Final Report - Review of Health Canada's Actions in the Recall of Alysena- 28
September 13, 2013
Prepared for Health Canada by Risk Sciences International (RSI)
Contents
- 1. Introduction
- 2. Background
- 3. Events in the Recall of Alysena™ 28
- 3.1 Alysena™ 28 Packaging Problem and Company Actions
- 3.2. Health Canada Actions
- 3.3 Considerations Relevant to Health Canada's Recall Response
- 3.4 Social Response: Media and Social Concern
- 3.4.1 Lack of understanding of events in the Alysena™ 28 recall
- 3.4.2 Misunderstanding of industry and Health Canada responsibilities for drug recall
- 3.4.3 Misunderstanding of intent of health risk levels and disagreement with designation that unplanned pregnancy is not a high health risk
- 3.4.4 Divergent expectations of public regulation
- 4. Analysis and Recommendations
- 4.1 Key Elements Contributing to Circumstances of the Recall
- 4.2 Evaluation of Recall Events and Response
- 4.3 Review Comments on Health Canada's Handling of the Recall
- 4.4 Recommendations
- 4.4.1 Recommendation 1: Strengthen Health Canada's authority in responding to hazards of drug products
- 4.4.2 Recommendation 2: Improve clarity and communication of roles and responsibility concerning a drug recall
- 4.4.3 Recommendation 3: Include consideration of level of social concern
- 4.4.4 Recommendation 4: Improve capacity to respond in a more timely fashion
- 5. References
- Appendix 1: Chronology of Alysena™ 28 Recall
- Appendix 2: Apotex Recall Notices
- Appendix 3: London Drugs Blog Post
- Appendix 4: Health Canada Recall Notices
- Appendix 5: Media Reports
NOTE:
- The purpose of this report was to assess whether Health Canada followed its own processes and procedures during the Alysena 28 recall, and whether those processes were sufficient. RSI's review was based on written information from the Department, including documents outlining the responsibilities of drug companies and Health Canada with respect to recalls and notifications of recalls. RSI also conducted interviews with a wide range of Health Canada personnel involved in the Alysena 28 recall.
1. Introduction
On April 3, 2013, Apotex sent notices of a Type II recall of a single lot of Alysena™ 28, its new generic oral contraceptive, to its wholesalers and distributors. The recall was prompted by the discovery of one package of the drug in which the blister pack contained two rows of active pills and two rows of placebo pills, instead of three rows of active pills and one row of placebo pills: taking two weeks of placebo pills rather than the usual one placed women at an additional risk of pregnancy. Due to Apotex's initiation of the recall before its notification of Health Canada, the intervention of a weekend in Health Canada's review process and several other factors, it was April 8 when Health Canada posted a drug recall notice on its website, after media announcements not only of the recall of the drug but also of the lack of earlier public notification by the Department.
Driven by media coverage, some of which was based on incomplete information on the drug recall, considerable public concern arose about the efficacy of the drug sold to consumers, the number of women who might be at risk of an unplanned pregnancy, and of Health Canada's handling of the recall. While 5 adverse drug reactions were reported between January 14 and February 24, 2013, more than 40 were received between April 9 and May 31, 2013; twenty-three of these reported a suspected pregnancy while using Alysena™ 28. Much of the media coverage of the recall focused on the perceived delay by Health Canada in notifying consumers of the recall and of the risk of unplanned pregnancy posed by the recalled products. There was also criticism of the discrepancy between the type II recall initially set by the company and the type I that was later assigned by Health Canada for vulnerable groups who, for medical reasons, should not get pregnant; the apparent escalation in the risk level suggested confusion and delay in attending to the issue, and the initial type II recall that was perceived to have led to a delay in communication suggested a lack of sensitivity to the seriousness of an unplanned pregnancy. Criticism was voiced by the Canadian Pharmacists Association and the Society of Obstetricians and Gynecologists of Canada.
In response to the public concern and criticism of the Department's handling of the issue, the former Minister of Health, the Honourable Leona Aglukkaq, announced an investigation into recall of Alysena™ 28. As quoted in the Toronto Star, Friday April 12, Minister Aglukkaq stated the following as the concern that prompted the investigation:
"I am concerned that Canadians may not have received important information in a timely manner. As a result, I have instructed Health Canada to look into the issue and assess whether processes were followed and that they are sufficient."
Risk Sciences International (RSI) was engaged to conduct this review of the recall of Alysena™ 28 ; this document reports the results of that investigation. The review of the recall process was based on key documents describing the responsibilities of drug companies and Health Canada in recalls and notifications of recalls, as well as on interviews with most of the Health Canada personnel who were closely involved in the recall of Alysena™ 28. RSI interviewed six individuals in Health Products and Food Branch Inspectorate, Regions and Programs Branch, Ontario Region, Therapeutic Products Directorate, and the Communications and Public Affairs Branch. These individuals provided the chronology of the events in which they were involved, as well as their assessments of the factors that led to the public and media concern; however the interviewees are not named and comments are not attributed to any individual in this report.
Section 2 of the report provides some background to the process followed by Health Canada in the recall. It describes the legislative and regulatory structure that assigns responsibilities for the safety of drugs in Canada, and the policy structure governing the process for and handling of recalls of drugs and the guidance documents that set out the expectations of industry and Health Canada in recalls. It then outlines the roles and responsibilities of Health Canada personnel in determining the recall strategy and ensuring that it is carried out according to requirements and expectations, and the terms and performance expectations for the processes to be followed.
Section 3 describes the events of the recall as recounted by the interviewees involved in the recall, and supplemented by documentation.
Section 4 contains an analysis of the key events that led to the social concern and a preliminary assessment of the response by Health Canada. It also makes a set of recommendations to improve procedures and considerations of actions to avoid.
Attached to this report are four Appendices Appendix 1 is a detailed chronology compiled from the timelines provided by all interviewees; Appendix 2 contains the Apotex recall notices; Appendix 3 is the drugstore company blog that communicated the Apotex recall notice and Appendix 4 contains Health Canada public communications on the recall. Appendix 5 is a set of media stories that were retrieved by Health Canada.
2. Background
2.1 Legislation and Regulations
Drugs are regulated in Canada by Health Canada under the authorities and provisions set out in the Food and Drugs Act (FDA), specifically Part I Sections 3 and 8 through 15, which address general considerations and prohibitions; and the Food and Drug Regulations, Part C, which specify the conditions that drugs and their manufacture, packaging, labeling, advertising and sale in Canada must meet.
The FDA prohibits the sale of drugs with deceptive claims or advertising, that do not comply with labeling regulations, that do not meet prescribed standards for that drug or that are manufactured in an unlicensed facility. The FDA is the legislative authority for the Food and Drug Regulations which specify the requirements for the manufacture, composition, packaging, importation, advertising and sale of drugs that can be approved in Canada. The Regulations assign the primary responsibility for ensuring the safety and effectiveness of drugs to industry, including conducting a recall of a drug; Health Canada has no authority to order the recall of a drug that it judges as posing an unacceptable risk. Manufacturers and importers that decide to recall a drug are required to notify Health Canada; Section C.01.051 of the Regulations set out the requirements for industry notification of Health Canada of a drug recall and the information that must be provided:
Where a manufacturer who sells a drug in dosage form or a person who imports into and sells in Canada a drug in dosage form commences a recall of the drug, the manufacturer or importer shall forthwith submit to the Director the following information:
- (a) the proper name of the drug, the common name of the drug if there is no proper name, the brand name of the drug and the lot number;
- (b) in the case of an imported drug, the names of the manufacturer and importer;
- (c) the quantity of the drug manufactured or imported;
- (d) the quantity of the drug distributed;
- (e) the quantity of the drug remaining on the premises of the manufacturer or importer;
- (f) the reasons for initiating the recall; and
- (g) a description of any other action taken by the manufacturer or importer with respect to the recall.
2.2 Policy and Guidance Documents
The statement (from Section C.01.051, above) that the manufacturer or importer must notify Health Canada "forthwith" has been problematic, as it does not provide a specific timeframe to be met by industry and that can be enforced by the Health Products and Food Branch Inspectorate (HPFBI) of Health Canada.
HPFBI has therefore developed a set of policy documents that specify the interpretation of these Regulations and that constitute their expectations of industry parties in drug recall processes. Three main documents set out the policy with respect to drug recalls: these are the Health Products and Food Branch Inspectorate Recall Policy (POL-0016, 2006); HPFBI Drug Recall Procedure (SOP-0331, 1993 revised in 2005 and currently under revision), describing the responsibilities of HPFBI and Operational Centre personnel in recalls; and HPFBI Guidelines for the Recall of Drugs and Natural Health Products (GUI-0039) which give guidance to industry parties on their responsibilities in initiating and conducting a drug recall.
The requirement (Food and Drug Regulations C.01.051) that the company notify Health Canada of a recall "forthwith" is interpreted as meaning that the responsible parties - which for the purpose of a drug recall are the "legal agents/ manufacturers/ distributors" of the drug - are to notify the HPFB Inspectorate at the Operational Centre responsible for their province within 24 hours of having made the decision to recall, providing the information specified in the Regulations. This is to be followed by a written report within three business days of initiating the action, that provides the information necessary for Health Canada to assess the risk to health, as well as information on the recall strategy, with timelines, a copy of the firm's recall communication, effectiveness checks that the company has carried out, and the proposed action to prevent recurrence of the problem or any corrective actions already carried out.
It is important to note that these policy expectations, while elaborated for the purposes of the practical implementation of the Food and Drug Regulations, are not set out in the Regulations and thus do not have the force of law. There is no legal requirement for company action in drug recalls beyond the notification requirements of Section C.01.051, and Health Canada has no powers to enforce its more precise interpretation of these or any other recall expectations. Health Canada can impose a fine of $5,000 to a company that fails to notify the Department of a recall 'forthwith', but it is difficult to prove a violation of this imprecise requirement.
2.3 Roles and Responsibilities for Drug Recalls
2.3.1 Industry Responsibilities
The expectations of industry in drug recalls, and guidance on the information that is required, are set out in HPFBI Guidelines for the Recall of Drugs and Natural Health Products (GUI-0039). Responsible parties are expected to take full responsibility for recalls and effectiveness checks, notifying Health Canada within 24 hours of making the decision to recall a product, as set out in POL-0016, providing information on the recall, with a fuller written report within three business days accompanied by more information. Responsible parties are also expected to make progress reports to the Inspectorate when requested, and to undertake recalls when requested to do so by Health Canada although, as noted above, Health Canada cannot compel a company to do so.
There is no specified timeframe for the company to decide on a recall after it receives a complaint or becomes aware of a problem. Health Canada recognizes that companies need time to investigate and verify complaints, but expects that the investigation will be timely.
2.3.2 Health Canada Responsibilities and the General Recall Process
Health Canada is responsible for enforcing the Food and Drugs Act and Regulations, a function that includes monitoring drug recalls and assessing the effectiveness of a responsible party's actions in carrying out the recall. The objectives of HPFBI in drug recalls are, as set out in the Policy Statement in POL-0016:
- remove noncompliant or potentially harmful health products from distribution
- fulfill voluntary and mandatory commitments to International parties (including distribution of Rapid Alert Notifications on recalled drug products)
- verify that manufacturers, importers and distributors establish and implement documented procedures that will enable them to carry out effective and timely investigation of reported problems and recalls and that wholesalers implement documented procedures for recalls
- that the Inspectorate is informed when recalls are initiated as per the Act and the Regulations and is provided with information about the results of recalls and the actions taken to prevent the recurrence of problems
- manufacturers, importers and distributors maintain records of incidents, complaints, adverse reactions and of actions taken in response to those reports or incidents.
The two primary sections of Health Canada with responsibilities for prescription drug recalls (with the exception of biologics) are the Compliance and Enforcement Coordination Division (CECD), specifically the Drug Compliance and Verification Investigation Unit, in the HPFB Inspectorate, and the Regional Centres of the Regions and Programs Bureau (RAPB) across the country. Two other sections may be involved: the Therapeutic Products Directorate (TPD) may, at the request of the Region or CECD, conduct a Health Risk Assessment to assess and verify the risk type, and the Communications Branch drafts communications, in consultation with CECD and TPD, and manages the approval and delivery of public communications that are required.
Personnel in CECD and the Regional Centres cooperate on handling drug recalls. The general responsibility of the Regional Centres is gathering information on the recall, interacting with the company on the progress of the recall, and leading the monitoring of the recall, including investigations and follow-up such as verifying effectiveness checks. CECD's main role is coordinating the recall, coordinating the request for a Health Risk Assessment, interacting with domestic and international partners as well as coordinating national communications and recording the progress of the recall for informing Departmental officials. The Inspector in the Regional Centre and CECD share responsibilities for preparing and distributing the Rapid Alert Notification (RAN), which is a form that is used for the "reporting and transmission of information related to the recall of drug products to the National Recall Coordinator, [CECD], and international partners" (FRM-0016, Rapid Alert Notification).
Initial notification of a recall may come to either CECD or a Regional Centre, and both are responsible for providing early alerts on the possibility of a Type I or Type II recall that they become aware of. However the usual process is that the company will inform the Supervisor in the region in which its headquarters are located that it has decided to initiate a drug recall. Normal industry practice is to engage with Health Canada, through the Regional Supervisor, before its formal notification, enabling discussions and agreement on the risk type and the recall strategy and timeline. This is also an opportunity to discuss the need for public notices that should be issued by the company or Health Canada, or both.
In its notification of Health Canada of a recall, the company will provide basic information on the reason for the recall, the risk type, and the recall strategy. The Supervisor reviews the information and assigns it to an Inspector, noting any considerations of the company's risk level or recall strategy that may be relevant; the Inspector evaluates the information in the file, verifying that all the information noted in SOP-331 is present. He or she then assigns a preliminary risk classification, contacting the company for additional information that is required, notifies CECD of the recall and notes any relevant factors such as the possibility that a Health Risk Assessment may be required. The request for a health risk assessment should follow the procedures described in SOP-0537, and should be accompanied by the information set out in FRM-0019, Health Hazard Evaluation Request. CECD then assumes its recall coordination responsibilities, while the Regional Inspector continues to work with the company through the recall process.
2.3.3 Recall responsibilities of Health Canada offices
The Drug Recall Procedures (SOP-0331, 2005) outline more detailed duties of the Regional Centres and CECD for drug recalls. From SOP 331, the following are the basic responsibilities for CECD and the Regional Centre.
CECD
- Providing early alert on the possibility of a Type I or Type II recall to the supervisor or inspector in RAPB ON (or other) Region.
- Coordinating recall activities
- Requesting a Health Risk Assessment from the review Directorates
- Notifying Mutual Recognition Agreement or Pharmaceutical Inspection Cooperation Scheme member countries of drug recalls
- Reviewing foreign enforcement reports of foreign recalls of drugs distributed in Canada
- Coordinating distribution of the Rapid Alert Notification for HPFB
- Collaborating with review Directorates in coordinating, preparing and issuing national or regional communications
- Maintaining a chronological record of events and follow-up activities associated with each recall associated with a Type I risk in the CECD
- Coordinating national communications
- Acting as media spokespersons as required
- Initiating and conducting situational reviews
- Providing information to the Director, CECD for completion of Enforcement Activities Report
- Sharing recall information with provincial government agencies, healthcare professional licensing bodies and other special interest groups as required
Regional Centres
- Providing early alert of Type I or Type II recall to designated inspector in CECD
- Interacting with recalling firm
- Obtaining recall information, evaluating firm's recall strategy and risk type assignment, and monitoring the progress of the recall
- Assigning the initial risk Type to the health risk and hence the recall priority
- Requesting a Health Risk Assessment (SOP 298)
- Assessing the need for a recall effectiveness check; conducting effectiveness checks in accordance with guidelines
- Preparing the Rapid Alert Notification, acquiring Supervisor/Manager sign-off and submitting to CECD within designated timelines
- Implementing follow-up investigations including enforcement actions if necessary
- Maintaining a chronological record of events and follow-up activities associated with the recall
- Participating in situational review with CECD and other Directorates
- Collaborating with CECD, as required, in drafting national communications
- Collaborating with CECD, as required, in acting as media spokespersons
Therapeutic Products Directorate
The conduct of Health Risk Assessments (HRAs) in support of drug recalls is coordinated by the Office of Risk Management (ORM) in the Therapeutic Products Directorate (TPD), which functions as a liaison between HPFBI and the specialist review bureaux that carry out the HRA.
An HRA may be requested in a number of situations, including "health risk and /or its potential severity posed by the incident or product is unknown, unclear or requires scientific clarification" (SOP-0537). SOP-0537 describes the shared responsibilities of HPFBI and the Regional Centres for requesting an HRA, and includes a list of the information that is necessary for the HRA and should be provided along with the request. ORM likes to hold a telephone conversation within half a day of receiving a request for an HRA for a drug recall with the personnel involved in HPHBI, the Region, and the review bureau, in order to determine the nature of the issue, its urgency and the timeframe for response, and to identify any further information that is required.
TPD assesses risks as Type I, II or III according to the severity of harm and the likelihood that it will occur, in a risk typology that is harmonized internationally (the definitions of the risk types and the applicable timelines are discussed in sections 2.4.1 and 2.4.2, below). The potential for serious harm is the primary consideration when assigning a risk classification, more than consideration of the population exposed. When there is a higher risk to a subpopulation than the general population, or a risk to a vulnerable group, a dual risk type will be assigned, and the risk management strategy is driven by the higher risk level.
An HRA for a Type II risk would normally be completed in 48 hours (or the equivalent of two business days), though the `clock` does not start until all the necessary information is provided; a request for an HRA for a Type II risk that requires additional information will be sent back to HPFBI for the remaining information and would be handled in the timeframe of a Type II risk when the file is complete.
TPD may recommend that a public advisory be issued if notification of the public is considered necessary for risk mitigation, regardless of the risk type that is assigned.
Communications Strategists
Public communications on drug recalls are handled by a Communications Strategist in the Communications and Public Affairs Branch (CPAB) who is assigned to the Inspectorate, and is responsible for advising on communications approaches and dissemination vehicles, drafting the message and getting it approved, and issuing the message. There are two standard types of communication products that are used to provide information to the public on drug recalls; these have separate mastheads, but require the same approval process, are posted in the same place on Health Canada's Healthy Canadians website (healthycanadians.gc.ca) and are distributed through the same channels.
- Public advisory. This is used when public communications are necessary for Type 1 risks, and has a template for standard information content that enables readers and the media to pick up specific information. The text in the public advisory includes a statement of the risk of the drug being recalled.
- Information update. This uses a less formal structure that allows for a more explanatory narrative such as the details of the reasons for a drug recall, Health Canada actions, and information on follow-up activities. The risk level is not necessarily included in an information update.
Both of these communication products are issued to Canada newswire and posted in the recalls database, which is shown on healthycanadians.gc.ca, and on the Drugs and Health Products pages of the Health Canada website. Public notices are also put on RSS feed and twitter, referring followers and other subscribers to the Health Canada website. Notices are also posted on the MedEffect listserv to inform health care professionals; where information is mostly relevant to health professionals a specific type of communication may be issued through MedEffect.
The usual process for preparing and issuing a public communication on a drug recall is that Inspectorate staff contact the Communications Strategists to discuss the most appropriate vehicle to reach the intended audience, such as one of the public communication types or a more targeted vehicle such as a "notice to hospitals." Based on the recommendations of the Inspectorate or TPD the Strategist prepares the message and sends a draft to the program area for approval, usually within an hour.
A Standard Operating Procedure for the Production of Risk Communications by the Public Affairs, Consultation and Communication Branch of Health Canada (now titled the Communications and Public Affairs Branch, CPAB), which is still in draft form, describes the process and expectations for risk communications. The full process of issuing a risk communication, including approvals, revision, translation and formatting, generally takes at least 24 hours. A standard risk communication must receive all necessary approvals by 3 p.m. to be issued that day, or 1 p.m. on Fridays; past that time the risk communication will be issued the following day. However, exceptions may be made for urgent risk communications, at the request of the Assistant Deputy Minister of CPAB or of the originating branch. The Draft Standard Operating Procedure for the Production of Risk Communications sets out shortened approval timelines and other procedures for preparing and issuing an urgent risk communication, which may be produced in three or four hours.
Program areas may also identify a risk situation that requires a risk communication to be prepared or issued outside of normal weekday working hours or over a weekend. The draft risk communications SOP includes procedures and requirements of both the CPAB and programs branches for the preparation and issuing of risk communications on evenings and weekends, and risk communications have been often been issued on evenings and weekends.
Some basic information is required for the creation of a communications product, including the product involved, the reason for the recall, the level of risk and who is at risk. Additional information that is helpful includes lot numbers and other more detailed information. In all cases of high risk, clear language is used in both the headline and the body of the message, with a clear description of the potential severity of the outcome of the risk. In cases where the risk level is higher for a particular group or vulnerable population, plain language is used to ensure that it is understood who the vulnerable groups are and what they should do if they are at risk.
Communications Strategists are aware of the sensitivity of certain issues: the risk communications SOP includes a "Protocol for Addressing Low Risk/ High Interest Health Issues" that includes issues related to pediatrics, pregnant or breastfeeding women or other vulnerable populations. This Protocol describes the steps that are to be taken when an urgent risk communication is required for a low risk/ high interest situation, including a recall. A brief interim statement may be posted when insufficient information is available to alert the public of a high interest risk, using standard language advising that more information will follow; subsequent messages are linked to this to ensure that people can find updated information.
2.4 Risk Classifications and Performance Standards for Drug Recalls
2.4.1 Risk Classification
Human health risk is classified as Type I, Type II or Type III according to an assessment of the likelihood and severity of adverse consequences. Some performance standards for drug recalls are associated with the risk type assigned. As defined in HPFB documents (POL-0016; GUI-0039), risks are classified according to the following criteria:
Type I: a situation in which there is a reasonable probability that the use of, or exposure to, a product will cause serious adverse health consequences or death.
Type II: a situation in which the use of, or exposure to, a product may cause temporary adverse health consequences or where the possibility of serious adverse consequences is remote
Type III: a situation in which the use of, or exposure to, a product is not likely to cause any adverse health consequences.
2.4.2 Timelines
SOP-331 (HPFBI Drug Recall Procedure) specifies timelines for the submission of the Rapid Alert Notification. The Operational Centre is to send a completed Rapid Alert Notification to the assigned CECD Inspector within 24 hours for a Type I risk, or within 5 working days for a Type II risk.
Timelines for completion of an HRA request are noted in SOP-0537. For an HRA for drugs the standard is 24 hours for a Type I risk, and 48 hours, or two business days, for a type II risk. An HRA for a Type III risk should be completed within seven business days.
No timelines are assigned for other recall actions in SOP-331 or in POL-0016; however interviewees noted that the timeline for responding is 24 hours for Type I risk and 48 hours for a Type II risk; this applies to review and turn-around time at major steps, such as from the Regional Centre Inspector to CECD.
The Communications and Public Affairs Branch service standard for the issuance of risk communications is 24 to 48 hours for a Type I Advisory, and three to five working days for other risk communications products, from the point at which all necessary information has been supplied. The draft Risk Communication SOP contains detailed timelines for each of the processes involved in issuing a communication, including approvals, translation, formatting and distribution and posting, for both routine and urgent risk communications.
2.4.3 Recall Types and Actions
The recall strategy is determined by a number of factors, in addition to the risk type assigned to the drug problem; actions such as the level of distribution to which the recall applies, the timelines, communications and effectiveness checks are decided on the basis of a number of factors related to the use of the drug (Guidelines for recall of Drugs and natural Health Products, GUI-0039; Product Recall Procedures, 1993). For example the depth of recall, which may target the wholesale, retail, user or consumer levels, and the communications required are not tied to a risk level. This is because the setting in which a drug is used may mean that recalls and communication are not needed at some levels, regardless of the risk type. An injectable drug used only in hospitals that is involved in a Type I recall requires a recall at wholesale and hospital levels but not at the retail level (e.g. store pharmacy), and communication should be directed at those levels and not at the general public.
There are general guidelines for the conduct of drug recalls according to the risk type assigned. For recalls for a Type I risk, the Regional Centre is to verify that the firm is recalling to the level agreed on and that its recall strategy is adequate to achieve this, and should also verify the company's effectiveness checks according to the guidelines in Section 5.2.3.2 of SOP-0331. The Regional Centre also recommends the issuance of a Health Canada public notice if the risk is not being adequately managed by the firm. A notice may be issued for Type I risks regardless of company actions.
For recalls for Type II risks, the Regional Centre should verify that the recall strategy extends to the wholesale level at minimum, and that all wholesale customers have been contacted. It also decides whether inspectors should conduct their own effectiveness check, based on the firm's compliance history, the extent of distribution of the recalled drug, the nature of the hazard and the population at risk, amount of product distributed; and will conduct effectiveness checks if necessary. A public notice may be issued for a type II risk if it is considered necessary to help the public avoid the risk.
There is no specific procedure on the issuing of public communication for a recall. The 1993 Product Recall Procedures note that a Public Warning is usually reserved for more urgent situations (class I and sometimes class II recalls, in the terminology of the 1993 document) but is specified in more detail in the recall strategy for the particular recall situation. The draft revision of SOP-0331 directs that the Regional Centre recommend to CECD that a public notice be issued for Type I risk recalls "where applicable," without explaining the situations in which a public notice would be applicable or providing any advice on communications for Type II or III recalls. CECD is responsible for "coordinating national communications," but the communications noted include Dear Healthcare Professional Letters, Question Period Notes and communications with international agencies; Public Warnings or other alerts or risk communication products are not mentioned. GUI-0039 notes that HPFBI encourages firms to issue a news release "when the recall strategy requires a public advisory," but that in some circumstances it will issue a release in cooperation with the firm or may issue its own, "when it believes the public needs to be alerted about a serious risk."
The Drug Recall Procedure is in the process of revision, with some provisions changing from one version to the next. The policy aspects of the 1993 Product Recall Procedures were superseded by Policy 0016 in 2006, while there is a draft revision of the 2005 "procedure or work instruction' (SOP-0331) that contains additional revisions. However, the guidance elements for Health Canada remain in the 1993 document HPFBI Guidelines for Recall of Drugs and Natural Health Products, and in GUI-0039.
2.5 Other Agencies' Recall Procedures and Requirements
It is instructive to compare Health Canada's powers and responsibilities with respect to drug recalls to those of other agencies that are responsible for health and safety-related recalls. There are different provisions in the legislation governing the regulation of drugs, foods and consumer products, in both the specificity of industry responsibilities in voluntary recall situations, and in the power to order a recall.
The Canadian Food Inspection Agency (CFIA), an agency that reports to the Minister of Agriculture and Agri-food, is responsible for enforcing food safety legislation, and the Consumer Product Safety Directorate is part of the Healthy Environments and Consumer Safety Branch of Health Canada. Both of these organizations are responsible for responding to complaints about health and safety risks associated with the products under their jurisdiction, ensuring that the appropriate mitigating and correction actions are implemented according to the risk posed by the product involved.
The CFIA is responsible for enforcing Part B of the Food and Drugs Regulations, which pertain to food; this section does not mention product recalls, as Section C 01.051 does in relation to drugs. However, the CFIA is given specific authority to order recalls of foods in the legislation under which it was created. The Canadian Food Inspection Agency Act (Canada, 1997) gives the CFIA the power to order recalls, setting out both the conditions for a recall order and establishing a penalty of $50,000 for contravening an order. Section 19 (1) of the CFIA Act states:
Where the Minister believes on reasonable grounds that a product regulated under an Act or provision that the Agency enforces or administers by virtue of section 11 poses a risk to public, animal or plant health, the Minister may, by notice served on any person selling, marketing or distributing the product, order that the product be recalled or sent to a place designated by the Minister.
As the Food and Drugs Regulations do not mention recalls of food products there is no legislative guidance on the specific responsibilities of a company that issues a voluntary recall. The CFIA has thus developed policy guides on its expectations of industry (manufacturers, importers, distributors and retailers) when they identify a food safety problem, in which it states that these parties should "notify the CFIA immediately when [they] suspect that [their] firm has sold, distributed or imported a product that may pose a serious risk to consumers" (CFIA Food recall and emergency response). A list is provided of the information that the firm should provide to the CFIA when it notifies the regulator of the safety problem (CFIA).
The Consumer Product Safety Program, administered by Health Canada's Consumer Product Safety Directorate and Regional Offices across the country, is governed by new legislation that gives greater powers to the Department. The Canadian Consumer Product Safety Act (CCPSA) gives the Minister of Health the power to issue recall orders, ordering manufacturers, importers, sellers or advertisers to halt the manufacture, importation, packaging, storing, advertising selling, labelling, testing or transportation ("stop order").
The CCPSA also provides greater clarity on industry responsibilities for the safety of consumer products when a safety incident is reported. Under "duties" of industry "in the event of an incident" (CCPSA S. 14) the Act specifies that industry must inform the department of an incident or complaint about a product "within two days after the day on which they became aware of the incident." The company must then provide a written report with full information on the incident within 10 days of becoming aware of the incident.
3. Events in the Recall of Alysena™ 28
A detailed account of actions taken by each party involved is given as a chronology table, found in Appendix 2.
3.1 Alysena™ 28 Packaging Problem and Company Actions
Alysena™ 28 is a generic version of the oral contraceptive Alesse, manufactured by Pfizer. It is a new drug that was manufactured and packaged at a plant in Spain and imported by Apotex, and was first placed on the market in Canada in December, 2012.
The events described in this section occurred before Health Canada was notified of the problem and before any Health Canada actions on the recall. They have been reconstructed from information provided to Health Canada by Apotex.
3.1.1 Original pre-production problem
The packaging problem is believed to have originated in the drug product manufacturing and packaging plant in Spain. The packaging problem occurred during the production run of the first lot of Alysena™ 28 packages. This first lot was placed on the market in Canada at the beginning of December 2012.
3.1.2 Notification of faulty package
Apotex received a complaint of a faulty package on Wednesday, March 20 along with a photograph of the blister package containing the wrong number of placebo pills: the packaged product contained two rows of placebo tablets (14 tablets) and two rows of active contraceptive tablets (14 tablets) in the blister pack. Correct packaging is one row of placebo tablets (7 tablets) and three rows of active contraceptive tablets (21 tablets) in the blister pack. On Monday March 25 Apotex received the faulty sample and verified the defect, and on March 28 it made the decision to recall the lot from which the defective sample had come. This was the Thursday before the long Easter weekend. Accordingly, it was on Monday April 1 that the company prepared the information package, April 2 when it finalized the risk as type II, and Wednesday April 3 when it sent the notice of recall to its wholesalers, distributors and retailers.
The "urgent drug recall" was described as a "type II recall . . . conducted in collaboration with the Health Products and Food Branch Inspectorate Canada." (as discussed below, this statement was inaccurate when the recall notice was first issued).) The "reason for market action" was given as: "Product lot identified in this recall notification may contain two rows of the placebo (white) Tablets instead of one row of placebo (white) Tablets." The Health Assessment was stated as "Ingestion of only 14 tablets active instead of 21 of oral contraceptive would most likely result in reduced efficacy for contraception and therefore possibility of unplanned pregnancy cannot be ruled out" (Apotex April 03, 2013).
According to HPFBI Policy 0016 (Recall Policy), Apotex should have notified the Health Canada Ontario Regional Office verbally or in writing within 24 hours of deciding to recall the affected lot of the drug. That would have been Friday March 29, which was the Good Friday statutory holiday; the next business day for the federal government was Tuesday April 2 ( April 1, Easter Monday, was a federal government holiday) but the company did not notify Health Canada until April 4, the day after they initiated the recall with a notice to their consignees.
3.2. Health Canada Actions
3.2.1 Initial notification receipt and response
The Ontario Regional Office received the first notice of Apotex's recall of one lot of Alysena™ 28 on the afternoon of Thursday, April 4. The supervisor reviewed the file and assigned it to an inspector within 1 hour and 20 minutes, flagged with a note that the Type II risk level might require review and validation; due to the involvement of pregnant women and potentially infants, it was considered that the case may require a higher priority than is strictly the case with Type II risk recalls. The inspector in turn reviewed the file, and in approximately 75 minutes forwarded it to HPFB Inspectorate in Ottawa. The email was sent late in the afternoon and the HPFBI Ottawa specialist opened it on the morning of Friday April 5. The file was forwarded to the Therapeutics Product Directorate on April 5 with a request for a consult to verify the risk level assigned by the company, and a subsequent conversation between HPFBI and TPD confirmed that TPD was reviewing the file. The Bureau of Pharmaceutical Science (BPS) and the Bureau of Metabolism, Oncology and Reproductive Science (BMORS) conducted the preliminary opinion on the company's risk classification for the recall of Alysena™ 28. No formal HRA was ever requested or performed.
Late on the afternoon of Friday April 5 TPD phoned HPFBI to inform them that their preliminary investigation suggested that the risk assessment would be assessed as a Type II risk for the general population, but as a Type I risk for vulnerable populations, specifically those women who should not get pregnant for medical reasons or because they are taking other drugs that could harm the fetus. TPD indicated that although the written report stating the Type I risk would not be issued until the following Monday, they recommended that a public communication be issued that Friday to inform consumers of the risk. The communications team was not informed of the preliminary recommendation by TPD, and a communication was not drafted or issued that day; the matter was left until the final assessment was issued on the following business day, Monday April 8.
3.2.2 Early media interest and coverage
Even though neither Apotex nor Health Canada had issued a public notice about the recall, a drug store chain posted information about it on a blog on Saturday, April 6 (shown in Appendix 3); this was picked up by a major media company and reported overnight into April 7. The headline of the blog post was "Health Canada and Apotex Urgent Drug Recall"; the news network article was headlined "Health Canada recalls birth control pill Alysena™ 28" (Global News, April 7 2013; this was inaccurate as Health Canada had not known of the recall in order to participate, and does not order recalls; these points are discussed in detail in Sections 3.4 and 4.1, below.) The news report went on to say that the pharmacy company "has chosen to be proactive in contacting their patients who are currently taking the Alysena™ 28 birth control pill." Media coverage continued through the week of April 8 – 12 with the story carried by major media networks.
3.2.3 Health Canada active response phase
On the morning of Monday April 8, Health Canada actions were taking place on several fronts. In the Ontario Regional Office, the inspector was following up with the company, gathering the information required for a Rapid Alert Notification, and asking the company for assurance that no other lots had been affected by the packaging problem at the plant. Communication between the Inspector and the company was taking place on plans to make media announcements
Late in the morning of April 8 TPD provided the formal result of the consult on the risk level, in which it confirmed that the recall was a Type II for the general population and a Type I for vulnerable groups, identified as those women who were advised not to become pregnant for medical reasons or because they were taking other drugs that could harm a fetus. With the Type I designation there is a recommendation that a Public Advisory be issued (draft revised SOP-0331).
HPFBI Ottawa notified the communications staff responsible for the Inspectorate; however communications personnel were already receiving media inquiries on the recall, and realized that there was pressure to issue a public communication right away. While the first draft of a formal advisory may be returned to the program for review and approval within an hour, the service standard is 24 to 48 hours for an urgent Type I Advisory to allow for revisions, approvals and translation. With this in mind, the decision was made to post a recall notice on the Healthy Canadians website, and tweet a link to that site for the media and other followers. This notice was posted around noon, and included a note on the Type I/ Type II risk levels, but did not identify the vulnerable groups to whom the Type I designation applied. The apparent discrepancy between the company's Type II assessment and Health Canada's updated, hybrid Type I/ Type II created challenges for Communications personnel in explaining why Health Canada had changed the company's assessment.
3.2.4 Recall escalation and completion
By April 9 the company issued an updated recall advisory reflecting Health Canada's hybrid Type I / Type II risk level. In addition, as Apotex was unable to confirm the root cause of the faulty packaging, and could not confirm how many, if any, that additional faulty packages might have been used by a consumer in the previous months or may still be in a pharmacy's stock, or that other lots may not have been affected, it expanded the recall to more lots, which expanded the scope of the recall in Canada from 7 to 11 provinces and territories. Updated recall notices were issued by Apotex on April 12, and by Health Canada on April 12 (reissued on April 15), asking consumers to return any packages of the drug they had to their pharmacist. A greater number of adverse drug reaction reports began to be received on April 9, and continued through to the end of May, with 40 received in that period including 23 that claimed a pregnancy while on Alysena™ 28.
On April 11 public concern and media coverage had grown increasingly critical of Apotex and Health Canada, due to the combined initial reporting about the perceived delay in responding and the subsequent expansion of the recall, and Minister Aglukkaq called for an investigation into the issue. "Health Minister Leona Aglukkak ordered an investigation after the Toronto-based company recalled defective birth control pills but allegedly failed to take steps to notify the public until five days later... . Aglukkak said she's 'concerned' Canadians may not have received important information in a timely manner. "I have instructed Health Canada to look into whether processes were followed and that they are sufficient" she said" (Appendix 5 p. 29).
By the end of the week the drugs had been taken off the shelves. At that time no faulty packages were verified in addition to the single package that prompted the initial recall action, though one additional faulty package was found in the investigation that followed in June and July.
HPFBI Ottawa had requested inspection reports on the manufacturing plant from the Spanish authority (it received the report on April 30 stating that the authority was satisfied with the company's Corrective Actions and Preventive Actions), and it reviewed the report on the investigation conducted by the manufacturer in Spain. On April 19 Health Canada declared that it had no objection to Apotex's release of newly manufactured lots of Alysena™ 28, on the basis of the company's report of the 200% visual inspection (that is, visual inspection of each package by two people) conducted of those lots.
3.3 Considerations Relevant to Health Canada's Recall Response
There were several considerations that Health Canada staff members needed to weigh in making decisions on various aspects of the recall. One was the setting of the risk level. TPD's risk type assignment is driven largely by the health risk rather than the number of people exposed, and there was no doubt that Type II was appropriate for the general population or that for a vulnerable group the risk was Type I; as noted above, the involvement of pregnant women and infants prompted the priority attention given by Regional Centre staff to the recall notification when it was received as a Type II risk. There was some consideration, however, of the appropriate balance to be struck in responding to a Type I risk to a vulnerable population when only a single defective package had so far been found; this suggested an issue with a limited scope, rather than the national scope full recall that was initiated by the company.
Many who were involved in this recall recognized that matters of contraceptives and unplanned pregnancies are very sensitive issues, even if they are appropriately classified as Type II risks. That sensitivity has led, in the past, to the issuing of public communications for contraceptive products that are not even sold in Canada, through foreign product alerts or other mechanisms, in order to inform consumers who may have gotten prescriptions elsewhere. At several points in the process of this recall it was noted that problems with contraceptives are sensitive issues, and a public communication was recommended regardless of the risk type that was assigned. None of the recall policy or procedure documents are prescriptive about public communications for specific recall types, leaving open the ability to recommend that one be issued. Authority to issue a Health Canada public notice may be unclear; it has shifted as procedures have been revised, from the Assistant Deputy Minister in the 1993 Product Recall Procedures to HPFBI's Compliance Enforcement and Coordination Division (CECD) in the draft revision of the 2005 SOP-0331.
3.4 Social Response: Media and Social Concern
There was significant concern generated by the reports in the media of the packaging error that could leave women at risk of an unplanned pregnancy. The concern among women was evident in the increase in the number of Adverse Drug Reaction (ADR) reports, 40 between April 9 and May 31, compared with five between January 14 and February 24. Twenty-three of these reports stated a suspected pregnancy while the woman was taking Alysena™ 28. Some drug stores developed a "crisis plan" that included contacting patients and offering free pregnancy tests, and setting up a call centre to answer questions. A class-action lawsuit against Apotex was launched, involving 60 women from across Canada, "among whom there have been 40 unwanted pregnancies and four abortions" (Appendix 5 pp 4-6).
Much of the media attention was focussed on the perceived delay by the company and Health Canada in notifying Canadians of the recall. Many news stories contained statements or headlines that "Canadian women were left in the dark for about a week about pill's effectiveness." (Appendix 5, p 59). Health Canada Media Relations received approximately 20 media inquiries on the recall between April 8 and August 16; requests from reporters in the early phase of the recall (April 8 – 12) related to the timelines for the Alysena ™ 28 recall, Good Manufacturing Practices, and inspections on foreign sites.
The essence of the message conveyed in much of the coverage of the recall was that Health Canada is responsible for ordering recalls of drugs when it identifies a problem that could put the public at risk, that it had initiated the recall of Alysena™ 28 on April 3 but had neglected to inform the public for five days. There are several critical misunderstandings embedded in this assumption, which are discussed in turn below; these include the assumption that Health Canada is responsible for, and has the authority to, order drug recalls, that in this particular case it had initiated the recall, and that it had chosen to wait for five days before issuing a public notice of the recall.
The concern among women was largely driven by extensive media coverage of the recall, with most attention given to the possibility that the perceived delay in public notification could result in unplanned pregnancies for thousands of women across Canada who may have received a faulty package of the pills over the previous few months. Most of the criticism was directed at Health Canada, rather than Apotex, with the common suggestion that the public regulator had failed in its duty to protect women's health, either through a failure to follow established recall procedures or through a lack of consideration for the significance of unplanned pregnancies.
Review of a number of representative news stories carried by major media outlets (including 55 articles retrieved by Health Canada, included in Appendix 5) suggests that the content of some of the coverage was based on inaccurate understandings of underlying factors, including the responsibilities for drug recall initiation and conduct and the meaning and intent of the health risk classification, and of the particular circumstances of the recall of Alysena™ 28. Some comments also suggest differences in the expectations of the scope of responsibility given to risk regulators in Canada.
3.4.1 Lack of understanding of events in the Alysena™ 28 recall
Early media reports were prompted by the blog posted by the pharmacy chain on April 6, which included a copy of the initial Apotex recall notice of April 3. That information was limited to the basic facts of the packaging error and the affected lot; however it also stated that the recall was being conducted in collaboration with Health Canada, which was not the case as Health Canada was not notified of the recall until late on the following day, Thursday April 4. This inaccuracy was made more explicit in the initial news network coverage, which was published under headlines such as "Health Canada recalls birth control pill Alysena™ 28" (Appendix 5 p. 57) and may have contributed to the common misinterpretation that Health Canada had known about the problem and the planned recall but had neglected to inform the public.
There was confusion and imprecise implications about the actions for which Health Canada and Apotex were responsible for and had carried out. For example one article stated that "Although Health Canada was informed of the problem last week and sent a recall notice to pharmacies and other retailers, a notice to the general public didn't go out until Monday." (Appendix 5 p. 38) (In fact the recall notice to pharmacies and other retailers was the initial recall notice sent by Apotex). Another headline read "Health Canada waited five days before ordering retailers to notify patients" (Appendix 5 p. 61).
Information in news reports was inconsistent, and the coverage was variable in descriptions of the scope of the problem. Some reports (including the pharmacy blog of April 6 and subsequent network articles of April 7) noted that the recall was initiated by the company and was prompted by the finding of a single faulty package, and that the concern stemmed from the suspicion that other faulty packages may be still on the market. However other stories did not make that distinction and were ambiguous about the scale of the problem: a number of news stories stated that packages in the affected lot ‘could contain' an extra row of inactive placebo pills, while others stated that ‘blister packs in this faulty lot contained 14 pink pills containing active drug and 14 white placebos' (for example, Appendix 5 p. 41). A few news stories stated that a batch of 50,000 faulty packages had been distributed (Appendix 5 p. 44). A story about the class-action law suit implied that Health Canada had attributed the problem to all packages in the lot, saying that "Health Canada warned women that the product contained two weeks' worth of placebos instead of one." (Global News April 22 2013).
The concern about an emerging widespread problem was magnified by suggestions that many women may unknowingly have used faulty packages for several months, as they may have been unaware that they had received the generic drug Alysena™ 28 as a substitute for the brand name drug with which they were familiar. News stories focused on the deep concern felt by some women who were taking Alysena™ 28 to prevent pregnancy when they learned of the problem with the drug, often without noting a faulty blister pack would be identifiable as it contained two rows (rather than one) of white placebo pills and only two (rather than three) pink active pills. One news story was titled "some women may not realize they're using recalled birth-control pill," and observed that "many Canadian women may unknowingly be affected by a massive recall of birth control pills that could put them at risk for unplanned pregnancies", while another stated that "thousands of women across Canada have been prescribed a faulty batch of Alysena™ 28... women in seven provinces have been taking the placebo pills for up to four months, thinking they were protected from unwanted pregnancies" (thestar.com April 11, 2013).
The subsequent expansion of the recall to include more lots and more regions in Canada may have fed the interpretation that Health Canada was struggling to catch up in its understanding of the true magnitude of the problem with the drug problem, particularly when this was perceived as associated with the adjustment in the risk type. Since Health Canada was not able to communicate about the challenge in obtaining clear information on the lots that may have been affected by the error, this interpretation of the recall expansions may not have been avoidable. As the recall process progressed, some stories began to note that Apotex had expanded the recall to include more lots, citing Health Canada communications as the source of this information.
3.4.2 Misunderstanding of industry and Health Canada responsibilities for drug recall
Only a few news stories reviewed showed an awareness of the distribution of responsibilities and authorities in drug recalls (such as that in Appendix 5 p. 30); many more implied inaccurate assumptions of these responsibilities.
The primary misunderstanding, which perhaps drove much of the criticism of Health Canada, was that Health Canada orders drug recalls and makes the primary decisions on the recall strategy and communications. Many statements implied that Health Canada ordered the recall, had issued the initial recall notice, and decided the recall and communications strategies. Few references were found to the primary responsibility of industry to identify problems with the drugs they manufacture, import into or sell in Canada, to assign the risk level and take responsibility for the recall strategy, including determining the levels contacted as directly involved in recalling the product.
Most of the criticism about the delay in issuing a public notice was directed to Health Canada, in accordance with the expectation that the recall process is initiated and managed by the Department.
3.4.3 Misunderstanding of intent of health risk levels and disagreement with designation that unplanned pregnancy is not a high health risk
The second major focus of criticism related to the assignment of a Type II risk to the potential for unplanned pregnancies as a consequence of the drug problem, and the criticism again targeted Health Canada. Much of the coverage referred in general terms to the seriousness for a woman and for her family of an unplanned pregnancy, and the fact that women rely on their contraceptives when they make a responsible choice to prevent or delay pregnancy. There were frequent references to the initial assignment of a Type II risk, as for example "The risk of accidental pregnancy was not deemed serious enough to trigger an urgent product recall" (Appendix 5 p. 32.)
As the recall progressed some news coverage began to quote Health Canada statements on the recall, for example noting that "Apotex, the drug's distributor, decided, "based on their own assessment," to issue a Type 2 recall, which requires drugstores to remove the product from their stock (but not alert patients), according to a Health Canada statement" (Appendix 5 p. 63).
There were also a number of health professionals quoted who were familiar with the risk typology used, but disagreed that an unplanned pregnancy should be considered a Type II risk. A pharmacist was quoted (Appendix 5 p. 32) as saying "Although an unplanned pregnancy might not meet the strict definition for a Class I recall, the serious nature of the problem warrants immediate notification . . . It could be catastrophic for a lot of people if it's not planned. I think there is an urgency [to this situation] that needs to be out there."
The Society of Obstetricians and Gynecologists issued a similar statement, but focussed its criticism on Health Canada; it said the "problem "leaves the perception that contraception for women is not considered a priority health issue. . . one would hope there's more enlightenment at Health Canada." (Globe and Mail April 11 2013).
3.4.4 Divergent expectations of public regulation
Largely implicit, though occasionally made clearly, were expectations that as a public risk regulator Health Canada is expected to be a public watchdog, protecting the public from industry. Some commentary described inadequate oversight of industry actions, including the conduct of drug recalls, while others implied that Health Canada orders drug recalls and determines the recall strategy. Both suggest an expectation that public regulators have and should exercise greater regulatory control, including the power to order a recall. It also suggests the belief that industry does not have responsibility in this regard and perhaps cannot be trusted to carry out if it did.
One clear articulation of this opinion was made by the Society of Obstetricians and Gynecologists, and the College of Family Physicians in the Canadian Medical Association Journal on May 2; this essay was very critical of Health Canada for "accepting the manufacturer's voluntary recall without notifying the public", and referenced "gaps that exist in drug industry oversight." This article also expressed what was implied in several other general media articles, that the events of the recall "damages Canadians' confidence in drug regulation," a function widely assigned to public regulators.
The high level of concern with the Alysena™ 28 recall led to further, more general criticism of Health Canada's regulatory oversight. A series of articles reported that the U.S. Food and Drug Administration had repeatedly warned Apotex of "significant violations in its manufacturing and quality control" (Appendix 5 pp 7- 10) which had not prompted concern or action by Health Canada. The articles cited a "regulatory disparity" in the rigour of the Canadian and US drug regulatory systems and quoted a health policy researcher as saying that "we should not have to go to the Americans to find out what's going on in plants here in Canada" and that the problems identified "should have Canadians questioning the safety of the drug supply."
4. Analysis and Recommendations
4.1 Key Elements Contributing to Circumstances of the Recall
There are three main sets of factors that contributed to the unusual amount of media coverage of the Alysena™ 28 recall and of the public concern with it.
The first is that several key events in the progress of the recall were not standard, and complicated Health Canada efforts to complete the actions for which they are responsible. The second is that Health Canada missed several opportunities to adjust to the non-standard conditions and initiate an earlier response to address public concerns about the drug recall. The third is that some of the criticism of the handling of the recall appear to have been based on mistaken assumptions, primarily that Health Canada is able to order recalls and establish risk levels; that it had known about and collaborated on Apotex's recall, risk classification and recall strategy; and that it had then neglected to inform the public of the recall until five days later.
In more detail, several discrete events can be identified as contributing to the high levels of concern about the recall and the criticism of its handling.
- Health Canada was not notified of the recall until one day after it had been initiated by the company through a recall notice to its wholesalers and distributors; the recall was underway before Health Canada knew about it.
- The company exceeded the recall notification expectation set out in Policy 0016 (Health Canada should be notified within 24 hours of the decision to recall) by three business days (six if the long weekend is counted).
- The Food and Drugs Act and/or the Food and Drug Regulations do not give clear direction to companies on notification requirements of drug recalls.
- The Food and Drugs Act and/or the Food and Drug Regulations do not give Health Canada authority to enforce any notification standard.
- A delay in public notification of the recall, and of the risk of the packaging error with the drug, was caused by the separation by the weekend of the business days on which Health Canada's recall process was initiated and on which action was implemented.
- The initial Type II level did not trigger a sense of urgency in all the Health Canada personnel involved in the response in order to issue an urgent advisory on Friday afternoon.
- The initial Type II level did not trigger a sense of urgency in all the Health Canada personnel involved in the response in order to issue an urgent advisory on Friday afternoon.
- The social sensitivity of contraceptive drugs and the perceived seriousness of an unplanned pregnancy were not fully recognized by all involved personnel to warrant the issuing of an early Public Alert before the preliminary opinion on the company's risk assignment was finalized. While not a Type I risk under the definition in the risk classification system, an unplanned pregnancy is a serious matter for many women and their families, and public advisories and other communications have been issued for Type II risks when appropriate for the situation.
- There is little guidance in the drug recall procedures that prescribes or precludes the issuing of a public communication for a type II risk if the issue is sensitive, or that explains the features of a sensitive risk issue.
- The draft Risk Communications SOP gives comprehensive guidance on the conditions under which an urgent public advisory may be issued, and on the factors that constitute a risk of ‘high public interest'. However this guidance is prepared for the Communications and Public Affairs Branch and is not referenced in the drug recall procedures documents.
- A drug store chain posted a notice of the recall on a blog, which was picked up and reported by a national news network.
- the reporting of the issue by the drug store company was not anticipated and is not standard, but it shone a spotlight on the absence of Health Canada notification.
- The initial pharmacy company blog post and the subsequent first news report attributed the recall in part or entirely to Health Canada, and alluded to the lack of Health Canada notification by stating that the pharmacy "had chosen to be proactive in advising its customers and patients" of the recall.
- A revision in the risk type to a Type I for vulnerable groups from a general Type II assigned by the drug company, four days after the initial recall notice was sent to pharmacies, suggested that the risk was more serious than had been declared and that Health Canada had not been sufficiently vigilant with industry to ensure that it assessed the risk appropriately
- In the context of the expansion of the recall, the revision of the initial Type II risk to hybrid Type I risk for a vulnerable group in this case appeared as a belated attempt at proper regulatory oversight of the drug company by Health Canada.
- In the context of the expansion of the recall, the revision of the initial Type II risk to hybrid Type I risk for a vulnerable group in this case appeared as a belated attempt at proper regulatory oversight of the drug company by Health Canada.
- The persistent inability by the drug company to confirm the root cause of the packaging problem and thus to estimate and clearly communicate about its likely extent led to expansions in the lots covered by the recall, fuelled a sense that there may have been many faulty packages that women across the country had been taking for months.
- Misunderstanding and imprecise description within the media and the public of the responsibilities of the drug company and Health Canada for initiating drug recalls, and of risk classification levels.
- It may be generally (incorrectly) assumed that drug recalls are ordered by Health Canada through its regulatory oversight and authority.
- The company's initial recall notices stated (incorrectly, at the time of release) that the recall was conducted in collaboration with Health Canada, implying that Health Canada had participated in its planning and conduct and had waited several days to notify the public of the recall.
4.2 Evaluation of Recall Events and Response
The events that occurred in the recall of Alysena™ 28 and the social context in which they were interpreted transformed the risk of the faulty packages to the women using the drug into a much broader risk issue that called into question the reliability of pharmaceutical drugs and their regulation in Canada. A significant amount of public concern over the recall was caused by the expansion of the recall to include more lots of the drug and to apply in almost all provinces and territories in Canada, which resulted from the inability of the company to ascertain the lots that might have been affected by the initial problem in the manufacturing plant in Spain. However, the factors described above lead to some general conclusions on the reasons that the recall received so much attention and that much of that attention focused on a critique of Health Canada, and of the significance of that attention.
First, it is clear that considerable concern was generated by the recall, resulting in a number of Adverse Drug Reaction reports, the launching of a class-action lawsuit against Apotex and pointed criticism of the recall process in general, and of Health Canada in particular, from health professionals' associations. The fact that such attention and concern were generated by a potential failure of a contraceptive attests to the significance of that issue among the public, and thus the importance of providing information to alert women using the drugs to the problem so they can take other precautions. The perceived delays in Health Canada's notification of the public led to a great deal of criticism directed at Health Canada; while much of this may have been founded on common misunderstandings of the control that regulators in Canada exercise over industry and the division of responsibilities between industry and regulators, it is also evident that public confidence in the regulation of drugs, and of the reliability of the drugs themselves, was shaken by the incident.
Second, the information on which the media coverage was based was incomplete and changing, especially in the first days of the recall; reports of this information, much of it placed in the context of an incomplete understanding of the responsibilities and authorities of Health Canada, resulted in public information that was inconsistent, ambiguous and in some cases inaccurate. The non-standard way that actions by parties outside of Health Canada unfolded in this case, particularly the late recall notification by the company, the posting of information on the recall on a drug store blog over the weekend and expansions of the recall necessitated by the company's difficulty in determining the extent of the packaging problem, meant that some of these challenges were difficult to avoid. Nevertheless, they do point to the importance of the manner in which information is delivered in the interpretation of an issue, and of the context in which those considered responsible for providing the information are judged.
Health Canada needs to be alert to the requirement to provide accurate and timely information on issues of high social concern. Public information on the responsibilities of Health Canada and industry in ensuring drug safety, which could be used by media and interested members of the public, was not found on the healthycanadians.gc.ca website, on which publicly-oriented information such as recall advisories is posted, and is not stated clearly on other pages of the department's main site. The Drugs and Health Products web page states that "Health Canada is responsible for compliance monitoring and enforcement activities related to health products in order to verify that regulatory requirements are being applied appropriately." but does not describe the nature of the responsibilities of the Health Canada and industry in drug regulation and recalls. The Drugs and Health Products web page includes links to the relevant policy documents, but it may be only the most dedicated reader who will navigate to the ‘compliance and enforcement' section, open the files and read the policy document to learn the distribution of responsibilities for drug recalls.
Third, the way that the information was posted on the blog of a drug store company, picked up by the media and transformed into a major news story during the course of a weekend, is evidence that traditional models of communications no longer apply. In an era of social media, 24-7 news media access to and publication of information and the ability of non-traditional news providers to publish information, greater agility in preparing and providing information and awareness of the issues that demand it will be increasingly important. Public risk regulators face new communications challenges in this changing environment: as Ragnar Lofstedt has noted recently in this regard, "due to the Internet, 24-7 news, and the social media, regulators are not the only ones with access to pertinent information and, as a result, they do not often have much time to craft messages or respond to queries before other groups obtain the information" (Lofstedt, 2013: 198). Regulators may thus find themselves taking "reactive rather than proactive communication approaches," which can have serious implications: "Proactive risk communication gains trust while reactive communication can destroy trust" (Lofstedt, 2013: 198). Illustrating the importance of proactive communication, the misunderstanding that Health Canada had initiated the recall but had neglected to notify the public enabled the Pharmacy company to ‘choose to be proactive' in advising its patients of the recall.
Fourth, the largely critical evaluation of the performance of Health Canada as a public risk regulator was based on the interpretation of the available information in the context of high expectations of pubic regulators that are not achievable with the present legislative and regulatory provisions. The Food and Drugs Act and the associated Food and Drug Regulations do not provide Health Canada with clear definitions of the requirements it is to enforce, or with the powers necessary to enforce them. The lack of such provisions leaves Health Canada vulnerable to incomplete compliance by companies, as public expectation of its performance and accountability far exceed its powers to achieve and enforce even the stated expectations of industry behavior.
4.3 Review Comments on Health Canada's Handling of the Recall
This review is concerned with evaluating Health Canada's handling of the Alysena™ 28 recall by Health Canada, with particular attention to the concerns, voiced in the media and by health professionals that Health Canada had failed to notify the public early enough in the recall process. While the preceding two sections have outlined the events that led to the high concern with the recall and analyzed their implications, this section focusses on the actions of Health Canada personnel and procedures in the recall action. The purpose is to consider whether Health Canada actions met existing performance expectations, whether any actions by Health Canada may have contributed to the concern caused by the recall, and whether there are any gaps in procedures or guidance that may require revision to improve the handling of drug recalls in the future.
- Factors beyond the control of Health Canada
- There is no means by which Health Canada personnel could have acquired earlier information on the recall, as it did not receive notice of the recall within the timeframe stipulated in the Recall Policy and GUI-0039. Health Canada has no authority to enforce companies' compliance with their responsibility to notify Health Canada within 24 hours of making a decision to initiate a recall as this is a policy interpretation of an imprecise regulatory requirement.
- There is no means by which Health Canada personnel could have acquired earlier information on the recall, as it did not receive notice of the recall within the timeframe stipulated in the Recall Policy and GUI-0039. Health Canada has no authority to enforce companies' compliance with their responsibility to notify Health Canada within 24 hours of making a decision to initiate a recall as this is a policy interpretation of an imprecise regulatory requirement.
- Aspects handled appropriately by Health Canada
- Health Canada personnel in the Regional Centre and HPFBI appropriately reviewed the company's recall notification file when it was received, and passed it on to the appropriate personnel and offices in a timely fashion.
- The revised Type I-Type II risk level reflecting the higher risk to a vulnerable population was appropriate and consistent with past practice. The time that was taken by TPD for the scientific review of the company's risk level assignment was within the guidelines in SOP—537 for a Type I risk (despite the fact that the request was received as a Type II risk). It is essential that sufficient time be allowed for a scientific Health Risk Assessment, in order to ensure the credibility of the assessment and the accountabilities of a science-based regulatory department.
- Problematic actions by Health Canada
- Health Canada personnel did not act on the opportunity to issue a public communication of the recall of the drug on the afternoon of Friday April 5, to respond to the social concern associated with unplanned pregnancies.
- Communications strategists, who are experienced in communicating about socially sensitive issues, were not informed of the recall until Monday April 8. A brief communication issued on Friday April 5 would have prevented the perceived delay in Health Canada's response; the anticipated revision in the risk level could have been addressed through the issuing of an alert stating that more information would follow, in accordance with past practice.
- Drug recall procedure documents do not provide detailed information on the circumstances in which a public communication may be issued and do not direct CECD personnel to consult communications strategists on the matter. They also do not refer to the risk communication SOP for information on the issuing of urgent communications and on socially sensitive risk issues.
- Aspects of the issue that were not foreseen by Health Canada
- Health Canada personnel did not anticipate that Apotex's issuing of a recall notice to its wholesalers and distributors on April 3, which set in motion the removal of the affected lot of the drug from pharmacy shelves, would reach the general media and the public in the five days before they had all the information they needed for a full public advisory.
4.4 Recommendations
This review has observed that in most instances Health Canada personnel met performance standards for a timely response to the information they received about Apotex's recall of Alysena™ 28, but that they failed to take the opportunity to issue a public communication to notify the public of the problem on Friday April 5, before news of the recall reached the public by other means and implied a delay in Health Canada's response.
However the review has also noted, from the broader perspective that may be more relevant to public expectations for the regulation of drugs in Canada, that a period of 15 days elapsed between the date the company became aware that its product could pose a risk to the public and the date on which it engaged the federal regulator in the management of that risk. Many of the issues raised by the recall of Alysena™ 28 by health professionals, the media and members of the public questioned the adequacy of regulatory oversight of the drug industry in Canada, a situation that may undermine Canadians' confidence in pharmaceutical drugs and in their regulation.
Therefore the following recommendations address both the ability of Health Canada to respond to information it receives on risks of pharmaceutical drug products, and the larger framework within which the Department's roles and responsibilities are set.
The recommendations come directly from the review of events around the Alysena™ 28 recall, and in all cases can be referred directly to citations or information provided in this evaluation. The first recommendation makes three suggestions for strengthening Health Canada's authority and engagement with the response to risks of drug products. Three recommendations address public communications on drug recalls, attesting to the influence of that function in the way the Alysena™ 28 recall unfolded, and to the importance of good communications in ensuring an accurate understanding of a drug safety issue and in maintaining public trust in the drug system in Canada. Two recommendations suggest revisions to fill gaps identified in procedures set out in guidance and procedure documents, as well as supporting material to help Health Canada make proactive responses to sensitive drug recall issues, while another recommends that accurate and complete information on the drug regulation framework be made more accessible for those who are interested.
4.4.1 Recommendation 1: Strengthen Health Canada's authority in responding to hazards of drug products
This recommendation has three components that address changes to the requirements of industry to engage with Health Canada when a risk is posed by a drug product. The first may be put in place relatively promptly, while the second and third are longer-term changes to bring the drug regulation regime into line with the provisions of comparable regimes in Canada.
Recommendation 1.1 Improve and clarify recall notification requirements
Much of the concern about the recall of Alysena™ 28 occurred because Health Canada was not notified about the recall until after it had been initiated. The legal responsibility of industry should be revised to state explicitly that Health Canada must be contacted within 24 hours of the decision to conduct a recall. In all instances the notification must occur before any recall action is initiated (the legislative requirements of industry notification of a drug recall and Health Canada's more precise policy elaboration of that requirement are discussed in section 2.1 and 2.2, above).
Health Canada personnel are constrained in their ability to establish and enforce clear responsibilities for industry on the conduct of a recall. This is due in part to the imprecise wording of the notification requirement in the Regulations, that companies must inform Health Canada "forthwith" when they "commence a recall." They are also constrained by their lack of authority to enforce the Department's more precise policy interpretation of that regulatory requirement, that companies must notify Health Canada within 24 hours of making the decision to recall a drug. It is evident from the response to the recall of Alysena™ 28 that the public, the media, and health professionals expect the public regulator of health products to have greater powers to engage with industry when the decision to recall a product is reached. The actions of Apotex clearly do not meet the expectations of the Canadian public, but it is not easy to prove that they were not in compliance with the current imprecise regulatory requirement.
Recommendation 1.2 Consider revising the notification requirement to within 24 hours of the company's becoming aware of a risk with a drug product.
Much of the commentary in response to the Alysena™ 28 recall called into question the adequacy of Health Canada' regulatory oversight of drugs and noted that apparent ‘gaps in regulation' undermine Canadians' confidence in drug products.
Under the current requirement that industry must notify Health Canada within 24 hours of making a decision to recall a drug, the period that may elapse between the identification of a risk and a decision to recall the drug is unspecified. The narrow scope of this requirement has further implications for drug regulation: it means that Health Canada may not be involved in discussions on the risk level assigned to the risk and the most appropriate risk management action and that if the decision does not involve a recall of the drug Health Canada may not be informed at all.
Making the trigger for notification of the regulator the identification of a risk with a drug rather than company's decision to initiate a recall will ensure that Health Canada will be engaged with the company in assessing the risk of drug problems and with the decision on the most appropriate risk management action, whether that is a recall or other measure. This extension of the notification requirement would bring Health Canada's regulatory oversight of drugs into line with that of the regulation of food and consumer products.
Recommendation 1.3 Consider strengthening the recall authority for marketed health products by Health Canada.
While not a direct focus of this evaluation, a review of recall authority across different regulatory units highlights a difference in recall authority between foods, consumer products and pharmaceutical products. In the interest of maximizing the effectiveness of drug safety programs in Canada, consideration might be given to strengthening recall authorities for pharmaceutical products to correspond to those currently in place for food and consumer products.
As discussed in section 2.5 of this report, legislation governing two regulatory regimes with similar mandates to Health Canada's oversight of pharmaceutical products includes the power to order a recall when regulatory authorities determine that a product may cause a risk to health or safety. These are the Canadian Food Inspection Agency Act, which created the Canadian Food Inspection Agency (CFIA), and the Canadian Consumer Product Safety Act, administered by Health Canada's Consumer Product Safety Program. The Food and Drugs Act does not give Health Canada that power, and the recall provisions in the Food and Drug Regulations pertain only to recalls that are initiated voluntarily by the drug company. Without the provision of mandatory recall of a drug product that poses an unacceptable risk, the drug regulation regime provides less robust oversight and weaker consumer protection than the food and consumer products regimes offer. The response of consumers and health professionals to the Alysena™ 28 recall indicates that Canadians expect a higher degree of regulatory control exerted by the federal regulator of drug products in Canada.
4.4.2 Recommendation 2: Improve clarity and communication of roles and responsibility concerning a drug recall
The Canadian public, media and pharmaceutical industry need clear and easily accessible information on the roles and responsibilities of all parties in drug recall actions. In this instance there was lack of clarity concerning the relative roles and responsibilities of Health Canada and Apotex in reaching the decision to recall Alysena™ 28 and in carrying out that decision (Sections 3.4.1 and 3.4.2 of this report describe some imprecise statements that Health Canada can order a drug recall and had ordered the recall of Alyesna 28™.)
Information on the framework of responsibilities for ensuring the safety of drugs and conducting drug recalls should be made more accessible on Health Canada's public website for those who are interested. The main responsibilities of all parties should be described on the Drugs and Health Products web page, and a link to that information and to recall policy documents should be provided on the drug recall database pages.
Health Canada should revise guidance documents for industry to describe their roles and responsibilities in this complex activity, including the absolute need for open and clear collaboration and coordination beginning before the recall activity is announced to the public and is initiated. The possibility should also be explored of giving guidance to industry on information to be included in recall notices. This could include information on the nature and extent of the problem, and on ensuring accuracy in references to the roles and responsibilities of industry and Health Canada in the recall action.
4.4.3 Recommendation 3: Include consideration of level of social concern
This recall was initiated by the recognition of a packaging error for Alysena™ 28 that substantially reduced its effectiveness in fertility regulation. An unanticipated pregnancy can represent a significant adverse impact on the life of a woman; however that did not appear to be considered either by Health Canada or industry. The initial Type II risk assigned by Apotex and the failure of Health Canada to issue an interim public communication on Friday April 5 suggested a lack of sensitivity to the significance of an unplanned pregnancy, and a consideration only of the strict physical health risks that were reflected in the later addition of a Type I risk for vulnerable individuals (discussed in section 3.4 above, on media and social concern).
The draft SOP for the Preparation of Risk Communication contains detailed information on the conditions under which an urgent risk communication may be issued, including outside of regular business hours and on weekends, and on the characteristics of risk issues of high social concern and the manner of addressing them. However there are no references to this SOP in any CECD directives on drug recalls, and the provisions it contains for the issuing of an urgent, preliminary public notice in recognition of the involvement of possible unplanned pregnancies were not followed in the Alysena™ 28 recall.
The draft SOP on risk communication should be finalized, and should be referenced in CECD procedural documents on drug recalls. Procedures should also be developed for CECD on the use of communications for recalls that are associated with socially sensitive issues such as unanticipated pregnancy, or health effects which may raise particular concern, and included, or referenced, in SOP-0331 and POL-016. Consistent with the risk communications SOP, these procedures should be clear that public communication should be issued for any drug recall about which there may be significant social concern, even if the risk is a Type II or Type III. The procedures should direct CECD to consult the HPFBI Communications Strategists for advice during the assessment process in situations in which a public communication would be helpful and on the issues of concern that should be addressed.
In addition, training should be provided to Health Canada personnel with responsibilities for drug recalls, on the social sensitivities of some drugs, and medical conditions or vulnerable groups that raise concern about them beyond risks to health, such as unanticipated pregnancy. This training material may be referenced in SOP-0331.
4.4.4 Recommendation 4: Improve capacity to respond in a more timely fashion
In this recall activity the presence of holidays and weekends substantially interfered with timely notification of the Canadian public and relevant health professionals about the ongoing recall activity for Alysena™ 28 . These gaps in the presence and role of Health Canada in making relevant information available to the public, the media and professional groups clearly hindered appropriate risk communication and led to the clear impression that Health Canada was shirking its responsibility in the recall (as expressed in the reports and commentary on Health Canada's apparent delay in notifying Canadians of the recall).
Guidance information should be developed, and included or referenced in relevant CECD procedure documents such as SOP-331, Drug Recall Procedure, on the potential for the publication of information by sources other than the established media, and on actions that are required and recommended for the provision of appropriate Health Canada information to the public. Current procedures should be revised to enable a response outside of normal business hours, and should include directives on appropriate consultation and collaboration among CECD, Regional Centres, TPD and Communications Strategists to ensure that information provided outside of normal business contexts is both timely and accurate. Health Canada should conduct an analysis of the capacity requirements for a response to risk issues outside normal business hours.
5. References
5.1 Health Canada Health Products and Food Branch Inspectorate documents
- Draft Standard Operating Procedure, Production of Risk Communications by the Public Affairs, Consultation and Communications Branch of Health Canada
- FRM-0016 Rapid Alert Notification
- FRM-019 Health Hazard Evaluation Request, 2006
- GUI-0039 Guidelines for Recall of Drugs and Natural Health Products, 2010
- POL-0016 Health Products and Food Branch Inspectorate Recall Policy, 2006
- Product Recall Procedures, 1993
- SOP-0331 HPFBI Drug Recall Procedure, 2005
- SOP-0331 HPFBI Drug Recall Procedure Revision 1, 2013
- SOP-0537 Requesting a Health Risk Assessment, 2013
5.2 Other references
- CFIA
- Health Canada Drugs and Health Products
- Justice Canada, Food and Drug Regulations
- Justice Canada, 1997. Canadian Food Inspection Agency Act 1997
- Justice Canada, 2008. Food and Drugs Act.
- Justice Canada, 2012. Canada Consumer Product Safety Act
- Lofstedt, Ragnar, 2013. Communicating Food Risks in an Era of Growing Public Distrust: Three Case Studies. Risk Analysis 33(2): 192-202
Appendix 1 - Chronology of Alysena™ 28 Recall
Date: unclear (pre-production test run of new generic drug)
- Apotex - packaging contractor in Spain allows defective test package into production
- RSI comment - Problem caused by human error, re-introducing rejected faulty test package into production line
Date: December 2012
- Apotex - markets product in Canada (only)
Date: March 20
- Apotex - received complaint + picture of faulty package
Date: March 25
- Apotex - received complaint sample, substantiated defect
- Performance Expectation - HC accepts that company needed time to contact packaging company, coordinate with 3rd party handling recall - expectation is that decision is timely and risk mitigation is driving concern
Date: March 28
- Apotex - made decision to recall
- Performance Expectation - HC accepts that company needed time to contact packaging company, coordinate with 3rd party handling recall - expectation is that decision is timely and risk mitigation is driving concern
Date: April 1
- Apotex - prepared recall notification package Assembled information package for HC
- Performance Expectation - HC accepts that company needed time to contact packaging company, coordinate with 3rd party handling recall - expectation is that decision is timely and risk mitigation is driving concern
- RSI comment - detailed recall info package not needed by HC at this point - could have notified earlier and provided detailed info later, per POL-0016.
Date: April 2
- Apotex - HRA finalized; type 2
- Performance Expectation - HC accepts that company needed time to contact packaging company, coordinate with 3rd party handling recall - expectation is that decision is timely and risk mitigation is driving concern
Date: April 3
- Apotex - sent recall letter to wholesalers and distributers stated "unplanned pregnancy cannot be ruled out"
beginning of Health Canada involvement
Date: April 4 Thursday
- Apotex - Notified HC Ontario Region supervisor
- HC - RAPB, ON Region - supervisor received notification email 2:16 pm
- Performance Expectation
- Apotex: company should notify HC within 24 hours of decision - would have been March 29 (Policy 016)
- HC: performance standard met. Timeline very fast, especially as recall still at type II risk which has 48 hour timeline
Date: April 4 Thursday
- HC - RAPB, ON Region - supervisor flags type 2 risk 2 needs confirmation: sensitive issue, but with a single package
- Performance Expectation
- Apotex: company should notify HC within 24 hours of decision - would have been March 29 (Policy 016)
- HC: performance standard met. Timeline very fast, especially as recall still at type II risk which has 48 hour timeline
- RSI comment - need to consider appropriate risk level when outcome may be serious or sensitive but only 1 faulty package confirmed
Date: April 4 Thursday
- HC - RAPB, ON Region - 3:39 pm supervisor assigns to inspector
- Performance Expectation
- Apotex: company should notify HC within 24 hours of decision - would have been March 29 (Policy 016)
- HC: performance standard met. Timeline very fast, especially as recall still at type II risk which has 48 hour timeline
Date: April 4 Thursday
- HC - RAPB, ON Region - 4:54 pm inspector refers file to HPFBI Ottawa
- Performance Expectation
- Apotex: company should notify HC within 24 hours of decision - would have been March 29 (Policy 016)
- HC: performance standard met. Timeline very fast, especially as recall still at type II risk which has 48 hour timeline
Date: April 5 Friday
- HC - HPFBI Ottawa - HPFBI reviews file; Sends to ORM in TPD, 10:03 am. Consult requested to verify risk type
Date: April 5 Friday
- HC - TPD - ORM refers consult to BPS and BMORS for risk type evaluation
Date: April 5 Friday
- HC - HPFBI Ottawa, HC - TPD- phone conversation, confirmation that TPD reviewing file
- Performance Expectation - file did not include the information normally required for an HRA
- RSI comment - involvement of contraceptive gave issue higher sensitivity and priority
Date: April 5 Friday
- HC - HPFBI Ottawa, HC - TPD
- Late afternoon. Phone conversation ORM (TPD), HPFBI Ottawa: preliminary investigation indicates type 1 risk for vulnerable pop's; formal clinical review board response to come Monday.
- TPD recommended public comm'n be issued that day to inform consumers of the risk. A communication was not issued that day, and the matter was left until the assessment would be finalized on the following business day (Monday).
- Performance Expectation - timeline for TPD HRA for Type II is 48 hours/ 2 business days
- RSI comment - public communication recommended to inform consumers of possibility of unplanned pregnancy with faulty package
Date: April 5 Friday
- HC - HPFBI Ottawa - 4:08 pm email to HPFBI Manager & Supervisor on informal indication of type I/ type II risk and that public comm'n may be needed
- RSI comment - Should public notice have been issued based on possible sensitivity regardless of type 2 risk, single package, and without a final health risk assessment from TPD?
Date: April 6 Saturday
- Media and Social - Drug store company posted notice of recall on blog.
- RSI comment
- this communication and others based on it - based on minimal information in Apotex recall notice
- Apotex recall directed to wholesalers, distributors and retailers
Date: April 7 Sunday
- Media and Social - Major news network publishes story from pharmacy company blog on "Health Canada recall"
Date: April 8 Monday
- HC - RAPB, ON Region - ongoing: asking company for assurance that no other lots were affected
- HC - HPFBI Ottawa - 10:15 email from HPFBI drug investigation specialist to Communications Strategists advising that PA may be needed
- Media and Social
- 10:47 email that recall has been in media already
- media inquiries received
Date: April 8 Monday
- HC - RAPB, ON Region - finalized RAN; forwarded to CECD
- HC - HPFBI Ottawa
- 11:23 written result of consult result received, confirming type 1 for vulnerable, type 2 general pop'n discussed April 5
- recommended Public Comm'n, and extending recall to patients
- Performance Expectation - risk classification assignment driven by potential for serious harm, rather than number of people exposed
Date: April 8 Monday
- Apotex - Apotex planned media announcement - reviewed with HC Region
- HC - RAPB, ON Region - notified Apotex HC was going to make announcement to media
- Performance Expectation - HC may ask company if they plan to issue public comm'n; companies usually advise inspectors of plans to post recall notice
Date: April 8 Monday
- HC Comm's
- around noon
- recall posted on Healthy Canadians website
- Recall info tweeted, with link to web notice.
- Notice included type 1 and type 2 but did not ID type 1 -vulnerable groups
- Cited product monograph advice
- Performance Expectation
3 assessments- standard met: public advisory not always posted if there is broad media coverage
- not met: PA should have been issued earlier regardless of risk type; have been issued previously for contraceptive recalls
- not met: contraceptive issues are sensitive and often get public comms
- RSI comment - posted recall notice and tweeted link to it rather than full PA in order to get something public ASAP - approvals, translation of formal PA takes 3-4 hours
Date: April 8
- HC Comm's - receipt of media inquiries
- Media and Social - media coverage
- RSI comment - Focused on HC not informing consumers, discrepancy in type I and II risk, lack of sensitivity to unplanned pregnancy. Content reflects incorrect understanding of scope of problem
Date: April 9
- Apotex - Issues updated advisory with type 2/ type 1 risk. Asks pharmacists to contact patients; advice on what to do if they have faulty package
- HC - RAPB, ON Region - Ongoing: receiving company updates of recall progress
- Performance Expectation - HC met. SOP gives service standard for proportion of consignees to contact
Date: April 9
- Media and Social - Adverse Drug Reaction reports begin: 40 from April 9- May 29; 31 from consumers, 24 claiming pregnancy while on Alysena™
- RSI comment - high level of concern; perhaps based on incomplete information on number of faulty packages
Date: April 11
- Apotex - provided risk of pregnancy due to missed doses
- RSI comment - Apotex risk description had been imprecise in initial recall notice
Date: April 11 and 12
- Apotex - ongoing - unable to verify that no other lots had been affected by faulty packaging due to plant's inability to reconcile package numbers
- HC - RAPB, ON Region, HC - HPFBI Ottawa, HC - TPD, HC Comm's - HC Minister "called an investigation into why women were not informed for five days after Health Canada authorities found out their birth control contained twice as many placebo pills as advertised."
Date: April 12
- Apotex - issues expanded recall notice as precaution due to inability to verify other lots - no evidence other lots affected
- HC Comm's - Issued Information Update: precautionary recall notice of additional lots recalled posted. No mention of risk type. Explains problem and repeats advice
Date: April 12
- HC - HPFBI Ottawa - QIAD requests previous inspection reports for packaging plant from European Medicines Agency
- Performance Expectation - met - under MRA inspections done by country of plant according to agreed standard
Date: April 12
- Apotex - Ongoing: recall process. With weekly updates on recall status
- Performance Expectation - Met. Takes time for patients to return product to pharmacies, pharmacies to return to their contact points.
Date: April 15
- Apotex - informed HC that no additional complaints of faulty packages for the original recall lot had been received
- HC Comm's - re-issue of April 12 recall notice on expanded recall lots. Includes type 1 and 2 risks with explanation of groups they apply to
Date: April 16
- HC - RAPB, ON Region
- effectiveness checks conducted through to April 23
- called 205 of 419 consignees; all aware of and acting on recall
- HC - HPFBI Ottawa - received investigation report from Spanish manufacturer
Date: April 17
- HC - HPFBI Ottawa - telecom with Spanish authority - planned inspection of plant April 29
Date: April 19
- Apotex
- provided report on 200% visual inspection of new lot of Alysena™ 28 for release on market in Canada
- HC - HPFBI Ottawa - reviewed Apotex report; had no objection to release of these newly manufactured and previously un-distributed lots with 200% visual inspection
- Performance Expectation - Recalled lots had all been removed from sale.
Date: April 30
- HC - HPFBI Ottawa - received report from Spanish authority - satisfied with company's CAPA
Date: May 2
- Media and Social - CMAJ release of Society of ObGyn and College of Fam, Phys.: criticized Health Canada for 'accepting manufacturer's voluntary recall without notifying public'; referenced thousands of women at risk of pregnancy.
- RSI comment - What information was this critique based on?
Date: ongoing
- Media and Social - recalled product being returned to 3rd party handling recall; packages to be inspected and final count of faulty packages made
Date: July 8
- HC - HPFBI Ottawa - received notification from Apotex that second faulty package discovered among recalled products
Appendix 2: Apotex Recall Notices
Apotex Recall Notice - April 3, 2013

Text description
Urgent Drug Recall
April 03, 2013
Apotex Advancing Generics
Apotex is notifying all direct account Wholesalers, Distributors, and Retailers that have received the impacted lot of a voluntary TYPE II recall being conducted up to the Retail level for the product listed below. This product is manufactured by a third party and distributed by Apotex. This voluntary TYPE II recall is being conducted in coordination with the Health Products Food Branch Inspectorate Canada
Product Name
ALYSENA 28
(Levonorgestrel and Ethinyl Estradiol Tablets USP)
DIN
02387883
Strength
100 µg / 20µg
PKG Format
28 x 1 BLS in a carton
UPC Code
771313219914
LOT Number
LF01899A
EXP. Date
10/2014
Reason for Market Action
Product lot identified in this recall notification may contain two rows of placebo (white) Tablets instead of one row of Placebo (white) Tablets.
Health Assessment
Ingestion of only 14 tablets of active instead of intended 21 of oral contraceptive would most likely result in reduced efficacy for contraception and therefore possibility of unplanned pregnancy cannot be ruled out.
Actions to Be Taken
- Stop distributing and quarantine above mentioned lot of ALYSENA 28 Tablets.
- Wholesalers/ Distributors are to conduct a sub-recall to retail customers to whom you have shipped the affected lot, by informing them of the recall, requesting that they remove the affected lot from sale and return the stock to the wholesaler from whom it was purchased. Your customers should not return stock directly back to Apotex or Stericycle.
- For customers who purchased the affected product DIRECTLY from Apotex, you are requested to conduct a physical count of affected inventory on hand. Note: Wholesaler/Distributors must also include in their count, inventory that was returned by their retail customers.
Record total inventory on hand on the Business Reply Form (BRF) included in this letter and fax to Stericycle to the Toll-Free fax number below. Note: If you have NO inventory on hand, you are still required to complete the BRF and fax it back to Stericycle documenting that no affected inventory on hand. - If this letter has been forwarded to a retail pharmacy or institution that has purchased affected inventory through a wholesaler/distributor:
The below mentioned return process DOES NOT apply to you, Retail customers, who purchased through a wholesaler/distributor, must return the affected inventory back to the establishment from where the inventory was purchased. For these retailers, please contact your wholesaler for specific return and credit instructions. DO NOT return your affected inventory back to Apotex or Stericycle. - Returning affected inventory to Stericycle. For customers who purchased affected product DIRECTLY from Apotex, place all affected product in a shipping carton, enclose a hard copy of the completed BRF, identify the product as "Recall Product" on the outside of the shipper and apply the prepaid shipping label provided. Contact Stericycle for additional return labels if required.
Return address:
Event #: 8415
Stericycle Inc.
25 Ironside Crescent,
Toronto, Ontario M1X 1G5
Toll-free FAX: 1-866-324-3734
Additional Information
A credit will be issued for affected stock upon receipt of inventory at Stericycle.
Please complete and return by fax the enclosed Business Reply Form to receive prompt credit to your account. Also, please identify the returned merchandise as "RECALL PRODUCT" on the outside of the shipper.
If further assistance is required for returns please contact Stericycle at : 1-866-367-4537
Sincerely,
Apotex
Apotex Recall Notice - April 9, 2013

Text description
Urgent Drug Recall (Update)
April 09, 2013
Apotex Advancing Generics
Apotex is updating all direct account Wholesalers, Distributors, and Retailers that have received the impacted lot below. The recall has been reclassified by Health Canada's Therapeutic Products Directorate as a voluntary TYPE I & TYPE II recall being conducted up to the Retail level and to a subset of the population. This product is manufactured by a third party and distributed by Apotex. This voluntary TYPE I & TYPE II recall is being conducted in coordination with the Health Products Food Branch Inspectorate Canada. Other lots or products are not impacted by this recall.
Product Name
ALYSENA™ 28 (Levonorgestrel and Ethinyl Estradiol Tablets USP)
DIN
02387883
Strength
100 µg / 20µg
PKG Format
28 x 1 BLS in a carton
UPC Code
771313219914
LOT Number
LF01899A
EXP. Date
10/2014
Recall Risk/Classification
- TYPE I: For patients which includes patients that should not get pregnant, whether for medical reasons or exposure to agents detrimental to a developing fetus (such as those on pregnancy prevention programmes while taking drugs that can cause harm to a developing fetus).
- TYPE II: General Population
Reason for Market Action
Product lot identified in this recall notification may contain two rows of placebo instead of one row (14 tablets instead of 7 tablets) and two rows of active contraceptive tablets (14 tablets instead of 21 tablets) in the blister pack.
Health Assessment
Ingestion of only 14 tablets of active instead of intended 21 of oral contraceptive would most likely result in reduced efficacy for contraception and therefore possibility of unplanned pregnancy cannot be ruled out.
Actions to Be Taken
- Stop distributing and quarantine above mentioned lot of ALYSENA™ 28 Tablets.
- Patients who have received ALYSENA™ 28 lot LF01899A or have questions regarding this recall please contact your pharmacy. Patients should not interrupt their therapy but should consult their health care provider for appropriate medical advice.
- Retail and hospital pharmacies that track lot numbers dispensed to patients please contact patients who have received ALYSENA™ 28 lot LF01899A, advise them of the recall, and recover any units of lot LF01899A for return.
For retail and hospital pharmacies that are unable to confirm the lot number of ALYSENA™ 28 lot LF01899A dispensed to patients, please contact patients who have received ALYSENA™ 28 lot LF01899A between December 4th, 2012 and receipt of this notification, advise them of the recall, and recover any units of lot LF01899A for return. - Wholesalers/ Distributors are to conduct a sub-recall to retail customers to whom you have shipped the affected lot, by informing them of the recall, requesting that they remove the affected lot from sale and return the stock to the wholesaler from whom it was purchased. Your customers should not return stock directly back to Apotex or Stericycle.
- Customers who purchased the affected product DIRECTLY from Apotex, you are requested to conduct a physical count of affected inventory on hand. Note: Wholesaler/Distributors must also include in their count, inventory that was returned by their retail customers.
Apotex Recall Notice - April 12, 2013

Text description
Urgent Drug Recall
April 12, 2013
Apotex Advancing Generics
Apotex is updating all direct account Wholesalers, Distributors, and Retailers that have received the impacted lots below. In addition to our recall letters issued on April 03, 2013 and April 09, 2013, Apotex is initiating a recall of all lots distributed to date on a precautionary basis. This product is manufactured by Laboratorios Leon Farma, S.A. and distributed by Apotex. This voluntary TYPE I and II recall is being conducted to the Patient Level, and in collaboration with Health Canada. Only the lots identified in this recall letter are impacted.
Product Name
ALYSENA™ 28 (Levonorgestrel and Ethinyl Estradiol Tablets USP)
DIN
02387883
Strength
100 µg / 20µg
PKG Format
28 x 1 BLS in a carton
UPC Code
771313219914
LOT Numbers
- LF01901A - EXP. Date 10/2014
- LF01900A - EXP. Date 10/2014
- LF01898A - EXP. Date 10/2014
- LF01894B - EXP. Date 10/2014
LOT Numbers
- LF01980A - EXP. Date 11/2014
- LF01982A - EXP. Date 11/2014
- LF01981A - EXP. Date 11/2014
- LF01979A - EXP. Date 11/2014
- LF02037A - EXP. Date 11/2014
- LF02036A - EXP. Date 11/2014
- LF02026A - EXP. Date 11/2014
Reason for Market Action
Apotex is recalling these additional lots of ALYSENA™ 28 as a precautionary measure, while it investigates why the original lot of ALYSENA™ 28, LF01899A, contained blisters with extra placebo tablets (white) in place of active tablets (pink). Blisters of ALYSENA™ 28 should have three rows of active tablets (pink) and only one row of placebo tablets (white).
Health Assessment
Ingestion of only 14 tablets of active instead of intended 21 of oral contraceptive would result in reduced efficacy for contraception and therefore possibility of unplanned pregnancy cannot be ruled out.
Actions To Be Taken
- Stop distributing and quarantine above mentioned lots of ALYSENA™ 28 Tablets.
- Individuals who have received impacted lots of ALYSENA™ 28 Tablets or have questions regarding this recall please contact your pharmacy. Individuals should not interrupt their therapy, use a non-hormonal method of birth control, contact their health care provider for medical advice and should return unopened packages to their pharmacist.
As an alternative to ALYSENA™ 28 tablets, individuals may continue their therapy with ALYSENA™ 21 tablets until the supply of ALYSENA™ 28 tablets resumes with new lots not impacted by this recall. The only difference between ALYSENA™ 28 and ALYSENA™ 21 is that ALYSENA™ 28 includes a row of seven placebo tablets (white), which are not a critical part of the therapy. Alternatively, if ALYSENA™ 21 tablets are not available and individuals urgently require tablets for immediate therapy, pharmacists may consider inspecting the blisters of ALYSENA™ 28 that are currently available to determine whether they contain all three rows of seven active tablets (pink), for a total of 21 tablets, in which case such blisters of ALYSENA™ 28 tablets may be used for an individual's current menstrual cycle only.
Appendix 3: London Drugs Blog Post
London Drugs
Urban Lifestyle Blog
April 6 2013
Health Canada and Apotex Urgent Drug Recall
If you had a prescription filled for the birth control pill, Alysena™ 28 day with Lot Number LF 01899A anywhere in Canada, you need to be aware of an urgent manufacturer recall. According to the manufacturer, Apotex, there was one unit of Alysena™ in Eastern Canada found to have an issue.
In a typical 28 day pack of Apotex pills of Alysena™ , the last week of pills are sugar (placebo) pills in order for the patient to remember to take the pills consistently. In Apotex's recall notice, Alysena™ 28 day Lot Number LF 01899A, the manufacturer has received one report of a package of pills with the first 7 days of the 28 day cycle of pills to also contain (placebo) sugar pills.
Please note this is currently a Type 2 recall from Health Canada and Apotex. A Type 2 recall requires drug wholesalers and pharmacies to remove inventory of the recalled product. Although this recall is currently a Type 2 recall and does not require pharmacy to contact patients, due to the potential seriousness of the recall, and in an abundance of caution, care and concern for our customers, London Drugs has chosen to be proactive in advising our customers and patients of the recall.
[Below is the excerpt of the Canada-wide manufacturer urgent recall.]
Appendix 4: Health Canada Recall Notices
Health Canada Recall Notice - April 8, 2013
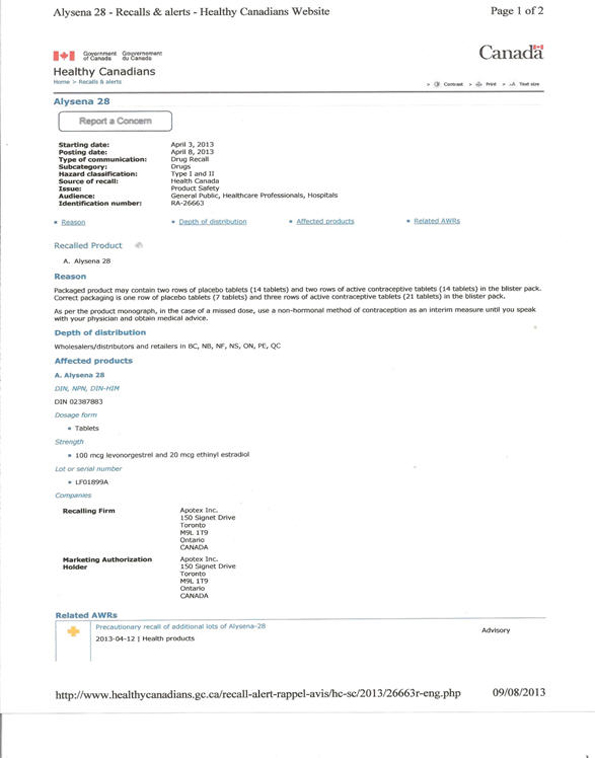
Text description
Alysena 28 - Recalls and alerts - Healthy Canadians Website
Government of Canada
Healthy Canadians
Home > Recalls and alerts
Alysena 28
Starting date:
April 3, 2013
Posting date:
April 8, 2013
Type of communication:
Drug Recall
Subcategory:
Drugs
Hazard classification:
Type I and II
Source of recall:
Health Canada
Issue:
Product Safety
Audience:
General Public, Healthcare Professionals, Hospitals
Identification number:
RA-26663
Recalled Product
A. Alysena 28
Reason
Packaged product may contain two rows of placebo tablets (14 tablets) and two rows of active contraceptive tablets (14 tablets) in the blister pack. Correct packaging is one row of placebo tablets (7 tablets) and three rows of active contraceptive tablets (21 tablets) in the blister pack.
As per the product monograph, in the case of a missed dose, use a non-hormonal method of contraception as an interim measure until you speak with your physician and obtain medical advice.
Depth of distribution
Wholesalers/distributors and retailers in BC, NB, NF, NS, ON, PE, QC
Affected products
A. Alysena 28
DIN, NPN, DIN-HIM
DIN 02387883
Dosage form
Tablets
Strength
100 mcg levonorgestrel and 20 mcg ethinyl estradiol
Lot or serial number
LF01899A
Companies
- Recalling Firm
- Apotex Inc.
150 Signet Drive
Toronto
M9L 1T9
Ontario
CANADA
- Apotex Inc.
- Marketing Authorization Holder
- Apotex Inc.
150 Signet Drive
Toronto
M9L 1T9
Ontario
CANADA
- Apotex Inc.
Apotex Recall Notice - April 15, 2013

Text description
Alysena 28 - Expanded (Additional lots) - Recalls and alerts - Healthy Canadians Website
Government of Canada
Healthy Canadians
Home > Recalls and alerts
Alysena 28 - Expanded (Additional lots)
Starting date:
April 12, 2013
Posting date:
April 15, 2013
Type of communication:
Drug Recall
Subcategory:
Drugs
Hazard classification:
Type I and II
Source of recall:
Health Canada
Issue:
Product Safety
Audience:
General Public, Healthcare Professionals, Hospitals
Identification number:
RA-26793
Recalled Products
A. Alysena 28
Reason
Further to the previous recall posted on April 8, 2013, Apotex is recalling additional lots of Alysena-28 birth control pills as a precautionary measure.
Lot LF01899A was originally recalled as packaged product may contain two rows of white (placebo) tablets (14 tablets) and two rows of pink (active) contraceptive tablets (14 tablets) in the blister pack.
Correct packaging is one row of white (placebo) tablets (7 tablets) and three rows of pink (active) contraceptive tablets (21 tablets) in the blister pack.
All lots distributed to date are being recalled as a precautionary measure while the firm investigates a packaging defect found in the original recalled lot (LF01899A).
Type I risk to health for patients who should not get pregnant, whether for medical reasons or exposure to agents detrimental to a developing fetus.
Type II risk for the general population.
Depth of distribution
Wholesalers/distributors and retailers in AB, BC, MB, NB, NF, NS, NT, ON, PE, QC, SK.
Affected products
A. Alysena 28
DIN, NPN, DIN-HIM
DIN 02387883
Dosage form
Tablets
Strength
100 mcg levonorgestrel and 20 mcg ethinyl estradiol
Lot or serial number
- LF01899A
- LF01898A
- LF01894B
- LF01901A
- LF01900A
- LF01980A
- LF01982A
- LF01981A
- LF01979A
- LF02037A
- LF02036A
- LF02026A
Companies
- Recalling Firm
- Apotex Inc.
150 Signet Drive
Toronto
M9L 1T9
Ontario
CANADA
- Apotex Inc.
- Marketing Authorization Holder
- Apotex Inc.
150 Signet Drive
Toronto
M9L 1T9
Ontario
CANADA
- Apotex Inc.
Appendix 5: Media Reports
Table of Contents
- 40 Canadiennes enceintes poursuivent un fabricant de pilules contraceptives
- Des femmes enceintes réclament 800 M$ PILULE DÉFECTUEUSE
- PHARMACEUTICALS Class action hits maker of recalled birth control pills
- Les usines d'Apotex sont sûres, dit Aglukkaq; La FDA a mis au jour des "déficiences récurrentes" dans le contrôle de la qualité
- Canuck drug maker warned
- Canuck drug maker warned
- PHARMACEUTICALS U.S. regulator flags problems at Canadian drug maker Apotex warned exports could be blocked
- Word spreading on birth control packaging flaw
- Word spreads on packaging flaw
- Apotex recalls all Alysena-28 birth-control pills as precaution -
- Double-check birth control prescriptions, health unit says
- Drug Recall
- Apotex widens recall of Alysena-28 birth control
- Contraceptif rappelé
- Nouveau rappel de pilule contraceptive
- Uh oh! Another birth control recall
- Birth control recall expanded; After extra placebo pills found, Apotex recalling 11 additional lots of Alysena-28
- Pill recall widens
- Wrong birth control dosage found in Alysena; Gynecology group urges women to use backup contraception if they took faulty pill in generic brand
- Ottawa to investigate birth-control pill recall Health
- Feds probe slow pill recall Public not notified promptly enough?
- Probe ordered over defective pills Birth control manufacturer allegedly failed to notify public
- Birth control pill recall investigated
- BIRTH CONTROL Minister orders inquiry into lack of urgency in Pill recall
- Health workers spread message of birth control pill recall; Recall One lot of generic birth control pills contained extra week of placebo
- Backup birth control urged for women who had faulty pill packs
- BIRTH CONTROL Some women may not realize they're taking recalled Pill
- La réaction de la ministre critiquée CONTRACEPTIF ALYSENA 28
- Women on faulty pill should use backup
- Women who took faulty 'pill' urged to used backup contraception
- Backup contraception urged amid recall; Gynecology group offers advice after faulty birth control pill packs recalled
- Gynecologists urge women to use backup contraception if they took faulty pills
- Women advised to use backup if they took faulty 'pill'
- Birth-control pill alysena 28 recalled
- Faulty-pill users need other birth control
- Rappel urgent de pilules contraceptives Alysena 28
- Use of recalled birth control pills puts women at risk of pregnancy
- Urgent rappel de pilules contraceptives
- Rappel urgent
- RAPPEL DES PILULES CONTRACEPTIVES ALYSENA 28
- Rappel de pilules contraceptives Alysena 28
- Health Canada recalls birth control pills; Extra placebo tablets placed in packages of Alysena 28 pills
- Birth Control Recall
- Birth control pills recalled; Canadian women were left in the dark for about a week about pill's effectiveness
- Birth control error sparks fear, anger; Health Canada waited five days before ordering retailers to notify patients
- MEDICINE Women not alerted immediately of birth-control recall
- Drug firm recalls Alysena-28 birth control pills
- Maker issues urgent recall of contraceptive Alysena over packaging mix-up
- Birth-control pill recalled
- Alysena birth-control pills recalled
- Recall of contraceptive pill issued
- National; Urgent recall issued on birth-control pill
- Birth control pill recalled after concerns of unplanned pregnancies
- Alysena 28 birth control pill recalled
- Rappel urgent du contraceptif Alysena 28
40 Canadiennes enceintes poursuivent un fabricant de pilules contraceptives
- Sourcetag
- 130518293081597
- Publication:
- Le Journal de Québec
- Date:
- 2013.05.18
- Edition:
- final
- Section:
- Nouvelles
- Page:
- 37
- Byline:
- AGENCE FRANCE-PRESSE
- Dateline:
- OTTAWA
- Word Count:
- 150
-- Un groupe de Canadiennes, dont 40 sont devenues enceintes alors qu'elles prenaient une pilule contraceptive, ont lancé un recours collectif contre une société pharmaceutique pour lui réclamer 800 millions de dollars, ont rapporté, hier, les médias. Soixante citoyennes de la province de l'Ontario, dont quatre ont subi des avortements, participent à cette action en justice contre la société Apotex, fabricant de la pilule Alysena, selon l'avocat des plaignantes, MeSandy Zaitseff, cité par le quotidien de référence The Globe and Mail. 50 000 boîtes d'Alysena avaient été rappelées le 8 avril, lorsqu'on avait découvert qu'un lot contenait deux rangées de pilules de placebo au lieu d'une. Cinq jours plus tard, le rappel a été étendu à 11 autres lots. La société pharmaceutique n'avait pas rendu public le premier rappel et la ministre fédérale de la Santé, Leona Aglukkaq, a demandé à ses services d'ouvrir une enquête sur cette absence d'information.
Des femmes enceintes réclament 800 M$ PILULE DÉFECTUEUSE
- Sourcetag
- 130518293080848
- Publication:
- Le Journal de Montréal
- Date:
- 2013.05.18
- Edition:
- final
- Section:
- Nouvelles
- Page:
- 41
- Byline:
- AFP
- Dateline:
- OTTAWA
- Word Count:
- 178
- Circulation:
- 245993
OTTAWA -- (AFP) Un groupe de Canadiennes, dont 40 sont tombées enceintes alors qu'elles prenaient une pilule contraceptive, ont lancé un recours collectif contre une société pharmaceutique pour lui réclamer 800 millions de dollars, ont rapporté hier les médias.
Soixante habitantes de l'Ontario, dont quatre ont subi des avortements, participent à cette action en justice contre la société Apotex, fabricant de la pilule Alysena, selon l'avocat des plaignantes, MeSandy Zaitseff, cité par le quotidien de référence The Globe and Mail. 50 000 boîtes d'Alysena avaient été rappelées le 8 avril dernier, lorsqu'on avait découvert qu'un lot contenait deux rangées de pilules de placebo au lieu d'une. Cinq jours plus tard, le rappel a été étendu à 11 autres lots.
La société pharmaceutique n'avait pas rendu public le premier rappel et la ministre fédérale de la Santé Leona Aglukkaq a demandé à ses services d'ouvrir une enquête sur cette absence d'information.
MeZaitseff a souligné que l'erreur d'emballage avait pour ses clientes des conséquences financières se chiffrant à des millions de dollars, et aussi leur posait de graves problèmes éthiques.
PHARMACEUTICALS Class action hits maker of recalled birth control pills
- Publication:
- GLOBE AND MAIL
- IDN:
- 131370086
- Date:
- 2013.05.17
- Page:
- A3
- Byline:
- Justin Fauteux
- Section:
- National News
- Edition:
- Metro
- Dateline:
- Toronto ONT
- Words:
- 281
- Word Count:
- 300
- Circulation:
- 321109
JUSTIN FAUTEUX TORONTO A group of Ontario women, including 40 who are pregnant, have launched an $800-million class action against Apotex Inc., the maker of the recalled Alysena birth control pill.
The lawsuit involves about 60 women, among whom there have been 40 unwanted pregnancies and four abortions, according to Sandy Zaitzeff, a lawyer with Thunder Bay firm Watkins Law Professional Corporation, which launched the suit.
"They're angry, they're distressed, they're worried," Mr. Zaitzeff said of his clients. "There's a lot of stress in something like this." A total of 50,000 packages of the Alysena birth control pill with the lot number LF01899A were recalled April 8 when it was discovered that some packs contained two rows of placebo pills instead of just one. On April 13, the recall was expanded to 11 other lot numbers.
Apotex did not notify customers of the initial recall for five days after the problem was identified, prompting federal Health Minister Leona Aglukkaq to order an investigation into the delay on April 11.
Mr. Zaitzeff said the first plaintiff came to his firm last month, and since then the numbers have grown. He added that his clients come from across Canada and that he expects the number of women involved in the case to increase.
"The damages are substantial," he said, adding that his clients are facing financial, ethical, moral and health issues. "An unwanted pregnancy - to raise a child today is millions of dollars. Just because somebody had sex with another individual in today's world does not mean that they should be burdened with the costs of raising a child." A request for comment from Apotex was not immediately returned.
ADDED SEARCH TERMS:
GEOGRAPHIC NAME: Ontario
SUBJECT TERM:birth control; damage suits; health; pharmaceutical industry; pregnancy; women
ORGANIZATION NAME: Apotex Inc.
Les usines d'Apotex sont sûres, dit Aglukkaq; La FDA a mis au jour des "déficiences récurrentes" dans le contrôle de la qualité
- Publication:
- Le Devoir
- Date:
- 2013.04.30
- Section:
- Actualités
- Page:
- A2
- Byline:
- Amélie Daoust-Boisvert
- Word Count:
- 224
- Circulation:
- 26658
Ottawa affirme que les médicaments génériques fabriqués dans deux usines d'Apotex en Ontario ne posent aucun risque pour la santé des Canadiens, même si la Food and Drug Administration (FDA) américaine a mis au jour des " déficiences récurrentes " dans leur système de contrôle de la qualité.
" Après avoir inspecté les installations, Santé Canada m'a indiqué n'avoir aucune preuve d'effet pour la santé des Canadiens ", a répondu lundi la ministre fédérale de la Santé, Leona Aglukkaq, à une question du NPD à la Chambre des communes.
En février dernier, la FDA a écrit au président d'Apotex inc. pour lui faire part de ses préoccupations concernant ses usines de Toronto et de Richmond Hill. Dans sa lettre, la FDA parle de " violations significatives des meilleures pratiques ". La FDA craint, entre autres choses, une déficience dans la prévention de la contamination microbienne. L'agence américaine a indiqué au Globe and Mail qu'elle révise actuellement la réponse d'Apotex à ses préoccupations.
Sandoz
L'an dernier, la fermeture de l'usine de médicaments génériques Sandoz de Boucherville après une inspection de la FDA avait considérablement compliqué l'approvisionnement pour certains médicaments.
Pour le NPD, il est très préoccupant que ce soient les autorités américaines qui sonnent l'alarme concernant les usines canadiennes de fabrication de médicaments.
C'est Apotex qui distribue au Canada le contraceptif Alysena, objet récent d'un rappel pour un problème de dosage.
Canuck drug maker warned
- Sourcetag
- 130427290132720
- Publication:
- The Toronto Sun
- Date:
- 2013.04.27
- Edition:
- Final
- Section:
- News
- Page:
- 29
- Byline:
- QMI AGENCY
- Word Count:
- 122
- Circulation:
- 133452
American regulators have repeatedly warned a Canadian drug maker of "significant violations" in its manufacturing and quality control--the latest coming two months before the same manufacturer recalled defective birth control pills and became the subject of a Health Canada investigation.
In a harshly worded letter dated Feb. 21, the U.S. Food and Drug Administration told Toronto-based Apotex it might block the import of some drugs until corrections are made. "FDA's inspections continue to find repeated deficiencies in your quality systems," the letter says.
Apotex has been under scrutiny for its handling of a recall earlier this month of Alysena-28 birth control pills that had too many placebo tablets and not enough active tablets.
Apotex hasn't returned QMI Agency's requests for comment.
Canuck drug maker warned
- Sourcetag
- 130427290132639
- Publication:
- The Ottawa Sun
- Date:
- 2013.04.27
- Edition:
- Final
- Section:
- News
- Page:
- 20
- Byline:
- QMI AGENCY
- Word Count:
- 124
- Circulation:
- 32186
American regulators have repeatedly warned a Canadian drug maker of "significant violations" in its manufacturing and quality control -- the latest coming two months before the same manufacturer recalled defective birth control pills and became the subject of a Health Canada investigation.
In a harshly worded letter dated Feb. 21, the U.S. Food and Drug Administration told Toronto-based Apotex it might block the import of some drugs until corrections are made. "FDA's inspections continue to find repeated deficiencies in your quality systems," the letter says.
Apotex has been under scrutiny for its handling of a recall earlier this month of Alysena-28 birth control pills that had too many placebo tablets and not enough active tablets.
Apotex hasn't returned QMI Agency's requests for comment.
PHARMACEUTICALS U.S. regulator flags problems at Canadian drug maker Apotex warned exports could be blocked
- Publication:
- GLOBE AND MAIL
- IDN:
- 131160100
- Date:
- 2013.04.26
- Page:
- A1
- Byline:
- CARLY WEEKS
- Section:
- National News
- Edition:
- Metro
- Dateline:
- Words:
- 761
- Word Count:
- 776
- Circulation:
- 321109
CARLY WEEKS U.S. regulators are threatening to block imports of some drugs produced by Canada's largest maker of generics after investigators raised questions about quality control at two of its Toronto-area manufacturing facilities.
In a February warning obtained by The Globe and Mail, the U.S. Food and Drug Administration said its inspectors have found "repeated deficiencies" in quality-control systems at Apotex Inc.'s facilities.
Foreign inspectors at the facilities found that Apotex released drugs into the U.S. market, despite defects discovered in part of the same batch.
They also found that the drug maker did not adequately control or detect microbial growth, which could compromise drug safety.
The warning did not cite specific drugs and did not say what health risks the lapses created for the public.
But the findings shed light on a regulatory disparity after the U.S. raised a flag and Canada didn't.
Apotex, which declined several requests for interviews, said in a statement that it is working with the FDA to address its concerns.
Health Canada said it is not concerned by the U.S. warnings, although Canadian inspectors have not inspected the facilities in question since 2011.
FDA investigators conducted the inspections last August and October at Apotex plants in Richmond Hill and Toronto.
The investigators gave the plants a failing grade and told Apotex it had to address the issues.
But in a warning letter sent to the company in February, 2013, the FDA said Apotex has not addressed the violations and that an import ban may be put on products coming out of those two plants.This is the second time Apotex has been warned by U.S. regulators in recent years. In 2009, the FDA imposed a two-year import ban on Apotex after inspectors found major deficiencies in manufacturing practices, notably failure to investigate how drugs that were rejected for public use became contaminated.
Apotex and Health Canada have been under pressure for their handling of a massive birth-control recall, which was triggered this month after a customer discovered an extra week of sugar pills in a package of Alysena. The dosage problem - which could dramatically increase the likelihood of pregnancy - was identified on April 3, but an urgent public warning was not issued by Health Canada until April 8. Spanish firm Laboratorios Leon Farma manufactured the pills, which were distributed in Canada by Apotex.
Federal Health Minister Leona Aglukkaq has ordered Health Canada to investigate the time lag and what factors led to the delayed warning.
Apotex has submitted its responses to the FDA, the company's statement said, and is "very confident that they will be resolved successfully in a short period of time." An FDA spokesman said the agency is reviewing Apotex's response.
The problems identified at Apotex's manufacturing facilities should have Canadians questioning the safety of the drug supply, said Joel Lexchin, a health-policy professor at York University and one of Canada's leading experts on drug safety.
"We should not have to go to the Americans to find out what's going on at plants here in Canada," Prof. Lexchin said.
In an e-mailed statement, Health Canada spokeswoman Blossom Leung said the department found "no evidence of impacts on health products destined for the Canadian market." Ms. Leung said the violations identified by the FDA relate to drugs that never made it to market, which means there is no cause for alarm. In its warning, the FDA criticized Apotex for failing to discover the source of contamination in numerous drug batches that were rejected for public use.
She added that Health Canada is overseeing the corrective actions being put in place and "continues to have oversight" on the company's progress.
The department is scheduled to inspect both plants in May and October of this year.
In the letter dated Feb. 21, 2013, the FDA said Apotex kept a batch of drugs on the market despite the fact a separate batch manufactured on the same filling line failed a sterility test. The inspection also cited instances where Apotex released questionable drugs into the market. In one case, an Apotex employee identified a "critical defect" with drug tablets and held them back. But the rest of the batch was released.
"Please explain the basis for your conclusion that the only affected part of the batch was the rejected portion," the FDA warning letter said. "Your firm's practice of rejecting portions of drug product batches is an indication that your firm does not have well-controlled manufacturing processes."
ADDED SEARCH TERMS:
GEOGRAPHIC NAME: Canada; United States
SUBJECT TERM:hazardous products; inspection; manufacturing; pharmaceutical industry; regulation
ORGANIZATION NAME: Apotex Inc.; Food and Drug Administration; Health Canada
Word spreading on birth control packaging flaw
- Idnumber
- 201304170070
- DocID:
- 166218933
- Publication:
- New Brunswick Telegraph-Journal
- Page:
- A3
- Date:
- 2013.04.17
- Section:
- Main
- Byline:
- Chris Morris Legislature Bureau
- Copyright:
- © 2013 Telegraph-Journal (New Brunswick)
- Word Count:
- 379
- Circulation:
- 33080
FREDERICTON - New Brunswick pharmacists are busy getting the word out about possibly flawed packages of a particular birth control drug following an expanded recall. Health Canada has issued an advisory that Apotex Inc. will be recalling 11 additional lots of Alysena birth control pills as a precautionary measure.
Apotex is making the additional recall while it investigates why one lot of Alysena-28 - LF01899A - contained packages with extra placebo pills in place of active pills.
Packages of Alysena-28 should have three rows of the pink active pills and only one row of white placebo pills.
The expanded recall concerns Alysena-21 packages which do not normally include a row of the placebo pills.
However, because the product is made in the same facility, Health Canada and Apotex are working together to verify this product as well.
Sam Lanctin of the New Brunswick Pharmaceutical Society said in an interview Monday the society has been busy advising pharmacists and public health officials about the recall.
He said it's important to make sure high school and university students also are aware of the recalls.
"It's about getting the word out," Lanctin said.
"I don't doubt some people will be shaken by the recall and I appreciate that. But overall we have a very safe health-care system and certainly with drugs and this type of recall, they are quite rare."
Canadians are encouraged to check to make sure their packages of Alysena-21 contain three rows of pink pills.
Consumers are also strongly encouraged to report any medical problems associated with the use of Alysena-28 or Alysena-21 to Health Canada.
The specific lots involved in the new recall are LF01901A, LF01980A, LF02037A, LF01900A, LF01982A, LF01981A, LF02026A, LF01898A, LF02036A, LF01894B and LF01979A.
Women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being, the Society of Obstetricians and Gynecologists of Canada has suggested.
And women may need to talk with their doctors about taking a pregnancy test, said Dr. Jennifer Blake, the organization's CEO.
The faulty lot of Alysena-28, which contained about 50,000 packets, was distributed across Canada.
Lanctin said there is no way of knowing how many packages, if any, ended up in New Brunswick. -with files from The Canadian Press
Word spreads on packaging flaw
- Idnumber
- 201304170088
- DocID:
- 166218772
- Publication:
- Times & Transcript (Moncton)
- Page:
- B4
- Date:
- 2013.04.17
- Section:
- International
- Byline:
- legislature bureau ?
- Copyright:
- © 2013 Times & Transcript (Moncton)
- Word Count:
- 245
- Circulation:
- 36505
FREDERICTON - New Brunswick pharmacists are busy getting the word out about possibly flawed packages of a particular birth control drug following an expanded recall. Health Canada has issued an advisory that Apotex Inc. will be recalling 11 additional lots of Alysena birth control pills as a precautionary measure.
Apotex is making the additional recall while it investigates why one lot of Alysena-28 - LF01899A - contained packages with extra placebo pills in place of active pills.
Packages of Alysena-28 should have three rows of the pink active pills and only one row of white placebo pills.
The expanded recall concerns Alysena-21 packages which do not normally include a row of the placebo pills.
However, because the product is made in the same facility, Health Canada and Apotex are working together to verify this product as well.
Sam Lanctin of the New Brunswick Pharmaceutical Society said in an interview Monday the society has been busy advising pharmacists and public health officials about the recall.
He said it's important to make sure high school and university students also are aware of the recalls. "It's about getting the word out," Lanctin said.
"I don't doubt some people will be shaken by the recall and I appreciate that. But overall we have a very safe health-care system and certainly with drugs and this type of recall, they are quite rare." Canadians are encouraged to check to make sure their packages of Alysena-21 contain three rows of pink pills.
Apotex recalls all Alysena-28 birth control pills as precaution -
- Publication:
- WINNIPEG FREE PRESS
- Date:
- 2013.04.17
- Page:
- B6
- Word Count:
- 289
- Circulation:
- 121150
A pharmaceutical company has recalled all lots of the birth control pill Alysena-28, including product sold at some Manitoba pharmacies.
A week ago, Apotex Inc. recalled one lot of Alysena-28 birth control pills (LF01899A) because the company incorrectly packaged extra white placebo bills with pink active pills.
Now, as a precaution, the company has pulled all lots of Alysena-28 off the shelves.
Packages of Alysena-28 should have three rows of the pink active pills and only one row of white placebo pills. The faulty blister pack that was returned to Apotex had 14 pink pills (active drug) and 14 white placebos.
Missing a week of the active pink tablets would place an individual at the same risk level of becoming pregnant as an individual who is not using contraception, an alert on Apotex's website states.
Gynecologists warn some women may not know they were taking the recalled contraceptive. That's because the drug is a generic version of the most popular birth control pill used in Canada, a drug called Alesse, which is not involved in the recall.
Some prescription-drug plans require pharmacists to replace higher-priced brand-name drugs with generic versions if a generic is available.
That means women might think they are taking Alesse -- because that is what their doctor prescribed -- but they are actually taking Alysena.
Canadians using this product should use a non-hormonal method of birth control, contact their health-care provider for medical advice and return unopened packages to their pharmacist.
Apotex has also established a toll-free recall information service at 1-866-367-4537 to provide patients and health-care professionals with additional support.
The additional recalled medication have the following lot numbers: LF01901A, LF01900A, LF01898A, LF01894B, LF01980A, LF01982A, LF01981A, LF01979A, LF02037A, LF02036A and LF02026A.
-- staff / The Canadian Press
Double-check birth control prescriptions, health unit says
- Idnumber
- 201304160015
- DocID:
- 166124724
- Publication:
- Windsor Star
- Page:
- A3
- Date:
- 2013.04.16
- Section:
- News
- Edition:
- Final
- Byline:
- Beatrice Fantoni
- Source:
- The Windsor Star; With Files From The Canadian Press
- Word Count:
- 268
- Circulation:
- 67530
The Windsor-Essex County Health Unit is encouraging locals to double-check their birth control prescriptions if they were issued by the WECHU clinic to make sure they aren't affected by the recent recall of a generic brand of birth control pill, Alysena-28.
The WECHU does not prescribe Alysena-28. Instead, it prescribes Alesse. However, it is possible that local pharmacists will substitute Alysena-28 for Alesse, since it's a cheaper generic version of the same contraceptive, the WECHU said in a news release Monday.
Alesse pills are not affected by the recall, but the health unit is urging patients who have been prescribed birth control pills at the clinic to double check which pill they have and whether it's part of the recalled lots.
Last week, Health Canada and the drug manufacturer Apotex recalled a series of lots of Alysena-28 pills because they mistakenly included too many placebo pills and too few active pills.
Packages of Alysena-28 should have three rows of the pink active pills and only one row of white placebo pills.
Canadians using this product should use a non-hormonal method of birth control, con-tact their health care provider for medical advice and return unopened packages to their pharmacist.
Alysena-21, which is manufactured at the same facility, is not part of the current recall as these packages don't normally include a row of the placebo pills. However, because the product is made in the same facility, Health Canada and Apotex are working together to verify this product as well.
The lots involved in the new recall are: LF01901A, LF01980A, LF02037A, LF01900A, LF01982A, LF01981A, LF02026A, LF01898A, LF02036A, LF01894B, and LF01979A.
Drug Recall
- Publication:
- CTV - CTV News
- Date:
- 2013.04.13
- Time:
- 23:00:00 ET
- End:
- 23:30:00 ET
- Word Count:
- 74
SANDIE RINALDO: A Toronto drug company has expanded its recall of potentially faulty birth control pills, while it investigates while some packages were found with too many placebo pills. A single lot of Alysena 28 was first recalled earlier this week. Today, Apotex added another eleven lots to the recall. The move is being described as precautionary. Health Canada has asked the company to explain why it took five days to alert the public.
Apotex widens recall of Alysena-28 birth control
- Idnumber
- 201304140080
- DocID:
- 166044799
- Publication:
- Toronto Star
- Page:
- A2
- Date:
- 2013.04.14
- Section:
- News
- Edition:
- ONT
- Byline:
- THE CANADIAN PRESS
- Copyright:
- © 2013 Torstar Corporation
- Word Count:
- 208
- Circulation:
- 331504
OTTAWA-Health Canada says Apotex Inc. will be recalling 11 additional lots of Alysena-28 birth control pills as a precautionary measure. Apotex is making the additional recall while it investigates why one lot - LF01899A - of the product contained packages with extra placebo pills in place of active pills. Packages of Alysena-28 should have three rows of the pink active pills and only one row of white placebo pills. Canadians using this product should use a non-hormonal method of birth control, contact their health-care provider for medical advice and return unopened packages to their pharmacist. Alysena-21, which is manufactured at the same facility, is not part of the current recall as these packages don't normally include a row of the placebo pills. However, because the product is made in the same facility, Health Canada and Apotex are working together to verify this product as well. In the meantime, Canadians are encouraged to check to make sure their packages of Alysena-21 contain three rows of pink pills. Consumers are also strongly encouraged to report any medical problems associated with the use of Alysena- 28 or Alysena-21 to Health Canada. The specific lots involved in the new recall are LF01901A, LF01980A, LF02037A, LF01900A, LF01982A, LF01981A, LF02026A, LF01898A, LF02036A, LF01894B and LF01979A.
Contraceptif rappelé
- Sourcetag
- 130414288297804
- Publication:
- Le Journal de Québec
- Date:
- 2013.04.14
- Edition:
- final
- Section:
- Nouvelles
- Page:
- 30
- Byline:
- AGENCE QMI
- Dateline:
- OTTAWA
- Word Count:
- 140
-- (QMI) Le contraceptif oral Alysena 28, distribué par l'entreprise Apotex inc., fera l'objet d'un rappel à titre préventif, a annoncé Santé Canada.
Le 9 avril, un lot de plaquettes de 28 comprimés avait été retiré du marché, car il contenait deux rangées de placebos, au lieu d'une seule. Le fabricant retire maintenant 11 nouveaux lots et cherche à déterminer pourquoi certaines plaquettes ne contenaient pas les 21 comprimés actifs habituels. Une plaquette de 28 pilules doit contenir sept placebos et 21 cachets actifs.
Vérifications
Les plaquettes de 21 comprimés ne font pas partie du rappel, puisqu'elles ne contiennent pas de placebos. Les femmes sont tout de même encouragées à vérifier si leur plaquette contient bien trois rangés de comprimés roses.
Voici les lots faisant l'objet d'un rappel : LF01901A, LF01900A, LF01898A, LF01894B, LF01980A, LF01982A, LF01981A, LF01979A, LF02037A, LF02036A, LF02026A.
Nouveau rappel de pilule contraceptive
- Publication:
- Le Soleil
- Date:
- 2013.04.14
- Section:
- Actualités
- Page:
- 28
- Source:
- La Presse
- Byline:
- Philippe Teisceira-Lessard
- Dateline:
- Montréal
- Word Count:
- 479
- Circulation:
- 88538
Santé Canada a annoncé vendredi le rappel par une pharmaceutique de 11 nouveaux lots de la populaire pilule contraceptive Alysena 28, de crainte qu'un problème d'emballage n'entraîne des grossesses imprévues.
Au début de la semaine, un lot avait déjà été rappelé parce que certains emballages contenaient plus de placebos que prévu. Plutôt que 21 comprimés contraceptifs et 7 comprimés placebos, des emballages contenaient 14 comprimés de chacun.
Le rappel supplémentaire a été effectué "à titre préventif" par Apotex Canada, qui fabrique le produit, selon Santé Canada. En date de vendredi, l'entreprise continuait à chercher "la raison pour laquelle" le premier lot avait été mal emballé.
Apotex n'a pas rappelé La Presse. Il n'a pas été possible de connaître le lien entre les 11 lots rappelés vendredi et ceux qui l'ont été plus tôt cette semaine.
Selon Elaine E. Jolly, professeure à la Faculté de médecine de l'Université d'Ottawa et gynécologue, l'Alysena est l'un des contraceptifs hormonaux les "plus populaires" sur le marché.
Placebo
"C'est très utilisé. C'est probablement l'un des meilleurs. Et c'est un problème sérieux s'il y a plus de sept placebos", a-t-elle affirmé au cours d'une entrevue téléphonique.
Comme il s'agit d'une pilule de "deuxième génération" - avec des concentrations d'hormones très basses - , le risque de tomber enceinte est réel si un seul comprimé est manqué, a expliqué la gynécologue.
La communauté scientifique s'interrogeait déjà quant à savoir si sept comprimés placebos par cycle étaient un nombre trop élevé. "Plusieurs docteurs demandent à leurs patientes de prendre le placebo ou d'arrêter la pilule pendant une période de seulement quatre jours" avant de recommencer, a-t-elle ajouté.
Si le débat continue sur ce point, il est certain que "si vous prenez le placebo pendant 8, 9 ou 10 jours, il y a un véritable risque de grossesse non désirée", selon Mme Jolly.
De tels rappels de médicaments sont "très rares", a indiqué la pharmacienne de garde. Même son de cloche du côté d'Elaine E. Jolly. C'est la première fois qu'elle entendait parler du rappel d'emballages de pilules contraceptives en raison du nombre de placebos qui s'y trouvent.
Apotex
Ce n'est pas la première fois qu'Apotex fait les manchettes pour des problèmes dans la distribution de ses médicaments. En 2011, des professionnels de la santé déploraient une grave pénurie de Misoprostol, un médicament très utilisé en obstétrique. Ils montraient du doigt une décision financière prise par Apotex, car le produit se vendait très peu cher. L'entreprise est aussi celle qui a fait tomber le brevet canadien de Pfizer sur le Lipitor, le médicament le plus vendu sur la planète.
Les produits rappelés portent les numéros de lot suivants : LF01901A, LF01980A, LF02037A, LF01900A, LF01982A, LF02036A, LF01898A, LF01981A, LF02026A, LF01894B, LF01979A et LF01899A.
Santé Canada conseille aux femmes qui ont ces produits entre les mains de communiquer avec leur médecin. D'ici là, elles devraient "utiliser une méthode contraceptive auxiliaire non hormonale".
Uh oh! Another birth control recall
- Sourcetag
- 130414288297663
- Publication:
- The Calgary Sun
- Date:
- 2013.04.14
- Edition:
- Final
- Section:
- News
- Page:
- 26
- Byline:
- QMI AGENCY
- Word Count:
- 138
- Circulation:
- 69586
Apotex Inc. is recalling more packages of its Alysena-28 birth control pills while it investigates why one lot of the product contained packages with extra placebo pills in place of active pills, Health Canada said Saturday.
The announcement came a day after Health Minister Leona Aglukkaq ordered an investigation after the Toronto-based company recalled defective birth control pills but allegedly failed to notify the public until five days later. In a statement to QMI Agency Friday, Aglukkaq said she's "concerned" Canadians may not have received important information in a timely manner. On April 8, Health Canada issued a recall of Alysena-28 because packages could contain two weeks of placebo pills and two weeks of contraceptive pills -- instead of one week's placebos and three weeks of contraceptives.
Apotex Inc. didn't issue a public advisory until the next day.
Birth control recall expanded; After extra placebo pills found, Apotex recalling 11 additional lots of Alysena-28
- Idnumber
- 201304140002
- DocID:
- 165992604
- Publication:
- Times Colonist (Victoria)
- Page:
- A10
- Date:
- 2013.04.14
- Section:
- Canada
- Edition:
- Final
- Source:
- The Canadian Press
- Dateline:
- OTTAWA
- Word Count:
- 208
Health Canada says Apotex Inc. will be recalling 11 additional lots of Alysena-28 birth control pills as a precautionary measure.
Apotex is making the additional recall while it investigates why one lot - LF01899A - of the product contained packages with extra placebo pills in place of active pills.
Packages of Alysena-28 should have three rows of the pink active pills and only one row of white placebo pills.
Canadians using this product should use a non-hormonal method of birth control, contact their health-care provider for medical advice and return unopened packages to their pharmacist.
Alysena-21, which is manufactured at the same facility, is not part of the current recall as these packages don't normally include a row of the placebo pills.
However, because the product is made in the same facility, Health Canada and Apotex are working together to verify this product as well.
In the meantime, Canadians are encouraged to check to make sure their packages of Alysena21 contain three rows of pink pills.
Consumers are also strongly encouraged to report any medical problems associated with the use of Alysena-28 or Alysena-21 to Health Canada.
The specific lots involved in the new recall are: LF01901A, LF01980A, LF02037A, LF01900A, LF01982A, LF01981A, LF02026A, LF01898A, LF02036A, LF01894B, and LF01979A.
Pill recall widens
- Sourcetag
- 130414288297562
- Publication:
- The Toronto Sun
- Date:
- 2013.04.14
- Edition:
- Final
- Section:
- News
- Page:
- 24
- Byline:
- QMI AGENCY
- Word Count:
- 197
- Circulation:
- 246898
Apotex Inc. is recalling more packages of its Alysena-28 birth control pills while it investigates why one lot of the product contained packages with extra placebo pills in place of active pills, Health Canada said Saturday.
The announcement came a day after Health Minister Leona Aglukkaq ordered an investigation after the Toronto-based company recalled defective birth control pills but allegedly failed to take steps to notify the public until five days later. In a statement to QMI Agency Friday, Aglukkaq said she's "concerned" Canadians may not have received important information in a timely manner. "I have instructed Health Canada to look into the issue," she said.
On April 8, Health Canada issued a recall of Alysena-28 because packages could contain two weeks of placebo pills and just two weeks of contraceptive pills -- instead of one week's worth of placebos and three weeks of contraceptives.
Apotex Inc. didn't issue a public advisory until the next day--even though it had sent an "urgent drug recall" letter to wholesalers, distributors and retailers April 3 regarding pill packages with serial/lot number of LF01899A that were sold in B.C., N.B., Nova Scotia, Newfoundland, Ontario, P.E.I. and Quebec.
Wrong birth control dosage found in Alysena; Gynecology group urges women to use backup contraception if they took faulty pill in generic brand
- Publication:
- The Telegram (St. John's)
- Date:
- 2013.04.13
- Section:
- Health & Fitness
- Page:
- D4
- Source:
- The Canadian Press
- Byline:
- Helen Branswell
- Dateline:
- TORONTO
- Illustration:
- Birth control pills Alysena28 in it's correct dosage. -Photo by The Canadian Press
- Word Count:
- 894
- Circulation:
- 41759
Women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being, the Society of Obstetricians and Gynecologists of Canada has suggested.
And women may need to talk with their doctors about taking a pregnancy test, said Dr. Jennifer Blake, the organization's CEO.
The advice is prompted by a major birth control recall involving the drug Alysena 28.
One lot of the product, bearing the number LF01899A, contained too little active drug and too much placebo, leaving women who took it vulnerable to becoming pregnant.
The lot, which contained about 50,000 faulty packets, was distributed across Canada. The company that sells the product in Canada, Apotex, believes it was not distributed to Alberta, Saskatchewan, Manitoba, the Northwest Territories and Nunavut, but it cannot say with certainty that is so, said Elie Betito, the company's director of public and government affairs.
Women using birth control pills take the drugs for 21 days each menstrual cycle. Because of the risk that they might not remember to resume taking their pills at the right time, many oral contraceptives are packaged with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
Blister packs in this faulty lot contained 14 pink pills containing active drug and 14 white placebos.
Lacking drug
And just that seven-day difference could be enough to result in a pregnancy, Blake said in an interview.
"That would mean two weeks without medication and that certainly gives enough time for the ovary to release an egg," she said. "And therefore if you have been taking that particular pill, Alysena, then it is important that you let your doctor know and, indeed, it may be important to follow up with a pregnancy test."
The problem is, some women may not know they were taking this contraceptive. That's because the drug is a generic version of the most popular birth control pill used in Canada, a drug called Alesse. (Alesse is not involved in this recall, Blake stressed.)
Some prescription drug plans require pharmacists to replace higher-priced brand-name drugs with generic versions if a generic is available.
That may make it difficult to get a sense of who was on this drug, Blake says.
It may also mean that women might think they are taking Alesse, because that is what their doctor prescribed, but they are actually taking Alysena.
"The problem is that it's a generic copy of the leading birth control pill. So that's a problem because we don't know how many women have had their prescription substituted for the generic. And women may not know they've had their prescription substituted, because the generic chose a very similar name and look to the leading birth control pill. And that does create confusion in everyone's mind."
Other methods
Blake said women who take this contraceptive should use other methods of birth control as well until they've gone a full month on a proper packet of the drug.
"I would regard it as being like a new start" (on oral contraceptives).
"If you have started a packet of Alysena this month, if that's what you're taking now, then you should be using an additional method and calling your doctor," she said. "If you took the faulty packet last month and you got your period, then that's very reassuring.
If you took the faulty packet last month and you did not get your period, then you certainly need to be getting a (pregnancy) test."
Blake said Plan B, the so-called morning-after pill, would also be an option for women who have had what amounts to unprotected sex because they were on a faulty pack of Alysena. But the drug only works within three to five days - and five is pushing it - after unprotected sex.
Alysena is manufactured in Spain by Leon Farma for Apotex. Betito said the company is working with the manufacturer to try to figure out what went wrong.
Seeking answers
Health Canada also wants answers to that question, said Dr. Supriya Sharma, senior medical adviser for Health Canada's health products and food branch.
"There does look like there was a problem in their good manufacturing processes. Otherwise this wouldn't have occurred," Sharma said. Apotex notified Health Canada of its intention to recall the product April 3.
As required, it submitted a full risk assessment to the department by late in the day April 4.
Health Canada finished assessing the file April 8, and posted a notice of the recall on its website.
The company felt the risk assessment for the recall was a Type 2 under Health Canada's rules - something that might cause temporary adverse health effects if used.
But Health Canada told the company it felt this merited a Type 1 designation.
A Type 1 risk is reserved for drugs that could cause serious injury or even death if used.
Sharma said that while the risk for many Alysena users was lower, some women using the drug might have been warned not to become pregnant because it might endanger their health or because they were on medication that would endanger the health of any fetus they conceived.
Recalls with a Type 1 designation involve notifying pharmacists and getting them to pass the message along to patients or to the prescribing doctor.
Ottawa to investigate birth-control pill recall Health
- Sourcetag
- 130413288165811
- Publication:
- The Kingston Whig-Standar
- Date:
- 2013.04.13
- Edition:
- Final
- Section:
- News
- Page:
- 13
- Byline:
- QMI AGENCY
- Word Count:
- 251
- Circulation:
- 27343
Health Minister Leona Aglukkaq has ordered an investigation after a manufacturer recalled defective birth control pills but allegedly failed to take steps to notify the public until five days later.
In a statement to QMI Agency Friday, Aglukkaq said she's "concerned" Canadians may not have received important information in a timely manner. "I have instructed Health Canada to look into the issue and assess whether processes were followed and that they are sufficient," she said.
On April 8, Health Canada issued a recall of Alysena-28 because packages could contain two weeks of placebo pills and just two weeks of contraceptive pills -- instead of one week's worth of placebos and three weeks of contraceptives.
Apotex Inc., based in Toronto, didn't issue a public advisory until the next day -- even though it had sent an "urgent drug recall" letter to wholesalers, distributors and retailers on April 3 regarding pill packages with serial/ lot number of LF01899A that were sold in British Columbia, New Brunswick, Nova Scotia, Newfoundland, Ontario, P.E.I. and Quebec.
The letter isn't available on the company's site, but was posted by the drugstore chain London Drugs on its blog on April 6 to advise its customers of the recall.
Under the Food and Drugs Act, it is the responsibility of the manufacturer or importer of a drug to notify Health Canada it has initiated a recall.
"Health Canada will be following up with the company to ensure that the recall was initiated properly," spokesman Blossom Leung said.
Feds probe slow pill recall Public not notified promptly enough?
- Sourcetag
- 130413288167293
- Publication:
- The Toronto Sun
- Date:
- 2013.04.13
- Edition:
- Final
- Section:
- News
- Page:
- 18
- Illustration:
- photo of LEONA AGLUKKAQ Investigation
- Byline:
- QMI AGENCY
- Word Count:
- 268
- Circulation:
- 133452
Health Minister Leona Aglukkaq has ordered an investigation after a manufacturer recalled defective birth control pills but allegedly failed to take steps to notify the public until five days later.
In a statement to QMI Agency Friday, Aglukkaq said she's "concerned" Canadians may not have received important information in a timely manner. "I have instructed Health Canada to look into the issue and assess whether processes were followed and that they are sufficient," she said.
On April 8, Health Canada issued a recall of Alysena-28 because packages could contain two weeks of placebo pills and just two weeks of contraceptive pills--instead of one week's worth of placebos and three weeks of contraceptives.
Apotex Inc., based in Toronto, didn't issue a public advisory until the next day -- even though it had sent an "urgent drug recall" letter to wholesalers, distributors and retailers on April 3 regarding pill packages with serial/lot number of LF01899A that were sold in B.C., New Brunswick, Nova Scotia, Newfoundland, Ontario, P.E.I. and Quebec.
The letter isn't available on the company's site but was posted by the drugstore chain London Drugs on its blog on April 6 to advise its customers of the recall.
Under the Food and Drugs Act, it is the responsibility of the manufacturer or importer of a drug to notify Health Canada it has initiated a recall.
"Health Canada will be following up with the company to ensure that the recall was initiated properly and will follow up to ensure that good manufacturing practices are followed," spokeswoman Blossom Leung said.
Apotex didn't return QMI Agency's request for comment.
Probe ordered over defective pills Birth control manufacturer allegedly failed to notify public
- Sourcetag
- 130413288167350
- Publication:
- The Ottawa Sun
- Date:
- 2013.04.13
- Edition:
- Final
- Section:
- Lifestyle
- Page:
- 31
- Illustration:
- photo of LEONA AGLUKKAQ
- Byline:
- QMI AGENCY
- Word Count:
- 255
- Circulation:
- 32186
Health Minister Leona Aglukkaq has ordered an investigation after a manufacturer recalled defective birth control pills but allegedly failed to take steps to notify the public until five days later.
In a statement to QMI Agency Friday, Aglukkaq said she's "concerned" Canadians may not have received important information in a timely manner. "I have instructed Health Canada to look into the issue and assess whether processes were followed and that they are sufficient," she said.
Last Monday, Health Canada issued a recall of Alysena-28 because packages could contain two weeks of placebo pills and just two weeks of contraceptive pills -- instead of one week's worth of placebos and three weeks of contraceptives.
Apotex Inc. , based in Toronto, didn't issue a public advisory until the next day -- even though it had sent an "urgent drug recall" letter to wholesalers, distributors and retailers on April 3 regarding pill packages with serial/lot No. LF01899A that were sold in British Columbia, New Brunswick, Nova Scotia, Newfoundland, Ontario, Prince Edward Island and Quebec.
The letter isn't available on the company's site but was posted by the drugstore chain London Drugs on its blog on April 6 to advise its customers of the recall.
Under the Food and Drugs Act, it's the responsibility of the manufacturer or importer of a drug to notify Health Canada it has initiated a recall.
"Health Canada will be following up with the company to ensure that the recall was initiated properly," spokesman Blossom Leung said. Apotex didn't return QMI Agency's request for comment.
Birth control pill recall investigated
- Sourcetag
- 130413288167511
- Publication:
- The Calgary Sun
- Date:
- 2013.04.13
- Edition:
- Final
- Section:
- News
- Page:
- 30
- Byline:
- QMI AGENCY
- Word Count:
- 128
- Circulation:
- 45600
Health Minister Leona Aglukkaq has ordered an investigation after a manufacturer recalled defective birth control pills but allegedly failed to take steps to notify the public until five days later.
In a statement to QMI Agency Friday, Aglukkaq said she's "concerned" Canadians may not have received important information in a timely manner. "I have instructed Health Canada to look into the issue and assess whether processes were followed and that they are sufficient," she said.
On April 8, Health Canada issued a recall of Alysena-28 because packages could contain two weeks of placebo pills and just two weeks of contraceptive pills -- instead of one week's worth of placebos and three weeks of contraceptives.
Apotex Inc., based in Toronto, didn't issue a public advisory until the next day.
BIRTH CONTROL Minister orders inquiry into lack of urgency in Pill recall
- Publication:
- GLOBE AND MAIL
- IDN:
- 131020094
- Date:
- 2013.04.12
- Page:
- L6
- Byline:
- CARLY WEEKS
- Section:
- Globe Life
- Edition:
- Metro
- Dateline:
- Words:
- 737
- Word Count:
- 748
- Circulation:
- 321109
CARLY WEEKS Federal Health Minister Leona Aglukkaq has ordered an investigation to determine why Canadian women were not immediately informed of a recall of potentially faulty birth control pills.
The department failed to notify Canadian women of the recall of Alysena 28, a generic birth control drug, for five days after the problem was identified. The risk of accidental pregnancy was not deemed serious enough to trigger an urgent product recall.
In a statement, Aglukkaq said she is "concerned" that women may not have received the information in a timely manner and has asked Health Canada to examine the issue and "assess whether processes were followed and that they are sufficient." Members of the medical community say they are angry and confused by what they describe as Health Canada's inadequate response. The incident risks undermining confidence in the safety of contraceptives, they say.
"This is just unprecedented," said Dr. Jennifer Blake, chief executive officer of the Society of Obstetricians and Gynecologists of Canada.
"We really regret and deplore that that has happened." Apotex Inc., the distributor of Alysena, learned on April 3 that the 28-day pill packs may contain an extra week of placebo, or sugar, pills. Typically, one week of sugar pills is included in 28-day packs to help women remember to take the medication every day. If women took two weeks of placebo pills instead of one, it would dramatically increase the risk of unplanned pregnancy.
After it learned of the problem, Apotex issued a "Class II" recall notice to pharmacies and other industry members. Health Canada learned of the issue the next day and posted the Class II recall notice, intended for health-care professionals, on its website over the weekend.
A Class II recall occurs when a product may cause temporary health issues or where the probability of serious problems is remote. But because Class II recalls are not considered life-or-death, the general public may not hear about them.
Last weekend, Health Canada conducted its own investigation and realized that some women who should not become pregnant for medical reasons might be at risk because of the potentially faulty pills.
For that reason, the department also issued a more serious Class I recall. Those types of recalls are reserved for instances when exposure to a product is reasonably likely to cause serious health problems or death.
On Monday, Health Canada posted the urgent Class I recall to its Healthy Canadians website, which e-mails details of product recalls to signed-up members of the media and the public. The department also tweeted the details of the recall.
Blossom Leung, a Health Canada spokeswoman, said the department would have encouraged pharmacists to contact women as soon as they learned of the recall last week.
However, the Canadian Pharmacists Association said the handling of this recall has led to widespread confusion among pharmacists.
Although some realized the severity right away, in many cases, they began reaching out to affected women only in the past few days.
"I really scratched my head on this one," said Phil Emberley, director of pharmacy innovation with the association.
Although an unplanned pregnancy might not meet the strict definition for a Class I recall, the serious nature of the problem warrants immediate notification, Emberley said. "It could be catastrophic for a lot of people if it's not planned," he said. "I think there is an urgency [to this situation] that needs to be out there." Leung said product-recall assessments are designed to take health risks into account, not lifestyle impacts, such as unplanned pregnancies.
Julianna Diller, a 19-year-old resident of Hamilton, said she takes Alysena and was informed of the recall only Tuesday. "It's pretty terrifying," she said. "I'm doing the smart thing and being a responsible teenage girl . . . and this kind of backfires." Emberley criticized the initial delay in spreading vital information about this recall and said action should be taken to prevent similar problems in the future. "This is a really, really unfortunate situation and I can't recall one quite this serious in quite some time," he said. "I think we really need a system where people assess the risk quickly and take a very broad view as to how this could affect Canadians and then do the right thing." The recalled pills carry the lot number LF01899A. Reports suggest that the batch contained about 50,000 packages.
ADDED SEARCH TERMS:
GEOGRAPHIC NAME: Canada
SUBJECT TERM:birth control; prescription drugs; public health; recalls; women
PERSONAL NAME: Leona Aglukkaq
ORGANIZATION NAME: Apotex Inc.; Health Canada
Health workers spread message of birth control pill recall; Recall One lot of generic birth control pills contained extra week of placebo
- Idnumber
- 201304120036
- DocID:
- 165845175
- Publication:
- New Brunswick Telegraph-Journal
- Page:
- C3
- Date:
- 2013.04.12
- Section:
- City
- Byline:
- Carolyn Thompson Telegraph-Journal
- Copyright:
- © 2013 Telegraph-Journal (New Brunswick)
- Word Count:
- 573
- Circulation:
- 33080
SAINT JOHN - The New Brunswick Pharmaceutical Society is hoping to have a presence in high schools to ensure teenagers are made aware of a widespread recall of birth control pills, says the organization's registrar.
"We've actually taken a step of trying to get a message out to high schools," Sam Lanctin said, adding that they're working with the department of public health and that the process hasn't yet been finalized.
Lanctin said it is possible that some teenagers may not be aware of the drug recall.
"Teenagers may not always be paying attention to the news," he said. "The flip side of that is that they're always connected and once one or two of them find out, this will probably spread like wildfire."
He said it's always better to err on the side of caution, adding that he hopes to get the message out several times to ensure everyone affected has been properly informed.
"We do think it will be another step to make sure that these people have another avenue to find out about this," he said. "Our message is basically if you've got any doubt, if you've got any issues at all, then contact your pharmacist or other health care provider."
The advice was prompted by the major birth control recall of the drug Alysena 28.
One lot of the product, bearing the number LF01899A, contained too little active drug and too much placebo, leaving women who took it vulnerable to becoming pregnant. The lot, which contained about 50,000 faulty packets, was distributed across Canada.
In Saint John, where teen pregnancy rates are some of the highest in the country, the recall has raised concerns over the possible consequences the recalled drug.
Terry-Lynne King, a nurse practitioner who works at the University of New Brunswick Saint John, said many university students rely on oral contraceptives - and the generic brand - as their method of birth control.
"I would expect that there are probably going to be some unintended pregnancies as a result of this error," she said.
She said while doctors may prescribe the brand pill, it is difficult to know when patients choose to purchase the cheaper generic form instead. They're relying on pharmacists to notify the students, but are encouraging anyone with questions or concerns to be tested for pregnancy.
"Young adults often do rely on the birth control pill to prevent pregnancy," she said. "We always encourage patients to be using backup methods - actually never to just solely rely on just birth control alone, because we know that there is a failure rate from the drug itself. Anywhere from one to two per cent."
Sharon Amirault, a member of the city's teen pregnancy committee and executive director at First Steps Housing Project, said many members of the vulnerable population in Saint John don't use oral contraceptives as their birth control method.
They often opt for a method that requires less daily adherence, such as an intrauterine device.
She added that most regular users would notice the unusual colours in the packaging.
"There are unwanted pregnancies for many, many reasons," she said, adding that the women who come to First Steps have all chosen to raise their children despite the circumstances of their pregnancy.
She did say there may be some of the more vulnerable population who could be affected, and that anyone concerned should see a doctor.
- With files from the Canadian Press
Backup birth control urged for women who had faulty pill packs
- Publication:
- The Chronicle-Herald
- Date:
- 2013.04.11
- Section:
- InFocus
- Page:
- B1
- Source:
- The Canadian Press
- Byline:
- Helen Branswell
- Illustration:
- This blister pack of Alysena28 contains the correct configuration of pills. The maker of Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive was incorrectly packaged with too many placebo pills. The lot contained 50,000 packets.(CP)
- Word Count:
- 610
- Circulation:
- 109164
TORONTO - Women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being, the Society of Obstetricians and Gynecologists of Canada suggested Wednesday.
And women may need to talk with their doctors about taking a pregnancy test, said Dr. Jennifer Blake, the organization's CEO.
The advice is prompted by a major birth control recall involving the drug Alysena 28. One lot of the product, bearing the number LF01899A, contained too little active drug and too much placebo, leaving women who took it vulnerable to becoming pregnant.
The lot, which contained about 50,000 faulty packets, was distributed across Canada. The company that sells the product in Canada, Apotex, believes it was not distributed to Alberta, Saskatchewan, Manitoba, Northwest Territories and Nunavut, but it cannot say with certainty that that is so, said Elie Betito, the company's director of public and government affairs.
Women using birth control pills take the drugs for 21 days each menstrual cycle. Because of the risk that they might not remember to resume taking their pills at the right time, many oral contraceptives are packaged with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
Blister packs in this faulty lot contained 14 pink pills containing active drug and 14 white placebos. And just that seven-day difference could be enough to result in a pregnancy, Blake said in an interview.
"That would mean two weeks without medication and that certainly gives enough time for the ovary to release an egg," she said. "And therefore if you have been taking that particular pill, Alysena, then it is important that you let your doctor know and indeed it may be important to follow up with a pregnancy test."
The problem is, some women may not know they were taking this contraceptive. That's because the drug is a generic version of the most popular birth control pill used in Canada, a drug called Alesse. (Alesse is not involved in this recall, Blake stressed.)
Some prescription drug plans require pharmacists to replace higher-priced brand-name drugs with generic versions if a generic is available. That may make it difficult to get a sense of who was on this drug, Blake says. It may also mean that women might think they are taking Alesse, because that is what their doctor prescribed, but they are actually taking Alysena.
"The problem is that it's a generic copy of the leading birth control bill. So that's a problem because we don't know how many women have had their prescription substituted for the generic. And women may not know they've had their prescription substituted, because the generic chose a very similar name and look to the leading birth control pill. And that does create confusion in everyone's mind."
Blake said women who take this contraceptive should be using other methods of birth control as well until they've gone a full month on a proper packet of the drug. "I would regard it as being like a new start" (on oral contraceptives).
Health Canada also wants answers to that question, said Dr. Supriya Sharma, senior medical adviser for Health Canada's health products and food branch.
"There does look like there was a problem in their good manufacturing processes. Otherwise this wouldn't have occurred," Sharma said.
Apotex notified Health Canada of its intention to recall the product on April 3. As required, it submitted a full risk assessment to the department by late in the day April 4. Health Canada finished assessing the file Monday morning, and posted a notice of the recall on its website.
Birth Control Some women may not realize they're taking recalled Pill
- Publication:
- GLOBE AND MAIL
- IDN:
- 131010075
- Date:
- 2013.04.11
- Page:
- L8
- Byline:
- CARLY WEEKS
- Section:
- Globe Life
- Edition:
- Metro
- Dateline:
- Words:
- 446
- Word Count:
- 456
- Circulation:
- 321109
CARLY WEEKS Many Canadian women may unknowingly be affected by a massive recall of birth control pills that could put them at risk for unplanned pregnancies.
Health Canada announced on Monday that Alysena 28, a generic birth-control pill distributed by Apotex Inc., is being recalled because it may contain an extra week of placebo pills instead of the medication.
But women who thought they were taking Alesse, the brand-name version of the pill, may actually have been given Alysena by their pharmacist.
It's a common practice known as "generic substitution," which allows pharmacists to dispense cheaper generic pills instead of the more expensive brand-name version.
"People who thought they were on Alesse might actually be on Alysena," said Dr. Jennifer Blake, chief executive officer of the Society of Obstetricians and Gynecologists of Canada.
Pharmacists are not required to tell patients when they are substituting a generic version and many women may not realize the difference because Alysena and Alesse have similar names, packaging and colours, Blake said. "It would say Alysena on [the package], but everything else would all look alike," she said.
Blake noted that the society was informed of this problem only on Tuesday, "which is itself a concern." The recall is being described by Blake and other health experts as unprecedented. She said the organization is hearing from women who are concerned about the possibility they may have become pregnant.
There are also concerns about the timing of the recall. Although Health Canada was informed of the problem last week and sent a recall notice to pharmacies and other retailers, a notice to the public didn't go out until Monday.
Phil Emberley, director of pharmacy innovation at the Canadian Pharmacists Association, said it's unclear how many women prescribed Alesse may have been given Alysena instead. Pharmacists have been reaching out to affected patients to alert them of the recall, he said.
Some provinces, such as Ontario, as well as drug plans require pharmacists to distribute generic versions of drugs to patients when available. But if pharmacists do substitute generic versions of drugs, they should be telling patients and ensuring they understand, Emberley said.
The Society of Obstetricians and Gynecologists of Canada maintains that generic birth-control pills should not be used to substitute for brand-name versions. In a 2008 statement, it noted that new generic versions of birth-control pills may not be as effective as the brand-name versions.
Options for Sexual Health, a non-profit organization based in Vancouver, says on its website that "there is not enough scientific evidence to support that any of the generic contraceptives are 'clinically equivalent' to the brand-name products."
ADDED SEARCH TERMS:
GEOGRAPHIC NAME: Canada
SUBJECT TERM:birth control; contraceptives; health; prescription drugs; recalls; women
ORGANIZATION NAME: Apotex Inc.
La réaction de la ministre critiquée CONTRACEPTIF ALYSENA 28
- Sourcetag
- 130411288032563
- Publication:
- Le Journal de Montréal
- Date:
- 2013.04.11
- Edition:
- final
- Section:
- Nouvelles
- Page:
- 39
- Byline:
- AGENCE QMI
- Dateline:
- OTTAWA
- Word Count:
- 97
- Circulation:
- 222520
OTTAWA -- (Agence QMI) Le NPD estime que le gouvernement conservateur a trop tardé avant d'émettre un rappel urgent pour le contraceptif oral Alysena 28, mettant à son avis en danger la santé de femmes et accroissant le risque de grossesse non planifiée.
"La ministre de la Santé aurait dû émettre un rappel obligatoire dès que Apotex, le fabricant d'Alysena, l'a informée des problèmes concernant le lot LF01899A de leur contraceptif, a affirmé la porte-parole en matière de santé, Libby Davies. Certaines femmes ont pu continuer à le prendre jusqu'à une semaine après la découverte du problème."
Women on faulty pill should use backup
- Idnumber
- 201304110064
- DocID:
- 165763559
- Publication:
- Hamilton Spectator
- Page:
- A12
- Date:
- 2013.04.11
- Section:
- Canada / World
- Edition:
- First
- Byline:
- The Canadian Press
- Copyright:
- © 2013 Torstar Corporation
- Word Count:
- 388
- Circulation:
- 93604
Women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being, the Society of Obstetricians and Gynecologists of Canada suggested Wednesday.
And women may need to talk with their doctors about taking a pregnancy test, said Dr. Jennifer Blake, the organization's CEO.
The advice is prompted by a major birth control recall involving the drug Alysena 28. One lot of the product, bearing the number LF01899A, contained too little active drug and too much placebo, leaving women who took it vulnerable to becoming pregnant.
The lot, which contained about 50,000 faulty packets, was distributed across Canada. The company that sells the product in Canada, Apotex, believes it was not distributed to Alberta, Saskatchewan, Manitoba, Northwest Territories or Nunavut, but it cannot say so with certainty, said company spokesperson Elie Betito.
Women using the pill take the birth prevention drugs for 21 days each menstrual cycle.
To make it easier to remember to take them every day, many oral contraceptives are packaged with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
Blister packs in this faulty lot contained 14 pink pills containing active drug and 14 white placebos. And just that seven-day difference could be enough to result in a pregnancy, Blake said in an interview.
"That would mean two weeks without medication and that certainly gives enough time for the ovary to release an egg," she said. "And therefore if you have been taking that particular pill, Alysena, then it is important that you let your doctor know and indeed it may be important to follow up with a pregnancy test."
The problem is, some women may not know they were taking this contraceptive. That's because the drug is a generic version of the most popular birth control pill used in Canada, a drug called Alesse (which is not involved in the recall.)
Some prescription drug plans require pharmacists to replace higher-priced brand-name drugs with generic versions if a generic is available. That may make it difficult to get a sense of who was on this drug, Blake says. It may also mean that women might think they are taking Alesse, because that is what their doctor prescribed, but they are actually taking Alysena.
Women who took faulty 'pill' urged to used backup contraception
- Publication:
- Red Deer Advocate
- Date:
- 2013.04.11
- Section:
- News
- Page:
- A3
- Source:
- BY THE CANADIAN PRESS
- Dateline:
- TORONTO
- Word Count:
- 472
- Circulation:
- 16045
Women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being, the Society of Obstetricians and Gynecologists of Canada suggested Wednesday.
And women may need to talk with their doctors about taking a pregnancy test, said Dr. Jennifer Blake, the organization's CEO.
The advice is prompted by a major birth control recall involving the drug Alysena 28. One lot of the product, bearing the number LF01899A, contained too little active drug and too much placebo, leaving women who took it vulnerable to becoming pregnant.
The lot, which contained about 50,000 faulty packets, was distributed across Canada. The company that sells the product in Canada, Apotex, believes it was not distributed to Alberta, Saskatchewan, Manitoba, Northwest Territories and Nunavut, but it cannot say with certainty that that is so, said Elie Betito, the company's director of public and government affairs.
Women using birth control pills take the drugs for 21 days each menstrual cycle. Because of the risk that they might not remember to resume taking their pills at the right time, many oral contraceptives are packaged with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
Blister packs in this faulty lot contained 14 pink pills containing active drug and 14 white placebos. And just that seven-day difference could be enough to result in a pregnancy, Blake said in an interview.
"That would mean two weeks without medication and that certainly gives enough time for the ovary to release an egg," she said. "And therefore if you have been taking that particular pill, Alysena, then it is important that you let your doctor know and indeed it may be important to follow up with a pregnancy test."
The problem is, some women may not know they were taking this contraceptive. That's because the drug is a generic version of the most popular birth control pill used in Canada, a drug called Alesse. (Alesse is not involved in this recall, Blake stressed.)
Some prescription drug plans require pharmacists to replace higher-priced brand-name drugs with generic versions if a generic is available. That may make it difficult to get a sense of who was on this drug, Blake says. It may also mean that women might think they are taking Alesse, because that is what their doctor prescribed, but they are actually taking Alysena.
"The problem is that it's a generic copy of the leading birth control bill. So that's a problem because we don't know how many women have had their prescription substituted for the generic. And women may not know they've had their prescription substituted, because the generic chose a very similar name and look to the leading birth control pill. And that does create confusion in everyone's mind."
Backup contraception urged amid recall; Gynecology group offers advice after faulty birth control pill packs recalled
- Idnumber
- 201304110012
- DocID:
- 165762056
- Publication:
- Waterloo Region Record
- Page:
- A6
- Date:
- 2013.04.11
- Section:
- NEWS
- Edition:
- First
- Byline:
- Helen Branswell
- Source:
- The Canadian Press
- Copyright:
- © 2013 Torstar Corporation
- Word Count:
- 355
- Circulation:
- 56519
The Society of Obstetricians and Gynecologists of Canada says women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being.
A senior official of the organization says women may also need to talk with their doctors about taking a pregnancy test.
The advice is prompted by a major birth control recall involving the drug Alysena 28.
A batch of about 50,000 faulty packets of the drug were distributed in Canada; the packages contained two weeks' worth of sugar pills, not one.
Dr. Jennifer Blake of the Society of Obstetricians and Gynecologists of Canada says missing a week of the drug could result in pregnancies. She says Plan B, the so-called morning-after pill, would be an option.
Women taking birth control pills swallow the drugs for 21 days each menstrual cycle. Because of the risk that they might not remember to resume taking their pills at the right time, many of these contraceptives are packed with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
But with these faulty packs of Alysena, there were two weeks of white placebos and two weeks of pink contraceptives.
Blake, the chief executive officer of the society, suggested the turn of event was upsetting, because Canada has had a variety of safe and reliable birth control options for women.
The drug, marketed by the drug firm Apotex, is a generic version of the most popular birth control pill in use, a drug called Alesse.
Alesse is not involved in this recall, Blake stresses.
Some drug plans require pharmacists to replace higher-priced brand name drugs with generic versions if a generic is available. That may make it difficult to get a sense of who was on this drug, Blake says.
Apotex is recalling about 50,000 packets in a lot of drugs that was distributed between Dec. 4 and 17, 2012, says Elie Betito, the company's director of public and governmental affairs. He says seven other batches have been released since the faulty batch was pushed into the system.
Gynecologists urge women to use backup contraception if they took faulty pills
- Idnumber
- 201304110051
- DocID:
- 165755174
- Publication:
- The Daily Gleaner (Fredericton)
- Page:
- A7
- Date:
- 2013.04.11
- Section:
- International
- Byline:
- HELEN BRANSWELL The Canadian Press
- Copyright:
- © 2013 The Daily Gleaner (Fredericton)
- Word Count:
- 301
TORONTO - Women who may have taken birth control pills from faulty packs should use a backup form of contraception for the time being, the Society of Obstetricians and Gynecologists of Canada suggested Wednesday.
And women may need to talk with their doctors about taking a pregnancy test, said Dr. Jennifer Blake, the organization's CEO.
The advice is prompted by a major birth control recall involving the drug Alysena 28.
One lot of the product, bearing the number LF01899A, contained too little active drug and too much placebo, leaving women who took it vulnerable to becoming pregnant.
The lot, which contained about 50,000 faulty packets, was distributed across Canada.
The company that sells the product in Canada, Apotex, believes it was not distributed to Alberta, Saskatchewan, Manitoba, Northwest Territories and Nunavut, but it cannot say with certainty that that is so, said Elie Betito, the company's director of public and government affairs.
Women using birth control pills take the drugs for 21 days each menstrual cycle.
Because of the risk that they might not remember to resume taking their pills at the right time, many oral contraceptives are packaged with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
Blister packs in this faulty lot contained 14 pink pills containing active drug and 14 white placebos. And just that seven-day difference could be enough to result in a pregnancy, Blake said in an interview.
"That would mean two weeks without medication and that certainly gives enough time for the ovary to release an egg," she said.
"And therefore if you have been taking that particular pill, Alysena, then it is important that you let your doctor know and indeed it may be important to follow up with a pregnancy test."
Women advised to use backup if they took faulty 'pill'
- Idnumber
- 201304110114
- DocID:
- 165749816
- Publication:
- Windsor Star
- Page:
- D5
- Date:
- 2013.04.11
- Section:
- Canada
- Edition:
- Final
- Byline:
- Helen Branswell
- Source:
- The Canadian Press
- Dateline:
- TORONTO
- Word Count:
- 192
- Circulation:
- 67530
The Society of Obstetricians and Gynecologists of Canada says women who may have taken birth control pills from faulty packs should use a backup form of contraception.
A senior official of the organization says women may also need to talk with their doctors about taking a pregnancy test. The advice is prompted by a major birth control recall involving the drug Alysena 28.
A batch of about 50,000 faulty packets of the drug were distributed in Canada; the pack-ages contained two weeks' worth of sugar pills, not one.
Dr. Jennifer Blake of the Society of Obstetricians and Gynecologists of Canada says missing a week of the drug could result in pregnancies.
She says Plan B, the so-called morning after pill, would be an option for women who have had what amounts to unprotected sex in the last three to five days. Women taking birth control pills swallow the drugs for 21 days each menstrual cycle. Because of the risk that they might not remember to resume taking their pills at the right time, many of these contraceptives are packed with a pill for each of the 28 days in a cycle.
Birth-control pill alysena 28 recalled
- Idnumber
- 201304110070
- DocID:
- 165750818
- Publication:
- The StarPhoenix (Saskatoon)
- Page:
- D4
- Date:
- 2013.04.11
- Section:
- News
- Edition:
- Final
- Byline:
- Helen Branswell
- Source:
- The Canadian Press
- Illustration:
- The Canadian Press / The maker of the birth-control pill Alysena 28 has issued an urgent recall.;
- Dateline:
- TORONTO
- Word Count:
- 250
- Circulation:
- 55325
The Society of Obstetricians and Gynecologists of Canada says women who may have taken birth-control pills from faulty packs should use a backup form of contraception for the time being.
A senior official of the organization says women may also need to talk with their doctors about taking a pregnancy test.
The advice is prompted by a major birth-control recall involving the drug Alysena 28.
A batch of about 50,000 faulty packets of the drug were distributed in Canada. The packages contained two weeks' worth of sugar pills, not one.
Dr. Jennifer Blake of the Society of Obstetricians and Gynecologists of Canada says missing a week of the drug could result in pregnancies.
Women taking birth-control pills swallow the drugs for 21 days each menstrual cycle. Because of the risk that they might not remember to resume taking their pills at the right time, many of these contraceptives are packed with a pill for each of the 28 days in a cycle - 21 are drug and seven are placebos.
But with these faulty packs of Alysena, there were two weeks of white placebos and two weeks of pink contraceptives.
Blake, the CEO of the society, suggested the turn of event was upsetting because Canada has had a variety of safe and reliable birth-control options for women.
"Certainly women using (Alysena 28) right now should be advised to use an additional method of protection, because they wouldn't have the appropriate level of protection from the faulty packages," she says.
Faulty-pill users need other birth control
- Idnumber
- 201304110046
- DocID:
- 165748631
- Publication:
- Ottawa Citizen
- Page:
- A5
- Date:
- 2013.04.11
- Section:
- News
- Edition:
- Final
- Source:
- Ottawa Citizen
- Word Count:
- 104
- Circulation:
- 128919
The Society of Obstetricians and Gynaecologists of Canada says women who took birth control pills from faulty packs should use a backup form of contraception. A senior official of the organization says women may also need to talk with their doctors about taking a pregnancy test. The advice is prompted by a major recall of the drug Alysena 28. A batch of about 50,000 faulty packets of the drug was distributed in Canada; packages contained two weeks of sugar pills, not one. Dr. Jennifer Blake of the Society of Obstetricians and Gynaecologists says missing a week of the drug could result in pregnancy.
Rappel urgent de pilules contraceptives Alysena 28
- Sourcetag
- 130410287772674
- Publication:
- Le Journal de Montréal
- Date:
- 2013.04.10
- Edition:
- final
- Section:
- Votre Vie
- Page:
- 53
- Byline:
- AGENCE QMI
- Dateline:
- OTTAWA
- Word Count:
- 86
- Circulation:
- 224324
OTTAWA -- (Agence QMI) Des pilules contraceptives Alysena 28, du fabricant Apotex, font l'objet d'un rappel par Santé Canada, puisqu'elles pourraient contenir moins de comprimés actifs que prévu.
L'important rappel concerne les plaquettes de 28 pilules de la marque, qui font partie du lot LF01899A. Celles-ci pourraient contenir 14 comprimés placebos et 14 comprimés actifs, d'où l'urgence de prévenir les femmes concernées. Santé Canada suggère aux femmes d'utiliser une autre méthode de contraception, non hormonale, comme mesure provisoire. Elles devraient aussi demander l'avis de leur médecin.
Use of recalled birth control pills puts women at risk of pregnancy
- Idnumber
- 201304100099
- DocID:
- 165671789
- Publication:
- Hamilton Spectator
- Page:
- A9
- Date:
- 2013.04.10
- Section:
- Canada / World
- Edition:
- First
- Byline:
- Marco Chown Oved Torstar News
- Illustration:
- In a recall notice distributed by Health Canada, Apotex Inc. said said its 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug. frole.com
- Copyright:
- © 2013 Torstar Corporation
- Word Count:
- 387
- Circulation:
- 92924
Women taking birth control pills recalled for having too many sugar pills in a monthly pack may have been in the dark for almost a week after the problem was discovered.
Apotex, the maker of Alysena-28 contraceptive pill, issued a voluntary recall to wholesalers, retailers and distributors on April 3. This "Type II recall" pulled the drug from the shelves but did not require contacting women taking the pill.
Health Canada upgraded the recall five days later, issuing the most serious, "Type I public recall" of the drug, which was sold in all provinces except Alberta, Saskatchewan and Manitoba.
According to the Health Canada website, a Type I recall occurs in "a situation in which there is a reasonable probability that the use of, or exposure to, a product will cause serious adverse health consequences or death."
A packet of 28 pills, to be taken daily, normally includes one week of white placebo pills and three weeks of pink active tablets. The affected lot, labelled with the code LF01899A, may contain up to two rows of white placebo pills.
"Ingestion of only 14 tablets of active instead of the intended 21 of oral contraceptive would most likely result in reduced efficacy for contraception and therefore possibility of unplanned pregnancy cannot be ruled out," wrote Apotex in its letter with the heading "Urgent Drug Recall."
Alysena 21, a version of the contraceptive that doesn't include the placebo pills, doesn't seem to be affected by the recall.
The letter instructs those in the drug supply chain to stop distributing and quarantine the tablets. It also requests that pharmacies remove the affected lot from sale and return all drugs to the wholesalers.
There is no mention of contacting patients.
People prescribed with Alysena-28 may have only discovered the error when Health Canada issued its drug recall Monday.
There is no mention of the recall on Apotex's website. Apotex did not return the Star's request for comment.
Shoppers Drug Mart representative Tammy Smitham said the pills have been on sale since December 2012 and were pulled from shelves last week when the first recall was issued. Pharmacists began calling patients Monday after receiving the second recall.
Health Canada advises patients to "use a nonhormonal method of contraception as an interim measure until you speak with your physician and obtain medical advice."
Urgent rappel de pilules contraceptives
- Publication:
- La Voix de l'Est
- Date:
- 2013.04.10
- Section:
- Actualité
- Page:
- 30
- Column:
- En bref
- Source:
- La Presse
- Word Count:
- 180
- Circulation:
- 15797
Importante mise en garde pour les femmes qui prennent la pilule contraceptive Alysena 28. Certaines plaquettes du produit actuellement en circulation au Québec contiennent un nombre trop élevé de placebos, ce qui peut les mettre à risque de tomber enceintes et de modifier leur cycle menstruel.
La société pharmaceutique Apotex a procédé hier au rappel urgent du lot LF1899A. Selon Santé Canada, certaines plaquettes alvéolaires dans ce lot peuvent contenir deux rangées (14 comprimés) de comprimés placebo et deux rangées (14 comprimés) de comprimés actifs, alors qu'elles devraient contenir seulement une rangée de comprimés placebo (7 comprimés). Santé Canada refuse d'indiquer le nombre de plaquettes dans le lot qui fait l'objet d'un rappel.
Leslie Meerburg, conseillère principale des relations avec les médias à l'agence gouvernementale fédérale, a aussi refusé d'indiquer si des Canadiennes étaient tombées enceintes à la suite de cette erreur. Les femmes qui ont en leur possession des plaquettes de contraceptifs qui font partie du lot rappelé doivent contacter leur médecin et utiliser une autre méthode de contraception en attendant de parler à un professionnel de la santé.
Rappel urgent
- Publication:
- Le Quotidien
- Date:
- 2013.04.10
- Section:
- Actualités
- Page:
- 37
- Source:
- La Presse
- Word Count:
- 180
- Circulation:
- 27420
Importante mise en garde pour les femmes qui prennent la pilule contraceptive Alysena 28. Certaines plaquettes du produit actuellement en circulation au Québec contiennent un nombre trop élevé de placebos, ce qui peut les mettre à risque de tomber enceinte et de modifier leur cycle menstruel. La société pharmaceutique Apotex a procédé hier au rappel urgent du lot LF1899A.
Selon Santé Canada, certaines plaquettes alvéolaires dans ce lot peuvent contenir deux rangées (14 comprimés) de comprimés placebo et deux rangées (14 comprimés) de comprimés actifs, alors qu'elles devraient contenir seulement une rangée de comprimés placebo (7 comprimés). Santé Canada refuse d'indiquer le nombre de plaquettes dans le lot qui fait l'objet d'un rappel.
Leslie Meerburg, conseillère principale des relations avec les médias à l'agence gouvernementale fédérale, a aussi refusé d'indiquer si des Canadiennes étaient tombées enceintes à la suite de cette erreur. Les femmes qui ont en leur possession des plaquettes de contraceptifs qui font partie du lot rappelé doivent contacter leur médecin et utiliser une autre méthode de contraception en attendant de parler à un professionnel de la santé.
RAPPEL DES PILULES CONTRACEPTIVES ALYSENA 28
- Publication:
- Le Droit
- Date:
- 2013.04.10
- Section:
- Actualités
- Page:
- 17
- Column:
- En bref
- Source:
- La Presse
- Word Count:
- 180
- Circulation:
- 35292
Importante mise en garde pour les femmes qui prennent la pilule contraceptive Alysena 28. Certaines plaquettes du produit actuellement en circulation au Québec contiennent un nombre trop élevé de placebos, ce qui peut les mettre à risque de tomber enceinte et de modifier leur cycle menstruel. La société pharmaceutique Apotex a procédé hier au rappel urgent du lot LF1899A. Selon Santé Canada, certaines plaquettes alvéolaires dans ce lot peuvent contenir deux rangées (14 comprimés) de comprimés placebo et deux rangées (14 comprimés) de comprimés actifs, alors qu'elles devraient contenir seulement une rangée de comprimés placebo (7 comprimés). Santé Canada refuse d'indiquer le nombre de plaquettes dans le lot qui fait l'objet d'un rappel. Leslie Meerburg, conseillère principale des relations avec les médias à l'agence gouvernementale fédérale, a aussi refusé d'indiquer si des Canadiennes étaient tombées enceintes à la suite de cette erreur. Les femmes qui ont en leur possession des plaquettes de contraceptifs qui font partie du lot rappelé doivent contacter leur médecin et utiliser une autre méthode de contraception en attendant de parler à un professionnel de la santé.
Rappel de pilules contraceptives Alysena 28
- Publication:
- La Presse
- Date:
- 2013.04.10
- Section:
- Santé
- Page:
- A13
- Byline:
- Daphné Cameron
- Word Count:
- 114
- Circulation:
- 203022
Importante mise en garde pour les femmes qui prennent la pilule contraceptive Alysena 28. Certaines plaquettes du produit actuellement en circulation au Québec contiennent un nombre trop élevé de placebos, ce qui peut les mettre à risque de tomber enceinte et de modifier leur cycle menstruel. La société pharmaceutique Apotex a procédé hier au rappel urgent du lot LF1 899A. Selon Santé Canada, certaines plaquettes alvéolaires dans ce lot peuvent contenir deux rangées (14 comprimés) de comprimés placebo et deux rangées (14 comprimés) de comprimés actifs, alors qu'elles devraient contenir seulement une rangée de comprimés placebo (7 comprimés). Santé Canada refuse d'indiquer le nombre de plaquettes dans le lot qui fait l'objet d'un rappel.
Health Canada recalls birth control pills; Extra placebo tablets placed in packages of Alysena 28 pills
- Publication:
- The Guardian (Charlottetown)
- Date:
- 2013.04.10
- Section:
- Front
- Page:
- A1
- Byline:
- Ryan Ross
- Word Count:
- 272
- Circulation:
- 18097
Island women who use Alysena 28 birth control pills should check their prescriptions after Health Canada issued a recall because of extra placebo tablets in the packaging.
The pills, which were sold in seven provinces, including P.E.I., normally contain one row of a week's worth of white placebo pills and three rows of pink contraceptive pills.
But the recalled pills contain an extra week's worth of placebo pills, which means a woman who takes every pill in the package will only get two weeks of the contraceptive tablets instead of three.
An advisory on pharmaceutical company Apotex's website said that in the case of a missed dose, women should use a non-hormonal contraceptive method as an interim measure until they talk to their pharmacist or doctor.
The company is asking all women who received lot LFO1899A of Alysena 28 to contact their doctor and return the pills to their pharmacist. Not all contraceptive prescriptions include placebos, but some do so there isn't a week off without any pills.
The idea is that women will be less likely to forget to take the contraceptive tablets if they take a pill every day.
Health Canada issued a Type II recall last week, which its website says is assigned in situations were there use of or exposure to a product "may cause temporary adverse health consequences or where the probability of serious adverse health consequences is remote."
The health agency upgraded the recall to a Type I on Tuesday, which Health Canada says involves the reasonable probability that use of or exposure to a product will cause serious adverse health consequences or death.
Birth Control Recall
- Publication:
- CTV - CTV News
- Date:
- 2013.04.09
- Time:
- 23:00:00 ET
- End:
- 23:30:00 ET
- Word Count:
- 424
LISA LAFLAMME: Health Canada has issued the urgent recall of a batch of birth control pills packaged improperly, more placebo than contraception. The foul-up has affected thousands of Canadian women on the pill, many now worried they could be pregnant. The risk is so real that today pharmacies were contacting customers, some offering free pregnancy tests and morning after pills. CTV's medical specialist Avis Favaro has the details.
AVIS FAVARO (Reporter): It's a drug recall that is making a lot of women nervous, involving a birth control pill called Alysena that may not stop a pregnancy.
SHANNON OGILVIE (Student): I immediately wanted to look at my packs because that's the brand I'm taking.
MYA THERIAULT GREEN (Mother): I've been taking the pills for about six months. I'm 38, and I'm married. I already have two kids, so a third one kind of would be a major change for me.
FAVARO: Normally the packages contain three rows of pink pills with active ingredients and one row of white pills, a placebo taken to replicate a woman's monthly cycle. But one woman in Atlantic Canada saw two rows of white pills in a package, meaning less contraceptive protection. The drug maker, Apotex, was alerted and it sent out this urgent drug recall April 3rd, asking pharmacies to stop selling one lot of Alysena, adding the possibility of unplanned pregnancy cannot be ruled out. Yet the alert makes no mention of contacting patients taking the pills.
EILEEN GAMOFF (Ellesmere Pharmacy): If I were taking these, or my daughter, or any woman, it would be very unsettling to find out there was a big error like this.
FAVARO: Almost a week later, Health Canada intervened, issuing a stronger alert requiring that patients be notified.
DR. SUBRIYA SHARMA (Health Canada): We're going to be following up with the company to find out if the recall was done appropriately and certainly we need to follow up with the company to find out why this issue occurred in the first place.
FAVARO: Still, some pharmacies are trying to give the women affected immediate reassurance.
JOHN TSE (London Drugs VP): If they're concerned we want to offer them a pregnancy test just in case, other individuals that may have a need, depending on the situation, the morning after pill.
FAVARO: And in a statement late today, Apotex says it is cooperating with Health Canada's investigation, adding these generic contraceptives actually originate from a factory in Spain. Lisa.
LAFLAMME: Interesting. So five days before telling their customers, Avis, thank you for this tonight.
Birth control pills recalled; Canadian women were left in the dark for about a week about pill's effectiveness
- Idnumber
- 201304100018
- DocID:
- 165670592
- Publication:
- Waterloo Region Record
- Page:
- A8
- Date:
- 2013.04.10
- Section:
- NEWS
- Edition:
- First
- Byline:
- Marco Chown Oved
- Source:
- News services
- Illustration:
- Shown here is a correct Alysena 28 blister pack. Apotex is recalling the product after some packs were found to contain 14 sugar pills instead of seven. Apotex website COPYRIGHT: © 2013 Torstar Corporation
- Word Count:
- 617
- Circulation:
- 56365
Women taking recalled birth control were kept in the dark for five days after the problem was discovered, prompting fears of unplanned pregnancies and panic on social media.
Apotex, the maker of the Alysena 28 contraceptive pill, issued a voluntary recall to wholesalers, retailers and distributors on April 3 after it discovered some packs contained 14 sugar pills instead of seven. This Type II recall pulled the drug from the shelves, but did not require contacting patients.
Health Canada upgraded the recall five days later on Monday, issuing the most serious Type I recall of the drug, which obligates retailers to contact women who were sold the pill.
As news of the recall spread Tuesday, hundreds of women across Canada took to social media, expressing outrage, worry and consternation, many of them saying they still hadn't been contacted.
One Twitter user, calling herself Mademoiselle Titam, summed up much of the outcry, writing "In Canada, taking the pill is like playing Russian roulette, especially with Alysena-28."
Health Canada says Type I recalls occurs in situations "in which there is a reasonable probability that the use of, or exposure to, a product will cause serious adverse health consequences or death."
"By taking the birth control pill, a woman has made the decision that she doesn't want to get pregnant," said Sarah Hobbs-Blyth, executive director of Planned Parenthood Toronto. "When a mistake like this happens, that decision has been taken away from them."
Carolyn Egan at the Birth Control Venereal Disease Information Centre says women unknowingly taking placebos are no longer protected from unwanted pregnancies.
"You think you're protected, but you aren't," she said. "You're in a pretty vulnerable situation until you're on your new package and you're on it for seven days."
The sexual health centre, like many others across the city, received calls from worried women on Tuesday.
They recommend women who are unsure if they're affected check the lot number on their pills - the affected lot is labelled with the code LF01899A - and call their pharmacy.
A packet of 28 pills, to be taken daily, normally includes one week of white placebo pills and three weeks of pink active tablets. The affected lot may contain up to two rows of white placebo pills.
"Ingestion of only 14 tablets of active instead of the intended 21 of oral contraceptive would most likely result in reduced efficacy for contraception and, therefore, possibility of unplanned pregnancy cannot be ruled out," wrote Apotex in its urgent drug recall issued last week.
The affected birth control product was sold in all provinces except Alberta, Saskatchewan and Manitoba since early December 2012.
Alysena 21, a version of the contraceptive that doesn't include the placebo pills, isn't included in the recall.
Women prescribed Alysena-28 were only required to be informed of the error when Health Canada issued its drug recall Monday afternoon. Apotex posted its first mention of the recall on their website late Tuesday.
Pharmacies had already begun calling affected customers.
Shoppers Drug Mart representative Tammy Smitham said the pills were pulled from shelves last week when the first recall was issued. Pharmacists began calling patients Monday after receiving the second recall.
London Drugs, a chain of pharmacies in western Canada, is offering two free pregnancy tests and a morning-after pill to anyone affected. Normally, morning-after pills cost $40.
Pharmacy vice-president John Tse said they also identified 350 women in British Columbia who received the affected lot and had pharmacists call them over the weekend - before the Health Canada recall was issued.
In its recall, Health Canada advises patients to "use a nonhormonal method of contraception as an interim measure until you speak with your physician and obtain medical advice."
Birth control error sparks fear, anger; Health Canada waited five days before ordering retailers to notify patients
- IDNumber
- 201304100082
- DocID:
- 165665205
- Publication:
- Toronto Star
- Page:
- A1
- Date:
- 2013.04.10
- Section:
- News
- Edition:
- ONT
- Byline:
- Marco Chown Oved Toronto Star
- Copyright:
- © 2013 Torstar Corporation
- Word Count:
- 620
- Circulation:
- 306395
Women taking recalled birth control were kept in the dark for five days after the problem was discovered, prompting fears of unplanned pregnancies and panic on social media.
Apotex, the maker of Alysena 28 contraceptive pill, issued a voluntary recall to wholesalers, retailers and distributors on April 3, after it was discovered some packs contained 14 sugar pills instead of seven. This Type II recall pulled the drug from the shelves but did not require contacting patients.
Health Canada upgraded the recall five days later on Monday, issuing a Type I recall, the most serious kind.
A Type 1 recall obligates retailers to contact women who were sold the pill.
As news of the recall spread Tuesday, hundreds of women across Canada took to social media, expressing outrage, worry and consternation, many of them saying they still hadn't been contacted.
One Twitter user, calling herself Mademoiselle Titam, summed up much of the outcry, writing "In Canada, taking the pill is like playing Russian roulette, especially with Alysena-28."
Health Canada says a Type I recall occurs in "a situation in which there is a reasonable probability that the use of, or exposure to, a product will cause serious adverse health consequences or death."
"By taking the birth control pill, a woman has made the decision that she doesn't want to get pregnant," said Sarah Hobbs-Blyth, executive director of Planned Parenthood Toronto. "When a mistake like this happens, that decision has been taken away from them."
Carolyn Egan at the Birth Control Venereal Disease Information Centre says women unknowingly taking placebos are no longer protected from unwanted pregnancies.
"You think you're protected, but you aren't," she said. "You're in a pretty vulnerable situation until you're on your new package and you're on it for seven days."
The sexual health centre, like many others across the city, received calls from worried women on Tuesday. They recommend that women who are unsure if they're affected check the lot number on their pills - the affected lot is labelled with the code LF01899A - and call their pharmacy.
A packet of 28 pills, to be taken daily, normally includes one week of white placebo pills and three weeks of pink active tablets. The affected lot may contain up to two rows of white placebo pills.
"Ingestion of only 14 tablets of active instead of intended 21 of oral contraceptive would most likely result in reduced efficacy for contraception and therefore possibility of unplanned pregnancy cannot be ruled out," wrote Apotex in its urgent drug recall issued last week.
The affected birth control has been sold in all provinces except Alberta, Saskatchewan and Manitoba since early December 2012.
Alysena 21, a version of the contraceptive that doesn't include the placebo pills, isn't included in the recall.
Women prescribed Alysena 28 were only required to be informed of the error when Health Canada issued its recall Monday afternoon.
Apotex posted its first mention of the recall on its website late Tuesday. Pharmacies had already begun calling affected customers.
Shoppers Drug Mart representative Tammy Smitham said the pills were pulled from shelves last week when the first recall was issued. Pharmacists began calling patients Monday after the second recall.
London Drugs, a chain of pharmacies in western Canada, is offering two free pregnancy tests and a morning-after pill to anyone affected. Normally, morning-after pills cost $40.
Pharmacy vice-president John Tse said they also identified 350 women in British Columbia who received the affected lot and had pharmacists call them over the weekend - before the Health Canada recall was issued.
In its recall, Health Canada advises patients to "use a nonhormonal method of contraception as an interim measure until you speak with your physician and obtain medical advice."
MEDICINE Women not alerted immediately of birth-control recall
- Publication:
- GLOBE AND MAIL
- IDN:
- 131000080
- Date:
- 2013.04.10
- Page:
- L8
- Byline:
- ERIN ANDERSSEN
- Section:
- Globe Life
- Edition:
- Metro
- Dateline:
- Words:
- 728
- Word Count:
- 728
- Circulation:
- 321109
ERIN ANDERSSEN Standing in her bathroom on Monday night, reading the fine print on her package of birth-control pills, Jacqueline Belo felt the panic many women will recognize: the queasy worry that you might be pregnant when you don't want to be. Belo, a hairdresser who lives in Burlington, Ont., had just heard about the recall of Alysena pills from a certain lot number - and hers matched. "I thought my heart was going to stop, to be honest," she says. "My life just flashed before my eyes." Belo, 30, doesn't know if she ever wants kids, but both she and her husband of three years know that the time isn't right. "I am definitely not ready to have kids," she says. "Obviously, I have been taking the precautions for a reason." And she's not the type to miss a pill by accident; "I take them the same time every day.
I don't fool around with it." But this wasn't Belo's mistake: On Monday, Health Canada upgraded the recall of 50,000 packages of Alysena 28-day birth control pills that arrived on drugstore shelves in mid-December. The recall was prompted when an Ontario customer returned a package that included two weeks of white placebo pills. The blister pack typically includes three weeks of pink pills, which contain the active ingredients of the drug, and one week of white sugar pills, which are included to help patients remember to continue taking them.
There may be questions, however, as to why word of the problem took more than three days to reach many patients.
Late last Friday afternoon, a voluntary recall for lot number LF01899A of Alysena 28 was announced. This batch had been distributed to retailers in British Columbia, Ontario, Quebec and the Atlantic provinces. But Apotex, the drug's manufacturer, decided, "based on their own assessment," to issue a Type 2 recall, which requires drugstores to remove the product from their stock (but not alert patients), according to a Health Canada statement.
On Monday morning, Health Canada raised the level of the recall out of concern for women who had been advised not to get pregnant for medical reasons or who might be using drugs that "could be harmful to a developing fetus." The updated recall advises women to check their packages and contact their doctors for advice, as well as to use an alternative birth control in the meantime.
The decision to delay the more serious recall is upsetting to women such as Belo, who would consider an unwanted pregnancy a serious health consequence. "If I didn't have the TV on last night, I wouldn't have known to check." As the recall was upgraded, pharmacists and doctors have begun calling patients who may have taken home a box from the lot number in question. London Drug, based in Vancouver, had already taken the step on Friday, tracking down 21 stores where the problem batch had been stocked, and calling every customer who might have received one. (Every patient under 18 was contacted in confidence through their doctor.) "It was a judgment call," said John Tse, the chain's vice-president of pharmacy. "You find one box - is there another? That's the unknown we can't answer." In addition, London Drugs has offered every affected customer a free pregnancy test or morning-after pill; a small number of women have asked for them.
Apotex says it's easy to know if you have a improperly packaged dose: Customers will have two rows of white pills in their blister pack. The pink pills, containing the active ingredient, work as usual, said Elie Betito, Apotex's director of public and government affairs.
Still, the company, which has since distributed seven more lots of product manufactured specifically for Canada from a factory in Europe, can't yet say how the error happened or how many packages were affected. As of Tuesday morning, it said it had learned of only one.
Since the batch went on shelves in December, however, some women may already have used them, creating a potential liability for Apotex: "We have had calls saying, 'My wife is pregnant and it's your fault,' but they don't have the package any more," Betito said.
Belo was lucky she had her package: It had three rows of pink pills.
ADDED SEARCH TERMS:
GEOGRAPHIC NAME: Canada
SUBJECT TERM:contraceptives; prescription drugs; recalls
ORGANIZATION NAME: Apotex; Health Canada
Drug firm recalls Alysena-28 birth control pills
- Publication:
- The Chronicle-Herald
- Date:
- 2013.04.09
- Section:
- NovaScotia
- Page:
- A9
- Word Count:
- 98
- Circulation:
- 109164
An Ontario drug company is warning consumers that a batch of its birth control pills could lead to unplanned pregnancies.
On Monday, Health Canada issued a Type 1 recall of Alysena-28, meaning it could have "serious adverse health consequences."
On April 3, Apotex Pharmachem Inc. in Brantford issued an "urgent drug recall," upon discovering that each month's prescription contained two weeks of placebo pills, rather than the normal week's worth. This reduces the number of hormone pills, containing estrogen and progestogen, which are necessary to prevent pregnancy.
Consumers are urged to check pill packages for lot number LF01899A.
Maker issues urgent recall of contraceptive Alysena over packaging mix-up
- Publication:
-
The Telegram (St. John's)
- Date:
- 2013.04.09
- Section:
- Family
- Page:
- B2
- Column:
- Briefs
- Source:
- CP
- Dateline:
- Ottawa
- Word Count:
- 131
- Circulation:
- 21646
The maker of the birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets instead of three.
Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy.
In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug. Apotex said blister packs in the affected lot - LF01899A - may instead contain 14 placebo pills and 14 active contraceptive tablets.
Alysena 28 is sold in British Columbia, New Brunswick, Newfoundland and Labrador, Nova Scotia, Ontario, P.E.I. and Quebec.
Birth-control pill recalled
- Idnumber
- 201304090094
- DocID:
- 165577847
- Publication:
- Times Colonist (Victoria)
- Page:
- D1 / Front
- Date:
- 2013.04.09
- Section:
- Life
- Edition:
- Final
- Source:
- The Canadian Press, With a File By Jeff Bell
- Dateline:
- OTTAWA
- Word Count:
- 162
The maker of birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets instead of three.
Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy.
London Drugs announced that customers may be offered the morning-after pill, as well as a pregnancy test. Each situation will be assessed individually, said John Tse, vice-president of pharmacy for London Drugs.
In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug.
Apotex said blister packs in the affected lot - LF01899A - may instead contain 14 placebo pills and 14 active contraceptive tablets.
Alysena 28 is sold in B.C., New Brunswick, Newfoundland and Labrador, Nova Scotia, Ontario, P.E.I. and Quebec.
Alysena birth-control pills recalled
- Idnumber
- 201304090034
- DocID:
- 165575928
- Publication:
- The Province
- Page:
- A15
- Date:
- 2013.04.09
- Section:
- News
- Edition:
- Final
- Source:
- The Canadian Press
- Dateline:
- OTTAWA
- Word Count:
- 131
- Circulation:
- 160739
The maker of the birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets instead of three.
Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy.
In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug.
Apotex said blister packs in the affected lot - LF01899A - may instead contain 14 placebo pills and 14 active contraceptive tablets. Alysena 28 is sold in B.C., New Brunswick, Newfoundland and Labrador, Nova Scotia, Ontario, P.E.I. and Quebec.
Recall of contraceptive pill issued
- Idnumber
- 201304090020
- DocID:
- 165570867
- Publication:
- Windsor Star
- Page:
- A5
- Date:
- 2013.04.09
- Section:
- News
- Edition:
- Final
- Source:
- The Canadian Press
- Dateline:
- OTTAWA
- Word Count:
- 131
- Circulation:
- 67530
The maker of the birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets in-stead of three.
Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy. In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug.
Apotex said blister packs in the affected lot - LF01899A - may instead contain 14 placebo pills and 14 active contraceptive tablets. Alysena 28 is sold in British Columbia, New Brunswick, Newfoundland and Labrador, Nova Scotia, Ontario, P.E.I. and Quebec.
National; Urgent recall issued on birth-control pill
- IDNumber
- 201304090038
- DocID:
- 165569376
- Publication:
- Ottawa Citizen
- Page:
- A3
- Date:
- 2013.04.09
- Section:
- News
- Edition:
- Final
- Source:
- Ottawa Citizen
- Word Count:
- 90
- Circulation:
- 128919
The maker of the birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets instead of three. Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy. In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28 day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug.
Birth control pill recalled after concerns of unplanned pregnancies
- Idnumber
- 201304090046
- DocID:
- 165569181
- Publication:
- National Post
- Page:
- A7
- Date:
- 2013.04.09
- Section:
- Canada
- Edition:
- National
- Source:
- The Canadian Press
- Word Count:
- 132
- Circulation:
- 194795
The maker of the birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets instead of three. Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy. In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug. Apotex said blister packs in the affected lot - LF01899A - may instead contain 14 placebo pills and 14 active contraceptive tablets. "I looked at it and said, 'There could be unwanted pregnancies out there," London Drugs vice-president John Tse told the CBC.
Alysena 28 birth control pill recalled
- Idnumber
- 201304090004
- DocID:
- 165569937
- Publication:
- Montreal Gazette
- Page:
-
A10
- Date:
- 2013.04.09
- Section:
- News
- Edition:
- Final
- Source:
- The Gazette
- Column:
- In Brief
- Dateline:
- OTTAWA
- Word Count:
- 131
- Circulation:
- 150159
The maker of the birth-control pill Alysena 28 has issued an urgent recall after it was discovered that one lot of the contraceptive may have been incorrectly packaged with only two weeks of drug-containing tablets instead of three. Women taking the incorrectly packaged pill would not receive the proper amount of contraceptive, raising the possibility of an unplanned pregnancy. In a recall notice distributed by Health Canada, Apotex Inc. said the product's 28-day blister packs should include 21 active birth-control pills plus seven placebo tablets that contain no drug. Apotex said blister packs in the affected lot - LF01899A - may instead contain 14 placebo pills and 14 active contraceptive tablets. Alysena 28 is sold in British Columbia, New Brunswick, Newfoundland and Labrador, Nova Scotia, Ontario, P.E.I. and Quebec.
Rappel urgent du contraceptif Alysena 28
- Publication:
- Le Soleil
- Date:
- 2013.04.09
- Section:
- Actualités
- Page:
- 18
- Column:
- En bref
- Source:
- La Presse Canadienne
- Word Count:
- 126
- Circulation:
- 81820
La compagnie pharmaceutique Apotex, fabricant du contraceptif oral Alysena 28, a publié un urgent rappel, hier, en raison d'une possible erreur d'empaquetage. Selon le site Internet Canadiens en santé du gouvernement du Canada, des plaquettes alvéolaires peuvent contenir deux rangées (14 comprimés) de comprimés placebo et deux rangées (14 comprimés) de comprimés actifs. Selon les spécifications, les plaquettes devraient contenir une rangée de comprimés placebo (sept comprimés) et trois rangées de comprimés actifs. Ainsi, les femmes utilisant le produit erronément empaqueté ne recevraient pas la quantité requise de contraceptif, soulevant le risque d'une grossesse non planifiée. Selon Apotex, un seul lot - LF01899A - serait visé par ce rappel. Le produit est distribué au Québec, en Ontario, dans les quatre provinces de l'Atlantique et en Colombie-Britannique.