Science Policy Note: Assessing Exposure from Pesticides in Food - A User's Guide
July 28, 2003
ISBN: 0-662-34734-X
Cat. No.: H113-13/2003-3E-PDF
(SPN2003-03)
Table of Contents
- Introduction
- A primer on pesticide exposure and risk from food
- Information sources: Where to find data, guidance, and other information on assessing exposure to pesticides in food
- Residue data and collection
- Magnitude of residue data for pesticides in foods
- Directive DIR98-02: Residue Chemistry Data Requirements
- CFIA monitoring data
- CGC monitoring data
- Health Canada monitoring data
- USDA Pesticide Data Program data
- U.S. FDA monitoring data
- Regulatory monitoring
- Incidence/level monitoring
- Total diet study
- U.S. market basket survey
- Using the FDA data: Decompositing
- U.S. state monitoring
- Quality assurance: Good laboratory practices
- Analytical Methods: Pesticide analytical manual
- Acute and chronic exposure estimates
- Consumption information
- Residue data and collection
- List of where to find information
- List of abbreviations
- References
- Glossary of terms
Introduction
The following policy document is intended to provide guidance and information to the Pest Management Regulatory Agency (PMRA) personnel and decision-makers and to the public. This document describes the processes used by PMRA scientists, in dietary risk assessments (DRA), to quantify the level of food residue exposures to consumers of all ages.
The PMRA has utilized, to the greatest extent possible, the policy and guidance outlined in the United States Environmental Protection Agency (U.S. EPA) document, Available information on assessing exposure from pesticides in food--a user's guide (U.S. EPA 2000). This was done, in part, to harmonize dietary risk assessment procedures for determination of the safety of pesticide residues in domestic and imported treated foods.
This endeavour, to harmonize DRA methodologies, is part of the North American Free Trade Agreement (NAFTA) goals within the Pesticides Technical Working Group Subcommittee.
The U.S. EPA has taken the lead in developing science policies related to the U.S. Food Quality Protection Act (FQPA). Harmonization of these policies between our agencies has been key to our ability to do joint reviews. Such policies play an increasingly important role in the evaluation and assessment of risks posed by pesticides, and improve the regulator's ability to make decisions that fully protect public health and sensitive subpopulations. These policies are vetted by the NAFTA Technical Working Group on Pesticides and have been approved for adoption, only after extensive consultation by scientific experts from governmental, academic and all nongovernmental interested parties. The consultation process utilized by the PMRA for science policy notices is outlined in a memo entitled: Memorandum to Registrants, Applicants and Agents, (January 25, 2001) and may be obtained from the PMRA web site at: http://www.hc-sc.gc.ca/pmra-arla/english/pdf/fqpa/fqpa_memo-e.pdf.
The PMRA is responsible for regulating the nature and amount of pesticide residues in food under the Food and Drugs Act and Regulations (FDAR). Section 4(a) and 4(d), of the Food and Drugs Act (FDA), authorizes PMRA to set a maximum residue limit (MRL) within Regulation B.15.002(1) of the Food and Drugs Regulations (FDR) or an exemption from the requirement of a MRL under Regulation B.15.002(2) of the FDR. The Agency performs various types of risk assessments to evaluate the safety of pesticides in food, including analyses to determine the nature and the amounts of pesticides that people might be exposed to over a single day. This paper discusses how the PMRA generally applies the statutory safety standard to dietary risk assessments for pesticide residues in foods.
The PMRA performs DRAs that include estimations of human exposure to pesticide residues in foods over a single day and lifetime exposures. These estimates require the use of magnitude of residue (MOR) data to calculate and quantify the degree to which humans will be exposed to residues from the use of pesticides approved for use in the registration and reevaluation/special review programs.
Exposures are determined for general and regional populations, as well as many subpopulations (infants, children, teenagers, adults, seniors, etc.), some of which may require MORs for specialized foods. The use of a pesticide on food is not supported for use in Canada, unless the DRAs are acceptable.
In addition to MOR data for domestically grown foods, MOR residue data are required for imported foods, since large amounts of foods consumed in Canada are imported from foreign countries.
The PMRA regulates pesticides to ensure that their use does not pose unreasonable risks to human health or the environment and that exposure to pesticide residues in food is safe. These determinations rely on the process of risk assessment. In assessing risk, the Agency considers all sources of exposure (e.g., food, drinking water, incidental exposure in and around the home, school, etc.) and the inherent toxicity of the pesticide.
The purpose of this user's guide is to provide the reader with a comprehensive discussion of guidance documents, policy documents, and databases that provide detailed, specific "how-to" information on assessing exposure to pesticides from the foods that we eat. To help the reader understand the context of this information, this guide first provides a basic overview of risk assessment for exposure resulting from pesticide residues in food.
As noted above, the PMRA has harmonized DRA methodologies for the determination of exposure of food residues, with those used by the U.S. EPA. While the Agency prepares PMRA versions of currently available U.S. EPA policy/guidance documents concerning DRA, the reader is referred to U.S. EPA documents, using the "Where to Find"-further information captions.
This guide does not address aggregate exposure and risk assessment, which is the process of combining exposure to a single pesticide from all sources of exposure: food, drinking water, and through nonoccupational sources such as homes and recreational areas. Furthermore, this guide does not address cumulative risk assessment, which is the process of combining exposure and risk from all pesticides with a common mechanism of toxicity.
The first section, "A Primer on Pesticide Exposure and Risk from Food", provides a very simple overview of the PMRA's approach to estimating risk and exposure from pesticide residues in food. Further general information on risk assessment and risk management, employed within the PMRA, may be found in the document: PMRA Guidance Document, A Framework for Risk Assessment and Risk Management at the Pest Management Regulatory Agency.
The following section, "Information Sources: Where-to-Find Data, Guidance, and Other Information on Assessing Exposure to Pesticides in Food", provides specifics on how to obtain or generate the data and/or information the PMRA uses in its assessments of exposure and risk from pesticides in food. The final section of this User's Guide provides a bibliography and glossary of terms; words shown in boldface type are defined in this Glossary at the end of this document.
A primer on pesticide exposure and risk from food
The risk that is posed by a pesticide in or on food depends on the toxicity of the pesticide and the amount of pesticide to which a person is exposed; this is expressed mathematically by the equation:
Risk = f (toxicity, exposure)
which in words means risk is a function of toxicity and exposure. More simply stated, risk is equal to toxicity multiplied by exposure. A pesticide with low toxicity and high exposure could pose a similar risk as a pesticide with high toxicity and low exposure.
Exposure and risk at a glance
To determine whether any risk can result from either short-term (i.e., acute) or longer term (i.e., chronic) exposure, one considers both the toxicity of the pesticide (which is sometimes referred to as hazard) and the amount of pesticide to which an individual may be exposed.
In the actual risk equations, which are discussed later on, toxicity is expressed as: an acute reference dose (ARfD) an acceptable daily intake (ADI) and a potency factor for cancer called the q*. Which toxicity expression the risk assessor uses depends on the duration of exposure (e.g., acute or chronic) and, in the case of a carcinogen, the method chosen for quantifying risk.
The risk posed by carcinogens can be quantified using an equation that assumes the pesticide's toxic effect occurs via a linear response, or it can be calculated using an equation that assumes a nonlinear response.
The amount of pesticide to which an individual is exposed (i.e., exposure) is determined by combining the amount of pesticide that is in or on the food (i.e., residue levels) and the amount and type of foods that people eat (i.e., food consumption).
Risk is estimated using a computer model that combines the toxicity, residue, and consumption information. This is further described in the "Risk" section below.
The rest of this Primer is divided into three sections: toxicity, exposure, and risk. Each elaborates on the principles introduced in this section. First, aggregate and cumulative assessments will be discussed.
Aggregate and cumulative assessments
The PMRA conducts aggregate exposure assessments, where all sources of exposure are considered in the risk assessment. These include exposures from:
- pesticide residues in food;
- pesticide residues in drinking water; and
- pesticide residues encountered through nonoccupational sources such as in the home, home turf uses, recreational areas, and schools.
This document only addresses exposure from pesticide residues found in food. Many other guidance and policy documents are under development for assessing exposure from foods, drinking water and conducting aggregate and cumulative assessments.
Toxicity
Risk = f (toxicity, exposure)
Noncancer endpoints
The PMRA assumes that noncancer toxicity endpoints exhibit a nonlinear response. In assessing risk resulting from exposure to pesticide residues in food, the toxicity for such effects is expressed as a reference dose (RfD). The RfD can be thought of as an amount of toxicant to which a person can be safely exposed to on a chronic lifetime exposure (ADI) or on a short term acute exposure (ARfD).
The RfD is calculated by dividing the dose in laboratory animals at which no harmful effects are observed by uncertainty and safety factors; these are discussed below. Separate reference doses are calculated for both acute and chronic effects. These calculations are shown just after the "Additional Safety Factor" section.
Safety and uncertainty factors
In determining acute and chronic reference doses, the respective No Observed Adverse Effect Levels (NOAELs) are divided by safety and uncertainty factors.
| Condition for safety and uncertainty factor | Magnitude of the factor | When factor is applied |
|---|---|---|
| Accounting for the potential variation within the human population (intraspecies) | 10-fold | Typically |
| Accounting for the potential differences between humans and animals as the animal data are translated to humans (interspecies) | 10-fold | Typically |
| Accounting for a gap in the toxicity database (i.e., a key study is missing) | 3-fold to 10-fold | When the nature of the toxicity database indicates its need |
| If a lowest observed adverse effect level (LOAEL) is used instead of a NOAEL |
In total, the safety and uncertainty factors applied to the NOAEL can range from the typical 100-fold inter- or intra-species factor to over 3000-fold for a pesticide where there are substantial concerns regarding the nature of toxicity database.
Additional safety factors
The use of an additional safety factor to assure the safety of infants and children reflects a similar policy used by the U.S. EPA, where this policy is an outcome of their FQPA. In determining acute and chronic reference doses, the PMRA conducts a case-by-case review of each chemical to determine whether an additional default 10-fold safety factor should be applied or whether another factor adequately protects infants and children. In addition, the PMRA may employ the use of additional safety factors to account for the severity of the endpoint. A sliding scale of concern is used, reserving higher safety factors (i.e., 10-fold) for endpoints that are severe, life-threatening and/or irreversible (e.g., mortality, malformations, failure to produce viable offspring, etc.) with lower safety factors for other serious endpoints (e.g., endocrine disruption, immunotoxicity, etc.).
Equations for acute effects
For acute (noncancer) toxicological effects (e.g., cholinesterase inhibition, which can occur following only one day of exposure), the toxicity portion of the risk equation is expressed as an ARfD. It is calculated as follows:
ARD = NOAEL / Uncertainty×Safety Factors
An ARfD is an estimate of the level of one-day exposure to a pesticide residue that is believed to have no significant deleterious effects. It is calculated by first determining the NOAEL from acute animal toxicity studies and dividing it by the appropriate uncertainty and safety factors. The ARD that the PMRA generates is equivalent to the acute Population Adjusted Dose (aPAD) that is generated by the U.S. EPA.
Equations for nonacute (chronic) effects
For toxicological effects that occur after repeated exposures (e.g., those effects that occur after exposure lasting a significant portion of the life-span), the toxicity portion of the risk equation is expressed as a chronic reference dose called the acceptable daily intake (ADI). It is calculated as follows:
ADI = NOAEL / Uncertainty×Safety Factors
A chronic reference dose is an estimate of the level of daily exposure to a pesticide residue, which, over a 70-year life-span, is believed to have no significant harmful effects. The ADI that the PMRA generates is equivalent to the chronic Populated Adjusted Dose (cPAD) that is generated by the U.S. EPA.
Cancer endpoints
Linear
For carcinogenic effects that are thought to occur through a linear response, the toxicity portion of the risk equation is expressed as a cancer potency factor, more commonly known as a q1*. A q1* is the relative strength of a carcinogen. Mathematically, it can be thought of as the slope of the dose-response curve. In reality, a q1* is a single number that is calculated from animal data using a sophisticated computer model that assumes linearity at low doses. The higher the q1* value, the more potent the chemical is as a carcinogen.
Nonlinear
For carcinogenic effects that are shown to exhibit a nonlinear response, the toxicity portion of the risk equation is expressed as Point of Departure or PoD. A PoD is simply the toxic dose that serves as the "starting point" in extrapolating a risk to the human population. The PoD can be either an observed dose (e.g., NOAEL) or it can be an interpolated value. Quite often, the PoD is equivalent to the NOAEL.
Exposure
Risk = f (toxicity, exposure)
The PMRA considers risks from "aggregate" exposures to a pesticide when establishing a pesticide MRL (maximum residue limit, i.e., the legally permitted level of a pesticide in a food). This means, that in addition to considering the exposures from food, the Agency must also consider other exposures for which reliable data are available. These include exposure from drinking water and nonoccupational sources such as pesticides used in and around the home, recreational areas, etc. This paper does not address aggregating exposures; it only discusses exposure from food.
Estimates of exposure from food are derived from two distinct pieces of information: the amount of a pesticide residue that is present in and on food (i.e., the residue level) and the types and amounts of foods that people eat (i.e., food consumption).
The amount of pesticide residue is termed the magnitude of the residue (MOR), and it is used to define the concentration permissible legally as an MRL based on residue studies performed according to the use pattern registered or to be registered. The MOR is also the concentration of any residue determined as part of monitoring residue data or of other residue chemistry studies, including: residue decline, residue degradation and many other scientific studies noted in the Residue Chemistry Guidelines (PMRA 1998a).
The exposure residue level is primarily developed from:
- the numerous crop field trials and monitoring programs (e.g., CFIA, CGC, PDP, U.S.FDA, EU) where the amount of pesticide residues on a given commodity is measured;
- use of information such as the percentage of a crop that is treated (percentage of crop treated or %CT); and
- commercial and consumer practice information such as washing, cooking, processing, and peeling practices.
Consumption information comes from the USDA's Continuing Survey of Food Intake by Individuals (CSFII), which provides survey data of what people eat in the United States (U.S.) and Canada.
Calculating the residue levels
Residue levels for use in acute exposure assessment are estimated a bit differently from residue levels that are to be used in chronic exposure assessments. In an acute exposure assessment, the risk assessor is attempting to estimate how much of a particular pesticide residue might be consumed in a single day. Acute exposure calculations tend to employ a full range of data including high-end residue values, high-end consumption and high-end % CT estimates. For a chronic exposure assessment, the risk assessor is attempting to estimate how much of a given pesticide residue might be consumed on a daily basis over the course of a lifetime. Consequently, the risk assessor tends to use average residue values, average consumption values, and average % CT estimates.
Consumption
Food consumption data are provided by USDA from its CSFII. The USDA has been conducting such food surveys since the 1930s by means of personal interviews in which interviewers ask individuals, who are selected statistically, to recall everything they ate and drank over the previous 24 hours.
In the late 1970s, Health Canada (HC) and the USDA conducted National Food Consumption Surveys, which were large and comprehensive surveys that sampled thousands of households to learn about what, and how much, people ate.
Over the course of the last 20+ years, people's dietary habits have changed, and the public health community has become more concerned with the unique patterns of children's exposure to pesticides through their diets. In 1993, the U.S. National Academy of Sciences (NAS) raised the concern that current food consumption data do not provide sufficient sample sizes to adequately estimate exposure to pesticide residues in the diets of children (NAS 1993). In 1996 the FQPA directed the USDA to "conduct surveys to document dietary exposure to pesticides among infants and children".
As a result of these concerns and changes in dietary habits, the U.S. EPA and the USDA have been working to update the food consumption information by periodically conducting the CSFII. This food survey data is also used by the PMRA since Canadian and American eating habits have been shown to be similar if not identical. In the next several months, the PMRA and the U.S. EPA will start using the latest CSFII information--that of a 1998-2000 survey, including 1998 data collected through a Children's Supplemental Survey, which was conducted to collect more information on what infants and young children eat. Where-to-Find food consumption information is listed in "Information Sources".
Risk
Risk = f (toxicity, exposure)
The basic algorithm or equation used to calculate risk resulting from exposure to pesticide residues in food depends on the duration of exposure (i.e., acute, chronic) and the type of response--linear or nonlinear. The PMRA assumes that all noncancer endpoints exhibit a nonlinear response. The basic equations used to calculate dietary risks are provided below. The actual dietary risk estimates are calculated using a sophisticated computer model that uses these algorithms. It is described below.
Tool for calculating risk
Risk resulting from exposure to pesticide residues in food, be it from acute exposure or chronic exposure, is calculated using a sophisticated computer software tool that combines food exposure data (both residue levels and % CT, and consumption) with toxicity to produce a risk value. The backbone of this model is USDA's food consumption survey information.
The model yields risk values for the general Canadian/U.S. population and 22 population subgroups, including infants, children, and nursing women. It has the ability to determine which crop/pesticide combinations contribute the highest exposures and, in turn, risks. Also, it can run probabilistic analyses for acute risk assessments.
The PMRA is currently using a model that was developed by Novigen Sciences, Inc. (2000); it is called the Dietary Exposure Evaluation Model or DEEM™.
Basic risk equations for noncancer endpoints
Provided below are the basic equations that are used to estimate risk resulting from exposure to pesticide residues in food for noncancer endpoints. The PMRA assumes that noncancer toxicity endpoints exhibit a nonlinear response.
Acute risk
Acute food risk is expressed as a percentage of the ARfD. If the calculated % ARfD is less than 100, the risk is generally considered to be acceptable.
% ARD =
Food Exposure(mg of residue/kg body weight/day) / ARD (mg residue/kg body weight/day)
× 100
Chronic risk
Chronic food risk is expressed as a percentage of the ADI. If the calculated % ADI is less than 100, the risk is generally considered to be acceptable.
% ADI =
Average Food Exposure (mg residue/kg body weight/day) / ADI (mg residue/kg body weight/ day)
×100
Basic risk equations for cancer endpoints
Cancer Risk =
Average Food Exposure (mg of residue/kg body weight/day) ×
q1* (mg of residue/kg body weight/day)-1
Linear
Linear cancer risk is expressed as a probability. For example, a calculated risk of 1 × 10-6 means that a person receiving a lifetime exposure to the pesticide increases his or her chance of developing cancer by one in a million. That is, for every one million exposed persons, one would expect, at the most (upper boundary), one more cancer than would otherwise occur, and it may be less. This probability is calculated using the relationship:
Nonlinear
Nonlinear cancer risk is calculated using the MOE approach where a margin of exposure (MOE) would be calculated. For nonlinear cancer risk assessment, the PMRA has not yet determined an appropriate target MOE. It is currently developing criteria by which to make that judgment.
The target MOE is a value incorporating safety factors and uncertainty factors, above which a calculated MOE is not considered to be of concern.
Information sources: Where to find data, guidance, and other information on assessing exposure to pesticides in food
Estimating exposure from pesticide residues found in or on food is a complex process. First, data must be obtained. The PMRA requires registrants to generate a large body of scientific data. The risk assessor gathers other information such as percentage of the crop treated and how the pesticide is used from existing sources. Then, the data must be evaluated by Agency scientists and transformed into exposure estimates.
The following sections provide a comprehensive discussion regarding the sources of these data and other information. The first part primarily discusses sources of actual pesticide residue data, while the second part presents sources of information on calculating acute and chronic exposure estimates using the gathered data.
Residue data and collection
Magnitude of residue data for pesticides in foods
The PMRA performs DRAs that include estimations of human exposure to pesticide residues in foods over a single day and lifetime exposures. These estimates require the use of MOR data to calculate and quantify the degree to which humans will be exposed to residues from the use of pesticides approved for use in the registration and reevaluation/special review programs.
Exposures are determined for general and regional populations as well as many subpopulations (infants, children, teenagers, adults, seniors, etc.), some of which may require MORs for specialized foods. The use of a pesticide on food is not supported for use in Canada, unless the DRAs are acceptable. In addition to MOR data for domestically grown foods, residue data are required for imported foods since large amounts of foods consumed in Canada are imported from many foreign countries.
Assessing the level of pesticide residue that is in or on the foods that we eat--for both fresh raw foods such as lettuce and apples and processed foods such as frozen french fries and canned beans--is a complex process that requires data from numerous sources. The registrants are required under the Pest Control Products Act (PCPA) to generate the basic residue data (i.e., the crop field trial data, which are discussed later). The PMRA obtains other data, which are often used to refine the basic residue data, from provincial and federal monitoring programs, other government sources, and voluntary submissions from registrants or other entities such as grower groups. Residue data sources are discussed later in this document.
Directive DIR98-02: Residue Chemistry Data Requirements
Under the authority of the PCPA, the PMRA requires registrants to submit a full battery of residue chemistry data that reflect pesticide residue concentrations in food and feeds. These data are used to estimate the Canadian population's level of exposure to pesticide residues in food and to set and enforce MRLs for pesticide residues in food.
Results of these studies provide the PMRA with, among other things, the information it needs to determine:
- the nature of the residue (i.e., what are the metabolites; how is the pesticide broken down by the plant or in livestock that is used for food), and
- the amount of the residues in food or feed (this is termed the MOR).
These crop field trial residue data may be considered "worst-case" because the testing guidelines require that the pesticide under investigation be applied at the maximum application rate using the maximum number of applications and the minimum preharvest interval (PHI). These worst-case residues reflect the most extreme use pattern allowed on the label. Because actual use (in practice) can be significantly less than label maximums, and for other reasons, the residue levels encountered by the consuming public are likely to be much lower.
Residue data are available from several sources including: residue trials submitted in support of registration and reevaluation programs, monitoring and surveillance programs and calculated anticipated residues.
Factors that may be considered in planning the types and numbers of samples to collect include:
- review of residue data provided to the PMRA in support of registration of pesticide uses on food,
- regional intelligence on pesticide use,
- dietary importance of the food,
- information on the amount of domestic or imported food that enters the Canadian food supply,
- absence of existing residue data for specific pesticide/commodity combinations,
- toxicity of the pesticide,
- the total potential exposure, and
- production volume/pesticide usage patterns.
CFIA monitoring data
To contribute to the safe supply of food to the Canadian consumers, the CFIA's chemical residue program monitors domestic and imported fresh and processed fruits and vegetables for approximately 270 pesticides. Meat, milk and eggs are also monitored for pesticide residues on a routine basis. The CFIA's chemical residue programs are similar to those described below, for the U.S. EPA and the U.S. FDA.
The monitoring program is conducted in support of setting MRLs, to discern residue trends, to respond to international commitments, to identify potential problem areas for surveillance activities and to assess the effectiveness of control programs.
The CFIA's chemical residue program is divided into three phases: monitoring, surveillance and compliance.
Monitoring or data gathering is designed to provide profile information on the occurrence of chemical residues and food additives in predefined sampling populations. Monitoring information is normally obtained through a statistically based selection of random samples from normal appearing populations. The sampled lots are not held and are passed into consumer channels before the results are known. No direct enforcement action is taken on the basis of monitoring alone.
Surveillance is designed to investigate suspected problems of potential violations to the FDR and/or health risk problem, suggested in the monitoring program and is directed at targeted populations (e.g. commodity or geographical area). If warranted, product is detained until test results indicate the appropriate course of action. The sampling approach to surveillance is referred to as biased or directed sampling, and follows Codex Alimentarius specifications. Violative results found during the surveillance phase that are subject to further compliance action must be verified by prescribed confirmatory techniques before any follow-up control action is taken. Education is an important element in correcting problems once identified.
Compliance action is taken as a regulatory control measure to prevent the marketing or remove from the market a product known to be contaminated or adulterated. The compliance action is always directed at a specific source. The product is detained until the test results indicate the appropriate disposition. The sampling approach for compliance testing is referred to as in-depth sampling and follows Codex Alimentarius specifications.
CGC monitoring data
The Canadian Grain Commission (CGC) monitors Canadian cereal grains, oilseeds and pulse crops for the presence of toxic substances such as pesticide residues. This is one of major aspects of the CGC program for safety assurance of Canadian grain. CGC grain safety monitoring data are shared with Health Canada for risk assessment purposes.
Chemical testing for pesticides in Canadian grain is carried out by the Commission's Grain Research Laboratory (GRL). The GRL employs a wide variety of analytical methodologies in order to target the wide range of pesticides of interest and concern to domestic and foreign customers.
Screening of export shipments allows the CGC to determine what pesticides occur as residues in Canadian grain and the levels that may arise in commercial shipments. The CGC provides pesticide residue testing services to grain marketers, processors and foreign importers for cargo certification and other safety assurance purposes.
Health Canada monitoring data
Total diet Studies
Market basket surveys are conducted in many countries as a means of determining the actual dietary exposure of the population to contaminants and pesticides as a result of the ingestion of food. These studies provide data that differ from regular surveillance data in that the food is cooked and prepared as for consumption, resulting in a more accurate reflection of exposure. Such surveys have been conducted in Canada by HC since 1969, albeit in different forms. The most recent study involved the preparation and analysis of 136 food composites representing 99% of the Canadian diet from each of six cities over a period from 1992 to 1996. Raw agricultural commodities (RACs) and processed commodities, including milk and soy-based infant formula have been analyzed. These studies are carried out as part of Health Canada's Food Directorate's programs, concerning the safety of foods.
Supplemental-Corroborative MOR data
Since many of Canada's food exports go to the U.S. and much of the food imported into Canada comes from the U.S., monitoring residue data, market basket surveys and total diet survey data generated in the U.S. is utilized by the PMRA in its dietary risk assessments to obtain accurate exposure estimates for pesticides in foods as consumed by humans. This approach has the added advantage of accessing other residue databases that cover Canadian domestic treated foods and imported treated foods from outside or within the U.S. The following MOR data are used for this purpose.
USDA Pesticide Data Program data
The USDA (United States Department of Agriculture) started the Pesticide Data Program (PDP) in May 1991 to collect data on pesticide residues in food; to date it has published its findings for calendar years 1991-1998. The PDP's sampling procedures were designed to capture residues in the food supply reasonably close to the time of consumption. The PDP has tested about 40 different commodities including fresh/frozen/canned fruit and vegetables, fruit juices, whole milk, grain, and corn syrup for more than 160 different pesticides.
The PDP continues to focus on the NAS's conclusions as indicated in the 1993 report Pesticides in the Diets of Infants and Children (NAS 1993). In that report, the Academy recommended that pesticide residue monitoring programs target foods highly consumed by children and that analytical testing methods used be standardized, validated, and subject to strict quality control and quality assurance programs (QA/QC). Consequently, since 1994, the PDP has modified its commodity testing profile to include not only fresh fruits and vegetables but also canned and frozen fruits/vegetables, fruit juices, whole milk, wheat, soybeans, oats, corn syrup, peanut butter, and poultry.
The PDP pesticide monitoring activities are a federal-state partnership, whereby 10 participating states, which represent about 50% of the U.S. population and all regions of the U.S., collect samples of fruit, vegetables, and other commodities. These samples are collected close to the point of consumption--at terminal markets and large chain store distribution centers immediately prior to distribution to supermarkets and grocery stores. This allows the capture of sample identity, takes into account pesticide degradation during transit and storage, and provides data on residues resulting from postharvest applications of fungicides and growth regulators.
The number of samples to be collected is apportioned according to state population or commodity production figures. Samples are randomly chosen without regard for commodity origin or variety. They reflect what is typically available to the consumer throughout the year. The PDP's statistically reliable sampling protocol is designed to select random samples that best represent pesticide residues in the food supply to allow for a realistic estimate of exposure to these chemicals. In addition, the PDP also conducts special surveys on single-serving sized food items to support acute exposure assessment.
Note that, even though the PDP samples are collected at a point in the channels-of-trade where residues are close to those at consumption, these are not "grocery store" or "dinner plate" levels, where residues may be reduced even further.
The PDP data are the PMRA's preferred monitoring data for use in assessing risk for exposure resulting from pesticide residues in food. The sampling protocol was developed in cooperation with the PMRA; the sampling frame is statistically designed to be representative; and the data generated are specifically designed to be used for risk assessment. Also, the Limits of Detection (LOD) are low and a significant number of samples are collected over multiple years.
Using the PDP data: Translating to other crops
The PDP data have been collected for about 40 different food crops. There are hundreds of food crops for which the PMRA conducts assessments for exposure resulting from pesticide residues in what we eat. To expand the utility of the PDP data beyond the 40 or so crops that are sampled, the PMRA has established a policy on translating PDP data to other similar crops when certain conditions are met.
Using the PDP Data: Decompositing
When a chemist in the laboratory is trying to determine the concentration of a pesticide residue in a particular commodity (e.g., apples), they often do this using a composite sample. In assessing acute exposure using a probabilistic technique, the pesticide concentration is needed in terms of an individual item such as an apple, not in terms of an average value for a large number of apples. The reason for this has to do with the nature of probabilistic analysis and what people actually eat.
To get such individual item residue data, the chemist could perform a residue analysis for each single apple. Alternatively, this individual item data could be ascertained through statistical adjustment of the composite data. This process uses statistical procedures for decompositing composite residue data into individual item data (i.e., residues for one apple vs. residues for a 5-lb (2.3 kg) sample of apples).
This methodology (see box on the next page), when applied, permits the use of monitoring data from PDP, U.S. FDA, and CFIA (which are collected and analyzed as composite samples) to be "statistically adjusted" such that they could be used in acute probabilistic exposure assessment.
Provided below are the sites for the original draft paper that describes the principle of decompositing in general and compares the earlier proposal with the additional methods.
U.S. FDA monitoring data
The U.S. FDA (United States Food and Drug Administration) operates an ongoing program of monitoring for pesticide residues in foods; the results of the program are published annually. The program consists of three components:
- regulatory monitoring, surveillance and compliance;
- incidence/level monitoring; and
- the Total Diet Study (TDS).
Regulatory monitoring
The U.S. FDA's regulatory monitoring program is directed toward enforcing tolerances in imported foods and in domestically produced foods shipped in interstate commerce. Under regulatory monitoring, U.S. FDA samples individual lots of domestically produced and imported foods and analyzes them for pesticide residues. Domestic shipments are collected as closely as possible to the point of production in the distribution system; import samples are collected at the point of entry into U.S. commerce. Emphasis is on the raw agricultural product, which is analyzed as the unwashed, whole (unpeeled), raw commodity. Processed foods are also included.
Domestic and imported food samples are collected for analysis. Most monitoring samples are collected at random by the U.S. FDA. Compliance samples (and in Canada, surveillance samples) are collected as follow-up to the finding of an illegal residue or when there is other evidence of a pesticide problem.
To analyze the large numbers of samples (which are collected and prepared as composites) for which the pesticide treatment history is usually unknown, analytical methods capable of simultaneously determining a number of pesticides are used. These multiresidue methods (MRMs) can detect and quantify about half of the approximately 400 pesticides with U.S. EPA tolerances, as well as many others that have no tolerances. The most commonly used MRMs can also detect many metabolites, impurities, and alteration products of pesticides. Single residue methods (SRMs) or selective MRMs are used to determine pesticides not covered by an MRM. An SRM usually measures one pesticide; a selective MRM measures a relatively small number of chemically related pesticides.
Incidence/level monitoring
A complementary approach to regulatory monitoring, known as incidence/level monitoring, has been used to increase the U.S. FDA's knowledge about particular pesticide/commodity combinations by analyzing certain foods to determine the presence and levels of selected pesticides. For example, from 1995 to 1997, a survey of triazines was done.
Total diet study
The U.S. FDA conducts an annual TDS program that provides data on pesticide residue levels that are present in table-ready foods. Because the study has been under way for more than 30 years, trends can be discerned, such as the decrease in dietary levels of the once-registered pesticide, DDT, and related residues.
As part of the TDS, U.S. FDA staffers shop in supermarkets or grocery stores four times a year, once in each of four geographical regions of the country. Shopping in three cities from each region, they buy the same 259 foods, including meat, selected from nationwide dietary survey data to typify the American diet. The purchased foods are called "market baskets".
Foods from the market baskets are then prepared as a consumer would prepare them. For example, a "beef and vegetable stew" is made from the collected ingredients, using a standard recipe. The prepared foods are analyzed for pesticide residues, and the results, together with USDA consumption studies, are used to estimate the dietary intakes of pesticide residues for 14 age-sex groups ranging from 6-month-old infants to 70+-year-old men and women.
The analytical methods used in the TDS are modified to permit measurement at levels 5-10 times lower than those normally used in regulatory monitoring. In general, residues present at or above one part per billion (ppb) can be measured.
U.S. market basket survey
As noted earlier for the Canadian MBS (Market Basket Survey), this type of survey is a study in which the level of pesticide residues in foods as purchased is measured. Market basket data are intended to characterize the difference between the level of the residue that is found on commodities in the field and the residues that remain on foods at the time of purchase by the consumer. MBSs make use of statistically defined sampling procedures. Generally, samples are collected at the point of sale to the consumer (e.g., supermarkets or convenience stores). Samples may be prepared as if for consumption (e.g., peeled or washed) but are not cooked as discussed above in the TDS program. In addition, these studies focus on analysis of single commodity servings such as apples, peaches, bananas, etc., whereas the TDS analyzes composited samples for these types of commodities.
Using the FDA data: Decompositing
As discussed in the section "Using the PDP Data: Decompositing", the decomposition technique also applies to CFIA and USFDA data, as appropriate. Please refer to that section for further information.
U.S. state monitoring
A few U.S. states (e.g., California and Florida) collect their own pesticide monitoring data. When these are available, they may be used by the PMRA in food exposure assessments related to foods imported into Canada from these U.S. states.
Quality assurance: Good laboratory practices
It is critically important to the functioning of the PMRA's pesticide regulatory system that the Agency and the public are able to trust the data on which decisions are based. Therefore, the PMRA has programs to assure that data submitted to the Agency in support of product registrations are reliable. For example, the PMRA establishes detailed guidelines describing how studies must be performed. In addition, the laboratories conducting the studies must follow the Good Laboratory Practices (GLP) Regulatory Directive DIR98-01 (PMRA 1998b).
The GLP Standards are a management tool to ensure that studies are conducted according to certain scientific standards. Each laboratory conforms with GLP requirements by implementing standard operating procedures (SOPs) and maintaining QA oversight through a QA/QC Unit that conducts internal audits of raw data and laboratory practices.
The mission of the PMRA's GLP program is to assure the quality and integrity of studies submitted to the Agency in support of pesticide product registration. The PMRA accomplishes this mission by conducting data audits to assure compliance with the GLP directive yearly. These studies that are being audited vary from chemical analyses of pesticides to long-term toxicity and carcinogenicity studies in mammals. Other audited studies may look at the effects of pesticides on the environment, residues of pesticides on commodities and the efficacy of public health antimicrobial products.
Once the Agency receives data supporting registration (e.g., residue chemistry, product chemistry, toxicology and environmental fate/effects data), scientists from appropriate scientific disciplines thoroughly review the data.
These reviews look not only a the substantive results, but also look for signs that the data may not be trustworthy, e.g., internal inconsistencies, discrepancies with tests run on similar products, or missing information on GLP compliance. If PMRA has concerns regarding the submitted data, additional data may be requested or the Agency may require that a laboratory audit be conducted.
Analytical Methods: Pesticide analytical manual
For each new pesticide MRL, the registrant must provide an analytical method that can be used for enforcement purposes. For an existing pesticide, the analytical method can be found in the U.S. FDA's Pesticide Analytical Manual (PAM) or similar analytical methodologies employed by the CFIA.
The CFIA and U.S. FDA are responsible under the Food and Drugs Act and the Federal Food, Drug, and Cosmetic Act (FFDCA), respectively, for enforcing tolerances established by the PMRA and the U.S. EPA. In meeting this responsibility, the CFIA or the U.S. FDA collects and analyzes food from commercial channels-of-trade. Analytical methods are used in laboratories to examine food for pesticide residues for regulatory purposes. The PAM manual is organized according to the scope of the analytical methods.
- Volume 1 contains multiresidue methods that are used by on a routine basis because of their efficiency and broad applicability, especially for analyzing foods of unknown pesticide treatment history.
- Volume 2 contains methods designed for analyzing commodities for residues of only a single compound (although some methods are capable of determining several related compounds). These methods are most often used when the likely residue is known and/or when the residue of interest cannot be determined by common MRM's.
Acute and chronic exposure estimates
As part of the PMRA's reevaluation program, the Agency reassess all existing MRLs, based on available information, according to new, more stringent standards. Among these new standards are specific determinations regarding the potential for increased sensitivity of infants, children, and other subpopulations to the pesticide; assessment of the potential for aggregate exposures from various sources (such as food, drinking water, and pesticide uses in and around the home); and cumulative assessments of pesticides with a common mechanism of toxicity. PMRA anticipates that refinements will be key to developing more realistic estimates of the actual residue levels on food as the PMRA proceeds through the aggregate, and particularly the cumulative, assessment of pesticides. More realistic residue estimates ultimately improve the Agency's ability to make informed regulatory decisions that fully protect public health and sensitive subpopulations, including infants and children.
As mentioned below, the PMRA develops these estimates of pesticide residue levels through a "tiered approach", where estimates of acute pesticide exposure are calculated slightly differently from those for chronic estimates.
The basic framework: The tiered approach
The PMRA uses a tiered approach in assessing acute and chronic risks from pesticides in food. Under this approach, acute exposure estimates are calculated differently from chronic exposure because in an acute assessment, the risk assessor is trying to estimate how much of a pesticide residue might be consumed in a single day, while in a chronic assessment, the risk assessor is trying to estimate how much of a pesticide residue might be consumed on a daily basis over the course of a lifetime. Acute exposure calculations tend to employ high-end residue values, high-end consumption and high-end % CT estimates. Chronic exposure calculations tend to use average residue values, average consumption values, and average % CT estimates.
Figure: Tiered Approach in Assessing Acute and Chronic Exposure Assessment
Text description
This figure describes the tiered approach used by the Pest Management Regulatory Agency (PMRA) in assessing acute and chronic risks from pesticides in food.
In assessing acute and chronic risks from pesticides in food, the PMRA uses a tiered approach where it performs an initial risk assessment using "worst-case" assumptions. For example, for tier 1 assessments, the PMRA would assume that for both acute and chronic risk assessments, the residues are at maximum residue levels and that 100% of the crop was treated. Generally speaking, the level of resources and data needed to refine exposure estimates increase with each tier.
The maximum residue level also referred to as the tolerance level are based on crop field trial data and are the highest residues that could possibly be found on food resulting from maximum use according to the label. MRL residues represent levels not likely to be found on foods in commerce
Lower tier (Tiers 1 and 2) exposure assessments use residue levels derived from guideline crop field trail data (tolerance or maximum residue levels) and can (for certain crops) use readily available usage information such as the percentage of crop that has been treated with a particular pesticide. These estimates tend to overestimate actual residue levels in food. Generally, if risks from pesticide residues are not of concern using lower tier exposure estimates, no further refinements are made. With the aggregate and cumulative assessments now required, it is likely that higher tier (tier 3 and 4) exposure estimates will be needed. In tier 3 and 4 assessments, the refinements considered include actual percent crop treated data, pesticide data program data, monitoring data, probabilistic assessment (acute only), residue decline studies, processing studies, and market basket data.

Maximum residue levels. These are based on crop field trial data and are used in setting MRLs. They are the highest residues that could possibly be found on food resulting from maximum use according to the label. MRL residues represent levels not likely to be found on foods in commerce.
The tiered approach to acute and chronic exposure assessment
In assessing acute and chronic risks from pesticides in food, the PMRA uses a tiered approach where it performs an initial risk assessment using "worst-case" assumptions. For example, at the first tier (Tier 1), the PMRA would assume that for both acute and chronic risk assessments, the residues are at tolerance levels and that 100% of the crop was treated. Generally speaking, the level of resources and data needed to refine exposure estimates increase with each tier.
Lower tier (Tiers 1 and 2) exposure assessments use residue levels derived from guideline crop field trial data (tolerance levels) and can (for certain crops) use readily available usage information such as the percentage of the crop that has been treated (% CT) with a particular pesticide. These estimates tend to overestimate actual pesticide residue levels in food. Generally, if risks from pesticide residues in food are not of concern using lower tier exposure estimates, no further refinements are made. With the aggregate and cumulative assessments now required, it is likely that higher tier (Tiers 3 and 4) exposure estimates will be needed.
In summary, the types of data that can be used in the tiering process include:
- percentage of crop treated;
- CFIA monitoring data;
- U.S. FDA monitoring data;
- European Union (EU) monitoring data;
- USDA PDP monitoring data;
- Total Diet Studies;
- other market basket (monitoring) studies;
- bridging studies;
- residue decline studies;
- residue degradation studies; and
- commercial and consumer practices such as washing, cooking, and peeling.
Acute exposure estimates
In assessing acute exposure estimates, the risk assessor is estimating how much of a particular pesticide residue might be consumed in a single day. General information on the acute exposure assessment policy and specific information on the types of data that can be used to estimate acute exposure can be found in Classification of Food Forms With Respect to Level of Blending (U.S. EPA 1999b) and Guidance for Refining Anticipated Residue Estimates for Use in Acute Dietary Probabilistic Risk Assessment (U.S. EPA 2000b)
Chronic exposure estimates
In assessing chronic exposure estimates, the risk assessor is calculating how much of a particular pesticide residue might be consumed on a daily basis over the course of a lifetime. Guidance on estimating chronic exposure resulting from exposure to pesticide residues in food can be found in Guidelines for the Use of Anticipated Residues in Dietary Exposure Assessment (U.S. EPA 1991a).
Limit of detection and limit of quantification
The limit of detection (LOD) is the minimum concentration that an analytical method can detect, and the limit of quantification (LOQ) is the minimum concentration that an analytical method can detect and reliably and consistently quantify. Note that the limit of quantification is the same as the limit of determination but the former term is used to avoid confusion with the limit of detection acronym, LOD.
Quite frequently, in analyzing food or other substances for pesticide residues, residues are not detected at concentrations above the LOD. Even though the laboratory equipment cannot detect a residue, a residue may be present, at some level below the LOD, and such residues may contribute to exposure resulting from pesticide residues in food.
In general, the PMRA utilizes a default value of one half the LOD (½ LOD) or one half the LOQ (½ LOQ) for commodities that have been treated with a pesticide but for which no detectable residues are measured. The policy for assigning values to nondetectable residues is intended to avoid underestimating exposure to potentially sensitive or highly exposed groups such as infants and children while attempting to approximate actual residue concentrations as closely as possible. Both biological information and empirical residue measurements support the PMRA's belief that these science policies are consistent with these goals.
The Agency utilizes statistical methods for handling data sets that contain both detected and nondetected (ND) residues; these are provided in the document listed below. The document also describes the PMRA's policy of performing a sensitivity analysis to determine the impact of using different assumptions (e.g., assuming NDs equal to LOD instead of ½ LOD) in evaluating nondetectable residues.
Percentage of crop treated
The Agency frequently uses information on how much of a crop is actually treated with a given pesticide to make as accurate an estimate of exposures as possible. The PMRA obtains this information from a variety of agricultural and nonagricultural data sources, including:
- Statistics Canada;
- Agriculture and Agri-Food Canada;
- various provincial surveys/censuses, and
- a variety of proprietary data sources.
These data sources contain pesticide information from all major crop producing states. PMRA scientists analyze all available information for dietary risk assessment, as appropriate.
Probabilistic analysis
One technique used to calculate acute exposure and risk in the more refined tiers--Tiers 3 and 4--is probabilistic analysis, where the entire range of residue data from the numerous crop field trial studies (or other sources) together with the range of consumption values is used to estimate the distribution of exposure for the population of concern and the probability of exposure to any particular level. This technique allows for a more realistic estimate of exposure. At this time, the probabilistic technique can be used only for acute assessment because of limitations in the consumption database.
Probabilistic analysis is in contrast to deterministic analysis, where only a single, high-end residue value (e.g., MRLs on foods) or a statistical tendency (e.g., average values from appropriate field trial data) is used with the range of consumption estimates. Such single-value risk estimates do not provide information on the variability and uncertainty that may be associated with a risk estimate. The Agency has traditionally used deterministic analyses involving point estimates of specific parameters to generate a single estimate of exposure and risk based on various assumptions about the concentration of pesticide in any given medium (e.g., food, water, air, etc.) and the amount of that medium consumed, breathed, or otherwise contacted.
Bridging study
A bridging study is one in which the study investigator examines the relationship between residue levels that occur as a result of maximum pesticide application (e.g., maximum rate, highest application frequency and shortest PHI) versus those expected to occur at the range of more typical rates. This relationship is then used to adjust the maximum residue levels originally obtained from the crop field trials.
The PMRA uses the residue data obtained from these field studies in conjunction with information on what fraction of the crop is treated at each rate to refine its exposure estimates. Specific guidance on conducting bridging studies can be found in the document listed below.
Residue decline study
A residue decline study is one in which the study investigator examines the relationship between residue levels at the time of application versus residue levels at the range of typical harvest times. That is, the investigator is looking at how quickly the pesticide being studied degrades between application and harvest. Because pesticides degrade and dissipate at different rates over time, it cannot be assumed that this relationship is linear (e.g., that doubling the preharvest interval would result in half the residue). In a residue decline study, samples from a single field trial are collected at multiple PHIs and analyzed to determine rates of residue disappearance/dissipation.
The PMRA uses the residue data obtained from these decline studies in conjunction with information on what fraction of the crop is harvested at each interval to refine its exposure estimates. Information from residue decline studies may be particularly useful when the pesticide of interest decays quickly and/or a large period of time elapses between the pesticide application date and the harvest date. Specific guidance on conducting residue decline studies can be found in the document listed below.
Residue Degradation Study
A residue degradation study is one in which the study investigator examines the relationship between residue levels at harvest versus the residue levels at consumer purchase. A residue degradation study is similar to a residue decline study; however, the time interval being studied is later. Residue degradation studies are designed to characterize the decreasing amounts of pesticide residues over time on commodities during storage or transportation. In a residue degradation study, samples are collected before storage or transportation begins and at different points in the "process" that correspond to times that consumers may purchase the food.
Information from a residue degradation study may be particularly useful when a substantial period of time elapses as during extended transportation or storage. The PMRA recognizes, for example, that some crops such as apples and potatoes can be typically stored for relatively long periods of time after harvest and before purchase by the consumer. Other items (e.g., tomatoes and bananas) may be typically picked green for ease of transport; of necessity, many days can, therefore, pass between harvest and consumption.
Consumer and commercial practices
Cooking and processing data permit better estimates of pesticide exposure by incorporating information on actual consumer and industry food preparation practices. Home processing such as cooking, washing, peeling, etc. can significantly reduce exposure to pesticide residues. For example, potatoes would likely be cooked prior to consumption, and oranges and bananas would typically be peeled. Commercial preparation practices, such as canning, washing, peeling, various cooking methods, etc., can also reduce exposure to pesticide residues.
In commercial processing studies, samples are collected from at least two points in the processing procedures (e.g., before processing/cooking, after washing, after peeling, at the end of processing, etc.) and a processing factor is calculated. The processing practices used in the study should reflect typical commercial practices (whether the raw agricultural commodity is typically washed, peeled, cooked or otherwise treated before canning, freezing, drying or other types of processing).
Consumption information
The food consumption data that PMRA uses in its risk assessment for exposure resulting from pesticide residues in food are provided by USDA from their periodic food consumption surveys.
List of where to find information
- Topic and Description
- Name of Document (as applicable)
Page - Acute exposure estimates. Policy on how to estimate acute exposure resulting from exposure to pesticide residues in food.
- Classification of food forms with respect to level of blending, HED Standard Operating Procedure 99.6 (U.S. EPA 1999b).
Page 34 - Acute exposure estimates. Descriptions of the types of data the PMRA can use to refine acute residue estimates.
- Guidance for refining anticipated residue estimates for use in acute dietary probabilistic risk assessment (U.S. EPA 2000b) http://www.epa.gov/fedrgstr/EPA-PEST/2000/June/Day-23/o-p15917.htm
Page 34 - Aggregate exposure. Guidance on conducting aggregate exposure assessment.
- Guidance for performing aggregate exposure and risk assessments, draft (U.S. EPA 1999a, revised November 28, 2001) http://www.epa.gov/pesticides/trac/science/#non-occupational
Page 5 - Analytical methods. Analytical methods (single residue and multiresidue methods) for determining the concentration of pesticide residues in food.
- PAM, Vols. 1 and 2 (U.S. HHS 1994, 1997) http://vm.cfsan.fda.gov/~frf/pami1.html
Page 31 - Bridging data. Detailed guidance on conducting bridging studies.
- Guidance for refining anticipated residue estimates for use in acute dietary probabilistic risk assessment (U.S. EPA 2000b) http://www.epa.gov/fedrgstr/EPA-PEST/2000/June/Day-23/o-p15917.htm
Page 40 - CFIA monitoring data. Where to find information, including data summaries from the monitoring programs, on CFIA's pesticide monitoring programs, including analytical methodology.
- CFIA reports may be obtained from: Bureau of Food Safety and Consumer Protection, Canadian Food Inspection Agency, 3rd floor West, 59 Camelot Drive, Ottawa, ON K1A 0Y9
Page 26 - CGC monitoring data. Where to find information, including data summaries from the monitoring programs, on CGC's pesticide monitoring programs.
- CGC reports may be obtained from:
Canadian Grain Commission,
1404-303 Main Street,
Winnipeg, MB R3R 2J2
tel: (204) 983-3345
fax (204) 983-0724
Page 27 - Chronic exposure estimates. Guidance on calculating chronic food exposure under the tiering system.
- Guidelines for the use of anticipated residues in dietary exposure assessment (U.S. EPA 1991)
Page 35 - Commercial and consumer practices. Information on how commercial and consumer practices can be factored into assessments for exposure resulting from pesticide residues from food.
- Guidance for refining anticipated residue estimates for use in acute dietary probabilistic risk assessment (U.S. EPA 2000b) http://www.epa.gov/fedrgstr/EPA-PEST/2000/June/Day-23/o-p15917.htm
- Data requirements. PMRA's data requirements for residue data on food.
Regulatory Directive DIR98-02 (PMRA 1998a), page 19, at Pest Management Regulatory Agency Publications Section - Decompositing. A paper discussing the "Allender" method for "decompositing," which is the process of statistically translating composite residue information into "individual item" residue information.
- Statistical methods for use of composite data in acute dietary exposure assessment(U.S. EPA 1999d) http://www.epa.gov/scipoly/sap/1999/may/hanssap.pdf
Page 23 - Decompositing. A paper comparing U.S. EPA's method for decomposition to two others: RDFgen, and MaxLIP.
- Office of Pesticide Programs' comparison of Allender, RDFgen, and MaxLIP decomposition procedures (U.S. EPA 2000a) http://www.epa.gov/scipoly/sap/index.htm
Page 23 - DEEM™. A full description of the DEEM™ model and how it operates.
- Background document for the sessions: dietary exposure evaluation model (DEEM™) and DEEM™ decompositing procedure and software (Novigen Sciences, Inc. 2000) http://www.epa.gov/scipoly/sap/index.htm
Page 15 - U.S. FDA monitoring data. Where to find information, including data summaries from the monitoring programs, on U.S. FDA's pesticide monitoring programs.
- For further information on the U.S. FDA monitoring program, including data summaries from the monitoring programs, contact:
U.S. Food and Drug Administration
Center for Food Safety and Applied Nutrition
200 C Street SW,
Washington, DC 20204
http://vm.cfsan.fda.gov/~dms/pesrpts.html
Page 26 - Food consumption information. Where to find information on the USDA food consumption surveys.
- For further information on USDA's food consumption surveys, contact:
Food Surveys Research Group
Beltsville Human Nutrition Research Center
Agricultural Research Service,
USDA 10300 Baltimore Ave., Building 005, Room 102,
BARC-West, Beltsville, MD 20705
tel: 301-504-0170
e-mail: amoshfegh@rbhnrc.usda.gov or visit the following web site: http://www.barc.usda.gov/bhnrc/foodsurvey/Fsrg1.html
Page 43 - Good laboratory practices. The standards that are to be used by laboratories in conducting studies to be used in setting tolerances, etc.
Regulatory Directive DIR98-01 (PMRA 1998b), page 30, Pest Management Regulatory Agency Publications Section - Guidelines. Guidelines residue chemistry data requirements.
- Residue chemistry guidelines (PMRA 1998a)
Page 19 - LOD and LOQ. The statistical methods for handling nondetectable pesticide residues in food.
- Assigning values to non-detected/ non-quantified pesticide residues in human health food exposure assessments March 23, 2000 (U.S. EPA 2000c)
Page 36 - Market basket studies. Information on how to conduct a market basket study for purposes of refining residues for use in acute exposure assessments.
- Guidance for refining anticipated residue estimates for use in acute dietary probabilistic risk assessment (U.S. EPA 2000b) http://www.epa.gov/fedrgstr/EPA-PEST/2000/June/Day-23/o-p15917.htm
Page 28 - Nondetectable residues. The statistical methods for handling nondetectable pesticide residues in food.
- The statistical methods for handling nondetects can be found in: Assigning values to nondetected/nonquantified pesticide residues in food(PMRA 2002a) or U.S. EPA web site: http://www.epa.gov/pesticides/trac/science/trac3b012.pdf
Page 36 - Percent of crop treated. Information on how percent of crop treated is determined and used in risk assessment.
- The role of the use-related information in pesticide risk assessment and risk management, draft document (U.S. EPA 1999c)
Page 37 - PDP. Obtaining information on USDA's PDP program or how to access data summaries.
- To obtain more information on the PDP program or to access summaries of the data contact USDA at: http://www.ams.usda.gov/science/pdp/index.htm
Page 21 - Probabilistic assessment. Guidance on the submission and review of probabilistic human health exposure assessments.
- Guidance for submission of probabilistic human health exposure assessments to the Office of Pesticide Programs, draft document (U.S. EPA 1998b) http://www.epa.gov/pesticides/trac/science/#monte
Note: This guidance document also provides a good overall discussion of the probabilistic methods.
Page 39 - Residue decline studies. Detailed guidance on conducting residue decline studies.
- Guidance for refining anticipated residue estimates for use in acute dietary probabilistic risk assessment(U.S. EPA 2000b) http://www.epa.gov/fedrgstr/EPA-PEST/2000/June/Day-23/o-p15917.htm
Page 41 - Residue degradation studies. Information on conducting residue degradation studies.
- Guidance for refining anticipated estimates for use in acute dietary probabilistic risk assessment (U.S. EPA 2000b) http://www.epa.gov/fedrgstr/EPA-PEST/2000/June/Day-23/o-p15917.htm
Page 42 - Tiering process. How PMRA assesses exposure to pesticide residues in food through the four-tier process.
- Classification of food forms with respect to level of blending. HED Standard Operating Procedure 99.6 (U.S. EPA 1999b)
Page 34 - Total diet study data. Where to find information, including data summaries from Health Canada's TDS programs.
- For further information on the HC total diet program contact:
Food Research Division, Health Canada,
Sir Frederick Banting Research Center,
Ross Avenue, Ottawa ON K1A 0L2
Page 26 - Translating PDP data to other crops. The conditions under which translating is appropriate and details on the translation policy.
- Translation of monitoring data HED Standard Operating Procedure 99.3 (3/26/99) (U.S. EPA 1999e)
Page 22 - User's guide. Listing of available EPA information on assessing exposure to pesticides in food.
- Available EPA Information on Assessing Exposure to Pesticides in Food--A User's Guide (Revised) July 12, 2000. Docket Number: OPP-00576A http://www.epa.gov/fedrgstr/EPA-PEST/2000/July/Day-12/6061.pdf
Page 1
List of abbreviations
- AAFC
- Agriculture and Agri-Food Canada
- ADI
- Acceptable Daily Intake
- aPAD
- Acute Population Adjusted Dose
- ARfD
- Acute Reference Dose
- CFIA
- Canadian Food Inspection Agency
- CFR
- Code of Federal Regulations (U.S. EPA)
- CGC
- Canadian Grain Commission
- cPAD
- Chronic Populated Adjusted Dose
- CSFII
- USDA's Continuing Survey of Food Intake by Individuals
- DEEM™
- Dietary Exposure Evaluation Model
- DRA
- Dietary Risk Assessment
- EU
- European Union
- FDAR
- Food and Drugs Act and Regulations (Canada)
- FDA
- Food and Drugs Act (Canada)
- FDR
- Food and Drug Regulations (Canada)
- FFDCA
- Federal Food, Drug, and Cosmetic Act (U.S. FDA)
- FIFRA
- The Federal Insecticide, Fungicide, and Rodenticide Act (U.S. EPA)
- FQPA
- The Food Quality Protection Act (U.S. EPA and U.S. FDA )
- GLP
- Good Laboratory Practices
- GRL
- Grain Research Laboratory (Winnipeg, Manitoba, Canada)
- HC
- Health Canada
- HED
- The Health Effects Division of the Office of Pesticide Programs (U.S. EPA)
- HHS
- United States Department of Health and Human Services
- LOAEL
- Lowest Observed Adverse Effect Level
- LOD
- Limit of Detection
- LOQ
- Limit of Quantification
- MBS
- Market Basket Survey
- MOE
- Margin of Exposure
- MOR
- Magnitude of Residue
- MRL
- Maximum Residue Limit
- MRM
- Multiresidue Method
- NAFTA
- North American Free Trade Agreement
- NARA
- National Archives and Records Administration (U.S.)
- NAS
- National Academy of Sciences (U.S.)
- ND
- Nondetects or nondetectable
- NOAEL
- No Observed Adverse Effect Level
- OPP
- Office of Pesticide Programs (U.S. EPA)
- OPPTS
- Office of Prevention, Pesticides, and Toxic Substances (U.S. EPA)
- PAM
- Pesticide Analytical Manual (U.S. FDA)
- PCPA
- Pest Control Products Act (Canada)
- PDP
- Pesticide Data Program (USDA)
- PHI
- Preharvest Interval
- PMRA
- Pest Management Regulatory Agency
- % CT
- Percentage of Crop Treated
- PoD
- Point of Departure
- ppb
- part per billion
- ppm
- part per million
- QA
- Quality Assurance
- QC
- Quality Control
- q1*
- Q-Star or Q1-Star
- RAC
- Raw Agricultural Commodity
- RfD
- Reference Dose
- SAP
- The FIFRA Scientific Advisory Panel (U.S. EPA)
- SOP
- Standard Operating Procedure
- SRM
- Single Residue Method
- TDS
- Total Diet Study
- U.S.
- United States
- USDA
- United States Department of Agriculture
- U.S. EPA
- United States Environmental Protection Agency
- U.S. FDA
- United States Food and Drug Administration
- U.S. HHS
- United States Department of Health and Human Services
References
- National Academy of Sciences (NAS). 1993. Pesticides in the diets of infants and children. National Academy Press, Washington, D.C.
- National Archives and Records Administration (NARA). 1999. Code of Federal Regulations. Protection of environment. Parts 150 to 189. July 1, 1999. U.S. Government Printing Office, Washington, D.C.
- Novigen Sciences, Inc. 2000. Background material for the March 2000 FIFRA Scientific Advisory Panel Meeting. Background document for the sessions: dietary exposure evaluation model (DEEM™) and DEEM™ decompositing procedure and software. February 29 to March 3, 2000.
- Pest Management Regulatory Agency (PMRA). 1998a. Residue chemistry guidelines. Dir98-02. Health Canada, Pest Management Regulatory Agency, Ottawa, Ont.
- Pest Management Regulatory Agency (PMRA). 1998b. Good laboratory practice. Dir98-01. Health Canada, Pest Management Regulatory Agency, Ottawa, Ont.
- Pest Management Regulatory Agency (PMRA). 2002. Assigning values to nondetected/nonquantified pesticide residues in foods; in preparation, expected release data: spring 2002. Health Canada, Pest Management Regulatory Agency, Ottawa, Ont.
- U.S. Department of Health and Human Services (U.S. HHS). 1994. Pesticide analytical manual. Vol. 1. Multiresidue methods. 3rd edition (Revised, September 1996; October 1997; and October 1999). January 1994. U.S. Government Printing Office, Washington, D.C.
- U.S. Department of Health and Human Services (U.S. HHS). 1997. Pesticide analytical manual. Vol. 2. February 1997. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1991. Guidelines for the use of anticipated residues in dietary exposure assessment. March 25, 1991. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1996. Residue chemistry test guidelines OPPTS 860; August 1996. (U.S. EPA 712-C-96-169). U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA).1998a. Memorandum from Margaret Stasikowski to Jim Jones, Anne Lindsay, and Lois Rossi. Supplementary guidance on use of OPPTS residue chemistry test guidelines 860.1500, Crop Field Trials (residue zone maps - Canadian extension). April 8, 1998. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1998b. Guidance for submission of probabilistic human health exposure assessments to the Office of Pesticide Programs. draft document. November 4, 1998. (63 FR 59780). U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1999a. Guidance for performing aggregate exposure and risk assessments, draft document. October 29, 1999. (65 FR 459). U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1999b. Memorandum from Margaret Stasikowski, Director Health Effects Division to Health Effects Division Staff. Classification of food forms with respect to level of blending. HED Standard Operating Procedure 99.6 (8/20/99); August 20, 1999. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1999c. The role of the use-related information in pesticide risk assessment and risk management, draft document. June 29, 1999. (64 FR 37977). U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1999d. Background document for the May 26, 1999 meeting of the FIFRA Scientific Advisory Panel. Statistical methods for use of composite data in acute dietary exposure assessment. May 26, 1999. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 1999e. Memorandum from Margaret Stasikowski, Director Health Effects Division to Health Effects Division Staff. Translation of monitoring data. HED Standard Operating Procedure 99.3 (3/26/99); March 26, 1999. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA) 2000a. Background document for the March 1, 2000 meeting of the FIFRA Scientific Advisory Panel. Office of Pesticide Programs' Comparison of Allender, RDFgen, and MaxLIP decomposition procedures. February 1, 2000. U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 2000b. Guidance for refining anticipated residue estimates for use in acute dietary probabilistic risk assessment. June 15, 2000. (65 FR 39147). U.S. Environmental Protection Agency, Washington, D.C.
- U.S. Environmental Protection Agency (U.S. EPA). 2000c. Assigning values to non-detected/non-quantified pesticide residues in human health food exposure assessments. March 23, 2000. (65 FR 17266). U.S. Environmental Protection Agency, Washington, D.C.
Glossary of terms
Bridging study.
Cancer potency.
Cancer potency
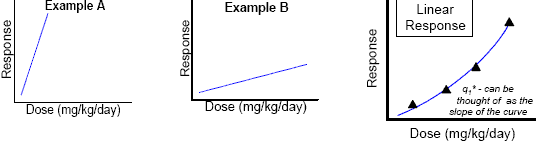
Text description
The figure describes cancer potency expressed as q1* using three dose response graphs.
The first two graphs illustrate the dose response of two example pesticides, example A and B. The potency of the pesticide in example A is greater than the potency of the pesticide in example B as reflected by the steeper dose-response curve in example A than in example B.
The third graph illustrates the measure of cancer potency as derived from the dose response data. A dose response curve is plotted and modelled to produce a linear response. The q1* represents the slope of the dose response curve in the low dose region.

Composite.
A method of sampling and analysis where a number of individual items (e.g., apples) is combined and/or blended into a single sample or analysis.
Crop field trials.
Decompositing.
A statistical procedure to estimate pesticide residue in a single food sample (e.g. , an apple) from residue data for a composite sample (e.g., average value for a bag of apples).
Deterministic analysis.
Dietary Exposure Evaluation Model (DEEM™).
Endpoint.
The type of toxic effect exhibited by a pesticide (e.g., if the pesticide affects the nervous system, the endpoint would be neurotoxicity). A pesticide may have more than one endpoint. Endpoints are determined for both acute and chronic exposures.
Limit of detection (LOD).
The minimum concentration that an analytical method, which includes the laboratory instrumentation (equipment), can detect or "see". A typical LOD might be 0.01 part per million (ppm).
Limit of quantification (LOQ).
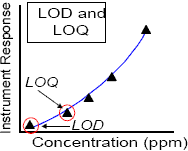
Limit of quantification (LOQ)

Text description
This figure is a 2-dimensional plot of concentration (in units of parts per million) along the abscissa, and instrument response (in dimensionless units) along the ordinate. The figure presents the relation between the limit of detection (LOD) and the limit of quantitation (LOQ). Two of five points on a concentration-response curve are identified in quadrant 1. These are the LOQ and the LOD, with the LOD located closest to the origin. There is a non-linear, monotonic increase in the concentration-response curve through the five points.
Linear vs. nonlinear response.
Lowest observed adverse effect level (LOAEL).
Margin of exposure (MOE).
Magnitude of the residue (MOR).
Maximum residue limits (MRLs).
Multiresidue methods (MRMs).
A nonlinear response
A nonlinear response
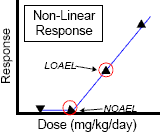
Text description
This figure is a 2-dimensional plot of dose (in units of milligrams per kilogram per day) along the abscissa, and response (in dimensionless units) along the ordinate. The figure presents a non-linear does-response curve. Two of the four points on the dose-response curve are identified in quadrant 1. These are the lowest observed adverse effect level (LOAEL), and the no observed adverse effect level (NOAEL). The dose-response curve is horizontal from the first to the second point, which is identified as the NOAEL. There is linear, proportional and monotonic increase in the dose-response curve to the third point, which is identified as the LOAEL, and then through the fourth point.
