Annual compliance and enforcement report fiscal year: 2019-2020
On this page
- Acronyms
- Appendix A: Examples of product types in "product categories"
Acronyms
- CBSA
- Canada Border Services Agency
- CCPSA
- Canada Consumer Product Safety Act
- CPSP
- Consumer Product Safety Program
- CVI
- Compliance Verification Inspection
- ESA
- Electrical Safety Authority
- FDA
- Food and Drugs Act
- PROFECO
- Procuraduría Federal del Consumidor (Mexico's Office of the Federal Prosecutor for the Consumer)
- US CPSC
- United States Consumer Product Safety Commission
- XRF
- X-Ray Fluorescence
1. What we do
1.1 Mission
Contribute towards a safe marketplace for consumer products and cosmetics by preventing, detecting and responding to adverse health occurrences to Canadians.
1.2 Vision
To be a world leader in preventing, detecting and responding to health hazards and safety risks posed by consumer products and cosmetics.
1.3 Program description
The Consumer Product Safety Program (CPSP) is responsible for the administration and enforcement of the Canada Consumer Product Safety Act (CCPSA), and regulations made under it, as well as cosmetic-related provisions of the Food and Drugs Act (FDA) and the Cosmetic Regulations.
Health Canada promotes, monitors, verifies, and enforces compliance with the CCPSA and the FDA. The CPSP reviews reports submitted by industry and consumers and regularly monitors the marketplace to look for potentially dangerous products. The CPSP gathers information, both domestically and internationally, about injuries, emerging issues and new science related to consumer product safety. It also conducts routine sampling and testing of products in the marketplace and works closely with the Canada Border Services Agency (CBSA) and other domestic and international regulators to verify the compliance of products being imported into Canada.
The CPSP uses this information to identify possible risks to Canadians posed by consumer products or cosmetics, and conducts risk assessments to identify issues that may result in serious injury. This allows the CPSP to target the Program's compliance and enforcement resources towards products that pose the greatest potential risk to Canadians. The CPSP uses a triage-based approach to identify product-related health and safety issues for follow up compliance and enforcement activity. The Program monitors issues that do not require immediate attention to be prepared to take action if the risk changes over time.
Finally, the CPSP provides credible and reliable information that facilitates public education and provides tools for informed decision-making by the public.
The CPSP's authorities do not include products excluded from the CCPSA in Schedule 1, such as explosives, drugs, food, medical devices, ammunition, natural health products, and tobacco products, nor do they include products regulated under the FDA beyond cosmetics.
1.4 What compliance and enforcement activities we do
The CPSP's compliance and enforcement activities focus on promoting and verifying compliance, as well as on correcting non-compliances with regulatory obligations, prohibitions, product standards, and reporting and notification requirements.
1.5 How compliance and enforcement activities are done
In Canada, the safety of consumer products and cosmetics is regulated through a post-market approach. There is no pre-approval required for industry members to sell their consumer products or cosmetics in Canada; however, industry is responsible for ensuring the compliance of theirproducts with the applicable legislation, including that the consumer products and cosmetics they manufacture, import, sell, or advertise in Canada do not pose a danger to human health or safety.
The CPSP directs its resources to where the human health risks are greatest. This approach is similar to risk-based approaches used by our major trading partners. Tools used by the CPSP to identify and manage risks include:
- Planning targeted inspections;
- Sampling and testing products;
- Following up on incidents and complaints involving consumer products and cosmetics;
- Reviewing incidents, media articles and injury data from other sources (ex. hospitals, poison control centres, Canadian fire marshals,fire commisioners, etc.) to identify areas of concern;
- Undertaking risk assessments and hazard analyses to assess emerging hazards, identifying and assessing product trends, and establishing methodologies, approaches and priorities to assess these risks;
- Communicating the risks to Canadians;
- Reviewing records from establishments, such as test reports;
- Negotiating and, if necessary, ordering corrective measures such as stopping sale and recalling a product; and
- Working with our other provincial, territorial, federal, and foreign counterparts (such as the ESA, CBSA and the US CPSC, respectively) to help dentify and prevent the importation of non-compliant consumer products and cosmetics into Canada.
2. Executive summary
Health Canada's Consumer Product Safety Program (CPSP) helps protect Canadians by assessing the health risks and safety hazards associated with consumer products and cosmetics. From April 1, 2019, to March 31, 2020, the CPSP conducted the following compliance and enforcement (C&E) activities:
- Carried out 252 planned product-based inspections across 10 different product categories;
- Carried out five planned mandatory incident reporting inspections;
- Responded to 586 referrals from the Canada Border Services Agency (CBSA) leading to the refusal of 123 (21%) shipments;
- Reviewed 2,339 consumer complaints and incident reports (1,384 from industry, 955 from consumers);
- Identified a total of 302 different non-compliant products requiring corrective action, 227 of which resulted in a recall (170 recalls being a result of reported incidents and 57 recalls being a result of the CPSP product inspections);
- Carried out 4,306 monitoring inspections to ensure recalled products were not for sale; and
- Received and processed 54,455 cosmetic notifications.
3. Compliance and enforcement key activities
3.1 Following-up on consumer complaints and mandatory incident reports
The CPSP reviews reports submitted by industry and consumers to look for potentially dangerous consumer products or unsafe cosmetics.
Under section 14 of the CCPSA, industry must report to Health Canada after it becomes aware of a health or safety incident involving consumer products. An incident can include (among other things listed in CCPSA s.14(1)(a) to (d)):
- An occurrence, a product defect, or incorrect or a lack of information that resulted or may result in death or serious negative impacts on health; or
- A product recall in another jurisdiction based on concerns about human health or safety.
When reports are received by the CPSP, they are reviewed and triaged to determine if action is required to address the risk. Product returns or complaints from consumers will often trigger mandatory reporting requirements for industry, which leads to the submission of reports describing the incident and any corrective measure(s) that the company is implementing to address the health or safety concerns.
There is no incident reporting requirement for cosmetics under the FDA and Cosmetic Regulations, although the Program does encourage consumers and industry members to report when they have health or safety concerns related to a cosmetic.
In 2019-2020, 2,339 reports were received by the Program: 1,384 from industry and 955 from consumers. Table 1 shows the most frequently reported product categories for those submitted reports.
| Product category | Percent (%) |
|---|---|
| Appliances | 24 |
| Housewares | 19 |
| Children's Products | 14 |
| Home and Automobile Maintenance | 11 |
| Electronics | 11 |
| Grooming Products and Accessories | 8 |
| Outdoor Living | 5 |
| Clothing, Textiles and Accessories | 5 |
| Sports, Recreation and Hobby | 4 |
| Totals may not equal 100% due to rounding. | |
Of the 2,339 reports received, 170 (7%) resulted in the posting of a recall table 2 shows the most frequently recalled product categories.
| Product category | Percent (%) |
|---|---|
| Housewares | 18 |
| Clothing, Textiles and Accessories | 16 |
| Home and Automobile Maintenance | 13 |
| Children's Products | 13 |
| Electronics | 11 |
| Sports, Recreation and Hobby | 9 |
| Appliances | 9 |
| Outdoor Living | 8 |
| Grooming Products and Accessories | 3 |
| Totals may not equal 100% due to rounding. | |
Findings this year are similar to the previous year, except that children's products dropped by 7%, and sports, recreation and hobby dropped by 4%. Electronics went up by 5% and the clothing, textiles and accessories went up by 7%. Click here for more information on all recalls and safety alerts published by the CPSP.
For more information on data reported to the CPSP by consumers and industry members, please see the following links to both the Consumer Product Safety Program Annual Surveillance Reports as well as the Quarterly Surveillance Reports: Annual Reports, Quarterly Infographic. The Annual Surveillance Reports provides a yearly summary of all reports received by the CPSP. The Quarterly Infographics provide a quick snapshot of the overall numbers and types of reports received for a 3-month period (January to March; April to June; July to September; October to December).
3.2 Collaboration with the United States
Canada actively collaborates with the United States Consumer Product Safety Commission (US CPSC) on consumer product safety as both countries share similar distribution networks, general approaches to consumer product health and safety standards, and enforcement activities. Information gathered on products of concern in the United States is used by the CPSP to determine if compliance or enforcement activities should be initiated in Canada. When Canadian establishments with a presence in both jurisdictions are initiating a product recall, the CPSP reminds them of their reporting requirements to the US CPSC, and vice-versa.
In 2019-2020, 69 joint recalls were posted by Health Canada and the US CPSC. The most frequent product types jointly recalled and their relative percentages are shown in table 3.
| Product category | Percent (%) |
|---|---|
| Housewares | 32 |
| Children's Products | 17 |
| Electronics | 16 |
| Sports, Recreation, and Hobby | 13 |
| Appliances | 7 |
| Clothing, Textiles, and Accessories | 7 |
| Outdoor Living | 4 |
| Home and Automotive Maintenance | 1 |
| Grooming Products and Accessories | 1 |
| Totals may not equal 100% due to rounding. | |
There was an increase by 15% over the last year for joint recalls between Canada and the United States. Figure 1 illustrates the overall number of joint recalls over the past eight fiscal years.
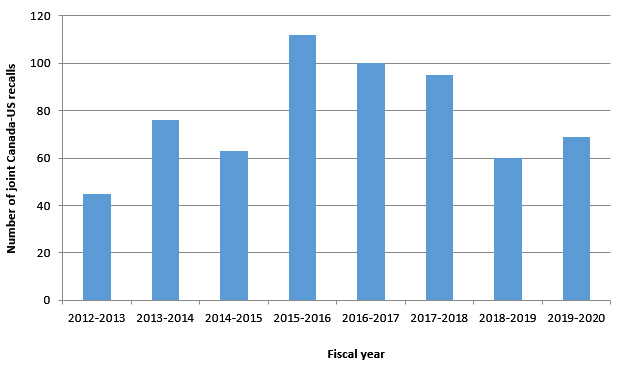
Figure 1: Text description
Graphic showing the number of joint Canada-US recalls from fiscal years 2012-2013 to 2019-2020. In 2012-2013 there were 45 recalls, in 2013-2014 there were 76 recalls, in 2014-2015 there were 63 recalls, in 2015-2016 there were 112 recalls, in 2016-2017 there were 100 recalls, in 2017-2018 there were 95 recalls, in 2018-2019 there were 60 recalls and in 2019-2020 there were 69 recalls.
The CPSP and the US CPSC continue to work towards posting recalls with Mexico's PROFECO. In 2019-2020, four recalls were posted jointly by all three regulators, compared to 2018-2019, where only 2 recalls were posted jointly.
3.3 Cosmetic notifications
Under section 30 of the Cosmetic Regulations, manufacturers and importers must notify Health Canada within 10 days after they first sell a cosmetic in Canada. Failure to notify may result in a product being denied entry into Canada or removed from sale. Over the course of 2019-2020, the Program received 54,455 cosmetic notifications, which is a slight decrease from the 56,755 notifications received in 2018-2019.
3.4 Targeted inspections
One of the key tools that the CPSP uses to support the goal of targeted oversight is planned, targeted inspections. The CPSP uses a risk-based approach to monitor and verify industry compliance with the regulations for consumer and cosmetic products on a cyclical basis. The sampling and inspecting for compliance is targeted to those regulated product categories and establishments where the information available to the Program suggests elevated levels of non-compliance. In many cases, inspectors can identify a higher probability of non-compliance based on previous market analysis or in the field using readily identifiable indicators, such as labelling or packaging issues, missing certification marks, or design issues that may lead to non-compliance. Due to this targeted approach, the CPSP expects higher levels of non-compliance than if the sampling and inspecting was carried out randomly.
3.4.1 Establishment inspections
The purpose of conducting inspections of Canadian establishments is to evaluate industry's ability to identify and report incidents to the CPSP and evaluate record-keeping practices. The results of these inspections help the CPSP assess how well a company's internal product safety quality system is functioning and informs how the Program can more effectively focus its resources. The CPSP's establishment inspections are evolving to include a stronger compliance education approach in hopes to improve industry compliance.
In 2019-2020, five Mandatory Incident Reporting inspections were completed to verify industry's compliance with the incident reporting requirements under section 14 of the CCPSA. Four additional inspections were delayed due to temporary Covid-19 business closures. Reporting on the delayed inspections numbers will be reflected in the next annual report.
3.4.2 Product inspections
The CPSP also conducts inspections targeted to specific product types to monitor and verify industry compliance with the CCPSA and the FDA. Compliance is verified by either conducting product sampling and testing, or by requesting documents, such as test reports or safety information, to evaluate compliance with the relevant legislative or regulatory requirements. Inspection projects are chosen every year based on multiple factors, such as emerging trends, the level of risk associated with a product, the type of product, previous levels of compliance for a product category, and the vulnerability of product users.
In 2019-2020, 10 product based projects for 10 different product categories were completed resulting in 252 inspections. In total, 337 products were assessed, leading to 132 enforcement actions, including 57 product recalls. Table 4 describes the 10 product categories and indicates the means by which compliance determinations were made and the corrective actions taken. For more details on the results of each project, see the Enforcement Summary Reports published on Health Canada's website. Publication of these reports is part of the Government of Canada's commitment to regulatory transparency and openness.
The product-based projects (Table 4) included in this annual report are those where the corrective actions for all products have been finalized by March 31, 2020. Other product-based projects that started in 2019-2020 but did not have corrective actions finalized by March 31, 2020 are not included. The results for these projects will be reflected in the next annual report.
| Start and completion date | Project | Number of inspections | Evaluation means (and applicable legislation) | Number of samples assessed | Findings of non-compliance (%) | Recall | Stop sale | Stop distribution | Trader commitment |
|---|---|---|---|---|---|---|---|---|---|
| Started in 2018-2019 and completed in 2019-2020 | CCCR, 2001 | 32 | Document review and sampling and testing (Consumer Chemicals and Containers Regulations, 2001) | 86 | 44 (51%) | 13 | 3 | 12 | 16 |
| Children's Sleepwear | 45 | Sampling and testing (Children's Sleepwear Regulations) | 92 | 30 (33%) | 16 | 3 | 3 | 8 | |
| Children's Jewellery | 32 | Sampling and testing (Children's Jewellery Regulations) | 22 | 5 (23%) | 4 | 1 | 0 | 0 | |
| Started in 2019-2020 and completed in 2019-2020 | Infant Feeding Bottle Nipples | 5 | Sampling and testing (Infant Feeding Bottle Nipples Regulations) |
4 | 0 | 0 | 0 | 0 | 0 |
| Pacifiers | 17 | Sampling and testing (Pacifiers Regulations) |
15 | 4 (27%) | 2 | 0 | 0 | 2 | |
| Expansion Gates | 14 | Sampling and testing (Expansion Gates and Expandable Enclosures Regulations, Surface Coating Materials Regulations) |
14 | 8 (57%) | 2 | 0 | 0 | 6 | |
| Toys - Small Parts/Boric Acid | 33 | Sampling and testing (Toys Regulations) |
30 | 9 (30%) | 8 | 1 | 0 | 0 | |
| Cribs and Cradles | 16 | Sampling and testing (Cribs, Cradles and Bassinets Regulations) |
16 | 16 (100%) | 0 | 1 | 7 | 8 | |
| Drawstrings | 39 | Onsite assessment (Canada Consumer Product Safety Act) |
39 | 16 (41%) | 12 | 0 | 0 | 4 | |
| Consumer Products Containing Lead (CPCL) - decals | 19 | Sampling and testing (Consumer Products Containing Lead Regulations) |
19 | 0 | 0 | 0 | 0 | 0 | |
| N/A | Total | 252 | N/A | 337 | 132 (39%) | 57 | 9 | 22 | 44 |
Based on the inspections projects done in 2019-2020, the following product-categories had the highest non-compliance levels:
- Cribs and Cradles with 100% non-compliance
- Expansion Gates with 57% non-compliance
- Products subject to the CCCR, 2001 with 51% non-compliance
- Drawstrings with 41% non-compliance
With respect to these product categories, high-risk non-compliances were identified, among other non-compliances, and a number of product recalls were required. In the cribs and cradles project, all products sampled were found to be non-compliant; the majority of these non-compliances were medium and low-risk non-compliances (for example, not meeting labelling requirements). No recalls were required this fiscal year for this product category. During the expansion gates project, two voluntary recalls (both for failing to meet the requirements for surface coatings) and six trader commitments were required to remedy the other identified non-compliances. The result of the project for expansion gates show some improvement in the compliance levels of the samples examined compared to fiscal year 2016-2017, which yielded a 0% compliance level.
As noted in table 4, a wide range of enforcement actions were taken with products subject to CCCR, 2001, to the Children's Sleepwear Regulations and to the Cribs and Cradles Regulations. Given the broad type and volume of products available for sale within these product categories, compliance projects are done regularly to verify compliance and educate industry on the relevant product requirements.
Overall, while working with industry to address all non-compliances, the CPSP continues to focus its more serious enforcement actions, such as recall, on products where a high risk has been identified.
Additional information on the Consumer Products Containing Lead (CPCL) decals Project
The lead limit of children's clothing and clothing accessories is regulated under the Consumer Products Containing Lead (Regulations) under the CCPSA. These Regulations were updated in 2018 to include new types of products such as decals on children's clothing and clothing accessories, child care articles and toys for older children. They came into force in May 2019.
Since decals on children's clothing and clothing accessories were not subject to the Regulations before May 2019, a project was undertaken for the first time in 2019-2020 to focus on decals on an assortment of children's clothing, clothing accessories and footwear for children under 7 years of age. This age range was chosen because younger children have a higher tendency to mouth items compared to older children. Compliance was determined by X-Ray Fluorescence (XRF) and wet chemistry testing, when required, at Health Canada's Product Safety Laboratory (PSL). Overall, the compliance level was determined to be 77%.
3.4.3 Recall monitoring inspections
Whether an establishment agrees to voluntarily recall a product or has been ordered to do so (for consumer products under the CCPSA only), it has the responsibility to contact ts supply chain accounts (the people to whom they sold the products) to ensure that the product is no longer offered for sale. Through recall monitoring inspections, the CPSP verifies that selected supply chain accounts have received the notification of the recall and that the necessary steps have been taken to remove the product from sale (including online sales); further minimizing the hazard in the Canadian marketplace. In 2019-2020, 4,306 recall monitoring inspections were conducted.
3.4.4 Import admissibility recommendations
The CPSP works with the CBSA to identify non-compliant products being shipped into the country. By stopping non-compliant products at the border, the CPSP is able to prevent these products from reaching the market, thereby reducing the potential for risk to Canadians. In 2019-2020, the CPSP received 586 referrals from the CBSA for shipments of products that were flagged as potentially non-compliant, 68% of which were for cosmetics. Of these referrals, 123 shipments were refused entry by the CBSA following recommendation from the CPSP; 79% of which involved cosmetic products. Shipments may be refused for a variety of reasons, including that the products are prohibited, or do not meet specific regulatory requirements.
4. Conclusion
The CPSP's compliance and enforcement activities described in this report focus on promoting and verifying compliance of consumer products with the CCPSA and cosmetics with the applicable requirements under the FDA, as well as on correcting non-compliances. From April 1, 2019, to March 31, 2020, the CPSP conducted 252 planned product-based inspections, responded to 586 referrals from the CBSA, reviewed 2,339 consumer complaints and incident reports and identified 302 different non-compliant products requiring corrective actions.
The CPSP will continue to implement its compliance and enforcement activities, supported by border oversight and international cooperation, to quickly identify and act on product safety risks while keeping Canadians informed about health and safety issues related to consumer products and cosmetics.
All information gathered from the activities conducted in 2019-2020 will be used to plan future years' compliance and enforcement activities. The information gathered through these compliance and enforcement activities helps the CPSP target existing and upcoming product safety risks as efficiently as possible.
5. Contact information
Any questions or comments on this report should be directed to hc.cps-spc.sc@canada.ca.
Appendix A: Examples of product types in "product categories"
- Appliances: Kitchen appliances; heating and cooling appliances; laundry and cleaning appliances.
- Children's Products: Nursery products; baby gear; toys.
- Clothing, Textiles, and Accessories: Clothing; household textiles; footwear.
- Electronics: Televisions and home theatres; electronic cables, batteries and chargers; computers and peripherals; cellphones and accessories.
- Home and Automobile Maintenance: Construction materials; tools.
- Housewares: Furniture; home décor; lighting; household cleaning; kitchenware.
- Outdoor Living: Outdoor furniture and decorations; pools and accessories; lawn and garden.
- Grooming Products and Accessories: Beauty and body care; beauty accessories; oral care.
- Sports, Recreation, and Hobby: Sports and outdoor activities; play structures; hobby or crafts.