Summary of Assessment of Public Health Risk to Canada Associated with Avian Influenza A(H7N9) Virus in China
The risk assessment is reviewed on a regular basis and updated as required.
8 March 2018
What's New?
- The event summary, epidemiologic curve and spatial distribution map for A(H7N9) have been updated to reflect cases reported since the last update to this risk assessment (June 30, 2017).
- A total of 34 new human cases of A(H7N9) were reported to the WHO since the last update, 31 in wave 5 and 3 in wave 6.
- Wave 6 monitoring began on October 1, 2017, with 3 cases reported from Guangdong Province, Xinjiang Uygur Autonomous Region, and Yunnan Province in China with symptom onset from 21 November 2017 – 3 February 2018.
Summary
- The public health risk posed by avian influenza A(H7N9) virus to Canada remains low.
- Human infection with avian influenza A(H7N9) in China follows a seasonal pattern with increases ("waves") typically between January and May each year.
- A total of 766 cases were reported in the (fifth) wave: the highest number of cases reported during any wave since emergence in March 2013. In addition, cases were more widely spatially distributed relative to previous waves, but there were no changes in the transmissibility, severity of infection, and overall risk assessment.
- The number of cases in the fifth wave peaked in January 2017, with 110 cases in that month alone. Since 10 January 2017, the A(H7N9) virus isolates from 32 human cases were found to be highly pathogenic avian influenza (HPAI) virus (Food and Agriculture Organization, 2018).
- On October 1, 2017, monitoring for the sixth wave of avian influenza A(H7N9) began. As of March 8, 2018, there have been 3 cases reported in wave 6.
- Infection is associated with exposure to infected poultry or contaminated environments, and most often results in clinically severe disease.
- In January 2017, highly pathogenic strains of avian influenza A(H7N9) virus were detected from live bird market samples and two human cases in Guangdong China (ECDC, 2017c).
- Approximately 7-9% of highly pathogenic avian influenza A(H7N9) viruses tested are known to have reduced susceptibility to at least one antiviral treatment with a neuraminidase inhibitor, including oseltamivir (European Centre for Disease Prevention and Control [ECDC], 2017b).
- Among all five epidemics, 14 and 26 of A(H7N9) cases were reported respectively as probable and possible human-to-human transmission (Zhou et al., 2018), and the likelihood of sustained human-to-human transmission remains low (ECDC, 2017b).
- As of March 5, 2018, the World Health Organization (WHO) identified four vaccine candidates for A(H7N9) virus namely; A/Guangdong/17SF003/2016, A/Hong Kong/125/2017, A/Shanghai/2/2013 and A/Anhui/1/2013. The A/Hunan/02650/2016 candidate vaccine is under preparation.
- The most likely risk to Canadians at this time remains the risk of travel-acquired infection and importation from China through exposure to infected poultry or contaminated environments.
- This is the summary of the report. You can order the full report.
Risk Assessment
Avian Influenza A(H7N9) virus is an emerging infectious zoonotic pathogen widely spread across several provinces in China. The virus is efficiently transmitted among poultry and sporadically in humans exposed to infected poultry or contaminated environments. Only rare and unsustained transmission has been observed among humans. To date, no animal reservoir for Avian Influenza A(H7N9) virus has been reported in Canada. The public health risk posed by avian influenza A(H7N9) virus to Canadians living in Canada is considered low based on available information at this time. However, the risk may change as new information arises.
Avian influenza A(H7N9) is a novel reassortant virus that was not previously known to circulate in humans.
Available information suggests that this virus does not have the ability to transmit easily among humans. Clusters that have been reported suggest that limited human-to-human transmission may occur when there is unprotected close contact with cases. Due to mutations in low pathogenic avian influenza (LPAI) strains at multiple basic amino acids cleavage sites, an indicator for high pathogenicity in poultry, new highly pathogenic avian influenza HPAI strains of A(H7N9) virus were detected in both poultry and human cases in China. However, there is no evidence of sustained human-to-human transmission of highly-pathogenic avian influenza A(H7N9) virus.
Being widespread in China, the most likely threat to Canadians from Avian influenza A(H7N9) remains a risk of importation to Canada from travelers infected through exposure to infected poultry or contaminated environments while in China. Canada reported the first travel-related human cases of A(H7N9) in North America in January 2015. Although travel-related cases have been reported in other countries, there is no indication that international spread has occurred. However, this information suggests a continued risk of sporadic imported cases with a history of travel to China. Increased travel to and from China is expected every year around the Lunar New Year. The Public Health Agency of Canada monitors the situation closely and updates the public health risk assessment and travel health notices accordingly.
Resistance to Antivirals
About one third of all A(H7N9) virus infections in humans since emergence were reported in China's fifth wave (CIDRAP, 2017). Additionally, about 7% of the viruses from A(H7N9) human cases in the fifth wave had a genetic marker which is linked to resistance to neuraminidase inhibitors like oseltamivir, one of the antivirals used to treat influenza infection. Almost all of the A(H7N9) isolates with neuraminidase resistance markers were isolated from humans treated with antivirals. This information suggests that neuraminidase resistance markers were developed after treatment and were not spread by poultry. Moreover, the additional testing of A(H7N9) viruses from the fifth wave is underway to assess in vitro susceptibility to the neuraminidase inhibitor class of antivirals. From testing of A(H7N9) viruses from previous waves of human infection, all tested viruses carried the S31N mutation on the M2 protein, indicating resistance to amantadine and rimantadine (WHO, 2017e). Continuous genetic testing of viruses isolated from pre- and post-treatment of humans is needed to rule out the possibility of circulation of viruses with neuraminidase inhibitor resistance mutations in humans. The WHO and member countries are closely monitoring the genetic changes in A(H7N9) viruses conferring resistance to neuraminidase inhibitors. However, currently there is no evidence to suggest changes in the clinical management of A(H7N9) human infection.
Transmission of A(H7N9) Virus in Birds
The Avian Influenza A(H7N9) viruses have been detected in wild and captive birds. However, the newly emerged HPAI A(H7N9) viruses are associated with higher infectiousness in poultry than the related low pathogenicity strains (ECDC, 2017b). Additionally, the HPAI viruses are also associated with a higher rate of molecular adaptation. These adaptation processes may cause infections of both resident and migratory birds, posing a risk of global spread through migratory birds. Since the emergence of LPAI A(H7N9) in 2013, this strain has been circulating in domestic poultry in China. In January 2017, the HPAI A(H7N9) virus emerged in poultry in Guangdong China and rapidly spread to other provinces. However, the majority of viruses detected in poultry during the fifth wave were LPAI viruses. This virus appears to be well adapted to poultry with limited transmission to wild birds.
Transmission of A(H7N9) Virus in Humans
Recently in China, a descriptive analysis was conducted on the 40 clusters of the laboratory-confirmed A(H7N9) cases reported during February 1, 2013 – June 30, 2017 (Zhou et al., 2018). Among clusters reported for all five epidemics, a distribution of 14 (35%) probable and 26 (65%) possible instances of human-to-human transmission was reported. Among 14 secondary cases with probable human-to-human transmission, four cases were associated with household exposure, while 10 cases were associated with health care settings. The proportion of clusters with probable human-to-human transmission did not differ significantly by epidemic (p = 0.842). A cluster of three infections in the fifth wave had possible and probable human-to-human transmission. Two similar clusters were identified during the fourth wave. In this study, similarities in number and size of clusters and in the proportion of clusters with probable human-to-human transmission across all waves suggest no change in human-to-human transmission risk.
Event Summary
The sixth wave began on October 1, 2017 and is ongoing. As of March 8, 2018, a total of 3 cases of A(H7N9) have been reported to the WHO for the sixth wave. These cases are spread across China in Guangdong Province, Xinjiang Uygur Autonomous Region, and Yunnan Province. The cases ranged in age from 59 to 72 years old, with a median age of 64 years old. Two of the three cases were male, and all three cases had exposure to poultry before symptom onset. One of the cases had a fatal infection. There is still no evidence of sustained human-to-human transmission (WHO, 2017a).
Figure 1. Global incidence of human cases of avian influenza A(H7N9) by month of symptom onset or earliest recorded date, March 2013 to February 28, 2018.
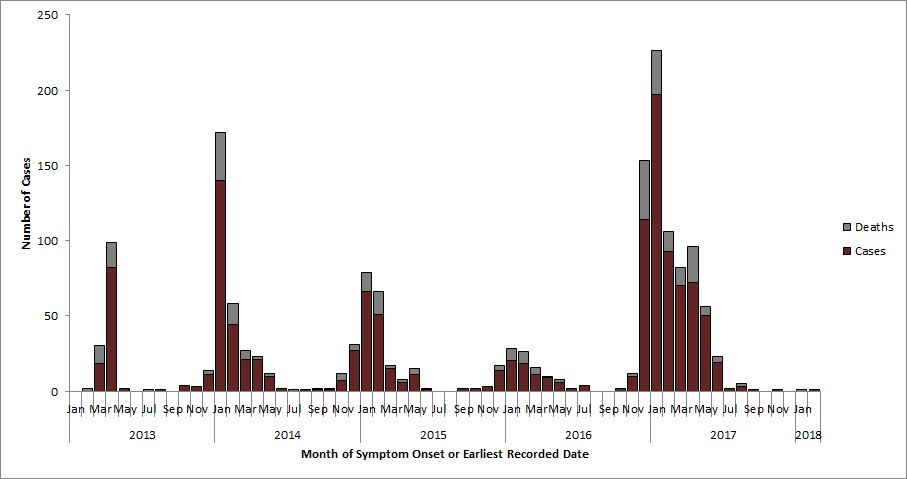
Text Equivalent - Figure 1
Global incidence of human cases of avian influenza A(H7N9) by month of symptom onset or earliest recorded date, March 2013 to February 28, 2018.
Epidemic curve of avian influenza A(H7N9) event. Since 2013 six waves of avian influenza A(H7N9) have occurred : Wave 1 (February 2013–September 2013), Wave 2 (October 2013-September 2014), Wave 3 (October 2014-September 2015), Wave 4 (October 2015- September 2016), Wave 5 (October 2016-September 2017), and Wave 6 (October 2017-present). The distribution of cases over each wave has followed a seasonal pattern with cases increasing in December and peaking in January. The highest number of cases has been observed in wave 5. To date, the epidemiological curve captures 1272 cases and 299 deaths: 103 cases and 32 deaths in wave 1; 258 cases and 61 deaths in wave 2; 187 cases and 47 deaths in wave 3; 89 cases and 27 deaths in wave 4; 633 cases and 131 deaths in wave 5; and 2 cases and 1 death in wave 6.
| Year | Month | Cases | Deaths |
|---|---|---|---|
| 2013 | Jan | 0 | 0 |
| 2013 | Feb | 0 | 2 |
| 2013 | Mar | 18 | 12 |
| 2013 | Apr | 82 | 17 |
| 2013 | May | 2 | 0 |
| 2013 | Jun | 0 | 0 |
| 2013 | Jul | 0 | 1 |
| 2013 | Aug | 1 | 0 |
| 2013 | Sep | 0 | 0 |
| 2013 | Oct | 4 | 0 |
| 2013 | Nov | 3 | 0 |
| 2013 | Dec | 11 | 3 |
| 2014 | Jan | 140 | 32 |
| 2014 | Feb | 44 | 14 |
| 2014 | Mar | 21 | 6 |
| 2014 | Apr | 21 | 2 |
| 2014 | May | 10 | 2 |
| 2014 | Jun | 2 | 0 |
| 2014 | Jul | 0 | 1 |
| 2014 | Aug | 1 | |
| 2014 | Sep | 1 | 1 |
| 2014 | Oct | 1 | 1 |
| 2014 | Nov | 7 | 5 |
| 2014 | Dec | 27 | 4 |
| 2015 | Jan | 66 | 13 |
| 2015 | Feb | 51 | 15 |
| 2015 | Mar | 15 | 2 |
| 2015 | Apr | 6 | 2 |
| 2015 | May | 11 | 4 |
| 2015 | Jun | 1 | 1 |
| 2015 | Jul | 0 | 0 |
| 2015 | Aug | 0 | 0 |
| 2015 | Sep | 2 | 0 |
| 2015 | Oct | 2 | 0 |
| 2015 | Nov | 3 | 0 |
| 2015 | Dec | 14 | 3 |
| 2016 | Jan | 20 | 8 |
| 2016 | Feb | 18 | 8 |
| 2016 | Mar | 11 | 5 |
| 2016 | Apr | 9 | 1 |
| 2016 | May | 6 | 2 |
| 2016 | Jun | 2 | 0 |
| 2016 | Jul | 4 | 0 |
| 2016 | Aug | 0 | 0 |
| 2016 | Sep | 0 | 0 |
| 2016 | Oct | 2 | 0 |
| 2016 | Nov | 10 | 2 |
| 2016 | Dec | 114 | 39 |
| 2017 | Jan | 197 | 29 |
| 2017 | Feb | 93 | 13 |
| 2017 | Mar | 70 | 12 |
| 2017 | Apr | 72 | 24 |
| 2017 | May | 50 | 6 |
| 2017 | June | 19 | 4 |
| 2017 | July | 2 | 0 |
| 2017 | Aug | 3 | 2 |
| 2017 | Sep | 1 | 0 |
| 2017 | Oct | 0 | 0 |
| 2017 | Nov | 1 | 0 |
| 2017 | Dec | 0 | 0 |
| 2018 | Jan | 0 | 1 |
| 2018 | Feb | 1 | 0 |
Figure 2. Spatial distribution of human cases of avian influenza A(H7N9), China, March 2013 to February 28, 2018.
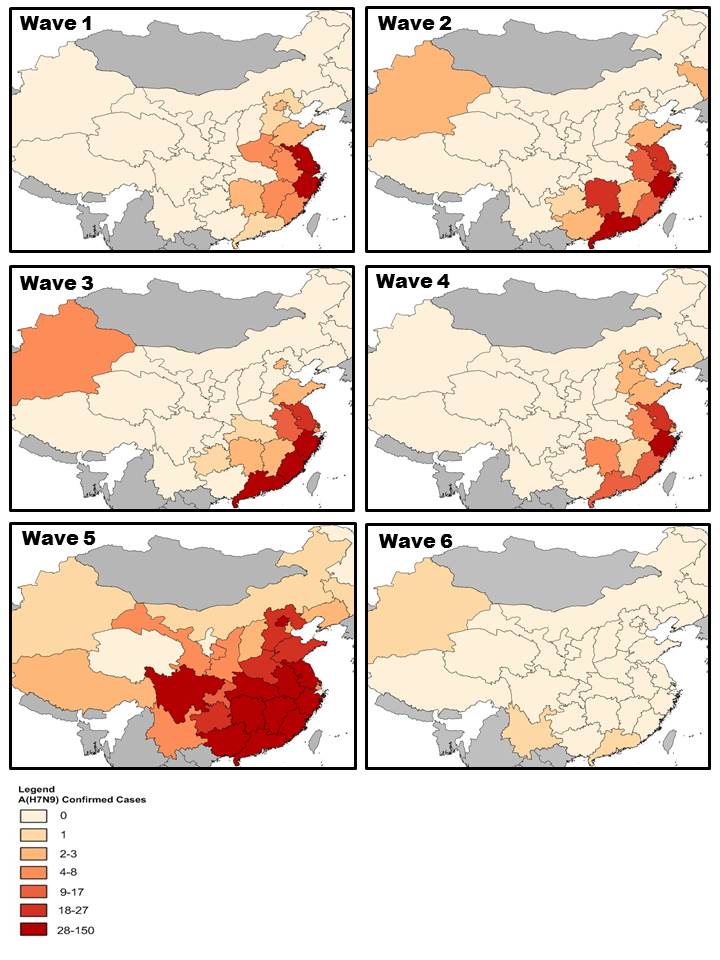
Note: Data for waves one to four were prepared using information compiled by the Public Health Agency of Canada's Global Public Health Intelligence Network (GPHIN) (n=809). Data for waves five and six were obtained from the WHO Influenza at the Human-Animal Interface Monthly Risk Assessment Summary (data as of 25 January 2018) and the WHO Event Information Site reports (data as of 8 March 2018) and includes 769 cases (766 from wave 5, 3 from wave 6).
Text Equivalent - Figure 2
Geographic distribution cases of A(H7N9) by wave. Across all six waves, cases have predominantly occurred in the southeast region of China. In wave 5, the occurrence of cases has become more intense and widespread in comparison to previous waves.
| Name | ID | Wave 1 | Wave 2 | Wave 3 | Wave 4 | Wave 5 | Wave 6 |
|---|---|---|---|---|---|---|---|
| Anhui | 1 | 4 | 14 | 16 | 5 | 62 | 0 |
| Beijing | 2 | 2 | 2 | 2 | 1 | 28 | 0 |
| Chongqing | 3 | 0 | 0 | 0 | 0 | 9 | 0 |
| Fujian | 4 | 0 | 17 | 41 | 12 | 35 | 0 |
| Gansu | 5 | 0 | 0 | 0 | 0 | 5 | 0 |
| Guangdong | 6 | 1 | 108 | 75 | 16 | 63 | 1 |
| Guangxi | 7 | 0 | 3 | 0 | 0 | 28 | 0 |
| Guizhou | 8 | 0 | 1 | 1 | 0 | 18 | 0 |
| Hainan | 9 | 0 | 0 | 0 | 0 | 0 | 0 |
| Hebei | 10 | 1 | 0 | 0 | 2 | 27 | 0 |
| Heilongjiang | 11 | 0 | 0 | 0 | 0 | 0 | 0 |
| Henan | 12 | 4 | 0 | 0 | 0 | 24 | 0 |
| Hubei | 13 | 0 | 0 | 1 | 0 | 29 | 0 |
| Hunan | 14 | 2 | 22 | 2 | 8 | 59 | 0 |
| Jiangsu | 15 | 28 | 27 | 23 | 26 | 148 | 0 |
| Jiangxi | 16 | 6 | 2 | 3 | 1 | 38 | 0 |
| Jilin | 17 | 0 | 2 | 0 | 0 | 1 | 0 |
| Liaoning | 18 | 0 | 0 | 0 | 1 | 3 | 0 |
| Nei Mongol | 19 | 0 | 0 | 0 | 0 | 1 | 0 |
| Ningxia Hui | 20 | 0 | 0 | 0 | 0 | 0 | 0 |
| Qinghai | 21 | 0 | 0 | 0 | 0 | 0 | 0 |
| Shaanxi | 22 | 0 | 0 | 0 | 0 | 7 | 0 |
| Shandong | 23 | 2 | 3 | 3 | 2 | 19 | 0 |
| Shanghai | 24 | 33 | 8 | 8 | 3 | 5 | 0 |
| Shanxi | 25 | 0 | 0 | 0 | 0 | 3 | 0 |
| Sichuan | 26 | 0 | 0 | 0 | 0 | 38 | 0 |
| Tianjin | 27 | 0 | 0 | 0 | 1 | 3 | 0 |
| Xinjiang Uygur | 28 | 0 | 2 | 8 | 0 | 1 | 1 |
| Xizang | 29 | 0 | 0 | 0 | 0 | 3 | 0 |
| Yunnan | 30 | 0 | 0 | 0 | 0 | 7 | 1 |
| Zhejiang | 31 | 46 | 93 | 45 | 34 | 91 | 0 |
The first human cases with the novel reassortant avian influenza A(H7N9) virus were reported to the World Health Organization (WHO) by China on March 31, 2013. Since then, additional cases have been reported in China from twenty Provinces (Anhui, Guangdong, Guangxi, Guizhou, Hebei, Henan, Hubei, Hunan, Fujian, Jiangsu, Jiangxi, Jilin, Liaoning, Shandong, Sichuan, Yunnan, Gansu, Shaanxi, Shanxi and Zhejiang), four Municipalities (Beijing, Chongqing, Tianjin and Shanghai) and in four autonomous regions/territories (Hong Kong, Macao, Tibet and Xinjiang Uyghur). Poultry and environmental samples have tested positive for avian influenza A(H7N9) in all above mentioned areas, except for Beijing (Food and Agricultural Organizations of the United Nations (FAO), 2017). Most human infections are believed to have occurred after exposure to infected poultry or contaminated environments. In addition, travel-related cases have been reported in Malaysia, Taiwan, and Canada.
Since March 2013, the WHO has reported over 1500 confirmed human cases, including at least 613 deaths (World Health Organization, 2017d). For the latest updates on cases and deaths please visit WHO's Disease Outbreak News website.
Since the virus first emerged in 2013, outbreaks of A(H7N9) in humans have occurred in six waves. The beginning of the first wave was marked by the detection of the novel avian influenza A(H7N9) in three patients from China in March 2013: two cases from Shanghai and one from Anhui. Subsequent cases were concentrated in the Eastern Chinese Provinces of Shanghai, Zhejiang, and Jiangsu. The first wave lasted to May 2013.
The second wave of human avian influenza A(H7N9) infections began in October 2013 and ended in the spring of 2014. A change in the epidemiologic profile of A(H7N9) was observed in the second wave. There was a geographic shift in the distribution of A(H7N9) cases to the Southern Chinese Province of Guangdong, suggesting the virus became more widespread in its domestic bird reservoir. Additionally, the second wave had significantly larger amplitude than the first wave, both in terms of number of cases and deaths.
The third wave of human infections began in October 2014 with declining number of cases by the spring of 2015. The majority of cases were reported during the months of January and February in Guangdong. On January 26, 2015, the first imported human case of avian influenza A(H7N9) from China was confirmed in Canada. A second case was confirmed on January 29, 2015. They were the first North Americans known to have been infected with this virus. The individuals were from British Columbia and travelled together to China. Neither case required hospitalization and both fully recovered. The number of cases and deaths reported in this third wave was similar to those observed in the second wave.
The fourth wave of influenza A(H7N9) was announced by the Food and Agriculture Organization (FAO) of the United Nations on October 15, 2015 after the report of two new cases of influenza A(H7N9) in China. A total of 121 cases including 34 deaths were reported during the fourth wave which is less than the total in each of the previous waves. There was no change in epidemiologic profile relative to waves two and three.
The fifth wave started on October 1, 2016 and ended in September 2017. Eight cases were reported between October 1 and November 30 2016, which was within the expected range for that time period (Zhou et al., 2017). Beginning December 1, 2016 there was a marked increase in the number of cases: a total of 766 cases were reported in wave 5, which is higher than the total number reported during each prior wave (Figure 1) (World Health Organization, 2017c).
Although the majority of human cases have resulted in clinically severe disease, human infections with avian influenza A(H7N9) can present with a variety of symptoms that range from mild illness to more severe disease. Children infected with the virus have mainly presented with mild or asymptomatic disease (Yi et al., 2015). Given the detection of several less severe cases through influenza-like illness (ILI) surveillance, along with continued reporting of severe cases, continued vigilance is warranted (World Health Organization, 2014b). The WHO continues to advise countries to move forward in strengthening influenza epidemiological and virological surveillance, reporting of human infections as applicable under the International Health Regulations (IHR) (2005), and other national health preparedness actions.
Summary of Literature
Evidence to date suggests a seasonal pattern to A(H7N9) infections in humans, peaking in the winter months and sporadic cases in the summer. The seasonal pattern observed is not unexpected as previous studies have reported greater circulation of avian influenza viruses in cooler weather and lower circulation in warmer temperatures (Q. Li et al., 2014).
The sex and age distribution of human cases of avian influenza A(H7N9) is skewed to middle aged or elderly males. This gender bias is not well understood, however it has been attributed by some researchers to cultural practices, such as live poultry markets (LPMs) predominantly being visited by males (Jernigan & Cox, 2015). There is a growing body of evidence that points to infected poultry, and contaminated environments including LPMs to be a key source of A(H7N9) infections in humans (Jernigan & Cox, 2015; J. Li et al., 2015; Spackman, Pantin-Jackwood, Swayne, Suarez, & Kapczynski, 2015; Yi et al., 2015). The vast majority (~85%) of all reported cases have had exposure to poultry or LPMs (Jernigan & Cox, 2015; Qin et al., 2015). Studies have shown that circulation of the virus may be amplified in LPMs with poultry-to-poultry transmission which may then serve as an ongoing source for A(H7N9) infection in humans (Jernigan & Cox, 2015). Poultry and environmental samples from several LPMs in China have tested positive for the virus with genomic sequences similar to those found in infected humans (Jernigan & Cox, 2015; Kang et al., 2015; Spackman et al., 2015; World Health Organization, 2017a; Yi et al., 2015). Since 10 January 2017, the highly pathogenic avian influenza (HPAI) type A(H7N9) virus was detected in 54 poultry or environmental samples; 42 chickens, 2 duck and 10 environmental samples (Food and Agriculture Organisation, 2018). Unlike human infections with A(H7N9), the virus has not been reported to cause severe disease in poultry. The absence of disease in poultry makes it challenging to detect and control the virus in the poultry population.
Although the vast majority of cases have been linked to poultry exposure, there have been 24 clusters (2-3 cases) reported in waves one to four associated with close contacts of infected cases (World Health Organization, 2013a; World Health Organization, 2015). Epidemiologic investigations suggest secondary transmission may have occurred in some instances in family members and caregivers exposed to infected cases (Y. Liu et al., 2015; World Health Organization, 2013a; World Health Organization, 2015; Yi et al., 2015). Of note, a healthcare associated cluster reported during wave 4 in Shantou, China in February 2015 points to a risk for healthcare workers (Farooqui et al., 2016; Y. Liu et al., 2015). A study published in May 2015 reported that secondary cases of A(H7N9) were younger and presented with milder disease than sporadic/index cases (Jernigan & Cox, 2015; Qin et al., 2015). These results further support other studies that indicate the detection of A(H7N9) cases to be biased towards older, more severe cases and are likely not capturing younger populations (Centre for Health Protection of the Department of Health HK, 2017b). Epidemiologic investigations of these clusters are suggestive of possible limited human-to-human transmission with no evidence of sustained transmission in the community.
Virus Characteristics
Avian influenza A(H7N9) is a triple reassortant avian influenza A virus with HA genes from Eurasian A(H7) avian influenza virus lineage, NA genes from avian influenza A(H11N9) and A(H7N9), and internal genes from A(H9N2), that emerged in poultry populations in China (European Centre for Disease Prevention and Control, 2017b; D. Liu et al., 2013; Wu et al., 2015). Until February 2017, this novel low pathogenic avian influenza virus did not appear to cause severe illness in birds. However, mutations in the hemagglutinin protein have since been detected that may make the virus more pathogenic in poultry. This is not expected to have an impact on severity of human illness (World Health Organization, 2017b). Mutations in A(H7N9) strains have been identified that could favour high affinity interaction with human receptors in the upper respiratory tract; however, there has been no evidence of sustained human-to-human transmission (World Health Organization, 2013b). In February 2017, a mutation in the neuraminidase protein was detected in virus samples from human cases in China that conferred resistance to oseltamivir (WHO press conference: Expert briefing on the current waves of avian influenza outbreaks - March 1, 7:30 EST 2017; World Health Organization, 2017b).
The virulence and transmissibility of avian influenza A(H7H9) virus has recently been reviewed (Subarao, 2018). Both the virulence and transmissibility of Highly Pathogenic Avian Influenza (HPAI) A(H7N9) is similar to Low Pathogenicity Avian Influenza (LPAI) prototype virus from 2013, A/Anhui/1/2013. Following replication in mammals, the avian influenza viruses often acquire mutations in the PB2 polymerase protein, which is associated with enhanced virulence and air-borne transmission (Subarao, 2018). Moreover, previous transmission studies of A(H7N9) viruses from 2013 using ferret models also indicate that the virus can transmit efficiently through direct contact but inefficiently through respiratory droplets (WHO, 2017e). The HPAI viruses with mutations in the PB2 protein have been recovered from ferrets that were experimentally infected with the A(H7N9) virus. These viruses were lethal in mice and as transmissible as the A/Anhui/1/2013 LPAI virus, but were more virulent in ferrets (Richard et al., 2013). These findings are consistent with a recent report about a human HPAI A(H7N9) isolate (Imai et al., 2017). These data suggest that the progeny of avian influenza A(H7N9) viruses have acquired mutations in the PB2 protein.
The majority of avian influenza A(H7N9) cases reported in the fifth wave were associated with the Yangtze River Delta lineage (Public Health England, 2018). This was in contrast with previous waves, where the majority of A(H7N9) cases were associated with strains from the Pearl River Delta lineage. This shift in the strains' lineage is not associated either with a change in virus transmissibility or significant change in demographics of cases and severity of infection, suggesting no change in risk assessment. However, change in the geographical distribution and number of human cases during the fifth wave in China suggest the importance of close monitoring of emergence of cases and considering the strains from the Yangtze River Delta lineage for candidate vaccine viruses.
Medical Countermeasures
The WHO Collaborating Centers have developed candidate influenza vaccine viruses (CVVs) for pandemic preparedness purposes. These include both A/Anhui/1/2013-like and A/Shanghai/2/2013-like viruses (World Health Organization, 2014a). Two new CVVs were recommended by the WHO in March 2017 (World Health Organization, 2017a). However, as per WHO influenza vaccine composition consultations, the recent highly pathogenic viruses first detected during wave five and some of the currently circulating low pathogenic viruses are genetically and antigenically distinct from the current A(H7N9) candidate vaccine viruses (European Centre for Disease Prevention and Control, 2017a). Sequences are shared through Global Initiative on Sharing All Influenza Data (GISAID) with WHO Collaborating Centers and other partners to improve diagnostics and for the development of candidate vaccines. The timely characterization of A(H7N9) viruses and the sharing of sequence information remain crucial for the development of A(H7N9) vaccine virus. There are currently no recommendations on the large-scale manufacture of avian influenza A(H7N9) vaccine (World Health Organization, 2013b).
Laboratory testing has confirmed that the avian influenza A(H7N9) virus is susceptible to the neuraminidase inhibitors oseltamivir and zanamivir, two antiviral medications that are available in the National Antiviral Stockpile and National Emergency Stockpile System should they be needed to treat Canadians. WHO has issued guidance on the use of post-exposure antiviral chemoprophylaxis for avian influenza A (H7N9) (PDF Document). Recent human cases with mutations conferring resistance to neuraminidase inhibitors were reported in wave 5.
The Agency's National Microbiology Laboratory (NML) has developed diagnostic assays (tests) allowing NML to rapidly detect the novel avian influenza A(H7N9) virus. These assays have been shared with the provincial/territorial laboratories allowing provinces to do their own testing should it be required.
Zoonotic Circulation in North America
Presently, this avian influenza A(H7N9) virus is considered a Foreign Animal Disease (FAD) agent as there may be consequences if this pathogen were to circulate in avian populations in Canada. No North American circulation of Eurasian strain A(H7N9) has been detected in wild or domestic birds. The North American high and low pathogenic avian influenza A(H7N9) detected in poultry in the United States differs from the Eurasian strain associated with the fifth wave in China (United States Department of Agriculture., 2017) (Alabama Department of Agriculture and Industries, 2017).
Recommendations for Canada
Surveillance
Under the authority of International Health Regulations, human infections with avian influenza A(H7N9) are notifiable through Canada's notification system. Health care professionals are encouraged to maintain vigilance for cases of avian influenza A(H7N9) infection and notify the appropriate local public health unit in their jurisdiction of any persons under investigation. The national case definitions for avian influenza A(H7N9) are to be used for the surveillance of avian influenza A(H7N9). For guidance on surveillance objectives and activities, please refer to the interim national surveillance guidelines for avian influenza A(H7N9).
Provinces and Territories are asked to report confirmed cases of avian influenza A(H7N9) infection to the Public Health Agency of Canada using the Emerging Pathogens and Severe Acute Respiratory Infection (SARI) Case Report Form.
Travel Health Notice
The Agency releases Travel Health Notices outlining potential risks to Canadian travelers and to the Canadian public, and recommends measures that can be taken to help reduce these risks. A travel health notice for avian influenza A(H7N9) is posted on the Government of Canada's website travel.gc.ca.
Infection Control
The Agency provides recommendations for infection prevention and control measures for patients presenting to hospitals, doctors offices and other clinics with suspected or confirmed infection with the avian influenza A(H7N9) virus in its Interim Guidance - Infection Prevention and Control Guidance for Acute Care Settings document. This guidance will be updated as new evidence becomes available.
Biosafety
Based on the clinical presentation of severe respiratory illness and death in humans, the potential for this virus to be a pandemic agent, and that the virus is currently considered a foreign animal disease agent, this avian influenza A(H7N9) virus is classified as a Risk Group 3 human and animal pathogen requiring Containment Level 3 for all proliferative in vitro or in vivo activities. Non-proliferative diagnostic/clinical activities can be conducted at Containment Level 2 with additional requirements. In the event of a non-negative human sample, it is strongly recommended that the work with the sample be stopped and the sample be transferred to the National Microbiology Laboratory (NML). In the event that a veterinary diagnostic laboratory detects a non-negative sample, the work is to be stopped and the sample be transferred to the National Centre for Foreign Animal Disease (NCFAD) as per the policy in the Foreign Animal Disease Diagnostic Laboratory Containment Standard. The biosafety advisory can be found on the PHAC website.
References
- Alabama Department of Agriculture and Industries. (2017). Press release: Additional poultry flocks test positive for low pathogenic avian influenza. Retrieved March/22, 2017, from http://www.agi.alabama.gov/s/avian-influenza/additional-poultry-flocks-test-positive-for-low-pathogenic-avian-influenza
- Centre for Health Protection of the Department of Health HK. (2017). Avian influenza report 2017 [wk 17-20]. Retrieved May 24, 2017, from http://www.chp.gov.hk/en/guideline1_year/29/134/332.html
- CIDRAP (2017) WHO: H7N9 antiviral resistance likely arose after treatment H7N9 Avian Influenza Avian Influenza (Bird Flu) (http://www.cidrap.umn.edu/news-perspective/2017/03/who-h7n9-antiviral-resistance-likely-arose-after-treatment)
- European Centre for Disease Prevention and Control. (2017a). Rapid risk assessment Influenza A(H7N9) virus in China – Implications for Public Health: Seventh Update, Stockholm: ECDC, 2017.
- European Centre for Disease Prevention and Control. (2017b). Rapid risk assessment: Human infection with avian influenza A(H7N9) virus fifth update No. 5). Stockholm: ECDC, 2017.
- European Centre for Disease Prevention and Control. (2017c) Mutation of avian influenza A(H7N9): now highly pathogenic for poultry but risk of human-to-human transmission remains low. ECDC Comment, February 2017 (https://ecdc.europa.eu/en/news-events/mutation-avian-influenza-ah7n9-now-highly-pathogenic-poultry-risk-human-human).
- Farooqui, A., Liu, W., Kelvin, D. J., H7N9 Shantou Working Group, Zeng, T., Liu, Y., et al. (2016). Probable hospital cluster of H7N9 influenza infection. The New England Journal of Medicine, 374(6), 596-598. doi:10.1056/NEJMc1505359 [doi]
- Food and Agricultural Organizations of the United Nations (FAO). (2017). H7N9 situation update-24 January 2017. Retrieved February/28, 2017, from http://www.fao.org/ag/againfo/programmes/en/empres/H7N9/situation_update.htm
- Food and Agricultural Organizations of the United Nations (FAO). (2018). H7N9 situation update-28 February 2018. From http://www.fao.org/ag/againfo/programmes/en/empres/h7n9/situation_update.html
- Imai, M., Watannabe, T., Kiso, M., et al. (2017). A Highly Pathogenic Avian H7N9 Influenza Virus Isolated From a Human Is Lethal in Some Ferrets Infected via Respiratory Droplets Cell Host Microbe, 22: 615-624.
- Jernigan, D. B., & Cox, N. J. (2015). H7N9: Preparing for the unexpected in influenza. Annual Review of Medicine, 66, 361-371. doi:10.1146/annurev-med-010714-112311 [doi]
- Kang, M., He, J., Song, T., Rutherford, S., Wu, J., Lin, J., et al. (2015). Environmental sampling for avian influenza A(H7N9) in live-poultry markets in guangdong, china. PloS One, 10(5), e0126335. doi:10.1371/journal.pone.0126335 [doi]
- Li, J., Chen, J., Yang, G., Zheng, Y. X., Mao, S. H., Zhu, W. P., et al. (2015). Case-control study of risk factors for human infection with avian influenza A(H7N9) virus in shanghai, china, 2013. Epidemiology and Infection, 143(9), 1826-1832. doi:10.1017/S0950268814003264 [doi]
- Li, Q., Zhou, L., Zhou, M., Chen, Z., Li, F., Wu, H., et al. (2014). Epidemiology of human infections with avian influenza A(H7N9) virus in china. The New England Journal of Medicine, 370(6), 520-532. doi:10.1056/NEJMoa1304617 [doi]
- Liu, D., Shi, W., Shi, Y., Wang, D., Xiao, H., Li, W., et al. (2013). Origin and diversity of novel avian influenza A(H7N9) viruses causing human infection: Phylogenetic, structural, and coalescent analyses. Lancet (London, England), 381(9881), 1926-1932. doi:10.1016/S0140-6736(13)60938-1 [doi]
- Liu, Y., Paquette, S. G., Zhang, L., Leon, A. J., Liu, W., Xiuming, W., et al. (2015). The third wave: H7N9 endemic reassortant viruses and patient clusters. Journal of Infection in Developing Countries, 9(2), 122-127. doi:10.3855/jidc.6759 [doi]
- Public Health England (2018). Risk assessment of avian influenza A(H7N9) – 6th update 17 Jan 2018, from https://www.gov.uk/government/publications/avian-influenza-a-h7n9-public-health-england-risk-assessment/risk-assessment-of-avian-influenza-ah7n9-sixth-update
- Qin, Y., Horby, P. W., Tsang, T. K., Chen, E., Gao, L., Ou, J., et al. (2015). Differences in the epidemiology of human cases of avian influenza A(H7N9) and A(H5N1) viruses infection. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America, 61(4), 563-571. doi:10.1093/cid/civ345 [doi]
- Richard M, Schrauwen EJ, de Graaf M, Bestebroer TM, Spronken MI, et al., (2013). Limited airborne transmission of H7N9 influenza A virus between ferrets. Nature. 26; 501 (7468): 560-3. doi: 10.1038/nature12476. Epub 2013 Aug 7.
- Spackman, E., Pantin-Jackwood, M., Swayne, D. E., Suarez, D. L., & Kapczynski, D. R. (2015). Impact of route of exposure and challenge dose on the pathogenesis of H7N9 low pathogenicity avian influenza virus in chickens. Virology, 477, 72-81. doi:10.1016/j.virol.2015.01.013 [doi]
- Subarao, Kanta (2018). Avian Influenza H7H9 viruses: a rare second warning. Cell Research, 28: 1-2. doi:10.1038/cr.2017.154; published online 1 December 2017
- United States Department of Agriculture. (2017). USDA issues update on highly pathogenic avian influenza in Tennessee. Retrieved March/01, 2017, from http://www.phac-aspc.gc.ca/eri-ire/h7n9/risk_assessment-evaluation_risque-eng.php
- WHO press conference: Expert briefing on the current waves of avian influenza outbreaks - march 1, 7:30 EST(2017). In World Health Organization (Ed.), Stockholm: World Health Organization. (2013a). Disease outbreak news: Human infection with influenza A(H7N9) virus in china. Retrieved March/04, 2014, from http://www.who.int/csr/don/2013_04_01/en/
- World Health Organization. (2013b). WHO recommendation on influenza A(H7N9) vaccine virus. Retrieved March/11, 2017, from http://www.who.int/influenza/human_animal_interface/influenza_h7n9/201309_h7n9_recommendation.pdf
- World Health Organization. (2014a). Summary of status of development and availability of avian influenza A(H7N9) candidate vaccine viruses and potency testing reagents. Retrieved March/11, 2017, from http://www.who.int/influenza/vaccines/virus/candidates_reagents/summary_a_h7n9_cvv_20140213.pdf
- World Health Organization. (2014b). WHO risk assessment: Human infection with avian influenza A(H7N9) (No. 4). Geneva: World Health Organization.
- World Health Organization. (2015). WHO risk assessment of human infections with avian influenza A(H7N9) virus (No. 8). Geneva: World Health Organization.
- World Health Organization. (2017a). Antigenic and genetic characteristics of zoonotic influenza viruses and development of candidate vaccine viruses for pandemic preparedness. (No. 21). Geneva: World Health Organization.
- World Health Organization. (2017b). Disease outbreak news: Human infection with avian influenza A(H7N9) virus-china. Retrieved February/28, 2017, from http://www.who.int/csr/don/27-february-2017-ah7n9-china/en/
- World Health Organization. (2017c). Influenza at the human-animal interface: Summary and assessment, 17 May 2017 to 15 June 2017 (Government No. 127). Geneva: World Health Organization.
- World Health Organization. (2017d). Influenza at the human-animal interface: Summary and assessment, 8 December 2017 to 25 January 2018 (Government No. 133). Geneva: World Health Organization.
- World Health Organization. (2017e). Analysis of recent scientific information on avian influenza A(H7N9) virus. Retrieved February 10, 2017, from http://www.who.int/influenza/human_animal_interface/avian_influenza/riskassessment_AH7N9_201702/en/
- Wu, D., Zou, S., Bai, T., Li, J., Zhao, X., Yang, L., et al. (2015). Poultry farms as a source of avian influenza A (H7N9) virus reassortment and human infection. Scientific Reports, 5, 7630. doi:10.1038/srep07630 [doi]
- Yi, L., Guan, D., Kang, M., Wu, J., Zeng, X., Lu, J., et al. (2015). Family clusters of avian influenza A(H7N9) virus infection in guangdong province, china. Journal of Clinical Microbiology, 53(1), 22-28. doi:10.1128/JCM.02322-14 [doi]
- Zhou, L., Ren, R., Yang, L., Bao, C., Wu, J., Wang, D., et al. (2017). Sudden increase in human infection with avian influenza A(H7N9) virus in china, September-December 2016. Western Pacific Surveillance and Response., 8(1)
- Zhou, L., Chen, E., Bao, C., Xiang, N., Wu, J., Wu, S., Shi, J., Wang, X., et al (2018). Clusters of human infection and human-to-human transmission of Avian Influenza A(H7N9) virus, 2013-2017. Emerging Infectious Diseases, 24 (2): 397-400. DOI: https://doi.org/10.3201/eid2402.171565