National Standards for Immunization Coverage Assessment: Recommendations from the Canadian Immunization Registry Network
1. Introduction/Background
Across Canada, publicly-funded immunization programs are provided for the routine administration of childhood vaccines against diphtheria, pertussis, tetanus, poliomyelitis, Haemophilus influenzae type b, measles, mumps, rubella, and hepatitis B infectionsFootnotei. Since 2000, new programs have been introduced in Canada based on recommendations from the National Advisory Committee on Immunization (NACI). Each vaccine is administered according to provincial and territorial immunization schedules which vary slightly between jurisdictions. To monitor the protection of a population against vaccine preventable diseases, coverage is considered an important health indicator. It is a sensitive measure and can be used as a proxy measurement to evaluate health services, systems, and interventionsFootnoteiiFootnoteiii. In addition, it is useful to monitor national immunization coverage trends over time, thus contributing to the evaluation of programs and progress towards the achievement of a targetFootnoteiv.
Immunization coverage information is often collected and reported at local, provincial/territorial, and national levels. National standards for the reporting of immunization coverage in Canada were last published in 2005Footnotev. These standards specify that coverage should be reported by the 2nd, 7th and 17th birthday as well as for school-age programs, by antigen and should be reported annually from immunization registries or every two years when using surveys.
Standards were developed to facilitate the collection of aggregate data, to enable the comparison of immunization coverage among jurisdictions, and to develop a standard approach to measuring immunization coverage in Canada. Current challenges that need to be overcome in terms of immunization coverage data collection and reporting include:
- Irregular and inconsistent data collection;
- Differences in methods of coverage assessment;
- A lack of standardized reporting definitions (e.g. numerator, denominator); and
- Heterogeneity among jurisdictional immunization schedules.
The purpose of defining national standards for immunization coverage reporting is to accomplish the following:
- Facilitate sharing of comparable immunization information among federal/provincial/territorial jurisdictions while enhancing the reporting capabilities of immunization registries or other information systems;
- Produce an aggregate measure of immunization coverage at the national level; and
- Contribute to the development of Provincial and Territorial (PT) standardized Immunization Registries, a key objective of the National Immunization StrategyFootnote vi.
Updating the national coverage reporting standards in 2012 and reviewing them in 2014 reflects the changes in priorities for coverage reporting in Canada over time. In addition, while past coverage standards focused mainly on reporting coverage estimates from survey data, this update revises the standards so that they are applicable to electronic immunization registries, surveys or alternative methods of data collection. Revisions to the standards were made through a consultative process with the provinces/territories.
This report presents the methods used to develop the 2005 and 2008 national coverage reporting standards and as well as the updated recommendations made by the CIRN in 2012 and revised in 2014. This document describes immunization coverage reporting standards for the 2 year old age group, in 2012 CIRN also decided to consider other age groups. In the future, it is hoped that a national network of immunization registries will be in place in Canada and these reporting standards will provide a “gold standard” for reporting vaccine coverage.
2. Methods
In 2004, a survey was completed by provincial/territorial representatives of the Canadian Immunization Registry Network (CIRN) on the standards used for provincial/territorial coverage reporting. The results of the survey were presented at the CIRN meeting, sponsored by Health Canada (HC), in July, 2004. During the meeting, key objectives for reporting immunization coverage were discussed, and recommendations for the reporting of national immunization coverage were drafted. In addition to the survey, a literature review of national and international coverage assessment methods contributed to these deliberations, the results of which are contained within the full report (available from the Public Health Agency of Canada, PHAC).
In October 2004, the Canadian Immunization Committee (CIC) requested that the Field Surveillance Officer (FSO) program address the improvement of national coverage reporting and prioritised this as a first collaborative project for the FSO’s. In 2005, a set of national coverage reporting standards for Canada was published in the CCDRFootnote v.
In Oct 2006, a survey was completed by the BC CDC and presented to CIRN which confirmed that the adoption of national coverage standards was inconsistent across the country. Based on these results CIRN identified a need to investigate:
- Reasons for the non-adoption of national coverage standards by jurisdictions;
- Success of these standards in reconciling differences between provincial coverage rates in order to increase comparability
In winter 2007, FSO through the FSO program in Alberta, Manitoba and Saskatchewan conducted a pilot project to investigate the feasibility of extracting and analyzing data from provincial immunization registries according to the 2005 national coverage reporting standards. The purpose of this pilot project was to help identify gaps in the existing reporting standards and to identify the reasons for the lack of consistent adoption. An analysis plan was developed following the existing national coverage reporting standards. Where gaps were identified, additional criteria were defined if deemed necessary to complete a full analysis. Some of these additional criteria included the definition of a common immunization schedule for participating provinces and providing definitions for terms such as valid doses, grace period etc., which were defined differently by these Provinces and Territories. When definitions were missing, current Provincial and Territorial definitions were used. At the end of the pilot study, each Province and Territory was asked to evaluate the process and the level of effort required to implement the 2005 standards. Coverage estimates produced from each province were also compared.
Results from the pilot study were presented to CIRN in June 2007 and a task group composed of FSOs and CIRN members was formed to revise the standards. In February 2012, CIRN met to review and revise all standards presented in the current document to confirm the recommendations. These standards were reviewed in fall 2014 by the CIRN and updates provided were included.
This document presents the final revised version of the 2014 national coverage reporting standards.
3. Results
Results and recommendations from the 2007 pilot project are as follows:
Key Results
- Overall coverage rates between provinces were comparable when using the same coverage reporting standards. Up-to-date coverage by 24months for MMR varied from 83% to 87% and for DTaP from 67 to 75%;
- Ability to apply national standards varied by registry. All the 2005 national reporting standards could be applied, however additional definitions developed for this project (e.g. valid doses, exemption/contraindication inclusions, uniform denominator) could not always be applied due to lack of available registry information or functionality; and
- The obstacles identified in applying the national standards included: limited time for additional programming of national reporting standards and lack of detail in national standards
Recommendations/Next Steps
- National standards need to be applicable to both registry data and survey data. Immunization registry data is richer and enables more elaborate reporting than survey data for which the 2005 reporting standards were intended
- Need clearer analysis methodology in national standards
- Need clear definitions for the following terms:
- Valid doses
- Reasons for not immunized (i.e. exemptions & contraindications)
- On-time and up-to-date coverage
- Denominator populations
- Provide clear direction on how to manage:
- Aboriginal group-specific data (First Nations receiving vaccine on reserve, First Nations off reserve, Inuit, Métis)
- Immunization schedules for national reporting
Recommendations for reporting of national immunization coverage (February 2012)
It was recommended that two broad coverage measures be reported nationally and include immunizations that are:
- up-to-date,
- on-time.
These can be calculated either using registry data or data from other sources (i.e. survey, physician billing data etc.). Within each measure, the definitions of some key elements for analysis are further defined and an example is provided to illustrate their use. Some of the elements common to the three outcomes are defined under a “general rules” section (Table 2) that follows the outcomes (Coverage Measure #1 & #2).
It was recommended that data be used to assess immunization coverage for school-based programs and adult immunization programs as well as from client registries.
The following immunization schedule outlines the immunization requirements for the recommended coverage outcomes.
Immunization Schedule
The immunization schedule for the purposes of national reporting should be based on the minimum common criteria for all provincial schedules at the time of reporting (e.g. yearly). Immunization schedules in each province/territory (PT) vary, but consider current NACI recommendations Footnote viii. As of February 2012, requirements are as follows:
Up-to-Date
Info
UP-TO-DATE Coverage at 2 years, 7 years and school-based programs
- Routine schedule
- By 24 months
- ≥ 4 DTaP-3IPVFootnote ix-4Hib (2,4,6,18 months)
- 1 MMR (12 months)
- 3 Pneumococcal Conjugate (2, 4, (6HR), 12-15 months)
- 1-4 Meningococcal Conjugate (by 12 months)
- 2 Varicella (12, 18 months)
- HB Infancy (3 doses) or pre-teen (2-3 doses)
- ≥ 1 Influenza
- 2 Rotavirus (2, 4 months)
- By 7 years
- ≥ 5 DTaP-4IPVFootnote ix-4Hib (2,4,6,18 months, 4-6 years)
- 2 MMR (12 AND 18 months OR 4-6 years)
- 3 Pneumococcal Conjugate (2, 4, (6HR), 12-15 months)
- 1-4 Meningococcal Conjugate (2, 4, 6, 12 months)
- 2 Varicella (12, 18 months)
- HB Infancy (3 doses) or pre-teen (2-3 doses)
- ≥ 1 Influenza
- By 17 years
- ≥ 6 DTaP-4IPVFootnote ix-4Hib (2,4,6,18 months, 4-6 years, 14-16 years)
- 2 MMR (12 AND 18 months OR 4-6 years)
- 3 Pneumococcal Conjugate (2, 4, (6HR), 12-15 months)
- 1-4+1 Meningococcal Conjugate (2, 4, 6, 12 months and pre-teen)
- 2 Varicella (12, 18 months)
- HB Infancy (3 doses) or pre-teen (2-3 doses)
- ≥ 1 Influenza
- 3 HPV (9-18 years at 0, 2, 6 months apart)
- By 24 months
- “Late starter” schedule
- Routine immunization schedule for children 24 months or <7 years of age not immunized in early infancy:
- DTaP-IPV:
- 4 doses (first visit, 2, 2, and 6-12 months later)
- Tdap:
- 1 dose at 14-16 years of age
- Hib:
- if 1st dose between 2-11 months: 3 doses
- if 1st dose between 12-14 months: 2 doses
- if 1st dose between 15-23 months: 1 dose
- MMR-Var:
- 2 doses (first visit and 2 months later)
- Pneumococcal Conjugate (2,4 months):
- if 1st dose between 2-6 months: 4 doses (ROUTINE)
- if 1st dose between 7-11 months: 3 doses
- if 1st dose between 12-21 months: 2 doses
- if 1st doses between 22-23 months: 1 dose
- Meningococcal Conjugate:
- if started at 12 months or later: 1 dose first visit + 1 dose at 12 years of age
- HB
- 3 doses (first visit, 2, and 6-12 months late
- HPV
- 3 doses (starting at 9 years of age)
- DTaP-IPV:
- Routine immunization schedule for children ≥ 7 years of age up to 17 years of age not immunized in early infancy
- Tdap:
- 4 doses (first visit, 2, 6-12 months, and 10 years later)
- IPV
- 3 doses (first visit, 2, and 6-12 months later)
- MMR-Var:
- 1-2 doses (first visit and 2 months later)
- Meningococcal Conjugate:
- 1 dose (first visit)
- HB
- 3 doses (first visit, 2, and/or 6-12 months later)
- HPV
- 3 doses (starting at 9 years of age, first visit, 2, and 6-12 months later)
- Tdap:
- Routine immunization schedule for children 24 months or <7 years of age not immunized in early infancy:
On-Time
Info
Same schedule as “up-to-date” coverage. However, spacing and timing of doses is determined by the eligible, due and overdue guidelinesFootnote vii.
- NOTE 1: For on-time coverage, not all antigens will be comparable across all Provinces and Territories. When assessing Hep B, Pneumo and Men C, the number of doses required by each age milestone will vary by Province and Territory, due to varying immunization schedules.
- NOTE 2: Children who do not start routine immunization during early infancy (i.e. later starters) are by definition not considered on-time for routine immunization. Therefore no alternate schedule is provided to calculate on-time immunization.
- Canadian Immunization Guide, November 2012.
- Publicly funded Immunization Programs in Canada - Routine Schedule for Infants and Children including special programs and catch-up programs, Public Health Agency of Canada in collaboration with the Canadian Nursing Coalition on Immunization (CNCI)
- National Eligible, Due, and Overdue Guidelines for Immunization Registries: Draft Recommendations from the Canadian Immunization Registry Network, Data Standards Task Group, Canada Communicable Disease Report (CCDR) March 15, 2004: Volume 30
Recommended outcomes for National Immunization Coverage Measurements
Coverage Measure #1: Up-to-date coverage (UTD)
Definition
Receipt of the full number of valid and required vaccine doses by the first or second birthday (See table 2 for definition of a valid dose). Valid doses must be received during the recommended period to maximize immunogenicity of each vaccine; as well as take into consideration the recommended time interval between each dose.
Intended use
A measure of population protection.
Numerator
The number of children who have received all valid and required doses by the 2nd birthday.
Rationale for age groups
Measuring coverage by the 2nd birthday is useful for national and international comparisons as this is a widespread age marker and to measure receipt of the full series of childhood vaccines.
Up-to-date Measures
The following three measures are recommended when reporting up-to-date coverage. Children who fulfill the following requirements by 2nd birthday are considered “up-to-date” and should be included in the numerator:
- By Antigen: An individual coverage estimate is reported for each of the following antigens and dose requirements, resulting in a single coverage measure per antigen
- By 2nd b-day: 4 doses each of D, T, aP, Hib, 3 doses each of IPV, and PCV-13.., 1 dose each of Mea, Mu, R, Men CVar and InfFootnote x
- By 7th b-day: DTaP (5 doses)-IPV (4)-Hib (4)+ 2 doses of Mea, 1 dose each of Mu and R
- By 17th b-day: DTaP (6 doses)-IPV (4)-Hib (4)+ 2 doses of Mea, 1 dose each of Mu and R
- Series of Antigens excluding new vaccinesFootnote xi: One coverage measure is reported which encompasses all of the following antigens and dose requirements, resulting in one up-to-date measure
- By 2nd b-day: DTaP (4 doses)-IPV (3)-Hib (4)+MMR (1 OR 2)+ PCV-13 (2-3 based on age at first dose) + MenC (1-3 based on age at first dose) + Var1 (2) + Inf (1)
- By 7th b-day: DTaP (5 doses)-IPV (4)-Hib (4)+MMR (2)
- By 17th b-day: DTaP (6 doses)-IPV (4)-Hib (4)+MMR (2)
- All antigensFootnote xii: One coverage measure is reported which encompasses all the following antigens and dose requirements, resulting in one up-to-date measure
- By 2nd b-day: DTaP (4 doses)-IPV (3)-Hib (4)+MMR (1 OR 2)+ PCV-13 (2-3 based on age at first dose) + MenC (1-3 based on age at first dose) + Var1 (2) + HB Infancy (3 or pre-teen 2-3) + Inf (1) + Rot (2)
- By 7th b-day: DTaP (5 doses)-IPV (4)-Hib (4)+MMR (2)+ PCV-13 (2-3 based on age at first dose) + MenC (1-3 based on age at first dose) + Var1 (2) + HB Infancy (3 or pre-teen 2-3) + Inf (1) + Rot (2)
- By 17th b-day: DTaP (6 doses)-IPV (4)-Hib (4)+MMR (2)+ PCV-13 (2-3 based on age at first dose) + MenC (1-3 based on age at first dose) + Var1 (2) + HB Infancy (3 or pre-teen 2-3) + Inf (1) + Rot (2) + HPV (3)
Denominator
For coverage by the 2nd birthday:
- All children with “active records”Footnote xiii in the birth cohort (i.e. population registry) who turn 2 years of age within the period of interest for assessment (i.e. all children who had their second birthday during the calendar year from January 1 – December 31). The denominator should include all children who have an exemption or contraindications (See Table 2 for definition of exemptions and contraindications).
Provinces/territories that rely on immunization registries for coverage assessment should undertake periodic evaluations to ascertain the completeness of their denominator population.
Coverage Calculation
The following calculation should be completed for each antigen or for combination of antigens:
Registry Data
- x
- # of children with active records in registry who have completed the routine childhood immunization schedule by 1st (or 2nd) birthday in province
- y
- Total # of children with active records in provincial registry who turn 1 or 2 within year or interest
Survey Data
- x
- # of children in survey sample who have completed the routine childhood immunization schedule by 1st (or 2nd) birthday
- y
- Total # of children whose parents have responded to the survey
Example 1
A child turns two years of age on March 15th, 2006. Coverage is being assessed in 2006; therefore this child is eligible and included in the birth cohort of interest for coverage assessment. The child received all recommended doses of each antigen, however some doses were invalid (e.g. 2nd dose DTaP-IPV-Hib was too early) and some doses were late (e.g. 4th dose received after 2nd birthday). This child would not be counted as “up-to-date” in the numerator.
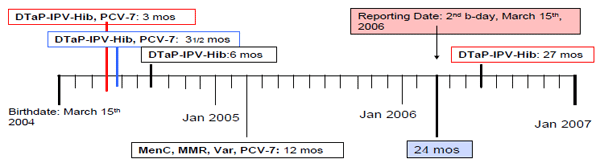
Example 1 figure description
This figure displays an example of how "up-to date" status is determined by immunization events. The time scale shown displays yearly intervals since the birthdate of a child born on March 15th, 2004. Each box along the time scale displays a single dose; which includes the antigens given and the age at which the immunization was delivered. The antigens given include diphtheria-tetanus-pertussis (DTaP), polio (IPV), Haemophilus influenza type b (Hib) at 3, 3.5,6, and 27 months of age, pneumococcal (PCV-7) at 3, 3.5, and 12 months of age, and measles-mumps-rubella (MMR), varicella (Var), pneumococcal (PCV-7), and meningococcal (MenC) at 12 months of age. In this example, the child would be eligible for immunization coverage assessment within the year of interest 2006 indicated by the reporting period box. This child received all recommended doses of each antigen, however the second dose of DTaP-IPV-Hib and PCV-7 at 3.5 months of age are invalid (e.g. given too early) and the fourth dose of DTaP-IPV-Hib is late (e.g. received after 2nd birthday). The 2nd birthday is indicated by the "24 months" box. Therefore this child would not be counted as "up-to-date" for PCV-7 and the DTaP-IPV-Hib series of antigens.
| By Single Antigen | Doses received by 2nd birthday | Up-to-date? | Reason |
|---|---|---|---|
| Diphtheria/Tetanus/Polio/Pertussis/ Hib | 3 | No | 3 doses received by 2nd birthday, but 1 dose received after 2nd birthday. 2nd dose invalid as received too early– |
| PCV-7 | 3 | No | dose 2 received too early, therefore invalid and not counted |
| Measles/mumps/rubella | 1 | Yes | 1 dose received before 2nd birthday |
| MenC | 1 | Yes | 1 dose received before 2nd birthday |
| Varicella | 1 | Yes | 1 dose received before 2nd birthday |
| By series of antigens minus new vaccines | |||
| DTaP-IPV, Hib, MMR | 3-3-1 | No | Dose 4 of DTaP-IPV, Hib received past 2nd birthday |
| By series of antigens including all vaccines | |||
| DTaP-IPV, Hib, MMR, Men C, PCV-7, Varicella | 3-3-1-1-3-1 | No | Dose 4 of DTaP-IPV, Hib received past 2nd birthday |
Coverage Measure #2: On-time coverage
Definition
Receipt of the full number of vaccine doses by the 1st and 2nd birthday. These guidelines also specify a 1 month leeway period after the recommended time of receipt of the dose. A 1 month leeway period allows for a child to be immunized one month after they are scheduled to receive the dose according to the immunization schedule (listed above). One month is defined as 4 weeks (28 days) or can be defined in calendar days according to the programming protocol being used. Doses must respect spacing criteria defined in the national eligible, due and overdue guidelines.
See Table 2 for detailed definition of “on-time period” and notes for programming
Intended use
On-time coverage is a measure of the success of immunization program delivery and resource administration.
Numerator
The number of children who have received all valid doses required to be on-time by the specified age milestones.
The following three measures are recommended when reporting on-time coverage. Children who fulfill the following requirements by the 1st and 2nd birthday are considered “on-time” and should be included in the numerator:
- By Antigen
- An individual coverage estimate is reported for each antigen, by each age milestone taking into account only the doses required for that antigen by that age. If even one dose of a particular antigen is late (or early) then the child will not be considered on-time for that antigen. - See immunization schedule (listed above)
- All antigens minus new vaccines
- One coverage measure is reported at each age milestone which encompasses all of the following antigens and dose requirements. - See immunization schedule (listed above)
- All antigens
- One coverage measure is reported which encompasses all the following antigens and dose requirements, resulting in one up-to-date measure - See immunization schedule (listed above)
Denominator
For coverage by the 1st or 2nd birthday:
- All children with “active records”Footnote xiii in the birth cohort (e.g. population registry) who turn 1 year or 2 years of age within the period of interest for assessment (i.e. all children who had their first or second birthday during the calendar year e.g. from January 1 – December 31). The denominator should include all children who have an exemption or contraindication (See Table 2 for definition of exemptions and contraindications)
Example 2
Scenario for a child not on-time
Child turns two years on March 15th, 2006 (i.e. they turn two years within the year of interest - 2006). All required doses of each antigen are received before the child’s 2nd birthday, however they are not all received at the correct time and with correct interval between doses.
Excerpt from the eligible, due and overdue guidelines:
- DTaP 3rd dose:
- Eligible
- 28 days from second dose
- Due
- 2 months from second dose
- Overdue
- 3 months from second dose
3rd dose of DTaP is only “on-time” if administered between 28 days from the 2nd dose to the end of the 6th month of life (i.e. by 3 months from 2nd dose). In the scenario below the 3rd dose was late, therefore child is not considered “on-time”.
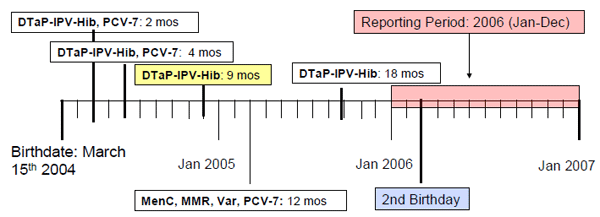
Example 2 figure description
This figure displays an example of how "on-time" status is determined by immunization events. The time scale shown displays yearly intervals since the birthdate of a child born on March 15th, 2004. Each box along the time scale displays a single dose; which includes the antigens given and the age at which the immunization was delivered. The antigens given include diphtheria-tetanus-pertussis (DTaP), polio (IPV), Haemophilus influenza type b (Hib) at 2, 4, 9 and 18 months of age, pneumococcal (PCV-7) at 2, 4, and 12 months of age, and measles-mumps-rubella (MMR), varicella (Var), and meningococcal (MenC) at 12 months of age. In this example, this child would be eligible for immunization coverage assessment within the year of interest 2006 indicated by the reporting period box. All required doses of each antigen are received before the child's 2nd birthday. However, they are not all received at the correct time and with correct intervals between doses. The third dose of DTaP-IPV Hib, PCV-7 given at 9 months of age is only "on-time" if administered between 28 days from the second dose to the end of the 6th month of life (e.g. by 3 months from 2nd dose). In this example the third dose was late; therefore this child would not be considered "on-time" for the DTaP-IPV-Hib series of antigens.
Table 2 - National Recommendations for Coverage Assessment Reporting Standards - General Terminology
1. Valid doses
- Definition
- A valid dose is considered protective for the individual and should be included in immunization coverage reporting. A valid dose is administered after the specified minimum number of days from the previous dose of this antigen or from birth. A dose is invalid if it is given earlier than the "eligible" period for each vaccine.
- Considerations / Rationale
- The National Eligible, Due and Overdue Guidelines should be used to define spacing between doses and eligibility for the first dose
2. Grace periods
- Definition
- Grace periods are used in reference to an allowable period prior to dose eligibility within which an administered dose can still be considered valid. For the purposes of national coverage reporting, valid doses currently will not incorporate a grace period for any antigens
- Considerations / Rationale
-
In the U.S, the Advisory Committee on Immunization Practices (ACIP) recommends a 4-day grace period, meaning that if doses are received 4 days prior to their due date they can still be considered valid and do not need to be re-administered. This rule is intended for coverage assessment purposes and not intended for use when scheduling appointments for receipt of immunization. Research conducted in British Columbia by the BC Centre for Disease Control has indicated that using a grace period does not have a significant impact on coverage.
Variations in valid dose criteria may currently exist as some Provinces and Territories incorporate grace periods and may have policies that allow for an early dose of MMR to be counted as valid. This is commonly based on a subjective decision prior to entering the dose into the immunization registry. In the future, if grace periods are to be employed, a decision should be made regarding a common grace period in each Province and Territory.
3. Exemptions and Contraindications
- Definition
- For national reporting all exemptions and contraindications should be included in the denominator without distinction. At this time, exemptions and contraindications cannot be measured in all Provinces and Territories.
- Considerations / Rationale
-
Recording of exemptions and contraindications is variable in each Province and Territory if recorded at all. In addition, children with a contraindication are difficult to capture because they do not present for immunization and will not be flagged in the registry. In the future, when registries are available through a national network in all Provinces and Territories, and exemptions and contraindications can be more easily tracked, definitions should be as follows:
- Exemptions
- Clients are exempt because of medical, religious, or philosophical reasons. A medical example would be immunity from a previous illness or vaccination.
- Contraindications
- Specific immunizations are intentionally not administered, and this fact is recorded in the Electronic Public Health System. A contraindication indicates that the immunizing agent should not be administered because the health risk outweighs the benefit to the recipient.
4. Aboriginal population-specific data (First Nations/Inuit/Métis)
- Definition
-
The Canadian Constitution recognizes three distinct groups of Aboriginal people — First Nations, Inuit and Métis (FN/I/M). The First Nations group includes First Nations living on and off reserve.
For national reporting, it is recommended that data from provinces/territories include identifiers for FN (on and off reserve)/I/M populations when collected. For example, without an identifier, coverage for FNs living on reserve may be underestimated if they receive their immunization off reserve.
Some provinces/territories have mechanisms in place to disaggregate data for FN/I/M from their immunization registries. In provinces/territories where data from these populations can be disaggregated in the registry, it is recommended that these data be included in the numerator and the denominator for coverage calculations. - Considerations / Rationale
-
Health Canada's First Nations and Inuit Health Branch (
FNIHB) deliver or support the delivery of immunization programs for First Nations on-reserve that are reflective of respective provincial immunization schedules. On-reserve immunization coverage data are reported by First Nations communities to FNIHB regional offices in Alberta, Saskatchewan, Manitoba, Ontario, Quebec and Atlantic region (inclusive of New Brunswick, Nova Scotia, Prince Edward Island and Newfoundland and Labrador). A mechanism to incorporate these data within provincial registries should be considered.
- FN (on and off reserve)/I/M immunization data in Provincial/Territorial registries are often incomplete and/or not collected in a standard mechanism across jurisdictions, which leads to an under estimation of coverage. This is due to several factors: When FN/I/M are included in Provincial/Territorial-registries, identifiers often don't exist, or may be incomplete (e.g. requires self-identification which is often under reported).
- In some provinces, immunization of First Nations who live on-reserve will only be recorded in the provincial registry if received off-reserve.
If First Nations-specific data are available in Provincial/Territorial-registries, 'First Nations' can be defined in different ways across jurisdictions due to lack of a standardized definition.
Disaggregated FN (on and off reserve)/I/M-specific data are required to monitor change and identify trends, evaluate effectiveness of programs and guide evidence-based decision making. There is a need to collect immunization data using a standardized and coordinated approach across jurisdictions.
5. Denominator
- Definition
-
The denominator for coverage calculations is defined as all children in the "population" registry of the age of interest and within the year of interest. The denominator should only be based on "active" records defined as those children residing in the province during the time period of interest for coverage assessment. In some Provinces and Territories this may include all those with active health card numbers during the time period of interest.
Records for individuals that are "active" at the time of analysis, but who in the past may have had non-continuous enrolment for health care benefits within the province should be included in the denominator. Source data for "population" registry could be e.g. a person registry database, physician billing database, health card number database - Considerations / Rationale
- Tracking in and out migration within a Province and Territory can be difficult and varies between Provinces and Territories. Some provinces have migration flags in their person registry databases to account for the moves while others do this manually. In addition, tracking non-continuous enrolment is not always complete as benefits are not transferred during brief periods of leave from a provinces
6. On-Time period
- Definition
-
A 1 month leeway period after the recommended time for receipt of a dose is allowed for on-time coverage reporting. A 1 month leeway period allows for a child to be immunized one month after the dose is scheduled according to the immunization schedule (listed above).
- Note
- For programming and comparison purposes, one month should be defined in terms of "real-time" calendar days, meaning that the variation in the number of days between months is acknowledged (e.g. 28-31days). For provinces already defining 1 month as 28 days, they may wish to continue using this definition as the difference is not expected to affect coverage significantly. Provincial analysis has demonstrated a minor difference in coverage (i.e. 0.1%) when analyzing on-time coverage by calendar days rather than by using a fixed 28 day interval.
- Alternate Schedules
- Children who do not start routine immunization during early infancy (i.e. later starters) are not considered on-time for immunization. However, alternate schedules are indicated in the immunization schedule (listed above), which late starters can follow to be considered up-to-date for immunization coverage.
- Considerations / Rationale
- On-time coverage is a measure of immunization delivery and of compliance with the routine immunization schedule in a specific province. On-time coverage may be an under estimation of the proportion of children fully protected against vaccine preventable diseases as children can still be considered fully immunized if a schedule for "late starters" is followed.
7. Frequency and time of year for reporting
- Definition
-
Each jurisdiction should assess and report immunization coverage annually. An annual national immunization report compiled by
FSO will include coverage estimates from both the NICS and the jurisdictional assessments with an addendum detailing the methodology and definition of age cohorts.
- Registry Data
- Assessment on an annual basis.
- Survey Data
- Assessment on a biennial basis. For children and adolescents, assessment should be done in the fall, to account for school-based requirements (applicable to 7 & 17 year olds).
- Considerations / Rationale
- Once immunization registries are fully functioning nationally, it is recommended that coverage data be reported from registry records on an annual basis. Registries should maintain the capacity to report more frequently as needed for program planning and evaluation, and for consideration of new immunization programs.
Conclusions
The draft standards for reporting of national immunization coverage are based on a comprehensive review of international and national reporting standards and were agreed upon by consensus among CIRN members. Defining standards for reporting of national immunization coverage is an important step towards achieving optimal information flow and comparability of jurisdictional and national immunization coverage estimates.
National standards may be updated again in the future and revisions are dependent on programmatic changes and other factors such as the introduction of new vaccine programs and the development of a national immunization registry network which may affect the way in which coverage is measured.
Abbreviations
- DTaP
- Diphtheria, Tetanus, acellular Pertussis
- HB
- Hepatitis B
- HiB
- Haemophilus influenzae type b
- MenC
- Meningococcal Conjugate
- MMR
- Measles, Mumps, Rubella
- Rot
- Rotavirus
- Tdap
- Tetanus, diptheria, acelluar Pertussis
- Var
- Varicella