignac B, Bonnet P, Muller J, Guenier JP. 1984. Quantitative evaluation of sensory irritating and neurobehavioural properties of aliphatic ketones in mice. Food Chem Toxicol. 22(7):545-549.
De Leonardis A, Lopez F, Nag A, Macciola V. 2013. Occurrence and persistence of diacetyl in unfermented and fermented milks. Eur Food Res Technol. 236(4):691-697.
Deacon MM, Pilny MD, John JA, Schwetz BA, Murray FJ, Yakel HO, Kuna RA. 1981. Embryo- and fetotoxicity of inhaled methyl ethyl ketone in rats. Toxicol Appl Pharmacol. 59(3):620-622.
Delzer GC, Ivahnenko T. 2003. Occurrence and temporal variability of methyl tert-butyl either (MTBE) and other volatile organic compounds in select sources of drinking water: results of the focused survey [PDF]. Water-resources investigations report 02-4084. Rapid City (ND): US Geological Survey.
Dietz FK, Rodriguez-Giaxola M, Traiger GJ, Stella VJ, Himmelstein KJ. 1981. Pharmacokinetics of 2-butanol and its metabolites in the rat. J Pharmacokinet Biopharm. 9(5):553-576.
DiVincenzo GD, Kaplan CJ, Dedinas J. 1976. Characterization of the metabolites of methyl n-butyl ketone, methyl iso-butyl ketone, and methyl ethyl ketone in guinea pig serum and their clearance. Toxicol Appl Pharmacol. 36(3):511-522.
Dodd DE, Garman RH, Pritts IM, Troup CM, Snellings WM, Ballantyne B. 1986. 2,4-Pentanedione: 9-day and 14-week vapor inhalation studies in Fischer-344 rats. Fundam Appl Toxicol. 7(2):329-339.
Doepker CL, Maier A, Willis A, Hermansky SJ. 2012. Toxicology of flavors in the food industry. In: Bingham E, Cohhrssen B, editors. Patty’s Toxicology. 6th ed., vol. 5. New York (NY): John Wiley & Sons, Inc. p. 133-168.
Dorado L, Montoya MR, Rodríguez Mellado JM. 1992. A contribution to the study of the structure-mutagenicity relationship for alpha-dicarbonyl compounds using the Ames test. Mutat Res. 269(2):301-306.
Duguay AB, Plaa GL. 1995. Tissue concentrations of methyl isobutyl ketone, methyl n-butyl ketone and their metabolites after oral or inhalation exposure. Toxicol Lett. 75(1-3):51-58.
[ECCC] Environment and Climate Change Canada. 2016a. Science approach document: ecological risk classification of organic substances. Gatineau (QC): ECCC.
[ECCC] Environment and Climate Change Canada. 2016b. Supporting documentation: data used to create substance-specific hazard and exposure profiles and assign risk classifications. Gatineau (QC): ECCC. Information in support of the science approach document: ecological risk classification of organic substances. Available from: substances@ec.gc.ca.
[ECCC] Environment and Climate Change Canada. 2017a. NAPS annual raw data 2006-2010 [Internet]. Gatineau (QC): Environment Canada, Environmental Technology Centre. [accessed 2017 February].
[ECCC] Environment and Climate Change Canada. 2017b. Data collected from a targeted information gathering initiative for assessments under the Chemicals Management Plan (October 2017). Data prepared by ECCC, Health Canada; Existing Substances Program.
[ECCC, HC] Environment and Climate Change Canada, Health Canada. [modified 2017 Mar 12]. Categorization of chemical substances. Ottawa (ON): Government of Canada.
[ECHA] European Chemicals Agency. c2007-2019a. Registered substances database; search results for CAS RN 107-87-9. Helsinki (FI): ECHA. [updated 2017 Aug 25; accessed 2017 Oct 13].
[ECHA] European Chemicals Agency. c2007-2019b. Registered substances database; search results for CAS RN 123-42-2. Helsinki (FI): ECHA. [updated 2017 September 21, 2017; accessed 2017 Oct 23].
[ECHA] European Chemicals Agency. c2007-2019c. Registered substances database; search results for CAS RN 110-12-3. Helsinki (FI): ECHA. [updated 2017 June 28; accessed 2017 Oct 13].
[ECHA] European Chemicals Agency. c2007-2019d. Registered substances database; registered dossier for pentane-2,4-dione, CAS RN 123-54-6. Helsinki (FI): ECHA. [updated 2017 Oct 2; accessed 2017 Oct 13].
[ECHA] European Chemicals Agency. c2007-2019e. Registered substances database; registered dossier for 4-methylpent-3-en-2-one, CAS RN 141-79-7. Helsinki (FI): ECHA. [updated 2017 Apr 14; accessed 2017 Oct 13].
[ECHA] European Chemicals Agency. 2019. Committee for Risk Assessment RAC Opinion proposing harmonised classification and labelling at EU level of 4-methylpentan-2-one; isobutyl methyl ketone. CAS RN 108-10-1. Adopted 20 September 2019. CLH-O-0000001412-86-295/F. Helsinki (FI): ECHA. [accessed 2020 Nov 19].
[EFSA] European Food Safety Authority. 2004. Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in contact with Food (AFC) on a request from the Commission related to Flavouring Group Evaluation 11 (FGE.11): aliphatic dialcohols, diketones, and hydroxyketones from chemical group 10 (Commission Regulation (EC) No 1565/2000 of 18 July 2000) [PDF]. Parma (IT): EFSA. EFSA J. 166:1-44.
[EFSA] European Food Safety Authority. 2012. Scientific opinion on Flavouring Group Evaluation 204 (FGE.204): consideration of genotoxicity data on representatives for 18 mono-unsaturated, aliphatic, α,β-unsaturated ketones and precursors from chemical subgroup 1.2.1 of FGE.19 by EFSA [PDF]. EFSA J. 10(12):2992. Parma (IT): EFSA.
[EFSA] European Food Safety Authority. 2015. Scientific opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs: executive summary [PDF]. EFSA J. 13(1):3978.
[EFSA] European Food Safety Authority. 2016. Safety and efficacy of secondary aliphatic saturated or unsaturated alcohols, ketones, ketals and esters with a second secondary or tertiary oxygenated functional group belonging to chemical group 10 when used as flavourings for all animal species. EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP), Rychen et al., 2016. Parma (IT): EFSA. EFSA J. 14(11):4618-4619.
Egilman DS, Schilling JH. 2012. Bronchiolitis obliterans and consumer exposure to butter-flavored microwave popcorn: a case series. Int J Occup Environ Health. 18(1):29-42.
Environment Canada. 2001. Data for selected substances collected under the Canadian Environmental Protection Act, 1999. Section 71: Notice with Respect to Certain Substances on the Domestic Substances List (DSL). Data prepared by: Environment Canada, Existing Substances Program.
Environment Canada. 2012. Canadian Environmental Protection Act, 1999: Notice with respect to certain substances on the Domestic Substances List [PDF]. Canada Gazette, Part I, vol. 146, no. 48, Supplement.
Environment Canada. 2013. DSL Inventory update data collected under theCanadian Environmental Protection Act, 1999, section 71: Notice with respect to certain substances on the Domestic Substances List. Data prepared by: Environment Canada, Health Canada; Existing Substances Program.
Environment Canada, Health Canada. 2002. Canadian Environmental Protection Act: Priority substances list health assessment supporting documentation report: 2-butoxyethanol. Ottawa (ON): Government of Canada. [accessed 2017 Sept]. Unpublished.
[EPI Suite] Estimation Program Interface Suite for Microsoft Windows [estimation model]. c2000-2012. Ver. 4.11. Washington (DC): US Environmental Protection Agency, Office of Pollution Prevention and Toxics; Syracuse (NY): Syracuse Research Corporation.
European Commission. 2014. Recommendation from the Scientific Committee on Occupational Exposure Limits for diacetyl. SCOEL/SUM/149. Brussels (BE): European Commission; Employment, Social Affairs and Inclusion. [accessed 2020 April].
European Commission. 2017. Cosmetic Ingredients and Substances (CosIng) Simple Search Database [database]. [updated 2017 Oct 20; accessed 2017 September].
Fasano WJ, McDougal JN. 2008. In vitro dermal absorption rate testing of certain chemicals of interest to the Occupational Safety and Health Administration: summary and evaluation of USEPA's mandated testing. Regul Toxicol Pharmacol. 51(2):181-194.
Farsalinos KE, Kistler KA, Gillman G, Voudris V. 2015. Evaluation of electronic cigarette liquids and aerosol for the presence of selected inhalation toxins. Nicotine Tob Res. 17(2):168-174.
Ficheux AS, Chevillotte G, Wesolek N, Morisset T, Dornic N, Bernard A, Bertho A, Romanet A, Leroy L, Mercat AC, et al. 2016. Consumption of cosmetic products by the French population second part: amount data. Food Chem Toxicol. 90:130-141.
Ficheux AS, Morisset T, Chevillotte G, Postic C, Roudot AC. 2014. Probabilistic assessment of exposure to nail cosmetics in French consumers. Food Chem Toxicol. 66:36-43.
Folkendt MM, Weiss-Lopez BE, Chauvel P Jr., True, NS. 1985. Gas-phase proton NMR studies of keto-enol tautomerism of acetylacetone, methyl acetoacetate, and ethyl acetoacetate. J Phys Chem. 89:3347-3352.
Furihata C, Matsushima T. 1987. Use of in vivo/in vitro unscheduled DNA synthesis for identification of organ-specific carcinogens. Crit Rev Toxicol. 17(3):245-277.
Furihata C, Yoshida S, Matsushima T. 1985. Potential initiating and promoting activities of diacetyl and glyoxal in rat stomach mucosa. Jpn J Cancer Res. 76(9):809-814.
Gaffney SH, Abelmann A, Pierce JS, Glynn ME, Henshaw JL, McCarthy LA, Lotter JT, Liong M, Finley BL. 2015. Naturally occurring diacetyl and 2,3-pentanedione concentrations associated with roasting and grinding unflavored coffee beans in a commercial setting. Toxicol Rep. 2:1171-1181.
García-Martínez T, de Lerma NL, Moreno J, Peinado RA, Millán MC, Mauricio JC. 2013. Sweet wine production by 2 osmotolerant Saccharomyces cerevisiae strains. J Food Sci. 78(6):M874-M879.
Gaunt IF, Brantom PG, Kiss IS, Grasso P, Gangolli SD. 1972. Short-term toxicity of acetoin (acetylmethylcarbinol) in rats. Food Cosmet Toxicol. 10(2):131-141.
Geller I, Gause E, Kaplan H, Hartmann RJ. 1979. Effects of acetone, methyl ethyl ketone and methyl isobutyl ketone on a match-to-sample task in the baboon. Pharmacol Biochem Behav. 11(4):401-406.
Giri A, Okamoto A, Okazaki E, Ohshima T. 2010. Headspace volatiles along with other instrumental and sensory analyses as indices of maturation of horse mackerel miso. J Food Sci. 75(8):S406-S417.
Glensvig D, Ports J. 2006. Mapping of perfume in toys and children’s articles [PDF]. Denmark: Danish Ministry of Environment. [accessed 2017 May].
Gloede E, Cichocki JA, Baldino JB, Morris JB. 2011. A validated hybrid computational fluid dynamics-physiologically based pharmacokinetic model for respiratory tract vapor absorption in the human and rat and its application to inhalation dosimetry of diacetyl. Toxicol Sci. 123(1):231-246.
Golder Associates. 1987. Testing of specific organic compounds in soils in background in urban areas. Draft working paper to Shell Canada Limited and Texaco Canada Limited.
Goravanahally MP, Hubbs AF, Fedan JS, Kashon ML, Battelli LA, Mercer RR, Goldsmith WT, Jackson MC, Cumpston A, Frazer DG, et al. 2014. Diacetyl increases sensory innervation and substance P production in rat trachea. Toxicol Pathol. 42(3):582-590.
Grady SJ. 2003. A national survey of methyl tert-butyl ether and other volatile organic compounds in drinking-water sources: results of the random survey [PDF]. Water-Resources Investigations Report 02-4079. East Hartford (CT): US Geological Survey.
Grady SJ, Casey GD. 2001. Occurrence and distribution of methyl tert-butyl ether and other volatile organic compounds in drinking water in the northeast and mid-Atlantic regions of the United States, 1993-98 [PDF]. Water-Resources Investigations Report 00-4228. East Hartford (CT): US Geological Survey.
Hall LW Jr., Hall WS, Bushong SJ, Herman RL. 1987. In situ striped bass (Morone saxatilis) contaminant and water quality studies in the Potomac River. Aquat Toxicol. 10(2-3):73-99.
[HAMN] Hamilton Air Monitoring Network. 2020. Air Quality Summary Reporting - Volatile Organic Compounds (VOCs).
Hansen, J, Hansen OC, Pommer K. 2004. Mapping of chemical substances in consumer products. Release of chemical substances from tents and tunnels for children [PDF, 360 KB]. Denmark: Danish Ministry of Environment. [accessed 2017 May].
Hansen PL, Tønning K, Malmgren-Hansen B, Jacobsen E. 2008. Survey and health assessment of chemical substances in hobby products for children [PDF, 372 KB]. Survey of Chemical Substances in Consumer Products, No. 93. Denmark: Danish Ministry of the Environment.
Health Canada. 1994. Human health risk assessment for priority substances [PDF, 213.5 KB]. Ottawa (ON): Minister of Supply and Services Canada. [accessed 2017 May 25]. Cat. No.: En40-215/41E.
Health Canada. 1998. Exposure factors for assessing total daily intake of priority substances by the general population of Canada. Unpublished report. Ottawa (ON): Health Canada, Environmental Health Directorate.
Health Canada. 2010a. Windsor exposure assessment study (2005-2006): data summary for volatile organic compound sampling. Ottawa (ON): Health Canada. 85 p.
Health Canada. 2010b. Regina indoor air quality study (2007): data summary for volatile organic compound sampling. Ottawa (ON): Health Canada. 164 p.
Health Canada. 2012. Halifax indoor air quality study 2009: VOC sampling data summary (draft). Ottawa (ON): Health Canada. 37 p.
Health Canada. 2013. Edmonton indoor air quality study 2010: Volatile Organic Compounds (VOC) data summary. Ottawa (ON): Health Canada. 50 p.
Health Canada. 2015a. Food Consumption Table derived from Statistics Canada, Canadian Community Health Survey, Cycle 2.2, Nutrition (2004), shared file. Ottawa.
Health Canada. 2015b. Keep calm and use hand sanitizer – how much and how often. Unpublished. Existing Substances Risk Assessment Bureau and New Substances Assessment and Control Bureau, Health Canada. Presented at Health Canada Science Forum 2015.
Health Canada. 2019. Fifth report on human biomonitoring of environmental chemicals in Canada: results of the Canadian Heath Measures Survey Cycle 5 (2016-2017). Ottawa (ON): Health Canada. [accessed 2020 Feb 04].
Health Canada. [modified 2020a Jul 14]. Vaping products regulations. Ottawa (ON): Government of Canada. [accessed 2020 Dec 04].
Health Canada. [modified 2020b Nov 11]. Industry guide to vaping products subject to the Canada Consumer Product Safety Act. Ottawa (ON): Government of Canada. [accessed 2020 Dec 21].
Health Canada. 2024. Supporting documentation: intake tables for Ketones Group. Ottawa (ON): Health Canada. Information in support of the assessment for the Ketones Grouping. Available from: eccc.substances.eccc@canada.ca.
Hjelm EW, Boman A, Fernström P, Hagberg M, Johanson G. 1991. Percutaneous uptake and kinetics of methyl isobutyl ketone (MIBK) in the guinea pig. Toxicol Lett. 56(1-2):79-86.
Hjelm EW, Hagberg M, Iregren A, Löf A. 1990. Exposure to methyl isobutyl ketone: toxicokinetics and occurrence of irritative and CNS symptoms in man. Int Arch Occup Environ Health. 62(1):19-26.
Holloway AL, Treacy J, Sidebottom H, Mellouki A, Daële V, Le Bras G, Barnes I. 2005. Rate coefficients for the reactions of OH radicals with the keto/enol tautomers of 2,4-pentanedione and 3-methyl-2,4-pentanedione, allyl alcohol and methyl vinyl ketone using the enols and methyl nitrite as photolytic sources of OH. J Photochem Photobiol A Chem. 176:183-190.
[HSDB] Hazardous Substances Data Bank [database]. 1983-. Bethesda (MD): National Library of Medicine (US). [accessed 2017 Oct 23].
Hubbs AF, Battelli LA, Goldsmith WT, Porter DW, Frazer D, Friend S, Schwegler-Berry D, Mercer RR, Reynolds JS, Grote A, et al. 2002. Necrosis of nasal and airway epithelium in rats inhaling vapors of artificial butter flavouring. Toxicol Appl Pharmacol. 185(2):128-135.
Hubbs AF, Cumpston AM, Goldsmith WT, Battelli LA, Kashon ML, Jackson MC, Frazer DG, Fedan JS, Goravanahally MP, Castranova V, et al. 2012. Respiratory and olfactory cytotoxicity of inhaled 2,3-pentanedione in Sprague-Dawley rats. Am J Pathol. 181(3):829-844.
Hubbs AF, Goldsmith WT, Kashon ML, Frazer D, Mercer RR, Battelli LA, Kullman GJ, Schwegler-Berry D, Friend S, Castranova V. 2008. Respiratory toxicologic pathology of inhaled diacetyl in Sprague-Dawley rats. Toxicol Pathol. 36(2):330-344.
Hubbs AF, Kreiss K, Cummings KJ, Fluharty KL, O’Connell R, Cole A, Dodd TM, Clingerman SM, Flesher JR, Lee R, et al. 2019. Flavorings-related lung disease: a brief review and new mechanistic data. Toxicol Pathol. 47(8):1012-1026.
Hughes BJ, Thomas J, Lynch AM, Borghoff SJ, Green S, Mensing T, Sarang SS, LeBaron MJ. 2016. Methyl isobutyl ketone-induced hepatocellular carcinogenesis in B6C3F1 mice: a constitutive androstane receptor (CAR)-mediated mode of action. Regul Toxicol Pharmacol. 81:421-429.
[IARC] International Agency for Research on Cancer. 2013. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans – Volume 101: Some Chemicals Present in Industrial and Consumer Products, Food and Drinking-water. IARC Monogr Eval Carcinog Risks Hum. 101. Lyon, France.
Imhof R, Glättli H, Bosset JO. 1995. Volatile organic compounds produced by thermophilic and mesophilic single strain dairy starter cultures. LWT - Food Sci Technol. 28(1):78-86.
[IPCS] International Programme on Chemical Safety. 1990. Environmental Health Criteria 117: Methyl isobutyl ketone. Geneva (CH): United Nations Environment Programme, International Labour Organization, World Health Organization. [accessed 2017 Oct 23].
[IPCS] International Programme on Chemical Safety. 1993. Environmental Health Criteria 143: Methyl ethyl ketone. Geneva (CH): United Nations Environment Programme, International Labour Organization, World Health Organization. [accessed 2017 Oct 23].
[IPCS] International Programme on Chemical Safety. 1997. Methyl isobutyl ketone. Geneva (CH): International Chemical Safety Cards, International Program on Chemical Safety, World Health Organization.
[IPCS] International Programme on Chemical Safety. 1999. Safety evaluation of aliphatic acyclic and alicyclic alpha-diketones and related alpha-hydroxy ketones. Prepared by the Fifty-first meeting of the Joint FAO/WHO Expert Committee on Food Additives, World Health Organization, Geneva, International Programme on Chemical Safety. (WHO Food Additive Series 42). [accessed 2017 Jan 20].
Iregren A, Tesarz M, Wigaeus-Hjelm E. 1993. Human experimental MIBK exposure: effects on heart rate, performance, and symptoms. Environ Res. 63(1):101-108.
[JBRC] Japan Bioassay Research Center. 2010a. Summary of inhalation carcinogenicity study of 2,4-pentanedione in F344 rats [PDF, 1.24 MB]. Japan Industrial Safety and Health Association. [accessed 2017 Jun 15].
[JBRC] Japan Bioassay Research Center. 2010b. Summary of inhalation carcinogenicity study of 2,4-Pentanedione in B6D2F1 mice [PDF, 1.24 MB]. Japan Industrial Safety and Health Association. [accessed 2017 Jun 15].
Juberg DR, Alfano K, Coughlin RJ, Thomson KM. 2001. An observational study of object mouthing behavior by young children. Pediatrics. 107(1):135-142.
Kato F, Araki A, Nozaki K, Matsushima T. 1989. Mutagenicity of aldehydes and diketones. Mutat Res. 216:366-367.
Katz GV, Renner ER Jr., Terhaar CJ. 1986. Sub-chronic inhalation toxicity of methyl isoamyl ketone in rats. Fundam Appl Toxicol. 6(3):498-505.
Klager S, Vallarino J, MacNaughton P, Christiani DC, Lu Q, Allen JG. 2017. Flavoring chemicals and aldehydes in e-cigarette emissions. Environ Sci Technol. 51(18):10806-10813.
Kim SB, Hayase F, Kato H. 1987. Desmutagenic effect of alpha-dicarbonyl and alpha-hydroxycarbonyl compounds against mutagenic heterocyclic amines. Mutat Res. 177(1):9-15.
Kreiss K, Gomaa A, Kullman G, Fedan K, Simoes EJ, Enright PL. 2002. Clinical bronchiolitis obliterans in workers at a microwave-popcorn plant. N Engl J Med. 347(5):330-338.
Larsen ST, Alarie Y, Hammer M, Nielsen GD. 2009. Acute airway effects of diacetyl in mice. Inhal Toxicol. 21(13):1123-1128.
Lawson AM, Speers RA, Zhu J. 1995. Chemical and sensory detection of diacetyl in strawberry juice (short communication) [PDF]. Nahrung. 39(4):323-327.
Li Y, Cakmak S, Zhu J. 2019. Profiles and monthly variations of selected volatile organic compounds in indoor air in Canadian homes: results of Canadian national indoor air survey 2012-2013. Environ Int. 126:134-144.
Liira J, Riihimäki V, Pfäffli P. 1988. Kinetics of methyl ethyl ketone in man: absorption, distribution and elimination in inhalation exposure. Int Arch Occup Environ Health. 60(3):195-200.
[LNHPD] Licensed Natural Health Products Database [database]. [modified 2021 Sep 08]. Ottawa (ON): Health Canada. [accessed 2022 Jun 22].
Loretz LG, Api AM, Barraj LM, Burdick J, Dressler WE, Gettings SD, Han Hsu H, Pan YHL, Re TA, Renskers KJ, et al. 2005. Exposure data for cosmetic products: lipstick, body lotion, and face cream. Food Chem Toxicol. 43(2):279-291.
Macciola V, Candela G, De Leonardis A. 2008. Rapid gas-chromatographic method for the determination of diacetyl in milk, fermented milk and butter. Food Control. 19(9):873-878.
MacEwen JD, Vernot EH, Haun CC. 1971. Effect of 90-day continuous exposure to methylisobutylketone on dogs, monkeys and rats. Aerospace Medical Research Laboratory Document No. AMRL-TR-71-65. NTIS No. AD Rep. 730291.
[MAI] Microbiological Associates, Inc. 1986. Sub-chronic toxicity of methyl isobutyl ketone in Sprague Dawley rats. Final report. Study No. 5221.04. Performed by Microbiological Associates, Inc. for Research Triangle Institute. Unpublished report dated July 15, 1986.
Malysheva MV. 1988. The effect of the methods of cutaneous administration of methyl isobutyl ketone on its toxicity. Gig Sanit. 10:79-80.
Marnett LJ, Hurd HK, Hollstein MC, Levin DE, Esterbauer H, Ames BN. 1985. Naturally occurring carbonyl compounds are mutagens in Salmonella tester strain TA104. Mutat Res. 148(1-2):24-34.
Messaadia L, El Dib G, Ferhati A, Chakir A. 2015. UV–visible spectra and gas-phase rate coefficients for the reaction of 2,3-pentanedione and 2,4-pentanedione with OH radicals. Chem Phys Lett. 626:73-79.
McCready D, Fontaine D. 2010. Refining ConsExpo evaporation and human exposure calculations for REACH. Human and Ecological Risk Assessment: An International Journal. 16(4):783-800.
McCoy MJ, Hoppe Parr KA, Anderson KE, Cornish J, Haapala M, Greivell J. 2017. Diacetyl and 2,3-pentanedione in breathing zone and area air during large-scale commercial coffee roasting, blending and grinding processes. Toxicol Rep. 4:113-122.
More SS, Raza A, Vince R. 2012. The butter flavorant, diacetyl, forms a covalent adduct with 2-deoxyguanosine, uncoils DNA, and leads to cell death. J Agric Food Chem. 60(12):3311-3317.
Morgan DL, Flake GP, Kirby PJ, Palmer SM. 2008. Respiratory toxicity of diacetyl in C57BL/6 mice. Toxicol Sci. 103(1):169-180.
Morgan DL, Jokinen MP, Price HC, Gwinn WM, Palmer SM, Flake GP. 2012. Bronchial and bronchiolar fibrosis in rats exposed to 2,3-pentanedione vapors: implications for bronchiolitis obliterans in humans. Toxicol Pathol. 40(3):448-465.
Morris JB, Hubbs AF. 2009. Inhalation dosimetry of diacetyl and butyric acid, 2 components of butter flavoring vapors. Toxicol Sci. 108(1):173-183.
Mujić I, Bavcon Kralj M, Jokić S, Jug T, Subarić D, Vidović S, Zivković J, Jarni K. 2014. Characterisation of volatiles in dried white varieties figs (Ficus carica L.). J Food Sci Technol. 51(9):1837-1846.
Munies R, Wurster DE. 1965. Investigation of some factors influencing percutaneous absorption III: absorption of methyl ethyl ketone. J Pharm Sci. 54(9):1281-1284.
Mutch RD Jr, Daigler J, Clarke JH. 1983. Clean-up of Shope’s landfill, Girard, PA. In: National Conference on Management of Uncontrolled Hazardous Waste Sites. Washington, DC. p. 296-300.
[NCI] National Chemical Inventories [software]. 2015. Issue 2. Columbus (OH): American Chemical Society, Chemical Abstracts Service.
Nemec M, Pitt J, Topping D, Gingell R, Pavkov K, Rauckman E, Harris S. 2004. Inhalation two-generation reproductive toxicity study of methyl isobutyl ketone in rats. Int J Toxicol. 23(2):127-143.
[NHPID] Natural Health Products Ingredients Database [database]. [modified 2022 Apr 09]. Ottawa (ON): Health Canada. [accessed 2022 Jun 22].
[NICNAS] National Industrial Chemicals Notification and Assessment Scheme. 2017. 2-Pentanone, 4-methyl-. Sydney (AU): Department of Health, National Industrial Chemicals Notification and Assessment Scheme. Human Health Tier III assessment for 2-Pentanone, 4-methyl- , CAS No. 108-10-1. [accessed 2017 Oct 13].
Nijssen LM, Ingen-Visscher CA van, Donders JJH, editors. 1963-2018. VCF Volatile Compounds in Food online database [database]. Version 16.6.1. Zeist (NL): Triskelion B.V. [updated 2018; accessed 2020 January].
Nilsson NH, Malmgren-Hansen B, Bernth N, Pedersen E, Pommer K. 2006. Survey of chemical substances in consumer products: survey and health assessment of chemicals substances in sex toys [PDF, 892 KB]. Report no. 77. [accessed 2017 May]. Denmark: Danish Ministry of Environment.
[NPRI] National Pollutant Release Inventory [database]. 2011-2017a. NPRI Datasets: Methyl ethyl ketone (78-93-3). Gatineau (QC): Environment and Climate Change Canada. Search results for 78-93-3. [modified 2019 Aug 20; accessed 2019 Nov 22].
[NPRI] National Pollutant Release Inventory [database]. 2011-2017b. NPRI Datasets: Methyl isobutyl ketone (108-10-1). Gatineau (QC): Environment and Climate Change Canada. Search results for 8-10-1. [modified 2019 Aug 20; accessed 2019 Nov 22].
[NPRI] National Pollutant Release Inventory. 2021. Pollutant release and transfer data reported by facilities, single year tabular format – National Pollutant Release Inventory (NPRI). NPRI Datasets for 2015, 2016, 2017, 2018, 2019 [Internet]. Gatineau (QC): Environment and Climate Change Canada. [accessed 2021 June].
[NTP] National Toxicology Program. 2007a. Chemical information review document for artificial butter flavoring and constituents. Diacetyl (CAS No. 431-03-8) and acetoin (CAS No. 513-86-0). Supporting nomination for toxicological evaluation by the National Toxicology Program, January 2007. Prepared by Integrated Laboratory Systems, Inc. Research Triangle Park (NC): US Department of Health and Human Services, National Toxicology Program. Contract No.: N01-ES-35515.
[NTP] National Toxicology Program. 2007b. NTP toxicology and carcinogenesis studies of Methyl Isobutyl Ketone (CAS NO. 108–10–1) in F344/N rats and B6C3F1 mice (inhalation studies). Research Triangle Park (NC): US Department of Health and Human Services, National Toxicology Program. Report No. 538: 1-236.
[NTP] National Toxicology Program. 2017. Tox-99: Acetoin and 2,3-pentanedione, Toxicity Report Tables & Curves. Research Triangle Park (NC): US Department of Health and Human Services, National Toxicology Program. [accessed 2017 Oct 13]. Results only.
[NTP] National Toxicology Program. 2018. NTP Technical Report on the toxicology and carcinogenesis studies of 2,3-butanedione in Wistar Han rats and B6C3F1/N mice (inhalation studies) [PDF]. NTP TR 593. Final. August 2018.
[NTP] National Toxicology Program. 2019. Acetylacetone (123-54-6). Chemical Effects in Biological Systems (CEBS). Research Triangle Park (NC): National Toxicology Program. Accessed 12 Dec 2019.
Nylén D, Borling P, Sørensen H. 2004. Survey of chemical substances in consumer products: Mapping of chemical substances in animal care products [PDF, 976 KB]. Report no. 44. [accessed 2017 May]. Denmark: Danish Ministry of Environment.
O’Donoghue JL. 2012a. Ketones of 4 or 5 carbons. In Patty’s Toxicology Sixth Edition, E. Bingham, and B. Cohrssen (eds.). John Wiley & Sons, Inc. p. 753-806.
O’Donoghue JL. 2012b. Ketones of 6 to 13 carbons. In Patty’s Toxicology Sixth Edition, E. Bingham, and B. Cohrssen (eds.). John Wiley & Sons, Inc. p. 807-914.
[OECD] Organisation for Economic Co-operation and Development. 1996. SIDS initial assessment report: Methyl isobutyl ketone: CAS No. 108-10-1 [PDF, 71.4 KB]. SIAM [SIDS Initial Assessment Meeting] 5: 1996 Oct. [accessed 2017 April].
[OECD] Organisation for Economic Co-operation and Development. 1997a. SIDS initial assessment report: Methyl ethyl ketone: CAS No. 78-93-3. SIAM [SIDS Initial Assessment Meeting] 6: 1997 June: Paris, France. [accessed 2017 May 5].
[OECD] Organisation for Economic Co-operation and Development. 1997b. SIDS initial assessment report: Mesityl oxide: CAS No. 141-79-7. SIAM [SIDS Initial Assessment Meeting] 6: 1997 June: Paris, France [updated 2010 Nov; accessed 2017 May 5].
[OECD] Organisation for Economic Co-operation and Development. 2000. SIDS initial assessment report: Diacetone alcohol, CAS No. 123-42-2. SIAM [SIDS Initial Assessment Meeting] 10; 2000 March; Japan [accessed 2017 April].
[OECD] Organisation for Economic Co-operation and Development. 2001. SIDS initial assessment report: 2,4-Pentanedione, CAS No. 123-54-6 [PDF, 465 KB]. SIAM [SIDS Initial Assessment Meeting] 13; 2001 Sept; Bern, Switzerland. [accessed 2017 Oct 13].
[OECD] Organisation for Economic Co-operation and Development. 2003. SIDS initial assessment report: 6-methylhept-5-en-2-one: CAS No. 110-93-0 [PDF, 531 KB]. SIAM [SIDS Initial Assessment Meeting] 17: 2003 November: Paris, France [accessed 2017 May 17].
OECD QSAR Toolbox [Read-across tool]. 2014. Version 3.3. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
OECD QSAR Toolbox [Read-across tool]. 2017. Ver. 4.1. Paris (FR): Organisation for Economic Co-operation and Development, Laboratory of Mathematical Chemistry.
Parmet AJ, von Essen S. 2002. Rapidly progressive, fixed airway obstructive disease in popcorn workers: a new occupational pulmonary illness? J Occup Environ Med. 44(3):216-218.
Paxéus, N. 1996. Organic pollutants in the effluents of large wastewater treatment plants in Sweden. Water Res. 30(5):1115-1122.
Pellizzari ED, Hartwell TD, Harris III BSH, Waddell RD, Whitaker DA, Erickson MD. 1982. Purgeable organic compounds in mother’s milk. Bull Environ Contam Toxicol. 28(3):322-328.
Perbellini L, Brugnone F, Mozzo P, Cocheo V, Caretta D. 1984. Methyl ethyl ketone exposure in industrial workers. Uptake and kinetics. Int Arch Occup Environ Health. 54(1):73-81.
[P&G] Procter & Gamble. c2017a. Febreze® PLUG. [accessed 2017 October].
[P&G] Procter & Gamble. c2017b. Febreze Set & Refresh Rain Air Freshener 5.5 ml. [accessed 2017 October].
Phillips RD, Moran EJ, Dodd DE, Fowler EH, Kary CD, O’Donoghue J. 1987. A 14-week vapor inhalation toxicity study of methyl isobutyl ketone. Fundam Appl Toxicol. 9(3):380-388.
Pierce JS, Abelmann A, Lotter JT, Comerford C, Keeton K, Finley BL. 2015. Characterization of naturally occurring airborne diacetyl concentrations associated with the preparation and consumption of unflavoured coffee. Toxicol Rep. 2:1200-1208.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2002. Children’s toys fact sheet: to assess the risks for the consumer [PDF, 303 KB]. Bilthoven (NL): RIVM. [accessed 2017 April-October]. Report No.: 612810012/2002.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2006. Cosmetics fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4 [PDF, 330 KB]. Bilthoven (NL): RIVM. [accessed 2017 April-October]. Report No.: 320104001/2006.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2007a. Do-it-yourself products fact sheet: to assess the risks for the consumer [PDF, 417 KB]. Bilthoven (NL): RIVM. [accessed 2017 April-October]. Report No.: 320104007/2007.
[RIVM] Rijksinstituut voor Volksgezondheid en Milieu [National Institute for Public Health and the Environment]. 2007b. Paint products fact sheet: to assess the risks for the consumer: updated version for ConsExpo 4 [PDF, 264 KB]. Bilthoven (NL): RIVM. [accessed 2017 April-October]. Report No.: 320104008/2007.
Rosati JA, Krebs KA, Liu X. 2007. Emissions from cooking microwave popcorn. Crit Rev in Food Sci Nutr. 47(8):701-709.
[RTECS] Registry of toxic effects of chemical substances.1987. Registry of toxic effects of chemical substances. 1985-86 ed., vol 1-6. Cincinnati (OH): National Institute for Occupational Safety and Health. DHSS (NIOSH) Publication No. 87-114.
Rychlik M, Grosch W. 1996. Identification and quantification of potent odorants formed by toasting of wheat bread. LWT - Food Sci Technol. 29(5-6):515-525.
Sabel GV, Clark TP. 1984. Volatile organic compounds as indicators of municipal solid waste leachate contamination. Waste Manag Res. 2(2):119-130.
Saillenfait AM, Gallissot F, Sabaté JP, Bourges-Abella N, Cadot R, Morel G, Lambert AM. 2006. Developmental toxicity of combined ethylbenzene and methylethylketone administered by inhalation to rats. Food Chem Toxicol. 44(8):1287-1298.
Sánchez-Peña CM, Luna G, García-González DL, Aparicio R. 2005. Characterization of French and Spanish dry-cured hams: influence of the volatiles from the muscles and the subcutaneous fat quantified by SPME-GC. Meat Sci. 69(4):635-645.
Sawhney BL, Kozloski RP. 1984. Organic pollutants in leachates from landfill sites. J Environ Qual. 13(3):349-352.
Schneider K, de Hoogd M, Madsen MP, Haxaire P, Bierwishch A, Kaiser E. 2020a. ERASSTRI – European risk assessment study on synthetic turf rubber infill – Part 1: analysis of infill samples. Sci Total Environ. 718:137174.
Schneider K, de Hoogd M, Haxaire P, Philipps A, Bierwishch A, Kaiser E. 2020b. ERASSTRI – European risk assessment study on synthetic turf rubber infill – Part 2: migration and monitoring studies. Sci Total Environ. 718:137173.
Schwetz BA, Leong BKJ, Gehring PJ. 1974. Embryo- and fetotoxicity of inhaled carbon tetrachloride, 1,1-dichloroethane and methyl ethyl ketone in rats. Toxicol Appl Pharmacol. 28(3):452-464.
Schwetz BA, Mast TJ, Weigel RJ, Dill JA, Morrissey RE. 1991. Developmental toxicity of inhaled methyl ethyl ketone in Swiss mice. Fundam Appl Toxicol. 16(4):742-748.
[SCOEL] European Commission. Scientific Committee on Occupational Exposure Limits. 2014. Recommendation from the Scientific Committee on Occupational Exposure Limits for diacetyl [PDF, 506.16 KB]. SCOEL/SUM/149. [accessed 2017 Oct 13].
Scott B, Moore L. 2000. Assessment of the risks to human health posed by azo colourants in toys, writing inks and paper products, and analysis of the advantages and drawbacks of restrictions on their marketing and use. Final report. United Kingdom Laboratory of the Government Chemist.
[SCREEN3] Screening Tool Program for Windows. 1996. Version 4.10. Research Triangle Park (NC): US Environmental Protection Agency, Office of Air Quality Planning and Standards Emissions, Monitoring, and Analysis Division.
[SDS] Safety Data Sheet. 2008. Permanent markers. La Mirada (CA): Kittrich Corporation. [accessed 2016 July].
[SDS] Safety Data Sheet. 2011. 3M™ Throttle Plate & Carb Cleaner. St. Paul (MN): 3M. [accessed 2020 April].
[SDS] Safety Data Sheet. 2012a. Homax Wall Texture Pro Grade- Orange Peel [PDF]. Bellingham (WA): Homax Group Inc. [accessed 2020 April].
[SDS] Safety Data Sheet. 2012b. 03405-0720 MOTOMASTER CARBURETOR CLEANER 312G [PDF]. Vaughan (ON): K-G SPRAY-PAK INC. [accessed 2020 April].
[SDS] Safety Data Sheet. 2012c. Sharpie Fine Point Marker, Sharpie Ultra Fine Point Marker, etc. Oak Brook (IL): Newell Rubbermaid. [accessed 2016 July].
[SDS] Safety Data Sheet. 2013. RectorSeal No.5. Houston (TX): RectorSeal. [accessed 2020 April].
[SDS] Safety Data Sheet. 2014a. Loctite® Stik 'n Seal Oil & Gas Resistant Adhesive [PDF]. Rocky Hill (CT): Henkel Corporation. [accessed 2020 April].
[SDS] Safety Data Sheet. 2014b. Berol, Expo Low Odor, Expo Click, etc. Oak Brook (IL): Newell Rubbermaid. [accessed 2016 July].
[SDS] Safety Data Sheet. 2015a. Oatey Clear Cleaner [PDF]. Cleveland (OH): Oatey Inc. [accessed 2017 November].
[SDS] Safety Data Sheet. 2015b. BIC Great Erase Bold Dry Erase Marker [PDF]. Shelton (CT): BicUSA Inc. [accessed 2020 April].
[SDS] Safety Data Sheet. 2015c. Turtle Wax Premium Scratch Repair Kit - Clear Coat Pen. Willowbrook (IL): Turtle Wax Inc. [accessed 2020 April].
[SDS] Safety Data Sheet. 2015d. Febreze Noticeables Air Freshener – Autumn Charm. Cincinnati (OH): Procter & Gamble, Fabric and Home Care Division. [accessed 2016 July].
[SDS] Safety Data Sheet. 2015e. Febreze Set & Refresh – Cozy Vanilla Sugar. Cincinnati (OH): Procter & Gamble, Fabric and Home Care Division. [accessed 2016 July].
[SDS] Safety Data Sheet. 2016a. Gun Metal Gray PDS Aerosol. Blaine (MN): Plasti Dip International. [accessed 2020 April].
[SDS] Safety Data Sheet. 2016b. NEVERWET 11 FL OZ 6PK RAIN REPELNT. Vernon Hills (IL): Rust-Oleum Corporation. [accessed 2021 September].
[SDS] Safety Data Sheet. 2016c. WATCO 6X946ML LACQUER CLEAR GLOSS WF [PDF]. Concord (ON): Rust-Oleum Corporation. [accessed 2017 November].
[SDS] Safety Data Sheet. 2016d. Vanilla Custard Flavor. Scotts Valley (CA): Perfumer’s Apprentice. [accessed 2016 July].
[SDS] Safety Data Sheet. 2017a. AUTORF +SSPR 6PK VINYL AND FABRIC SAND [PDF]. Vernon Hills (IL): Rust-Oleum Corporation. [accessed 2020 April].
[SDS] Safety Data Sheet. 2017b. Epoxy Thinner. Montreal (QC): Swing Paints Ltd. [accessed 2017 November].
[SDS] Safety Data Sheet. 2018a. Furniture Refinisher 1801. Montreal (QC): Swing Paints Ltd. [accessed 2019 November].
[SDS] Safety Data Sheet. 2018b. Vacubond Solvent Cement (Clear) [PDF]. Markham (ON): Sluyter Company Ltd. [accessed 2020 April].
[SDS] Safety Data Sheet. 2018c. 3M™ Bondo® Glazing & Spot Putty 907, 907M, 907C, 907ES, 907W, 937, 937C. St. Paul (MN): 3M. [accessed 2020 April].
[SDS] Safety Data Sheet. 2018d. TRMCLD 6X426g GRAFFITI REMOVER [PDF]. Concord (ON): Rust-Oleum Canada (ROCA). [accessed 2020 April].
[SDS] Safety Data Sheet. 2019a. Methyl Ethyl Ketone (MEK). Montreal (QC): Recochem Inc. [accessed 2020 April].
[SDS] Safety Data Sheet. 2019b. DUPLI-COLOR® Adhesion Promoter. Ciudad de México (MX): Compania Sherwin-Williams. [accessed 2020 April].
[SDS] Safety Data Sheet. 2019c. 3M™ Bondo® MEKP Liquid Hardener. London (ON): 3M Canada Company. [accessed 2020 April].
[SDS] Safety Data Sheet. 2019d. Berryman B-12 Chemtool Carburetor, Choke & Throttle Body Cleaner. Arlington (TX): Berryman Products, Inc. [accessed 2020 April].
[SDS] Safety Data Sheet. 2019e. 3 Primer (Clear) [PDF]. Markham (ON): Sluyter Company Ltd. [accessed 2020 April].
[SDS] Safety Data Sheet. 2019f. Maple Sugar Fragrance Oil [PDF]. Mississauga (ON): New Directions Aromatics Inc. [accessed 2020 April].
[SDS] Safety Data Sheet. 2020. KRYLON® FARM AND IMPLEMENT International Harvest Red. Mississauga (ON): Krylon Products Group. [accessed 2020 April].
[SDS] Safety Data Sheet. 2021. DUPLI-COLOR™ Truck Bed Liner Black [PDF]. Cleveland (OH): Dupli-Color Products Company. [accessed 2021 August].
Shane BS, Troxclair AM, McMillin DJ, Henry CB. 1988. Comparative mutagenicity of 9 brands of coffee to Salmonella typhimurium TA100, TA102, and TA104. Environ Mol Mutag. 11(2):195-206.
Sheldon LS, Hites RA. 1978. Organic compounds in the Delaware River. Environ Sci Technol. 12(10):1188-1194.
Shimoda M, Yoshimura T, Ishikawa H, Hayakawa I, Osajima Y. 2000. Volatile compounds of human milk. J Fac Agr. 45(1):199-206.
Silverman L, Schulte HF, First MW. 1946. Further studies on sensory response to certain industrial solvent vapors. J Ind Hyg Toxicol. 28(6):262-266.
Smyth HF Jr. 1956. Hygienic standards for daily inhalation. Am Ind Hyg Assoc Q. 17(2):129-185.
Smyth HF Jr, Seaton J, Fischer L. 1942. Response of guinea pigs and rats to repeated inhalation of vapors of mesityl oxide and isophorone. J Ind Hyg Toxicol. 24(3):46-50.
Smyth HF Jr, Carpenter CP, Weil CS. 1951. Range-finding toxicity data: list IV. AMA Arch Ind Hyg Occup Med. 4(2):119-122.
Sparks LE, Tichenor BA, Chang J, Guo Z. 1996. Gas-phase mass transfer model for predicting volatile organic compound (VOC) emission rates from indoor pollutant sources. Indoor Air(1). 6:31-40.
Spencer JN, Holmboe ES, Kirshenbaum MR, Firth DW, Pinto PB. 1982. Solvent effects on the tautomeric equilibrium of 2,4-pentadione. Can J Chem. 60:1178-1182.
Stoner GD, Shimkin MB, Kniazeff AJ, Weisburger JH, Weisburger EK, Gori GB. 1973. Test for carcinogenicity of food additives and chemotherapeutic agents by the pulmonary tumor response in strain A mice. Cancer Res. 33(12):3069-3085.
Svendsen N, Pedersen SF, Hansen OC, Pedersen E, Bernth N. 2005. Survey and release of chemical substances in “slimy” toys [PDF, 892 KB]. [accessed 2017 May]. Denmark: Danish Ministry of Environment.
[TDS] Technical Data Sheet. 2015. Klenk’s Epoxy Thinner. Montreal (QC): Swing Paints. [last modified 2015 Aug 11; accessed 2017 Nov].
TERA. 2004. Report of the Peer Consultation Meeting on Methyl Ethyl Ketone. Submission by the VCCEP Task Group of the American Chemistry Council Ketones Panel for the Voluntary Children's Chemical Evaluation Program (VCCEP). Toxicology Excellence for Risk Assessment (TERA) Center, Cincinatti, Ohio.
TERA. 2017. Read across approach for the Methyl Iso Ketone group of chemicals. Toxicology Excellence for Risk Assessment (TERA) Center, Cincinatti, Ohio. Unpublished.
Tomicic C, Berode M, Oppliger A, Castella V, Leyvraz F, Praz-Christinaz SM, Danuser B. 2011. Sex differences in urinary levels of several biological indicators of exposure: a human volunteer study. Toxicol Lett. 202(3):218-225.
Toso B, Procida G, Stefanon B. 2002. Determination of volatile compounds in cows’ milk using headspace GC-MS. J Dairy Res. 69(4):569-577.
Tyl RW, Ballantyne B, Fisher LC, Tarasi DJ, Dodd DE. 1989. Dominant lethal assay of 2,4-pentanedione vapour in Fischer 344 rats. Toxicol Ind Health. 5(3):463-477.
Tyl RW, Ballantyne B, Pritts IM, Garman RH, Fisher LC, France KA, McNeil DJ. 1990. An evaluation of the developmental toxicity of 2, 4-pentanedione in the Fischer 344 rat by vapor exposure. Toxicol Ind Health. 6(3-4):461-474.
Tyl RW, France KA, Fisher LC, Pritts IM, Tyler TR, Phillips RD, Moran EJ. 1987. Developmental toxicity evaluation of inhaled methyl isobutyl ketone in Fischer 344 rats and CD-1 mice. Fundam Appl Toxicol. 8(3):310-327.
[US EPA] United States Environmental Protection Agency. 1988. Superfund Record of Decision: Kin-Buc Landfill, NJ (September 1988). EPA/ROD/R02-88/068.
[US EPA] United States Environmental Protection Agency. 1992. Screening procedures for estimating the air quality impact of stationary sources. Revised. Office of Air and Radiation, Office of Air Quality Planning and Standards. EPA-454/R-92-019.
[US EPA] United States Environmental Protection Agency. 1994. Methods for Derivation of Inhalation Reference Concentrations and Application of Inhalation Dosimetry. Research Triangle Park (NC): US EPA, Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, Office of Research and Development.
[US EPA] United States Environmental Protection Agency. 1999. Estimate of stack heights and exit gas velocities for TRI facilities in OPPT's Risk-Screening Environmental Indicators Model [PDF]. United States Environmental Protection Agency; Office of Pollution Prevention and Toxics; Economics, Exposure, and Technology Division.
[US EPA] United States Environmental Protection Agency. 2003a. Toxicological review of methyl ethyl ketone in support of summary information on the Integrated Risk Information System (IRIS) [PDF, 1.07 MB]. National Center for Environmental Assessment, Washington, DC. EPA 635/R-03/009. Bethesda (MD): US National Library of Medicine. [accessed 2017 Jan 4]
[US EPA] United States Environmental Protection Agency. 2003b. Toxicological review of methyl isobutyl ketone CAS NO. 108-10-1 in support of summary information on the Integrated Risk Information System (IRIS) [PDF, 586 KB]. National Center for Environmental Assessment, Washington, DC. EPA EPA/635/R-03/002. [accessed 2016 Aug 18].
[US EPA] United States Environmental Protection Agency. 2005. PARAMS model. Washington (DC): US Environmental Protection Agency. [last revised 2011 Aug 18; accessed Oct 2017].
[US EPA] United States Environmental Protection Agency. 2006. Inert reassessment – mesityl oxide (CAS Reg. No. 141-79-7) [PDF]. Washington (DC): US EPA, Office of Pollution Prevention and Toxics. [accessed 2020 January].
[US EPA] United States Environmental Protection Agency. 2009a. A scoping-level field monitoring study of synthetic turf fields and playgrounds. Washington (DC): US EPA, Office of Research and Development. EPA/600/R-09/135.
[US EPA] United States Environmental Protection Agency. 2009b. Risk Assessment Guidance for Superfund Volume I: Human Health Evaluation Manual (Part F, Supplemental Guidance for Inhalation Risk Assessment). Washington (DC): US EPA, Office of Superfund Remediation and Technology Innovation.
[US EPA] United States Environmental Protection Agency. 2010. High Production Volume Information System. Washington (DC): US EPA, Office of Pollution Prevention and Toxics. 2-Pentanone. Submission ID 24977107. [accessed 2020 Nov 17].
[US EPA] United States Environmental Protection Agency. 2011. Exposure factors handbook: 2011 edition. Washington (DC): Office of Research and Development.
[US EPA] United States Environmental Protection Agency. 2019. Consumer Exposure Model v2.1. Washington (DC): US EPA, Office of Pollution Prevention and Toxics.
[US EPA AcToR]. United States Environmental Protection Agency Aggregated Computational Toxicology Resource System. 2015. 2-Pentanone. CAS RN. 107-87-9. [accessed 1 Feb 2017].
[US EPA & CDC/ATSDR] United States Environmental Protectional Agency, Centers for Disease Control and Prevention/Agency for Toxic Substances & Disease Registry. 2019. Synthetic turf field recycled tire crumb rubber research under the Federal Research Action Plan: final report part 1 – tire crumb rubber characterization (volume 1) [PDF]. EPA/600/R-19/051.
Valero E, Villamiel M, Miralles B, Sanz J, Martinez-Castro I. 2001. Changes in flavour and volatile components during storage of whole and skimmed UHT milk. Food Chem. 72(1):51-58.
Van Engelen JGM, Park MVDZ, Janssen PJCM, Omen AG, Brandon EFA, Bouma K, Sips AJAM, Van Raaij MTM. 2008. Chemicals in toys. A general methodology for assessment of chemical safety of toys with a focus on elements [PDF]. RIVM report 320003001/2008.
Van Rooy FGBGJ, Rooyackers JM, Prokop M, Houba R, Smit LAM, Heederik DJJ. 2007. Bronchiolitis obliterans syndrome in chemical workers producing diacetyl for food flavorings. Am J Respir Crit Care Med. 176(5):498-504.
Versar. 1986. Standard scenarios for estimating exposure to chemical substances during use of consumer products. Vol. II. Consumer use of used motor oil. Springfield (VA): Versar, Inc. Prepared for US Environmental Protection Agency.
[VCCEP] Voluntary Children’s Chemical Evaluation Program. 2003. Methyl ethyl ketone (CAS No. 78-93-3). VCCEP Submission. Prepared by: American Chemistry Council’s Ketones Panel. [accessed 2016 November].
Wilkinson SC, Williams FM. 2001. In vitro dermal absorption of liquids. Contract Research Report 350/2001. Newcastle-upon-Tyne (UK): University of Newcastle, Department of Environmental and Occupational Medicine, Skin Research Group.
[WHO] World Health Organization. 1999a. Safety evaluation of certain food additives: aliphatic acyclic and alicyclic alpha-diketones and related alpha-hydroxyketones. Geneva (CH): World Health Organization, International Programme on Chemical Safety. (WHO Food Additive Series 42). Prepared by the Fifty-first meeting of the Joint FAO/WHO Expert Committee on Food Additives.
[WHO] World Health Organization. 1999b. Safety evaluation of certain food additives: saturated aliphatic acyclic secondary alcohols, ketones, and related saturated and unsaturated esters. Geneva (CH): World Health Organization, International Programme on Chemical Safety. (WHO Food Additive Series 42). Prepared by the Fifty-first meeting of the Joint FAO/WHO Expert Committee on Food Additives.
[WHO] World Health Organization. 2003. Safety evaluation of certain food additives: aliphatic secondary alcohols, ketones and related esters. Geneva (CH): World Health Organization, International Programme on Chemical Safety. (WHO Food Additive Series 50). Prepared by the Fifty-ninth meeting of the Joint FAO/WHO Expert Committee on Food Additives.
Won D, Lusztyk E. 2011. Data gathering on chemicals released to indoor air of residences from building materials and furnishings. Final report. Ottawa (ON): National Research Council Canada. 158 p. Report No.: B3332.2.
Won D, Yang, W. 2012. Material emission information from 105 building materials and consumer products. Final report. Ottawa (ON): National Research Council Canada. 164 p. Report No.: B3341.1.
Won D, Nong G, Yang W, Schleibinger H. 2013. Material emissions data: 52 building materials tested for 124 compounds. Ottawa (ON): National Research Council Canada. 300 p. Report No.: A1-000342.
Won D, Nong G, Yang W, Collins P. 2014. Material emissions testing: VOCs from wood, paint, and insulation materials. Ottawa (ON): National Research Council Canada. Report No.: A1-000342.2.
Won D, Nong G, Yang W, Lusztyk E. 2015. VOC emissions from evaporative sources in residential garages. Ottawa (ON): National Research Council Canada. 235 p. Contract No.: A1-000342.
Won D, Yang W, So S, Aubin D. 2019. Investigation of sources and indoor levels of CMP3 chemicals. Phase 1 - analysis of indoor air samples from 2 field studies. Ottawa (ON): National Research Council. Report A1-014282.1. Unpublished.
[WSDE] Washington State Department of Ecology [database]. 2020. Search results for CAS RN [78-93-3]. Lacey (WA): Department of Ecology State of Washington. [accessed 2016 Jul 4].
Yu H, Chen S. 2010. Identification of characteristic aroma-active compounds in steamed mangrove crab (Scylla serrata). Food Res Int. 43(8):2081-2086.
Yue J, Zheng Y, Liu Z, Deng Y, Jing Y, Luo Y, Yu W, Zhao Y. 2015. Characterization of volatile compounds in microfiltered pasteurized milk using solid-phase microextraction and GCxGC-TOFMS. Int J Food Prop. 18:2193-2212.
Zaccone EJ, Goldsmith WT, Shimko MJ, Wells JR, Schwegler-Berry D, Willard PA, Case SL, Thompson JA, Fedan JS. 2015. Diacetyl and 2,3-pentanedione exposure of human cultured airway epithelial cells: ion transport effects and metabolism of butter flavoring agents. Toxicol Appl Pharmacol. 289(3):542-549.
Zaccone EJ, Thompson JA, Ponnoth DS, Cumpston AM, Goldsmith WT, Jackson MC, Kashon ML, Frazer DG, Hubbs AF, Shimko MJ, et al. 2013. Popcorn flavoring effects on reactivity of rat airways in vivo and in vitro. J Toxicol Environ Health A. 76(11):669-689.
Zakhari S, Levy P, Liebowitz M, Aviado DM. 1977. Acute oral, intraperitoneal, and inhalation toxicity of methyl isobutyl ketone in the mouse. In: L. Goldberg, editor. Isopropanol and ketones in the environment. Cleveland (OH): CRC Press. p. 101-104 (Chapter 11).
Zhou S, Barnes I, Zhu T, Bejan I, Albu M, Benter T. 2008. Atmospheric Chemistry of acetylacetone. Environ Sci Technol. 42:7905-7910.
Zhu J, Cao X-L, Beauchamp R. 2001. Determination of 2-butoxyethanol emissions from selected consumer products and its application in assessment of inhalation exposure associated with cleaning tasks. Environ Int. 26(7-8):589-597.
Zhu J, Newhook R, Marro L, Chan CC. 2005. Selected volatile organic compounds in residential air in the city of Ottawa, Canada. Environ Sci Technol. 39(11):3964-3971.
Zhu J, Wong SL, Cakmak S. 2013. Nationally representative levels of selected volatile organic compounds in Canadian residential indoor air: population-based survey. Environ Sci Technol. 47(23):13276-13283.
Appendices
Appendix A. Read-across tables
| Chemical name | 2-Butanol | MEK | MPK |
|---|---|---|---|
| Role | Analogue | Target chemical | Target chemical |
| CAS RN# | 78-92-2 | 78-93-3 | 107-87-9 |
| Chemical structure |  MW: 74.12 g/mol Log Kow: 0.61 |
 MW: 72.11 g/mol Log Kow: 0.29 |
 MW: 86.13 g/mol Log Kow: 0.91 |
| Toxicokinetics & metabolism | Rapidly converted to MEK | Rapidly absorbed via oral, inhalation, and dermal routes. Reduced to 2-butanol, which is rapidly oxidized back to MEK (US EPA 2003a). | Absorption expected to be similarly rapid and extensive relative to MEK and 2-butanol. |
| Acute toxicity | N/A | Oral LD50 2,737 mg/kg bw (rat), 4,044 mg/kg bw (mice) Inhalation LC50 >23,600 mg/m3 (rats, mice, guinea pigs) Neurobehavioural LOAEC = 4,800 mg/m3 (4 hours, mice) |
Oral LD50 = >1,600 to <3,200 mg/kg bw (rat) Inhalation LC50 >25mg/L Dermal LD50 = 8,000 mg/kg bw Neurobehavioural LOAEC = 3,500 mg/m3 (4 hours, mice) |
| Short-term inhalation studies | N/A | Neurobehavioural LOAECs = 300 to 600 mg/m3 (humans, baboons), 4,800 mg/m3 (rats) | NA |
| Repeated-dose toxicity (oral) | NOAEL = 1,771 mg/kg bw/day (8-week, female rat) | Neurotoxicity (non-acute effects) NOAEL = 173 mg/kg bw/day (rats) NOAEL = 1,723 mg/kg bw/day (read-across from 2-butanola) |
NOAEL = 454 mg/kg bw/day (13-month, male rats) |
| Repeated-dose toxicity (inhalation) | N/A | NOAEC = 14,750 mg/m3 (90-day, rats) | NOAEC = 5,300 mg/m3 (13-week, rat) Neurobehavioural LOAEC = 5,000 mg/m3 (up to 51 days, rat) |
| Repeat-dose toxicity (dermal) | NA | NA | NOAEL = 454 mg/kg bw/day (from MPK oral study) |
| Reproductive and/or developmental toxicity (oral) | Reproductive toxicity (limited)b NOAEL = 5,089 (males), 4,571 (females) mg/kg bw/day (rats) Developmental and maternal NOAEL = 1,771 mg/kg bw/day Developmental LED05 = 657 mg/kg bw/day (multigeneration 8-weeks, rats) |
Developmental toxicity LED05 = 639 mg/kg bw/day (read-across from 2-butanola) | LED05 = 763 mg/kg bw/day (read-across from 2-butanola) |
| Reproductive and/or developmental toxicity (inhalation) | N/A | Developmental toxicity LEC10 = 5,202 mg/m3 (intermittent exposure, mice) | Reproductive toxicity NOAEC = 5,000 mg/m3 NOAECdev = 5,000 mg/m3 and 5,300 mg/m3 |
| Reproductive and/or developmental toxicity (dermal) | N/A | LED05 = 639 mg/kg bw/day (read-across from 2-butanol oral studya) | LED05 = 763 mg/kg bw/day (read-across from 2-butanol oral studya) |
| Genotoxicity | Not genotoxic | Negative in vitro and in vivo assays | Negative in vitro assays, negative (Q)SAR model predictions for structural alerts |
| Carcinogenicity | N/A | Negative/ inconclusive (EPA 2003). | Negative (Q)SAR model predictions for carcinogenicity |
Abbreviations: NA, not available; N/A, not applicable; Log Kow, octanol-water partition coefficient; MW, molecular weight; LC50, the concentration of a substance that is estimated to be lethal to 50% of the test organisms; LD50, the dose of a substance that is estimated to be lethal to 50% of the test organisms; LEC10, a lower limit on an effective concentration using a 10% response level; LED05, lowest effective dose (95% lower confidence limit on the effective dose [ED]); LOAEC, lowest observed adverse effect concentration; LOAEL, lowest observed adverse effect level; LOEL, lowest observed effect level; NOAEC, no observed adverse effect concentration; NOAEL, no observed adverse effect level.
a Molar adjustment calculated from read-across value. Molecular weights are 74.12 g/mol for 2-butanol, 72.11 g/mol for MEK, and 86.13 g/mol for MPK.
b As noted by the US EPA (2003a), this study did not include the evaluation of certain parameters routinely measured in studies of more current design. Deficiencies included the lack of measurements of estrous cyclicity, sperm parameters, weights of uterus, epididymides, seminal vesicles, and brain; and less than complete clinical chemistry/hematology and histopathology.
| Chemical name | MIBK | MIAK | DAA |
|---|---|---|---|
| Role | Target chemical | Target chemical | Target chemical |
| CAS RN# | 108-10-1 | 110-12-3 | 123-42-2 |
| Chemical structure |  MW = 100.16 g/mol |
 MW = 114.18 g/mol |
 MW = 116.16 g/mol |
| Acute toxicity Oral | Rat LD50 = 2,080 to 4,570 mg/kg bw Mice LD50 = 1,900 to 2,850 mg/kg bw | Rat LD50 = 5,657 mg/kg bw | Rat LD50 = 3,002 mg/kg bw |
| Acute toxicity inhalation | Rat LC50 = 8,200 to 16,;400 mg/m3 Mice LC50 (1.25h) = 20,500 to 74,200 mg/m3 Juvenile baboons LOAEC = 200 mg/m3 (1- to 7-day exposure), CNS depression Human volunteers LOAEC = 200 mg/m3 (2-hour exposure), CNS depression |
Rat LC50 (6h)= 17,806 mg/m³ |
Rat LC50 (4h) = 7,600 mg/m3 |
| Acute toxicity Dermal | Rabbit LD50 = >20,000 mg/kg bw | NA | Rat LD50 (24h) >1,875 mg/kg bw |
| Short-term (14-day) inhalation | NOAEC = 410 mg/m³ (increased relative kidney weight and hyaline droplet-related tubular nephrosis) | NOAEC = 1,775 mg/m3 (maternal toxicity as CNS depression in developmental toxicity study) | NOAEC = 968 mg/m3 (from oral 15-day developmental study) |
| Short-term dermal (14-day) | NA | NOEL = 137 mg/kg bw/day (from inhalation developmental study) | NOEL = 1,000 mg/kg bw/day (from oral 15-day developmental study) |
| Repeated-dose toxicity (oral) | LOEL/NOAEL = 250 mg/kg bw/day (13-week; hepatic and renal effects at 1,000 mg/kg -bw/day) LOAEL = 101 mg/kg bw/day (2-year; chronic nephropathy in females) |
NOEL = 52 mg/kg bw/day (from inhalation NOAEC) | NOAEL = 100 mg/kg bw/day (45-day; kidney changes and altered hematological and blood chemistry) |
| Repeated-dose toxicity (inhalation) | LOAEC = 410 mg/m³ (increased incidence in renal tubule hyperplasia and chronic nephropathy [female] and mineralization of renal papilla [male]) LOAEC = 1,843 mg/m3 (2-year; chronic nephropathy in females) |
NOAEC = 934 mg/m3 (96-day; increased liver and kidney weight and hepatocytic hypertrophy in liver and tubular epithelium regeneration in kidneys in both males and females at 4,670 mg/m3) | NOAEC = 1,035 mg/m3 (6-week; liver weight changes without histopathological alterations at 4,685 mg/m3) |
| Repeated-dose toxicity (dermal) | LOAEL = 300 mg/kg bw/day (4-month; lowest dose tested; morphological changes in several tissues) LOEL/NOAEL = 250 mg/kg bw/day (from MIBK oral) |
NOEL = 52 mg/kg bw/day (from inhalation NOAEC) | NOAEL = 100 mg/kg bw/day (from oral NOAEL) |
| Developmental and/or reproductive toxicity (oral) | NOAEL = 259 mg/kg bw/day (read-across from DAA) | NOAECdev = 271 mg/m3 (RTR from inhalation NOAEC) NOAECmat= 137 mg/m3 (from inhalation NOAEC) |
NOAELdev = 300 mg/kg bw/day (reproductive and developmental effects at 1,000 mg/kg bw/day NOAELmat = 100 mg/kg bw/day (kidney changes and altered hematological and blood chemistry) |
| Developmental and/or reproductive toxicity (inhalation) | NOAECdev/mat of 4,106 mg/m3 (based on transient decreased body weight and food consumption, changes to reproductive organ weights in parents, and acute CNS depressive effects in pups) | NOAECmat= 1,775 mg/m3 (based on CNS depression at 3,505 mg/m3) NOAECmat= 3,505 mg/m3 (based on decreased body weight and reduced food consumption at 7,005 mg/m3) |
NOAECdev = 968 mg/m3 (from oral 45-day NOAEL) NOAECdev/mat = 3,182 mg/m3 (from oral study GD6 to 20) |
| Developmental and/or reproductive toxicity (dermal) | NOAEL = 259 mg/kg bw/day (read-across from DAA) | NOAECdev = 271 mg/m3 (from inhalation NOAEC) NOAECmat= 137 mg/m3 (from inhalation NOAEC) |
NOAELdev = 300 mg/kg/day NOAELmat = 100 mg/kg/day |
| Genetic toxicity | Negative | Negative | Negative |
| Carcinogenicity | Positive | Possible | Possible |
Abbreviations: MW, molecular weight; NA, not available; Dev, developmental toxicity; LC50, the concentration of a substance that is estimated to be lethal to 50% of the test organisms; LD50, the dose of a substance that is estimated to be lethal to 50% of the test organisms; LOAEC, lowest observed adverse effect concentration; LOAEL, lowest observed adverse effect level; LOEL, lowest observed effect level; NOAEC, no observed adverse effect concentration; Mat, maternal toxicity; NOAEL, no observed adverse effect level; NOEL, no observed effect level.
| Chemical name | Diacetyl | Acetoin | 2,3-PD |
|---|---|---|---|
| Role | Target chemical | Target chemical | Target chemical |
| CAS RN# | 431-03-8 | 513-86-0 | 600-14-6 |
| Chemical structure |  MW = 86.09 g/mol |
 MW = 88.10 g/mol |
 MW = 100.12 g/mol |
| Acute toxicity (lethality) | Oral LD50 = 3,000 mg/kg in rats | Oral LD50 >5,000 mg/kg | Oral LD50 = 3,000 mg/kg in rats |
| Short-term toxicity | LOAEC = 92.9 ppm (333 mg/m3) as a single 15-minute pulse | NA | NOAEC = 112 ppm (459 mg/m3) (respiratory and olfactory cytotoxicity). |
| Repeated-dose toxicity (inhalation, <2 weeks) | LOAEC = 100 ppm (358 mg/m3) the lowest test concentration in rats. LOAEC = 50 ppm (176 mg/m3) (read across from 2,3-PD) | NA | LOAEC = 50 ppm (205 mg/m3) (histopathological changes in rats and mice, 10 to 12 days). |
| Sub-chronic toxicity (90-day oral) | NOAEL = 90 mg/kg bw/day | NOAEL= 330 mg/kg bw/day (drinking water) (decreased body weight and anemia) | NOAEL = 105 mg/kg bw/day (read-across from diacetyl) NOAEL = 24 mg/kg bw/day (from non-respiratory tract effects in 90-day inhalation study) |
| Sub-chronic toxicity (90-day inhalation) | NOAEC = 12.5 ppm (45 mg/m3) | NOAEC = 800 ppm (2,883 mg/m3), the highest test concentration (NTP 2017) | NOAEC = 12.5 ppm (51 mg/m3) (histopathological effects in respiratory system in rats and mice; 14 weeks) |
| Sub-chronic toxicity (dermal) | NOAEL = 90 mg/kg bw/day (from oral exposure) | NOAEL = 330 mg/kg bw/day (extrapolation from oral) | NOAEL = 105 mg/kg bw/day (read-across from diacetyl) |
| Chronic toxicity (inhalation) | BMCL10= 30.23 mg/m3 based on squamous cell papilloma and carcinoma (adjusted for continuous exposure) | NA | NA |
| Developmental toxicity (oral) | NOAEL = 1,600 mg/kg bw/day | NOAEL = 1,640 mg/kg bw/day (read-across from diacetyl) | NA |
| Genetic toxicity | Likely to be genotoxic | Possible due to its similarity to diacetyl | Likely to be genotoxic (read-across from diacetyl) |
| Carcinogenicity | Some evidence for carcinogenicity in chronic animal studies | Possible due to its similarity to diacetyl | Some evidence for carcinogenicity in chronic animal studies (read-across from diacetyl) |
Abbreviations: NA, not available; MW, molecular weight; BMCL10, a lower one-sided 95% confidence limit for the benchmark concentration (BMCL) predicted to result in a 10% incidence of tumours; LD50, the dose of a substance that is estimated to be lethal to 50% of the test organisms; LOAEC, lowest observed-adverse-effect concentration; LOAEL, lowest-observed-adverse-effect level; NOAEC; no-observed-adverse-effect concentration; NOAEL, no-observed-adverse-effect level.
| Chemical name | MO | 6-methyl-5-heptene-2-one (MHE) |
|---|---|---|
| Role | Target chemical | Analogue |
| CAS RN# | 141-79-7 | 110-93-0 |
| Chemical structure |  MW = 98.1 g/mol Log Kow: 1.2 to 1.7 |
 MW = 126.2 g/mol Log Kow: 2.06 |
| Repeated-dose toxicity (inhalation, 49-day exposure) | LOAEC = 124 mg/m3 (reductions in body weight and body weight gain, and histopathology in nasal passage); no NOAEC was identified. | NA |
| Repeated-dose toxicity (inhalation, 6 weeks) | NOAEC = 200 mg/m3 (histopathological effects in kidney, lung, and liver) | NA |
| Repeated-dose toxicity (oral) | NOAEL = 15.0 mg/kg bw/day (route-to-route extrapolation from 6-week inhalation studya) NOAEL = 39 mg/kg bw/day (read-across from MHE by adjusting the molecular weight) |
NOAEL = 50 mg/kg bw/day (increases in platelet counts in female rats in a 90-day study) LOAEL = 200 mg/kg bw/day (OECD 2003) |
| Developmental toxicity (inhalation) | No developmental toxicity was noted up to 1,211 mg/m3 | NA |
| Developmental toxicity (oral) | No developmental toxicity was noted up to 95.2 mg/kg bw/day (route-to-route extrapolation from inhalation studya) NOAEL = 165 mg/kg bw/day (read-across from MHE by adjusting the molecular weight) |
NOAEL = 200 mg/kg bw/day (maternal and prenatal developmental toxicity in a prenatal developmental toxicity study in rats) LOAEL = 1,000 mg/kg bw/day. No fetal malformations up to 1,000 mg/kg bw/day (OECD 2003) |
| Reproductive toxicity (inhalation) | NOAEC = 413 mg/ m3 (decreased number of litters at 1211 mg/ m3) | NA |
| Reproductive toxicity (oral) | NOAEL = 32.5 mg/kg bw/day (route-to-route extrapolation from combined repeated-dose and reproductive/developmental toxicity study) NOAEL = 165 mg/kg bw/day (read-across from MHE by adjusting the molecular weight) |
NOAEL = 200 mg/kg bw/day (testicular toxicity in male rats in 90-day study) LOAEL= 1,000 mg/kg bw/day No effects in female rats (OECD 2003) |
| Genetic toxicity | Negative | Negative |
| Carcinogenicity | NA | NA |
Abbreviations: NA, not available; MW, molecular weight; Log Kow, octanol-water partition coefficient, LOAEC, lowest observed adverse effect concentration; LOAEL, lowest observed adverse effect level; NOAEC; no observed adverse effect concentration; NOAEL, no observed adverse effect level.
a This concentration was converted to oral dose using parameters from Health Canada (1994), with a rat average body weight of 0.35 kg and an inhalation rate of 0.11 m3/day, that is, 200 mg/m3 Í 0.11 m3/day Í 8h/24h Í 5 days/7 days / 0.35 kg = 15.0 mg/kg bw/d for repeated-dose toxicity; or 413 mg/m3Í 0.11 m3/day Í 6h/24h / 0.35 kg = 32.5 mg/kg bw/d for reproductive toxicity.
Appendix B. Parameters for exposures near facilities
The SCREEN3 model (SCREEN3 1996) was used to estimate ambient air concentrations near facilities that reported releases of MEK and MIBK to air. It was based on the Industrial Source Complex (ISC) model (for assessing pollutant concentrations from various sources in an industry complex). SCREEN3 is designed to estimate maximum concentrations of chemicals at chosen receptor heights and at various distances from a release source for a given continuous emission event. The maximum calculated exposure concentration is selected using a built-in meteorological data matrix of different combinations of meteorological conditions, including wind speed, turbulence, and humidity. The driver for air dispersion in the SCREEN3 model is wind. This model directly predicts concentrations resulting from point, area, and volume source releases.
The table below outlines the input parameters for the SCREEN3 model used to estimate ambient air concentrations of MEK from releases to air by selected reporting facilities in 2016.
| Variables | Input value | Input value |
|---|---|---|
| Source release typea | Area | Point |
| Effective emission area(m2)b | 95 550 | NR |
| Emission rate (g/s•m2)c | 1.06 x 10-5 | NR |
| Source release height (m) | 10 | NR |
| Stack height (m)d | NR | 9.14 |
| Stack inside diameterb (m) | NR | 0.5 |
| Emission rate (g/s)c | NR | 4.02 |
| Stack gas exit velocityd (m/s) | NR | 10.44 |
| Stack gas exit temperature (K)e | NR | 293 |
| Receptor height above ground (m)f | 1.74 | 1.74 |
| Variable wind adjustment factorg | 0.4 (daily average), 0.2 (yearly average) | 0.4 (daily average), 0.2 (yearly average) |
| Urban/rural optionb | Urban | Urban |
| Type of meteorology | Full | Full |
| Minimum and maximum distance (m) | 0 to 500 | 0 to 200 |
Abbreviations: NR, not a required input for the source release type selected
a Release type was selected on the basis of data from on-site releases in the NPRI
b Professional judgment and based on aerial photo analysis
c Estimates based on quantities reported by facilities under the NPRI, assuming continuous release (NPRI 2021)
d US EPA (1999)
e Regulatory default from the SCREEN3 model
f Curry et al. (1993)
g US EPA (1992) and professional judgment
The table below outlines the input parameters for the SCREEN3 model used to estimate ambient air concentrations of MIBK from releases to air by selected reporting facilities in 2015.
| Variables | Input value | Input value |
|---|---|---|
| Source release typea | Area | Point |
| Effective emission area(m2)b | 592,500 | NR |
| Emission rate (g/s•m2)c | 5.45 x 10-8 | NR |
| Source release height (m) | 10 | NR |
| Stack height (m)d | NR | 18.59 |
| Stack inside diameterb (m) | NR | 0.5 |
| Emission rate (g/s)c | NR | 0.03 |
| Stack gas exit velocityd (m/s) | NR | 11.89 |
| Stack gas exit temperature (K)e | NR | 293 |
| Receptor height above ground (m)f | 1.74 | 1.74 |
| Variable wind adjustment factorg | 0.4 (daily average), 0.2 (yearly average) | 0.4 (daily average), 0.2 (yearly average) |
| Urban/rural optionb | Urban | Urban |
| Type of meteorology | Full | Full |
| Minimum and maximum distance (m) | 0 to 700 | 0 to 700 |
Abbreviations: NR, not a required input for the source release type selected
a Release type selected on the basis of data from on-site releases in the NPRI
b Professional judgment and based on aerial photo analysis
c Estimates based on quantities reported by facilities under the NPRI, assuming continuous release (NPRI 2021)
d US EPA (1999)
e Regulatory default from the SCREEN3 model
f Curry et al. (1993)
g US EPA (1992) and professional judgment
Appendix C. Ketones in food
| Substance name | US per capita intake (µg/day) | US per capita intake (µg/kg bw/day) | Europe per capita intake (µg/day) | Europe per capita intake (µg/kg bw/day) |
|---|---|---|---|---|
| MEK | 36 | 0.6 | 110 | 2 |
| MPK | 42 | 0.7 | 140 | 2 |
| MIBK | 2 | 0.03 | 7 | 0.12 |
| Diacetyl | 8,000 | 133 | 3,300 | 55 |
| 2,3-PD | 80 | 1 | 220 | 4 |
| Acetoin | 1,800 | 30 | 2,800 | 47 |
| MO | NA | NA | 0.40 | 0.0067 |
Abbreviations: NA, not applicable
Per capita intake estimates (µg/day) were derived by the JECFA using the maximized survey-derived daily intake (MSDI) approach, assuming that the reported annual production amount for the various substances in the US and Europe was consumed by just 10% of the population (“eaters only”), and that only 60% of the annual production amount was reported in the poundage surveys. A body weight of 60 kg was used to derive intake estimates in µg/kg bw/day (International Organization of the Flavor Industry 1995; US National Academy of Sciences 1989, both cited in WHO 1999b).
Quantitative exposure estimates for the natural occurrence of all 10 substances were derived using the consumption data for comparable food categories from the Canadian Community Health Survey (CCHS) 2.2 Food Consumption Table (Health Canada 2015a). A summary of the data identified for each of the substances on their concentrations in foods from the literature and from the Volatile Compounds in Food (VCF) database (Nijssen et al. 1963-2018) can be found in Table C-2, Table C-3, Table C-4, and Table C-5. Quantitative data were not available for all food items; therefore, maximum reported values were used for various food items to estimate potential high-end exposures to all substances from food for the general population of Canada. Estimated exposures for all 10 substances from their natural occurrence in food are presented in Table C-6.
| Food category | MEK (µg/g) | MPK (µg/g) |
|---|---|---|
| Beers and coolers | 0.06 | 0.02 |
| Spirits and liqueurs | 2.00 | 1.20 |
| Fruit juices | 0.20 | 0.10 |
| Coffee; powder items are reconstituted | NA | 4.70 |
| Tea, including iced tea | 8.00 | NA |
| Milks | NA | 0.10b |
| Creams | 0.07 | 0.05 |
| Cheeses | 67.10 | 14.71 |
| Yoghurts | 7.00 | 0.01 |
| Bananas | NA | 27.00 |
| Cherries | NA | 0.01 |
| Pears | 1.00 | 2.40 |
| Pineapple | NA | 0.01 |
| Plums and prunes | NA | 2.00 |
| Strawberries | NA | 3.10 |
| Other fruits (blueberries, dates, kiwis, fruit salads) | 0.02 | 3.30 |
| Vegetables, excluding potatoes | 6.00 | 20.00 |
| Potatoes, fried | 0.13 | 0.07 |
| Chicken, turkey and other birds | 0.10 | 0.23 |
| Pork, fresh and ham | 0.72c | NA |
| Fish | 0.60 | NA |
| Shellfish | NA | 0.12 |
| Eggs | 0.01 | NA |
| Nuts, seeds and peanut butter | 0.10 | 7.60 |
| Legumes | 0.05 | NA |
| Butter | 0.16 | 0.95 |
| Other fats and spreads | 1.51 | 0.03 |
| Sugars, syrups and preserves | 0.08 | 0.03 |
| Savory snacks | 0.13 | NA |
Abbreviations: NA, not available
a Data from the Volatile Compounds in Food (VCF) Database (Nijssen et al. 1963-2018), unless specified otherwise.
b Yue et al. 2015
c Sánchez-Peña et al. 2005
| Food category | MIBK (µg/g) | MIAK(µg/g) | DAA (µg/g) |
|---|---|---|---|
| Beers and coolers | 0.12 | NA | NA |
| Coffee; powder items are reconstituted | 6.50 | 0.50 | NA |
| Milks | 0.016b | NA | 0.00015c |
| Plums and prunes | 0.01 | NA | NA |
| Other fruits (blueberries, dates, kiwis, fruit salads) | 0.02 | 0.001 | 0.408 |
| Chicken, turkey and other birds | 0.0004 | NA | 0.07 |
| Eggs | 0.003 | NA | NA |
| Other fats and spreads | 0.38 | NA | NA |
| Sugars, syrups and preserves | NA | NA | 2.70 |
| Other ingredients for recipes (for example, spices, baking ingredients) | NA | NA | 28.50 |
Abbreviations: NA, not available
a Data from the Volatile Compounds in Food (VCF) Database (Nijssen et al. 1963-2018), unless specified otherwise.
b Yue et al. 2015
c Toso et al. 2002
| Food category | Diacetyl (µg/g) | 2,3-PD (µg/g) | Acetoin (µg/g) |
|---|---|---|---|
| Beers and coolers | 0.20 | 0.30 | 9.00 |
| Spirits and liqueurs | 9.77b | 0.60 | 335.00 |
| Wines | 4.10 | NA | 234.00l |
| Fruit juices | 3.15c | NA | 0.35 |
| Coffee; powder items are reconstituted | 58.70 | 39.60 | 4.90 |
| Tea, including iced tea | 1.00 | 2.00 | NA |
| Milks | 29.3d | 0.20j | NA |
| Creams | 30.00 | NA | NA |
| Cheeses | 4.20 | NA | 40.00 |
| Yoghurts | 43.00e | NA | 28.00 |
| Pasta, rice, cereal grains and flours | 0.19 | 0.25 | 0.75 |
| White breads | 0.92 | 0.14 | NA |
| Wholemeal breads | 1.52 | 0.14k | 1.22m |
| Other breads | 0.33 | 0.04 | NA |
| Apples | 0.40 | NA | 3.50 |
| Cherries | NA | NA | 4.70 |
| Pears | NA | NA | 0.11 |
| Strawberries | 0.20 | NA | 0.49 |
| Other fruits (blueberries, dates, kiwis, fruit salads) | 0.60f | NA | 22.63 |
| Vegetables, excluding potatoes | 0.79g | NA | 6.40 |
| Potatoes, fried | 0.31 | NA | NA |
| Beef | 23.00 | 6.50 | 8.40 |
| Veal | 23.00 | 6.50 | 8.40 |
| Chicken, turkey and other birds | 0.90 | NA | 2.40 |
| Livers and liver pates | 27.81 | NA | NA |
| Luncheon meats, canned and cold cuts | 27.81 | NA | NA |
| Pork, fresh and ham | 0.36 | NA | NA |
| Fish | 0.0017 | 0.69 | 0.31 |
| Shellfish | 0.10h | NA | 0.16 |
| Nuts, seeds and peanut butter | 0.09 | 0.23 | NA |
| Butter | 21.00e | 0.05e | 2.00 |
| Margarines, tub | NA | 0.01 | NA |
| Margarines, block | NA | 0.01 | NA |
| Other fats and spreads | 0.60 | 0.01 | 0.04 |
| Confectionary, chocolate bars | 0.40 | NA | 17.00 |
| Sugars, syrups and preserves | 2.60 | NA | 26.00 |
| Savory snacks | NA | 0.20 | NA |
| Soups without vegetables | 0.00056i | 0.42i | NA |
| Gravies | 0.09 | NA | NA |
| Seasonings, salt, pepper, vinegar | 197.00 | NA | 1020 |
| Other ingredients for recipes (for example, spices, baking ingredients) | 238.00 | 109.00 | 951.00 |
Abbreviations: NA, not available
a Data from the Volatile Compounds in Food (VCF) Database (Nijssen et al. 1963-2018), unless specified otherwise.
b Cardoso et al. 2003
c Lawson et al. 1995
d De Leonardis et al. 2013
e Macciola et al. 2008
f Mujic et al. 2014
g Annan et al. 2005
h Yu and Chen 2010
i Giri et al. 2010
j Imhof et al. 1995
k Rychlik and Grosch 1996
l Garcia-Martinez et al. 2013
m Birch et al. 2013
| Food category | 2,4-PD (µg/g) | MO (µg/g) |
|---|---|---|
| Wines | NA | 0.00005 |
| Fruit juices | 0.01 | NA |
| Other fruits (blueberries, dates, kiwis, fruit salads) | NA | 0.01 |
| Vegetables, excluding potatoes | NA | 0.04 |
| Chicken, turkey and other birds | 0.004 | NA |
| Other fats and spreads | NA | 3.00 |
| Other ingredients for recipes (for example, spices, baking ingredients) | NA | 2.40 |
Abbreviations: NA, not available
a Data from the Volatile Compounds in Food (VCF) Database (Nijssen et al. 1963-2018), unless specified otherwise.
| Substance | 0 to 6 monthsa | 6 to 12 months | 1 year | 2 to 3 years | 4 to 8 years | 9 to 13 years | 14 to 18 years | 19+ years |
|---|---|---|---|---|---|---|---|---|
| MEK | 0 | 112 | 185 | 174 | 130 | 82 | 66 | 67 |
| MPK | 0 | 146 | 216 | 192 | 137 | 86 | 68 | 98 |
| MIBK | 0 | 0.5 | 0.9 | 0.7 | 0.4 | 0.8 | 5 | 32 |
| MIAK | 0 | 0 | 0.001 | 0.001 | 0.0004 | 0.04 | 0.4 | 3 |
| DAA | 0 | 0.04 | 3 | 4 | 4 | 3 | 2 | 2 |
| Diacetyl | 0 | 996 | 1,625 | 1,118 | 671 | 370 | 281 | 445 |
| 2,3-PD | 0 | 6.5 | 22 | 22 | 20 | 16 | 40 | 208 |
| Acetoin | 0 | 173 | 346 | 369 | 293 | 189 | 161 | 308 |
| 2,4-PD | 0 | 0.04 | 0.1 | 0.1 | 0.08 | 0.04 | 0.03 | 0.02 |
| MO | 0 | 0.3 | 1 | 2 | 1 | 0.9 | 0.7 | 0.6 |
a Infants 0 to 6 months old are assumed to be exclusively human milk-fed or formula-fed.
Appendix D. Parameters used to estimate human exposures from the use of products available to consumers
Cosmetic exposures were estimated using ConsExpo Web (2016). Exposure estimates were calculated using default body weights of 70.9 kg (20 years and older), 59.4 kg (12 to 19 years old), 31 kg (5 to 11 years old), 15.5 kg (6 months to 4 years old), and 7.5 kg (0 to 6 months old) (Health Canada 1998). The estimated inhalation and dermal exposure parameters for cosmetics are described in Table D-1. Dermal intakes are only presented for DAA and 2,4-PD. An inhalation rate of 16.2 mg/m3 was used to calculate the internal dose for the DIY products for DAA (Health Canada 1998). Unless otherwise specified, the defaults come from the relevant ConsExpo Fact Sheet for the scenario presented.
| Product (substance) | Assumptionsa |
|---|---|
| Top coat (assumed to be applied on finger- and toenails) (MEK) | Concentration of MEK: 55.7% (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017 and November 2019; unreferenced) Inhalation – Exposure to vapour, evaporation model, Exposure duration: 18 minutesProduct amount: 0.33 g (Ficheux et al. 2014) Room volume: 1 m3 (close to the face) Ventilation rate: 1 change per hour Mass transfer coefficient: 6.401 m/hr (Sparks method) Release area mode: constant Release area: 26.2 cm2 (based on data from Ficheux et al. 2014 and assuming that both finger- and toenails are painted) Molecular weight matrix: 124 g/mol |
| Nail polish/nail adhesive (two coats on finger- and toenails) (MEK, MPK, MIBK) | Concentration: MEK = 94.7%, MPK = 1% to 3%, MIBK 1% to 3% (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017, and November 2019; unreferenced) Inhalation – Exposure to vapour, evaporation modelExposure duration: 35 minutes Product amount: 0.8 g for 20 years and older and teens, and 0.27 g for 6 months to 4 years old (Ficheux et al. 2014) Room volume: 1 m3 (close to the face) Ventilation rate: 1 change per hour Mass transfer coefficient: MEK: 6.401 m/hr (20 years and older), 9.082 m/hr (6 months to 4 years old) (Sparks method); MPK: 10 m/hr; MIBK: 10 m/hr Release area mode: constant Release area: 26.2 cm2 (12 years and older), 8.8 cm2 (6 months to 4 years old) (based on data from Ficheux et al. 2014 and assuming that both finger- and toenails are painted) Molecular weight matrix: 124 g/mol |
| Nail polish (two coats on finger- and toenails) (DAA) | Concentration of DAA: 10% (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017 and November 2019; unreferenced) Inhalation – Exposure to vapour, evaporation model Amount on the skin (g/use): 0.16 (12 years and older), 0.06 (6 months to 4 years) (Ficheux et al. 2014) |
| Nail polish remover (MEK, MIBK) | Concentration of MEK: 84% (20 years and older), 79.6% (6 months to 4 years); MIBK (10% to 30%) (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017 and November 2019; unreferenced) Inhalation – Exposure to vapour, evaporation modelExposure duration: 8 minutes Product amount: 5.36 g (12 years and older), 1.82 g (6 months to 4 years) Room volume: 1 m3 (close to the face) Ventilation rate: 1 change per hour Mass transfer coefficient: MEK: 5.080 m/hr (20 years and older), 7.209 m/hr (6 months to 4 years) (Sparks method); MIBK: 10 m/hr (ConsExpo default) Release area mode: constant Release area: 34 cm2 (12 years and older), 25 cm2 (6 months to 4 years) (based on data from Ficheux et al. 2014 and assuming that both finger- and toenails are painted) Molecular weight matrix: 75 g/mol |
| Face moisturizer (diacetyl) | Concentration: Diacetyl: 0.1% (personal communication, email from the CHPSD, HC, to the ESRAB, HC, April 2020; unreferenced) Inhalation – Exposure to vapour, evaporation model (RIVM)Frequency: 2/day (20 years and older), 1/day (5 to 11 years) Exposure duration: 12 hours (20 years and older), 24 hours (5 to 11 years) Product amount: 1.5 g (20 years and older), 1.1 g (5 to 11 years) Molecular weight matrix: 1000 g/mol Room volume: 20 m3 Ventilation rate: 0.6 change per hour Mass transfer coefficient: 10 m/hr Release area mode: constant Release area: 638 cm2 (20 years and older), 605 cm2 (5 to 11 years) Emission duration: 12 hours (20 years and older ), 24 hours (5 to 11 years) |
| Body moisturizer (diacetyl) | Concentration: Diacetyl: 0.04% to 0.1% (given that no concentration data were available for body moisturizers, it is presumed that diacetyl is present at the same levels as in face moisturizers) Inhalation – Exposure to vapour, evaporation modelFrequency: 1 Exposure duration: 20 minutes (time in bathroom) Product amount: 10 g (12 years and older) (Ficheux et al. 2016) Molecular weight matrix: 1000 g/mol Room volume: 10 m3 Ventilation rate: 2 changes per hour Mass transfer coefficient: 10 m/hr Release area mode: constant Release area: 17 530 cm2 (20 years and older) Emission duration: 20 minutes |
| Rubbing alcohol (MIBK) | Concentration of MIBK: 0.98% (personal communication, email from the NNHPD, HC, to the ESRAB, HC, August 16, 2016; unreferenced) No scenario identified in ConsExpo, professional judgment was used. Inhalation – Exposure to vapour, instantaneous release model Surface area: it is assumed that an area of 5 cm x 5 cm = 25 cm2 is covered Amount on the skin (g/use): product amount of hand sanitizer 0.7 g (Health Canada 2015b) is adjusted for 910 cm2 surface area to an area of 25 cm2 = 0.02 g |
| Temporary tattoos (MIBK) | Concentration of MIBK: 1% to 3% (personal communication, email from the CHPSD, HC, to the ESRAB, HC, November 2019; unreferenced) Inhalation – Exposure to vapour, instantaneous releaseExposure duration: 5 minutes Product amount: 0.0036 g (professional judgment based on Scott and Moore 2000, and personal communication, email from the CHPSD, HC to the ESRAB, HC, December 2019; unreferenced Room volume: 1 m3 Ventilation rate: 1 change per hour |
| Eyeliner stickers (DAA) | Concentration of DAA: 30% (personal communication, emails from the CHPSD, HC, to the ESRAB, HC, August 2016, April 2017 and November 2019; unreferenced) Dermal: Exposure duration: 12 hours Product amount: 27 mg/use (Loretz et al. 2005) Room volume: 1 m3 (close to the face) Mass transfer coefficient: 6.844 m/hr (Sparks method) Release area mode: constant Release area: 5 cm2 (professional judgment) Ventilation rate: 1 change per hour Molecular weight matrix: 124 g/mol |
a Unless specified, a retention factor of 1 was used.
| Product (substance) | Assumptionsa |
|---|---|
| Mouthing pacifier and/or teether | Concentration of MEK residue: 500 ug/g (WSDE 2020) Oral: Etoy = qproduct × ftime × fsurface / bw where, Etoy = exposure from mouthing toy (µg/kg bw per day) Pacifier: qproduct = amount of substance that leaches from product over 24 hours (µg) = 22,500 µg (500 ug/g × 45 g)b ftime = fraction of the day that the product is mouthed (that is, sucking time) = 0.2 (infants) and 0.32 (toddler) (Juberg et al. 2001 as cited in EFSA 2015) fsurface = fraction of the product surface that is mouthed = 0.5 (Lassen et al. 2011 as cited in EFSA 2015) Teether: qproduct = amount of substance that leaches from product over 24 hours (µg) = 68,000 µg (500 ug/g × 136 g)b ftime = fraction of the day that the product is mouthed (that is, sucking time) = 0.02 (Juberg et al. 2001 as cited in EFSA 2015) to 0.05 (Van Engelen et al. 2008) (infants and toddler) fsurface = fraction of the product surface that is mouthed = 0.5 (RIVM 2002) |
a Approach from EFSA (2015), body weights of 7.5 kg and 15.5 kg for infants (0 to 6 months old) and toddlers (6 months to 4 years old), respectively (Health Canada 1998).
b Assuming that all of the MEK (500 ug/g) could be released. Weight of pacifier and teether from examining product weights based on product labels.
Other products
Sentinel exposure scenarios were used to estimate the potential exposure to substances in the Ketones Group. Exposures were estimated on the basis of the assumed weight, 70.9 kg of an adult (Health Canada 1998), and use behaviours of an adult (20 years and older). Exposures were estimated using ConsExpo Web (ConsExpo 2016) or algorithms (see below for more details). Scenario-specific assumptions are provided in Table D-3. The PARAMs model was used to estimate mass transfer coefficients (Sparks method) (US EPA 2005). Refer to Table D-4 for the defaults used in the PARAMs model.
| Exposure scenario | Assumptions |
|---|---|
| Paint or coating remover or stripper (for example, lacquer remover) (MEK) | Concentration of MEK: 20% to 40% (SDS 2018a) Scenario: paint remover in DIY Fact Sheet (RIVM 2007a). Assumes that work would be done in a garage. Inhalation – Exposure to vapour, evaporation model |
| Adhesive remover, degreaser (MEK) | Concentration of MEK: 100% (SDS 2019a) Scenario: adhesive remover used in the house, based on DIY Fact Sheet (RIVM 2007a) and US EPA 2011 defaults. Inhalation – Exposure to vapour, evaporation model |
| Paint or coating thinners (dilutes lacquer or other coating) (MEK) | Concentration: MEK = 100% (3% diluted in product) (SDS 2019a) Scenario: general coating (floor) in Do-It-Yourself Fact Sheet (RIVM 2007a). Assumes that work is done in a garage. The final concentration of MEK in the product was based on product information stating that lacquers should be diluted with pure MEK at no more than 4 oz per gallon (3%) Inhalation – Exposure to vapour, evaporation model |
| Liquid paint (solvent-rich paint for truck bed) (MEK, MIBK) | Concentration: MEK = 10% to 20% (SDS 2021), MIBK = 10% to 15% (SDS 2021) Scenario: Brush or roller paint with solvent-rich paint (truck bed paint) in Paint Fact Sheet (RIVM 2007b). Assumes that work is done in a garage. Inhalation – Exposure to vapour, evaporation model Ventilation rate: 1.5 changes per hour |
| Liquid paint for steel (high-solid paint) (MPK) | Concentration of MPK: 1% to 10% (ECCC 2017b) Scenario: Brush or roller paint with high-solid paint in Paint Fact Sheet (RIVM 2007b). Assumes that work is done in a garage. Inhalation – Exposure to vapour, evaporation model |
| Spray products (for example, spray paint) (MEK, MIBK, DAA) | Concentration: MEK = 1% to 75% (SDS 2016a, 2019b), MIBK = 0.4% to 25% (SDS 2017a, 2020), DAA = 1% to 5% (SDS 2012a) Scenario: spray can scenario from Paint Fact Sheet (RIVM 2007b), but the exposure to vapour–evaporation model was used since substances are volatile. Assumes that work is done in a garage. Some aerosol spray cans are ~400 g in size; therefore, the product amount is adjusted to 400 g, and the application and exposure duration are increased by 5 minutes each. Inhalation – Exposure to vapour, evaporation model Dermal: |
| PVC cement/primer (MEK) | Model: ConsExpo exposure to vapour—evaporation, using defaults from the universal wood glue in a bottle scenario, where relevant (RIVM 2007a) Concentration of MEK:
Room volume: 20 m3 (RIVM 2007a) Ventilation rate: 0.6 changes per hour (RIVM 2007a) Molecular weight matrix: 3,000 g/mol (RIVM 2007a) Product amount for:
Emission duration for:
Release area:
|
| Multi-purpose adhesives (MEK) | Concentration of MEK: 3% to 100% (SDS 2019c, 2014a) Scenario: tube glue (contact glue) in the Do-It-Yourself Fact Sheet (RIVM 2007a) Inhalation – Exposure to vapour, evaporation model |
| Water repellent/protectant (for example, windshield cleaner/protectant spray) (MEK) | Concentration of MEK = 10% to 25% (SDS 2016b) Scenario: Glass cleaner spray in Cleaning Products Fact Sheet (RIVM 2007a) Inhalation – Exposure to vapour, instantaneous release model |
| Wood lacquer (MIBK) | Concentration: MIBK = 2.5% to 10% (SDS 2016c) Scenario: general coating on a floor (in a garage) in the DIY Fact Sheet (RIVM 2007a) Inhalation – Exposure to vapour, evaporation model |
| Filler/putty from tube (automotive) (MIBK) | Concentration of MIBK: 1% to 5% (SDS 2018c) Model: filler/putty from tube (in a garage) in the DIY Fact Sheet (RIVM 2007a). Assumes that work is done in a garage. Inhalation – Exposure to vapour, evaporation model |
| Automotive coating repair pen | Concentration of MIAK: 3% to 7% in an automotive coating repair pen (SDS 2015c) Inhalation – Exposure to vapour, evaporation model |
| Automotive choke cleaner (MEK, DAA) | Concentration: MEK = 1%–5% (SDS 2019d), DAA = 1% to 30% (SDS 2011, 2012b) Scenario: spray paint (in a garage) in Paint Fact Sheet (RIVM 2007b). Assumes that work is done in a garage. Inhalation – Exposure to vapour, evaporation model Dermal – Direct product contact |
| Pipe thread sealant (DAA) | Concentration: 20% to 30% (SDS 2013) Scenario: universal/wood glue in a bottle as per DIY Fact Sheet (RIVM 2007a) Inhalation – Exposure to vapour, evaporation model Dermal – Direct product contact |
| Paint/marker remover, spray (DAA) | Concentration of DAA: 7% (SDS 2018d) Model: spray paint (in unknown room) in Paint Fact Sheet (RIVM 2007b). Inhalation – Exposure to vapour, evaporation model Dermal – Direct product contact |
| Paint thinner (for epoxy paints) (DAA) | Concentration: DAA = 5% to 10% (SDS 2017b) Scenario: epoxy thinner, to clean brushes or surfaces. Assumes that this could be done where epoxy is being used in the home (unspecified room) Inhalation – Exposure to vapour, evaporation model Dermal – instant application |
| Markers (MIBK, DAA) | Concentration: MIBK = 10% to 30% in dry erase markers (SDS 2015b), DAA = 60% to 100% in permanent markers (SDS 2008, 2012c, 2014b) Scenario: markers (dry erase or permanent) Inhalation: used scenario from Children’s Products Fact Sheet (RIVM 2002) – for MIBK only Dermal or oral (ink scenario from the Arts & Creative Materials Institute [ACMI] approach; personal communication from the ACMI to the ESRAB, HC, 2009; unreferenced) Acute or per event scenario: Intake (mg/kg bw/event = [Concentration of substance in marker (w/w) × estimated amount of ink per exposure (50 mg) × fraction absorbed] / body weight (kg) Amount of ink per exposure = 50 mg (Hansen et al. 2008) Chronic scenario: Intake (mg/kg bw/day): [(Concentration of substance in marker (w/w) × ink laydown rate (µg/cm) × 25 cm ink line/day)/1000 µg/mg] / body weight (kg) Ink laydown rate = 100 µg/cm (90th percentile level for ink laydown of writing instruments) (personal communication, from the Duke Medical Centre to the ESRAB, HC, 2009; unreferenced) |
| Essential oil as air freshener (inhalation) (2,3-PD) | Concentration of 2,3-PD: 0.1% to 1% (SDS 2019f) Scenario: essential oil as air freshener (in a living room) in Cosmetics Fact Sheet (RIVM 2006) Inhalation – Exposure to vapour, constant rate model |
| Plug-in air freshener (inhalation) (2,3-PD) | Concentration of 2,3-PD: 0.1% to 1% (SDS 2015d) Scenario: plug-in air freshener Inhalation – Exposure to vapour, instantaneous release scenario Frequency: all day, every day |
| Gel air freshener (inhalation) (2,3-PD) | Concentration of 2,3-PD: 1% to 5% (SDS 2015e) Scenario: gel air freshener Inhalation – Exposure to vapour, instantaneous release scenario Frequency: all day, every day |
| Parameter | Value | Additional Information |
|---|---|---|
| Density of air (g/cm3) | 0.0011774 | At 25 degrees Celsius, atmospheric pressure of 760 mmHg, and relative humidity of 50% |
| Viscosity of air (g/cm/s) | 1.86E-04 | At 25 degrees Celsius |
| Velocity of air (cm/s) | 10 | (Sparks et al. 1996; McGready and Fontaine 2010) |
| Diffusivity in air (cm2/s) | MEK: 9.19E-02 MPK: 8.22E-02 MIBK: 7.49E-02 MIAK: 6.92E-02 DAA: 7.24E-02 Diacetyl: 8.91E-02 2,3-PD: 8.02E-02 2,4-PD: 8.02E-02 |
At 25 degrees Celsius |
| Length of surface for various scenarios | Nail polish: 20 cm (20 years and older), 7 cm (6 months to 4 years) Nail polish remover: 40 cm (20 years and older), 14 cm (6 months to 4 years) Paint or coating remover or stripper (for example, lacquer remover): 2.5 m Adhesive remover, degreaser: 1 to 5 m Paint or coating thinners: 3 to 5 m Truck bed paint: 3 to 5 m Spray paint: 1 to 2 m Multi-purpose adhesive: 10 to 20 cm Wood finish (floor): 3 to 5 m Markers: 25 cm Graffiti remover (spray): 1 to 2 m Eyeliner stickers: 8 cm General coating: 3 to 5 m Filler/putty: 2 to 5 cm |
Values estimated by taking into account the release area listed in the ConsExpo Fact Sheets for each specific scenario (if scenario was not available in ConsExpo, professional judgment was used) |
Appendix E. Human inhalation exposures to diacetyl from microwave popcorn
The approach used in Zhu et al. (2001) and the Priority Substances List Assessment for 2-butoxyethanol (Environment Canada, Health Canada 2002) was used to convert the data from Rosati et al. (2007) on the emission of diacetyl from a bag of microwaved popcorn based on chamber studies to a concentration in air in a standard room. Details on the assumptions used are outlined below.
It was assumed that the emission of diacetyl from microwave popcorn follows first order decay and that there are no sink effects in the chamber. Given these assumptions, the concentration of the substance in an emission chamber (or a room) is related to the emission factor as follows:
Equation 1: C = [(EF0 × A) / (V × (N − k))] × [exp(-kt) − exp(-Nt)]
where:
C = the concentration of diacetyl in the chamber at any time [mg/m3]
EF0 = the initial emission factor for diacetyl from the product sample [mg/m2/h]
A = the emitting surface area of the product sample [m2]
N = the number of air changes per hour in the chamber [h-1]
V = the volume of the chamber [m3]
t = the duration of the emission [h]
and k = emission decay constant [h-1]
For a slowly depleting or nearly constant emission source, the decay constant k approaches zero. When the test time t becomes infinite or a steady-state equilibrium is reached, equation 1 can be rewritten as:
Equation 2: C = (EF0 × A) / (V × N)
It was assumed that steady-state equilibrium was achieved during product testing. For calculating the initial emission factors, equation 2 was rearranged to:
Equation 3: EF0 = (C × N × V) / A [mg/m2/h] = [mg/m3] [h-1][m3] / [m2]
where N is the number of air changes per hour in the chamber, V is the cell volume, and A is the emitting surface area, and the average concentrations of diacetyl emitted from the bag of microwaved popcorn is 778.9 µg/bag (Rosati et al. 2007).
Step 1: Calculate the initial emission factor using equation 3 (data from Rosati et al. 2007).
| Range of concentration (mg/m3) | N = ACH (h-1) | V = volume of chamber (m3) | A - emitting SAa (m2) | EF0 (mg/m2/h) |
|---|---|---|---|---|
| 5.8 | 2.3 | 0.515 | 0.1 | 68.70 |
| 0.02 | 2.3 | 0.515 | 0.1 | 0.237 |
a Assumes that the emitting surface area is the surface area of a popcorn bag, 1000 cm2 (Borkowski 2007).
Step 2: Calculate the emission decay constant using k = EF0*A/W, where W is the total evaporable amount of the substance in the source (W = average amount emitted from the microwaved popcorn bag, 0.779 mg).
| EF0 (mg/m2/h) | A (m2) | W (mg) | k (h-1) |
|---|---|---|---|
| 68.7 | 0.1 | 0.779 | 8.82 |
| 0.237 | 0.1 | 0.779 | 0.03 |
Step 3: Calculate the concentration in a standard room using equation 1.
| EF0 (mg/m2/h) | A (m2) | V (m3) | N (h-1) | k (h-1) | t (h) | Concentration (mg/m3) |
|---|---|---|---|---|---|---|
| 68.7 | 0.1 | 17.4 | 0.5 | 8.82 | 1 | 0.0288 |
| 0.237 | 0.1 | 17.4 | 0.5 | 8.82 | 1 | 0.0001 |
For 5.8 mg diacetyl/m3:
| Time (minutes) | Concentration (mg/m3) |
|---|---|
| 0 | 0 |
| 10 | 0.0328 |
| 20 | 0.0377 |
| 30 | 0.0364 |
| 40 | 0.0339 |
| 50 | 0.0313 |
| 60 | 0.0288 |
| 120 | 0.0175 |
| 180 | 0.0106 |
| 240 | 0.0064 |
| 300 | 0.0039 |
| 360 | 0.0024 |
| Average after 6 hours | 0.02 |
For 0.02 mg diacetyl/m3:
| Time (minutes) | Concentration (mg/m3) |
|---|---|
| 0 | 0 |
| 10 | 0.00011 |
| 20 | 0.00013 |
| 30 | 0.00013 |
| 40 | 0.00012 |
| 50 | 0.00011 |
| 60 | 0.00010 |
| 120 | 0.00006 |
| 180 | 0.00004 |
| 240 | 0.00002 |
| 300 | 0.00001 |
| 360 | 0.00001 |
| Average after 6 hours | 0.00007 |
Appendix F. Parameters used for route-to-route extrapolation
| Substance | Study parameters | Original dose | Converted dose | Formula |
|---|---|---|---|---|
| MIBK | 2-year; 6h/day; 5 days/week | 1843 mg/m3 | 101 mg/kg bw/day | 1843 × 0.31a × (6h/24h)b × (5d/7d)c |
| MIAK | 69 exposures spanning 96 calendar days; 6h/day; | 934 mg/m3 | 52 mg/kg bw/day | 934 × 0.31a × (6h/24h)b × (69d/96d)d |
| 2.3-PD | RA from diacetyl 90-day study | 90 mg/kg bw/day | 105 mg/kg bw/day | 90 × (100.12 / 86.09)e |
| 2.3-PD | 14-week; 6h/day; 5 days/week | 51 mg/m3 | 12 mg/kg bw/day | 51 × 1.33f × (6h/24h)b × (5d/7d)c |
| 2.3-PD | 14-week; 6h/day; 5 days/week | 102 mg/m3 | 24 mg/kg bw/day | 102 × 1.33f × (6h/24h)b × (5d/7d)c |
| 2.4-PD | 6h/day; GD6–15 | 827 mg/m3 | 64 mg/kg bw/day | 827 × 0.31a × (6h/24h)b |
| 2.4-PD | 6h/day; 5 days/week; 14-week | 417 mg/m3 | 23 mg/kg bw/day | 417 × 0.31a × (6h/24h)b × (5d/7d)c |
| MO | 6-week; 8h/day; 5 days/week | 200 mg/m3 | 15 mg/kg bw/day | 200 × 0.31a × (8h/24h)b × (5d/7d)c |
a Considering a standard rat body weight of 0.35 kg and a volume of inhalation of 0.11 m3/day, resulting in a constant of 0.31 m3/day kg (from Health Canada 1994).
b Exposure duration in a day
c Duration of the study
d Number of exposures
e Molecular weight (substance/read-across)
f Considering a standard mice body weight of 0.03 kg and a volume of inhalation of 0.04 m3/day, resulting in a constant of 1.33 m3/day kg (from Health Canada 1994).
