FluWatch report: November 11, 2018 to November 17, 2018 (Week 46)
Download the alternative format
(PDF format, 815 KB, 7 pages)
Organization: Public Health Agency of Canada
Date published: 2018-11-23
Related Topics
Overall Summary
- Influenza activity continued to increase in week 46. The influenza season has started earlier than in recent years.The number of regions in Canada reporting localized influenza activity in week 46 increased slightly compared to the previous week.
- Influenza A is the most common influenza virus circulating in Canada, and the majority of these viruses are A(H1N1)pdm09.
- The number of influenza-associated hospitalizations continued to increase in week 46. In particular, the number of pediatric hospitalizations is significantly higher than in recent years.
- The number of influenza outbreaks remains within expected levels. However, the settings in which outbreaks have been reported this season suggests that younger age-groups are being affected more than in previous seasons.
On this page
- Influenza/ILI Activity (geographic spread)
- Laboratory Confirmed Influenza Detections
- Syndromic/Influenza-like Illness Surveillance
- Participatory Syndromic Surveillance
- Influenza Outbreak Surveillance
- Severe Outcomes Influenza Surveillance
- Antiviral Resistance
- Provincial and International Influenza Reports
Influenza/Influenza-like Illness Activity (geographic spread)
During week 46, the following influenza activity levels increased slightly compared to the previous week (Figure 1):
- Five regions reported localized activity in Alta. (2) and Ont. (2), and Que. (1).
- Sporadic activity was reported by 25 regions, in B.C.(5), Alta.(3), Sask.(3), Man.(2), Ont.(2), Que.(5), N.B.(1), P.E.I.(1), N.L.(1), N.W.T(1) and Nvt.(1).
- No activity was reported by 23 regions.
Figure 1 – Map of overall influenza/ILI activity by province and territory, Canada, week 2018-46
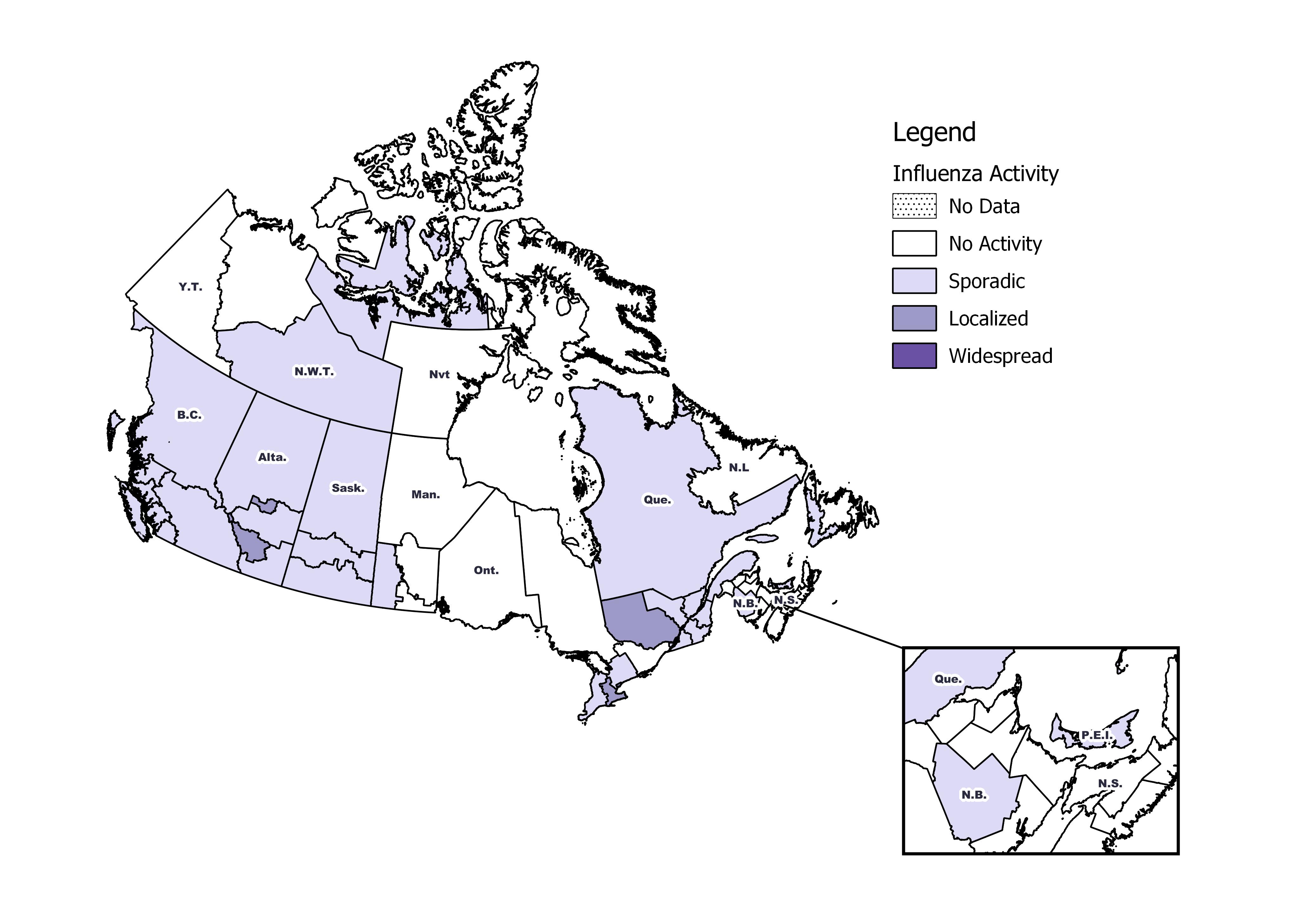
Figure 1 - Text description
| Province | Influenza Surveillance Region | Activity Level |
|---|---|---|
| N.L. | Eastern | No Activity |
| N.L. | Labrador-Grenfell | No Activity |
| N.L. | Central | No Activity |
| N.L. | Western | Sporadic |
| P.E.I. | Prince Edward Island | Sporadic |
| N.S. | Zone 1 - Western | No Activity |
| N.S. | Zone 2 - Northern | No Activity |
| N.S. | Zone 3 - Eastern | No Activity |
| N.S. | Zone 4 - Central | No Activity |
| N.B. | Region 1 | No Activity |
| N.B. | Region 2 | No Activity |
| N.B. | Region 3 | Sporadic |
| N.B. | Region 4 | No Activity |
| N.B. | Region 5 | No Activity |
| N.B. | Region 6 | No Activity |
| N.B. | Region 7 | No Activity |
| Que. | Nord-est | Sporadic |
| Que. | Québec et Chaudieres-Appalaches | Sporadic |
| Que. | Centre-du-Québec | Sporadic |
| Que. | Montréal et Laval | Sporadic |
| Que. | Ouest-du-Québec | Localized |
| Que. | Montérégie | Sporadic |
| Ont. | Central East | Sporadic |
| Ont. | Central West | Localized |
| Ont. | Eastern | No Activity |
| Ont. | North East | No Activity |
| Ont. | North West | No Activity |
| Ont. | South West | Sporadic |
| Ont. | Toronto | Localized |
| Man. | Northern Regional | No Activity |
| Man. | Prairie Mountain | Sporadic |
| Man. | Interlake-Eastern | No Activity |
| Man. | Winnipeg | Sporadic |
| Man. | Southern Health | No Activity |
| Sask. | North | Sporadic |
| Sask. | Central | Sporadic |
| Sask. | South | Sporadic |
| Alta. | North Zone | Sporadic |
| Alta. | Edmonton | Localized |
| Alta. | Central Zone | Sporadic |
| Alta. | Calgary | Localized |
| Alta. | South Zone | Sporadic |
| B.C. | Interior | Sporadic |
| B.C. | Fraser | Sporadic |
| B.C. | Vancouver Coastal | Sporadic |
| B.C. | Vancouver Island | Sporadic |
| B.C. | Northern | Sporadic |
| Y.T. | Yukon | No Activity |
| N.W.T. | North | No Activity |
| N.W.T. | South | Sporadic |
| Nvt. | Qikiqtaaluk | No Activity |
| Nvt. | Kivalliq | No Activity |
| Nvt. | Kitimeot | Sporadic |
Laboratory Confirmed Influenza Detections
In week 46, the following results were reported from sentinel laboratories across Canada (Figure 2):
- The percentage of tests positive for influenza continued to increase to 13.8% of tests positive.
- The percentage of tests positive for influenza A is higher for this time of year compared to the same period during the previous eight seasons.
- In week 46, 642 laboratory detections of influenza were reported, of which 99% were influenza A.
To date this season 2,063 laboratory-confirmed influenza detections have been reported (Figure 3):
- 98% have been influenza A
- Among the 1,203 influenza A viruses subtyped, 91% have been A(H1N1)pdm09
To date this season, detailed information on age and type/subtype has been received for 2,069 laboratory-confirmed influenza cases (Table 1):
- Adults 20-44 years of age represent the largest proportion of cases (25%), followed by individuals 5-19 years of age (20%).
For more detailed weekly and cumulative influenza data, see the text descriptions for Figures 2 and 3 or the Respiratory Virus Detections in Canada Report.
Figure 2 – Number of positive influenza tests and percentage of tests positive, by type, subtype and report week, Canada, 2018-35 to 2018-46
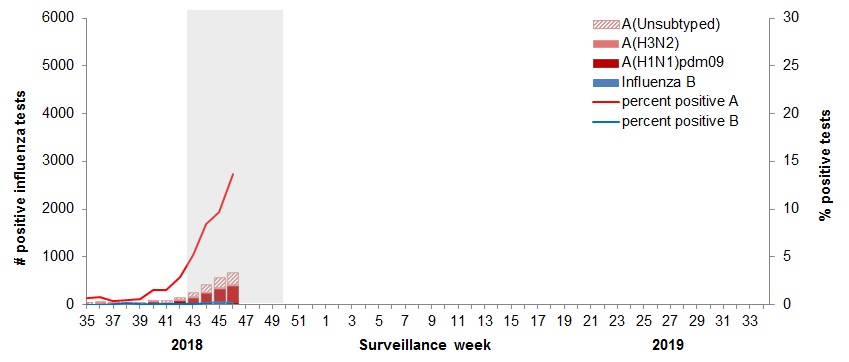
The shaded area indicates weeks where the positivity rate was at least 5% and a minimum of 15 positive tests were observed, signalling the start and end of seasonal influenza activity.
Figure 2 - Text description
| Surveillance Week | A(Unsubtyped) | A(H3) | A(H1)pdm09 | Influenza B |
|---|---|---|---|---|
| 35 | 3 | 2 | 7 | 0 |
| 36 | 4 | 7 | 4 | 0 |
| 37 | 3 | 2 | 3 | 1 |
| 38 | 6 | 3 | 2 | 3 |
| 39 | 11 | 5 | 1 | 3 |
| 40 | 16 | 7 | 29 | 3 |
| 41 | 27 | 6 | 21 | 3 |
| 42 | 40 | 19 | 55 | 2 |
| 43 | 83 | 23 | 128 | 4 |
| 44 | 170 | 13 | 213 | 6 |
| 45 | 201 | 17 | 276 | 15 |
| 46 | 249 | 10 | 375 | 8 |
Figure 3 – Cumulative numbers of positive influenza specimens by type/subtype and province/territory, Canada, weeks 2018-35 to 2018-46
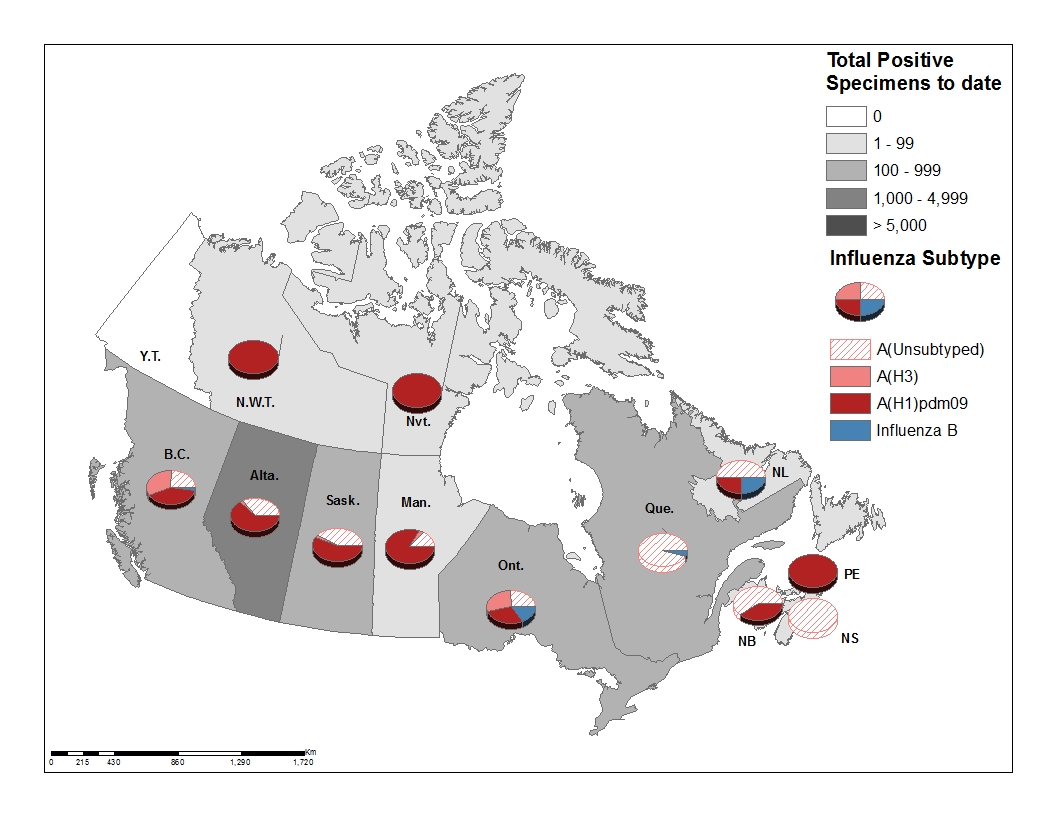
Figure 3 - Text description
| Reporting provincesTable Figure 3 - Footnote 1 |
Week (November 11, 2018 to November 17, 2018) | Cumulative (August 26, 2018 to November 17, 2018) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A | B | A & B Total |
|||||||
| A Total |
A (H1N1)pdm09 |
A (H3N2) |
A(UnS)Table Figure 3 - Footnote 3 | B Total |
A Total |
A (H1N1)pdm09 |
A (H3N2) |
A(UnS)Table Figure 3 - Footnote 3 | B Total |
||
| BC | 19 | 13 | 0 | 6 | 1 | 111 | 47 | 36 | 28 | 3 | 114 |
| AB | 451 | 293 | 3 | 155 | 1 | 1354 | 846 | 31 | 477 | 12 | 1366 |
| SK | 0 | 0 | 0 | 0 | 0 | 140 | 78 | 3 | 59 | 0 | 140 |
| MB | 18 | 9 | 0 | 9 | 0 | 75 | 62 | 3 | 10 | 0 | 75 |
| ON | 23 | 13 | 7 | 3 | 3 | 121 | 45 | 39 | 37 | 22 | 143 |
| QC | 70 | 0 | 0 | 70 | 3 | 190 | 0 | 0 | 190 | 10 | 200 |
| NB | 1 | 1 | 0 | 0 | 0 | 5 | 2 | 0 | 3 | 0 | 5 |
| NS | 1 | 0 | 0 | 1 | 0 | 6 | 0 | 0 | 6 | 0 | 6 |
| PEI | 1 | 1 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 3 |
| NL | 1 | 0 | 0 | 1 | 0 | 3 | 1 | 0 | 2 | 1 | 4 |
| YT | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| N.W.T | 4 | 4 | 0 | 0 | 0 | 4 | 4 | 0 | 0 | 0 | 4 |
| NU | 1 | 1 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 3 |
| Canada | 590 | 335 | 10 | 245 | 8 | 2015 | 1091 | 112 | 812 | 48 | 2063 |
| PercentageTable Figure 3 - Footnote 2 | 99% | 57% | 2% | 42% | 1% | 98% | 54% | 6% | 40% | 2% | 100% |
Discrepancies in values in Figures 2 and 3 may be attributable to differing data sources. Cumulative data includes updates to previous weeks. |
|||||||||||
| Age groups (years) | Cumulative (August 26, 2018 to November 17, 2018) | ||||||
|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A and B | |||||
| A Total | A(H1) pdm09 | A(H3) | A (UnS)Footnote 1 | Total | # | % | |
| 0-4 | 387 | 246 | 3 | 138 | 5 | 392 | 19% |
| 5-19 | 411 | 213 | 4 | 194 | 7 | 418 | 20% |
| 20-44 | 519 | 272 | 12 | 235 | 8 | 527 | 25% |
| 45-64 | 371 | 195 | 28 | 148 | 7 | 378 | 18% |
| 65+ | 342 | 140 | 124 | 78 | 12 | 354 | 17% |
| Total | 2034 | 1066 | 171 | 793 | 39 | 2069 | 100% |
|
|||||||
Syndromic/Influenza-like Illness Surveillance
Healthcare Professionals Sentinel Syndromic Surveillance
In week 46, 1.7% of visits to healthcare professionals were due to influenza-like illness (ILI) (Figure 4). The percentage of visits for ILI is within expected levels.
Figure 4 - Percentage of visits for ILI reported by sentinels by report week, Canada, weeks 2018-35 to 2018-46
Number of Sentinels Reporting in Week 46: 84
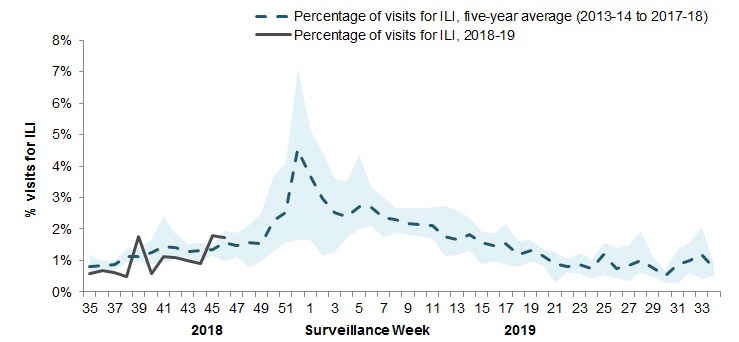
The shaded area represents the maximum and minimum percentage of visits for ILI reported by week from seasons 2013-14 to 2017-18
Figure 4 - Text description
| Report week | 2018-19 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0.6% | 0.6% | 0.4% | 0.9% |
| 36 | 0.7% | 0.8% | 0.5% | 1.0% |
| 37 | 0.6% | 0.9% | 0.7% | 1.1% |
| 38 | 0.5% | 1.0% | 0.7% | 1.4% |
| 39 | 1.8% | 1.1% | 0.8% | 1.5% |
| 40 | 0.6% | 1.6% | 0.9% | 3.4% |
| 41 | 1.1% | 1.6% | 1.1% | 2.1% |
| 42 | 1.2% | 1.4% | 0.8% | 1.8% |
| 43 | 1,0% | 1,4% | 0,7% | 1,8% |
| 44 | 0,9% | 1,4% | 1,1% | 1,6% |
| 45 | 1,8% | 1,5% | 1,2% | 1,8% |
| 46 | 1,7% | 1,7% | 1,1% | 2,2% |
Participatory Syndromic Surveillance
In week 46, 2,183 participants reported to FluWatchers, of which 43 (2.0%) reported symptoms of cough and fever (Figure 5).
Among the 43 participants who reported fever and cough:
- 16% consulted a healthcare professional
- 79% reported days missed from work or school, resulting in a combined total of 87 missed days of work or school.
Figure 5 – Percentage of participants reporting cough and fever, Canada, weeks 2018-40 to 2018-46
Number of Participants Reporting in Week 46: 2,183
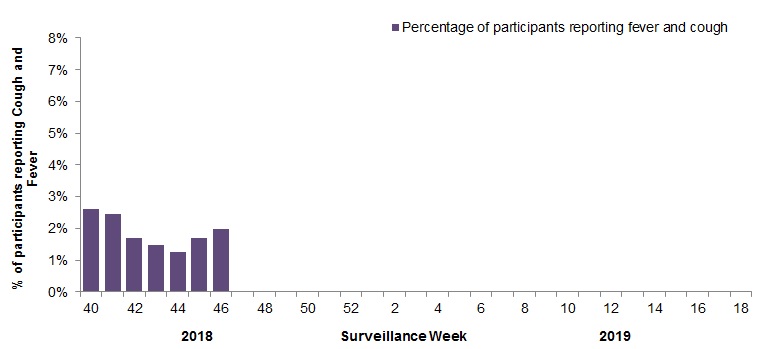
Figure 5 - Text description
| Surveillance week | % cough and fever |
|---|---|
| 40 | 2.6% |
| 41 | 2.5% |
| 42 | 1.7% |
| 43 | 1.5% |
| 44 | 1.3% |
| 45 | 1.7% |
| 46 | 2.0% |
Influenza Outbreak Surveillance
In week 46, five new laboratory-confirmed influenza outbreaks were reported in long-term care (3) and acute care (2) facilities.
To date this season, 26 influenza outbreaks have been reported (Figure 6):
- 14 outbreaks were in LTCF, five were in schools, and four were in other settings.
- All of the 23 outbreaks for which the influenza type was available were associated with influenza A.
- Among the 15 outbreaks for which the influenza A subtype was available:
- Eight were associated with influenza A(H1N1)pdm09, reported from schools, acute care facilities and other settings;
- Four were associated with A(H3N2), reported from LTCF and other settings;
- Three were associated with both A(H1N1)pdm09 and A(H3N2), reported from LTCF.
- Compared to previous seasons, a greater proportion of outbreaks have been reported in settings other than LTCF, suggesting an increased burden of disease in younger age-groups.
No new ILI outbreaks were reported in week 46.
To date this season, 23 ILI outbreaks have been reported; 21 occurred in LTCF, one in a school, and one in an acute care facility.
Figure 6 – Number of new outbreaks of laboratory-confirmed influenza by report week, Canada, weeks 2018-35 to 2018-46
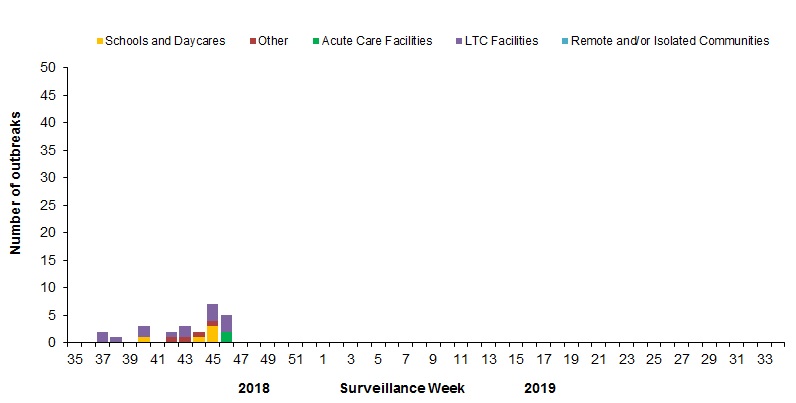
Figure 6 - Text description
| Surveillance Week | Acute Care Facilities | Long Term Care Facilities | Other | Schools and Daycares | Remote and/or Isolated Communities |
|---|---|---|---|---|---|
| 35 | 0 | 0 | 0 | 0 | 0 |
| 36 | 0 | 0 | 0 | 0 | 0 |
| 37 | 0 | 0 | 0 | 0 | 0 |
| 38 | 0 | 1 | 0 | 0 | 0 |
| 39 | 0 | 0 | 0 | 0 | 0 |
| 40 | 0 | 2 | 0 | 1 | 0 |
| 41 | 0 | 0 | 0 | 0 | 0 |
| 42 | 0 | 1 | 1 | 0 | 0 |
| 43 | 0 | 2 | 1 | 0 | 0 |
| 44 | 0 | 0 | 1 | 1 | 0 |
| 45 | 0 | 3 | 1 | 3 | 0 |
| 46 | 2 | 3 | 0 | 0 | 0 |
Severe Outcomes Influenza Surveillance
Provincial/Territorial Influenza Hospitalizations and Deaths
In week 46, 62 influenza-associated hospitalizations were reported, which is a slight increase compared to the previous week.
To date this season, mothe than 300 influenza-assocaited hospitalizations have been reported by participating provinces and territoriesFootnote 1.
Hospitalizations (Table 2):
- 99% (310) were associated with influenza A
- The highest estimated rates of hospitalization are among children under 5 years of age, and adults 65 years of age and older.
Intensive Care Unit (ICU) cases and deaths:
- To date this season 36 ICU admissions and <5 deaths have been reported.
| Age Groups | Cumulative (August 26, 2018 to November 17, 2018) | ||
|---|---|---|---|
| Influenza A | Influenza B | Rate per 100,000 population | |
| 0-4 | 61 | 10 | 7.77 |
| 5-19 | 39 | 0 | 2.31 |
| 20-44 | 47 | 0 | 1.20 |
| 45-64 | 79 | 0 | 2.39 |
| 65+ | 84 | <5 | 5.18 |
| Total | 310 | <5 | |
| % | 99% | 1% | |
Pediatric Influenza Hospitalizations and Deaths
TIn week 46, 39 pediatric (≤16 years of age) hospitalizations with influenza have been reported by the Immunization Monitoring Program Active (IMPACT) network. Pediatric hospitalizations reported by IMPACT are at levels not normally seen until late December (4-6 weeks earlier than in recent seasons)(Figure 7).
To date this season, 108 pediatric hospitalizations have been reported (Figure 8):
- All but one case have been associated with influenza A.
- Among the 83 cases for which the influenza subtype was available, 82 were associated with A(H1N1)pdm09.
- 91 of the 108 cases (84%) were between 6 months and 9 years of age.
To date this season, 10 ICU admissions, and no deaths have been reported.
Figure 7 – Number of pediatric (≤16 years of age) hospitalizations reported by the IMPACT network, by week, Canada, weeks 2018-35 to 2018-46

Figure 7 - Text description
| Surveillance week | 2018-19 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0 | 0 | 0 | 1 |
| 36 | 0 | 0 | 0 | 1 |
| 37 | 0 | 0 | 0 | 2 |
| 38 | 0 | 1 | 0 | 2 |
| 39 | 0 | 1 | 0 | 3 |
| 40 | 1 | 0 | 0 | 2 |
| 41 | 1 | 1 | 0 | 3 |
| 42 | 6 | 1 | 0 | 4 |
| 43 | 11 | 1 | 0 | 3 |
| 44 | 19 | 3 | 1 | 6 |
| 45 | 32 | 3 | 2 | 4 |
| 46 | 39 | 5 | 1 | 13 |
Figure 8 - Cumulative numbers of pediatric hospitalizations (≤16 years of age) with influenza by age-group reported by the IMPACT network, Canada, weeks 2018-35 to 2018-46

Figure 8 - Text description
| Age Group | Total |
|---|---|
| 0-5 mo | 11 |
| 6-23 mo | 30 |
| 2-4 yr | 29 |
| 5-9 yr | 32 |
| 10-16 yr | 6 |
Adult Influenza Hospitalizations and Deaths
Surveillance of laboratory-confirmed influenza-associated adult (≥16 years of age) hospitalizations by the Canadian Immunization Research Network (CIRN) Serious Outcomes Surveillance (SOS) network began on November 1st for the 2018-19 season.
To date this season, seven hospitalizations have been reported, of which six were associated with influenza A and one with influenza B.
Influenza Strain Characterizations
Since September 1, 2018, the National Microbiology Laboratory (NML) has characterized 60 influenza viruses (11 A(H3N2), 48 A(H1N1) and one B) that were received from Canadian laboratories.
Genetic Characterization of Influenza A (H3N2):
Ten influenza A(H3N2) viruses did not grow to sufficient hemagglutination titer for antigenic characterization by hemagglutination inhibition (HI) assay. Therefore, NML has performed genetic characterization to determine the genetic group identity of these viruses.
Sequence analysis of the HA gene of the viruses showed that:
- One virus belonged to genetic group 3C.2a.
- Nine viruses belonged to subclade 3C.2a1.
A/Singapore/INFIMH-16-0019/2016-like virus belongs to genetic group 3C.2a1 and is the influenza A(H3N2) component of the 2018-19 Northern Hemisphere influenza vaccine
Antigenic Characterization:
Influenza A(H3N2):
- One influenza A(H3N2) virus was antigenically characterized as A/Singapore/INFIMH-16-0019/2016-like by HI testing using antiserum raised against egg-propagated A/Singapore/INFIMH-16-0019/2016.
- A/Singapore/INFIMH-16-0019/2016-like virus is the influenza A(H3N2) component of the 2018-19 Northern Hemisphere influenza vaccine.
- The influenza A (H3N2) virus characterized belonged to genetic group 3C.2a1.
Influenza A(H1N1):
- 48 A(H1N1) viruses characterized were antigenically similar to A/Michigan/45/2015, which is the influenza A(H1N1) component of the 2018-19 Northern Hemisphere influenza vaccine.
Influenza B:
Influenza B viruses can be divided into two antigenically distinct lineages represented by B/Yamagata/16/88 and B/Victoria/2/87 viruses. The recommended influenza B components for the 2018-19 Northern Hemisphere influenza vaccine are B/Colorado/06/2017 (Victoria lineage) and B/Phuket/3073/2013 (Yamagata lineage).
- One influenza B viruses was characterized as B/Phuket/3073/2013-like, which belongs to the Yamagata lineage and is included as an influenza B component of the 2018-19 Northern Hemisphere quadrivalent influenza vaccine.
Antiviral Resistance
Antiviral Resistance – Amantadine:
59 influenza A (11 A(H3N2) and 48 A(H1N1)) viruses were tested for resistance to amantadine and it was found that:
- The 11 A(H3N2) viruses tested were resistant to amantadine.
- The 48 A(H1N1) viruses tested were resistant to amantadine.
Antiviral Resistance – Oseltamivir:
59 influenza viruses (10 A(H3N2), 48 A(H1N1) and 1 B) were tested for resistance to oseltamivir and it was found that:
- The 10 A(H3N2) viruses tested were sensitive to oseltamivir
- The 48 A(H1N1) viruses tested were sensitive to oseltamivir
- The B virus was sensitive to oseltamivir
Antiviral Resistance – Zanamivir:
59 influenza viruses (10 A(H3N2), 48 H1N1 and 1 B) were tested for resistance to zanamivir and it was found that:
- The 10 A(H3N2) viruses were sensitive to zanamivir.
- The 48 A(H1N1) viruses were sensitive to zanamivir.
- The B virus was sensitive to zanamivir.