FluWatch report: May 5 to May 11, 2019 (Week 19)
Download the alternative format
(PDF format, 1.17 MB, 10 pages)
Organization: Public Health Agency of Canada
Date published: 2019-05-17
Related Topics
Overall Summary
- Influenza activity continues to decline overall.
- Influenza A(H3N2) was the predominant subtype this week (83% of subtyped influenza A detections); however, influenza A(H1N1)pdm09 remains the predominant subtype to date this season (71% of subtyped influenza A detections).
- Detections of influenza A continue to be greater than those of influenza B. There is currently very little influenza B circulation compared to previous seasons.
On this page
- Influenza/ILI Activity (geographic spread)
- Laboratory Confirmed Influenza Detections
- Syndromic/Influenza-like Illness Surveillance
- Participatory Syndromic Surveillance
- Influenza Outbreak Surveillance
- Severe Outcomes Influenza Surveillance
- Influenza Strain Characterizations
- Antiviral Resistance
- Vaccine Monitoring
- Provincial and International Influenza Reports
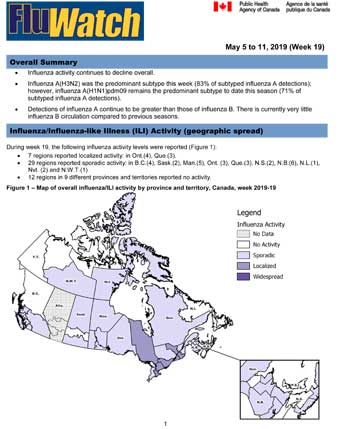
Influenza/Influenza-like Illness Activity (geographic spread)
During week 19, the following influenza activity levels were reported (Figure 1):
- 7 regions reported localized activity: in Ont.(4), Que.(3).
- 29 regions reported sporadic activity: in B.C.(4), Sask.(2), Man.(5), Ont. (3), Que.(3), N.S.(2), N.B.(6), N.L.(1), Nvt. (2) and N.W.T.(1)
- 12 regions in 9 different provinces and territories reported no activity.
Figure 1 – Map of overall influenza/ILI activity by province and territory, Canada, week 2019-19
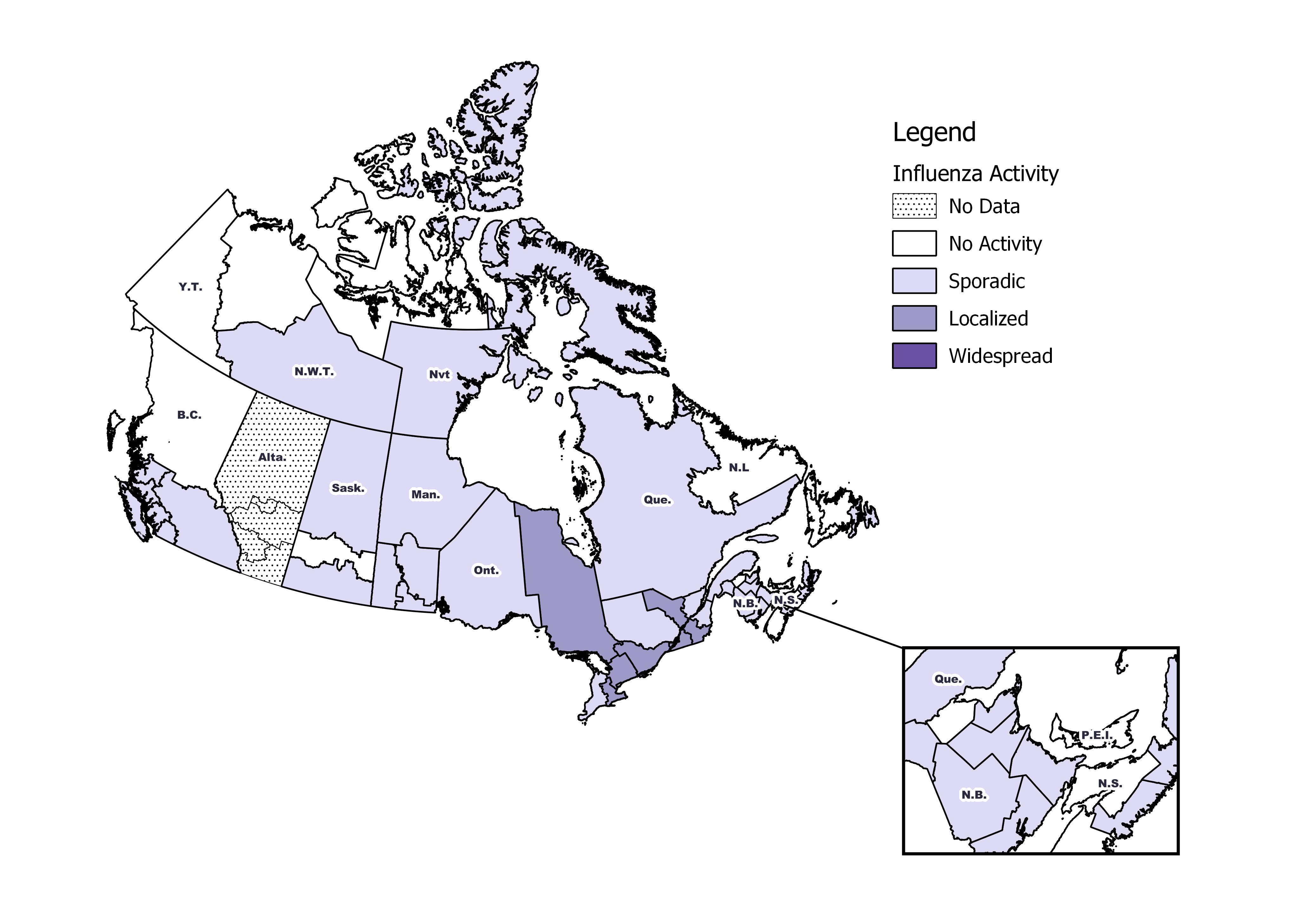
Figure 1 - Text equivalent
| Province | Influenza Surveillance Region | Activity Level |
|---|---|---|
| N.L. | Eastern | Sporadic |
| N.L. | Labrador-Grenfell | No Activity |
| N.L. | Central | No Activity |
| N.L. | Western | No Activity |
| P.E.I. | Prince Edward Island | No Activity |
| N.S. | Zone 1 - Western | No Activity |
| N.S. | Zone 2 - Northern | No Activity |
| N.S. | Zone 3 - Eastern | Sporadic |
| N.S. | Zone 4 - Central | Sporadic |
| N.B. | Region 1 | Sporadic |
| N.B. | Region 2 | Sporadic |
| N.B. | Region 3 | Sporadic |
| N.B. | Region 4 | Sporadic |
| N.B. | Region 5 | No Activity |
| N.B. | Region 6 | Sporadic |
| N.B. | Region 7 | Sporadic |
| Que. | Nord-est | Sporadic |
| Que. | Québec et Chaudieres-Appalaches | Sporadic |
| Que. | Centre-du-Québec | Localized |
| Que. | Montréal et Laval | Localized |
| Que. | Ouest-du-Québec | Sporadic |
| Que. | Montérégie | Localized |
| Ont. | Central East | Localized |
| Ont. | Central West | Localized |
| Ont. | Eastern | Localized |
| Ont. | North East | Localized |
| Ont. | North West | Sporadic |
| Ont. | South West | Sporadic |
| Ont. | Toronto | Sporadic |
| Man. | Northern Regional | Sporadic |
| Man. | Prairie Mountain | Sporadic |
| Man. | Interlake-Eastern | Sporadic |
| Man. | Winnipeg | Sporadic |
| Man. | Southern Health | Sporadic |
| Sask. | North | Sporadic |
| Sask. | Central | No Activity |
| Sask. | South | Sporadic |
| Alta. | North Zone | No Data |
| Alta. | Edmonton | No Data |
| Alta. | Central Zone | No Data |
| Alta. | Calgary | No Data |
| Alta. | South Zone | No Data |
| B.C. | Interior | Sporadic |
| B.C. | Fraser | Sporadic |
| B.C. | Vancouver Coastal | Sporadic |
| B.C. | Vancouver Island | Sporadic |
| B.C. | Northern | No Activity |
| Y.T. | Yukon | No Activity |
| N.W.T. | North | No Activity |
| N.W.T. | South | Sporadic |
| Nvt. | Qikiqtaaluk | Sporadic |
| Nvt. | Kivalliq | Sporadic |
| Nvt. | Kitimeot | No Activity |
Laboratory-Confirmed Influenza Detections
In week 19, the following results were reported from sentinel laboratories across (Figures 2 and 3):
- The percentage of tests positive for influenza increased slightly from 11% to 12% in week 19.
- A total of 592 laboratory detections of influenza were reported, of which 76% were influenza A.
- Influenza A(H3N2) accounted for 83% of subtyped influenza A detections.
To date this season, 47,143 laboratory-confirmed influenza detections have been reported:
- 96% have been influenza A.
- Among the 15,938 influenza A viruses subtyped, 71% have been A(H1N1)pdm09.
- Influenza B often circulates later in the season in Canada (Feb-Apr). Fewer influenza B detections have been reported this season compared to recent seasons at this time of year. The percentage of tests positive for influenza B in week 19 was similar to the previous week.
To date this season, detailed information on age and type/subtype has been received for 37,844 laboratory-confirmed influenza cases (Table 1):
- 83% of all influenza A(H1N1)pdm09 detections have been reported in individuals younger than 65 years of age.
- 58% of all influenza A(H3N2) detections have been reported in adults 65 years of age and older.
For more detailed weekly and cumulative influenza data, see the text descriptions for Figures 2 and 3 or the Respiratory Virus Detections in Canada Report.
Figure 2 – Number of positive influenza tests and percentage of tests positive, by type, subtype and report week, Canada, weeks 2018-35 to 2019-19
The shaded area indicates weeks where the positivity rate was at least 5% and a minimum of 15 positive tests were observed, signalling the period of seasonal influenza activity.
Data for week 14 excludes subtyping results from one jurisdiction due to batch reporting of subtype information. The results for week 14 should be interpreted with caution.
Figure 2 - Text equivalent
| Surveillance Week | A(Unsubtyped) | A(H3N2) | A(H1N1)pdm09 | Influenza B |
|---|---|---|---|---|
| 35 | 3 | 2 | 7 | 0 |
| 36 | 4 | 7 | 4 | 0 |
| 37 | 3 | 2 | 3 | 1 |
| 38 | 6 | 3 | 2 | 3 |
| 39 | 11 | 5 | 1 | 3 |
| 40 | 16 | 7 | 29 | 3 |
| 41 | 27 | 6 | 21 | 3 |
| 42 | 40 | 19 | 55 | 2 |
| 43 | 83 | 23 | 128 | 4 |
| 44 | 169 | 13 | 214 | 6 |
| 45 | 244 | 18 | 295 | 15 |
| 46 | 346 | 10 | 404 | 9 |
| 47 | 449 | 17 | 507 | 8 |
| 48 | 679 | 29 | 632 | 10 |
| 49 | 851 | 35 | 785 | 16 |
| 50 | 1368 | 35 | 828 | 14 |
| 51 | 1890 | 54 | 953 | 21 |
| 52 | 2292 | 55 | 903 | 26 |
| 1 | 2387 | 58 | 893 | 31 |
| 2 | 1985 | 93 | 1061 | 43 |
| 3 | 1613 | 101 | 585 | 40 |
| 4 | 1483 | 93 | 532 | 48 |
| 5 | 1449 | 112 | 433 | 43 |
| 6 | 1234 | 118 | 315 | 42 |
| 7 | 1116 | 128 | 244 | 33 |
| 8 | 1066 | 236 | 216 | 40 |
| 9 | 1172 | 285 | 213 | 64 |
| 10 | 1085 | 335 | 268 | 71 |
| 11 | 990 | 357 | 197 | 90 |
| 12 | 959 | 380 | 155 | 131 |
| 13 | 939 | 387 | 128 | 167 |
| 14 | 1091 | 292 | 41 | 169 |
| 15 | 756 | 514 | 121 | 157 |
| 16 | 614 | 277 | 42 | 175 |
| 17 | 448 | 244 | 55 | 194 |
| 18 | 275 | 160 | 29 | 153 |
| 19 | 282 | 150 | 31 | 146 |
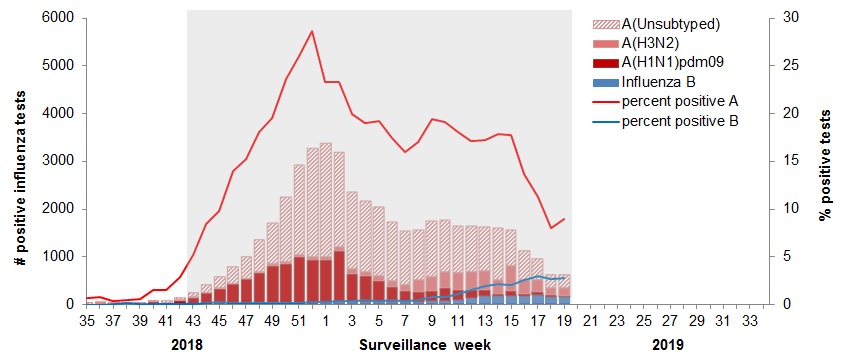
Figure 3 – Cumulative numbers of positive influenza specimens by type/subtype and province/territory, Canada, weeks 2018-35 to 2019-19
Figure 3 - Text equivalent
| Reporting provincesTable Figure 3 - Footnote 1 | Week (May 5 to May 11, 2019) | Cumulative (August 26, 2018 to May 11, 2019) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A | B | A & B Total |
|||||||
| A Total |
A (H1N1)pdm09 |
A (H3N2) |
A(UnS)Table Figure 3 - Footnote 3 | B Total |
A Total |
A (H1N1)pdm09 |
A (H3N2) |
A(UnS)Table Figure 3 - Footnote 3 | B Total |
||
| BC | 58 | 3 | 30 | 25 | 6 | 6990 | 3153 | 1529 | 1608 | 229 | 6519 |
| AB | 95 | 15 | 62 | 18 | 21 | 6938 | 3633 | 1136 | 2169 | 295 | 6519 |
| SK | 8 | 2 | 5 | 1 | 0 | 2386 | 1387 | 136 | 863 | 94 | 2480 |
| MB | 11 | 0 | 5 | 6 | 6 | 1152 | 322 | 100 | 730 | 52 | 1202 |
| ON | 69 | 9 | 44 | 16 | 5 | 6236 | 2078 | 1642 | 2516 | 234 | 6470 |
| QC | 171 | 0 | 0 | 171 | 79 | 16947 | 0 | 0 | 16947 | 730 | 17677 |
| NB | 26 | 1 | 0 | 25 | 23 | 2836 | 231 | 34 | 2571 | 300 | 3136 |
| NS | 2 | 0 | 0 | 2 | 1 | 784 | 0 | 0 | 784 | 18 | 802 |
| PEI | 0 | 0 | 0 | 0 | 0 | 278 | 221 | 56 | 1 | 1 | 279 |
| NL | 9 | 0 | 0 | 9 | 0 | 1025 | 1 | 0 | 1024 | 4 | 1029 |
| YT | 1 | 0 | 1 | 0 | 0 | 115 | 65 | 15 | 35 | 0 | 115 |
| N.W.T | 1 | 0 | 1 | 0 | 0 | 179 | 175 | 4 | 0 | 2 | 181 |
| NU | 0 | 0 | 0 | 0 | 0 | 20 | 20 | 0 | 0 | 0 | 20 |
| Canada | 451 | 30 | 148 | 273 | 141 | 45186 | 11286 | 4652 | 29248 | 1957 | 47143 |
| PercentageTable Figure 3 - Footnote 2 | 76% | 7% | 33% | 61% | 24% | 96% | 25% | 10% | 65% | 4% | 100% |
Discrepancies in values in Figures 2 and 3 may be attributable to differing data sources. Cumulative data includes updates to previous weeks. |
|||||||||||
| Age groups (years) | Cumulative (August 26, 2018 to May 11, 2019) | ||||||
|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A and B | |||||
| A Total | A(H1N1) pdm09 | A(H3N2) | A (UnS)Table 1 – Note 1 | Total | # | % | |
| 0-4 | 6707 | 1689 | 248 | 4770 | 286 | 6993 | 18% |
| 5-19 | 5127 | 1389 | 471 | 3267 | 482 | 5609 | 15% |
| 20-44 | 6847 | 2012 | 596 | 4239 | 308 | 7155 | 19% |
| 45-64 | 6964 | 1963 | 632 | 4369 | 115 | 7079 | 19% |
| 65+ | 10761 | 1480 | 2667 | 6614 | 247 | 11008 | 29% |
| Total | 36406 | 8533 | 4614 | 23259 | 1438 | 37844 | 100% |
|
|||||||
Syndromic / Influenza-like Illness Surveillance
Healthcare Professionals Sentinel Syndromic Surveillance
In week 19, 0.8% of visits to healthcare professionals were due to influenza-like illness (ILI) (Figure 4).
Figure 4 – Percentage of visits for ILI reported by sentinels by report week, Canada, weeks 2018-35 to 2019-19
Number of Sentinels Reporting in Week 19: 72
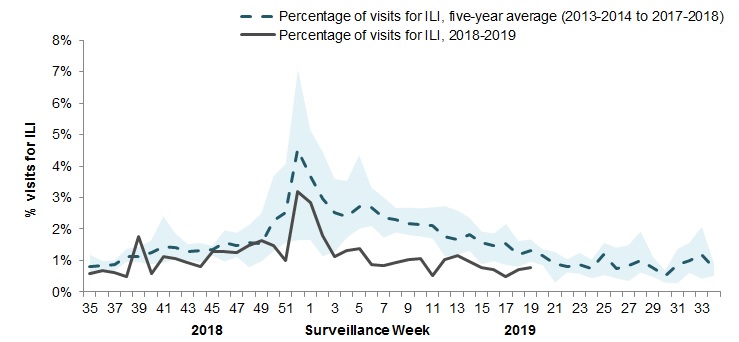
The shaded area represents the maximum and minimum percentage of visits for ILI reported by week from seasons 2013-2014 to 2017-2018
Figure 4 - Text equivalent
| Surveillance Week | 2018-19 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0.6% | 0.8% | 0.5% | 1.2% |
| 36 | 0.7% | 0.8% | 0.7% | 1.0% |
| 37 | 0.6% | 0.9% | 0.7% | 1.0% |
| 38 | 0.5% | 1.1% | 1.0% | 1.4% |
| 39 | 1.8% | 1.1% | 0.9% | 1.4% |
| 40 | 0.6% | 1.3% | 0.9% | 1.6% |
| 41 | 1.1% | 1.4% | 0.9% | 2.4% |
| 42 | 1.0% | 1.4% | 1.0% | 1.9% |
| 43 | 0.9% | 1.3% | 1.0% | 1.5% |
| 44 | 0.8% | 1.3% | 0.9% | 1.6% |
| 45 | 1.3% | 1.3% | 1.2% | 1.5% |
| 46 | 1.3% | 1.6% | 1.0% | 2.0% |
| 47 | 1.3% | 1.5% | 1.1% | 1.9% |
| 48 | 1.5% | 1.6% | 0.8% | 2.1% |
| 49 | 1.6% | 1.5% | 1.0% | 2.5% |
| 50 | 1.5% | 2.3% | 1.3% | 3.7% |
| 51 | 1.0% | 2.5% | 1.6% | 4.1% |
| 52 | 3.2% | 4.5% | 1.7% | 7.1% |
| 1 | 2.8% | 3.7% | 1.7% | 5.2% |
| 2 | 1.8% | 3.0% | 1.1% | 4.5% |
| 3 | 1.1% | 2.5% | 1.3% | 3.6% |
| 4 | 1.3% | 2.4% | 1.7% | 3.5% |
| 5 | 1.4% | 2.7% | 2.0% | 4.4% |
| 6 | 0.9% | 2.7% | 2.1% | 3.3% |
| 7 | 0.8% | 2.4% | 1.7% | 3.0% |
| 8 | 0.9% | 2.3% | 1.9% | 2.7% |
| 9 | 1.0% | 2.2% | 1.8% | 2.7% |
| 10 | 1.1% | 2.1% | 1.8% | 2.7% |
| 11 | 0.5% | 2.1% | 1.7% | 2.7% |
| 12 | 1.0% | 1.8% | 1.1% | 2.7% |
| 13 | 1.1% | 1.7% | 1.2% | 2.6% |
| 14 | 1.0% | 1.8% | 1.3% | 2.4% |
| 15 | 0.8% | 1.6% | 0.9% | 1.9% |
| 16 | 0.7% | 1.5% | 1.0% | 1.9% |
| 17 | 0.5% | 1.5% | 0.9% | 2.2% |
| 18 | 0.7% | 1.2% | 0.8% | 1.6% |
| 19 | 0.8% | 1.3% | 1.0% | 1.7% |
Participatory Syndromic Surveillance
FluWatchers surveillance has ended for the 2018-19 influenza season. On average 2,097 participants reported to FluWatchers each week, resulting in 64,672 questionnaires completed this season. The proportion of participants reporting fever and cough peaked in week 51 at 3.9% (Figure 5). Approximately 63% of FluWatchers participants reported being vaccinated for influenza in the 2018-19 season.
Figure 5 - Percentage of participants reporting cough and fever, Canada, weeks 2018-40 to 2019-18
Number of Participants Reporting in Week 18: 1,951
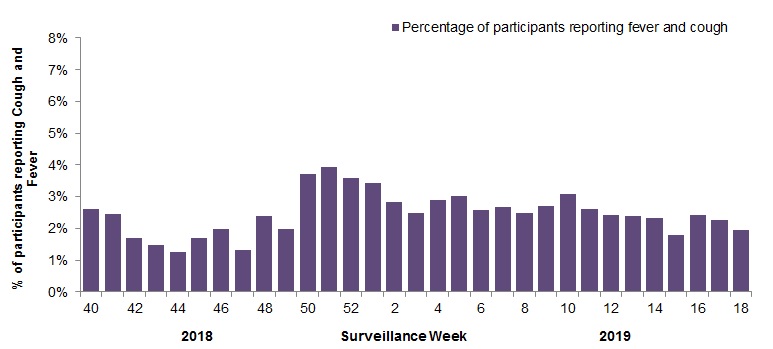
Figure 5 - Text equivalent
| Surveillance Week | % cough and fever |
|---|---|
| 40 | 2.6% |
| 41 | 2.5% |
| 42 | 1.7% |
| 43 | 1.5% |
| 44 | 1.3% |
| 45 | 1.7% |
| 46 | 2.0% |
| 47 | 1.3% |
| 48 | 2.4% |
| 49 | 2.0% |
| 50 | 3.7% |
| 51 | 3.9% |
| 52 | 3.6% |
| 1 | 3.4% |
| 2 | 2.8% |
| 3 | 2.5% |
| 4 | 2.9% |
| 5 | 3.0% |
| 6 | 2.6% |
| 7 | 2.7% |
| 8 | 2.5% |
| 9 | 2.7% |
| 10 | 3.1% |
| 11 | 2.6% |
| 12 | 2.4% |
| 13 | 2.4% |
| 14 | 2.3% |
| 15 | 1.8% |
| 16 | 2.4% |
| 17 | 2.3% |
| 18 | 1.9% |
Influenza Outbreak Surveillance
In week 19, 11 new laboratory-confirmed influenza outbreaks were reported: long-term care facilities (LTCF) (8) acute care facilities (1) and other settings (2). Four new ILI outbreaks were reported in LTCF. Among the outbreaks with available information (11), 10 were associated with influenza A (of which 2 were associated with A(H3N2)), and one outbreak was associated with influenza B.
To date this season, 838 laboratory-confirmed influenza outbreaks have been reported (Figure 6):
- 496 (59%) outbreaks were in LTCF, 30 were in schools and daycares, 120 in acute care facilities, and 192 were in other settings.
- Among the 752 outbreaks for which the influenza type was available, 98% (734) were associated with influenza A.
- Among the 323 outbreaks for which the influenza A subtype was available, 59% (192) were associated with influenza A(H3N2);
To date this season, 157 ILI outbreaks have been reported; 91 occurred in LTCF, 61 in schools, one in other settings and four in acute care facilities.
Figure 6 – Number of new outbreaks of laboratory-confirmed influenza by report week, Canada, weeks 2018-35 to 2019-19
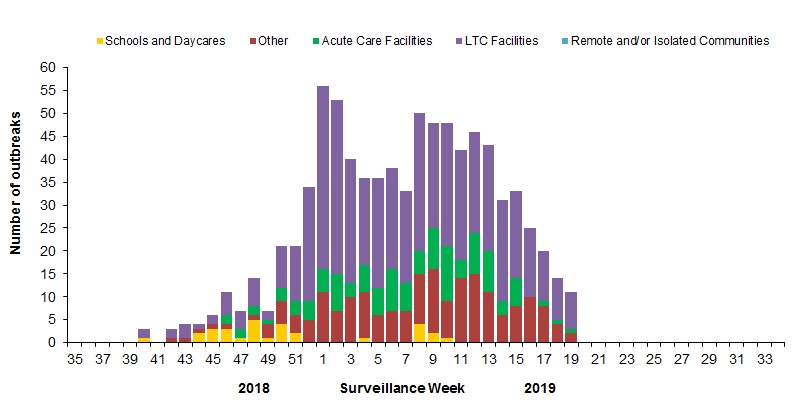
Figure 6 - Text equivalent
| Surveillance Week | Acute Care Facilities | Long Term Care Facilities | Other | Schools and Daycares | Remote and/or Isolated Communities |
|---|---|---|---|---|---|
| 35 | 0 | 0 | 0 | 0 | 0 |
| 36 | 0 | 0 | 0 | 0 | 0 |
| 37 | 0 | 0 | 0 | 0 | 0 |
| 38 | 0 | 0 | 0 | 0 | 0 |
| 39 | 0 | 0 | 0 | 0 | 0 |
| 40 | 0 | 2 | 0 | 1 | 0 |
| 41 | 0 | 0 | 0 | 0 | 0 |
| 42 | 0 | 2 | 1 | 0 | 0 |
| 43 | 0 | 3 | 1 | 0 | 0 |
| 44 | 0 | 1 | 1 | 2 | 0 |
| 45 | 0 | 2 | 1 | 3 | 0 |
| 46 | 2 | 5 | 1 | 3 | 0 |
| 47 | 2 | 4 | 0 | 1 | 0 |
| 48 | 2 | 6 | 1 | 5 | 0 |
| 49 | 1 | 2 | 3 | 1 | 0 |
| 50 | 3 | 9 | 5 | 4 | 0 |
| 51 | 3 | 12 | 4 | 2 | 0 |
| 52 | 4 | 25 | 5 | 0 | 0 |
| 1 | 5 | 40 | 11 | 0 | 0 |
| 2 | 8 | 38 | 7 | 0 | 0 |
| 3 | 3 | 27 | 10 | 0 | 0 |
| 4 | 6 | 19 | 10 | 1 | 0 |
| 5 | 6 | 24 | 6 | 0 | 0 |
| 6 | 9 | 22 | 7 | 0 | 0 |
| 7 | 6 | 20 | 7 | 0 | 0 |
| 8 | 5 | 30 | 11 | 4 | 0 |
| 9 | 9 | 23 | 14 | 2 | 0 |
| 10 | 12 | 26 | 8 | 1 | 0 |
| 11 | 4 | 24 | 14 | 0 | 0 |
| 12 | 9 | 22 | 15 | 0 | 0 |
| 13 | 8 | 23 | 11 | 0 | 0 |
| 14 | 3 | 22 | 6 | 0 | 0 |
| 15 | 6 | 19 | 8 | 0 | 0 |
| 16 | 0 | 15 | 10 | 0 | 0 |
| 17 | 1 | 11 | 8 | 0 | 0 |
| 18 | 1 | 9 | 4 | 0 | 0 |
| 19 | 1 | 8 | 2 | 0 | 0 |
Severe Outcomes Influenza Surveillance
Provincial/Territorial Influenza Hospitalizations and Deaths
To date this season, 3,092 influenza-associated hospitalizations have been reported by participating provinces and territoriesFootnote 1. Note that data from one participating jurisdiction has not been available since surveillance week 13, so these figures are an underestimate of cases for this season.
Hospitalizations (Table 2):
- 97.6% (3,018) were associated with influenza A
- Among the 1,704 cases for which the influenza subtype was available, 1,414 (83%) were associated with A(H1N1)pdm09.
- The highest estimated rate of hospitalization is among adults over 65 years of age.
Intensive Care Unit (ICU) cases and deaths:
- To date this season 555 ICU admissions and 182 deaths have been reported.
- 41% (227) of reported ICU admissions were in adults aged 45-64 years.
- All but eight ICU admissions were associated with influenza A.
- All but one of the deaths were associated with influenza A.
| Age Groups (years) | Cumulative (August 26, 2018 to May 11, 2019) | ||
|---|---|---|---|
| Influenza A | Influenza B | Rate per 100,000 population | |
| 0-4 | 394 | 23 | 87.61 |
| 5-19 | 235 | 20 | 18.38 |
| 20-44 | 354 | 10 | 12.79 |
| 45-64 | 776 | 7 | 35.98 |
| 65+ | 1259 | 14 | 104.63 |
| Total | 3018 | 74 | |
| % | 97.6% | 2.4% | |
|
|||
Pediatric Influenza Hospitalizations and Deaths
To date this season, 1,280 pediatric hospitalizations have been reported (Figure 7 & 8):
- 66% of cases were in children under 5 years of age.
- 93% (1,192) of cases have been associated with influenza A.
- Among the 375 cases for which the influenza subtype was available, 305 (81%) were associated with A(H1N1)pdm09.
To date this season, 246 ICU admissions, and 10 deaths have been reported.
- 58% (142) of ICU admissions were in children under 5 years of age.
- All but 13 (95%) of the ICU admissions have been associated with influenza A; 84% of the 105 cases for which the influenza A subtype was available were associated with A(H1N1)pdm09.
- 80% (8) of deaths occurred in children 2 to 4 years of age.
- All deaths have been associated with influenza A.
Figure 7 – Number of pediatric (≤16 years of age) hospitalizations reported by the IMPACT network, by week, Canada, weeks 2018-35 to 2019-19
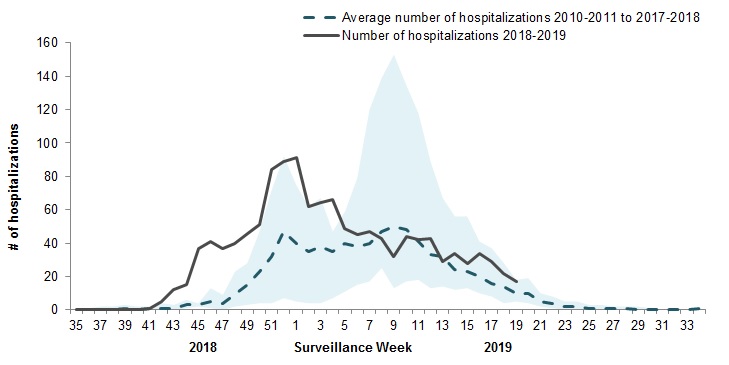
The shaded area represents the maximum and minimum number of cases reported by week from seasons 2010-11 to 2017-18
Figure 7 - Text equivalent
| Surveillance week | 2018-19 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0 | 0 | 0 | 0 |
| 36 | 0 | 0 | 0 | 1 |
| 37 | 0 | 0 | 0 | 2 |
| 38 | 0 | 0 | 0 | 2 |
| 39 | 0 | 1 | 0 | 3 |
| 40 | 0 | 0 | 0 | 2 |
| 41 | 1 | 1 | 0 | 2 |
| 42 | 5 | 1 | 0 | 4 |
| 43 | 12 | 1 | 0 | 3 |
| 44 | 15 | 3 | 1 | 6 |
| 45 | 37 | 3 | 2 | 4 |
| 46 | 41 | 5 | 1 | 13 |
| 47 | 37 | 4 | 0 | 9 |
| 48 | 40 | 9 | 2 | 23 |
| 49 | 46 | 15 | 3 | 28 |
| 50 | 51 | 23 | 4 | 47 |
| 51 | 84 | 32 | 4 | 72 |
| 52 | 89 | 47 | 7 | 92 |
| 1 | 91 | 40 | 5 | 75 |
| 2 | 62 | 35 | 4 | 62 |
| 3 | 64 | 38 | 4 | 67 |
| 4 | 66 | 35 | 7 | 47 |
| 5 | 49 | 40 | 11 | 59 |
| 6 | 46 | 38 | 15 | 79 |
| 7 | 48 | 40 | 17 | 120 |
| 8 | 42 | 47 | 25 | 139 |
| 9 | 34 | 50 | 13 | 153 |
| 10 | 45 | 48 | 17 | 135 |
| 11 | 42 | 41 | 18 | 118 |
| 12 | 42 | 33 | 13 | 89 |
| 13 | 29 | 32 | 14 | 67 |
| 14 | 34 | 24 | 12 | 56 |
| 15 | 28 | 23 | 13 | 56 |
| 16 | 34 | 20 | 10 | 41 |
| 17 | 29 | 16 | 8 | 37 |
| 18 | 22 | 14 | 4 | 28 |
| 19 | 17 | 10 | 5 | 18 |
Figure 8 - Cumulative numbers of pediatric hospitalizations (≤16 years of age) with influenza by age-group reported by the IMPACT network, Canada, weeks 2018-35 to 2019-19

Figure 8 - Text equivalent
| Age Group | Total |
|---|---|
| 0-5 mo | 138 |
| 6-23 mo | 318 |
| 2-4 yr | 389 |
| 5-9 yr | 285 |
| 10-16 yr | 150 |
Adult Influenza Hospitalizations and Deaths
Surveillance of laboratory-confirmed influenza-associated adult (≥16 years of age) hospitalizations by the Canadian Immunization Research Network (CIRN) Serious Outcomes Surveillance (SOS) network began on November 1st for the 2018-19 season.
To date this season, 942 hospitalizations, 115 ICU admissions and 54 deaths have been reported (Figure 9):
- 873 (93%) hospitalizations were associated with influenza A.
- A greater proportion of hospitalizations have been reported among adults ≥65 years of age (60%) compared to adults <65 years of age (40%).
- Among the 204 cases for which the influenza subtype was available, 107 (52%) were associated with A(H1N1)pdm09.
- 86% of hospitalized cases reported more than one type of comorbid condition.
- The most commonly reported comorbidity was endocrine disorders, which were reported in 87% of hospitalized cases.
Figure 9 - Cumulative numbers of adult hospitalizations (>20 years of age) with influenza by age-group reported by CIRN, Canada, 2018-19, weeks 2018-44 to 2019-19

Figure 9 - Text equivalent
| Age Group | Total |
|---|---|
| 20-34 yr | 92 |
| 35-49 yr | 84 |
| 50-64 yr | 199 |
| 65-79 yr | 271 |
| 80+ yr | 296 |
Influenza Strain Characterizations
From September 1, 2018 to 16 May, 2019, the National Microbiology Laboratory (NML) has characterized 2 170 influenza viruses (440 A(H3N2), 1,594 A(H1N1) and 136 B) that were received from Canadian laboratories.
Genetic Characterization of Influenza A(H3N2):
191 influenza A(H3N2) viruses did not grow to sufficient hemagglutination titer for antigenic characterization by hemagglutination inhibition (HI) assay. Therefore, NML has performed genetic characterization to determine the genetic group identity of these viruses.
Sequence analysis of the HA gene of the viruses showed that:
- 20 viruses belonged to genetic group 3C.2a.
- 168 viruses belonged to subclade 3C.2a1.
- Four viruses belonged to 3C.3a.
- One isolate could not be sequenced.
A/Singapore/INFIMH-16-0019/2016-like virus belongs to genetic group 3C.2a1 and is the influenza A(H3N2) component of the 2018-19 Northern Hemisphere influenza vaccine.
Antigenic Characterization:
Influenza A (H3N2):
- 148 influenza A(H3N2) viruses were antigenically characterized as A/Singapore/INFIMH-16-0019/2016-like by HI testing using antiserum raised against egg-propagated A/Singapore/INFIMH-16-0019/2016.
- 99 viruses showed reduced titer with ferret antisera raised against egg-propagated A/Singapore/INFIMH-16-0019/2016.
- A/Singapore/INFIMH-16-0019/2016-like virus is the influenza A(H3N2) component of the 2018-19 Northern Hemisphere influenza vaccine.
- 101 influenza A(H3N2) viruses characterized belonged to genetic group 3C.2a1. 20 viruses belonged to genetic group 3C.2a and 116 to 3C.3a. Sequencing is pending for the remaining isolates.
Influenza A(H1N1):
- 1,551 A(H1N1) viruses characterized were antigenically similar to A/Michigan/45/2015, which is the influenza A(H1N1) component of the 2018-19 Northern Hemisphere influenza vaccine.
- 43 viruses showed reduced titer with ferret antisera raised against cell culture-propagated A/Michigan/45/2015
Influenza B:
Influenza B viruses can be divided into two antigenically distinct lineages represented by B/Yamagata/16/88 and B/Victoria/2/87 viruses. The recommended influenza B components for the 2018-19 Northern Hemisphere influenza vaccine are B/Colorado/06/2017 (Victoria lineage) and B/Phuket/3073/2013 (Yamagata lineage).
- 25 influenza B viruses were characterized as B/Colorado/06/2017, which belong to the Victoria lineage and are included as an influenza B component of the 2018-19 Northern Hemisphere influenza vaccine.
- 88 viruses showed reduced titer with ferret antisera raised against cell culture-propagated B/Colorado/06/2017. Sequence analysis showed that 87 viruses that showed reduced titer had a three amino acid deletion (162-164) in the HA gene.
- 23 influenza B viruses were characterized as B/Phuket/3073/2013-like, which belongs to the Yamagata lineage and is included as an influenza B component of the 2018-19 Northern Hemisphere quadrivalent influenza vaccine.
Antiviral Resistance
Antiviral Resistance – Amantadine:
449 influenza A (89 A(H3N2) and 360 A(H1N1)) viruses were tested for resistance to amantadine and it was found that:
- All 449 influenza A viruses were resistant to amantadine.
Antiviral Resistance – Oseltamivir:
1,262 influenza viruses (159 A(H3N2), 1,024 A(H1N1) and 79 B) were tested for resistance to oseltamivir and it was found that:
- All 159 A(H3N2) viruses were sensitive to oseltamivir.
- Of the 1,024 A(H1N1) viruses tested, 1,020 were sensitive to oseltamivir and four viruses were resistant to oseltamivir with a H275Y mutation.
- All 79 B viruses were sensitive to oseltamivir.
Antiviral Resistance – Zanamivir:
1,261 influenza viruses (159 A(H3N2), 1,023 H1N1 and 79 B) were tested for resistance to zanamivir and it was found that:
- All 1,261 influenza viruses were sensitive to zanamivir.
Vaccine Monitoring
Vaccine monitoring refers to activities related to the monitoring of influenza vaccine coverage and effectiveness.
Vaccine Coverage
The Seasonal Influenza Immunization Coverage Survey is an annual telephone survey conducted between January and February that collects information related to the influenza vaccine in Canada. This survey measures vaccine coverage, which is the percentage of people who received the annual seasonal influenza vaccine in a specific influenza season.
In the 2018-19 influenza season, coverage was:
- 34% among adults aged 18 to 64 years.
- 31% among adults aged 18-64 wihout chronic diseases.
- 43% among adults aged 18 to 64 years with chronic diseases.
- 70% among seniors (aged 65 years and older).
| Age group (years) | Male | Female | Combined | |||
|---|---|---|---|---|---|---|
| Total | Vaccine Coverage (%) | Total | Vaccine Coverage (%) | Total | Vaccine Coverage (%) | |
| All adults (≥18) | 1568 | 36.6 | 2150 | 46.8 | 3726 | 41.8 |
| 18-64 | 1252 | 28.6 | 1640 | 39.9 | 2898 | 34.3 |
| without chronic diseases | 948 | 25.8 | 1171 | 36.1 | 2124 | 30.8 |
| with chronic diseases | 304 | 36.3 | 465 | 48.5 | 770 | 42.8 |
| ≥65 | 316 | 69.0 | 510 | 70.9 | 828 | 69.9 |
|
||||||
Vaccine Effectiveness
Vaccine effectiveness (VE) is a measure of how well the influenza vaccine is able to prevent influenza illness. Throughout the influenza season, surveillance networks estimate how well the influenza vaccine is working. Estimates can vary depending on several factors such as the study methods; the population, setting and outcomes that are being studied; the type and mix of vaccine products; the stage of the season and the kinds of influenza viruses that are circulating when the study is conducted.
The community-based Canadian Sentinel Practitioner Surveillance Network (SPSN) published an interim VE estimate in January 2019 for A(H1N1)pdm09. Subsequently, given an atypical late-season wave of influenza A(H3N2),SPSN has undertaken an additional interim analysis to assess effectiveness of the 2018/19 influenza vaccine against medically-attended outpatient A(H3N2) illness. Vaccine effectiveness (VE) monitoring methods and results are available at the SPSN website.
Based on data collected as of March 30th, 2019 from more than 2800 patients from B.C., Alta., Ont., and Que., the 2018/19 northern hemisphere vaccine effectiveness has varied depending on the strain.
- A(H1N1)pdm09: In the first interim analysis, VE against A(H1N1)pdm09 was 72% (95% CI: 60 to 81) overall, with substantial protection observed in all age groups. In the most recent analysis as of March 30th, estimates against A(H1N1)pmd09 have remained stable at approximately 70%.
- A(H3N2): In the most recent analysis, VE against A(H3N2) was 23% (95% CI: -9 to 46) overall. As the confidence interval crosses zero, this estimate does not provide evidence of vaccine protection against medically-attended outpatient A(H3N2) illness.
The SPSN continues to monitor and will further update VE estimates at end of season.