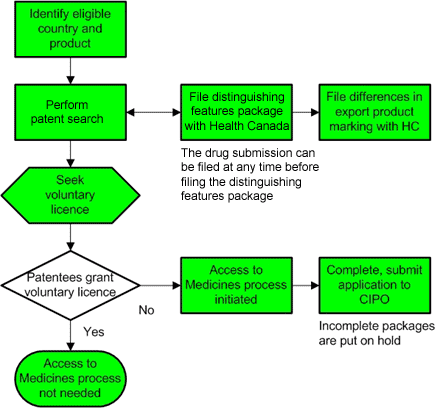
Process map for preparing to submit an application under Canada's Access to Medicines Regime
This process map provides a visual overview of the steps that need to be followed by a pharmaceutical company that is preparing to submit an application for a compulsory licence under Canada's Access to Medicines Regime. For details about these steps, return to Preparing to Submit an Application.