Food and Drugs Act Liaison Office: Report on Activities, April 2014 - March 2015
Table of Contents
Introduction
The Food and Drugs Act Liaison Office (FDALO) provides an avenue for staff and external stakeholders to obtain information and dispute resolution services related to the Food and Drugs Act. During 2014 - 2015, FDALO continued its core work of dispute resolution and building stakeholder relations competence within the Health Products and Food Branch of Health Canada (HPFB). In addition, FDALO led the redesign of the Branch's reconsideration process for human drug submissions, a process the Office will manage in the future. This annual report summarizes the activities of FDALO in 2014-2015 and provides insight into stakeholder experiences based on the Office's unique perspective within Health Canada.
Summary of Accomplishments
Managing the Reconsideration Process
In 2014, the Office was asked by Senior Management to review the reconsideration processes in the Health Products and Food Branch (HPFB). The reconsideration process is an administrative recourse within Health Canada for human drug sponsors who disagree with a licensing decision. FDALO was asked to find ways to enhance the impartiality and transparency of the reconsideration process which have been questioned by external stakeholders. This initiative is part of Health Canada's overall commitment to greater transparency and openness to further strengthen trust in its regulatory decisions.
While there are multiple reconsideration processes at Health Canada, the first phase of the review was to specifically revise the reconsideration process for pre-market human drugs decisions rendered by the Therapeutics Products Directorate (TPD) and Biologics and Genetics Therapies Directorate (BGTD). A working group led by FDALO redesigned the reconsideration process. Within the revised model, FDALO, as an impartial and arms-length Office located in the Communications and Public Affairs Branch (CPAB), became responsible for administering the reconsideration process. This process is outlined in the revised Guidance Document: Reconsideration of Decisions Issued for Human Drug Submissions.
Input regarding the revised reconsideration model was sought from external stakeholders in the Fall of 2014. Stakeholder feedback was generally supportive or neutral. In light of the stakeholder feedback, FDALO finalized the guidance document, which was posted on the Health Canada Web site. On April 1, 2015 FDALO began to manage all reconsideration requests received on or after that date.
Over time, FDALO is expected to take responsibility for redesigning and managing the reconsideration processes in other regulatory program areas at HPFB. The Natural and Non-Prescription Health Products Directorate (NNHPD) will be the next to undergo a review.

Download the alternative format
(PDF format, 401 KB, 15 pages)
Case Management
In 2014-2015, the FDALO team remained focused on delivering on its original mandate of providing expedient and informal dispute resolution services for regulated parties and regulatory staff over Food and Drugs Act matters. FDALO managed 100 cases in 2014-2015 (See Figure 1), which is comparable to the previous year. A previously-identified trend has continued: more businesses are contacting the Office, and they represented 49% of all cases in the past fiscal year.
FDALO's services are available to both external stakeholders as well as Health Canada staff. Either of these parties can request impartial dispute resolution assistance. In 2014-2015, nine Health Canada staff approached FDALO for assistance in managing difficult stakeholder relationships.
We categorize cases we receive into two broad types: Information Seeking and Issues Management. Issues Management cases typically involve a complaint. This year, Issues Management represented 64% of all cases. Issues Management cases make the best use of FDALO's dispute resolution expertise. More detail about our cases is provided in a later section.
While the types of cases that FDALO manages differ widely in terms of scientific content and areas of dispute, in each case the stakeholder's basic desire is to have a satisfactory service experience. We share their objective, which is in keeping with the recommendations of the Red Tape Reduction Commission. FDALO's efforts to improve the service experience of stakeholders also satisfy the requirements set out in recent Treasury Board policies including the Interpretation Policy and Policy on Service.
Building Competency
Finally, FDALO continues to play a role at Health Canada in building staff competencies to manage stakeholder relations within a complex and ever-changing regulatory system. This past year, we delivered three sessions of "Making the Most of Difficult Communications with Stakeholders", a two-day experiential training program which has proven popular with participants. Since our creation seven years ago, we have trained over 655 Health Canada and portfolio staff (including the Public Health Agency of Canada and the Pest Management Regulatory Agency), as well as over 140 Community of Federal Regulators staff from across government.
What We Heard
FDALO's role gives us a perspective on how stakeholders feel about the department's administration of the Food and Drugs Act in any given year. We report on both improvements that have been noted, as well as suggestions which may identify opportunities for future improvements.
The trends noted in this section of the report are not necessarily based on a cluster of feedback or complaints related to a particular issue. Rather, trends are identified based on both cases we receive and anecdotal information we glean in our interactions with various stakeholders.
Stakeholders Noted Improvements
Stakeholders offered positive feedback in 2014-2015 about two changes Health Canada made to the administration of the Food and Drugs Act.
Streamlining Regulation of Similar Risk Products
In July 2013, the department transferred the review of non-prescription drugs and disinfectants to what is now known as the Natural and Non-prescription Health Products Directorate (NNHPD). This aligned review uses similar approaches and practices among products of similar risk in order to increase efficiencies and consistency in decision-making.
Stakeholders from the various business sectors have noted significant improvements since this change. They say there is better communication and greater harmonization in the regulatory approach for these products, many of which have a similar risk profile. NNHPD efforts in this regard have been positively received, including the Non-prescription Drug Monograph Attestation Pilot, launched in August 2014. This pilot project eliminates the need for a Category IV Drug Submission review and allows approval of non-prescription drug products supported entirely by one of eight product monographs. This expedited regulatory process for non-prescription drugs is consistent with the process for product licensing that is in place for natural health products. The reduction in administrative burden has been noted with approval by a number of stakeholders.
Therapeutic Products Division Minimizes Generic Drug Review Backlog
Generic drug submissions increased significantly when patent protection ended for a record number of 'blockbuster' drugs between 2008 and 2013. This created a significant backlog in the review and licensing of generic drugs. TPD took several steps through this period to retire the backlog and ultimately achieved this result in November 2013.
Manufacturers of generic drugs have expressed satisfaction that TPD has been able to maintain its performance targets for generic drug reviews over the 2014-2015 period and avoided any significant backlog.
Stakeholders Requested Improvements
The following trends are regulatory issues that led to complaints and requests for improvements.
Publication of Products at Time of DIN Approval
Some regulated parties have expressed concern about the department's decision to publicly publish all products at the time they are granted a drug identification number (DIN).
This initiative was originally launched as part of the Department's commitment to openness and transparency in the drug approval process. As well, it supports the recently-passed Plain Language Labelling Regulations which require companies to carry out a brand name assessment for any new drug product to minimize the risk of their product being confused with another drug product. This assessment requires that companies carry out a name assessment of all approved drug products that could be marketed in Canada.
This publication of products immediately upon approval concerns industry associations and companies whose products do not have patent protection. These include sunscreens, acne therapy, anti-dandruff products, fluoride-containing anti-cavity products, tooth whiteners etc. Their concern is that publishing their products at the time of approval will allow competitors and counterfeiters to replicate their non-patented products before they have an opportunity to launch the products and secure market share. Companies felt they were not given sufficient advance notice of the decision to post DINs at the time of approval, and did not therefore have an opportunity to mitigate the impact on their business processes.
FDALO brought these concerns to the attention of senior management for their consideration.
Appropriate Regulatory Oversight for Consumer Health Products
Although generally supportive of the creation of NNHPD, some regulated parties who manufacture or import cosmetics, cosmetic-like drugs and other personal care products continue to complain of what they consider as administrative burden and lack of efficiency in review related to some of their products. These products, specifically sunscreens (both primary sunscreens and secondary sunscreens such as make-up foundation or lip balm), can be classified as non-prescription drugs, natural health products or cosmetics depending on the active ingredient and labelling claims. Each classification has different regulatory requirements, including some which these companies consider as onerous as those applied to prescription drugs.
Industry cites two recent examples. First, sunscreen manufacturers were included in the departmental notification sent to all drug manufacturers and importers to post anticipated and actual drug shortages and discontinuances in the Canadian Drug Shortage Website. Second, Good Manufacturing Practices (GMP) standards were introduced for active pharmaceutical ingredients (APIs). In both instances, members of this industry sector appealed to the department to modify the requirements for cosmetic-like drugs so that an appropriate level of regulatory oversight would be carried out.
FDALO played an instrumental role in bringing these concerns to the attention of Senior Management. The department made changes to ensure that the administrative and compliance burden for this sector were proportional to the risks presented by their products. The department also launched a consultation between November 2014 and February 2015 on a "Consumer Health Products Framework" that seeks to establish a consistent and aligned approach to the regulation of health products intended for consumer use (disinfectants, natural health products, non-prescription drug products and cosmetics). This initiative was welcomed by industry.
Timeframes for Reviewing Foreign Site GMP Compliance
Companies who sell drugs made outside Canada are required to submit evidence to Health Canada to demonstrate Good Manufacturing Practices (GMP) compliance of those foreign facilities. A company cannot manufacture or import any drug product for the Canadian market without receiving a valid Canadian GMP compliance rating. Furthermore, as GMP is an important measure of an establishment's commitment to quality, the Therapeutic Products Directorate (TPD) requires a valid GMP compliance rating for a proposed manufacturing site prior to accepting a drug submission into review. According to departmental performance standards, the process (outlined in the guidance document GUI-0080) should be completed within 250 days. Companies have complained that long review times for a foreign site GMP rating can cause significant delays in bringing new therapeutic products to the Canadian market, but over the past year, stakeholders have said that GMP foreign site review timeframes have improved considerably and are being completed well within the 250 day performance standard. Nonetheless, they complain there is no predictability or certainty regarding when a company can expect to receive its compliance rating. This causes difficulty in business planning. Stakeholders would like to see more predictability around the completion of foreign site reviews, and have suggested a system which differentiates between low-risk and high-risk drug manufacturing sites would be preferable to the present "one size fits all" approach.
Changes to NHP Licenses Cause Export Difficulties
Since the Natural Health Products Regulations were introduced in 2004, NNHPD has made many changes to streamline the product licensing process. It developed monographs to attest to the safety and efficacy of many commonly used NHPs. In 2013, NNHPD implemented a three-class system of product review that matches the review process to what is known about a product. For example, low risk products that comply with a monograph can attest to meeting that monograph, whereas medium and higher risk products require a full review for safety and efficacy.
The "as per monograph" attestation process has many benefits for industry in addition to an expedited licensing timeframe. Companies can make post-license changes that are fully within the parameters of the monograph without the need to file an amendment to their original application or notify the department, as applicable. However, the change by NNHPD to expedite the licensing process has resulted in issuing low risk product licenses that no longer contain detailed information about the product such as the quantity of medicinal ingredients and the source of the ingredients. Canadian companies wanting to export their products have found that some foreign regulatory authorities require details about what specifically has been approved by Health Canada before allowing the import and sale of products, so these new licenses are not adequate.
FDALO requested that NNHPD revisit this issue given the potential impact on the export of Canadian natural health products. After consultation with industry and internal discussions, NNHPD determined that the efficiencies gained in the expedited licensing process would be lost if it reverted to an extensive review of all applications in order to issue detailed product licenses. The solution proposed was that companies can obtain an International Trade Certificate (ITC) from an independent third party that speaks to the detailed regulatory status of an NHP license issued by Health Canada. A similar process was put in place in 2013 when Health Canada transferred the responsibility of ITC issuance to independent third-parties who would carry out the function according to criteria set out by Health Canada.
No Reconsideration Possibility for Partial Refusal
One case this year illustrated an issue that occurs periodically in human drug submissions. A company filed a submission to have a drug approved for several indications of use. The department assessed the safety and efficacy data submitted and determined that it only supported some of the indications. The department gave the company two choices. The first involved receiving a Notice of Non-compliance (NON) to allow the company an opportunity to produce the missing data before the review process would be resumed. The second choice was to have the company withdraw the indications requiring further safety and efficacy data, and have the department issue a Notice of Compliance (NOC) for only some of the indications. The company could choose to file a new application at a later date for the outstanding indications once it had obtained additional data.
The company was frustrated that the options presented did not allow it to request a reconsideration of the department's decision that the safety and efficacy data originally submitted was insufficient for all four indications. Neither choice formalized the department's rejection of two indications. In this instance, the company strenuously disagreed with the department's decision requiring additional safety and efficacy data and wanted to request a reconsideration process of the department's refusal. However, because of the way in which the department presented the company the two choices for bringing the submission to a close, a reconsideration process could not be offered. Other companies have encountered this situation in the past.
The department will examine this issue further to determine if there are alternative ways to managing decisions of this nature.
Who Contacted Us
Both businesses and individuals contact FDALO, but businesses are the most common type of stakeholder contacting FDALO. They are also less likely than individuals to contact FDALO simply seeking information.
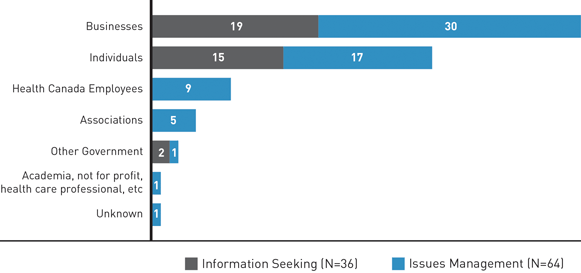
Figure 1: Types of Stakeholders and the Nature of the Cases - Text Equivalent
| Information Seeking | Issues Management | |
|---|---|---|
| Individuals | 15 | 17 |
| Businesses | 19 | 30 |
| Health Canada employees | 0 | 9 |
| Academia, not for profit, health care professionals, etc. | 0 | 1 |
| Other Government | 2 | 1 |
| Associations | 0 | 5 |
| Unknown | 0 | 1 |
| Total | 36 | 64 |
Some larger companies and associations who do a high volume of work with the department have begun to use FDALO services on a repeat basis. As well, we are becoming better known for our expertise in "issues management" (or complaint resolution) as we are receiving more requests to manage complaints than routine requests for information. Issues management cases maximize the use of our skills as dispute resolution practitioners.
Themes Observed
The cases we have received have been further categorized into one or more of the following four themes:
- Communication issues (For example, information-seeking inquiries, unreturned calls, unclear correspondence or correspondence that does not address the stakeholder's concerns.)
- Policy issues (For example, disagreements with the interpretation or application of the law, policies or regulations, such as product classification, risk assessment, policy coherence.)
- Procedural issues (For example, dissatisfaction with the processes used in regulatory decision-making, such as timeliness, openness, transparency, predictability, advance notice of changes to rule making.)
- Interpersonal issues (For example, stakeholder treatment by staff, or staff requests for assistance in dealing with difficult stakeholder communications.)
Issues Management cases in 2014-2015 were most likely to relate to policy and procedural concerns, such as those outlined in this report.
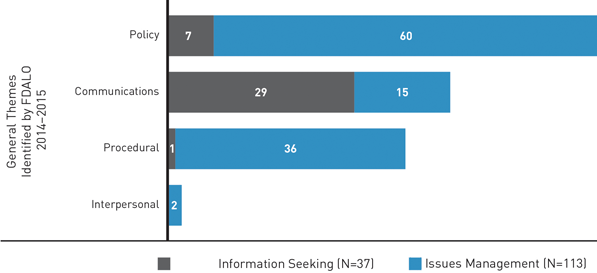
Figure 2: General Themes of Cases - Text Equivalent
| Information Seeking Cases | Issues Management Cases | |
|---|---|---|
| Individual cases may fall into more than one theme. | ||
| Communication Issues | 29 | 15 |
| Policy Issues | 7 | 60 |
| Procedural Issues | 1 | 36 |
| Interpersonal Issues | 0 | 2 |
| Total | 37 | 113 |
A significant proportion of complaints in 2014-2015 related to two specific issues. One concerned the Department's position related to electronic cigarettes (7 cases). The second involved the licensing process for medical marihuana producers (17 cases). FDALO no longer addresses medical marihuana complaints as it does not have a mandate to deal with matters that fall under the Controlled Drugs and Substances Act.
FDALO continues to play an important role in obtaining clarification and assistance for stakeholders when products are seized or refused entry into Canada because of an alleged contravention of the Food and Drugs Act. In some instances, FDALO is able to act as intermediary between the stakeholder and the department to help resolve the issues blocking an importation by ensuring the department is satisfied that regulatory requirements have been met. In other instances, FDALO helps the stakeholder understand the rationale and legal authority for the refusal.
Directorates Involved in our Cases
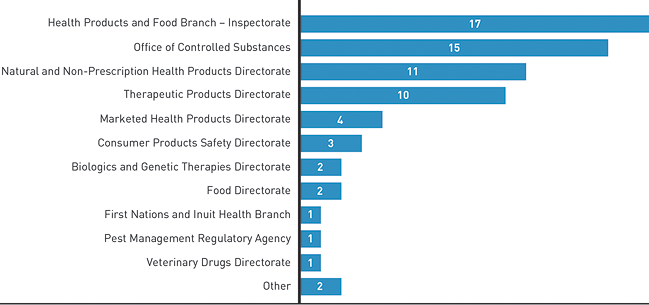
Figure 3: Work Units Implicated in Issues Management Cases - Text Equivalent
Horizontal bar graph detailing which work units were involved in resolving complaint cases.
- Health Products and Food Branch Inspectorate: 17;
- Office of Controlled Substances: 15;
- Natural and Non-Prescription Health Products Directorate: 11;
- Therapeutic Products Directorate: 10;
- Marketed Health Products Directorate: 4;
- Consumer Products Safety Directorate: 3;
- Biologics and Genetic Therapies Directorate: 2;
- Food Directorate: 2;
- First Nations and Inuit Health Branch: 1;
- Pest Management Regulatory Agency: 1;
- Veterinary Drugs Directorate: 1;
- Other: 2.
The bulk of FDALO's Issues Management cases relates to the HPFB Inspectorate, Office of Controlled Substances (OCS), NNHPD, and TPD. The most common Information Seeking cases involved the HPFB Inspectorate, NNHPD, and TPD.
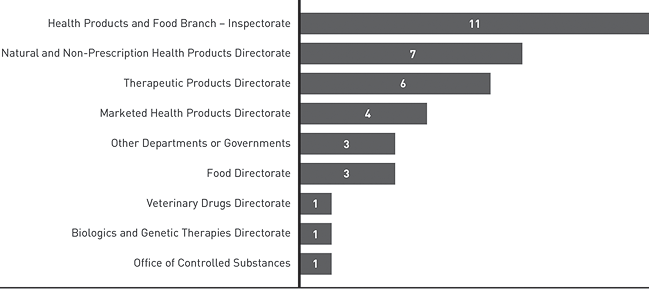
Figure 4: Case Referrals in Information Seeking Cases - Text Equivalent
Horizontal bar graph detailing to whom the Food and Drugs Act Liaison Office made referrals.
- Health Products and Food Branch Inspectorate: 11;
- Natural and Non-Prescription Health Products Directorate: 7;
- Therapeutic Products Directorate: 6;
- Marketed Health Products Directorate: 4;
- Other Departments of Governments: 3;
- Food Directorate: 3;
- Veterinary Drugs Directorate: 1;
- Biologics and Genetic Therapies Directorate: 1;
- Office of Controlled Substances: 1.
In some cases, the Directorates have been directly involved in responding to the stakeholders or have worked with FDALO to respond to the stakeholder inquiries. In other cases, FDALO works directly with the stakeholder to resolve the matter without the involvement of the Directorates. FDALO may offer coaching to stakeholders so that they may have more productive conversations with staff.
A very large proportion of FDALO's cases originate in Ontario.
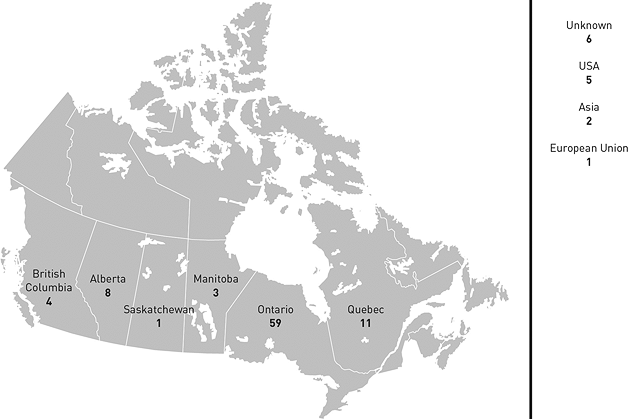
Figure 5: Geographic Origin of Cases - Text Equivalent
Map of Canada detailing geographic origin of cases opened by the Food and Drugs Act Liaison Office.
- Ontario: 59;
- Quebec: 11;
- British Columbia: 4;
- Nova Scotia: 0;
- Alberta: 8;
- Saskatchewan: 1;
- Northwest Territories: 0;
- New Brunswick: 0;
- Prince Edward Island: 0;
- Manitoba: 3;
- Newfoundland: 0;
- Yukon: 0;
- Nunavut: 0;
- United States of America: 5;
- European Union: 1;
- Asia: 2;
- Unknown: 6.
Moving Ahead
In the coming year, the FDALO team will continue to foster positive relations with stakeholders through case management, new training programs for staff on managing communications with stakeholders, and support for the department's Public Service renewal efforts under the government-wide Blueprint 2020 initiative.
On April 1, 2015 we began our new role as process administrators under the new Guidance Document: Reconsideration of Decisions Issued for Human Drug Submissions. We are moving carefully to ensure that we deliver this service in a timely and professional way. FDALO has also struck a new working group with the Natural and Non-prescription Health Products Directorate (NNHPD) to begin the process of building a streamlined reconsideration process for this directorate that will respond to requests from both of these regulatory streams.