Archived - Health Product InfoWatch – July 2018

Download the alternative format
(PDF format, 424 KB, 6 pages)
Health Products and Food Branch
Marketed Health Products Directorate
Health Product InfoWatch Editorial Team
ISSN 2368-8025
Pub. no 170363
Contents
- Health products mentioned in this issue
- Monthly recap
- New information
- Scope
- Helpful links
- Suggestions?
- Copyright
Health products mentioned in this issue
Pharmaceuticals and Biologics
Blincyto (blinatumomab)
Clozapine
Demulen 30
Dolutegravir-containing medicines (Tivicay, Triumeq and Juluca)
Imatinib mesylate (Gleevec and generics)
Imbruvica (ibrutinib)
Sunscreens
Medical Devices
Fentanyl-detection test strips
Natural Health Products
Jamp-Glucose 50 and Jamp-Glucose 75
Other
Monthly recap of health product safety information
The following is a list of health product advisories, type I recalls as well as summaries of completed safety reviews published in June 2018 by Health Canada.
Blincyto (blinatumomab)
Blincyto has recently been authorized with an additional option of preparing a 7-day infusion bag containing benzyl alcohol for patients weighing greater than or equal to 22 kg. It is not recommended for use in patients weighing less than 22 kg. Serious and fatal adverse reactions, including “gasping syndrome”, can occur in pediatric patients, particularly in neonates and infants treated with Blincyto containing benzyl alcohol as a preservative. When preparing bags of Blincyto solution for infusion in neonates, infants and patients weighing less than 22 kg, healthcare professionals are advised to only utilize preservative-free saline. The Canadian product monograph has been updated to reflect this new safety information.
Health Professional Risk Communication – Blincyto (blinatumomab)
Clozapine
This updated summary safety review, originally published in March 2018, evaluated the risk of agranulocytosis associated with clozapine. Health Canada's review concluded that the monitoring measures in place for agranulocytosis are acceptable, but that the risk should still be monitored. Health Canada reminded healthcare professionals to follow the recommendations regarding switching of a patient from one brand of clozapine to another as highlighted in the current Canadian product monograph and in a previously issued risk communication.
Summary Safety Review – Clozapine
Demulen 30
Pfizer Canada Inc. received complaints of broken or chipped pills involving Demulen 30. Health Canada reminded women to check their packages of oral contraceptives and to report problems when noticed.
Advisory – Demulen 30 birth control pills
Dolutegravir-containing medicines (Tivicay, Triumeq and Juluca)
Serious cases of neural tube defects in infants born to women with exposure to dolutegravir at the time of conception were identified in an ongoing observational study in Botswana. In the same study, no infant born to a woman who started dolutegravir during pregnancy had neural tube defects. Healthcare professionals are advised to avoid prescribing dolutegravir in women of childbearing potential who are trying to become pregnant unless a suitable alternative treatment option is not available. The risks and benefits of dolutegravir treatment should be considered when prescribing it to women of childbearing potential.
Health Professional Risk Communication – Dolutegravir-containing medicines (Tivicay, Triumeq and Juluca)
Information Update – Dolutegravir-containing medicines (Tivicay, Triumeq and Juluca)
Fentanyl-detection test strips
Health Canada reminded Canadians of the potential limitations of fentanyl test strips when used to detect fentanyl or other deadly substances in street drugs before consuming them. No fentanyl test strips are specifically designed to check street drugs before consumption. A preliminary study of a fentanyl test strip product by the Department indicated that false negatives could occur. Health Canada has undertaken additional assessments of the same fentanyl test strip and found that while the product detected fentanyl every time, it was not reliable for detecting some fentanyl analogs, including carfentanil. Health Canada is working with companies to include warnings on the packages of these products, to better inform consumers about the risks of unreliable results when using the fentanyl test strips to check street drugs they plan to consume.
Information Update – Fentanyl-detection test strips
Imatinib mesylate (Gleevec and generics)
This safety review evaluated the risk of tendinopathy associated with the use of imatinib mesylate (Gleevec and generics). Health Canada's review of the available information did not find a link. Health Canada encourages the reporting of any side effects related to the use of this health product.
Summary Safety Review – Imatinib mesylate (Gleevec and generics)
Jamp-Glucose 50 and Jamp-Glucose 75
Six lots of Jamp-Glucose 50 and Jamp-Glucose 75 were recalled because they contained less glucose than was shown on their labels, which could lead to false negative results. Healthcare professionals are advised to immediately stop using the affected lots and return them to the wholesalers. As well, they should contact the patients who had negative oral glucose challenge or tolerance test results involving the affected lots, and consider repeat testing, or use an alternate diagnostic test for diabetes as appropriate.
Health Professional Risk Communication – Jamp-Glucose 50 and Jamp-Glucose 75
Sunscreens
Health Canada received a higher than expected number of reports of skin reactions suspected of being associated with Banana Boat sunscreen products last summer. As a result of these reports, Health Canada tested a wide range of sunscreen brands and released a summary of the results. Health Canada’s testing did not identify any serious concerns with the quality of these products. Although reactions observed are not the result of product quality issues, other factors that could have contributed to the reported reactions are under review. Health Canada is completing a comprehensive scientific and clinical safety review of sunscreens and their risk of causing skin reactions. A summary of the safety review will be released when available. Health Canada also reminded Canadians to protect themselves and their families from the sun and provided sunscreen safety tips.
Information Update – Sunscreens (testing results)
Information Update – Sunscreens (safety tips)
Unauthorized health products
Health Canada advised Canadians about various unauthorized health products being sold at retail locations across Canada or online that may pose serious health risks.
Advisory – Unauthorized health products
New health product safety information
The following topics have been selected to raise awareness and, in some cases, to stimulate reporting of similar adverse reactions.
Product Monograph Update
The following safety labelling updates, which were recently made to the Canadian product monograph, have been selected for your awareness. A complete list of safety labelling updates is available on Health Canada's Product Monograph Brand Safety Updates page. Canadian product monographs can be accessed through Health Canada's Drug Product Database.
Imbruvica (ibrutinib)
The risk of ventricular tachyarrhythmia has been included in the Warnings and Precautions and Post-Market Adverse Drug Reactions sections of the Canadian product monograph for Imbruvica.
Key messages for healthcare professionals: Footnote 1
- Events of ventricular tachyarrhythmia, including some with a fatal outcome, have been reported in patients treated with Imbruvica, particularly in patients with cardiac risk factors, hypertension, acute infections, and a previous history of cardiac arrhythmia.
- Healthcare professionals should periodically monitor for cardiac arrhythmia in all patients treated with Imbruvica. Patients who develop arrhythmic symptoms such as palpitations, light-headedness, syncope, chest discomfort or new onset of dyspnea, should be evaluated clinically and an electrocardiogram should be performed if indicated.
- Cardiac arrhythmias should be managed appropriately. If they persist, healthcare professionals should consider the benefits and risks of Imbruvica treatment and follow the dose modification guidelines indicated in the product monograph.
Reference
- Footnote 1
-
Imbruvica (ibrutinib) [product monograph]. Toronto (ON): Janssen Inc. 2018
Vaccine safety biannual summary
Health Canada and the Public Health Agency of Canada (PHAC) share the responsibility of monitoring of the safety of vaccines in Canada.
Market authorization holders are required to report serious adverse events following immunization to the Canada Vigilance Program in Health Canada. The Canada Vigilance Program also receives voluntary reports from healthcare professionals and consumers.
Provincial and territorial public health authorities report adverse events following immunization (AEFIs) from publicly-funded vaccine programs to the Canadian Adverse Events Following Immunization Surveillance System (CAEFISS) in PHAC to monitor the safety of immunization programs.
Report for July 1, 2017 to December 31, 2017
Key Messages:
- No new safety signals (potential safety issues) were identified during this period.
- From July 1, 2017 to December 31, 2017, the Canada Vigilance Program received 270 reportsFootnote * of adverse events following immunization for which vaccines were a suspected cause.
This biannual vaccine safety summary includes adverse events following immunization reports received by the Canada Vigilance Program between July 1, 2017 and December 31, 2017. To access reports published by the Canadian Adverse Events Following Immunization Surveillance System (CAEFISS), please visit the CAEFISS Web site.
- From July 1, 2017 to December 31, 2017, the Canada Vigilance Program received 270 reportsFootnote * of adverse events following immunization for which vaccines were a suspected cause.

Figure 1 - Text Equivalent
The figure shows the total number of adverse events following immunization reports received from July 1, 2017 to December 31, 2017, by type of originating reporter.
| Originating reporter | Percentage (%) |
| Physicians | 24.44 |
| Pharmacists | 30.37 |
| Other health professional | 19.63 |
| Consumer or other non health professional | 25.19 |
| Unknown | 0.37 |
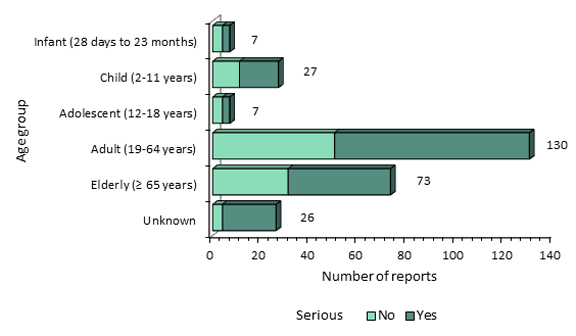
Figure 2 - Text Equivalent
The figure shows the total number of adverse events following immunization reports received from July 1, 2017 to December 31, 2017, by age group and severity:
| Age | Serious | Number of reports |
| Infant (28 days to 23 months) | No | 4 |
| Infant (28 days to 23 months) | Yes | 3 |
| Child (2-11 years) | No | 11 |
| Child (2-11 years) | Yes | 16 |
| Adolescent (12-18 years) | No | 4 |
| Adolescent (12-18 years) | Yes | 3 |
| Adult (19-64 years) | No | 50 |
| Adult (19-64 years) | Yes | 80 |
| Elderly (≥ 65 years) | No | 31 |
| Elderly (≥ 65 years) | Yes | 42 |
| Unknown | No | 4 |
| Unknown | Yes | 22 |
- There were 166 (61%) serious reports, all of which were individually assessed. Most of these involved patients with underlying medical conditions and were unlikely related to the vaccination.
- The highest number of reports (serious and non-serious) involved influenza vaccines (46%), followed by herpes zoster (19%) and pneumococcal vaccines (12%). The majority of reports for influenza vaccines occurred between October 1, 2017 and December 31, 2017, which is expected during the Influenza Immunization Awareness Campaign in Canada.
- There were 6 reports with an outcome of death. These reports involved patients between 19 and 70 years of age: 2 females, 3 males and 1 with unknown gender. One report was for Zostavax from a healthcare professional and 5 were for influenza vaccines from social media extracted by the company. The information provided was not sufficient to adequately assess the causal association with the vaccines.
- The most frequently reported adverse events (serious and non-serious) included pyrexia, malaise, injection site erythema, nausea, and pain. These are common adverse events following vaccination and are captured in the Canadian product monographs.
- No new safety signals (potential safety issues) were identified during this period.
- Health Canada, in collaboration with the Public Health Agency of Canada, will continue to closely monitor the safety of vaccines authorized in Canada and will take appropriate action if any new health risks are identified.
For additional information, contact the Marketed Health Products Directorate.
Note that because of updated information received by the Canada Vigilance Program, there may be differences in the number of reports and adverse events retrieved at different dates.
Footnotes
Scope
This monthly publication is intended primarily for healthcare professionals and includes information on pharmaceuticals, biologics, medical devices and natural health products. It provides a summary of key health product safety information published in the previous month by Health Canada, as well as a selection of new health product safety information meant to raise awareness. New information contained in this issue is not comprehensive but rather represents a selection of clinically relevant items warranting enhanced dissemination.
Reporting Adverse Reactions
Canada Vigilance Program
Telephone: 1-866-234-2345
Fax or mail: Form available on MedEffect Canada
For more information on how to report an adverse reaction, visit the Adverse Reaction and Medical Device Problem Reporting page.
Helpful links
- MedEffectTM Canada
- Recalls and Safety Alerts Database
- Summary Safety Reviews
- New Safety Reviews
- Canada Vigilance Adverse Reaction Online Database
- Drug Product Database
- Medical Devices Active Licence Listing
- Licensed Natural Health Products Database
- The Drug and Health Product Register
- Drug Shortages Canada
Suggestions?
Your comments are important to us. Let us know what you think by reaching us at HC.infowatch-infovigilance.SC@canada.ca
Health Canada
Marketed Health Products Directorate
Address Locator 1906C
Ottawa ON K1A 0K9
Telephone: 613-954-6522
Fax: 613-952-7738
Copyright
© 2018 Her Majesty the Queen in Right of Canada. This publication may be reproduced without permission provided the source is fully acknowledged. The use of this publication for advertising purposes is prohibited. Health Canada does not assume liability for the accuracy or authenticity of the information submitted in case reports.
Adverse reactions (ARs) to health products are considered to be suspicions, as a definite causal association often cannot be determined. Spontaneous reports of ARs cannot be used to estimate the incidence of ARs because ARs remain underreported and patient exposure is unknown.
Due to time constraints relating to the production of this publication, information published may not reflect the most current information.