Laboratory exposure to human pathogens and toxins, Canada 2020

 Download this article as a PDF
Download this article as a PDF Published by: The Public Health Agency of Canada
Issue: Volume 47 No. 10, October 2021: Influenza Vaccine
Date published: October 2021
ISSN: 1481-8531
Submit a manuscript
About CCDR
Browse
Volume 47 No. 10, October 2021: Influenza Vaccine
Surveillance
Surveillance of laboratory exposures to human pathogens and toxins, Canada 2020
Nicole Atchessi1, Megan Striha1, Rojiemiahd Edjoc1, Emily Thompson1, Maryem El Jaouhari1, Marianne Heisz1
Affiliation
1 Centre for Biosecurity, Public Health Agency of Canada, Ottawa, ON
Correspondence
Suggested citation
Atchessi N, Striha M, Edjoc R, Thompson E, El Jaouhari M, Heisz M. Surveillance of laboratory exposures to human pathogens and toxins, Canada 2020. Can Commun Dis Rep 2021;47(10):422–9. https://doi.org/10.14745/ccdr.v47i10a04
Keywords: laboratory exposures, laboratory incidents, laboratory-acquired infections, human pathogens and toxins, surveillance, Laboratory Incident Notification Canada, Centre for Biosecurity
Abstract
Background: The Laboratory Incident Notification Canada surveillance system monitors laboratory incidents reported under the Human Pathogens and Toxins Act and the Human Pathogens and Toxins Regulations. The objective of this report is to describe laboratory exposures that were reported in Canada in 2020 and the individuals who were affected.
Methods: Laboratory incident exposures occurring in licensed Canadian laboratories in 2020 were analyzed. The exposure incident rate was calculated and the descriptive statistics were performed. Exposure incidents were analyzed by sector, activity type, occurrence type, root cause and pathogen/toxin. Affected persons were analyzed by education, route of exposure sector, role and laboratory experience. The time between the incident and the reporting date was also analyzed.
Results: Forty-two incidents involving 57 individuals were reported to Laboratory Incident Notification Canada in 2020. There were no suspected or confirmed laboratory acquired infections. The annual incident exposure rate was 4.2 incidents per 100 active licenses. Most exposure incidents occurred during microbiology activities (n=22, 52.4%) and/or were reported by the hospital sector (n=19, 45.2%). Procedural issues (n=16, 27.1%) and sharps-related incidents (n=13, 22.0%) were the most common occurrences. Most affected individuals were exposed via inhalation (n=28, 49.1%) and worked as technicians or technologists (n=36, 63.2%). Issues with standard operating procedures was the most common root cause (n=24, 27.0%), followed by human interactions (n=21, 23.6%). The median number of days between the incident and the reporting date was six days.
Conclusion: The rate of laboratory incidents were lower in 2020 than 2019, although the ongoing pandemic may have contributed to this decrease because of the closure of non-essential workplaces, including laboratories, for a portion of the year. The most common occurrence type was procedural while issues with not complying to standard operating procedures and human interactions as the most cited root causes.
Introduction
Laboratory work with human pathogens and toxins (HPTs) poses an inherent risk to the security of laboratory personnel. While safety practices and regulations of HPTs have evolved considerably over the years, accidental or deliberate exposure to human pathogens and toxins in laboratory settings remain a biosafety and biosecurity concern, both within Canada and abroad.
In response to the reporting requirements for incidents involving HPTs outlined by the 2009 Human Pathogens and Toxins Act (HPTA)Footnote 1, the Laboratory Incident Notification Canada (LINC) surveillance system was launched in December 2015. The LINC system is unique in that it is one of the first comprehensive national surveillance systems to provide a systematic framework for reporting HPT exposures and laboratory-acquired infections (LAIs) across various settings. A total of 247 exposure incidents have been reported between 2016 and 2019, involving a total of 539 individuals among private, public, hospital, and academic sector laboratoriesFootnote 2Footnote 3Footnote 4Footnote 5. In contrast, national reporting requirements for LAIs among other countries is often voluntary or conducted via retrospective surveyFootnote 6Footnote 7Footnote 8Footnote 9.
The Public Health Agency of Canada's Centre for Biosecurity is mandated to protect the health and safety of the public against risks posed by HPTs through the administration and enforcement of the HPTA and the Human Pathogen and Toxins Regulations (HPTR). Under the HPTA, all Canadian laboratory facilities conducting controlled activities with HPTs are required to obtain a license, unless otherwise exempted. Under the HPTA, all licensed facilities are required to report laboratory incidents involving risk group 2 (RG2) pathogens or above in the following instances:
- Exposures and laboratory-acquired infections/intoxication
- Inadvertent release, production, or possession of an HPT
- Missing, stolen or lost HPT, including security sensitive biological agents (SSBA) not received within 24 hours of the expected date and time of receipt
- Changes in biocontainment
Canadian Biosafety Standard (CBS) Second Edition categorizes pathogens among four RGs, dependent upon a pathogen's risk to the individual and to the communityFootnote 10. The RG2 pathogens pose a low risk to public health, but a moderate risk to an individual's health. These pathogens can cause serious disease in humans but are unlikely to do so. The RG3 pathogens pose a low risk to public health, but a high risk to an individual's health, and are likely to cause serious disease in humans. Finally, RG4 pathogens pose a high risk to both public and individual health and are likely to cause serious disease in humans that often leads to death.
The 2020 Annual Report marks the fifth year of the program and would normally be the year at which a baseline on incident reporting is established. However, due to the unprecedented response to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic by the Public Health Agency of Canada and the associated resource re-allocation, the development of a baseline will occur in the 2022 annual report, to be released in 2023.
As with previous years, this annual report aims to describe the distribution of laboratory incidents reported to LINC across years with special attention to exposures, LAIs and factors associated with these exposures at the license (by sector of exposures, HPT, occurrence type) and person (number of affected persons, education, main role, type of activity, years of experience, route of exposure, root causes) level.
Methods
Data sources
The Biosecurity Portal, LINC's external interface, receives notification and follow-up report(s) of laboratory incidents, which are then captured by the internal Customer Relationship Management system. For this report, exposure incidents that took place from January 1, 2020 to December 31, 2020 were extracted from the Customer Relationship Management system. Incidents that did not have a known occurrence date were also included if they were reported during this period. Data of the most recent follow-up reports were used for analysis, while the data of initial reports were used where corresponding follow-up reports and/or data were not present as of the data extraction date, February 8, 2021. Extracted data were cleaned by investigation of any outliers and removal of duplicate entries. It should be noted that while licensed facilities are obligated to report laboratory incidents, the rate of non-reporting is currently unknown and a confounder in this analysis.
Within the scope of the HPTA/HPTR, an exposure incident was defined as a laboratory incident that may have resulted in intoxication/infection or had resulted in suspected or confirmed LAIFootnote 1Footnote 10. A non-exposure incident referred to inadvertent possession or production of an HPT that is a higher RG than the lab is licensed to work with, release of a pathogen or toxin (to which no laboratory personnel are exposed), or a missing, lost or stolen pathogen or toxin or a security-sensitive biological agent not being received within 24 hours of expected arrival.
Analysis
Data from reports submitted to the LINC surveillance system were extracted to Microsoft Excel 2016 for analysis and R 4.0.2 was used to perform descriptive statistics with cross-validation using SAS EG 7.1. All exposure incidents were first subdivided into ruled out incidents and confirmed incidents, with confirmed and suspected LAIs included in the latter. Reports can be ruled out for a variety of reasons, including if no exposure was found to have occurred, if the exposure involved an RG1 HPT or an HPT in its natural environment such as a primary specimen (neither are mandated by the HPTA and these reports are considered voluntary) or if duplicate reports are received. Affected persons in confirmed incidents were also subdivided into confirmed or ruled out individuals. Among confirmed exposure incidents, the numbers of incidents were analyzed against parameters obtained at two levels of reporting. At the level of the active license holder, the distributions of incidents by sector, main activity, root cause, occurrence type, and implicated pathogen/toxin reported were examined as well as reporting delays. At the level of persons affected in these incidents, the distributions of their highest level of education, years of experience, route of exposure, sector and regular role were examined. Particular attention was given to exposures involving SARS-COV-2 because of its status as an emerging pathogen and its role in the ongoing coronavirus disease 2019 (COVID-19) pandemic.
A comparison of exposure incidents and a measure of the exposure incident rate per 100 active licenses from 2016 to 2020 were also performed. The incident rate was described in greater detail in a previous reportFootnote 5. Active licenses are licenses that were considered active during 2020 and were able to report an incident. Given the unavailability of the number of active licenses for December 31, 2020 owing to the impact of the pandemic on normal operations, and given the low fluctuation over the year (25–50 licenses each year), the number of active licenses on April 2020 was used for the calculation of the exposure incidence rate. The median time between the date of occurrence and the date of submission of the exposure incidents was also calculated. Median values were chosen compared to mean values owing to the presence of extreme outliers.
Results
Between January 1, 2020 and December 31, 2020, LINC received 96 laboratory incident reports: 56 exposure reports, 27 non-exposure reports and 13 other reports (Figure 1). All 13 other reports described changes within the laboratory that could affect biocontainment. There were 14 exposure reports and one non-exposure report ruled out, leaving 42 exposure incidents and 26 non-exposure incidents (Figure 2). There were no suspected or confirmed LAIs in 2020. From the exposure reports, 79 people were identified as having been exposed in laboratory incidents. Upon further investigation, 22 of those people were ruled out, leaving a total of 57 exposed people in 2020.
Figure 1: Types of incidents reported to Laboratory Incident Notification Canada and exposure incidents included in analysis, Canada 2020
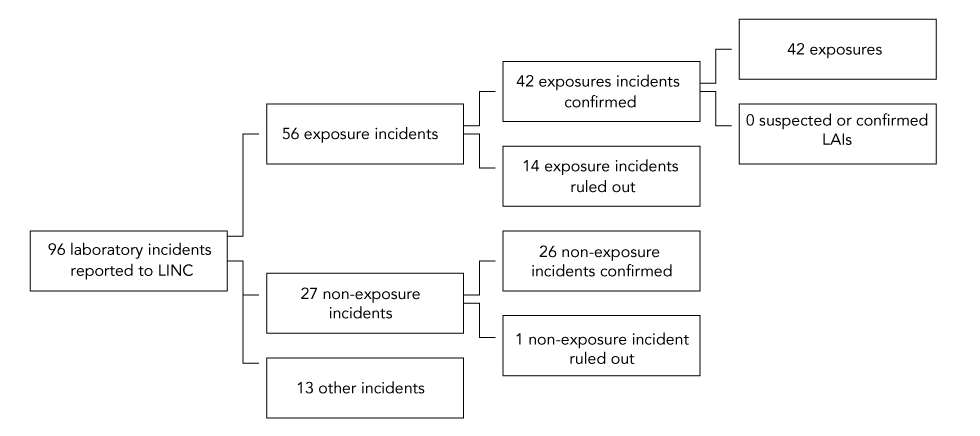
Text description: Figure 1
This figure depicts the breakdown of incidents reported to the Laboratory Incident Notification Canada surveillance system in 2020. Of the 96 incidents reported there were 56 exposure incidents, 27 non-exposure incidents, and 13 other incidents. Among the 56 exposure incidents, one was ruled out from analysis and 55 resulted in exposures. There were no LAIs suspected or confirmed in 2020.
Figure 2: Confirmed exposure incidents, suspected and confirmed laboratory acquired infections and active licenses, Canada 2016–2020
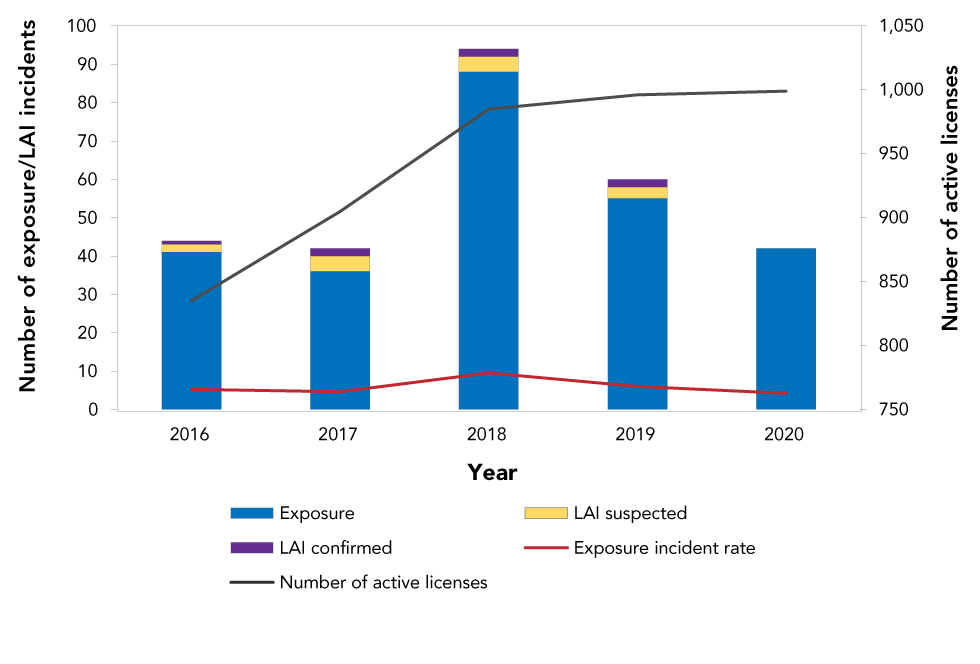
Text description: Figure 2
| Year | Type | Number of exposures/LAI incidents |
|---|---|---|
| 2016 | Exposure | 41 |
| 2016 | LAI suspected | 2 |
| 2016 | LAI confirmed | 1 |
| 2017 | Exposure | 36 |
| 2017 | LAI suspected | 4 |
| 2017 | LAI confirmed | 2 |
| 2018 | Exposure | 88 |
| 2018 | LAI suspected | 4 |
| 2018 | LAI confirmed | 2 |
| 2019 | Exposure | 55 |
| 2019 | LAI suspected | 3 |
| 2019 | LAI confirmed | 2 |
| 2020 | Exposure | 42 |
| 2020 | LAI suspected | 0 |
| 2020 | LAI confirmed | 0 |
| Year | Number of active licenses | Exposures | Exposure incident rate |
|---|---|---|---|
| 2016 | 835 | 44 | 5.3 |
| 2017 | 905 | 42 | 4.6 |
| 2018 | 985 | 94 | 9.5 |
| 2019 | 996 | 60 | 6.0 |
| 2020 | 999 | 42 | 4.2 |
There were 999 active licenses held in Canada permitting the use of HPTs in 2020. The exposure incident rate was 4.2 incidents per 100 active licenses in 2020. The total number of incidents and the rate of incidents per 100 active licenses was lower in 2020 than in 2019 (60 exposure incidents and 6.0 per 100 active licenses) (Figure 2).
Exposure incidents by main activity and sector
Microbiology was the most common activity being performed during exposure incidents (n=22, 52.4%), followed by in vivo animal research (n=5, 11.9%). Other activities include cell culture, autopsy/necropsy, maintenance, animal care, molecular investigation, microscopy or other (n=15, 35.7%). Definitions of activities are available in Appendix A1.
Most exposure incidents occurred in the hospital sector (n=19, 45.2%) followed by the academic sector (n=16, 38.1%) (Figure 3). The hospital sector had the highest number of exposure incidents per 100 active licenses (10.4 per 100), while the environmental health sector had the lowest with no incidents reported in 2020.
Figure 3: Confirmed exposures incidents and active licenses by sector reported to Laboratory Incident Notification Canada, Canada 2020
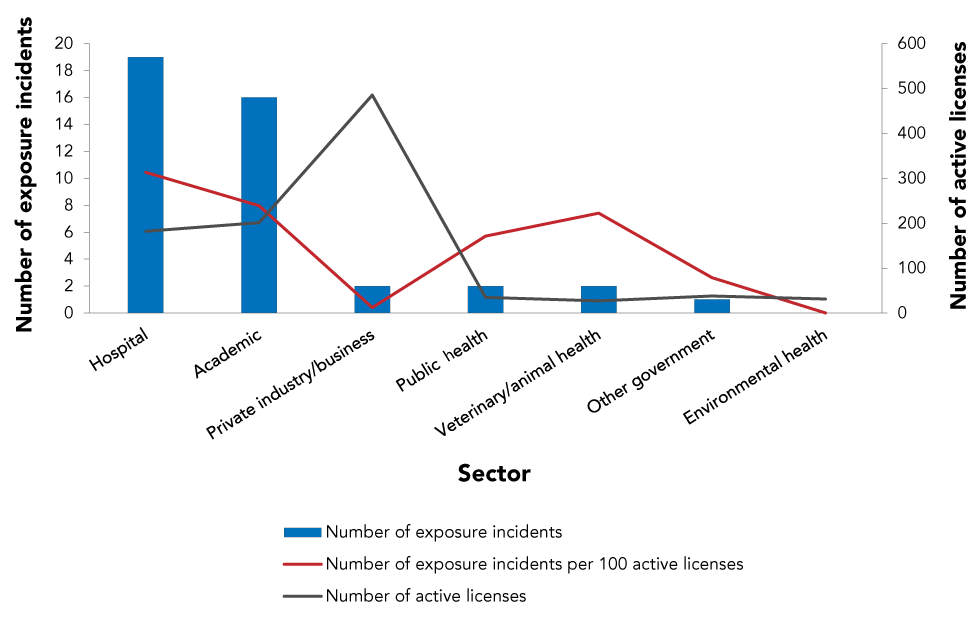
Text description: Figure 3
| Sector | Number of exposure incidents | Percent | Number of active licenses | Number of exposure incidents per 100 active licenses |
|---|---|---|---|---|
| Hospital | 19 | 45.2% | 182 | 10.4 |
| Academic | 16 | 38.1% | 201 | 8.0 |
| Private industry/business | 2 | 4.8% | 485 | 0.4 |
| Public health | 2 | 4.8% | 35 | 5.7 |
| Veterinary/animal health | 2 | 4.8% | 27 | 7.4 |
| Other government | 1 | 2.4% | 38 | 2.6 |
| Environmental health | 0 | 0.0% | 31 | 0.0 |
| Sum | 42 | 100.0% | 999 | Not applicable |
Implicated human pathogens and toxins
Among the 42 implicated biological agents, most were non-SSBA (n=37, 88.1%) and human risk group 2 (n=23, 54.8%) (Table 1). Bacteria were the most commonly implicated agent (n=17, 40.5%), while parasites and prions were the least frequently implicated (n=1, 2.3% each). Neisseria meningitidis was the most common RG2 agent (n=6, 14.3%), followed by lentiviral vectors (n=3, 7.1%). Blastomyces (Ajellomyces) dermatitidis was the most common RG3 agent (n=7, 16.7%), followed by SARS-CoV-2 (n=4, 9.5%) (data not shown).
| Biological agent type by risk group | Non-SSBA | SSBA | Total | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| RG2 | 23 | 55 | 0 | 0 | 23 | 55 |
| Bacteria | 12 | 29 | 0 | 0 | 12 | 29 |
| Fungus | 0 | 0 | 0 | 0 | 0 | 0 |
| Parasite | 1 | 2 | 0 | 0 | 1 | 2 |
| Prion | 1 | 2 | 0 | 0 | 1 | 2 |
| Toxin | 3 | 7 | 0 | 0 | 3 | 7 |
| Virus | 6 | 14 | 0 | 0 | 6 | 14 |
| Unknown | 0 | 0 | 0 | 0 | 0 | 0 |
| RG3 | 14 | 33 | 4 | 10 | 18 | 43 |
| Bacteria | 2 | 5 | 3 | 7 | 5 | 12 |
| Fungus | 7 | 17 | 1 | 2 | 8 | 19 |
| Parasite | 0 | 0 | 0 | 0 | 0 | 0 |
| Prion | 0 | 0 | 0 | 0 | 0 | 0 |
| Toxin | 0 | 0 | 0 | 0 | 0 | 0 |
| Virus | 5 | 12 | 0 | 0 | 5 | 12 |
| Unknown | 0 | 0 | 0 | 0 | 0 | 0 |
| Unknown | 0 | 0 | 0 | 0 | 1 | 2 |
| Bacteria | 0 | 0 | 0 | 0 | 0 | 0 |
| Fungus | 0 | 0 | 0 | 0 | 0 | 0 |
| Parasite | 0 | 0 | 0 | 0 | 0 | 0 |
| Prion | 0 | 0 | 0 | 0 | 0 | 0 |
| Toxin | 0 | 0 | 0 | 0 | 0 | 0 |
| Virus | 0 | 0 | 0 | 0 | 0 | 0 |
| Unknown | 0 | 0 | 0 | 0 | 1 | 2 |
| Total | 37 | 88 | 4 | 10 | 42 | 100 |
Occurrence types
The 42 exposure reports cited 58 incident occurrence types. Procedural (n=16, 27.1%) and sharps-related incidents (n=13, 22.0%) were the most common (Figure 4). Definitions are given in Appendix B1.
Figure 4: Reported occurrence types involved in reported exposure incidents, Canada 2020 (N=58)
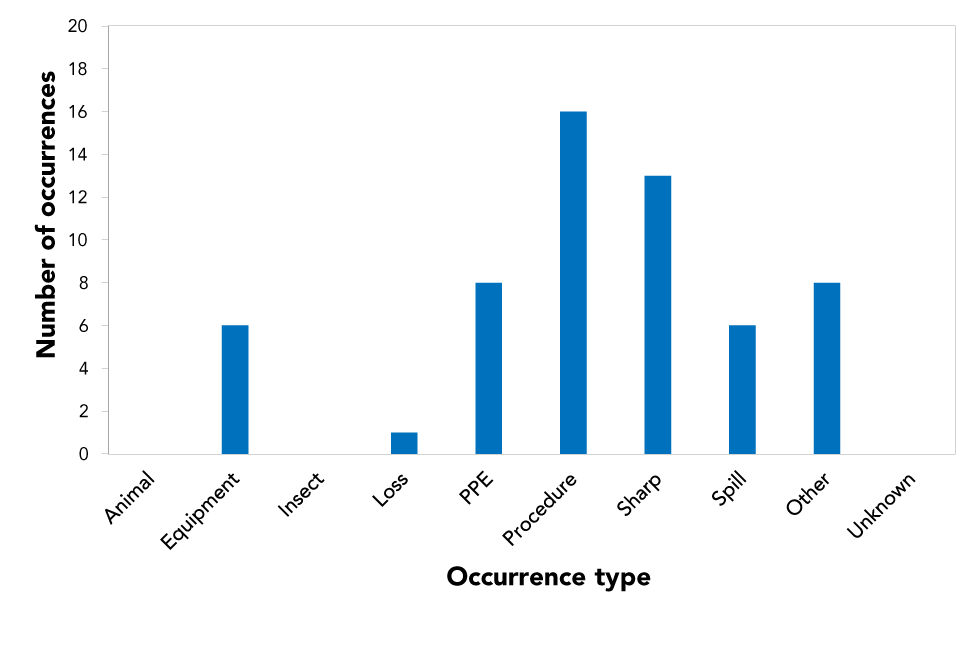
Text description: Figure 4
| Occurrence type | Number of occurrences |
|---|---|
| Animal | 0 |
| Equipment | 6 |
| Insect | 0 |
| Loss | 1 |
| PPE | 8 |
| Procedure | 16 |
| Sharp | 13 |
| Spill | 6 |
| Other | 8 |
| Unknown | 0 |
Exposed individuals
In total, 57 individuals were exposed through the 42 confirmed exposure reports. Most exposed individuals had a technical or trades college diploma as their highest level of education (n=24, 42.1%), followed by a Bachelor's degree (n=12, 21.1%) or a Master's degree (n=11, 19.3%). Other highest levels include high school (n=2, 3.5%), a MD/PhD (n=1, 1.8%) and a postdoctoral fellow (n=1, 1.8%). The remaining six individuals had other (n=3, 5.3%) or unknown (n=3, 5.3%) highest level of education (data not shown).
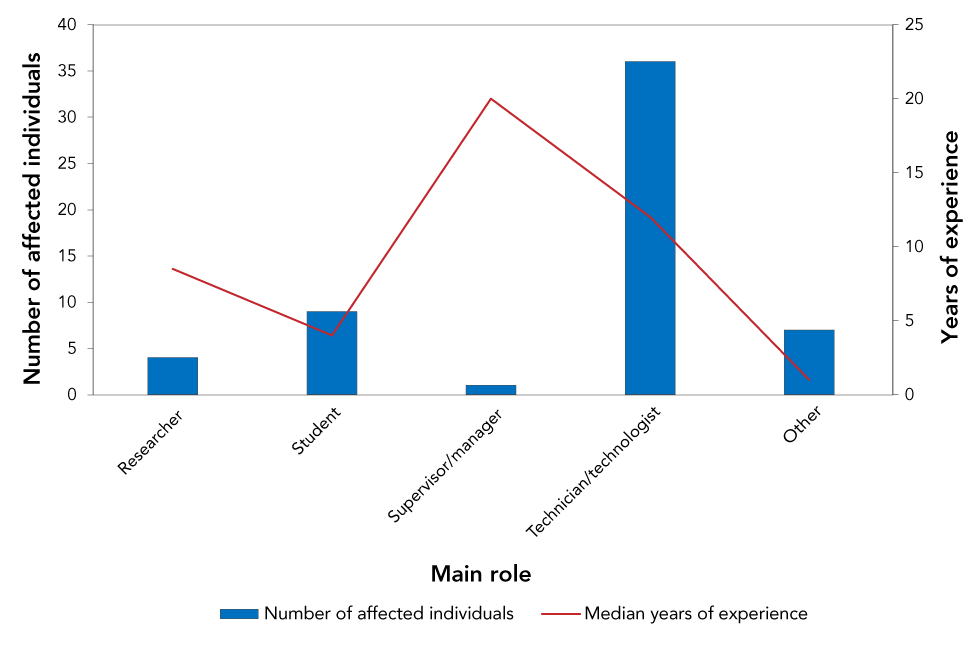
Consequently, most of the exposed individuals worked as technicians or technologists (n=36, 63.2%), students (n=9, 15.8%) and researchers (n=4, 7.0%). One exposed person was a supervisor or manager (1.8%), and the rest had other roles (n=7, 12.3%) (Figure 5).
Figure 5: Individuals affected in exposure incidents reported by number of years of laboratory experience and main roleFigure 5 Footnote a, Canada 2020 (N=57)
Text description: Figure 5
| Main role | Median years of experience | Number of affected individuals |
|---|---|---|
| Researcher | 8.5 | 4 |
| Student | 4 | 9 |
| Supervisor/manager | 20 | 1 |
| Technician/technologist | 12 | 36 |
| Other | 1 | 7 |
Among the 57 exposed individuals (not shown), most were exposed through inhalation (n=32, 56.1%) or sharps (n=9, 15.8%). Other routes of exposure include absorption (n=3, 5.3%) and ingestion (n=2, 3.5%). The rest were other (n=11, 19.3%) routes of exposure (data not shown).
Root causes and areas for laboratory safety improvement
In total, there were 89 root causes identified in the 42 exposure reports (Table 2). Issues with standard operating procedures (SOP) was the most common root cause (n=24, 27.0%), followed by human interactions (n=21, 23.6%) and equipment issues (n=12, 13.5%).
| Root cause | Examples of areas of concern | Citations | |
|---|---|---|---|
| n | %Table 2 Footnote a | ||
| Communication | Communication did not occur but should have | 8 | 9 |
| Communication was unclear, ambiguous, etc. | |||
| Equipment | Equipment quality control needed improvement | 12 | 13 |
| Equipment failed | |||
| Equipment was not appropriate for purpose | |||
| Human interaction | A violation (cutting a corner, not follow correct procedure, deviating from standard operating procedure) | 21 | 24 |
| An error (a mistake, lapse of concentration, or slip of any kind) | |||
| Management and oversight | Supervision needed improvement | 10 | 11 |
| Lack of auditing of standards, policies and procedures | |||
| Risk assessment needed improvement | |||
| Training | Training not in place but should have been in place | 9 | 10 |
| Training not appropriate for task/activity | |||
| Staff were not qualified or proficient in performing task | |||
| Standard operating procedure | Documents were followed as written but not correct for activity/task | 24 | 27 |
| Procedures that should have been in place were not in place | |||
| Documents were not followed correctly | |||
| Other | Not applicable | 5 | 6 |
Time between the incident and the reporting date
Exposure incident reports are to be submitted to LINC without delay. In 2020, of the 41 incident exposure reports that included the incident date, 23 (56.1%) were submitted to LINC within one week of the incident. The median number of days between the incident and the reporting date was six days in 2020, up slightly from a median of four days in 2019 (Figure 6).
Figure 6: Time between the date of the incident and the date report was submitted to Laboratory Incident Notification Canada, Canada 2016–2020
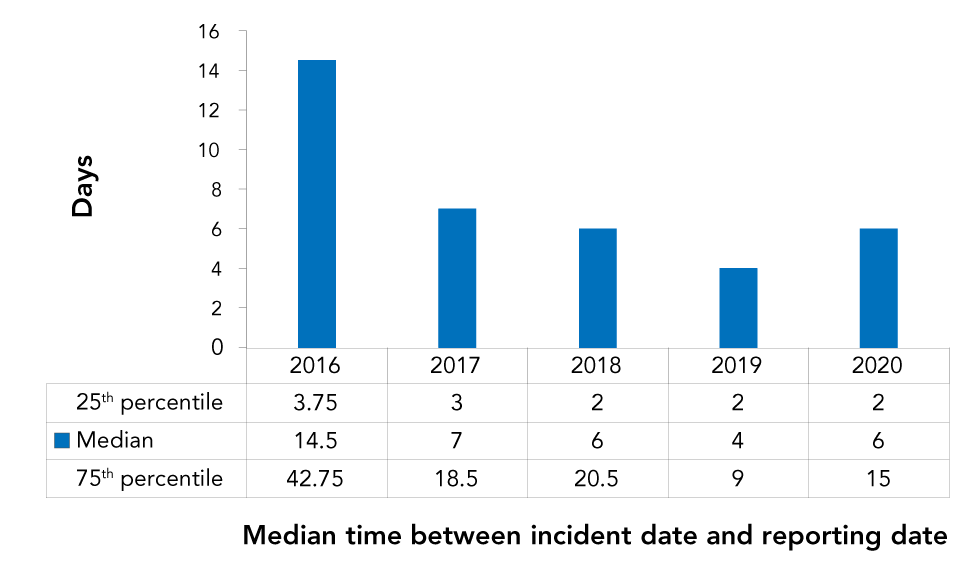
Text description: Figure 6
| Reporting | 2016 | 2017 | 2018 | 2019 | 2020 |
|---|---|---|---|---|---|
| MIN | 0 | 0 | 0 | 0 | 0 |
| 25th percentile | 3.75 | 3 | 2 | 2 | 2 |
| Median | 14.5 | 7 | 6 | 4 | 6 |
| 75th percentile | 42.75 | 18.5 | 20.5 | 9 | 15 |
| MAX | 456 | 137 | 380 | 37 | 76 |
Discussion
In 2020, 42 laboratory exposures to HPTs had been reported to LINC, a decrease from the 60 reported in 2019. The reports did not include any LAI and were submitted within a median delay of six days. Reports on RG2, non-SSBA agents as well as bacteria were the most common types of HPTs involved in exposure incidents. Neisseria meningitides and lentiviral vector exposures were more common among RG2 HPTs, whereas Blastomyces (Ajellomyces) dermatitidis and SARS-CoV-2 exposures were more common among RG3.
Similar to 2019, exposures were mainly due to procedure breaches and sharps, and occurred mostly in academic and hospital sectors while performing microbiology activities. In total, 57 individuals, predominantly technicians or technologists, were exposed to an HPT. Lack of awareness or compliance with standard operating procedures and human interactions were the main root causes identified.
Number of exposures and exposure incident rate have followed the same trend over the past five years
At the onset of the LINC program in 2016, the number of exposure incidents reported had increased, with a peak reached in 2018. The increase was concomitant to the rise of the number of licenses granted to laboratories over the same period. After 2018, despite the number of licenses remaining stable, the number of incidents started to decrease. The exposure incident rate followed a trend similar to the number of licenses, meaning that the increase from 2016 to 2018 and the decrease from 2018 to 2020 were not due to a change in the number of licenses granted to laboratories. The initial rise of the exposure incident rate from 2016 to 2018 was likely the result of the actions engaged by the LINC surveillance system to facilitate reporting and enhance clarity on regulatory requirementsFootnote 5. Regarding the decrease from 2018 to 2020, when an exposure incident occurs in a licensed laboratory, an incident response is actioned by the Centre for Biosecurity with the final goal of identifying root causes and encouraging corrective actions. This feedback may have raised the awareness of licensed parties and may be partially responsible for the decline in reports in recent years. Further information on incident reporting specifics can be found in the incident reporting guidelines published in 2017Footnote 11. In addition, stay at home orders and other pandemic responses likely led to a reduction in laboratory activities for a portion of 2020, possibly leading to fewer reports.
Exposure incidents involving SARS-CoV-2 reported to Laboratory Incident Notification Canada did not include exposure incidents occurring during diagnostic activities
The reporting of exposure incidents in a laboratory setting through activities involving HPTs in their natural environment is not mandatory under the HPTA. Pathogens and toxins are considered to be in their natural environment if they are collected directly from humans or animals (e.g. blood, serum, tissue, urine, feces, saliva, milk, etc.) or from the environment (e.g. water, soil). Consequently, exposure incidents occurring during diagnostic activities involving SARS-CoV-2 were not systematically reported to LINC and were not included in this report. Four of the 42 exposure incidents reported to LINC involved SARS-CoV-2. These incidents occurred during research activities and were therefore mandatory. Although such reporting was voluntary, laboratory workers are encouraged to report exposure incidents involving HPTs in the HPTs' natural environment. This reporting enables the collection of data at the national level that can be used to detect real-time trends and potential patterns of concern, and to facilitate early responses in order to prevent and/or mitigate biosafety risks.
Delay of notification of exposure incidents has improved over the past five years
According to the Notification and Reporting under the HPTA and HPTR Guidelines and the HPTA, notification reports of exposure incidents have to be submitted to LINC without delayFootnote 11. From 2016 to 2019, the median time of submission of exposure incidents decreased from two weeks to four days. Such a decrease maybe explained by the LINC surveillance system actions to facilitate reporting and inform laboratories regarding submission timeliness recommendations. However, in the past year (2020), the median time of report submission increased slightly, from four to six days. This change was possibly attributable to an increase of the workload of laboratories and to disruptions of work caused by the ongoing pandemic. A comparison of time of submission was not done internationally, since exposure reports in other countries were done on a voluntary basis or through surveysFootnote 8Footnote 12Footnote 13.
Strengths and limitations
The main strength of this study is the centralized and mandatory reporting process of laboratory incidents in laboratories across Canada. Further, the LINC allows for an almost real-time identification of causes of incidents and potential areas of improvement that could be addressed in conjunction with laboratories to ensure risks are mitigated in a timely manner. For example, the most exposed individuals were found to be technicians, due to lack of compliance to SOPs. This information could be used by licensed facilities to examine current protocols that are related to SOP compliance to reduce the risk of exposures of laboratory workers in the future. NewslettersFootnote 14 and e-blasts prepared by the LINC team discuss common safety issues and areas for improvement as they arise, which are shared with stakeholders. In addition, there is constant communication between the Centre for Biosecurity and regulated parties. Further follow-up with regulated parties are planned to communicate these results to ensure incidents involving SOP compliance are addressed and adhered to.
There are several limitations of this study. First, non-reporting is a possible confounder in this analysis. The magnitude and significance of non-reporting is currently unmeasured; however, we continually encourage license holders to report laboratory exposure incidents without delay. Second, the exclusion of reports with missing dates from the analysis of the "time to reporting" calculation is another limitation. Given that the proportion of missing values was lower than 10%, the estimation of the median time to reporting likely had only a minor impact. Another limitation is that the number of licenses was used as a proxy of the laboratory workforce for the calculation of the exposure incidence rateFootnote 5. Further, the number of active licenses from December 2020 was unavailable due to the effects of the pandemic. Instead, the number of active licenses for April 2020 was used, as the number of licenses usually fluctuates minimally throughout the year. We will continue to address these limitations through constant communication with stakeholders, by ways of newsletters and e-blasts and biosafety advisories.
Conclusion
The rate of laboratory exposure incidents was lower in 2020 than 2019. The ongoing pandemic may have contributed to this decrease because of the closure of laboratories (and other non-essential workplaces) for a portion of the year. The most common occurrence type was procedural, while issues with non-compliance with SOP and human interactions were the most cited root causes.
Authors' statement
NA — Methodology, investigation, writing: original draft, review and editing
MS — Methodology, investigation, writing: original draft, review and editing
RE — Conceptualization, methodology, investigation, writing: original draft, review and editing, supervision
ET — Writing–original draft, review and editing
MEJ — Writing–original draft, review and editing
MH — Writing–review and editing
Competing interests
None.
Acknowledgements
We would like to express our gratitude to our regulated parties for their continued support and contribution regarding incident reporting across Canada. We would also like to say a special thanks to the staff of the Centre of Biosecurity for their continued input, support and expertise.
Funding
None.
Appendices
| Main activity | Definition |
|---|---|
| Animal care | Activities such as attending to the daily care of animals and providing animals with treatment |
| Autopsy or necropsy | Post-mortem surgical examinations for purposes such as determining cause of death or to evaluate disease or injury for research or educational purposes |
| Cell culture | The process of growing cells under controlled conditions; it can also involve the removal of cells from an animal or plant |
| Education or training | Education or training of students and/or personnel on laboratory techniques and procedures |
| In vivo animal research | Experimentation with live, non-human animals |
| Maintenance | The upkeep, repair, and/or routine and general cleaning of equipment and facilities |
| Microbiology | Activities involving the manipulation, isolation, or analysis of microorganisms in their viable or infectious state |
| Molecular investigations | Activities involving the manipulation of genetic material from microorganisms or other infectious material for further analysis |
| Serology | Diagnostic examination and/or scientific study of immunological reactions and properties of blood serum |
| Hematology | Scientific study of the physiology of blood |
| Occurrence type | Definition |
|---|---|
| Spill | Any unintended release of an agent from its container |
| Loss of containment | Includes malfunction or misuse of containment devices or equipment and other type of failures that results in the agent being spilled outside of, or released from containment |
| Sharps-related | Needle stick, cut with scalpel, blade or other sharps injury (i.e. broken glass) |
| Animal-related | Includes animal bites or scratches, as well as other exposure incidents resulting from animal behavior (i.e. animal movement resulting in a needle stick) |
| Insect-related | Includes insect bites |
| PPE-related | Includes either inadequate PPE for the activity or failure of the PPE in some way |
| Equipment-related | Includes failure of equipment, incorrect equipment for the activity, or misuse of equipment |
| Procedure-related | Includes instances when written procedures were not followed, were inadequate or absent, or were incorrect for the activity |
