Mpox: For health professionals
On this page
- What's unique to the 2024 multi-country outbreak
- What's unique to the 2022 multi-country outbreak
- What health professionals need to know about mpox
- Agent of disease
- Transmission
- Clinical manifestations
- Risk assessments
- Diagnosis
- Management and treatment
- Reporting an adverse reaction following vaccination
- Infection prevention and control
- Surveillance
- Evidence
- PHAC Expert Panel on Mpox in Canada
- Webinars about mpox
- Additional resources
What's unique to the 2024 multi-country outbreak
On August 14, 2024, the WHO Director General issued a statement declaring that the increase in mpox (previously known as monkeypox) cases in the Democratic Republic of the Congo (DRC) and a growing number of countries in Africa, as well as the emergence of a new strain of clade I, clade Ib, monkeypox virus (MPXV) constitute a public health emergency of international concern (PHEIC). The PHEIC was declared over on September 5, 2025, based on sustained declines of cases and deaths in the DRC and other affected African countries including Burundi, Sierra Leone, and Uganda.
The new strain (clade Ib) that emerged in the DRC's South Kivu province around September 2023 is a member of clade I. Current evidence is still emerging, but suggests that it is more common among youth and adults 15 years and older and is less severe and possibly more transmissible than clade Ia.
Clade Ia is endemic in the DRC, and there has been an increase in clade Ia cases in the DRC, Central African Republic and Republic of Congo this year. Clade Ia is most common among children, spreads by multiple modes of transmission and is historically the most severe.
Currently, transmission of both clade Ia and Ib MPXV occurs mainly through direct and intimate contact (for example, sexual contact) with a person who has mpox. However, cases of transmission not linked to sexual exposure have also been reported, including family members, and persons associated with close nonsexual contact. Although, airborne transmission of mpox is possible, current data continue to support a minimal role of spread through the air for clade Ia and Ib MPXV.
Although the risk to people in Canada from clade I MPXV remains low at this time, the Government of Canada is closely monitoring the mpox situation in the DRC and neighbouring countries, while working with domestic and international partners to determine opportunities for support.
What's unique to the 2022 multi-country outbreak
Since May 2022, many non-endemic countries, including Canada, reported an unusually large number of cases of clade IIb MPXV, with sustained chains of local transmission. Prior to this outbreak, mpox was rarely observed outside of endemic countries of West and Central Africa, except for sporadic cases or clusters of cases, in relation with travel to endemic countries or with importation of reservoir animals.
In Canada, transmission of clade IIb MPXV occurs mainly through direct and intimate contact (for example, sexual contact) with a person who has mpox. However, cases of transmission not linked to sexual exposure have been reported. Although airborne transmission is possible, current data continue to support minimal role of spread through the air for clade II MPXV.
While MPXV can infect anyone, most cases reported in Canada in the 2022 outbreak have been among gay, bisexual, and other men who have sex with men.
On July 23rd, 2022, the World Health Organization (WHO) officially declared mpox to be a Public Health Emergency of International Concern (PHEIC).
During the multi-country 2022 outbreak, not all cases have been presenting in the typical fashion historically described in the literature. Unique manifestations described during this outbreak include:
- asynchronous generalized rash
- exclusively oral, genital, or perianal lesions
- presentation with a single or only a few lesions
- pharyngitis manifesting as sore throat (may be the only initial presentation)
- symptoms of proctitis (for example, rectal pain)
Historically, systemic symptoms were reported to begin prior to skin lesions. However, now they are also reported to begin concurrently with, or after the appearance of skin lesions.
Some of the leading causes of hospitalization include the following complications:
- myocarditis
- severe pharyngitis (may compromise airway and swallowing)
- severe proctitis (may hinder defecation)
- uncontrolled genital pain leading to urinary retention
- bacterial superinfection of skin lesions
- corneal infections that can lead to loss of vision
- encephalitis
Although rare, death due to MPXV infection can occur and has been reported during the 2022 multi-country outbreak.
The MPXV can infect several different animal species. Therefore, although most cases are currently due to human-to-human transmission, there is concern that the virus could spread to animals in non-endemic regions and establish a new reservoir. To date, only a small number of instances of human-to-animal transmission has been reported in household settings, but this is an area that requires further study.
What health professionals need to know about mpox
Mpox is a viral zoonotic infectious disease caused by the MPXV (MPXV) an Orthopoxvirus. Person-to-person transmission can occur through direct contact with an infected person or shared contaminated objects. Human-to-human transmission is the major driver of the 2022 and 2024 multi-country outbreak.
While outbreaks have occurred in endemic areas, sustained multi-country outbreaks with very large numbers of affected persons have not been previously reported. Outside of endemic areas, there was a single zoonotic outbreak in 2003 in the United States, linked to pet prairie dogs who were infected after contact with rodents imported from Africa for the pet trade. As noted, there has been a second worldwide outbreak beginning in 2022 that affected many non-endemic countries, that to a small degree is still ongoing. Historically, mpox has presented with a characteristic rash, which could be preceded by systemic manifestations such as fever. Lymphadenopathy is a feature that helps to differentiate mpox from other diseases with a similar rash (such as varicella zoster, herpes simplex, secondary syphilis, chancroid and hand-foot-and-mouth disease).
Presentation of cases has not always been typical. Please refer to What's unique to the 2022 multi-country outbreak and the What's unique to the 2024 section for additional details.
Management is mainly focused on supportive care, as there is limited data on the clinical effectiveness of specific treatments for MPXV infections in humans. Some existing treatments for smallpox may have a role to play in mpox management. Smallpox vaccines have been used for pre-exposure and post-exposure prevention. According to reports from the 2003 United States outbreak, post-exposure use of smallpox vaccine prevented the diseases among the vaccine recipients.
Agent of disease
The monkeypox virus is part of the Poxviridae family. Specifically, it is a member of the Orthopoxvirus genus, which also includes variola virus (the virus that causes smallpox), vaccinia virus and cowpox virus.
There are 2 known subtypes of MPXV (called clades). On August 12, 2022, the World Health Organization introduced a new nomenclature for the MPXV clades:
- Clade one (I) (formerly known as Congo Basin or Central African clade), which is further divided into 2 subcladesFootnote 1:
- Clade Ia
- Clade Ib
- Clade two (II) (formerly known as West African clade), which is further divided into 2 subclades:
- Clade IIa
- Clade IIb
The original Congo-Basin clade Ia virus has the highest case fatality rate. The other clades including clade Ib and II are associated with a relatively less severe symptoms and outcomes, although evidence is still emerging with respect to clade Ib.
Clade IIb has been implicated in the 2022 multi-country outbreak.
Clade Ib has been implicated in the 2024 multi-country outbreak.
Transmission
Mpox has typically been characterized as a zoonotic infection. Its main reservoirs are unknown but are thought to be small mammals from endemic regions of Africa, such as:
- dormice
- Gambian pouched rats
- rope squirrels
- sun squirrels
Humans and non-human primates are thought to be incidental hosts.
People are contagious from the onset of first symptoms until the scabs have fallen off on their own and the skin is healed. This also includes the healing of all mucosal surfaces (mouth, throat, eyes, vagina and anorectal area). People may also be contagious up to 4 days before symptoms begin, but it is not currently known how often transmission happens before symptoms begin.
Modelling studies based on linked infection pairs with known exposures times have suggested that transmission can occur up to 4 days before symptom onset.
Infection is thought to occur when the virus enters the body through skin, the respiratory tract, or mucous membranes.
This can occur through the following routes:
Animal-to-human
- through direct contact with an infected animal's skin lesions, body fluids (such as blood and saliva) or mucosal surfaces, including:
- contact from providing care and handling
- through bites or scratches
- preparing or eating undercooked meat (for example, bushmeat)
Person-to-person
- through direct contact with skin lesions or scabs, body fluids (such as blood, saliva and semen) or mucosal surfaces (such as eyes, mouth, throat, genitalia and anorectal area)
- for example, contact from providing care or sexual contact
- Although spread through the air is possible, current data continue to support a minimal role of spread through the air for clade I and II mpox. However, this possibility should continue to be examined given ongoing viral evolution.
- from an infected pregnant person to the fetus
Fomites
- through direct, unprotected contact with:
- surfaces,
- materials (for example, clothing or linens and towels),
- objects (for example, razors, utensils, needles, sex toys, toothbrushes) that have been in contact with a person or animal with mpox.
Clinical manifestations
The incubation period can range from 3 to 21 days. During the 2022 multi-country outbreak, the incubation period has generally ranged from 7 to 10 days.
Mpox illness is usually self-resolving within a period of 2 to 4 weeks. The spectrum of severity varies from a single skin lesion to disseminated, multi-organ infection. At this time, it is not known with certainty if asymptomatic infections occur.
Mpox presents with either systemic symptoms, skin or mucosal lesions, or both. Systemic symptoms typically occur 0 to 5 days before the appearance of lesions. However, they may also occur during or after the onset of skin lesions (as documented in the 2022 outbreak).
Signs and symptoms of mpox may include:
- Skin or mucosal lesions (often painful)
- fever
- chills
- lymphadenopathy (localized or generalized)
- sore throat
- rectal symptoms (for example, rectal pain)
- fatigue
- headache
- musculoskeletal manifestations such as myalgia, arthralgia and back pain
- gastrointestinal symptoms (for example, vomiting, diarrhea)
In the 2022 multi-country outbreak, pharyngitis symptoms (for example, sore throat) and proctitis symptoms (for example, rectal pain) have frequently been reported.
Skin lesions
Lesions generally (but not always) appear 1 to 3 days after the onset of fever and last for 2 to 4 weeks. Mpox lesions can be painful and may become itchy during the healing phase. Lesions can be located anywhere on the body, including the:
- mouth and pharynx
- genitals
- anal and perianal area
- hands (including the palms)
- feet (including the soles)
As the illness progresses, lesions change from flat lesions (macules) to raised lesions (papules), to vesicles and then pustules (see Figure 1). At the latest stage, they will form ulcers that eventually scab over. Figure 2 shows photographs of mpox skin lesion progression.
Mpox lesions in the same body area tend to evolve at the same time (synchronously). However, individuals may have an atypical or asynchronous rash.
Photos of mpox rash
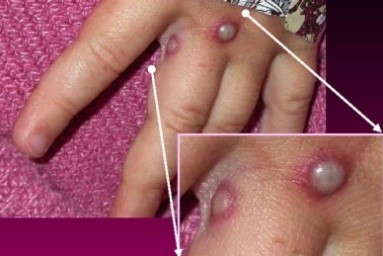
Reproduced with permission from Canadian Medical Association Journal.
Figure 1: Text description
This photograph depicts the hand of a child with mpox skin lesions. There are two round, well-defined, pustular lesions. They are surrounded by red skin and located at the proximal end of the third digit. The photograph includes a magnification of the lesions in the bottom right corner.
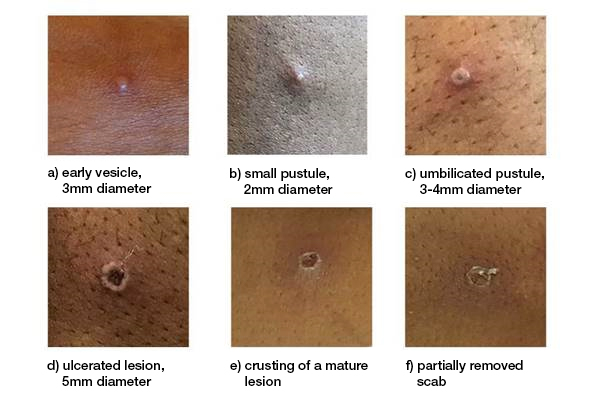
Reproduced with permission from United Kingdom Health Security Agency.
Figure 2: Text description
This figure depicts a series of 6 photographs which illustrate the evolution of a mpox lesion throughout the progression of the disease. The images are arranged such that there are 3 photographs at the top and 3 at the bottom. The progression of the rash can be followed by looking at the photographs in order from the top left corner to the bottom right corner, moving from left to right. Each photograph focuses on a single lesion.
- photograph in the top left corner depicts a small and rounded vesicle. The skin surrounding the vesicle is reddened.
- photograph in the top center shows a pustule with an erythematous base.
- photograph in the top right corner depicts an umbilicated pustule (a pustule with a central depression).
- photograph in the bottom left corner depicts an ulcerated lesion.
- photograph in the bottom center depicts an ulcerated lesion that has begun to crust over.
- photograph in the bottom right corner depicts a lesion with a scab peeling off, revealing healed skin underneath
Severe cases can occur but are rarely fatal. Notably, mpox infection during pregnancy may increase risk of maternal and fetal morbidity and mortality. Young children and immunocompromised individuals are also more likely to experience severe disease.
Case fatality rate (CFR) depends on viral clade, host factors (such as immune status and age) and structural factors (such as availability of health care).
Clade Ia has a CFR of around 3.6% in 2024 and early evidence suggests that CFR with clade Ib infection are lower than with clade Ia. Clade II, when occurring in endemic areas, has historically had a CFR of approximately 1 to 3%. When outbreaks of clade II have occurred in non-endemic countries, the CFR has been lower. For instance, there are no recorded fatalities in Canada thus far.
Note: Mortality data for clade Ia may also reflect differences in population and availability of supportive care.
Mpox complications
Young children and immunocompromised individuals might be at increased risk of complications, which may include:
- proctitis
- pharyngitis
- bacterial superinfection
- corneal infection (may lead to vision loss)
- sepsis
- pneumonia
- myocarditis
- encephalitis
- death
Co-infections
Mpox may present with concurrent sexually transmissible and blood-borne infections (STBBIs). Hence, healthcare providers should be vigilant and offer testing to mpox cases to rule out STBBIs such as syphilis, gonorrhea, chlamydia, herpes simplex and HIV.
Risk assessments
The Public Health Agency of Canada assesses risks to public health to:
- identify emerging threats
- describe, anticipate or estimate public health risks
- develop recommended actions
- explore future outcomes of risk
Public health assessments support planning, responses and decision making to improve health outcomes for people living in Canada or Canadians abroad.
Risk assessments for public health professionals
Diagnosis
To confirm the diagnosis of a MPXV infection, one or more of the following diagnostic markers must be positive:
- presence of MPXV DNA by PCR
- isolation of MPXV from viral culture
Consult your public health laboratory before sending specimens to receive proper instructions on specimen handling and transport. See biosafety advisory: Monkeypox virus (MPXV).
Wear the adequate personal protective equipment when evaluating and sampling the patient.
The best specimens for PCR and isolation are skin lesion material, including:
- swabs of lesion surface or lesion fluid
- lesion crusts (scabs)
- roofs of multiple lesions (no need to de-roof the lesion before swabbing)
Skin lesion material should be placed in an empty, sterile container for transport.
Although the preferred transport container for a MPXV specimen is an empty, sterile container, formalin-fixed tissues and paraffin-embedded tissues can be sent for PCR, but not for viral culture. We do not recommend use of Viral Transport Media (VTM or similar) but will accept these for diagnostic testing if the sample has already been taken.
Currently, serology is not being used as a diagnostic modality.
The National Microbiology Laboratory (NML) offers validated testing for mpox within the Special Pathogens program.
In response to the emergence of clade I MPXV in the Democratic Republic of Congo and other countries, the Special Pathogens Program has developed new clade I differentiating molecular assay(s) to distinguish between clades I and II.
Contact the National Microbiology Laboratory (NML) at nml.sp-lnm.asp@phac-aspc.gc.ca if you are unable to collect lesion swabs to discuss other possible viable sample types. You can also contact your local public health laboratory to inquire about other appropriate specimen types.
Given the evolving situation, NML will continue to sequence all mpox samples that test positive in relation to any one of the below criteria:
- Clade I positivity on PCR testing
- Patient presentation of unique clinical, travel, or contact history
Test requisitions forms:
Differential diagnosis
The mpox rash can be difficult to distinguish from a number of other infectious and non-infectious rashes.
A possible distinguishing feature of mpox is the presence of localized or generalized lymphadenopathy. Refer to the What's unique to the 2022 multi-country outbreak section for more information.
Smallpox can be similar to mpox in its presentation, but since its global eradication in 1980, it is not considered in the differential diagnosis unless there has been a serious laboratory breach.
Other infections to be considered in the differential diagnosis can include:
- Varicella zoster (chickenpox, shingles)
- herpes simplex
- lymphogranuloma venereum
- gonorrhea
- hand-foot-mouth disease
- molluscum contagiosum
- syphilis
- human papillomavirus infection
- chancroid
- orf (rare)
Co-infections of MPXV with sexually transmissible and blood-borne infections (STBBIs) have been frequently reported during the 2022 outbreak, therefore clinicians should be vigilant and consider testing for HIV, syphilis, gonorrhea and other STBBIs in anyone suspected to have acquired mpox through sexual contact.
Management and treatment
Supportive care is a central part of mpox management as there is limited data on the clinical effectiveness of specific treatments for mpox infections in humans. Consult an infectious disease physician to discuss therapeutic options for suspected or confirmed cases.
Some existing treatments for smallpox, such as TPOXX (tecovirimat monohydrate capsules) may have a role to play in select instances. TPOXX is an oral antiviral agent that is indicated for the treatment of human smallpox disease in adults and pediatric patients weighing at least 13 kg. It does not currently have an approved Health Canada indication for mpox or other Orthopoxviruses. However, recommendations for its off-label use can be found at: CADTH Health Technology Review on Tecovirimat (Tpoxx): Update (PDF).
Vaccination
Imvamune® is an authorized vaccine indicated for immunization against smallpox, mpox and related Orthopoxvirus infection and disease in adults 18 years of age and older who are at high risk for exposure. Although evidence is still emerging at this time, Imvamune is thought to offer protection against clade Ia, Ib and IIb.
The National Advisory Committee on Immunization (NACI) recommends post-exposure vaccination with Imvamune® to individuals who have had high risk exposure(s) to a probable or confirmed case of mpox, or within a setting where transmission is happening, if they have not received both doses of pre-exposure vaccination. NACI also recommends routine immunization for those at high risk of mpox. This guidance was last updated on May 24, 2024. For the full statement, see the NACI statement Interim guidance on the use of Imvamune®in the context of a routine immunization program. A summary of updated NACI interim guidance as of May 24, 2024, is also available.
Learn more about storage temperatures, shelf life, shipment and supportive temperature excursion information for the Imvamune vaccine in Canada.
Vaccination for travellers
Currently, the mpox vaccine is not routinely recommended for travellers without known exposure to mpox, unless they:
- meet the high risk criteria or
- are Canadian healthcare professionals in advance of deployment to support the mpox clade I outbreak in countries where there is a level 2 travel health notice for mpox
Healthcare workers being deployed to these regions should receive 2 doses of Imvamune® administered at least 28 days apart, in advance of deployment, given the heightened mpox epidemiology in these regions and potential increased risk of exposure to the virus.
Access to products
The PHAC National Emergency Strategic Stockpile (NESS) maintains a supply of vaccines and therapeutics and continues to work with provinces and territories to support collective preparedness and, as required, outbreak management. Health care facilities and local or regional health departments should direct requests for these vaccines and therapeutics to their respective provincial or territorial ministry of health.
For more information on these products, refer to the Product Monographs available on Health Canada's Drug Product Database.
Reporting an adverse reaction following vaccination
Healthcare providers are required to report possible reactions following vaccination to their local public health authority. The public health authority then reports them to PHAC.
Learn more about:
Infection prevention and control
Airborne, droplet and contact precautions are recommended in a healthcare setting.
For guidance on infection prevention and control for mpox, refer to the Interim guidance on infection prevention and control for patients with suspected, probable or confirmed mpox within healthcare settings – November 2024.
Surveillance
If you suspect a case of mpox, follow your provincial, territorial or local public health authority's reporting requirements. Ensure you're using the correct case report form for your jurisdiction.
Mechanisms are in place for provincial and territorial public health authorities to securely transfer their de-identified public health surveillance data to PHAC.
We're working with provincial and territorial public health partners to monitor cases of mpox in Canada. To learn more about this, refer to the Mpox: Update web page.
Download the case report form (PDF, 1,385 KB)
Evidence
To help health professionals and other decision makers respond to mpox, PHAC has commissioned the McMaster Health Forum to produce a rapid living evidence profile. The profile identifies, assesses the quality, categorizes and briefly summarizes the emerging scientific literature. It includes key findings on epidemiology, prevention and control, clinical presentation and treatment.
PHAC Expert Panel on Mpox in Canada
As part of the federal government's continued commitment to engaging scientific and public health expertise from diverse disciplinary perspectives and using the best available evidence, PHAC's Chief Public Health Officer of Canada and the Chief Science Officer have established PHAC's Expert Panel on Mpox in Canada.
Learn more about the PHAC Expert Panel on Mpox in Canada.
Webinars about mpox
There is a series of webinars about mpox in Canada that aim to improve awareness of the ongoing outbreak. These webinars are organized and hosted by the National Collaborating Centre for Infectious Diseases (NCCID) in partnership with PHAC and the Association of Medical Microbiology and Infectious Disease (AMMI) Canada.
These webinars are available for health professionals to watch on the NCCID website:
- mpox in Canada: what clinicians need to know
- mpox: Infection prevention and control
- mpox: Diagnostics
- mpox: Perspectives from the US
Additional resources
- Mpox: Public health management of cases and contacts in Canada
- Mpox vaccination clinic resources
- National case definition
- Mpox (monkeypox) and animals
Related links
- World Health Organization: Monkeypox fact sheet
- Centers for Disease Control and Prevention: Mpox
- European Centre for Disease Prevention and Control. Epidemiological update: Monkeypox multi-country outbreak. 25 May 2022
- World Health Organization: Emergency events, monkeypox
- World Health Organization: Weekly epidemiological record (WER) no.11, 16 March 2018, Emergence of monkeypox in West Africa and Central Africa 1970-2017
- World Health Organization: Monkeypox outbreak toolbox
- World Health Organization: Online training module: Mpox: Introductory course for African outbreak contexts
- World Health Organization: Online training module: Mpox: Epidemiology, preparedness and response for African outbreak contexts
Endnotes
- Endnotes 1
-
Vakaniaki EH, Kacita C, Kinganda-Lusamaki E, O'Toole Á, Wawina-Bokalanga T, Mukadi-Bamuleka D, et al. Sustained Human Outbreak of a New MPXV Clade I Lineage in the Eastern Democratic Republic of the Congo. Nature Medicine. 2024:1-11. Available at: https://www.nature.com/articles/s41591-024-03130-3