Centre for Biosecurity Annual Report 2018-2019 - Maximizing Impact
Download in PDF format
(2.8 MB, 25 pages)
Organization: Public Health Agency of Canada
Date published: August 2020
Applying Lessons Learned to Strengthen Canada’s Biosafety and Biosecurity
Public Health Agency of Canada
Biosecurity Program
Annual Report 2018–19
Table of Contents
- 2018–19 Results: Regulatory Oversight
- Maximizing impact in Compliance Monitoring and Verification
- Maximizing impact in Program Administration and Service Delivery
- Maximizing impact in Compliance Promotion
- Maximizing impact in Intragovernmental Collaboration
- Maximizing Impact in International Engagement and Capacity-Building
At the Public Health Agency of Canada’s Biosecurity Program, our work has one ultimate measure of impact: the safety of Canadians.
In 2018–19, we took action on multiple fronts to maximize that impact, including applying stakeholder feedback and lessons learned to enhance, streamline and optimize our effectiveness at protecting Canadians from pathogen- and toxin-related risks.
This report tells the story of those efforts, reflecting our commitment to the highest standards of biosafety and biosecurity — and to maintaining the culture of continuous improvement expected from a modern, efficient and agile regulatory organization.
The Public Health Agency of Canada’s Biosecurity Program protects Canadians from the health and safety risks associated with the use of human and indigenous terrestrial animal pathogens and select biological toxins.
Our Mission
To deliver a strong and comprehensive safety and security regime that prevents, detects and responds to the risks associated with the use of pathogens and toxins, and fosters an enhanced national biosafety culture.
How We Work
Organizations across a wide range of sectors use pathogens (microorganisms, nucleic acids or proteins capable of causing disease in humans or animals) and toxins (poisonous substances produced by or derived from microorganisms) for many different purposes: teaching and research at academic institutions, disease diagnosis at hospitals and public health facilities, vaccine development in the pharmaceutical industry, quality control in the food industry, and more.
Our program upholds Canada’s standards for the safe, secure use of pathogens and toxins by administering and enforcing the Human Pathogens and Toxins Act (HPTA) and Human Pathogens and Toxins Regulations (HPTR), as well as certain provisions of the Health of Animals Act (HAA) and Health of Animals Regulations (HAR) related to importing indigenous terrestrial animal pathogens.
Our mandate has two sides: to strengthen biosafety (preventing unintentional exposures to or releases of pathogens and toxins) and promote biosecurity (preventing the loss, theft or misuse of pathogens and toxins).
To achieve that mandate, we set clear expectations and provide guidance to help regulated parties comply with regulatory requirements. We also monitor and verify compliance to determine whether the activities being conducted with pathogens and toxins in labs across Canada are performed in a safe manner and do not pose a risk to national security.
Our comprehensive, national approach to regulating pathogens and toxins includes:
- Issuing Pathogen and Toxin Licences
- Issuing HPTA Security Clearances for individuals accessing high-risk pathogens
- Monitoring, verifying and enforcing compliance through inspections, document reviews and border surveillance activities
- Monitoring laboratory incident and exposure notifications
- Conducting pathogen risk assessments
- Developing standards, guidance and resources
- Conducting stakeholder outreach and engagement
We support the advancement of global health priorities in biosafety and biosecurity, working collaboratively with international partners to protect Canadians from pathogen- and toxin-related risks emerging outside Canada. In addition, we offer guidance and tools to state parties around the world to help build their biosafety and biosecurity capacity so they can fulfil their obligations including the Biological and Toxins Weapons Convention (BTWC) and United Nations Security Council Resolution (UNSCR) 1540.
Regulatory Authorizations
Since the HPTA and HPTR came fully into effect in December 2015, we have issued hundreds of Pathogen and Toxin Licences and HPTA Security Clearances to regulated parties:
986
Licence distribution by Risk group:
- RG2: 916
- RG3: 65
- SSBA Toxin: 3
- RG4: 2
439
Active HPTA Security
Clearances
(as of March 31, 2019)
| Sector | Number of Licences |
|---|---|
| Industry | 464 |
| Hospital | 192 |
| Academic | 201 |
| Other Government | 38 |
| Public Health | 34 |
| Vet/Animal Health | 27 |
| Environmental Health | 30 |
| Citizen Science | 0 |
| Total | 986 |
Which activities need a licence?
Conducting a controlled activity with a Risk Group 2 or higher human pathogen or a Schedule 1 toxin in Canada requires a Pathogen and Toxin Licence, with some exemptions and exclusions as defined in the Human Pathogens and Toxins Act and associated Regulations.
Controlled Activities:
- Possessing, handling or using,
- Producing,
- Storing,
- Permitting access to,
- Transferring,
- Importing or exporting,
- Releasing or abandoning,
- Disposing of human pathogens or toxins
| Risk Group 1 | Risk Group 2 | Risk Group 3 | Risk Group 4 | |
|---|---|---|---|---|
| Individual Risk | Low | Moderate | High | High |
| Community Risk | Low | Low | Low | High |
| Examples | Lactobacillus spp. | Salmonella bacteria, Hepatitis A virus. | Mycobacterium tuberculosis, severe acute respiratory syndrome (SARS) virus | Ebola virus, Marburg virus |
| The HPTA applies to all persons/facilities in Canada conducting controlled activities with RG2, RG3 or RG4 human pathogens and select toxins. | ||||
2018–19 Results: Regulatory Oversight
We take a risk-based approach to licensing and compliance monitoring, verification and enforcement. That means as the risks inherent in activities involving pathogens and toxins increase, the requirements placed on our regulated parties — and our processes for addressing compliance-related issues — become more stringent.
We use the best available evidence and science to determine the biosafety and biosecurity requirements for the controlled activities conducted by our regulated parties. That allows us to prioritize inspections and other compliance monitoring and verification activities in a way that is commensurate with risk, and take proportionate enforcement actions when needed.
Pathogen and Toxin Licences
We issue Pathogen and Toxin Licences to organizations that work with pathogens or select biological toxins in Canada. A licence is the document that authorizes the conduct of controlled activities under the Human Pathogens and Toxins Act (HPTA). It also serves as a permit that authorizes the importation and subsequent transfer of indigenous terrestrial animal pathogens under the Health of Animals Act (HAA). Each licence has specific conditions based on the risk group (RG) classification of the pathogens or status of the toxins involved as well as the nature of the work being done.
346 licence applications processed (including new applications, renewals, amendments and cancellations) in 2018–19
Pathogen and Toxin Licence applications processed per quarter
- Q1 (April – June 2018): 91
- Q2 (July – Sept. 2018): 76
- Q3 (Oct. – Dec. 2018): 85
- Q4 (Jan. – Mar. 2019): 94
| Target | Result | |
|---|---|---|
| Issuance of Pathogen and Toxin Licences within 80 business days from receipt of a completed application | 80% | 99% |
HPTA Security Clearances
Every person who works with or has access to security sensitive biological agents (SSBAs) requires an HPTA Security Clearance. SSBAs are a subset of RG3 and RG4 pathogensand regulated toxins above a “trigger quantity” that pose an increased biosecurity risk due to their potential for use as biological weapons.
52 HPTA Security Clearances issued in 2018–19
HPTA Security Clearances issued per quarter
- Q1 (April – June 2018): 14
- Q2 (July – Sept. 2018): 10
- Q3 (Oct. – Dec. 2018): 9
- Q4 (Jan. – Mar. 2019): 19
| Target | Result | |
|---|---|---|
| Acknowledgment of receipt of a completed HPTA Security Clearance application within five business days | 80% | 59% |
| Issuance of HPTA Security Clearances within 80 business days from receipt of a completed application | 80% | 53% |
Although we did not meet our service standards over the entire fiscal year, we did achieve our targets toward the end of the year. The significant improvements seen in early 2019 were driven by a root-cause analysis that helped us find ways to make our quality-control processes more efficient.
Compliance Monitoring, Verification and Enforcement
Inspections
Inspections are done to verify regulated parties’ compliance with the mandatory requirements of the HPTA, Human Pathogens and Toxins Regulations (HPTR) and Health of Animals Regulations (HAR), as well as all applicable requirements of the Canadian Biosafety Standard (CBS).
78 licences inspected in 2018–19
Licences inspected
| Active Licences end of 2017-18Table 5 - Footnote * | Target | Result | ||
|---|---|---|---|---|
| RG2 | 913 | 5% | 46 | 55 |
| SSBA Toxins | 2 | 33% | 0Table 5 - Footnote ** | N/A |
| RG3 | 62 | 33% | 20 | 21 |
| RG4 | 2 | 100% | 2 | 2 |
The frequency of inspection is determined by the risks associated with pathogens or toxins, which correspond to a type of licence. Because RG3, SSBA and RG4 pathogens and toxins pose higher risk, facilities working with them must be inspected at least once during the term of their licence. That means RG3/SSBA licenses are inspected once every three years and RG4 licences are inspected annually. As the activities authorized by an RG2 licence pose lower relative risk, a percentage are inspected each year - 5% for 2018-19. Inspections are determined by an algorithm that prioritizes site selection based on several risk criteria.
Inspection findings
468 deficiencies with applicable Canadian Biosafety Standard requirements identified by inspectors in 2018–19
| RG2 | RG3 |
|---|---|
Records and documentation |
|
| Training Example: No annual refresher training on emergency response procedures |
Work practices Example: No verification of the integrity of primary containment devices |
| Surface finishes and casework Example: Inadequate replacement, removal or repair of worn-down surfaces and materials |
Performance and verification tests Example: Integrity of primary containment devices not tested in accordance with procedures |
8 non-compliances with HPTA/HPTR requirements noted by inspectors in 2018–19
Document Reviews
Plan for Administrative Oversight
All licence applicants looking to conduct scientific research with pathogens and toxins must submit a Plan for Administrative Oversight (PAO) describing how biosafety and biosecurity risks will be managed.
157 PAOs with a compliant rating since 2016
| Target | Result | |
|---|---|---|
| Percentage of PAOs with a compliant rating | 95% of all PAOs by March 2020 | 50% of all PAOs as of March 2019 |
We are getting closer to achieving our 2020 PAO target, with the number of assessed and compliant PAOs increasing from just 2% in 2016–17 to 40% in 2017–18 and now 50% in 2018–19.
Biosecurity plans
All facilities require a biosecurity plan that outlines the security measures in place to prevent the loss, theft, misuse, diversion or intentional release of pathogens and toxins.
| Licences Assessed | Reviews | |
|---|---|---|
| Plans reviewed | 19 | 24 |
Compliance document reviews
For any licensed organization, we can request documents with a focus on areas inspections have identified with more non-compliances and/or potential for risk, such as pathogen inventories or decontamination practices. The information included in those documents can help us establish a compliance history for that organization.
| Licences Assessed | Reviews | |
|---|---|---|
| Documents reviewed | 20 | 41 |
Enforcement and Overall Compliance
We approach compliance and enforcement in a progressive manner: our primary objective is to manage risk and guide the regulated party toward compliance through a graduated continuum of enforcement activities. However, some situations may be more appropriately addressed with stronger, more immediate actions. We consider several factors when selecting the most appropriate enforcement activity to address non-compliance. For more information, please see PHAC’s Regulatory Compliance and Enforcement Framework.
We monitor and report on how licensed laboratories implement required corrective actions identified through the program's compliance monitoring and verification activities.
| Target | Result | |
|---|---|---|
| Percentage of compliance issues in Canadian laboratories successfully responded to within established timelines | 85% | 88% |
If an organization fails to implement required corrective actions within established timelines or otherwise not meet its regulatory requirements or licence conditions, we may take additional enforcement action, including but not limited to Notices of Non-Compliance and Inspector Orders issued under the Act. In 2018-19, we issued 39 Notices of Non-Compliance and 2 Orders.
Pathogen Risk Assessments
Pathogen risk assessments (PRAs) evaluate the risks associated with pathogens that are uncharacterized or may have been modified. These assessments help us determine if a pathogen does or does not fall under HPTA requirements. They also help determine the appropriate risk group classification for a pathogen, which informs licensing as well as risk-mitigation strategies.
159 PRAs completed in 2018–19
Laboratory Incident Reporting
Licensed facilities are required to report three types of incidents to the Laboratory Incident Notification Canada (LINC) surveillance system:
- Exposure incidents including those that have caused disease (confirmed laboratory-acquired infection) or may have caused disease (suspected laboratory-acquired infection)
- Non-exposure incidents including inadvertent possession, production or release, or the loss, theft or inadvertent release of a pathogen or toxin, including SSBAs
- Other reports including changes affecting containment, exemption from a risk group (risk group reduction) or the prohibition of a holder of an HPTA Security Clearance from accessing a part of the facility
This allows for near-real-time detection of trends and patterns in lab incidents, which we use to identify risk factors and address emerging public health and safety risks.
235 incident notifications received in 2018–19
For more information on incident notifications, please see the 2018 LINC annual report.
Compliance Promotion
We aim to provide our regulated parties and stakeholders with a range of tools, resources, and guidance to support them in identifying and mitigating biosafety and biosecurity risks,and fulfilling their obligations under the HPTA and HPTR.
- 2
- New training courses added to e-learning
- 16,193
-
biosafety and biosecurity training course completions
- 2,907
-
Pathogen Safety Data Sheet mobile app downloads
- 696
-
Canadian Biosafety Standard mobile app downloads
- 4
-
Quarterly newsletters
- 8
-
e-Blasts
- 5
-
New webinars
- 1,445
-
Total webinar participants
- 3
-
New biosafety guidelines/directives
- 2
-
Pre-licensing inspections
- 2
-
Booths at conferences (Biosafety Symposium, Canada-Wide Science Fair)
Maximizing Impact in Compliance Monitoring and Verification
More precise criteria for a more efficient risk-based inspection approach
More than 900 organizations across Canada hold RG2 licences. To deploy inspectors effectively and focus on the facilities where compliance monitoring is needed most, we updated the risk-based approach we use to select and prioritize the RG2 sites to be inspected.
Each year’s RG2 inspection sites are determined by an algorithm that considers a variety of risk factors, including an organization’s compliance history and the robustness of its biosafety and biosecurity program. In keeping with our culture of continuous improvement, in early 2019 we undertook a planned evaluation of our site-selection process, including the criteria used to rank an organization’s level of risk.
To create more precise criteria on which to evaluate risk, we cross-referenced information from inspections conducted between December 2015 and January 2019 against other compliance-related data. Comparing the actual deficiencies we observed during those inspections against the original risk ranking assigned to each organization, we modified the weighting of our site-selection criteria. We also added a new criterion related to the role of a facility’s biological safety officer. With these new criteria, we can better determine when and why a particular facility should be inspected.
Approved in March 2019, the new site-selection criteria for RG2 inspections were implemented officially for the start of the 2019–20 inspection schedule. Going forward, we will be able to more precisely prioritize facilities whose profile could present a higher risk to both individual and public health, while making the best possible use of our inspection resources.
Innovation in assessing compliance
Another way we brought efficiency to our risk-based approach in 2018–19 was by piloting a document review process for RG2 licences. We specifically targeted a subset of the RG2-licensed organizations with no existing compliance history, reviewing their documentation related to pathogen and toxin inventories as well as containment, decontamination and other work practices. This pilot allowed us to gain a baseline compliance history for these organizations without having to expend resources for on-site inspections. It also helped us establish a baseline level of awareness of our program among these regulated parties and further promote biosafety and biosecurity best practices.
Using national data to identify lab safety risks
When five exposure incidents involving Matrix-Assisted Laser Desorption/Ionization-Time Of Flight (MALDI-TOF) mass spectrometry (MS) devices were reported to us over a nine-month period through the Laboratory Incident Notification Canada (LINC) system, we launched a national investigation to determine the cause and how to prevent similar incidents from happening in the future. Our study revealed the MALDI-TOF MS had the potential to misidentify dangerous pathogens, which could lead to exposure risks for lab personnel. The results of our investigation were published in the May 2018 issue of the Canada Communicable Disease Report, providing the labs using these devices with the baseline data needed to inform their mitigation strategies. Using the lessons learned from the study, we engaged with the device manufacturers to improve how the devices are used in practice. This will help enhance biosafety at a national level by ultimately reducing exposure incidents.
Equipping our inspectors to excel
Well-trained, equipped and knowledgeable inspectors are key to promoting and facilitating biosafety in labs across Canada. We continued to enhance our inspectors' training and tools in 2018–19, including updating our training policy, adding new training courses and changing the training schedule to better align with how inspectors are designated. On the technology front, we piloted the use of tablet devices so inspectors could capture their findings electronically. This has improved consistency and shortened times to generate reports by reducing the need to scan or retype documents post-inspection.
BSOs are a big deal
Biological safety officers (BSOs) oversee biosafety and biosecurity at labs across the country. They facilitate regulatory compliance by verifying their organizations' licence applications, communicating with PHAC, and promoting and monitoring compliance. We work closely with them to disseminate best practices — and because they are so important to biosafety, we need to know whenever a regulated party's BSO changes (within 30 days of any change).
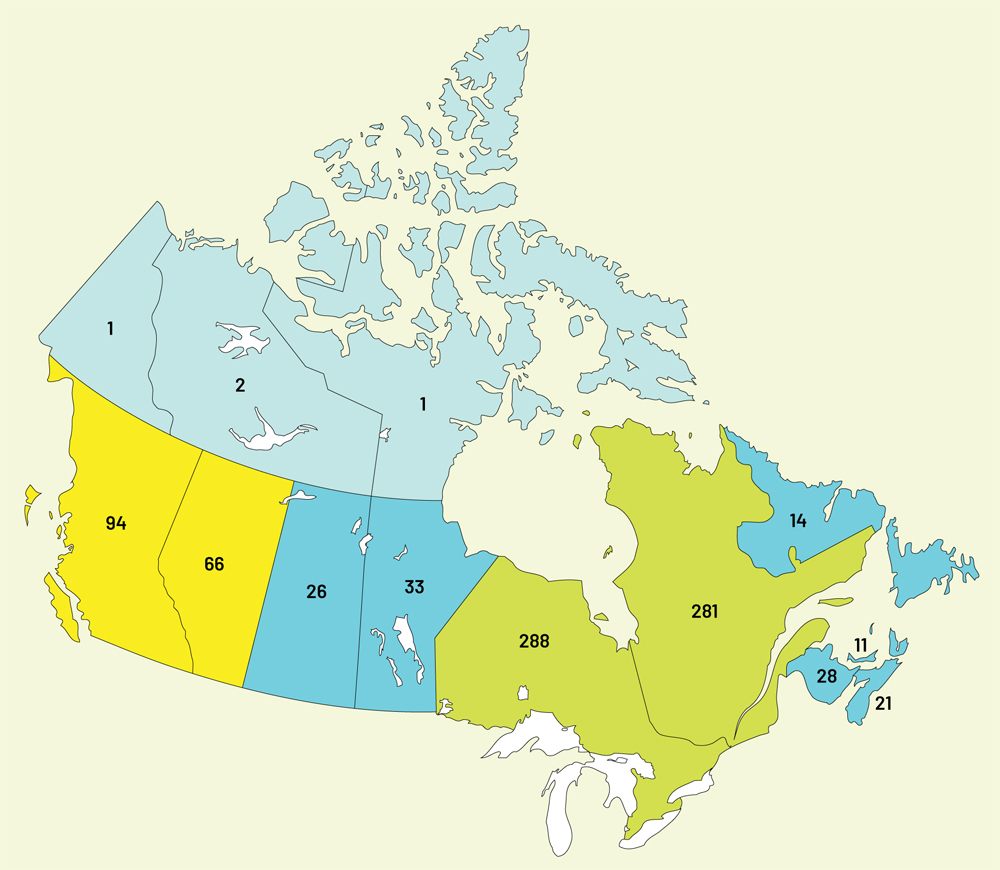
866 BSOs across Canada
Descriptive Text
| BSO Distribution by Province | Q4 |
|---|---|
| Alberta | 66 |
| British Columbia | 94 |
| Manitoba | 33 |
| New Brunswick | 28 |
| Newfoundland and Laborador | 14 |
| Northwest Territories | 2 |
| Nova Scotia | 21 |
| Nunavut | 1 |
| Ontario | 288 |
| Prince Edward Island | 11 |
| Quebec | 281 |
| Saskatchewan | 26 |
| Yukon | 1 |
| Total | 866 |
Maximizing Impact in Program Administration and Service Delivery
Preparing for the peak
In early 2016, after the HPTR came fully into effect, our team processed the licence applications and documentation of more than 860 regulated parties. The majority of the licences issued at that time carried a five-year term, meaning most will need to be renewed in 2021 —a considerable influx of licensing activity to manage over a short period of time. Applying lessons learned in 2016, we took proactive steps to ensure that the coming “peak” of renewals will be managed in a smart, strategic and efficient way.
As a first step, we analyzed licence application data from 2016, looking for trends that could shed light on how the 2021 renewal peak might unfold. Based on that analysis, we made improvements to how and when regulated parties receive automated renewal notifications, and developed a new communications plan (including newsletters and a webinar dedicated to licence renewals) to better inform regulated parties about the process. We also improved our web-based licence application system: if there are no changes to the licensed activities or the information on the licence, a renewal can be submitted with just a few clicks, greatly reducing the administrative burden placed on our regulated parties.
Roughly 30 reduced-term RG2 HPTA licences came up for renewal in 2018, giving us the perfect opportunity to test these new approaches. During the pilot, we noticed some regulated parties were submitting new applications instead of renewals, which are more complex and take longer to process. In response, we created clearer instructions for the renewal notices, further streamlining and accelerating the renewal process.
Thanks to these and other modifications to our systems and protocols, not one licence lapsed during the pilot. That strong result positions us well for the next phase of the pilot in summer 2019, when RG3 licence renewals will be processed — and for the peak still to come.
Process refinements to improve regulatory compliance
We keep track of how our regulated parties implement required corrective actions identified through the program's compliance monitoring and verification activities. Following a detailed analysis of the previous year’s compliance data, we made several changes that led to 88 percent of corrective actions being implemented by deadline, exceeding our 85 percent target and marking a 6 percent improvement from 2017–18. Among these changes were simple fixes like factoring long weekends and holidays into our deadlines, emphasizing the importance of due dates in post-inspection closing meetings, and sending automated reminder emails to regulated parties 15 days ahead of their due dates.
More answers, fewer calls
When regulated parties can quickly access information about pathogens and toxins, they are more likely to comply with regulatory requirements. In July 2018, we launched ePATHogen, a publicly searchable database of nearly 8,000 pathogens and toxins. ePATHogen lists the associated human and animal risk groups for each pathogen/toxin, along with details on containment considerations and regulatory authorities. Having this kind of information at their fingertips allows stakeholders to easily determine if they require a Pathogen and Toxin Licence, without having to call us for support. Since the launch of ePATHogen, monthly inquiries about risk group classifications have declined by 40 percent — allowing our team to focus on handling more complex inquiries.
Reducing Administrative Burden
Regulated parties use the Biosecurity Portal, a centralized electronic system, to apply for licences and communicate reportable incidents to us. Based on user feedback, in 2018–19, we made a number of changes to the portal to streamline and accelerate the licensing process. For example, we now allow regulated parties to change licence-holder information directly through the portal without having to request a form from our team (which would then need to be filled in, scanned and sent back by email) — saving a significant amount of time and effort for all involved.
Maximizing Impact in Compliance Promotion
A two-way dialogue on biosecurity
Ongoing engagement with stakeholders is one of our most effective tools for promoting compliance and encouraging a strong culture of biosafety and biosecurity. While biosafety standards and requirements are generally well understood, through our interactions with our regulated parties, we noted a lack of familiarity with those related to biosecurity. Listening to their needs, in 2018–19 we introduced a suite of new guidance documents and training sessions to facilitate meeting their biosecurity requirements.
The topics focused on in our stakeholder engagement are based largely on the feedback received in the field by inspectors and following our webinars and other outreach activities. Through these interactions, we became aware that while many regulated parties are well trained on protecting their own safety in the lab, far fewer have a solid grasp on the principles of biosecurity. To meet this pressing need, we delivered a webinarFootnote 1 titled Biosecurity Awareness in February 2019 (and again in March). This webinar helped regulated parties better understand what biosecurity is, what's at stake when a pathogen or toxin is stolen or lost, and the steps they can take to mitigate biosecurity risks. In addition, we took part in a regulatory panel at the 2018 Canadian Biosafety Symposium, giving attendees the opportunity to ask questions about this important issue.
During our inspections, we also saw that regulated parties were having difficulty completing their biosecurity plans. Because these plans are so critical — they outline the measures they have put in place to prevent pathogens and toxins from being lost, stolen or misused — we released additional and clearer guidance on that topic. That included publishing a new Canadian Biosafety Guideline, Conducting a Biosecurity Risk Assessment, in July 2018 on how to identify and address biosecurity weaknesses.
Testing new guidelines with the research community
In 2018–19, we began development of a new Canadian Biosafety Guideline to help life science researchers identify, assess and mitigate “dual-use” risks in their labs. A pathogen or toxin with “dual-use potential” could be used for legitimate scientific applications or intentionally misused as a biological weapon. In the life sciences, any asset related to biological material, including knowledge, technologies or products, could have dual-use potential. To make sure the new guidelines are clear, easily understood and meet the needs of their intended users, we posted the draft online for a period of five months in 2018 to solicit feedback. The input we received from life science researchers, biosafety/biosecurity professionals, holders of Pathogen and Toxin Licences, and the general public will help inform the final version of the guidelines.
Making sure our stakeholders are heard
Our program has always relied on a broad range of engagement channels: webinars, online courses, guidelines, newsletter articles, e-blasts, workshops, kiosks at conferences and events, and more. To make those interactions as strong as possible, we developed a new Stakeholder Engagement Strategy in March 2019that formalizes how we reach out to our stakeholders. The strategy puts in place a continuous, structured mechanism for collecting sector-based intelligence and feedback from regulated parties. It also establishes a governance framework for using the collected data to better plan and prioritize future outreach and engagement activities.
The result is a three-year forward plan of engagement activities that will be updated semi-annually, taking into account stakeholder feedback and our own operational lessons learned. For example, if we hear regulated parties are having difficulty using one of our tools, we'll adjust the plan accordingly and make informed decisions about which outreach activities will have the most impact.
Tools to help regulated parties prepare for inspection
Knowledge and compliance go hand in hand. By downloading the CBS Biosafety App — available for free as either a desktop or mobile app — regulated parties can quickly and easily see which requirements of the Canadian Biosafety Standard apply to their labs. They can then develop customized checklists to support their own internal audits or to help better prepare for visits from our inspectors. Plus, the app provides access to the Canadian Biosafety Handbook and related guidelines and directives, and allows users to search keywords across these products.
Supporting government-wide openness and transparency
The Government of Canada is committed to making more data and information available to Canadians than ever before. Our Regulatory Openness and Transparency Framework (ROTF) Action Plan 2017–20is our contribution to that goal, outlining on a year-by-year basis the steps we will take to improve public access to timely, useful and relevant health, safety and scientific information. ROTF activities in 2018–19 were rooted in three pillars: “inform and engage”, “enable” and “enforce”. Our focus was largely based on the action plan’s “enable” pillar, providing regulated parties with the information they need to fulfill their regulatory responsibilities. That included making public our pathogen risk assessment methodology and publishing the ePATHogen online database.
Life science research outside of regulated labs
Not all life sciences laboratories work with pathogens and toxins that are regulated under the HPTA/HPTR. We actively engage with these labs to raise awareness about the regulations and the resources they can access to help them work safely. We also encourage them to keep their work limited to non-regulated pathogens and toxins, which helps them maintain biosafety and biosecurity while avoiding having to take on extra regulatory burden. We frequently work with the do-it-yourself (DIY) biology community to foster safe innovation in community laboratories. Plus, as a member of the National Ethics and Safety Committee of the Canadian-Wide Science Fair, we are able to raise students' awareness of biosafety and biosecurity and help them stay safe as they work with microorganisms in their projects.
Maximizing impact in Intragovernmental Collaboration
Sharing systems and data across the whole of government
The Government of Canada’s “one enterprise” approach aims to better align initiatives, systems and solutions across federal departments and agencies. That includes sharing IT infrastructure and tools to meet common needs. In 2018–19, we played our part by working more closely with partners across the Health Portfolio, including sharing access to our case-management system.
We rely on our case-management system to administer licensing and compliance-related activities — and have been actively updating and improving the electronic system since its launch in December 2015.The lessons we have learned along the way are now being leveraged by other health-focused agencies across the Government of Canada.
For example, many of our regulated parties are also regulated by the Canadian Food Inspection Agency (CFIA), which regulates the import and transfer of certain animal pathogens and toxins. In many cases, both agencies need to collect the same basic organizational details from regulated parties. In 2018–19, we entered into an agreement to give CFIA staff joint access to our case-management system. It now serves as a central repository for that common data, which helps make both our program and CFIA more efficient and demonstrated good value for money in eliminating the need for two separate systems. Further, Health Canada’s Controlled Substances and Cannabis Branch was able to leverage the system to meet their needs and support implementation of the new Cannabis Act in the fall of 2018.
The regulatory ecosystem
Our program is just one part of a broader ecosystem of federal regulators and enforcement agencies, all of which have a role to play in maintaining Canada’s biosafety and biosecurity. Some of the other agencies and departments we work with include:
- Canadian Food Inspection Agency
- Health Canada
- Transport Canada
- Global Affairs Canada
- Environment and Climate Change Canada
- Canada Border Services Agency
- Royal Canadian Mounted Police
- Canadian Security Intelligence Service
- Treasury Board Secretariat
Aligning pathogen oversight across the agri-food sector
Our case-management system is not the only way we've become more closely aligned with CFIA. Canadian agri-food researchers have noted misalignment and duplication between the regulatory regimes for human and animal pathogen oversight (PHAC and CFIA, respectively). This makes it burdensome for them to comply with their regulatory obligations and could even deter them from conducting innovative research in Canada. To address these issues, we have been working with CFIA and other departments to explore how federal regulations can be better aligned and to consider risk-based exemptions for researchers facing duplicative requirements. The aim is to develop a regulatory review roadmap for the agri-food and aquaculture sector by December 2020, which will help minimize regulatory burden and bottlenecks while continuing to maintain appropriate health safeguards.
Two inspection bodies, one voice
PHAC is home to two inspection bodies: our Biosecurity Program and the Travelling Public Program (TPP), which aims to reduce the public health risks associated with food, water and sanitation on planes, trains, ships and other modes of transportation. In 2018–19, staff from both groups participated in and observed each other's inspections and shared best practices. Systems and processes in both programs were then updated and aligned, including training practices, compliance and enforcement policies, standard operating procedures, and even the apparel worn by inspectors — resulting in a more unified approach for all inspectors representing the Centre for Biosecurity.
Maximizing Impact in International Engagement and Capacity-Building
Making the world a safer place
Canada has long been respected as a global leader in biosafety and biosecurity. That position comes with great responsibility — and by sharing our own tools and processes with regulatory authorities in other countries, we can help make the world safer and more secure today and for the long term.
Building international capacity for biosafety and biosecurity reduces the risk of global incidents that could affect Canada domestically, such as the need to manage an infectious disease outbreak. As a lead country in the Global Health Security Agenda (GHSA) Action Package Prevent 3 (APP3) program, we leveraged our strong global reputation to develop a new version of our Analytical Approach to Biosafety and Biosecurity policy tool in 2018–19. This modular, scalable policy toolkit provides practical, step-by-step guidance to help other countries independently establish or strengthen their national policies and oversight frameworks for pathogen biosafety and biosecurity.
Using the feedback collected during consultations held in 2017–18, including pilot workshops in Mali, Sierra Leone, Laos and Trinidad & Tobago, we finalized the second edition of the Analytical Approach tool. Published in September 2019 with an improved methodology, this version of the tool will be heavily marketed and promoted — and shared broadly with our international partners and other regulators.
Through this tool, Canada is able to strategically influence the development of high-impact biosafety and biosecurity programs and policies all over the world. In doing so, we are directly contributing to the achievement of the United Nations Sustainable Development Goal 3, Good Health and Wellbeing. One of the many focus areas for this goal is strengthening the capacity of all countries for early warning, risk reduction and management of national and global health risks. Our four pilot countries will also help spread best practices throughout their own regions, further increasing global capacity to sustainably manage biosafety and biosecurity risks.
Keeping up the global fight against polio
In the move toward a polio-free world, effective containment of the poliovirus to a minimum number of labs is critical. Strengthening existing containment requirements and reducing the number of containment facilities around the world helps reduce the risk of the disease re-emerging. As Canada’s national authority for containment, we continued to meet our international commitments, which include implementing the World Health Organization (WHO) Global Polio Eradication Initiative’s Global Action Plan (GAPIII). We are proud to report that, as a result of our targeted outreach and engagement with regulated parties, the number of Canadian labs working with poliovirus has gone from 23 in 2009 to just five in 2018–19.
To meet Canada’s WHO reporting requirements and establish our country’s inventory of potentially infectious poliovirus material, we distributed three surveys to stakeholders throughout the year. We also contributed to the GAPIII 2019–23 strategic plan and provided key insights to the WHO on GAPIII polio containment requirements.
Our draft cost analysis for implementing GAPIII containment requirements is considered the “gold standard” and has helped other countries understand the financial and resource costs involved. In addition, we participated in numerous international meetings to promote sustainable containment requirements to facilitate the continued protection of Canadians and people around the world.
High marks from international evaluators
The Joint External Evaluation (JEE) was developed by the WHO and GHSA in 2016 to assess countries' ability to prevent, detect and respond to public health risks under the International Health Regulations. Canada's evaluation took place in June 2018, part of which looked at our country's progress toward whole-of-government oversight for biosafety and biosecurity. Canada obtained the highest possible score in this area, with evaluators remarking on the strength of our licensing regime and legislation. Canada also scored 4/5 on biosafety and biosecurity training and practices, with areas for improvement identified that we are now acting upon. These high marks put Canada in an excellent position to influence biosafety and biosecurity around the world — for example, as a partner to regulators in regions with lower JEE scores, whose regimes we could help improve.
A champion of international regulatory collaboration
PHAC is the secretariat for the International Experts Groups of Biosafety and Biosecurity Regulators (IEGBBR), an assembly of regulatory authorities from 11 countries with strong oversight systems for biosafety and biosecurity. The IEGBBR holds face-to-face meetings every 18 months to two years, and the seventh such meeting took place in Ottawa in September 2018. One of the main focuses of this meeting was do-it-yourself (DIY) biology. DIY researchers presented on new technologies and trends as well as the challenges they face related to biosafety and biosecurity — underscoring the continued importance of building international capacity in those areas. Through the discussions that followed, IEGBBR members gained a better understanding of different national perspectives (including the needs of developing versus developed countries), fostering regulatory convergence and a path to strengthened biosecurity and biosafety capacities.
Keeping an eye on emerging technologies
Science is about innovation — and technologies and methodologies in the life sciences are advancing at an increasingly rapid pace around the world. Although many of these technologies have the potential to greatly improve human life, they might also come with risks and consequences that are not yet well understood. Our attendance at meetings, conferences and the annual International Genetically Engineered Machine (iGEM) Competition (where teams of undergraduate students test novel synthetic biological systems in living cells) gives us the opportunity to monitor emerging technologies, influence responsible research and network with technology experts.
Closing Statement
Making evidence-based decisions to maximize efficiency was core to our work in 2018–19and reflects our mindset of continuous improvement.
By being a responsive, modern regulator, we are better able to support our regulated community become or remain compliant with biosafety and biosecurity standards. Through compliance promotion and enforcement we can foster a culture of innovation while protecting the health and safety of Canadians.
- Footnote 1
-
We delivered many more webinars throughout the year on a wide range of topics, including:What to Expect When You Are Inspected, Reporting Under the HPTA/HPTR, Licence Renewal, and Local Risk Assessment.
