FluWatch report: October 13 to October 19, 2019 (Week 42)

Download the alternative format
(PDF format, 384 KB, 8 pages)
Organization: Public Health Agency of Canada
Date published: 2019-10-25
Related Topics
Overall Summary
- Influenza activity remains at interseasonal levels at the national level.
- The number of regions in Canada reporting influenza activity in week 42 was similar to the previous week.
- Influenza A(H3N2) is the most common influenza virus circulating in Canada.
- Weekly reporting of laboratory detections of respiratory viruses continues via our Respiratory Virus Detections Surveillance System.
On this page
- Influenza/ILI Activity (geographic spread)
- Laboratory Confirmed Influenza Detections
- Syndromic/Influenza-like Illness Surveillance
- FluWatchers
- Influenza Outbreak Surveillance
- Severe Outcomes Influenza Surveillance
- Influenza Strain Characterizations
- Antiviral Resistance
- Vaccine Monitoring
- Provincial and International Influenza Reports
Influenza/Influenza-like Illness Activity (geographic spread)
During week 42, the number of regions reporting influenza activity was similar to the previous week. The following influenza activity levels were reported (Figure 1):
- 20 regions (38%) reported sporadic activity, within 7 provinces and territories.
Figure 1 – Map of influenza/ILI activity by province and territory, Canada, week 2019-42
Number of Regions Reporting in Week 42: 53 out of 53

Figure 1 - Text equivalent
| Province | Influenza Surveillance Region | Activity Level |
|---|---|---|
| N.L. | Eastern | No Activity |
| N.L. | Labrador-Grenfell | No Activity |
| N.L. | Central | No Activity |
| N.L. | Western | No Activity |
| P.E.I. | Prince Edward Island | No Activity |
| N.S. | Zone 1 - Western | No Activity |
| N.S. | Zone 2 - Northern | No Activity |
| N.S. | Zone 3 - Eastern | No Activity |
| N.S. | Zone 4 - Central | Sporadic |
| N.B. | Region 1 | No Activity |
| N.B. | Region 2 | No Activity |
| N.B. | Region 3 | No Activity |
| N.B. | Region 4 | No Activity |
| N.B. | Region 5 | No Activity |
| N.B. | Region 6 | No Activity |
| N.B. | Region 7 | No Activity |
| Que. | Nord-est | Sporadic |
| Que. | Québec et Chaudieres-Appalaches | No Activity |
| Que. | Centre-du-Québec | Sporadic |
| Que. | Montréal et Laval | Sporadic |
| Que. | Ouest-du-Québec | Sporadic |
| Que. | Montérégie | Sporadic |
| Ont. | Central East | Sporadic |
| Ont. | Central West | No Activity |
| Ont. | Eastern | No Activity |
| Ont. | North East | No Activity |
| Ont. | North West | No Activity |
| Ont. | South West | Sporadic |
| Ont. | Toronto | No Activity |
| Man. | Northern Regional | Sporadic |
| Man. | Prairie Mountain | No Activity |
| Man. | Interlake-Eastern | No Activity |
| Man. | Winnipeg | No Activity |
| Man. | Southern Health | Sporadic |
| Sask. | North | No Activity |
| Sask. | Central | Sporadic |
| Sask. | South | Sporadic |
| Alta. | North Zone | Sporadic |
| Alta. | Edmonton | Sporadic |
| Alta. | Central Zone | No Activity |
| Alta. | Calgary | Sporadic |
| Alta. | South Zone | No Activity |
| B.C. | Interior | Sporadic |
| B.C. | Fraser | Sporadic |
| B.C. | Vancouver Coastal | Sporadic |
| B.C. | Vancouver Island | Sporadic |
| B.C. | Northern | No Activity |
| Y.T. | Yukon | Sporadic |
| N.W.T. | North | No Activity |
| N.W.T. | South | No Activity |
| Nvt. | Qikiqtaaluk | No Activity |
| Nvt. | Kivalliq | No Activity |
| Nvt. | Kitimeot | No Activity |
Laboratory-Confirmed Influenza Detections
In week 42, detections of influenza continued to increase. The following results were reported from sentinel laboratories across Canada (Figures 2 and 3):
- A total of 86 laboratory detections of influenza were reported, of which 84% (72) were influenza A.
- The percentage of tests positive for influenza increased, but remains at interseasonal levels, at 1.9%.
- Among subtyped influenza A detections 11 out of 12 were influenza A(H3N2).
To date this season (weeks 35 to 42), 358 laboratory detections of influenza were reported:
- 89% (319) were influenza A.
- Among subtyped influenza A detections (131), 92% were influenza A(H3N2).
Detailed information on age and type/subtype has been received for 263 laboratory-confirmed influenza cases. To date this season (weeks 35 to 42):
- 88% (231) were cases of influenza A and 12% (32) were influenza B.
- Among the 107 cases of influenza A for which the subtype was known, 92% of cases were A(H3N2).
- The majority of influenza A cases reported to date were in adults 65 years of age and older (55%). The majority of cases of influenza B were in younger age-groups; 78% of cases (25 of 32) were under 45 years of age.
For more detailed weekly and cumulative influenza data, see the text descriptions for Figures 2 and 3 or the Respiratory Virus Detections in Canada Report.
Figure 2 - Number of positive influenza tests and percentage of tests positive, by type, subtype and report week, Canada, weeks 2019-35 to 2019-42
Number of Laboratories Reporting in Week 42: 33 out of 34
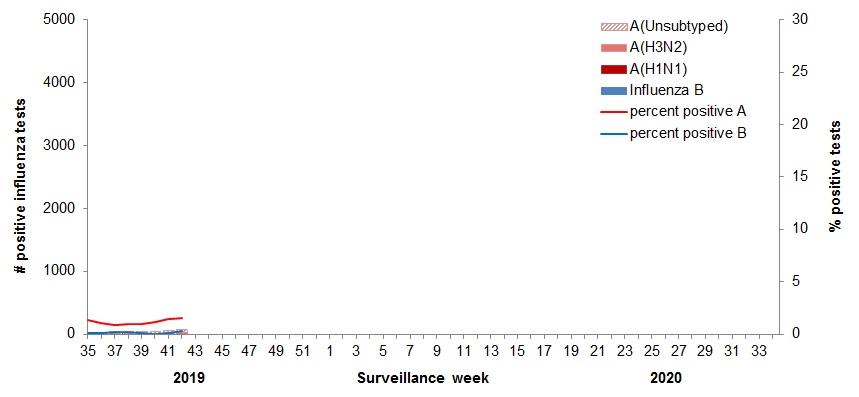
Figure 2 - Text equivalent
| Surveillance Week | A(Unsubtyped) | A(H3N2) | A(H1N1)pdm09 | Influenza B | Percent Positive A | Percent Positive B |
|---|---|---|---|---|---|---|
| 35 | 10 | 16 | 0 | 2 | 1.3 | 0.1 |
| 36 | 11 | 13 | 2 | 2 | 1.1 | 0.1 |
| 37 | 5 | 17 | 2 | 5 | 0.9 | 0.2 |
| 38 | 11 | 15 | 3 | 6 | 1.0 | 0.2 |
| 39 | 11 | 21 | 2 | 3 | 1.0 | 0.1 |
| 40 | 34 | 9 | 1 | 2 | 1.2 | 0.1 |
| 41 | 34 | 18 | 0 | 5 | 1.4 | 0.1 |
| 42 | 54 | 11 | 1 | 14 | 1.6 | 0.3 |
Figure 3 – Distribution of positive influenza specimens by type/subtype and province/territory*, Canada, weeks 2019-35 to 2019-42
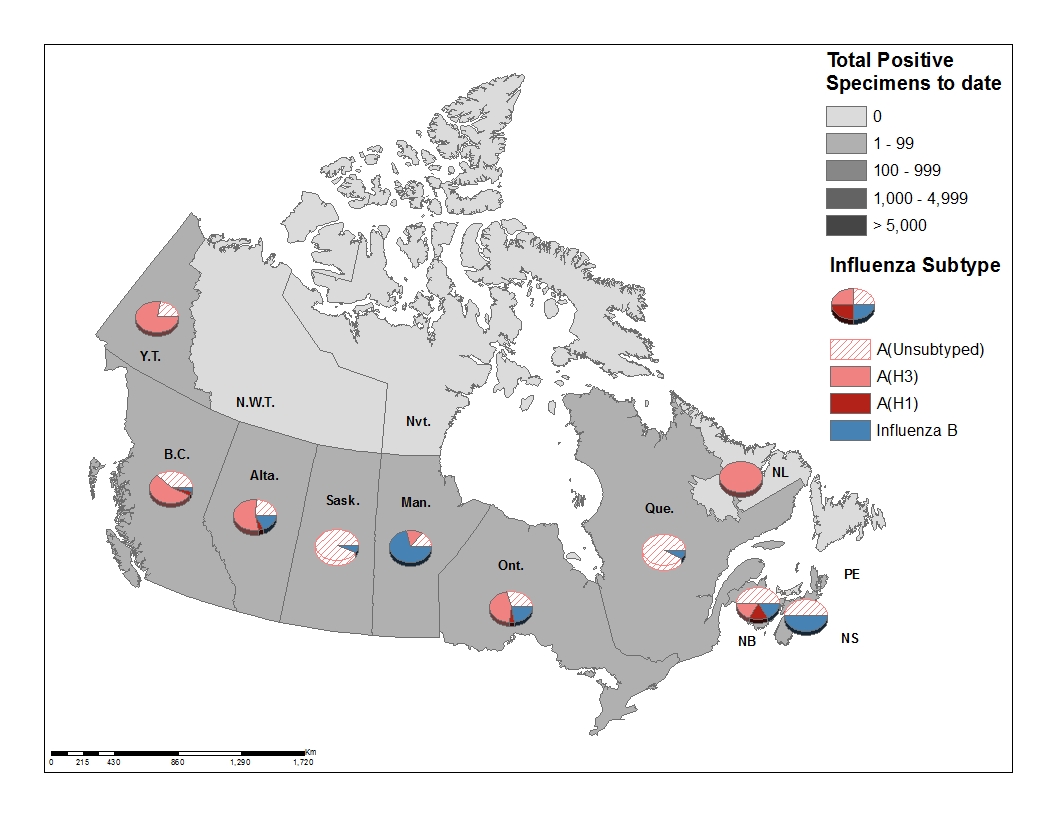
Figure 3 - Text equivalent
| ProvincesTable Figure 3 - Footnote 1 | Cumulative (August 25. 2019 to October 19, 2019) | |||||
|---|---|---|---|---|---|---|
| A Total | A(H1N1) | A(H3N2) | A(UnS)Table Figure 3 - Footnote 3 | B Total | Total | |
| B.C. | 110 | 4 | 52 | 36 | 3 | 113 |
| Alta. | 64 | 4 | 42 | 18 | 13 | 77 |
| Sask. | 16 | 0 | 0 | 14 | 1 | 17 |
| Man. | 2 | 0 | 1 | 1 | 5 | 7 |
| Ont. | 29 | 2 | 16 | 11 | 8 | 37 |
| Que. | 82 | 0 | 0 | 82 | 7 | 89 |
| N.B. | 5 | 1 | 1 | 3 | 1 | 6 |
| N.S. | 1 | 0 | 0 | 1 | 1 | 2 |
| P.E.I. | 0 | 0 | 0 | 0 | 0 | 0 |
| N.L. | 1 | 0 | 1 | 0 | 0 | 1 |
| Y.T. | 9 | 0 | 7 | 2 | 0 | 9 |
| N.W.T | 0 | 0 | 0 | 0 | 0 | 0 |
| Nvt. | 0 | 0 | 0 | 0 | 0 | 0 |
| Canada | 319 | 11 | 120 | 168 | 39 | 358 |
| PercentageTable Figure 3 - Footnote 2 | 89% | 3% | 38% | 53% | 11% | 100% |
|
||||||
Syndromic / Influenza-like Illness Surveillance
Healthcare Professionals Sentinel Syndromic Surveillance
In week 42, 1.4%, of visits to healthcare professionals were due to influenza-like illness (ILI) which is similar to the average for this time of year (Figure 4).
Figure 4 – Percentage of visits for ILI reported by sentinels by report week, Canada, weeks 2019-35 to 2019-42
Number of Sentinels Reporting in Week 42: 82
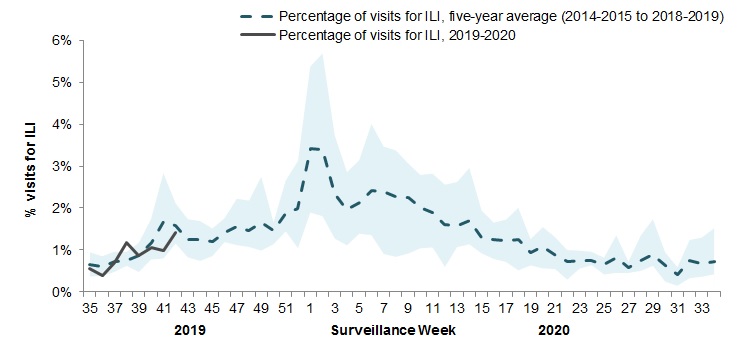
The shaded area represents the maximum and minimum percentage of visits for ILI reported by week from seasons 2014-2015 to 2018-2019
Figure 4 - Text equivalent
| Surveillance Week | 2019-20 | Average | Min | Max |
|---|---|---|---|---|
| 35 | 0.6% | 0.6% | 0.4% | 0.9% |
| 36 | 0.4% | 0.6% | 0.4% | 0.9% |
| 37 | 0.7% | 0.7% | 0.5% | 1.0% |
| 38 | 1.2% | 0.7% | 0.6% | 1.0% |
| 39 | 0.9% | 0.9% | 0.5% | 1.2% |
| 40 | 1.0% | 1.2% | 0.8% | 1.7% |
| 41 | 1.0% | 1.7% | 0.8% | 2.8% |
| 42 | 1.4% | 1.6% | 1.2% | 2.1% |
FluWatchers
In week 42, 2, 957 participants reported to FluWatchers, of which 1.7% (51) reported symptoms of cough and fever (Figure 5).
Among the 51 participants who reported cough and fever:
- 18% consulted a healthcare professional;
- 75% reported days missed from work or school, resulting in a combined total of 127 missed days of work or school.
If you are interested in becoming a FluWatcher. sign up today.
Figure 5 – Percentage of FluWatchers participants reporting cough and fever, Canada, weeks 2019-40 to 2019-42
Number of Participants Reporting in Week 42: 2,957
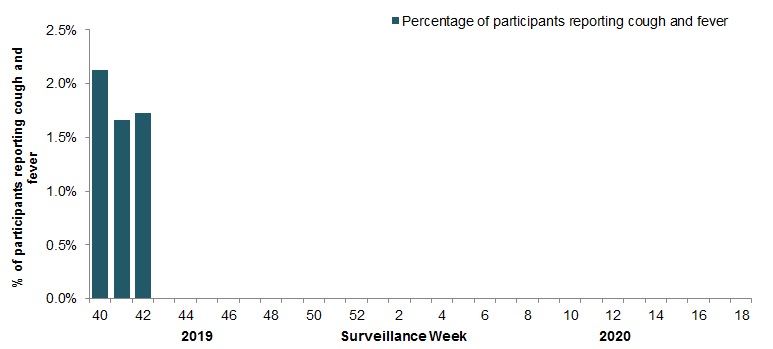
Figure 5 - Text equivalent
| Surveillance Week | % cough and fever |
|---|---|
| 40 | 2.1% |
| 41 | 1.7% |
| 42 | 1.7% |
Online Figure – Geographic distribution of FluWatchers participants reporting cough and fever, Canada, week 2019-42
Click on the map to access the link
Influenza Outbreak Surveillance
In week 42, no new outbreaks were reported.
To date this season, a total of 7 laboratory-confirmed influenza outbreaks have been reported; six in long-term care facilities and one in another type of facility. All reported outbreaks were due to influenza A. Two ILI outbreaks in schools and daycares have also been reported.
Number of provinces and territories reporting in week 42: 13 out of 13
Severe Outcomes Influenza Surveillance
Provincial/Territorial Influenza Hospitalizations and Deaths
In week 42, six influenza-associated hospitalizations were reported by participating provinces and territoriesFootnote 1.
To date this season. 28 influenza-associated hospitalizations were reported by participating provinces and territoriesFootnote 1.
- 82% of the cases were influenza A.
- Of the cases for which subtype was reported (22), 82% were associated with influenza A(H3N2).
- The majority of cases (54%) were ≥ 65 years of age.
Less than five ICU admissions and no deaths have been reported.
Number of provinces and territories reporting in week 42: 9 out of 9- Footnote 1
-
Influenza-associated hospitalizations are reported by N.L., P.E.I. N.S., N.B., Man., Alb., Y.T. and N.W.T. Only hospitalizations that require intensive medical care are reported by Sask. The cumulative rate of hospitalizations is calculated using the population by age-group in participating provinces and territories.
Pediatric Influenza Hospitalizations and Deaths
In week 42, less than five pediatric (≤16 years of age) laboratory-confirmed influenza-associated hospitalizations were reported by the Immunization Monitoring Program Active (IMPACT) network.
To date this season, seven pediatric hospitalizations have been reported by the IMPACT network; four cases associated with influenza A and three with influenza B.
Adult Influenza Hospitalizations and Deaths
Surveillance of laboratory-confirmed influenza-associated adult (≥16 years of age) hospitalizations by the Canadian Immunization Research Network (CIRN) Serious Outcomes Surveillance (SOS) network has not yet begun for the 2019-20 season.
Influenza Strain Characterizations
From September 1 to October 24, 2019, the National Microbiology Laboratory (NML) has characterized 14 influenza viruses (9 A(H3N2), 1 A(H1N1) and 4 influenza B) that were received from Canadian laboratories.
Influenza A(H3N2)
Over recent years, circulating strains of A(H3N2) have evolved, and are increasingly difficult to characterize by hemagglutination inhibition (HI) assay. Genetic characterization is established by sequencing the hemagglutinin (HA) gene of the influenza viruses to compare their genetic properties.
Antigenic Characterization:
- Four influenza A(H3N2) viruses were antigenically characterized as A/Kansas/14/2017-like by HI assay using antiserum raised against cell culture-propagated A Kansas/14/2017.
Genetic Characterization:
All nine A(H3N2) viruses this season have been genetically characterized, based on sequence analysis of the HA gene.
- Among the four A(H3N2) viruses antigenically characterized as A/Kansas/14/2017-like by HI testing.
- All viruses belonged to genetic group 3C.2a1b.
- Among the five A(H3N2) viruses which did not grow to sufficient hemagglutination titer for antigenic characterization by HI assay:
- All viruses belonged to genetic subclade 3C.2a1b.
A/Kansas/14/2017 belongs to genetic group 3C.3a and is the influenza A(H3N2) component of the 2019-20 Northern Hemisphere influenza vaccine.
Influenza A(H1N1)
- One A(H1N1) virus characterized was antigenically similar to A/Brisbane/02/2018 by HI testing using antiserum raised against egg-propagated A/Brisbane/02/2018.
A/Brisbane/02/2018 is the influenza A(H1N1) component of the 2019-20 Northern Hemisphere influenza vaccine.
Influenza B
- One influenza B virus characterized was antigenically similar to B/Colorado/06/2017 by HI assay using antiserum raised against cell culture-propagated B/Colorado/06/2017.
- Three influenza B viruses showed reduced titer by HI assay. Sequence analysis showed that :
- Two of these viruses had a three amino acid deletion (162-164) in the HA gene.
- Sequence data is pending for the other virus.
The recommended influenza B components for the 2019-20 Northern Hemisphere influenza vaccine are B/Colorado/06/2017 (Victoria lineage) and B/Phuket/3073/2013 (Yamagata lineage). B/Phuket/3073/2013 is included in the quadrivalent influenza vaccine.
Antiviral Resistance
The National Microbiology Laboratory (NML) also tests influenza viruses received from Canadian laboratories for antiviral resistance.
Oseltamivir:
Fourteen influenza viruses (9 H3N2, 2 H1N1 and 3 B) were tested for resistance to oseltamivir :
- All influenza viruses tested were sensitive to oseltamivir.
Zanamivir:
Fourteen influenza viruses (9 H3N2, 2 H1N1 and 3 B) were tested for resistance to zanamivir :
- All influenza viruses tested were sensitive to zanamivir.
Amantadine:
High levels of resistance to amantadine persist among influenza A(H1N1) and influenza A(H3N2) viruses. All viruses tested this season were resistant.
Vaccine Monitoring
Vaccine monitoring refers to activities related to the monitoring of influenza vaccine coverage and effectiveness.
Vaccine Coverage
Influenza vaccine coverage estimates for the 2019-20 season are anticipated to be available in February or March 2020.
Vaccine Effectiveness
Influenza vaccine effectiveness estimates for the 2019-20 season are anticipated to be available in February or March 2020.
Provincial and International Surveillance Links
- British Columbia – Influenza Surveillance; Vaccine Effectiveness Monitoring
- Alberta – Influenza surveillance
- Saskatchewan – Influenza Reports
- Manitoba – Seasonal Influenza Reports
- Ontario – Ontario Respiratory Pathogen Bulletin
- Québec – Système de surveillance de la grippe (available in French only)
- New Brunswick – Influenza Surveillance Reports
- Prince Edward Island – Influenza Summary
- Nova Scotia – Respiratory Watch Report
- Newfoundland and Labrador – Surveillance and Disease Reports
- Yukon – Information on Pandemic, Influenza, Seasonal Flu, Avian Flu and H1N1
- Northwest Territories – Influenza/ Flu Information
- Nunavut – Influenza Information
- World Health Organization – FluNet (Global Influenza Surveillance Network)
- Pan American Health Organization – Influenza situation report
- U.S. Centers for Disease Prevention & Control (CDC) - Weekly Influenza Summary Update
- ECDC – Surveillance reports and disease data on seasonal influenza
- United Kingdom – Weekly Influenza Activity Reports
- Hong Kong Centre for Health Protection - Flu Express
- Australia – Influenza Surveillance Report and Activity Updates
- New Zealand – Influenza Weekly Update
Notes
The data in the FluWatch report represent surveillance data available at the time of writing. All data are preliminary and may change as more reports are received.
To learn more about the FluWatch program, see the Overview of influenza monitoring in Canada page.
For more information on the flu, see our Flu (influenza) web page.
We would like to thank all the Fluwatch surveillance partners participating in this year's influenza surveillance program.
This report is available on the Government of Canada Influenza webpage.
Ce rapport est disponible dans les deux langues officielles.