Archived - Frequency of HIV screening and testing

 Download this article as a PDF (278 KB - 8 pages)
Download this article as a PDF (278 KB - 8 pages) Published by: The Public Health Agency of Canada
Issue: Volume 42-8: Current travel risks
Date published: August 4, 2016
ISSN: 1481-8531
Submit a manuscript
About CCDR
Browse
Volume 42-8, August 4, 2016: Current travel risks
Systematic review
Canadian and international recommendations on the frequency of HIV screening and testing: A systematic review
Austin T1, Traversy GP1, Ha S1, Timmerman K1*
Affiliation
1 Centre for Communicable Diseases and Infection Control, Public Health Agency of Canada, Ottawa, ON
Correspondence
Suggested citation
Austin T, Traversy GP, Ha S, Timmerman K. Canadian and international recommendations on the frequency of HIV screening and testing: A systematic review. Can Comm Dis Rep 2016;42:161-8. https://doi.org/10.14745/ccdr.v42i08a03
Abstract
Background: In 2014, the Public Health Agency of Canada estimated that 21% of the people living with HIV in Canada were unaware of their infection. Increased screening and testing for HIV is crucial to reducing the number of undiagnosed infections. To ensure the best use of available resources, it is important to determine the optimal intervals for HIV screening and testing.
Objective: To conduct a systematic review of the recommendations for the frequency of HIV screening and testing in different populations.
Methods: To identify eligible guidelines, a comprehensive two-tiered search strategy of journals and websites of governments and non-governmental organizations and a three-tiered screening strategy (title, abstract and full content screen) were used. Guidelines were eligible for inclusion if they, a) were published between 2000 and 2015 in English or French, and b) provided guidance on HIV screening/testing intervals for at least one population.
Results: Of the 609 documents retrieved from the search, 34 guidelines met the eligibility criteria. The most frequently mentioned populations were pregnant women, men who have sex with men (MSM) and the general population. Overall, there was consensus on at least annual testing for MSM, intravenous drug users, individuals with HIV-positive sex partners, individuals with multiple partners, sex workers and their clients, migrants from HIV-endemic countries and indigenous peoples. Of the 20 guidelines that provided recommendations for pregnant women, the most common recommendation (n=9) was to test as early as possible during each pregnancy; four guidelines recommended screening during the first prenatal visit; three recommended routine HIV testing; and four suggested retesting in the third trimester regardless of maternal risk of HIV infection. Consensus on HIV testing of the general public, incarcerated people and individuals diagnosed with other sexually transmitted infections (STIs) was lacking. Four guidelines cited a lack of data for not providing specific recommendations in the general population.
Conclusions: Additional evidence is needed to refine the recommendations for pregnant women and inform the optimal timing of HIV testing, especially in the general population, individuals diagnosed with other STIs and incarcerated people.
Introduction
Diagnosed and treated human immunodeficiency virus (HIV) infection is considered a chronic diseaseFootnote 1. Early detection and treatment of HIV is important not only for the individuals who are infected but also to prevent transmission of the virusFootnote 2. Clinical trials have shown that early initiation of HIV treatment reduces viral load, thereby decreasing infectivity and potentially preventing HIV transmissionFootnote 3Footnote 4.
Low rates of screening and testing have been identified as a potential limiting factor in the success of HIV-prevention strategiesFootnote 5Footnote 6. Approximately 30-50% of new infections are a result of individuals who are unaware of their infectionFootnote 7Footnote 8. Research among men who have sex with men (MSM), injection drug users (IDUs) and heterosexual men and women indicates that once individuals learn of their HIV-positive status they are more likely to take steps to minimize the likelihood of transmissionFootnote 9. However, it was estimated that at the end of 2014, about 21% of people living with HIV in Canada were unaware of their infectionFootnote 10.
The Public Health Agency of Canada (PHAC)'s HIV Screening and Testing Guide recommends HIV screening as part of routine care and annual testing for individuals involved in high-risk practicesFootnote 2. In addition to MSM and IDU, other commonly identified risk groups for HIV infection include individuals with HIV-positive sex partners and individuals with multiple or anonymous partnersFootnote 2Footnote 11Footnote 12Footnote 13Footnote 14Footnote 15.
However, the benefits and frequency of HIV testing must be weighed against costs, and there is a lack of clarity as to the ideal frequency of testing in other populations.
The objective of this systematic review was to address the question: What are the recommended intervals for HIV screening and testing among various population groups in Canada and elsewhere?
Methods
Search strategy
A research librarian-designed comprehensive search of electronic databases identified guidelines published in peer-reviewed journals. Government and non-governmental organization (NGO) websites were also searched to identify any guidelines that may have been posted but not published in the scientific literature. The electronic databases searched included MEDLINE, Embase, Scopus, Cochrane Library and the Canadian Electronic Library (CEL). See Appendix 1 for a complete list of the government and NGO websites searched.
The search terms were the same for both types of searches: "HIV testing frequency," "HIV testing interval," "HIV guideline," "HIV testing guideline," "HIV screening," "HIV screening frequency," "HIV screening guideline," "HIV screening and testing guideline," "HIV screening and testing recommendations," "HIV screening recommendations," "HIV testing recommendations," "STI guidelines," "STI testing intervals" and "STI testing frequency." Search strings for the different databases are identified in Appendix 2.
Inclusion and exclusion criteria are listed in Table 1. The search was restricted to guidelines published or posted within the last 15 years to capture the influence of new HIV prevention methods (e.g., treatment as prevention [TasP]).
| Item | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| Nature of recommendation | Provides position, recommendations or guidance on HIV testing intervals or frequency of testing (all populations and subgroups) Multiple recommendations published from the same organization (e.g., updates or addendums) | No mention of testing intervals or frequency of HIV testing Recommendations related to individuals who already have HIV (e.g., TB/HIV co-infection or HIV treatment/management) |
| Language of publication | English, French | Languages other than English and French |
| Date of publication | Recommendations published from January 2000 to August 2015 | Guidelines published prior to January 2000 |
Guideline selection
We conducted a three-tiered screening process: title screen, abstract screen and full content screen. Three authors (TA, GT and SH) independently screened the titles. Titles with the terms "guideline," "strategy," "directive," "recommendation," "guidance" or "position" were included. Guidelines on HIV treatment or the management of opportunistic infections and co-infected populations were excluded. Two authors (TA, GT) independently screened the abstracts and excluded those that did not refer to HIV screening and testing. Disagreements between reviewers at either stage were resolved through discussion with a third reviewer (KT) and a fourth, if required. Two authors (TA, GT) then completed the full content screen. Guidelines that did not provide specific information on the recommended frequency or intervals of HIV screening and testing for any population group were excluded. Only primary source guidelines were included. Guidelines were considered duplicates if the same recommendation was published in multiple locations or if an article summarized or endorsed a guideline.
Data extraction
Data were extracted on the country of publication, population screened and testing frequency. Guidelines were categorized as Canadian, foreign, or international (spanning multiple countries, such as those from the World Health Organization). The population groups initially targeted in this review were the general population, MSM and IDU, but other population groups were also noted.
Results
A total of 609 documents were retrieved from the search. After duplicates were removed and inclusion/exclusion criteria applied, the final review included 34 guidelines (Figure 1).
Figure 1: Screening methodology and final results flow chart
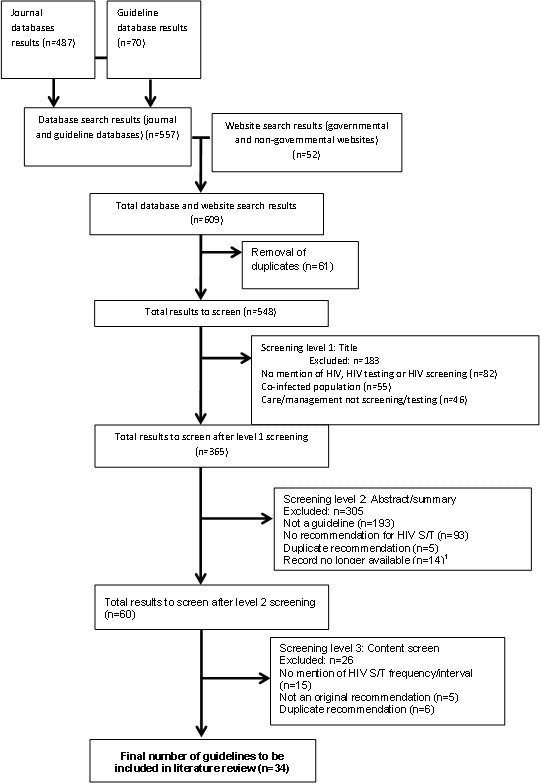
Text description: Figure 1
Figure 1: Screening methodology and final results flow chart
Figure 1 is a flow diagram showing the method for the retrieval and assessment of documents to identify the guidelines for the study. At the top are two boxes showing the sources of searches and the results. Box 1 identifies the journal databases results (n = 487) and Box 2 identifies the Guideline database results (n = 70). This then leads to Box 3 showing the combined Database search results (journal and guideline database) (n = 557). This is then linked with Box 4 that identifies the website search results (governmental and non-governmental websites) (n = 52). This then leads to Box 5 showing the combined database and website search results (n = 609). Box 6 identifies the removal of duplicates (n = 61), that then leads to Box 7 that identifies the total results to screen (n = 548). This leads to Box 8 that identifies: Screening level 1 of the Title, which excludes: n = 183 due to: No mention of HIV, HIV testing or HIV screening (n = 82), Co-infected population (n = 55), or Care/management not screening/testing (n=46) This leads to Box 9: Total results to screen after level 1 screening (n = 365). This is followed by Box 10 that identifies the Screen level 2 of the Abstract or summary which excluded: n = 305 due to: Not a guideline (n = 193), No recommendation for HIV screening/testing (n = 93). Duplicate recommendation (n = 5). And Record no longer available (n = 14). This leads to Box 11 for the Total results to screen after level 2 screening (n = 60). This is followed by Box 12 that identifies the Screening level 3: Content Screen that excluded: n = 26 due to: No mention of HIV screening/testing frequency/interval (n = 15), Not an original recommendation (n = 5), or a duplicate recommendation (n = 6). Box 13 identifies the final number of guidelines to be included in literature review (n = 34).
The characteristics of included guidelines are summarized in Table 2. Two-thirds (65%) came from the USA and Europe. In addition to the general population, MSM and IDU, several other key populations emerged: pregnant women, migrants from HIV-endemic regions, indigenous peoples, adolescents, incarcerated individuals, partners of unknown HIV status and others.
| Type | Characteristic | Number (%) |
|---|---|---|
| Geographic region | United States | 12 (35) |
| Europe | 10 (29) | |
| Canada | 5 (15) | |
| Africa | 2 (6) | |
| Australia | 2 (6) | |
| World Health Organization | 2 (6) | |
| Asia | 1 (3) | |
| Key populations mentioned | Pregnant | 20 (59) |
| MSM | 19 (56) | |
| General population | 14 (41) | |
| IDU | 13 (38) | |
| Multiple partners | 7 (21) | |
| HIV-positive sex partner | 7 (21) | |
| Other STI diagnosis | 5 (15) | |
| Sex workers and their clients | 4 (12) | |
| Migrants from HIV-endemic countries | 4 (12) | |
| Indigenous peoples | 3 (9) | |
| Adolescents | 3 (9) | |
| Incarcerated individuals | 3 (9) | |
| Transgender men and women | 2 (6) | |
| Partner with unknown HIV status | 2 (6) | |
All 34 Canadian, foreign and international guidelines are summarized in Table 3. Of these, 9 provided recommendations only for high-risk groups and the remaining 25 provided recommendations for other risk groups (e.g., pregnant women and the general population). Five guidelines were updates to previous guidelines.
| Source | General | Pregnant | Multiple partners | HIV+ partner | MSM/ MSM sex partner | IDU/IDU sex partner | Incarcerated individuals | Adolescents | Transgender men/women | Partner of unknown HIV status | Sex workers and their clients | Migrants from HIV- endemic countries | Indigenous peoples | Other STIs | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Canadian | Quebec (2011)Footnote 19 | NSR | First prenatal visit | More frequent | ALA | ALA | ALA | Annual risk evaluation | Annual risk evaluation | NSR | NSR | NSR | NSR | Annual risk evaluation | 3-6 months |
| PHAC (2012)Footnote 2 | Normalized testing | First prenatal visit; repeat if high risk | NSRTable 3 Footnote 2 | NSRTable 3 Footnote 2 | NSRTable 3 Footnote 2 | NSRTable 3 Footnote 2 | NSRTable 3 Footnote 2 | NSR | NSR | NSR | NSRTable 3 Footnote 2 | NSR | NSR | NSR | |
| Ontario (2012)Footnote 32 | After high risk exposure | NSR | ALA | ALA | ALA | ALA | NSR | NSR | NSR | ALA | NSR | ALA | ALA | NSR | |
| Saskatchewan (2014)Footnote 22 | Every 5 years | Routine prenatal care | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| British Columbia (2014)Footnote 20 | Routine; every 5 years | First prenatal visit; repeat if high risk | ALA | ALA | ALA | ALA | After incarceration | NSR | NSR | NSR | ALA | ALA | ALA | NSR | |
| Foreign | CDC (multiple years)Footnote 13Footnote 17Footnote 24Footnote 25Footnote 31Footnote 33Footnote 34 | Routine; re-test based on riskFootnote 13Footnote 17Footnote 25 | Each pregnancyFootnote 31; retest in 3rd trimesterFootnote 13Footnote 17Footnote 25Footnote 31 | ALAFootnote 13 | ALAFootnote 13 | ALAFootnote 13Footnote 17Footnote 31 | ALAFootnote 13 | RoutineFootnote 34 | Re-test based on riskFootnote 17 | Re-test based on behavioural historyFootnote 17 | NSR | ALAFootnote 13 | NSR | NSR | Retest with new complaintFootnote 13 |
| Seattle/King County (2001)Footnote 35 | NSR | NSR | NSR | NSR | ALA | ALA | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| UK (2006, 2008)Footnote 21Footnote 36 | Routine; retest after windowFootnote 21Footnote 36 | Beginning of pregnancyFootnote 21 | NSR | NSR | ALAFootnote 21 | ALAFootnote 21 | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| Liberia (2007)Footnote 37 | Re-test after window | Re-test in 3rd trimester | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| Afghanistan (2008)Footnote 28 | Additional research needed | NSR | 6-12 months | 6-12 months | 6-12 months | 6-12 months | NSR | NSR | NSR | NSR | 6-12 months | NSR | NSR | NSR | |
| ACP/HIV Medicine Association (2009)Footnote 38 | Routine | Each pregnancy | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| France (2009)Footnote 29 | Routine; Additional research needed | NSR | NSR | NSR | Annually (if multiple partners) | Annually | NSR | NSR | NSR | NSR | NSR | Annually (if sex partner is from HIV endemic region) | NSR | NSR | |
| Central African Republic (2010)Footnote 39 | Routine testing at STI clinics | Beginning and end of each pregnancy | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| STIs in Gay Men Action Group (2010, 2014)Footnote 40Footnote 41 | NSR | NSR | NSR | NSR | ALAFootnote 40Footnote 41 | 3-6 months (if MSM)Footnote 41 | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| NICE (2011)Footnote 27Footnote 42 | Routine at specialised clinicsFootnote 27 | NSR | NSR | RoutineFootnote 27 | AnnuallyFootnote 42; routineFootnote 27 | RoutineFootnote 27 | NSR | NSR | NSR | NSR | NSR | RoutineFootnote 27 | NSR | RoutineFootnote 27 | |
| New York (2011)Footnote 43 | NSR | Re-test 3rd trimester | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| USPSTF (2014)Footnote 18 | No retest for low riskTable 3 Footnote 3 | Every pregnancy | NSRTable 3 Footnote 3 | NSRTable 3 Footnote 3 | NSRTable 3 Footnote 3 | NSRTable 3 Footnote 3 | NSRTable 3 Footnote 3 | Screen for risk | NSR | NSR | NSRTable 3 Footnote 3 | NSRTable 3 Footnote 3 | NSRTable 3 Footnote 3 | NSR | |
| ACOG (2015)Footnote 44 | NSR | Beginning of pregnancy; retest in 3rd trimester | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| InternationalTable 3 Footnote 1Footnote 30 | Europe (2008, 2014)Footnote 16Footnote 30 | Routine; Additional research neededFootnote 16Footnote 30 | RoutineFootnote 16 | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR |
| WHO (2009, 2010)Footnote 15Footnote 45 | NSR | Beginning of pregnancy; repeat if high riskFootnote 15Footnote 45 | ALAFootnote 45 | ALAFootnote 15Footnote 45 | ALAFootnote 15Footnote 45 | NSR | NSR | ALAFootnote 45 | ALAFootnote 45 | ALAFootnote 15Footnote 45 | NSR | NSR | Re-test with new complaintFootnote 45 | ||
| EMCDDA (2010)Footnote 14 | NSR | NSR | NSR | NSR | NSR | 6-12 months | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | |
| ECDC (2010, 2015)Footnote 23Footnote 26 | NSR | RoutineFootnote 23 | NSR | NSR | RoutineFootnote 23Footnote 26 | NSR | NSR | NSR | NSR | NSR | NSR | NSR | NSR | RoutineFootnote 23 | |
More recent guidelines have focused on routine testing (i.e., not recommending a specific testing interval but rather providing a recommendation to test everyone) with more frequent testing for individuals who engage in high-risk behavioursFootnote 2Footnote 16Footnote 17Footnote 18.
The most frequently mentioned populations were pregnant women, MSM and the general population. Of all 20 guidelines that provided recommendations for pregnant women, the most common recommendation (n=9) was to test as early as possible during each pregnancy; 4 guidelines recommended screening during the first prenatal visitFootnote 2Footnote 19Footnote 20Footnote 21; 3 recommended routine HIV testingFootnote 16Footnote 22Footnote 23; and 4 suggested retesting in the third trimester regardless of maternal risk of HIV infectionFootnote 13Footnote 17Footnote 24Footnote 25.
Of the 19 guidelines that considered MSM, 14 provided a specific testing frequency, 3 recommended routine testing without specific testing intervalsFootnote 23Footnote 26Footnote 27 and 2 cited insufficient evidence to determine a testing interval but recommended that MSM be screened more frequentlyFootnote 2Footnote 18. Altogether 14 recommended testing at least annually.
Testing at least annually was also the most common recommendation for IDU (11 of 13 guidelines), individuals with HIV-positive sex partners (6 of 7), individuals with multiple partners (4 of 7), sex workers and their clients (4 of 4), migrants from HIV-endemic countries (3 of 4) and indigenous peoples (2 of 3). Overall, the recommendations for frequency of testing higher-risk populations varied little.
Recommendations for the general population varied slightly. Some (2 of 14) focused on a specific time, whereas the majority (8 of 14) focused on routine or normalized testing without providing a specific interval (e.g., PHAC, European Union, Central African Republic). Four guidelines cited a lack of data as reason for not providing specific recommendations in the general population Footnote 16Footnote 28Footnote 29Footnote 30.
The guidelines disagree about whether sufficient evidence exists to formulate testing frequency recommendations in certain populationsFootnote 2Footnote 18Footnote 28Footnote 29. This is the case for both populations in which there is consistency across guideline recommendations (e.g., MSM and IDU), and for populations in which there was some consistency or no guidelines (e.g., incarcerated people).
Guidelines for people diagnosed with STIs commonly recommend routine HIV testing (n=3) or re-testing with each new STI diagnosis (n=2). The emergence of this population in this review highlights STI diagnoses as a potential proxy for high-risk sexual behaviour and identifying individuals at higher risk for HIV infection.
The least frequently mentioned populations included incarcerated individuals, adolescents, individuals with partners of unknown HIV status, and transgender men and women.
Discussion
This review identified 34 guidelines on the frequency of HIV testing. In addition to testing frequency recommendations for high-risk groups, several guidelines also included recommendations for the general population and pregnant women, highlighting a shift from risk-based and targeted-testingFootnote 13Footnote 24Footnote 33Footnote 46 to incorporating HIV testing into routine careFootnote 2Footnote 13Footnote 16Footnote 18Footnote 21Footnote 32Footnote 39. There was good consensus that testing at least annually is recommended in higher-risk populations.
Most guidelines suggest testing early in pregnancy, and some recommend testing again in the third trimester. There is a lack of consensus on some subgroups (i.e., incarcerated individuals, the general population and individuals diagnosed with other STIs), and there appears to be insufficient evidence to make recommendations for the general population and incarcerated people. Differences in the recommendations for population groups may be a result of the varying types of evidence used to inform the guidelines.
Several factors should be considered when interpreting these results. The strengths of this review include a thorough search strategy, and consistent, objective assessment and data extraction of the studies. There are also a number of limitations. Guidelines that may have been published in languages other than French and English were not included in this review. Of note, there were few published guidelines from Asia and Africa. Since these regions have high HIV incidence and prevalence rates, guidelines from these regions were either not captured by our search parameters or there is a lack of guidance on the optimal intervals for HIV screening and testing in these regions.
Research is needed to examine and critically appraise the evidence for the frequency of HIV testing recommendations in various populations. Specific research could be aimed at identifying the optimal testing interval for the general population, for adolescents, for transgender men and women and for incarcerated peopleFootnote 47Footnote 48 as well as the optimal frequency of testing for indigenous peoples, ethnocultural communities with high incidences of HIV and domestic migrant workers.
In summary, HIV screening and testing is an extremely important tool within the continuum of HIV care. Although many guidelines have been developed to identify the ideal frequency of testing for different populations, there are inconsistencies among them and the evidence base for some populations appear to be lacking. Additional evidence to inform the optimal frequency of HIV screening and testing in different populations could strengthen the global efforts to eradicate this disease.
Acknowledgements
We would like to thank Jun Wu and Margaret Gale-Rowe for their constructive feedback on the draft manuscript. We would also like to thank Ella Westhaver, the research librarian who helped us design and carry out our systematic guideline search, and Jessica Yau for her contributions to the revision of this manuscript.
Conflict of interest
None.
Funding
This study was supported by the Public Health Agency of Canada. The authors have no additional sources of external funding to declare.
Appendix 1: Websites searched
The website search was divided into national and international sites and government and non-governmental/stakeholder sites.
| Type of website | Organization |
|---|---|
| National governmental |
|
| International governmental |
|
| National non-governmental / stakeholder |
|
| International non-governmental / stakeholder |
|
Appendix 2: Database Search Strings
MEDLINE Search String
Database(s): Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations, Ovid MEDLINE(R) Daily, Ovid MEDLINE(R) and Ovid OLDMEDLINE(R) 1946 to Present
| No. | Searches | Results |
|---|---|---|
| 1 | (hiv or human immunodeficiency or human immune deficiency or vih).ti. | 188944 |
| 2 | exp *HIV Infections/ or exp *HIV/ or exp HIV Infections/ep | 246368 |
| 3 | (hiv positive* or hiv+ or vih positi* or vih+).ti. | 161297 |
| 4 | 1 or 2 or 3 | 272101 |
| 5 | exp Mass Screening/ | 106834 |
| 6 | (frequency or schedule or interval?).mp. [mp=title, abstract, original title, name of substance word, subject heading word, keyword heading word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier] | 1348682 |
| 7 | (guideline? or recommendation? or policy).mp. [mp=title, abstract, original title, name of substance word, subject heading word, keyword heading word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier] | 614880 |
| 8 | ((Testing or screening) adj5 (frequency or interval or guideline? or recommendation?)).ti,hw. | 2834 |
| 9 | 4 and 7 and 8 | 178 |
| 10 | 4 and 5 and 6 and 7 | 94 |
| 11 | 9 or 10 | 265 |
| 12 | 10 not 9 | 87 |
| 13 | ((Testing or screening) adj6 (frequency or interval or guideline? or recommendation?)).ti,hw. | 4611 |
| 14 | 4 and 7 and 13 | 238 |
| 15 | 14 not 9 | 60 |
| 16 | limit 14 to (yr="2000 -Current" and (english or french)) | 182 |
Scopus Search String
Scopus OECD ( TITLE ( hiv OR aids OR vih) AND ( TITLE-ABS-KEY ( ( hiv OR aids OR vih) W/6 ( testing OR screening) W/6 ( frequency OR interval* OR guidelin* OR recommendation*))) AND TITLE-ABS-KEY ( guidelin* OR recommendation*) PUBYEAR > 2000) AND ( TITLE-ABS-KEY ( spain OR slovakia OR poland OR portugal OR greece OR germany OR france OR finland OR denmark OR "Czech Republic" OR canad* OR belgium OR austria OR australia OR norway OR "New Zealand" OR netherlands OR mexico OR luxembourg OR korea OR japan OR italy OR iceland OR hungary OR ireland OR "United States" OR great-britain OR "United Kingdom" OR turkey OR switzerland OR sweden)) AND ( LIMIT-TO ( LANGUAGE, "English") OR LIMIT-TO ( LANGUAGE, "French")) 284
Embase Search String
| No. | Searches | Results |
|---|---|---|
| 1 | (Spain or Slovakia or Poland or Portugal or Greece or Germany or France or Finland or Denmark or Czech-Republic or Canada or Belgium or Austria or Australia or Norway or New-Zealand or Netherlands or Mexico or Luxembourg or Korea or Japan or Italy or Iceland or Hungary or Ireland or United-States or Great-Britain or Turkey or Switzerland or Sweden).mp. | 3697234 |
| 2 | (hiv or human immunodeficiency or human immune deficiency or vih).ti. | 213771 |
| 3 | exp *Human immunodeficiency virus/ | 75032 |
| 4 | Human immunodeficiency virus infection/ep | 32614 |
| 5 | *Human immunodeficiency virus infection/ | 154397 |
| 6 | 2 or 3 or 4 or 5 | 258779 |
| 7 | exp mass screening/ | 174825 |
| 8 | (frequency or schedule or interval?).mp. | 1558540 |
| 9 | (guideline? or recommendation? or policy).mp. | 917993 |
| 10 | 6 and 7 and 8 and 9 | 62 |
| 11 | ((Testing or screening) adj6 (frequency or interval or guideline? or recommendation?)).ti,hw. | 6001 |
| 12 | 6 and 9 and 11 | 313 |
| 13 | 1 and 12 | 123 |
| 14 | 12 not 13 | 190 |
| 15 | limit 12 to ((english or french) and yr="2000 -Current") | 245 |
Cochrane Library Search String
"hiv".ti and "interval".ti and "guideline".ti and "recommendation".ti Publicaton Year from 2000 to 2015 (Word variation have been searched)
Canadian Electronic Library Search String
Canadian Electronic Library: Canadian Publishers Collection, Canadian Public Policy Collection, Canadian Health Research Collection
Title:HIV AND all:screening OR Testing