Archived - Benzathine penicillin G and early syphilis

 Download this article as a PDF (373 KB - 7 pages)
Download this article as a PDF (373 KB - 7 pages) Published by: The Public Health Agency of Canada
Issue: Volume 42-2: Update on STIs
Date published: February 4, 2016
ISSN: 1481-8531
Submit a manuscript
About CCDR
Browse
Volume 42-2, February 4, 2016: Update on STIs
Systematic review
Benzathine penicillin G for the management of early syphilis among HIV co-infected persons: A systematic review
Niragira O1, Ha S1, Pogany L1*, Singh A2
Affiliations
1 Centre for Communicable Diseases and Infection Control, Infectious Disease Prevention and Control Branch, Public Health Agency of Canada, Ottawa, ON
2 University of Alberta, Department of Medicine, Edmonton, AB
Correspondence
Suggested citation
Niragira O, Ha S, Pogany L, Singh A. Benzathine penicillin G for the management of early syphilis among HIV co-infected persons: A systematic review Can Comm Dis Rep 2016;42-2:30-36. https://doi.org/10.14745/ccdr.v42i02a02
Abstract
Background: The optimal treatment for syphilis in people who are human immunodeficiency virus (HIV) positive is controversial.
Objective: To assess the efficacy of three doses versus a single dose of long acting Benzathine Penicillin G (BP-G) for the effective management of early syphilis among HIV co-infected populations.
Methods: A systematic search of the published literature was conducted using MEDLINE and EMBASE databases to identify clinical and observational studies published between January 2010 and May 2015. Inclusion criteria were: publication in English or French, populations co-infected with HIV and early syphilis, treatment with BP-G and outcomes related to syphilis treatment. All articles underwent a risk of bias assessment and data extraction was completed on all included studies.
Results: Seven studies were eligible for final inclusion, data extraction and analysis. The evidence from the final included studies was from non-randomized controlled trials. In general, no significant differences were found between groups treated with one versus two or more doses of BP-G; but there was a trend toward longer time to treatment failure with three doses. Differences in methodology limit the ability to draw any firm conclusions on the relative efficacy between these two treatment regimens.
Conclusion: Insufficient data exist to ascertain whether or not there is an added benefit from additional doses of BP-G for the treatment of early syphilis with HIV co-infection. A high-quality, randomized controlled trial is needed to definitively answer this question.
Introduction
The effective management of early syphilis (primary, secondary and early latent stages) among human immunodeficiency virus (HIV) co-infected persons is an important public health issue due to the resurgence of syphilis in Canada and globally Footnote 1Footnote 2Footnote 3Footnote 4Footnote 5. Between 2000 and 2012, the rate of infectious syphilis in Canada increased from 1.84 to 8.85 cases per 100,000 persons Footnote 4. Similar increases were also noted in Europe and the United States Footnote 1Footnote 5.
The prevalence of early syphilis is higher among HIV co-infected persons than in the general population Footnote 6Footnote 7Footnote 8. In addition, case reports and case series suggest that co-infection with HIV can result in atypical or more severe manifestations of syphilis. HIV-positive persons are more likely to present with multiple, larger ulcers, primary and secondary infections can overlap and cerebrospinal fluid (CSF) abnormalities, ocular and ophthalmologic manifestations are more common Footnote 9Footnote 10Footnote 11Footnote 12Footnote 13. Intercurrent syphilis may also temporarily increase HIV viral load and reduce CD4 lymphocyte count Footnote 14Footnote 15.
Furthermore, the optimal treatment for HIV-positive individuals co-infected with early syphilis has been controversial since early reports of treatment failures in the late 1980s which followed standard recommended treatment regimens Footnote 16. Since that time, a number of studies have been completed, but their small sample sizes, largely observational nature and methodological heterogeneity has prevented the development of strong evidenced-based recommendations about optimal treatment in this situation. Current guidelines have been criticized for not guiding physicians Footnote 17 and ambiguity in recommendations has resulted in varied clinical practice Footnote 18. As a result, the optimal antibiotic regimen among individuals with early syphilis remains controversial and, at times, the guidance is unclear Footnote 19Footnote 20.
Findings from a systematic review published in 2011 suggest that inadequate high-quality evidence exists to fully understand the efficacy of additional doses of BP-G for the management of early syphilis in individuals with HIV as measured by serologic response to treatment Footnote 19Footnote 20. Further, it is unclear whether improved outcomes would be achieved from additional doses of BP-G and whether this would outweigh the requirement of individuals to adhere to additional painful injections scheduled over multiple clinical visits Footnote 19.
In order to address these issues, a systematic review of recent evidence was carried out to compare the efficacy of one versus three doses of BP-G in the treatment of adults with HIV and early syphilis infection.
Methods
Search strategy
A search of the published literature was conducted using the following electronic databases: MEDLINE, EMBASE, Cochrane Library, Clinical trials.gov, Canadian Agency for Drugs Technologies in Health and Scopus. Hand searches were also completed to identify any relevant studies that may have been missed in the initial search. Key words used for the search were: "syphilis" or "Treponema pallidum", "human immunodeficiency virus" or "HIV" and "follow-up" or "treatment".
Eligibility criteria
Eligibility criteria were designed to update a previous systematic review published in 2011 Footnote 19. Studies were eligible for inclusion if they were published between January 2010 and May 2015 in English or French. Studies were required to report upon early syphilis in adult populations who are HIV-positive and interventions comparing doses of BP-G. All eligible articles were required to report outcomes related to the biologic cure of early syphilis.
Study selection
A two-step process was used to exclude less relevant publications. Two authors (ON, SH) independently screened titles followed by abstracts. Any discrepancies were resolved in discussion with a third author (LP). Publications were excluded if they did not have the following key words: "HIV" or "human immunodeficiency virus" and "early syphilis" and "treatment" or "antibiotic" or "management" or "follow-up" or "therapy".
An a priori decision was made to use the ACROBAT-NRSI to assess risk of bias in included observational studies Footnote 21 the Cochrane risk of bias assessment tool for randomized controlled studies (RCT) Footnote 22 and for accessing methodological quality of systematic reviews (AMSTAR) Footnote 23. Overall quality appraisal of each study was performed by two reviewers (ON, SH) and a third author (LP) was consulted to resolve any disagreement.
Data extraction
Two authors (ON, SH) extracted information on the study design, target population, sample size and intervention. The lack of consistency of reported data did not allow for meta-analysis. Therefore, results were summarized in a narrative format.
Results
A total number of 328 citations were identified through different databases and hand searches (Figure 1). After the title screening, 21 citations were retained for abstracts screening. Finally, seven publications were excluded based on language Footnote 24 study design Footnote 25Footnote 26 or absence of treatment data Footnote 27Footnote 28Footnote 29Footnote 30. The remaining 14 studies were eligible for full text retrieval. Seven studies were excluded for the following reasons: not early syphilis Footnote 31Footnote 32Footnote 33 or unspecified treatment information Footnote 34Footnote 35Footnote 36. Seven final publications were included for data extraction and synthesis. Key characteristics of the included studies are reported in Table 1 (excludes two systematic reviews).
Figure 1: Inclusion and exclusion of publications
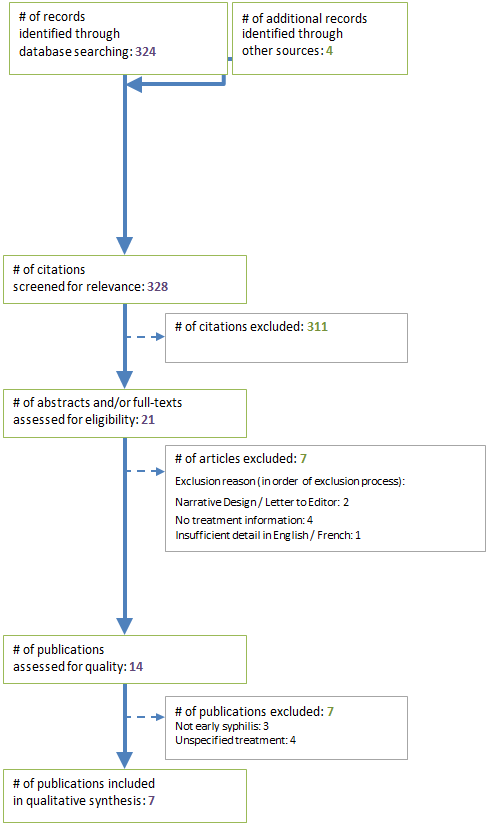
Text description: Figure 1
Figure 1: Inclusion and exclusion of publications
This figure is a flow diagram showing how publications were assessed for inclusion or exclusion into the study. At the top are two boxes that show there were 324 records were identified through database searching and 4 through other sources. An arrow points to the screening boxes. Two boxes identify that 328 citations were screened for relevance and 311 citations were excluded. An arrow then points to the eligibility boxes that show 21 abstracts and or full-texts were assessed for eligibility and 7 were excluded. The exclusion reason (in order of exclusion process) were: narrative Design or letter to Editor: 2, No treatment information: 4 and Insufficient detail in English or French: 1. An arrow points to the next box noting 14 publications were assessed for quality, and 7 were excluded; 3 because they did not address early syphilis and 4 because of unspecified treatment. An arrow points to the final box that notes there were 7 publications included in the qualitative synthesis.
Study characteristics
Among the included publications, there were two systematic reviews and five studies of observational design (two prospective cohort studies and three retrospective studies). The systematic review by Blank et al. was focused on HIV co-infected population Footnote 19; however, the review by Clement et al., focused more broadly on syphilis with some discussion of HIV co-infection Footnote 20. Studies were conducted in Taiwan Footnote 37 the United States Footnote 38 and Europe Footnote 39Footnote 40Footnote 41. The participants in the studies were predominantly male. In four studies, the intervention was one or three doses of BP-G Footnote 37Footnote 39Footnote 40Footnote 41. A single study compared one dose of BP-G with two or more doses of BP-G Footnote 38 (Table 1).
| Author | NumberTable 1 - Note 1 | Country of study | HIV treatment | Pharmacological regimen | Follow-up period | Outcome measure |
|---|---|---|---|---|---|---|
| Cousins Footnote 39 2012 | 62 (3 dose) Unclear (1 dose) |
United Kingdom | Unspecified: Some patients receiving antiretroviral therapy (ART) | 1 dose: BP-G 2.54 MU versus 3 doses: BP-G 2.4 MU |
12 months | Serological cure (4-fold decrease in serum RPR or serofast for 12 months) |
| Knaute Footnote 40 2012 | 88 | Switzerland | Not specified | 1 dose: BP-G 2.4 MU versus 3 doses: BP-G 2.4 MU |
3, 6, 8, 9, 12, 18, 24 months | Serological Response (VDRL, TPPA, Pathozyme IgM) |
| Tittes Footnote 41 2013 | 84 | Austria | 44% (35/80) were on ART | 1 dose: BP-G 2.4 MU versus 3 doses: BP-G 2.4 MU |
3, 6 and 12 months | Serological cure (4-fold decrease in VDRL within 6 months) Time to cure (days) |
| Yang Footnote 45 2014 | 420 | Taiwan | 63.2% (362) on combination ART | 1 dose: BP-G 2.4 MU versus 3 doses: BP-G 2.4 MU |
6 and 12 months | Serological response (4-fold decrease or greater in RPR titres at 12 month follow-up) |
| Ganesan Footnote 38 2015 | 286 (393 infectionsTable 1 - Note 2) | United States | 59% used Highly active retroviral therapy (H) at first syphilis case | 1 dose: BP-G 2.4 MU versus ≥2 doses: BP-G 2.4 MU |
3, 6, 9, 12, 18, 24 months | Serologic response Seroconversion |
Risk of bias of included studies
There were no high-quality intervention studies available to include in the update and therefore the overall body of evidence includes important biases (Table 1). The possibility of the introduction of selection biases through lack of randomization is important. Of additional concern, the publications generally did not adjust for the known confounder of HIV treatment status or other comparable measures (e.g., CD4 count). In addition, the publications often did not stratify participants by stage of syphilis and HIV status (including treatment) and there were often considerable missing data (Table 2).
| Author | BP-GTable 2 - Note 1 doses | Serological response at 12 monthsTable 2 - Note 2 | HIV association with serological response | ||
|---|---|---|---|---|---|
| Response | p value | Response | p value | ||
| Cousins Footnote 39 2012 | 1 dose | 78.9% (95% CI 68.0-89.8) |
p > 0.05 | N/A | - |
| 3 doses | 64.1% (95% CI 45.0-73.2) |
- | N/A | - | |
| Knaute Footnote 40 2012 | 1 dose | 100% | - | HIV-negative (reference) | - |
| 3 doses | 100% | - | CD4 ≥500 HR 1.27 (95% CI 0.79-2.04) CD4 < 500 HR 0.83 (95% CI 0.60-1.14) |
p = 0.332 p = 0.241 |
|
| Tittes Footnote 41 2013 | 1 dose | 88% | - | No correlation between viral suppression and outcome - data not shown |
p = 0.18 |
| 3 doses | 97% | - | |||
| Yang Footnote 45 2014 | 1 dose | 66.2% (95% CI 59.6-72.4) |
p = 0.24 | CD4 ≤ 200 (reference) | - |
| 3 doses | 71.8% (95% CI 64.7-78.2) |
- | 200 < CD4 ≤ 350 ORADJ 1.05 (95% CI 0.54-2.07) CD4 > 350 ORADJ 1.51 (95% CI 0.69-3.51) |
p = 0.88 p = 0.30 |
|
| Ganesan Footnote 38 2015 | 1 dose | 92% | - | CD4 (per 100-cell increase) HR 1.07 (95% CI 1.01-1.12) |
p = 0.02 |
| ≥2 doses | 92% | - | |||
Outcomes
Overall, four observational studies not included in the 2011 and 2014 systematic reviews included sample sizes of 62 to 350 subjects and concluded that there were no differences in serologic response at 12 (n=2 or 3 studies) or 24 months (n=1 study) if one or three doses BP-G were given Footnote 38Footnote 39Footnote 40Footnote 41. The fifth and largest, observational study conducted to date (n=573) in Taiwan hypothesized that one dose was not inferior to three weekly doses of BP-G and set the significant difference between regimens as 10% of participants with a serologic response at 12 months. Yang et al. were unable to demonstrate that one dose was not inferior to three doses of BP-G Footnote 37.
Serologic response
Serologic response to syphilis treatment is monitored by serial testing using the rapid plasma reagin (RPR). The RPR is a non-specific test that detects both IgM and IgG antibodies and is a measure of response to treatment Footnote 42. In the included studies, serologic response was commonly defined as ≥4 fold reduction in RPR titres at 12 months.
Among the included publications, four did not show a benefit from additional doses beyond the standard therapy of 2.4 million units in a single dose of BP-G Footnote 37Footnote 39Footnote 40Footnote 41Footnote 43. Yang et al.'s study was suggestive of a positive effect from additional doses: the effect was statistically significant when analyzed by last known results (p = 0.04) but not when analyzed per protocol (p= 0.24) Footnote 37.
The time to the first episode of serological failure appeared shorter in the group that received one dose: 1,184 (40 months) days for one-dose group and 1,436 days (almost 48 months) in the three-dose group, suggesting benefit from a three-dose regimen Footnote 37. In the same study, a Kaplan-Meier survival plot model showed a statistically significant longer mean time to failure in the group that received three-dose regimen (p = 0.03) Footnote 37.
HIV status
Co-infection with HIV impacts the manifestations and potential response to treatment of early syphilis. Therefore, the use of anti-retroviral therapy is an important characteristic to include when assessing the efficacy of syphilis therapy. Tittes et al. found a slower response to one-dose therapy within an HIV-positive group compared to an HIV-negative group, but there was no significant benefit when the two groups were treated with the three-dose regimen Footnote 41. Ganesan et al. found similar findings when comparing populations with CD4 <500 cells/μL compared to CD4 ≥ 500 cells/μL (p = 0.012) and the Knaute et al publication reported a statistically significant association between higher CD4 count and time to response to therapy Footnote 38Footnote 40.
Harms
The included publications did not document harms of therapy. Up to 10% of the population will report allergies to penicillin Footnote 44 potentially posing challenges to the use of BP-G. In addition, the intramuscular route of administration is painful and the additional health care visits are a potential burden to both the health care system as well as the patient.
Discussion
This review confirms that there is limited evidence to definitively guide management decisions in HIV-infected persons with early syphilis infection. The foundational 2011 systematic review concluded that the optimal treatment regimens remain unknown Footnote 19 and, although limited in its focus on HIV-positive individuals, the 2014 systematic review Footnote 20 concluded that in the absence of compelling data, individuals with HIV infections should be treated similarly to uninfected patients. This review of additional studies not included in these systematic reviews is consistent with the recommendation to use single-dose BP-G for HIV co-infected patients based on serologic response as the treatment outcome.
Of note, however, was the finding by Yang et al., which suggested a possible benefit of three doses of BP-G by reporting a longer time to serologic treatment failure in this group Footnote 37. However, the authors described a number of limitations to the study that could potentially explain the difference in their findings relative to other studies: 1) the study was not a RCT; 2) the decision to use one vs three doses was made by physicians assessing the patients; 3) 25% had missing RPR titres on follow-up testing; 4) the researchers could not definitely differentiate between re-infection and treatment failure; and, 5) the majority of patients were men who have sex with men (MSM) which limited the generalizability of their findings to other populations. In later correspondence, the same authors reported that the proportion of HIV co-infected patients with early syphilis treated with three doses at the eight centres in Taiwan declined from 60.2% in 2007-2009 to 25% in 2012 after a change in the Centre for Disease Control and Prevention (CDC) guidelines recommending single-dose BP-G Footnote 45.
Finally, results suggested that individuals with the highest CD4 counts had lower risk of poor serologic response to treatment Footnote 40Footnote 45.
Strengths and limitations
Strength of this review is that it is built on a previous systematic review and that it took all precautions to minimize bias. However, a number of limitations need to be considered when interpreting the results. Included studies were limited by their design and the potential impact of selection bias through the lack of randomization to dosing schedules contributes important potential biases to the results. The lack of adjustment for HIV treatment/status (anti-retroviral therapy and CD4 count) further limits the ability to extrapolate results to the clinical setting. These are known and significant associations Footnote 37 but the included publications were not analyzed with a stratified or multivariate approach Footnote 37Footnote 39Footnote 40Footnote 41. Studies were not representative of the entire population at risk for syphilis and HIV co-infection. More than 80% (and as high as 99%) of included study participants were male, of which the majority appeared to be MSM. Ideally, an adequately-powered RCT with clearly stratified HIV populations including women and with sufficiently long follow-up periods should be conducted to more definitely answer this question.
Conclusion
Our review found that the recent publications do not demonstrate a clear benefit to additional doses of BP-G for the treatment of individuals with early syphilis and HIV co-infection; more definitive trails are needed.
Acknowledgements
The authors would like to acknowledge Ella Westhaver for her support with systematic searches through electronic databases and the technical contribution of the Expert Working Group for the Canadian Guidelines for Sexually Transmitted Infections.
Conflict of interest
None to declare.
Funding
This systematic review was funded by the Public Health Agency of Canada.