FluWatch report: March 27 to April 2, 2016 (week 13)
Overall Summary
- Influenza activity peaked nationally in week 10; however, lower but sustained activity is being reported throughout the country.
- Most regions of Canada reported sporadic or localized influenza activity.
- Compared to the previous two weeks, there was an increase in the number of provincial and territorial hospitalizations reported in week 13 but the number reported (353) remains below the week 10 peak.
- The number of influenza B detections is increasing, while influenza A detections are decreasing. Overall, laboratory detections of influenza are steadily decreasing.
- Hospitalizations, ICU admissions and deaths among the pediatric population, while declining, remain above expected levels based on the past several influenza seasons.
- For more information on the flu, see our Flu(influenza) web page.
Are you a primary health care practitioner (General Practitioner, Nurse Practitioner or Registered Nurse) interested in becoming a FluWatch sentinel for the 2015-16 influenza season? Contact us at FluWatch@phac-aspc.gc.ca
On this page
- Influenza/ILI Activity (geographic spread)
- Laboratory Confirmed Influenza Detections
- Influenza-like Illness Consultation Rate
- Pharmacy Surveillance
- Influenza Outbreak Surveillance
- Sentinel Pediatric Hospital Influenza Surveillance
- Provincial/Territorial Influenza Hospitalizations and Deaths
- Influenza Strain Characterizations
- Antiviral Resistance
- International Influenza Reports
- FluWatch definitions for the 2015-2016 season

Download the alternative format
(PDF format, 863 KB, 9 pages)
Related Topics
Influenza/Influenza-like Illness Activity (geographic spread)
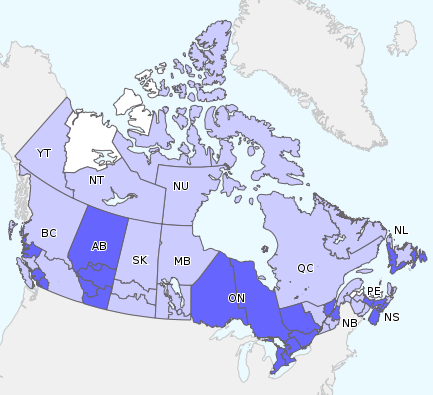
In week 13, influenza activity was reported in all but four regions of Canada. A total of 23 regions reported localized activity levels across the country. Sporadic influenza/ILI activity was reported in 26 regions across Canada.
Figure 1. Map of overall influenza/ILI activity level by province and territory, Canada, week 13
Note: Influenza/ILI activity levels, as represented on this map, are assigned and reported by Provincial and Territorial Ministries of Health, based on laboratory confirmations, sentinel ILI rates and reported outbreaks. Please refer to detailed definitions at the end of the report. Maps from previous weeks, including any retrospective updates, are available in the mapping feature found in the Weekly Influenza Reports.
Figure 1 - Text Description
In week 13, influenza activity was reported in all but four regions of Canada. A total of 23 regions reported localized activity levels across the country. Sporadic influenza/ILI activity was reported in 26 regions across Canada.
Laboratory Confirmed Influenza Detections
In week 13, the percent positive for influenza continued to decrease from the previous week [from 30% in week 12 to 28% in week 13] (Figure 2). Compared to the previous five seasons, the percent positive (28%) reported in week 13 was above the five year average for that week and exceeded the expected levels (range 12.0%-17.9%). With the late start to the 2015-16 influenza season, these above normal levels are not unexpected.
Figure 2. Number of positive influenza tests and percentage of tests positive, by type, subtype and report week, Canada, 2015-16
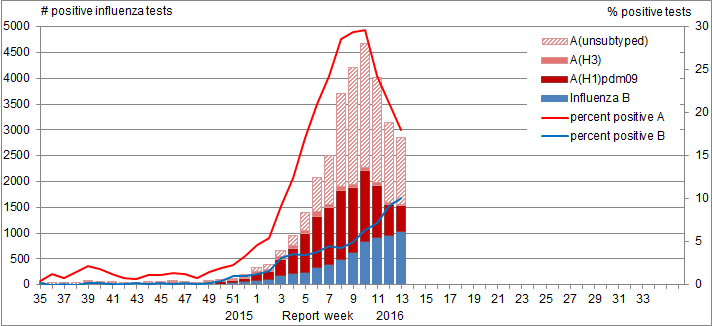
Figure 2 - Text Description
In week 13, the percent positive for influenza continued to decrease from the previous week [from 30% in week 12 to 28% in week 13].
In week 13, there were 2,837 positive influenza tests reported. The number of positive influenza B tests reported is accounting for an increasing proportion of all positive influenza tests reported. This week, influenza B accounted for 36% of all positive tests. To date, 80% of influenza detections have been influenza A and among those subtyped, the vast majority have been influenza A(H1N1) [91% (10155/11124)].
Figure 3. Cumulative numbers of positive influenza specimens by type/subtype and province, Canada, 2015-16
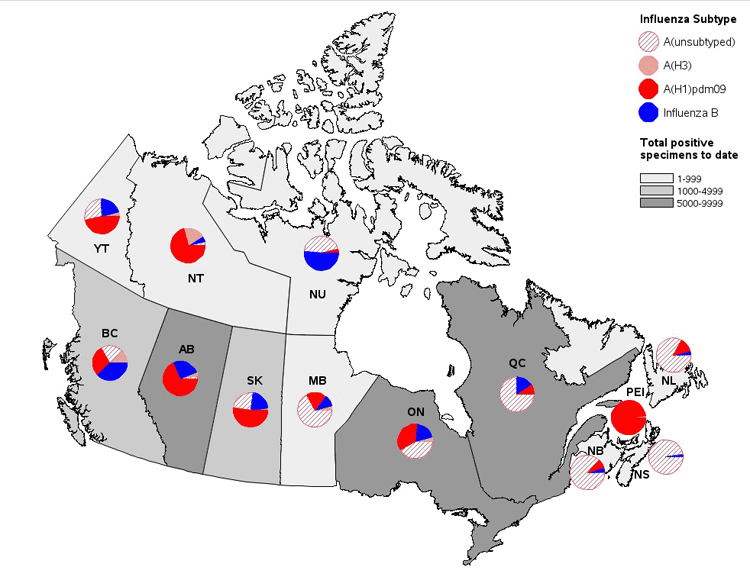
Note: Specimens from NT, YT, and NU are sent to reference laboratories in other provinces. Cumulative data includes updates to previous weeks.
Figure 3 - Text Description
| Reporting provincesTable Figure 3 - Footnote 1 |
Weekly (March 27 to April 2, 2016) | Cumulative (August 30, 2015 to April 2, 2016) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A | B | A & B Total |
|||||||
| A Total |
A (H1)pdm09 |
A (H3) |
A Table Figure 3 - Footnote UnS | B Total |
A Total |
A (H1)pdm09 |
A (H3) |
ATable Figure 3 - Footnote UnS | B Total |
||
| BC | 163 | 82 | <5 | Table Figure 3 - Footnote x | 36 | 1746 | 805 | 335 | 606 | 1066 | 2737 |
| AB | 134 | 105 | <5 | Table Figure 3 - Footnote x | 139 | 3791 | 3494 | 168 | 129 | 1328 | 5119 |
| SK | 49 | 33 | <5 | Table Figure 3 - Footnote x | 49 | 2174 | 1419 | 46 | 709 | 567 | 2741 |
| MB | 76 | 11 | 0 | 65 | 26 | 841 | 170 | 36 | 635 | 103 | 944 |
| ON | 435 | 133 | 6 | 296 | 261 | 6998 | 3033 | 343 | 3622 | 1741 | 8739 |
| QC | 574 | 50 | 0 | 524 | 427 | 7924 | 930 | 5 | 6989 | 1467 | 9391 |
| NB | 158 | 10 | 0 | 148 | 10 | 734 | 64 | <5 | Table Figure 3 - Footnote x | 32 | 766 |
| NS | 41 | 0 | 0 | 41 | <5 | 232 | 0 | <5 | Table Figure 3 - Footnote x | 5 | 237 |
| PE | 0 | 0 | 0 | 0 | 0 | 51 | 50 | <5 | Table Figure 3 - Footnote x | 0 | 51 |
| NL | 64 | 0 | 0 | 64 | <5 | 408 | 58 | <5 | Table Figure 3 - Footnote x | 14 | 422 |
| YT | 6 | <5 | 0 | <5 | <5 | 71 | 42 | <5 | Table Figure 3 - Footnote x | 19 | 90 |
| NT | <5 | <5 | 0 | <5 | 0 | 119 | 89 | 25 | 5 | 6 | 125 |
| NU | <5 | 0 | 0 | <5 | 0 | 13 | <5 | Table Figure 3 - Footnote x | 12 | 14 | 27 |
| Canada | 1706 | 428 | 14 | 1264 | 952 | 25102 | 10155 | 969 | 13978 | 6362 | 31464 |
| Percentage Table Figure 3 - Footnote 2 | 64% | 25% | 1% | 74% | 36% | 80% | 40% | 4% | 56% | 20% | 100% |
|
|||||||||||
To date this season, detailed information on age and type/subtype has been received for 27,158 cases. Children and teenagers (0-19 years of age) accounted for almost one third of all influenza cases (32%). Children (0-19), young adults (20-44) and middle-aged adults (45-64) accounted for approximately an equal proportion of influenza A(H1N1) cases (28%).
| Age groups (years) | Weekly (March 27 to April 2, 2016) | Cumulative (August 30, 2015 to April 2, 2016) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A | B | Influenza A and B | ||||||||
| A Total | A(H1) pdm09 | A(H3) | A UnSTable 1 - Footnote 3 | Total | A Total | A(H1) pdm09 | A(H3) | A UnSTable 1 - Footnote 3 | Total | # | % | |
| <5 | 171 | 30 | <5 | Table 1 - Footnote x | 129 | 4127 | 1650 | 63 | 2414 | 895 | 5023 | 18% |
| 5-19 | 56 | 9 | 0 | 47 | 196 | 2226 | 984 | 92 | 1150 | 1660 | 3887 | 14% |
| 20-44 | 209 | 52 | <5 | Table 1 - Footnote x | 155 | 5387 | 2595 | 146 | 2646 | 1403 | 6790 | 25% |
| 45-64 | 287 | 79 | 0 | 208 | 73 | 5747 | 2549 | 175 | 3023 | 646 | 6393 | 24% |
| 65+ | 262 | 60 | <5 | Table 1 - Footnote x | 118 | 4233 | 1437 | 376 | 2420 | 832 | 5065 | 19% |
| Total | 985 | 230 | 6 | 749 | 671 | 21720 | 9215 | 852 | 11653 | 5436 | 27158 | 100% |
| PercentageTable 1 - Footnote 2 | 59% | 23% | 1% | 76% | 41% | 80% | 42% | 4% | 54% | 20% | ||
|
||||||||||||
For data on other respiratory virus detections see the Respiratory Virus Detections in Canada Report on the Public Health Agency of Canada website.
Influenza-like Illness Consultation Rate
The national ILI consultation rate increased slightly from the previous week from 43.6 per 1,000 patient visits in week 12, to 45.2 per 1,000 patient visits in week 13. The highest ILI consultation rate was found in the 0-4 years age group (69.7 per 1,000) and the lowest was found in the ≥65 years age group (35.2 per 1,000) (Figure 4).
Figure 4. Influenza-like-illness (ILI) consultation rates by age group and week, Canada, 2015-16

Delays in the reporting of data may cause data to change retrospectively. In BC, AB, and SK, data are compiled by a provincial sentinel surveillance program for reporting to FluWatch. Not all sentinel physicians report every week.
Figure 4 - Text Description
Influenza-like illness consultation rate by age-group in week 13 for the 2015-16 season:
Age 0-4: 69.7; Age 5-19: 54.7; Age 20-64: 42.3; Age 65+: 35.2
Pharmacy Surveillance
During week 13, the proportion of prescriptions for antivirals increased slightly to 78.6 antiviral prescriptions per 100,000 total prescriptions, which is slightly higher than the five year historical average for week 13. The proportion of prescriptions for antivirals remains highest among children. In week 13, the proportion reported among children was 162.4 per 100,000 total prescriptions.
Figure 5. Proportion of prescription sales for influenza antivirals by age group and week, Canada, 2015-16
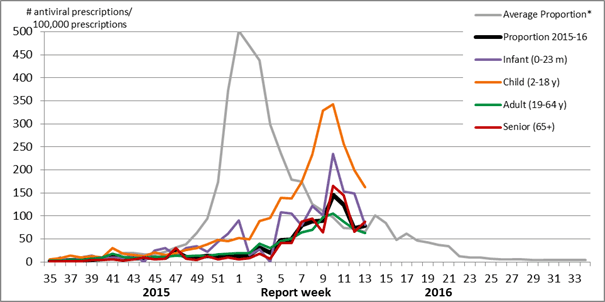
Note: Pharmacy sales data are provided to the Public Health Agency of Canada by Rx Canada Inc. and sourced from major retail drug chains representing over 3,000 stores nationwide (excluding Nunavut) in 85% of Health Regions. Data provided include the number of new antiviral prescriptions (for Tamiflu[oseltamivir] and Relenza [zanamivir]) and the total number of new prescriptions dispensed by Province/Territory and age group.
* The average weekly proportion includes data from April 2011 to March 2015.
Figure 5 - Text Description
Proportion of antiviral prescriptions per 100,000 total prescriptions
Average National Rate (Yrs 10-11 to 14-15): 65.4.; Rate wk 13: 78.6
Proportion of antiviral prescriptions by age-group in week 13 for the 2015-16 season:
Infant: 80.7; child: 162.4; adult: 62.3; senior: 88.0
Influenza Outbreak Surveillance
In week 13, 21 new laboratory confirmed influenza outbreaks were reported: 12 in long-term care facilities (LTCF), five in hospitals and four in institutions or community settings. Of the outbreaks with known strains or subtypes, three outbreaks were due to influenza B, two outbreaks were due to influenza A(H1N1), one outbreak was due to influenza A(H3N2) and three were due to influenza A(UnS). Additionally, one ILI outbreak was reported in a school.
To date this season, 354 outbreaks have been reported. At week 13 in the 2014-15 season, 1,527 outbreaks were reported and in the 2013-14 season,184 outbreaks were reported.
Figure 6. Overall number of new laboratory-confirmed influenza outbreaksFigure 5 - Footnote 1 by report week, Canada, 2015-2016
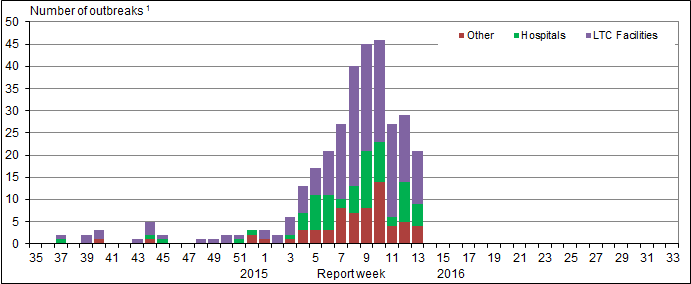
Figure 6 - Text Description
| Report week | Hospitals | Long Term Care Facilities | Other |
|---|---|---|---|
| 35 | 0 | 0 | 0 |
| 36 | 0 | 0 | 0 |
| 37 | 1 | 1 | 0 |
| 38 | 0 | 0 | 0 |
| 39 | 0 | 2 | 0 |
| 40 | 0 | 2 | 1 |
| 41 | 0 | 0 | 0 |
| 42 | 0 | 0 | 0 |
| 43 | 0 | 1 | 0 |
| 44 | 1 | 3 | 1 |
| 45 | 1 | 1 | 0 |
| 46 | 0 | 0 | 0 |
| 47 | 0 | 0 | 0 |
| 48 | 0 | 1 | 0 |
| 49 | 0 | 1 | 0 |
| 50 | 0 | 2 | 0 |
| 51 | 1 | 1 | 0 |
| 52 | 1 | 0 | 2 |
| 1 | 0 | 2 | 1 |
| 2 | 0 | 2 | 0 |
| 3 | 1 | 4 | 1 |
| 4 | 4 | 6 | 3 |
| 5 | 8 | 6 | 3 |
| 6 | 8 | 10 | 3 |
| 7 | 2 | 17 | 8 |
| 8 | 6 | 27 | 7 |
| 9 | 13 | 24 | 8 |
| 10 | 9 | 23 | 14 |
| 11 | 2 | 21 | 4 |
| 12 | 9 | 15 | 5 |
| 13 | 5 | 12 | 4 |
Sentinel Pediatric Hospital Influenza Surveillance
Paediatric Influenza Hospitalizations and Deaths
Hospitalizations reported by the the Immunization Monitoring Program Active (IMPACT) network continue to decrease (Figure 7). In week 13, 61 hospitalizations were reported. The largest proportion of hospitalizations were in children aged 0-2 years, accounting for 38% of the hospitalizations. The majority of hospitalizations in week 13 were due to influenza A (57%); however, the proportion of weekly hospitalizations due to influenza B has been increasing steadily since week 08.
To date this season, 1,075 laboratory-confirmed influenza-associated pediatric (≤16 years of age) hospitalizations have been reported by the IMPACT network: 822 hospitalized cases (76%) were due to influenza A and 253 cases (24%) were due to influenza B. The greatest proportion of hospitalized cases were in children aged 0-2 years (41 %). To date, 180 intensive care unit (ICU) admissions have been reported. Children aged 2 to 4 and 5 to 9 years each accounted for 27% of ICU admissions. A total of 113 ICU cases (62%) reported at least one underlying condition or comorbidity. Eight influenza-associated deaths have been reported.
| Age Groups | Cumulative (30 Aug. 2015 to 2 April 2016) | |||||
|---|---|---|---|---|---|---|
| Influenza A | Influenza B | Influenza A and B (#(%)) | ||||
| A Total | A(H1) pdm09 | A(H3) | A (UnS) | B Total | ||
| 0-5m | 110 | 32 | 5 | 73 | 19 | 129 (12%) |
| 6-23m | 251 | 72 | 7 | 172 | 57 | 308 (29%) |
| 2-4y | 236 | 76 | <5 | Table 2 - Footnote x | 61 | 297 (28%) |
| 5-9y | 172 | 46 | <5 | Table 2 - Footnote x | 81 | 253 (24%) |
| 10-16y | 53 | 18 | <5 | Table 2 - Footnote x | 35 | 88 (8%) |
| Total | 822 | 244 | 20 | 558 | 253 | 1075 (100%) |
|
||||||
Figure 7. Number of cases of influenza reported by sentinel hospital networks, by week, Canada, 2015-16, paediatric and adult hospitalizations (≤16 years of age, IMPACT; ≥16 years of age, CIRN-SOS)
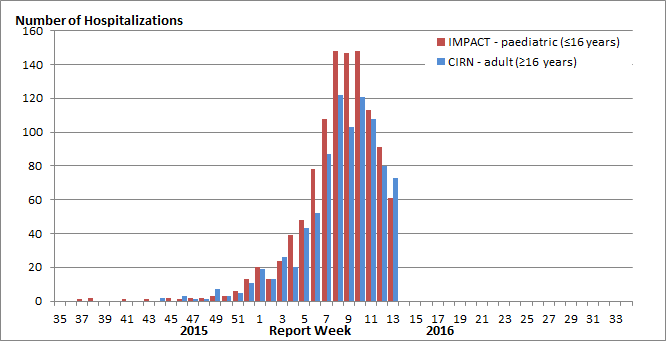
Not included in Table 2 and Figure 7 are two IMPACT cases that were due to co-infections of influenza A and B.
Figure 7 - Text Description
| Report week | IMPACT | CIRN-SOS |
|---|---|---|
| 35 | 0 | 0 |
| 36 | 0 | 0 |
| 37 | 1 | 0 |
| 38 | 2 | 0 |
| 39 | 0 | 0 |
| 40 | 0 | 0 |
| 41 | 1 | 0 |
| 42 | 0 | 0 |
| 43 | 1 | 0 |
| 44 | 0 | 2 |
| 45 | 2 | 0 |
| 46 | 1 | 3 |
| 47 | 2 | 1 |
| 48 | 2 | 1 |
| 49 | 3 | 7 |
| 50 | 3 | 3 |
| 51 | 6 | 5 |
| 52 | 13 | 11 |
| 1 | 20 | 19 |
| 2 | 13 | 13 |
| 3 | 24 | 26 |
| 4 | 39 | 20 |
| 5 | 48 | 43 |
| 6 | 78 | 52 |
| 7 | 108 | 87 |
| 8 | 148 | 122 |
| 9 | 147 | 103 |
| 10 | 148 | 121 |
| 11 | 113 | 108 |
| 12 | 91 | 80 |
| 13 | 61 | 73 |
Adult Influenza Hospitalizations and Deaths
In week 13, 73 hospitalizations were reported by the Canadian Immunization Research Network Serious Outcome Surveillance (CIRN-SOS) (Figure 7). The largest proportion of hospitalizations was in adults 65+ years of age (51%) and due to influenza A (73%).
To date this season, 898 laboratory-confirmed influenza-associated adult (≥16 years of age) hospitalizations have been reported by CIRN-SOS (Table 3). The majority of hospitalized cases were due to influenza A (85%) and the largest reported proportion was among adults ≥65 years of age (49%). One hundred and thirty intensive care unit (ICU) admissions have been reported. A total of 69 ICU cases reported to have at least one underlying condition or comorbidity. A total of 32 deaths have been reported this season with the majority of deaths reported in adults ≥65 years of age (63%).
| Age groups (years) | Cumulative (1 Nov. 2015 to 2 April 2016) | |||||
|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A and B | ||||
| A Total | A(H1) pdm09 | A(H3) | A(UnS) | Total | # (%) | |
| 16-20 | Table 3 - Footnote x | <5 | 0 | <5 | <5 | 6 (1%) |
| 20-44 | 118 | 35 | <5 | Table 3 - Footnote x | 27 | 145 (16%) |
| 45-64 | 274 | 78 | <5 | Table 3 - Footnote x | 29 | 303 (34%) |
| 65+ | 363 | 81 | 21 | 261 | 76 | 439 (49%) |
| Unknown | <5 | 0 | 0 | <5 | <5 | 5 (1%) |
| Total | 764 | 200 | 25 | 539 | 134 | 898 |
| % | 85% | 26% | 3% | 71% | 15% | 100% |
|
||||||
Figure 8. Percentage of hospitalizations, ICU admissions and deaths with influenza reported by age-group (≥16 year of age), Canada 2015-16
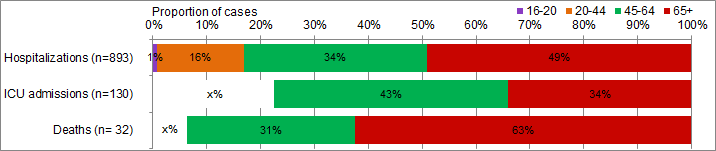
Note: The number of hospitalizations reported through CIRN-SOS and IMPACT represents a subset of all influenza-associated adult and paediatric hospitalizations in Canada. Delays in the reporting of data may cause data to change retrospectively.
x - Suppressed to prevent residual disclosure.
Figure 8 - Text Description
| Age-group (years) | Hospitalizations (n=893) | ICU admissions (n=130) | Deaths (n=32) |
|---|---|---|---|
| 16-20 | 0.7% | Table Figure 8 - Footnote x% | Table Figure 8 - Footnote x% |
| 20-44 | 16.2% | Table Figure 8 - Footnote x% | Table Figure 8 - Footnote x% |
| 45-64 | 33.9% | 43.1% | 31.3% |
| 65+ | 49.2% | 33.8% | 62.5% |
|
|||
Provincial/Territorial Influenza Hospitalizations and Deaths
Since the start of the 2015-16 season, 3,990 laboratory-confirmed influenza-associated hospitalizations have been reported. A total of 3,401 hospitalizations (85%) were due to influenza A and 589 (15%) were due to influenza B. Among cases for which the subtype of influenza A was reported, 93% (1601/1715) were influenza A(H1N1). The largest proportion (30%) of hospitalized cases were ≥65 years of age, followed closely by adults 45-64 years of age (27%). Three hundred and ninety-four ICU admissions have been reported of which 225 (57%) were due to influenza A(H1N1) and 183 (46%) were in the 45-64 age group. A total of 161 deaths have been reported; all but 14 were associated with influenza A. Adults ≥65 years of age represent 49% of reported deaths.
Figure 9. Percentage of hospitalizations, ICU admissions and deaths with influenza reported by age-group, Canada 2015-16

Figure 9 - Text Description
| Age-group (years) | Hospitalizations (n=3990) | ICU admissions (n=394) | Deaths (n=161) |
|---|---|---|---|
| 0-4 | 19.7% | 9.1% | 3.1% |
| 5-19 | 9.1% | 7.1% | 3.1% |
| 20-44 | 14.0% | 17.8% | 8.1% |
| 45-64 | 27.1% | 46.4% | 36.6% |
| 65+ | 30.1% | 19.5% | 49.1% |
See additional data on Reported Influenza Hospitalizations and Deaths in Canada: 2011-12 to 2015-16 on the Public Health Agency of Canada website.
Influenza Strain Characterizations
During the 2015-16 influenza season, the National Microbiology Laboratory (NML) has characterized 1442 influenza viruses [159 A(H3N2), 857 A(H1N1) and 426 influenza B].
Influenza A (H3N2): When tested by hemagglutination inhibition (HI) assays, 40 H3N2 viruses were antigenically characterized as A/Switzerland/9715293/2013-like using antiserum raised against cell-propagated A/Switzerland/9715293/2013.
Sequence analysis was done on 119 H3N2 viruses. All viruses belonged to a genetic group for which most viruses were antigenically related to A/Switzerland/9715293/2013. A/Switzerland/9715293/2013 is the A(H3N2) component of the 2015-16 Northern Hemisphere's vaccine.
Influenza A (H1N1): A total of 857 H1N1 viruses characterized were antigenically similar to A/California/7/2009, the A(H1N1) component of the 2015-16 influenza vaccine.
Influenza B: A total of 112 influenza B viruses characterized were antigenically similar to the vaccine strain B/Phuket/3073/2013. A total of 314 influenza B viruses were characterized as B/Brisbane/60/2008-like, one of the influenza B components of the 2015-16 Northern Hemisphere quadrivalent influenza vaccine.
The recommended components for the 2015-2016 northern hemisphere trivalent influenza vaccine include: an A/California/7/2009(H1N1)pdm09-like virus, an A/Switzerland/9715293/2013(H3N2)-like virus, and a B/Phuket/3073/2013 -like virus (Yamagata lineage). For quadrivalent vaccines, the addition of a B/Brisbane/60/2008-like virus (Victoria lineage) is recommended.
The NML receives a proportion of the influenza positive specimens from provincial laboratories for strain characterization and antiviral resistance testing. Characterization data reflect the results of haemagglutination inhibition testing compared to the reference influenza strains recommended by WHO.
Antiviral Resistance
During the 2015-16 season, the National Microbiology Laboratory (NML) has tested 976 influenza viruses for resistance to oseltamivir, 979 for resistance to zanamivir and 1,027 influenza viruses for resistance to amantadine. All but seven tested viruses were sensitive to oseltamivir. The seven H1N1 viruses resistant to oseltamivir had a H275Y mutation. All viruses tested for resistance were sensitive to zanamivir. All but one influenza A viruses were resistant to amantadine (Table 4).
| Virus type and subtype | Oseltamivir | Zanamivir | Amantadine | |||
|---|---|---|---|---|---|---|
| # tested | # resistant (%) | # tested | # resistant (%) | # tested | # resistant (%) | |
| A (H3N2) | 139 | 0 | 139 | 0 | 173 | 172 (99.4%) |
| A (H1N1) | 600 | 7 | 603 | 0 | 854 | 854 (100%) |
| B | 237 | 0 | 237 | 0 | NATable 4 - Footnote * | NATable 4 - Footnote * |
| Total | 976 | 7 | 979 | 0 | 1027 | 1026 |
|
||||||
International Influenza Reports
- World Health Organization influenza update
- World Health Organization FluNet
- WHO Influenza at the human-animal interface
- Centers for Disease Control and Prevention seasonal influenza report
- European Centre for Disease Prevention and Control - epidemiological data
- South Africa Influenza surveillance report
- New Zealand Public Health Surveillance
- Australia Influenza Report
- Pan-American Health Organization Influenza Situation Report
FluWatch definitions for the 2015-2016 season
Abbreviations: Newfoundland/Labrador (NL), Prince Edward Island (PE), New Brunswick (NB), Nova Scotia (NS), Quebec (QC), Ontario (ON), Manitoba (MB), Saskatchewan (SK), Alberta (AB), British Columbia (BC), Yukon (YT), Northwest Territories (NT), Nunavut (NU).
Influenza-like-illness (ILI): Acute onset of respiratory illness with fever and cough and with one or more of the following - sore throat, arthralgia, myalgia, or prostration which is likely due to influenza. In children under 5, gastrointestinal symptoms may also be present. In patients under 5 or 65 and older, fever may not be prominent.
ILI/Influenza outbreaks
- Schools:
-
Greater than 10% absenteeism (or absenteeism that is higher (e.g. >5-10%) than expected level as determined by school or public health authority) which is likely due to ILI.
Note: it is recommended that ILI school outbreaks be laboratory confirmed at the beginning of influenza season as it may be the first indication of community transmission in an area. - Hospitals and residential institutions:
- two or more cases of ILI within a seven-day period, including at least one laboratory confirmed case. Institutional outbreaks should be reported within 24 hours of identification. Residential institutions include but not limited to long-term care facilities ( LTCF) and prisons.
- Workplace:
- Greater than 10% absenteeism on any day which is most likely due to ILI.
- Other settings:
- two or more cases of ILI within a seven-day period, including at least one laboratory confirmed case; i.e. closed communities.
Note that reporting of outbreaks of influenza/ILI from different types of facilities differs between jurisdictions.
Influenza/ILI activity level
1 = No activity: no laboratory-confirmed influenza detections in the reporting week, however, sporadically occurring ILI may be reported
3 = Localized:
- evidence of increased ILIFootnote * and
- lab confirmed influenza detection(s) together with
- outbreaks in schools, hospitals, residential institutions and/or other types of facilities occurring in less than 50% of the influenza surveillance regionFootnote †
4 = Widespread:
- evidence of increased ILIFootnote * and
- lab confirmed influenza detection(s) together with
- outbreaks in schools, hospitals, residential institutions and/or other types of facilities occurring in greater than or equal to 50% of the influenza surveillance regionFootnote †
Note: ILI data may be reported through sentinel physicians, emergency room visits or health line telephone calls.