Handling complaints about medical devices (GUI-0065): About complaints
On this page
- Introduction
- What is a complaint
- Keys elements to handling complaints effectively
- What the process looks like
- Your responsibilities
- Written procedures
Introduction
The Medical Devices Regulations (regulations) require you to keep records of reported problems related to the performance or safety of your medical device. They also direct you to:
- document how you address those problems
- develop written procedures for investigating complaints
- ensure your procedures enable you to conduct an effective and timely investigation
What is a complaint
A complaint alleges or implies that a medical device has deficiencies concerning its identity, quality, durability, reliability, safety, effectiveness or performance. It is made after a device is released for distribution or sale.
A complaint may be written or verbal and may be submitted by fax, mail, email, telephone, in person or on a website. It may come from many different sources, including:
- end-users
- such as patients, consumers and health care professionals
- reports from sales or service representatives, scientific literature, legal documents or internal analysis
- importers, distributors and retailers
- medical associations
- regulatory agencies
- such as Health Canada
- service requests and warranty claims
Complaints may lead to repairs, servicing or changes in a manufacturer's instructions for use, or to a recall. They include reports of incidents or problems with the medical device, its label or its packaging.
Note: Complaints can stem from someone returning a product or asking for servicing under warranty, and at first may not be identified as a complaint. As a result, you should be clear when deciding what is and is not a complaint.
Key elements to handling complaints effectively
To address complaints effectively, you should:
- have written procedures for documenting the complaint handling process
- have trained personnel who carry out the procedures and have the background, education, training and experience to perform their roles
- collect and screen complaints
- evaluate complaints to determine whether further investigation or/and an incident report is needed
- Identify the criteria for risk to health and a risk based timetable to investigate the problem
- collaborate and promptly share information about the safety and performance of the device within your company and to affected parties outside the company
- may include manufacturer, importer, distributor, users and regulatory authority
- keep records to document the complaint and all actions taken in response to it or the reason for not taking action
- report to all affected parties outside the company when necessary
- trend reports for regular review
What the process looks like
The key elements of an effective system for handling complaints are part of a 5-stage process. Each stage has a sequence of steps and decisions that are to be carried out which may overlap depending on the situation.
Figure 1 shows the 5 stages.
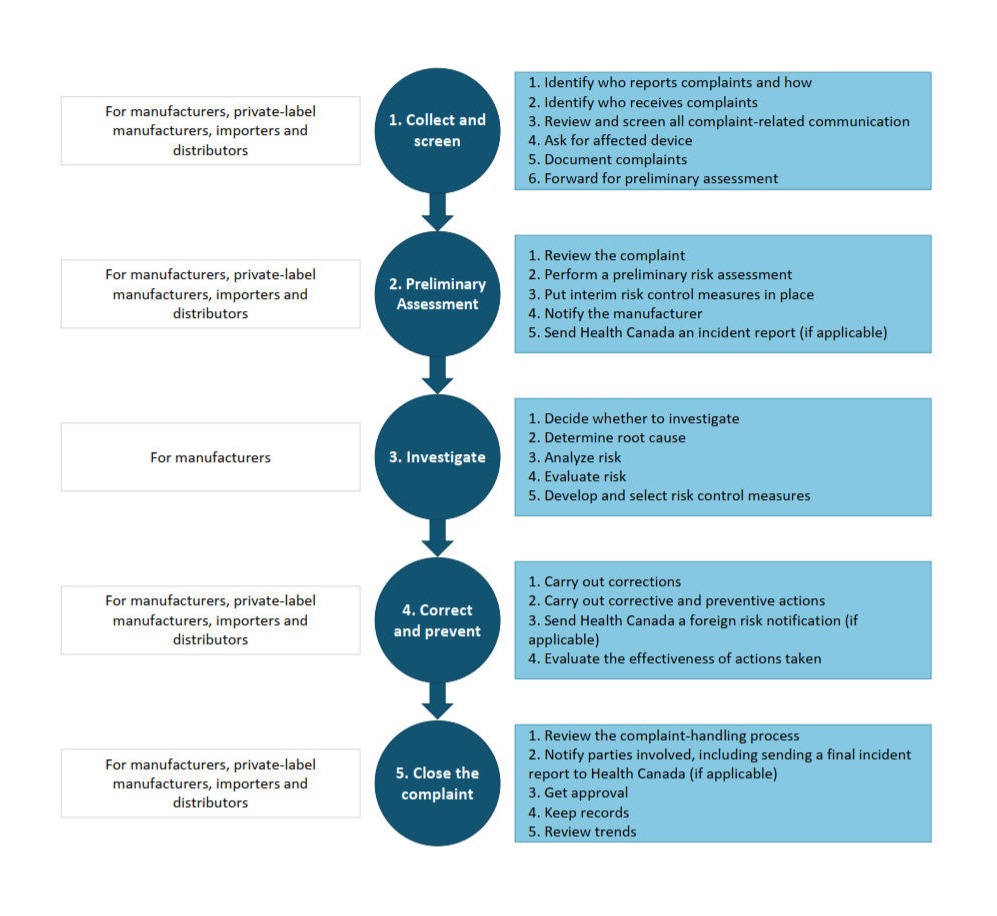
Figure 1: Text description
These are the steps to a 5-stage process for handling complaints.
Stage 1 is titled ''Collect and Screen.'' It is for manufacturers, private-label manufacturers, importers and distributors. This stage has 6 steps. Step 1 is identifying who reports the complaints and how they are reported. Step 2 is identifying who receives complaints at the company. Step 3 is reviewing and screening all complaint-related communications. Step 4 is about asking for the affected device involved in the complaint. Step 5 pertains to documenting complaints. Step 6 is forwarding the complaint for a preliminary assessment.
Stage 2 is titled ''Preliminary Assessment.'' It is for manufacturers, private-label manufacturers, importers and distributors. This stage is organized into 5 steps. Step 1 is reviewing the complaint. Step 2 is performing a preliminary risk assessment. Step 3 is putting interim risk control measures in place to reduce the risk(s). Step 4 is notifying the manufacturer about the complaint. Step 5 is sending Health Canada an incident report, if applicable.
Stage 3 is titled ''Investigate.'' It is for manufacturers. Advancing through this stage involves 4 steps. Step 1 is deciding whether to investigate the complaint. Step 2 is determining the root cause. Step 3 is analyzing the risk posed by the problem followed by Step 4 which is evaluating the risk. Step 5 is about developing and selecting risk control measures.
Stage 4 is titled ''Correct and prevent.'' It is for manufacturers, private-label manufacturers, importers and distributors. This stage consists of 4 steps. Step 1 is carrying out corrections promptly. Step 2 is carrying out corrective and preventive actions. Step 3 is sending Health Canada a foreign risk notification, if applicable. Step 4 is evaluating the effectiveness of actions taken.
Stage 5 is titled ''Close the complaint.'' It is for manufacturers, private-label manufacturers, importers and distributors. This stage breaks down into 5 steps. Step 1 is reviewing the complaint-handling process to ensure all steps and actions are completed. Step 2 is notifying parties involved, including sending a final incident report to Health Canada, if applicable. Step 3 is getting approval for the closure of the complaint file. Step 4 is about keeping complete records. Step 5 is reviewing complaints on a regular basis to detect trends in failure.
Your responsibilities
Whether you are a manufacturer, private-label manufacturer, importer or distributor, you have a role to play in handling complaints. You must:
- understand your role and responsibilities
- define your relationship with the other companies in the medical device distribution chain
- document your role, responsibilities and relationship with other companies in your procedures
Table 1 lists the responsibilities in handling complaints.
| Responsibilities | |||
|---|---|---|---|
| Manufacturer | Private-label manufacturer and importer | Distributor | |
| Collect, document and verify complaint | Yes | Yes | Yes |
| Carry out preliminary risk assessment and put interim risk control measures in place | Yes | Yes (at a minimum for activities for which you are responsible) | Yes (at a minimum for activities for which you are responsible) |
| Notify the manufacturer | Not applicable unless another company is delegated to investigate complaints | Yes (notify the supplier if different from the manufacturer) | Yes (notify the supplier if different from the manufacturer) |
| Send Health Canada a preliminary incident report | Yes (if needed) | Yes (if needed) | Not applicable |
| Conduct root cause investigation and risk assessment | Yes | Not applicable (unless the cause originates from your company) | Not applicable (unless the cause originates from your company) |
| Develop and select risk control measures (for example: corrections, corrective and preventive actions) | Yes | Not applicable (unless the cause originates from your company) | Not applicable (unless the cause originates from your company) |
| Carry out risk control measures | Yes | Yes | Yes |
| Send Health Canada a foreign risk notification | Yes – Class II to IV (if needed) | Yes – Class II to IV (if needed) | Not applicable |
| Evaluate the effectiveness of risk control measures | Yes | Not applicable (unless the cause originates from your company) | Not applicable (unless the cause originates from your company) |
| Provide feedback on risk control measures to the manufacturer | Not applicable | Yes | Yes |
| Send Health Canada a final incident report | Yes (if needed) | Yes (if needed) | Not applicable |
| Communicate with complainant and, if necessary, the person reporting the complaint (end-user, private-label manufacturer, importer or distributor) | Yes | Yes | Yes |
| Conduct final review and file closure | Yes | Yes | Yes |
| Maintain complaint handling records | Yes | Yes | Yes |
| Conduct periodic trend analysis of complaints | Yes | Yes | Yes |
All parties in the distribution chain must work together and share information on a complaint quickly (from the first report, throughout the investigation, to file closure). |
|||
If you are the manufacturer
You are responsible for your medical device on the Canadian market. This means being responsible for:
- the safety and effectiveness of the device
- any other risks related to how the device is handled, stored and used when instructions for use are followed
- the identification of possible misuse and how to protect against the risk of misuse
Note: You are considered a manufacturer if your company name or trademark appears on the medical device label. If more than one company name appears on the label, the relationship of each company name to the device must be made clear. For a Class II, III or IV medical device, the manufacturer's name will also be on the device licence.
You must be ready to address complaints made to you from both:
- end-users
- such as patients, consumers, health care professionals and facilities
- other sources
- such as importers, distributors, private-label manufacturers and regulatory agencies
To do so, you should collect complaints through a single point of contact such as a role, function or group within each division or location.
Once you become aware of a potential risk, you must take the lead in investigating and resolving the complaint. Steps may include root cause analysis, risk assessment and corrective and preventive actions.
Your process, when applicable, must also include steps to evaluate your complaint against incident reporting criteria; actions taken to address the risk, and assessment against foreign risk notification and recall criteria to ensure you meet requirements for other reportable actions to Health Canada.
For more information, consult:
- Incident reporting for medical devices: Guidance document
- Foreign Risk Notification for medical devices: Guidance document
- Guide for recalling medical devices (GUI-0054)
You may also want to inform the person who made the complaint, of the risk control measures you plan to carry out. This communication is a key part of the feedback loop that enables you to manage risk posed by a device.
If you are the private-label manufacturer, importer or distributor
You are a vital link between those who use a medical device and the manufacturer who makes it. The manufacturer and the end-user depend on you to collect, evaluate and share complaints about the safety and performance of a device with the manufacturer without delay. To do this, you should have:
- a method for collecting complaints made to you such as a complaint log or file
- a process for forwarding complaints to the manufacturer for investigation
Note: If you do not have a direct link to the manufacturer, you may send complaints to your immediate supplier. Your supplier should then forward them to the manufacturer or their supplier.
As a private-label manufacturer or importer, you should also assess the criteria for incident reporting to determine whether the incident is reportable to Health Canada.
For more information, consult:
Once the manufacturer tells you that a device poses a potentially serious risk, you must take actions within your control. This means implementing the manufacturer's risk control measures. These measures may include, for example:
- amending the label
- returning the device
- stopping the sale of the device
- providing more instructions for use
- advising end-users to stop using the device
- improving storage and/or transportation controls
- repairing the device at the end-user's facility
You may also evaluate the manufacturer's risk control measures to assess their effectiveness and report the results back to the manufacturer.
If someone (like distributor or end-user) reported the complaint to you, you should communicate the efforts taken to address the problem with them).
Note: Risk control measures may meet the definition of a recall. If you are a private-label manufacturer or importer, you must notify Health Canada if you recall a device that is likely to cause injury to the health of a patient, user or other person, or could cause serious injury to the health of a patient, user or other person. Consult the following guidance document for the information you must report to Health Canada:
Written procedures
You must document the complaint-handling process in written procedures. The procedures can be in an electronic or paper-based format or a combination of both. You may document the process in one procedure or several. You must be able to show Health Canada that:
- you have documented the entire process
- you are following the procedure or procedures
The procedure(s) should:
- use a consistent structure for organizing and presenting the information
- define what is a complaint and the key activities involved in the process of addressing it
- assign responsibility for the key activities in the procedure(s) to those who have the knowledge and authority to ensure they are carried out effectively and in a timely manner
- describe in detail the steps involved in handling a complaint, from receipt to closure of the file
- specify measurable time frames for completing the steps based on the risk level (the higher the risk, the shorter the time frame)
- include a mechanism to ensure time frames are met, from receipt to closure of the file
- require that actions taken to address the complaint be documented
- list the records or forms associated with the complaint handling process as well as where and how these records are maintained
Consult the following guidelines for help with developing your procedure(s):
Guide for recalling medical devices (GUI-0054): Defining a recall, documented processes
You must follow your procedure(s) as soon as you receive a complaint. This involves:
- performing the needed tasks or activities
- documenting or updating records as needed
- ensuring each employee responsible for any procedural task or activity:
- has access to the procedure
- has had the appropriate training
- understands their responsibility and
- providing management oversight to assure the procedures are followed
If you rely on a third party to handle complaints, then a written quality agreement or contract between you and the third party should be available. This agreement should specify the roles and responsibilities of each party and assures compliance with the Medical Devices Regulations (for example, timelines for reporting of serious incidents). The quality agreement is signed and dated by all responsible parties involved.