FluWatch report: February 16 to 22, 2020 (week 08)

Download the alternative format
(PDF format, 1.46 MB, 14 pages)
Organization: Public Health Agency of Canada
Date published: 2020-02-28
Related Topics
Overall Summary
- Influenza activity remained high in week 08; the majority of indicators remained similar or increased slightly from the previous week.
- Influenza A(H1N1) is currently the dominant influenza A subtype circulating in Canada, representing 78% of subtyped influenza A specimens in week 08.
- The highest cumulative hospitalization rates are among children under 5 years of age and adults 65 years of age and older.
- The Sentinel Practitioner Surveillance Network (SPSN) has published interim estimates of vaccine effectiveness which indicate that the 2019/2020 influenza vaccine has provided substantial protection against medically-attended influenza illness in the early part of the season in Canada. Within-season vaccine effectiveness (VE) was estimated to be 58% for any influenza, 44% for influenza A(H1N1), 62% for influenza A(H3N2), and 69% for influenza B.
On this page
- Influenza/ILI Activity (geographic spread)
- Laboratory Confirmed Influenza Detections
- Syndromic/Influenza-like Illness Surveillance
- FluWatchers
- Influenza Outbreak Surveillance
- Severe Outcomes Influenza Surveillance
- Influenza Strain Characterizations
- Antiviral Resistance
- Vaccine Monitoring
- Provincial and International Influenza Reports
Influenza/Influenza-like Illness (ILI) Activity (geographic spread)
During week 08, influenza activity was reported in all regions in all reporting provinces and territories. Among these regions, 55% reported sporadic activity, 43% reported localized activity, and 2% reported widespread activity (Figure 1).
Figure 1 – Map of influenza/ILI activity by province and territory, Canada, week 2020-08
Number of Regions Reporting in Week 08: 53 out of 53

Figure 1 - Text equivalent
| Province | Influenza Surveillance Region | Activity Level |
|---|---|---|
| N.L. | Eastern | Sporadic |
| N.L. | Labrador-Grenfell | Localized |
| N.L. | Central | Localized |
| N.L. | Western | Sporadic |
| P.E.I. | Prince Edward Island | Localized |
| N.S. | Zone 1 - Western | Localized |
| N.S. | Zone 2 - Northern | Localized |
| N.S. | Zone 3 - Eastern | Sporadic |
| N.S. | Zone 4 - Central | Localized |
| N.B. | Region 1 | Sporadic |
| N.B. | Region 2 | Localized |
| N.B. | Region 3 | Localized |
| N.B. | Region 4 | Sporadic |
| N.B. | Region 5 | Sporadic |
| N.B. | Region 6 | Localized |
| N.B. | Region 7 | Localized |
| Que. | Nord-est | Localized |
| Que. | Québec et Chaudieres-Appalaches | Sporadic |
| Que. | Centre-du-Québec | Localized |
| Que. | Montréal et Laval | Localized |
| Que. | Ouest-du-Québec | Localized |
| Que. | Montérégie | Sporadic |
| Ont. | Central East | Localized |
| Ont. | Central West | Localized |
| Ont. | Eastern | Localized |
| Ont. | North East | Localized |
| Ont. | North West | Sporadic |
| Ont. | South West | Localized |
| Ont. | Toronto | Localized |
| Man. | Northern Regional | Sporadic |
| Man. | Prairie Mountain | Sporadic |
| Man. | Interlake-Eastern | Sporadic |
| Man. | Winnipeg | Sporadic |
| Man. | Southern Health | Sporadic |
| Sask. | North | Sporadic |
| Sask. | Central | Sporadic |
| Sask. | South | Localized |
| Alta. | North Zone | Sporadic |
| Alta. | Edmonton | Sporadic |
| Alta. | Central Zone | Localized |
| Alta. | Calgary | Sporadic |
| Alta. | South Zone | Sporadic |
| B.C. | Interior | Sporadic |
| B.C. | Fraser | Sporadic |
| B.C. | Vancouver Coastal | Localized |
| B.C. | Vancouver Island | Widespread |
| B.C. | Northern | Sporadic |
| Y.T. | Yukon | Sporadic |
| N.W.T. | North | Sporadic |
| N.W.T. | South | Sporadic |
| Nvt. | Qikiqtaaluk | Sporadic |
| Nvt. | Kivalliq | Sporadic |
| Nvt. | Kitimeot | Sporadic |
Laboratory-Confirmed Influenza Detections
In week 08, the percentage of laboratory tests positive for influenza was similar to the previous three weeks at 29% and remains similar to the peak percentages reported since late December. Influenza A and B continue to co-circulate.
The following results were reported from sentinel laboratories across Canada (Figures 2 and 3):
- The percentage of tests positive for influenza B was 12% in week 08. This continues to be two times the average (5.6%) for this time of year.
- The percentage of tests positive for influenza A was 17% in week 08, which is similar to the previous six weeks, and below average for this time of year.
- Among subtyped influenza A detections, influenza A(H1N1) accounted for 78% of detections, a slight decrease in proportion to the previous two weeks.
To date this season (weeks 35 to 08), 42,291 laboratory detections of influenza were reported
- 57% (24,139) were influenza A.
- Among subtyped influenza A detections (5,610), A(H1N1) is the predominant subtype this season (65%).
Detailed information on age and type/subtype has been received for 32,801 laboratory-confirmed influenza cases (Table 1). To date this season (weeks 35 to 08):
- Among cases of influenza A(H3N2) (1,735), the largest proportion was in adults 65 years of age and older (46%).
- Cases of influenza B (14,856) were primarily in younger age groups; 57% of cases were under 19 years of age and 30% between 20 and 44 years of age.
- Among cases of influenza A(H1N1) (2,609), 30% of cases were in adults 65 years of age and older, with approximately equal proportions in adults 20-44 years and 45-64 years (~25%).
For more detailed weekly and cumulative influenza data, see the text descriptions for Figures 2 and 3 or the Respiratory Virus Detections in Canada Report.
Figure 2 - Number of positive influenza tests and percentage of tests positive, by type, subtype and report week, Canada, weeks 2019-35 to 2020-08
Number of Laboratories Reporting in Week 08: 34 out of 36
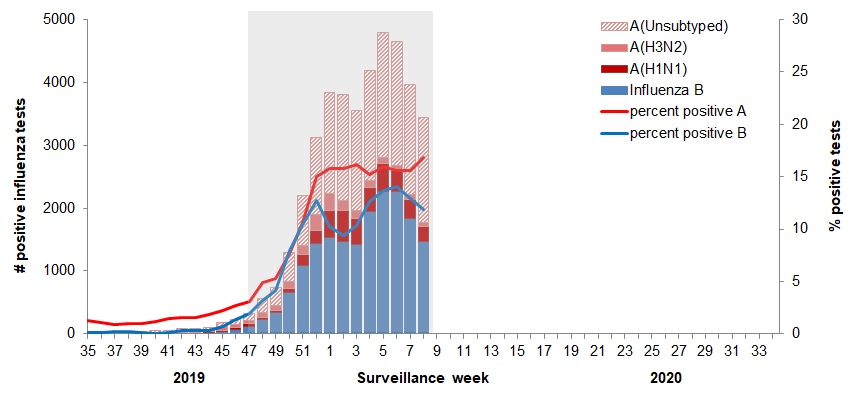
The shaded area indicates weeks where the positivity rate was at least 5% and a minimum of 15 positive tests were observed, signalling the period of seasonal influenza activity.
Figure 2 - Text equivalent
| Surveillance Week | A(Unsubtyped) | A(H3N2) | A(H1N1)pdm09 | Influenza B | Percent Positive A | Percent Positive B |
|---|---|---|---|---|---|---|
| 35 | 10 | 16 | 0 | 2 | 1.3 | 0.1 |
| 36 | 11 | 13 | 2 | 2 | 1.1 | 0.1 |
| 37 | 5 | 17 | 2 | 5 | 0.9 | 0.2 |
| 38 | 11 | 15 | 3 | 6 | 1.0 | 0.2 |
| 39 | 11 | 21 | 2 | 3 | 1.0 | 0.1 |
| 40 | 34 | 9 | 1 | 2 | 1.2 | 0.1 |
| 41 | 34 | 18 | 0 | 5 | 1.4 | 0.1 |
| 42 | 54 | 12 | 1 | 14 | 1.6 | 0.3 |
| 43 | 44 | 13 | 7 | 17 | 1.6 | 0.3 |
| 44 | 43 | 23 | 16 | 17 | 1.8 | 0.3 |
| 45 | 57 | 57 | 20 | 39 | 2.2 | 0.7 |
| 46 | 82 | 43 | 23 | 77 | 2.7 | 1.4 |
| 47 | 118 | 49 | 33 | 124 | 3.1 | 1.9 |
| 48 | 225 | 67 | 42 | 223 | 4.9 | 3.2 |
| 49 | 281 | 79 | 41 | 336 | 5.3 | 4.1 |
| 50 | 463 | 100 | 73 | 654 | 7.7 | 8.0 |
| 51 | 794 | 149 | 169 | 1094 | 10.6 | 10.4 |
| 52 | 1223 | 267 | 197 | 1439 | 15.0 | 12.7 |
| 1 | 1620 | 261 | 431 | 1533 | 15.8 | 10.3 |
| 2 | 1690 | 165 | 493 | 1463 | 15.8 | 9.4 |
| 3 | 1587 | 131 | 414 | 1418 | 16.2 | 10.3 |
| 4 | 1736 | 126 | 368 | 1952 | 15.2 | 12.7 |
| 5 | 1987 | 96 | 437 | 2269 | 16.0 | 13.7 |
| 6 | 1982 | 70 | 337 | 2265 | 15.6 | 14.1 |
| 7 | 1742 | 74 | 303 | 1841 | 15.6 | 12.9 |
| 8 | 1675 | 67 | 241 | 1466 | 16.8 | 11.8 |
Figure 3 – Distribution of positive influenza specimens by type/subtype and province/territoryFootnote *, Canada, weeks 2019-35 to 2020-08
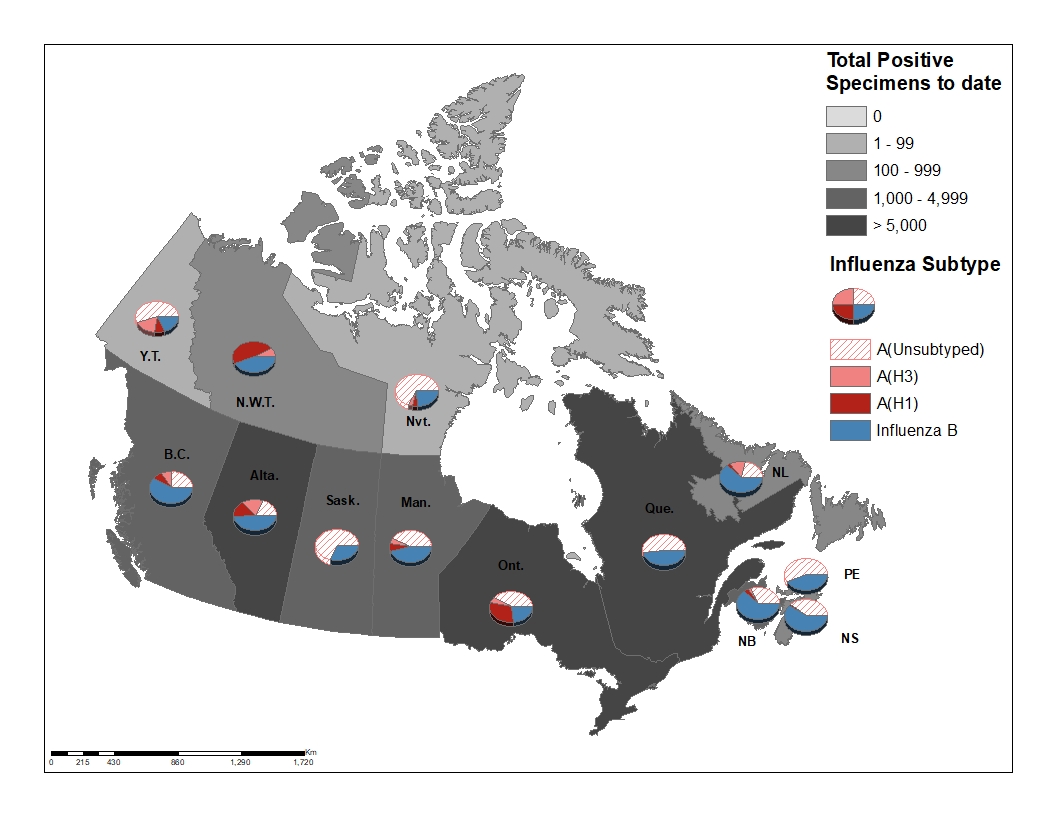
- Footnote *
-
Specimens from NWT, YT, and Nvt are sent to reference laboratories in other provinces.
Figure 3 - Text equivalent
| ProvincesTable Figure 3 - Footnote 1 | Cumulative (August 25, 2019 to February 22, 2020) | |||||
|---|---|---|---|---|---|---|
| A Total | A(H1N1) | A(H3N2) | A(UnS)Table Figure 3 - Footnote 3 | B Total | A & B Total | |
| B.C. | 2119 | 220 | 235 | 747 | 1555 | 3674 |
| Alta. | 3256 | 990 | 1162 | 1104 | 2972 | 6228 |
| Sask. | 1196 | 0 | 0 | 1196 | 556 | 1752 |
| Man. | 1061 | 129 | 102 | 830 | 811 | 1872 |
| Ont. | 5625 | 2208 | 383 | 3034 | 1841 | 7466 |
| Que. | 9948 | 0 | 0 | 9948 | 9054 | 19002 |
| N.B. | 550 | 36 | 22 | 492 | 900 | 1450 |
| N.S. | 77 | 2 | 1 | 74 | 123 | 200 |
| P.E.I. | 76 | 0 | 0 | 76 | 60 | 136 |
| N.L. | 79 | 4 | 31 | 44 | 179 | 258 |
| Y.T. | 42 | 4 | 8 | 30 | 8 | 50 |
| N.W.T | 68 | 57 | 10 | 1 | 74 | 142 |
| Nvt. | 42 | 3 | 3 | 36 | 19 | 61 |
| Canada | 24139 | 3653 | 1957 | 17612 | 18152 | 42291 |
| PercentageTable Figure 3 - Footnote 2 | 57% | 15% | 8% | 73% | 43% | 100% |
|
||||||
| Age groups (years) |
Cumulative (August 25, 2019 to February 22, 2020) | ||||||
|---|---|---|---|---|---|---|---|
| Influenza A | B | Influenza A and B | |||||
| A Total | A(H1N1) | A(H3N2) | A (Un subtyped)Table 1 Footnote 1 | Total | # | % | |
| 0-4 | 2901 | 305 | 172 | 2424 | 3423 | 6324 | 19% |
| 5-19 | 2004 | 186 | 224 | 1594 | 5108 | 7112 | 22% |
| 20-44 | 3797 | 663 | 280 | 2854 | 4443 | 8240 | 25% |
| 45-64 | 3508 | 666 | 258 | 2584 | 833 | 4341 | 13% |
| 65+ | 5735 | 789 | 801 | 4145 | 1049 | 6784 | 21% |
| Total | 17945 | 2609 | 1735 | 13601 | 14856 | 32801 | 100% |
|
|||||||
Syndromic / Influenza-like Illness Surveillance
Healthcare Professionals Sentinel Syndromic Surveillance
In week 08, 1.5% of visits to healthcare professionals were due to influenza-like illness (ILI) which is below the average for this time of year (Figure 4).
Figure 4 – Percentage of visits for ILI reported by sentinels by report week, Canada, weeks 2019-35 to 2020-08
Number of Sentinels Reporting in Week 08: 79
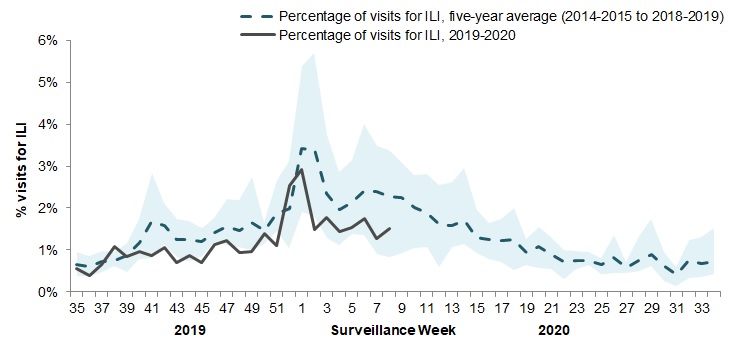
The shaded area represents the maximum and minimum percentage of visits for ILI reported by week from seasons 2014-2015 to 2018-2019
Figure 4 - Text equivalent
| Surveillance Week | 2019-20 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0.6% | 0.6% | 0.4% | 0.9% |
| 36 | 0.4% | 0.6% | 0.4% | 0.9% |
| 37 | 0.7% | 0.7% | 0.5% | 1.0% |
| 38 | 1.1% | 0.7% | 0.6% | 1.0% |
| 39 | 0.8% | 0.9% | 0.5% | 1.2% |
| 40 | 1.0% | 1.2% | 0.8% | 1.7% |
| 41 | 0.9% | 1.7% | 0.8% | 2.8% |
| 42 | 1.1% | 1.6% | 1.2% | 2.1% |
| 43 | 0.7% | 1.2% | 0.8% | 1.7% |
| 44 | 0.9% | 1.2% | 0.7% | 1.7% |
| 45 | 0.7% | 1.2% | 0.9% | 1.5% |
| 46 | 1.1% | 1.4% | 1.2% | 1.8% |
| 47 | 1.2% | 1.6% | 1.1% | 2.2% |
| 48 | 0.9% | 1.5% | 1.1% | 2.2% |
| 49 | 1.0% | 1.7% | 1.0% | 2.8% |
| 50 | 1.4% | 1.5% | 1.1% | 1.7% |
| 51 | 1.1% | 1.9% | 1.4% | 2.7% |
| 52 | 2.5% | 2.0% | 1.0% | 3.1% |
| 1 | 2.9% | 3.4% | 1.9% | 5.4% |
| 2 | 1.5% | 3.4% | 1.8% | 5.7% |
| 3 | 1.8% | 2.3% | 1.3% | 3.7% |
| 4 | 1.4% | 2.0% | 1.1% | 2.9% |
| 5 | 1.5% | 2.1% | 1.4% | 3.1% |
| 6 | 1.8% | 2.4% | 1.4% | 4.0% |
| 7 | 1.3% | 2.4% | 0.9% | 3.5% |
| 8 | 1.5% | 2.3% | 0.8% | 3.4% |
FluWatchers
The proportion of FluWatchers participants reporting symptoms of cough and fever increased in week 08 compared to the previous week. In week 08, 3,131 participants reported to FluWatchers, of which 2.8% (86) reported symptoms of cough and fever (Figure 5).
Among the 86 participants who reported cough and fever:
- 20% consulted a healthcare professional;
- 78% reported days missed from work or school, resulting in a combined total of 215 missed days of work or school.
If you are interested in becoming a FluWatcher, sign up today.
Figure 5 - Percentage of participants reporting fever and cough, Canada, weeks 2019-40 to 2020-08
Number of Participants Reporting in Week 08: 3,131
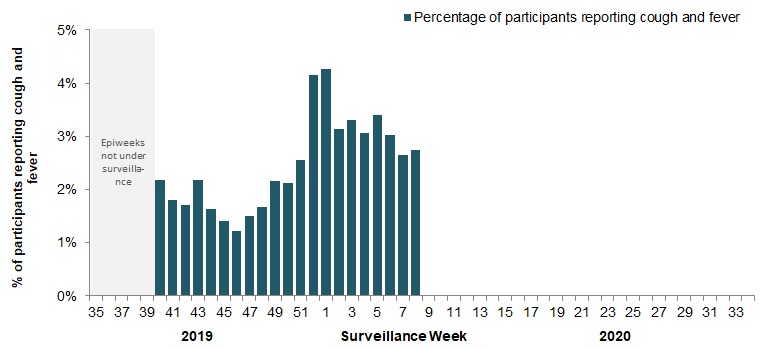
Figure 5 - Text equivalent
| Surveillance Week | % cough and fever |
|---|---|
| 40 | 2.2% |
| 41 | 1.8% |
| 42 | 1.7% |
| 43 | 2.2% |
| 44 | 1.6% |
| 45 | 1.4% |
| 46 | 1.2% |
| 47 | 1.5% |
| 48 | 1.7% |
| 49 | 2.2% |
| 50 | 2.1% |
| 51 | 2.6% |
| 52 | 4.1% |
| 1 | 4.3% |
| 2 | 3.1% |
| 3 | 3.3% |
| 4 | 3.1% |
| 5 | 3.4% |
| 6 | 3.0% |
| 7 | 2.7% |
| 8 | 2.7% |
Online Figure – Geographic distribution of FluWatchers participants reporting cough and fever, Canada, week 2020-08
Click on the map to access the link
Influenza Outbreak Surveillance
In week 08, a total of 30 outbreaks were reported: 22 in long term care facilities, three in facilities categorized as ‘other’, which includes facilities such as private personal care homes, correctional facilities, and colleges/universities, four in acute care facilities and one in a school/daycare (Figure 6). In addition, 19 ILI outbreaks were reported: 17 in schools/daycares and 2 in facilities categorized as ‘other’ .
To date this season, a total of 757 laboratory-confirmed influenza outbreaks have been reported; 62% (473) in long-term care facilities, 25% (190) in facilities categorized as ‘other’, 10% (79) in acute care facilities, and 2% (15) in schools/daycares. Of the 720 outbreaks where influenza type was reported, 88% (632) were due to influenza A. Among the 272 outbreaks for which the influenza A subtype was reported, 50% were associated with A(H3N2) and 50% were associated with A(H1N1). To date this season, 140 ILI outbreaks have also been reported; 98%(137) in schools/daycares and 2%(3) in facilities categorized as ‘other’.
Figure 6 – Number of new outbreaks of laboratory-confirmed influenza by report week, Canada, weeks 2019-35 to 2020-08
Number of provinces and territories reporting in week 08: 13 out of 13
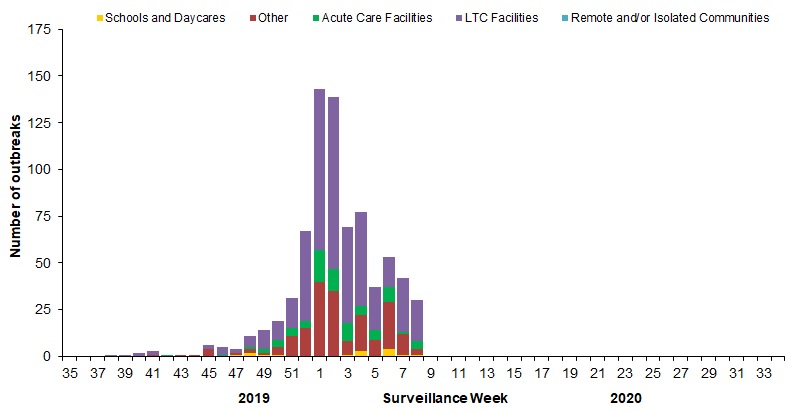
Figure 6 - Text equivalent
| Surveillance Week | Acute Care Facilities | Long Term Care Facilities | Other | Schools and Daycares | Remote and/or Isolated Communities |
|---|---|---|---|---|---|
| 35 | 0 | 0 | 0 | 0 | 0 |
| 36 | 0 | 0 | 0 | 0 | 0 |
| 37 | 0 | 0 | 0 | 0 | 0 |
| 38 | 0 | 1 | 0 | 0 | 0 |
| 39 | 0 | 1 | 0 | 0 | 0 |
| 40 | 0 | 2 | 0 | 0 | 0 |
| 41 | 0 | 2 | 1 | 0 | 0 |
| 42 | 1 | 0 | 0 | 0 | 0 |
| 43 | 0 | 0 | 1 | 0 | 0 |
| 44 | 0 | 0 | 1 | 0 | 0 |
| 45 | 0 | 2 | 4 | 0 | 0 |
| 46 | 1 | 4 | 0 | 0 | 0 |
| 47 | 0 | 2 | 1 | 1 | 0 |
| 48 | 1 | 6 | 2 | 2 | 0 |
| 49 | 2 | 10 | 1 | 1 | 0 |
| 50 | 4 | 10 | 4 | 1 | 0 |
| 51 | 4 | 16 | 11 | 0 | 0 |
| 52 | 4 | 48 | 15 | 0 | 0 |
| 1 | 17 | 86 | 40 | 0 | 0 |
| 2 | 12 | 92 | 35 | 0 | 0 |
| 3 | 10 | 51 | 7 | 1 | 0 |
| 4 | 5 | 50 | 19 | 3 | 0 |
| 5 | 5 | 23 | 9 | 0 | 0 |
| 6 | 8 | 16 | 25 | 4 | 0 |
| 7 | 1 | 29 | 11 | 1 | 0 |
| 8 | 4 | 22 | 3 | 1 | 0 |
Severe Outcomes Influenza Surveillance
Provincial/Territorial Influenza Hospitalizations and Deaths
To date this season, 1,795 influenza-associated hospitalizations were reported by participating provinces and territories1.
- 66% of the cases were associated with influenza A.
- Of the 826 cases for which subtype was reported, 60% were associated with influenza A(H3N2).
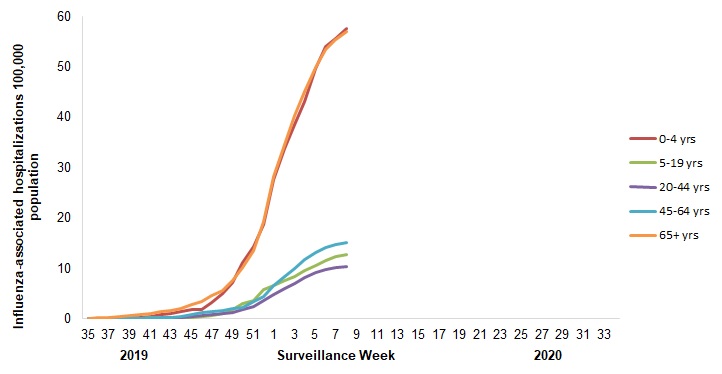
- The highest cumulative hospitalization rates up to week 08 were among children under 5 years of age and adults 65 years of age and older (57/100,000 population).
211 ICU admissions and 74 deaths have been reported.
- 65% of the ICU admissions and 69% of the deaths were associated with influenza A.
Figure 7 – Cumulative rates of influenza-associated hospitalizations by age-group and surveillance week, Canada, participating provinces and territoriesFootnote 1 weeks 2019-35 to 2020-08
Number of provinces and territories reporting in week 08: 9 out of 9
- Footnote ‡
-
Influenza-associated hospitalizations are reported by Alberta, Manitoba, New Brunswick, Newfoundland and Labrador, Northwest Territories, Nova Scotia, Prince Edward Island and Yukon. Only hospitalizations that require intensive medical care are reported by Saskatchewan.
Figure 7 - Text equivalent
| Surveillance Week | 0-4 yrs | 5-19 yrs | 20-44 yrs | 45-64 yrs | 65+ yrs |
|---|---|---|---|---|---|
| 35 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| 36 | 0.0 | 0.0 | 0.0 | 0.0 | 0.2 |
| 37 | 0.0 | 0.0 | 0.0 | 0.1 | 0.2 |
| 38 | 0.0 | 0.0 | 0.0 | 0.1 | 0.4 |
| 39 | 0.2 | 0.0 | 0.0 | 0.1 | 0.6 |
| 40 | 0.2 | 0.0 | 0.0 | 0.1 | 0.8 |
| 41 | 0.4 | 0.2 | 0.1 | 0.2 | 1.1 |
| 42 | 0.8 | 0.2 | 0.1 | 0.2 | 1.3 |
| 43 | 1.0 | 0.2 | 0.2 | 0.3 | 1.6 |
| 44 | 1.5 | 0.2 | 0.2 | 0.5 | 2.1 |
| 45 | 1.9 | 0.4 | 0.4 | 0.8 | 2.8 |
| 46 | 1.9 | 0.4 | 0.6 | 1.1 | 3.3 |
| 47 | 3.1 | 0.6 | 0.8 | 1.5 | 4.6 |
| 48 | 5.0 | 1.1 | 1.0 | 1.6 | 5.7 |
| 49 | 7.1 | 1.9 | 1.3 | 2.0 | 7.5 |
| 50 | 11.1 | 3.0 | 1.8 | 2.3 | 10.2 |
| 51 | 14.3 | 3.7 | 2.5 | 3.5 | 13.6 |
| 52 | 18.7 | 5.8 | 3.7 | 4.5 | 19.3 |
| 1 | 27.7 | 6.5 | 4.8 | 6.6 | 28.1 |
| 2 | 33.8 | 7.6 | 5.9 | 8.4 | 34.5 |
| 3 | 38.6 | 8.4 | 6.9 | 10.0 | 40.4 |
| 4 | 43.0 | 9.6 | 8.1 | 11.8 | 45.1 |
| 5 | 49.7 | 10.6 | 9.1 | 13.2 | 49.9 |
| 6 | 53.9 | 11.5 | 9.8 | 14.2 | 53.5 |
| 7 | 55.6 | 12.3 | 10.2 | 14.8 | 55.3 |
| 8 | 57.5 | 12.8 | 10.4 | 15.1 | 56.9 |
Pediatric Influenza Hospitalizations and Deaths
In week 08, 84 pediatric (≤16 years of age) laboratory-confirmed influenza-associated hospitalizations were reported by the Immunization Monitoring Program Active (IMPACT) network (Figure 8). The number of cases due to influenza B remains high; however, in recent weeks, a growing proportion of cases have been due to influenza A.
The elevated number of cases this season compared to previous seasons is likely due to the concurrent circulation of influenza A and B. The number of influenza A-associated pediatric hospitalizations is above the average for this time of year which is expected when A(H1N1) is the predominant circulating virus. The number of hospitalizations with influenza B remains well above average compared to previous seasons.
To date this season (weeks 35 to 08):
- 1037 pediatric hospitalizations have been reported by the IMPACT network, of which 51% (531) were associated with influenza A and 49% (506) with influenza B.
- The largest proportion of hospitalizations (65%) were among children under 5 years of age (Figure 9).
- 155 ICU admissions were reported, of which 52% were associated with influenza A.
- Less than five pediatric deaths have been reported.
Figure 8 – Number of pediatric (≤16 years of age) hospitalizations reported by the IMPACT network, by week, Canada, weeks 2019-35 to 2020-08
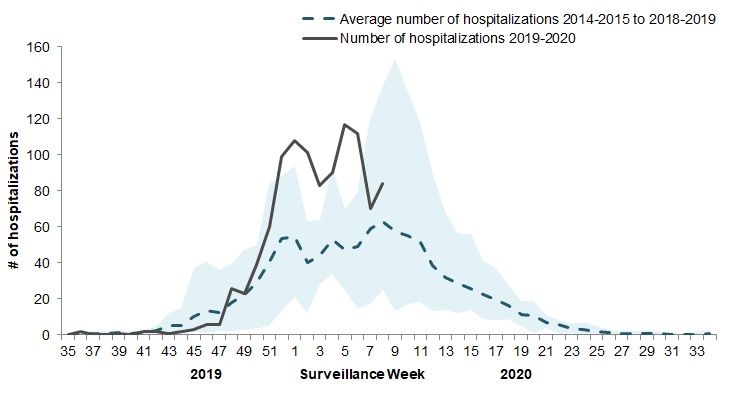
The shaded area represents the maximum and minimum number of cases reported by week from seasons 2014-15 to 2018-19
Figure 8 - Text equivalent
| Surveillance week | 2019-2020 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0 | 0 | 0 | 1 |
| 36 | 2 | 0 | 0 | 1 |
| 37 | 0 | 1 | 0 | 2 |
| 38 | 0 | 1 | 0 | 2 |
| 39 | 1 | 1 | 0 | 3 |
| 40 | 0 | 1 | 0 | 2 |
| 41 | 2 | 1 | 0 | 3 |
| 42 | 2 | 2 | 0 | 5 |
| 43 | 1 | 5 | 2 | 12 |
| 44 | 2 | 5 | 1 | 15 |
| 45 | 3 | 10 | 2 | 37 |
| 46 | 6 | 13 | 1 | 41 |
| 47 | 6 | 13 | 2 | 36 |
| 48 | 26 | 18 | 2 | 40 |
| 49 | 23 | 22 | 3 | 47 |
| 50 | 39 | 29 | 4 | 50 |
| 51 | 60 | 41 | 5 | 84 |
| 52 | 99 | 54 | 14 | 89 |
| 1 | 108 | 55 | 21 | 94 |
| 2 | 101 | 40 | 12 | 63 |
| 3 | 83 | 44 | 28 | 64 |
| 4 | 90 | 53 | 34 | 93 |
| 5 | 117 | 47 | 25 | 70 |
| 6 | 112 | 49 | 15 | 79 |
| 7 | 70 | 59 | 17 | 120 |
| 8 | 84 | 63 | 25 | 139 |
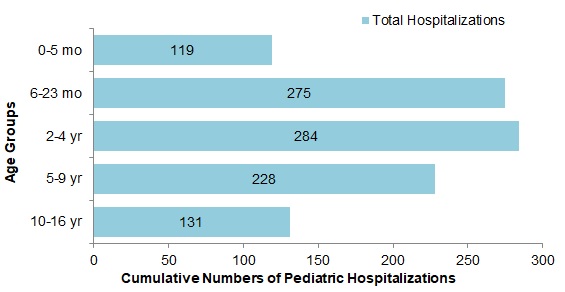
Figure 9 - Text Description
| Age Group | Total |
|---|---|
| 0-5 mo | 119 |
| 6-23 mo | 275 |
| 2-4 yr | 284 |
| 5-9 yr | 228 |
| 10-16 yr | 131 |
Adult Influenza Hospitalizations and Deaths
Surveillance of laboratory-confirmed influenza-associated adult (≥16 years of age) hospitalizations by the Canadian Immunization Research Network (CIRN) Serious Outcomes Surveillance (SOS) network began on November 1st for the 2019-20 season.
To date this season, 627 hospitalizations, 60 intensive care unit admissions, and 28 deaths have been reported (Figure 10).
- The majority of hospitalizations have been due to influenza A (82%), and among those subtyped (162) 91% were influenza A(H1N1).
- Among the 511 cases with influenza A, the largest proportion of hospitalizations were in adults 65-79 years of age (35%) and adults 80 years of age and older (31%). Among the 114 cases with influenza B, 31% were between 16 and 34 years of age (Figure 11).
- 89% of hospitalized cases reported at least one type of comorbid condition.
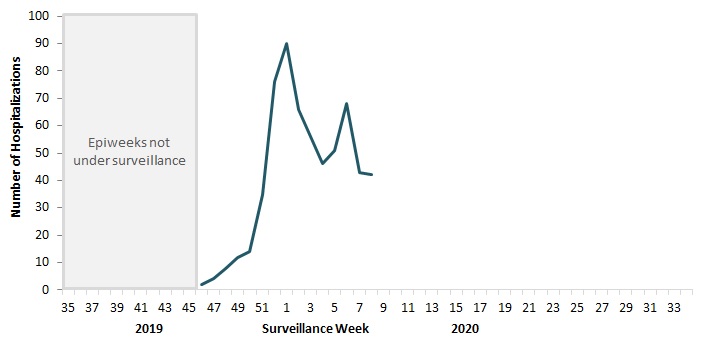
Figure 10 - Text Description
| Surveillance Week | Number of Hospitalizations |
|---|---|
| 35 | No data |
| 36 | No data |
| 37 | No data |
| 38 | No data |
| 39 | No data |
| 40 | No data |
| 41 | No data |
| 42 | No data |
| 43 | No data |
| 44 | No data |
| 45 | No data |
| 46 | 2 |
| 47 | 4 |
| 48 | 9 |
| 49 | 12 |
| 50 | 14 |
| 51 | 35 |
| 52 | 76 |
| 1 | 90 |
| 2 | 66 |
| 3 | 56 |
| 4 | 46 |
| 5 | 51 |
| 6 | 68 |
| 7 | 43 |
| 8 | 42 |
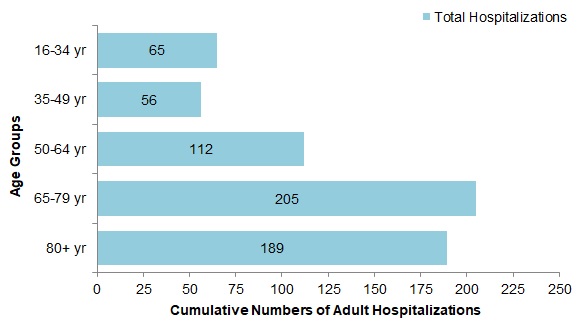
Figure 11 - Text Description
| Age Group | Total hospitalizations |
|---|---|
| 16-34 yr | 65 |
| 35-49 yr | 56 |
| 50-64 yr | 112 |
| 65-79 yr | 205 |
| 80+ yr | 189 |
Influenza Strain Characterizations
From September 1, 2019 to February 27, 2020, the National Microbiology Laboratory (NML) has characterized 1157 influenza viruses (406 A(H1N1), 159 A(H3N2) and 433 influenza B) that were received from Canadian laboratories.
Influenza A(H3N2)
Over recent years, circulating strains of A(H3N2) have evolved, and are increasingly difficult to characterize by hemagglutination inhibition (HI) assay. Genetic characterization is established by sequencing the hemagglutinin (HA) gene of the influenza viruses to compare their genetic properties.
Antigenic Characterization:
Among the 55 influenza A(H3N2) viruses antigenically characterized to date, the majority (80%) showed reduced titer by HI assay to A Kansas/14/2017 using antiserum raised against egg-propagated A Kansas/14/2017. Eleven viruses were characterized as A Kansas/14/2017-like (Figure 12a).
Genetic Characterization:
Nearly all (98%) of the 135 A(H3N2) viruses genetically characterized this season belonged to genetic group 3C.2a1b based on sequence analysis of the HA gene. Three viruses belonged to the genetic group 3C.3a (Figure 13).
Group 3C.2a1b viruses analysed represent:
- 90% (28 out of 31) viruses that were also antigenically characterized.
- 100% (104 out of 104) viruses which did not grow to sufficient hemagglutination titer for antigenic characterization by HI assay.
A/Kansas/14/2017 belongs to genetic group 3C.3a and is the influenza A(H3N2) component of the 2019-20 Northern Hemisphere influenza vaccine.
Influenza A(H1N1)
Among the 406 A(H1N1) viruses characterized to date, 52% were antigenically similar to A/Brisbane/02/2018 by HI testing using antiserum raised against egg-propagated A/Brisbane/02/2018 (Figure 12 b).
A/Brisbane/02/2018 is the influenza A(H1N1)component of the 2019-20 Northern Hemisphere influenza vaccine.
Influenza B
Antigenic Characterization:
Among the 182 influenza B viruses antigenically characterized this season, the vast majority (180) belonged to the B/Victoria lineage. Two viruses were antigenically characterized as similar to B/Phuket/3073/2013 (B/Yamagata lineage).
The majority (89%, 161) of B/Victoria lineage viruses showed reduced titer by HI assay to B/Colorado/06/2017 using antiserum raised against cell culture-propagated B/Colorado/06/2017 (Figure 12c).
Sequence analysis of 150 B/Victoria lineage viruses with reduced titre to B/Colorado/06/2017 showed that 100% had a three amino acid deletion (162-164) in the HA gene and belong to the genetic subclade V1A-3Del. Sequencing is pending for the remaining viruses.
Genetic Characterization:
Genetic characterization was also performed on 251 B/Victoria lineage viruses. All of these viruses had a three amino acid deletion (162-164) in the HA gene and belong to the genetic subclade V1A-3Del. Viruses belonging to genetic subclade V1A-3Del are antigenically distinct from the vaccine strain B/Colorado/06/2017, which belongs to genetic subclade V1A.1.
The recommended influenza B components for the 2019-20 Northern Hemisphere influenza vaccine are B/Colorado/06/2017 (Victoria lineage) and B/Phuket/3073/2013 (Yamagata lineage). B/Phuket/3073/2013 is included in the quadrivalent influenza vaccine. The vaccine strain B/Colorado/06/2017 belongs to genetic subclade V1A.1.
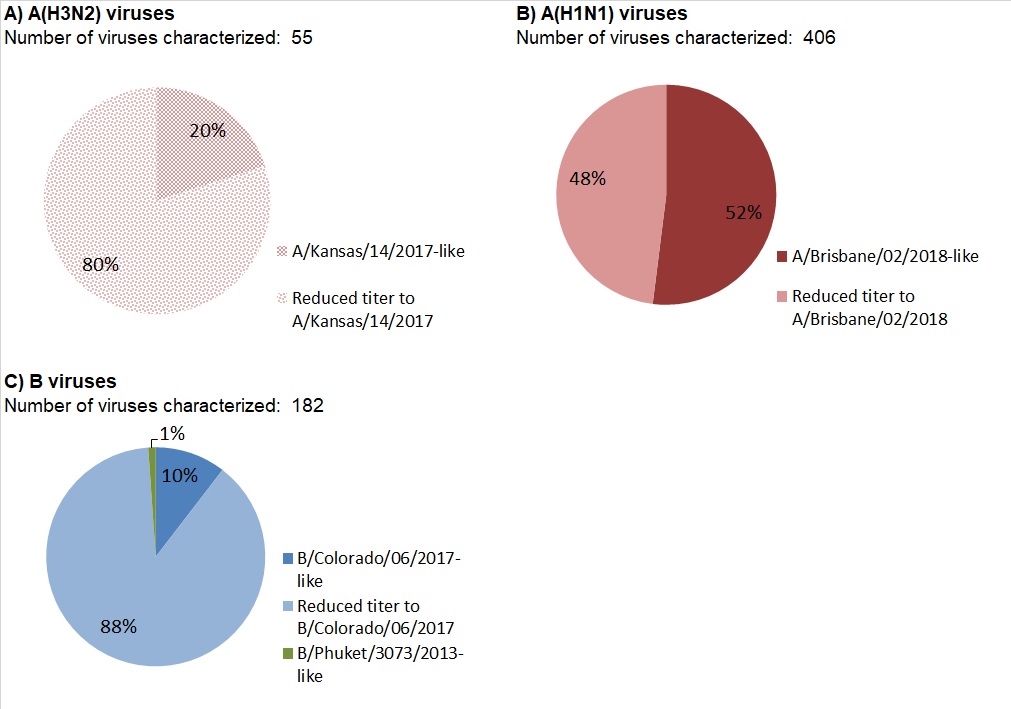
Figure 12 - Text Description
| Number of viruses characterized: 55 | ||
| Antigenic phenotype of A(H3N2) virus | Number of viruses | Percentage |
|---|---|---|
| A/Kansas/14/2017-like | 11 | 20% |
| Reduced titer to A/Kansas/14/2017 | 44 | 80% |
| Number of viruses characterized: 406 | ||
| Antigenic phenotype of A(H1N1) virus | Number of viruses | Percentage |
|---|---|---|
| A/Brisbane/02/2018-like | 211 | 52% |
| Reduced titer to A/Brisbane/02/2018 | 195 | 48% |
| Number of viruses characterized: 182 | ||
| Antigenic phenotype of influenza B virus | Number of viruses | Percentage |
|---|---|---|
| B/Colorado/06/2017-like | 19 | 10% |
| Reduced titer to B/Colorado/06/2017 | 161 | 88% |
| B/Phuket/3073/2013-like | 2 | 1% |
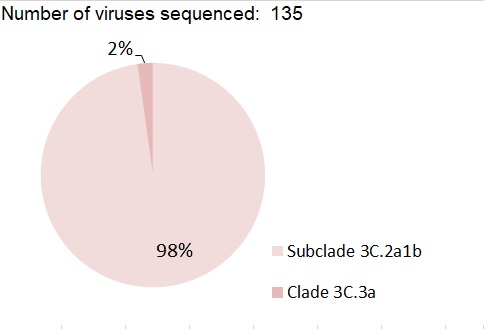
Figure 13 - Text Description
| Number of viruses sequenced: 135 | ||
| Genetic Clade of A(H3N2) virus | Number of viruses | Percentage |
|---|---|---|
| Subclade 3C.2a1b | 132 | 98% |
| Clade 3C.3a | 3 | 2% |
Antiviral Resistance
The National Microbiology Laboratory (NML) also tests influenza viruses received from Canadian laboratories for antiviral resistance. From September 1, 2019 to February 27, 2020, the following results were reported:
Oseltamivir:
554 influenza viruses (148 A(H3N2), 192 A(H1N1) and 214 B) were tested for resistance to oseltamivir:
- All influenza A(H3N2) and B viruses were sensitive to oseltamivir.
- Among the A(H1N1) viruses tested, 191 (99%) were sensitive to oseltamivir and one virus was resistant to oseltamivir with the H275Y mutation in the neuraminidase gene.
Zanamivir:
553 influenza viruses (147 A(H3N2), 192 A(H1N1) and 214 B) were tested for resistance to zanamivir:
- All influenza viruses tested were sensitive to zanamivir.
Amantadine:
High levels of resistance to amantadine persist among influenza A(H1N1) and influenza A(H3N2) viruses. All viruses tested this season were resistant.
Vaccine Monitoring
Vaccine monitoring refers to activities related to the monitoring of influenza vaccine coverage and effectiveness.
Vaccine Effectiveness
The Canadian Sentinel Practitioner Surveillance Network (SPSN) provides estimates of the effectiveness of the seasonal influenza vaccine in preventing medically-attended illness due to laboratory-confirmed influenza among Canadians.
Based on data collected between November 1, 2019 and February 1, 2020, vaccine effectiveness (VE) was estimated to be 58% for any influenza, 44% for influenza A(H1N1), 62% for influenza A(H3N2), and 69% for influenza B. Substantial protection was observed among children 1 to 19 years of age against both influenza A and B. A good level of protection was also observed among working age adults (20-64 yrs) across all influenza types (Table 2). VE among adults 65 years and older, although imprecise due to small numbers, was lower at 18% (95% CI -59 to 58). The SPSN interim estimates are published and available online.
More information on the network and past VE findings can be viewed on the SPSN website.
Updated influenza vaccine effectiveness estimates will be published at the end of the 2019/2020 influenza season. At that time, sufficient data will likely be available to estimate VE by age-group, including adults 65 years and older with greater precision, as well as for influenza A subtypes.
| All ages | 1-19 years | 20-64 years | ||
|---|---|---|---|---|
| VE(%) (CI)Table 2 - Footnote * | N | VE(%) (CI)Table 2 - Footnote * | VE(%) (CI)Table 2 - Footnote * | |
| All Influenza | 58 (47, 66) | 2808 | 74 (59, 84) | 55 (41, 66) |
| Influenza A | 49 (34, 60) | 2128 | 70 (44, 84) | 45 (25, 59) |
| Influenza A(H1N1) | 44 (26, 58) | 1948 | ||
| Influenza A(H3N2) | 62 (37, 77) | 1561 | ||
| Influenza B | 69 (57, 77) | 2080 | 77 (59, 87) | 68 (51, 79) |
CI: 95% confidence interval
|
||||
Vaccine Coverage
Influenza vaccine coverage estimates for the 2019-20 season are anticipated to be available in March 2020.
Notes
The data in the FluWatch report represent surveillance data available at the time of writing. All data are preliminary and may change as more reports are received.
To learn more about the FluWatch program, see the Overview of influenza monitoring in Canada page.
For more information on the flu, see our Flu (influenza) web page.
We would like to thank all the Fluwatch surveillance partners participating in this year's influenza surveillance program.
This report is available on the Government of Canada Influenza webpage.
Ce rapport est disponible dans les deux langues officielles.