Sexually transmitted and blood-borne infections guideline development

 Download this article as a PDF
Download this article as a PDFPublished by: The Public Health Agency of Canada
Issue: Volume 46-11/12: Oral Health in Canada
Date published: November 5, 2020
ISSN: 1481-8531
Submit a manuscript
About CCDR
Browse
Volume 46-11/12, November 5, 2020: Oral Health in Canada
Overview
Translating evidence into practice with the National Advisory Committee on Sexually Transmitted and Blood-Borne Infections
Shamila Shanmugasegaram1, Stephan Gadient1, Margaret Gale-Rowe1
Affiliation
1 Centre for Communicable Diseases and Infection Control, Public Health Agency of Canada, Ottawa, ON
Correspondence
Suggested citation
Shanmugasegaram S, Gadient S, Gale-Rowe M. Translating evidence into practice with the National Advisory Committee on Sexually Transmitted and Blood-Borne Infections. Can Commun Dis Rep 2020;46(11/12):398-402. https://doi.org/10.14745/ccdr.v46i1112a06
Keywords: evidence-based guidelines, external advisory body, guideline methodology, infection prevention and control, sexually transmitted and blood-borne infections
Abstract
For over 30 years, the Government of Canada has developed guidelines on sexually transmitted and blood-borne infections (STBBI) with a group of subject matter experts. This expert group provided advice to the Public Health Agency of Canada (PHAC) from 2004 to 2019; transitioning to the National Advisory Committee on STBBI (NAC-STBBI) in 2019. NAC-STBBI supports PHAC's mandate to prevent and control infectious diseases by providing advice for the development of STBBI guidelines. The methodology for developing the NAC-STBBI recommendations is evolving to a more rigorous, systematic and transparent process that is consistent with current standards in guideline development. It is also informed by-and aligned with-the methods of several other major guideline developers. The methodology incorporates the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach, as appropriate, when conducting evidence reviews and developing recommendations. Recommendations will be published on the canada.ca website with the supporting NAC-STBBI Statement detailing the methodology and evidence used to develop them. This process will ensure that PHAC provides trustworthy evidence-based STBBI recommendations to primary care providers and public health professionals.
Introduction
Sexually transmitted and blood-borne infections (STBBI) remain a public health threat to Canadians. Rates of chlamydia, gonorrhea and syphilis have increased steadily in recent yearsFootnote 1, and multiple provinces and territories declared outbreaks of syphilis in 2018Footnote 2 and 2019Footnote 3. The Public Health Agency of Canada (PHAC) provides national leadership for the prevention and control of STBBI through the development of evidence-based public health guidelines. The recommendations in these guidelines are developed-with PHAC support-by the National Advisory Committee on STBBI (NAC-STBBI), an external advisory body of subject matter experts from across Canada. This article describes the new STBBI recommendation development process followed by PHAC and NAC-STBBI.
Background
An expert group has provided advice to the Government of Canada for more than 30 years. The first advisory committee-the Expert Interdisciplinary Advisory Committee on Sexually Transmitted Diseases (STD) in Children and Youth-was established in 1986 by Health and Welfare Canada to provide advice and guidance for the prevention and control of STD. In 1988, this committee published the first Canadian Guidelines for the Treatment of Sexually Transmitted Diseases in Neonates, Children, Adolescents and AdultsFootnote 4. While the Advisory Committee was disbanded in 1991, the Expert Working Group on STD was struck in 1998 under the authority of Health Canada's Laboratory Centre for Disease Control for the purpose of developing guidelines.
Following the creation of PHAC in 2004, the Expert Working Group on STD began providing advice to PHAC, and the name was changed to the Expert Working Group for the Canadian Guidelines on Sexually Transmitted Infections. In 2019, the Group transitioned to a formal external advisory body that is based on the principles and requirements set out under federal government legislation and policiesFootnote 5.
Mandate and membership
NAC-STBBI provides PHAC with ongoing, timely advice and recommendations for the development of STBBI guidelines, in support of its mandate to prevent and control infectious diseases in Canada. PHAC retains all decision-making authority and decides how it will use the recommendations and advice of the external advisory body.
NAC-STBBI carries out its mandate as follows: reviewing the epidemiology and scientific literature on STBBI as well as the evidence on specific prevention strategies, diagnosis and treatment; providing advice based on the best available literature evidence or, where there is a paucity of literature evidence, based on expert knowledge and practice; and advising PHAC about current and emerging issues relating to STBBI.
NAC-STBBI consists of 15 voting members with expertise in the areas of healthcare epidemiology, infectious disease, medical microbiology, laboratory diagnostics, pharmacology, obstetrics and gynecology, paediatrics, primary care, psychology, and public health. Recruitment ensures members have a range of knowledge, expertise and experience, as well as varied perspectives. Consideration is also given to geographic representation given that challenges may differ across Canada. PHAC ("the Secretariat") assesses and manages competing interests for NAC-STBBI members who must declare any conflicts upon joining, on an annual basis and prior to each meeting to maintain impartiality of the committee.
Recommendation development process
The methodology for developing the NAC-STBBI recommendations is evolving to a more rigorous, systematic and transparent process to formulate trustworthy recommendations. This new approach was informed by best practice standards in guideline developmentFootnote 6Footnote 7Footnote 8Footnote 9 and the methodology of several other major guideline developersFootnote 10Footnote 11Footnote 12Footnote 13Footnote 14Footnote 15Footnote 16. A manual has been drafted outlining the methodology, which is summarized herein and illustrated in Figure 1.
Figure 1: Recommendation development process
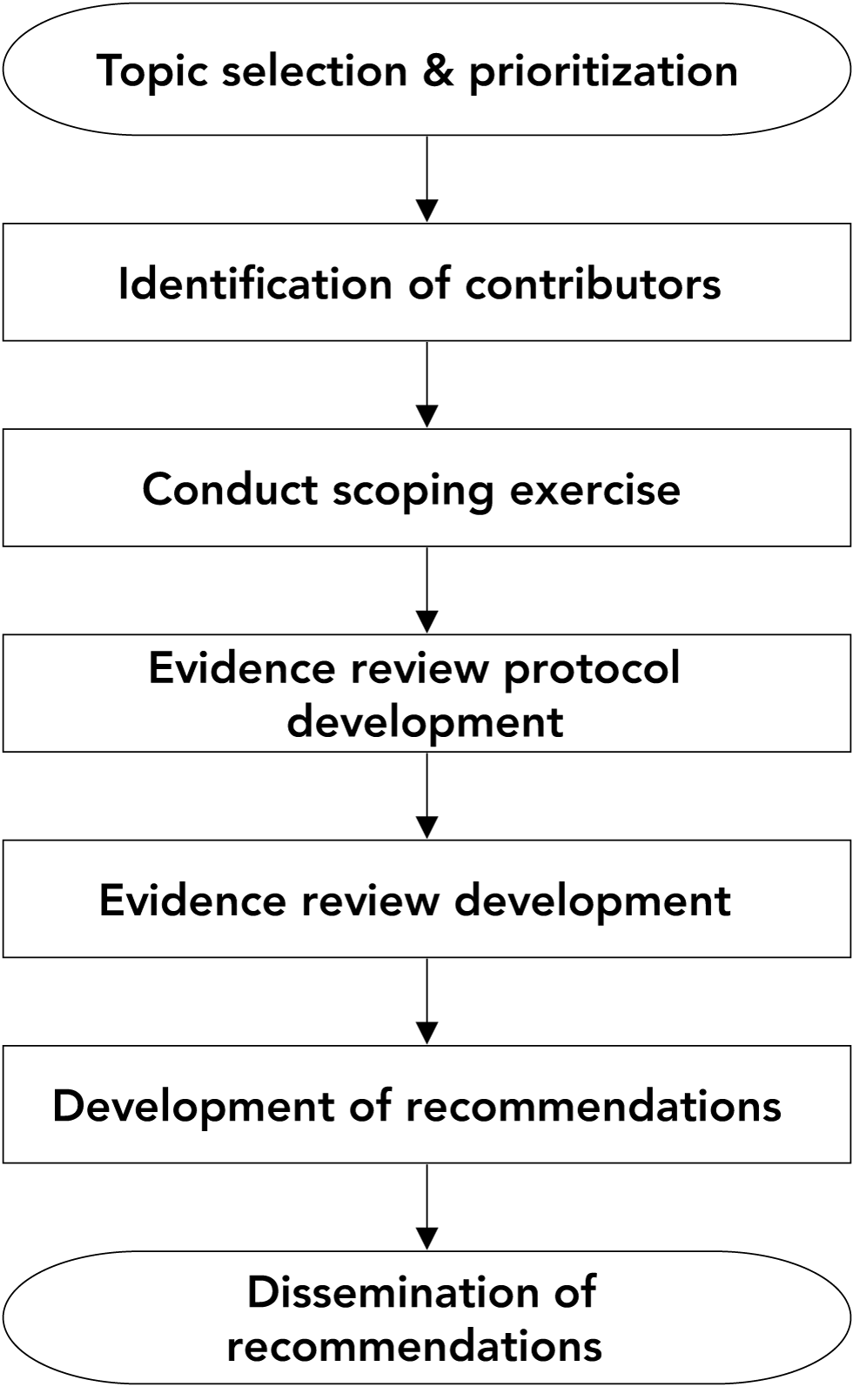
Text description: Figure 1
Figure 1: Recommendation development process
Figure 1 is a basic flow diagram outlining the order of steps to develop recommendations for the Public Health Agency of Canada's Sexually Transmitted and Blood-Borne Infections guidelines. The diagram begins with the topic selection and prioritization exercise, followed by the identification of contributors. The next steps in the process include conducting a scoping exercise, followed by the development of an evidence review protocol and the evidence review. The flow diagram concludes with the development and dissemination of the recommendations.
The Secretariat conducts a topic selection and prioritization exercise annually (or on an as-needed basis) to help determine which areas to focus on to update or reaffirm existing recommendations or develop new recommendations during the upcoming fiscal year. Figure 2 illustrates the topic selection and prioritization exercise. This exercise involves the following:
- Soliciting topics for development of recommendations
- Screening topics against the scope of the STBBI guidelines and PHAC mandates and priorities
- Completing an assessment tool which includes questions on the availability of evidence on the topic and epidemiology to help NAC-STBBI rank the topics in order of priority
- Ranking (repeated, if necessary) topics and discussing the results (NAC-STBBI)
- Deciding (the Secretariat) on the final list of topics for recommendation development, seeking PHAC approvals and sharing this list with NAC-STBBI. The topics that have not been prioritized for recommendation development will be considered again during the next topic prioritization exercise along with any new suggestions
Figure 2: Topic selection and prioritization exercise
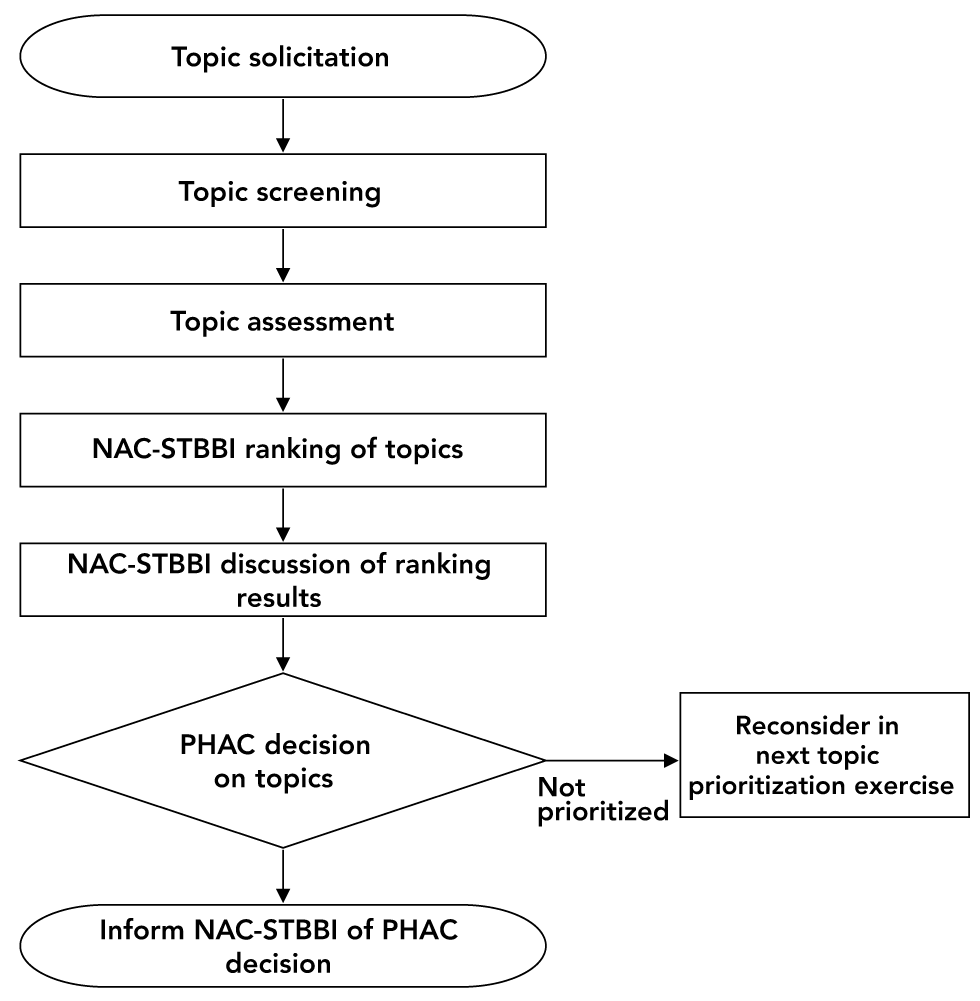
Text description: Figure 2
Figure 2: Topic selection and prioritization exercise
Figure 2 is a basic flow diagram outlining the topic selection and prioritization exercise process. The process begins with the solicitation of topics, followed by topic screening and then topic assessment. The National Advisory Committee on Sexually Transmitted and Blood-Borne Infections (NAC-STBBI) then ranks the topics. The results of the ranking exercise are discussed by NAC-STBBI, and the Public Health Agency of Canada (PHAC) then decides on the topics to be addressed. Topics that are not prioritized are reconsidered in the next prioritization exercise. NAC-STBBI is then informed of PHAC's decision.
A working group (WG) composed of experts from NAC-STBBI is formed for each prioritized topic. Other potential contributors include external experts (if necessary) and relevant stakeholders. The WG receives methodological and technical support from the Secretariat. A scoping exercise is conducted to identify relevant systematic reviews, guidelines and any major studies published or in progress since the release of the existing PHAC recommendations (if applicable) to help the WG develop the research (key and contextual) questions, inclusion and exclusion criteria, and analytic framework. The findings from the scoping exercise also help the WG determine the following: whether a systematic review is necessary; whether to use/update an existing systematic review or conduct a new systematic review; and whether to adopt, adapt or develop de novo recommendations for the topic under considerationFootnote 17Footnote 18.
The WG develops the key questions using the population, intervention, comparator and outcomes (PICO) framework and determines the inclusion and exclusion criteria for the evidence reviewFootnote 6. Outcomes (both beneficial and harmful) that are important for decision-making are identified from the scoping exercise and a targeted search of the literature (if necessary) in combination with feedback from the WG members and other contributors. The WG members rate the relative importance of outcomes for decision-making (critical, important or not important) based on the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approachFootnote 9. The WG also develops contextual questions and the analytic framework.
A systematic review is conducted independently by an external evidence review team. A librarian prepares the search strategy for the systematic review according to protocol parameters (e.g. study designs, time frame and databases) and it is reviewed by a second librarian and the WG. The systematic review protocol is drafted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis Protocols (PRISMA-P)Footnote 19Footnote 20 and includes the research questions, inclusion and exclusion criteria, analytic framework, and search strategy. The final version of the protocol is registered with the International Prospective Register of Systematic Reviews (PROSPERO) databaseFootnote 21.
When conducting a systematic review, the steps undertaken by the external evidence review team will include the following: screening the titles, abstracts and selected full-text articles based on predefined inclusion and exclusion criteria; extracting data; assessing the risk of bias; performing quantitative and/or qualitative synthesis; and preparing the GRADE evidence tablesFootnote 6Footnote 9. The quality/certainty of evidence is graded separately across studies (as high, moderate, low or very low) for each important outcomeFootnote 9. The systematic review article is prepared based on the PRISMA checklist and the PRISMA flow diagram is used to show the study selection processFootnote 22Footnote 23Footnote 24Footnote 25. The final version of the article is published in a peer-reviewed journal.
The GRADE Evidence to Decision framework is drafted to help the WG use evidence in a structured and transparent way to develop recommendationsFootnote 9Footnote 26Footnote 27Footnote 28Footnote 29Footnote 30. The overall quality of evidence is assessed across outcomesFootnote 9Footnote 26Footnote 27Footnote 28Footnote 29Footnote 30.
Other types of evidence reviews, such as narrative reviews or rapid reviews, may be conducted for certain guidance products. When evidence from the literature is very limited, recommendations are developed based on expert opinion using a systematic and transparent approach.
The WG drafts recommendations using the GRADE wording for direction and strength, as appropriateFootnote 9Footnote 14, and presents them to NAC-STBBI for discussion and voting. NAC-STBBI Statement, drafted by the WG, includes the need for the recommendations, the methodology used, the evidence considered, the final recommendations and a summary of the NAC-STBBI deliberations. The Statement is reviewed by NAC-STBBI and published on the canada.ca website after PHAC approvals. The relevant PHAC STBBI guides (formerly the Canadian Guidelines on Sexually Transmitted Infections) are updated with the recommendations.
Conclusion
The methodology for developing the NAC-STBBI recommendations is evolving to meet best practice standards and will continue to be improved and refined as appropriate. The new methodology combined with the ongoing support and expert advice of NAC-STBBI will ensure that PHAC provides trustworthy evidence-based STBBI recommendations to primary care providers and public health professionals.
Authors' statement
- SS - Conceptualization, methodology, writing of original draft, review and editing
- SG - Conceptualization, writing of original draft, review and editing
- MGR - Conceptualization, review and editing
Competing interests
None.
Acknowledgements
The authors would like to thank current and former members of the National Advisory Committee on Sexually Transmitted and Blood-Borne Infections (NAC-STBBI) and all those who have contributed to the development of the STBBI guidelines. The authors would also like to thank U Auguste and CG Healy for reviewing the final version of the article.
Funding
The National Advisory Committee on Sexually Transmitted and Blood-Borne Infections is supported by the Public Health Agency of Canada.
