Original quantitative research – A cost-utility analysis of the impact of electronic nicotine delivery systems on health care costs and outcomes in Canada

HPCDP Journal Home
Published by: The Public Health Agency of Canada
Date published: January 2022
ISSN: 2368-738X
Submit a manuscript
About HPCDP
Browse
Previous | Table of Contents | Next
Catherine M. Pound, MD, MScAuthor reference footnote 1Autheference or rfootnote 2; Doug Coyle, PhDAuthor reference footnote 2
https://doi.org/10.24095/hpcdp.42.1.05
This article has been peer reviewed.
Author references
Correspondence
Catherine Pound, Division of Pediatric Medicine, Children’s Hospital of Eastern Ontario, 401 Smyth Road, Ottawa, ON K1H 8L1; Tel: 613-737-7600, ext. 2701; Email: cpound@cheo.on.ca
Suggested citation
Pound CM, Coyle D. A cost-utility analysis of the impact of electronic nicotine delivery systems on health care costs and outcomes in Canada. Health Promot Chronic Dis Prev Can. 2022;42(1):29-36. https://doi.org/10.24095/hpcdp.42.1.05
Abstract
Introduction: We determined the impact of electronic nicotine delivery systems (ENDS) on health outcomes and costs in Canada, based on their effect on smoking cessation and smoking initiation rates.
Methods: We used gender-specific Markov models to estimate lifetime discounted life years, quality-adjusted life years (QALYs) and smoking-related health care costs for cohorts of males and females aged 15 to 19 years, in scenarios in which (1) ENDS are available (status quo); (2) ENDS are completely unavailable; and (3) ENDS are available for smoking cessation through health care provider prescription, in addition to currently recognized smoking cessation tools. Analysis was from the perspective of a publicly funded health care system.
Results: Outcomes are expressed per 1000 individuals and based on expected values obtained through a Monte Carlo simulation of 10 000 replications. For males aged 15 to 19 years, life years, QALYs and smoking-related health care costs were 41 553, 35 871 and CAD 79 645 964, respectively, when ENDS were available; 41 568, 35 894 and CAD 79 645 960 when ENDS were unavailable; and 41 570, 35 897 and CAD 79 605 869 when ENDS were available through prescription only. For females, life years, QALYs and smoking-related health care costs were 43 596, 37 416 and CAD 69 242 856, respectively, when ENDS were available; 43 610, 37 438 and CAD 69 085 926 when ENDS were unavailable; and 43 611, 37 438 and CAD 69 076 034 when ENDS were available through prescription only. Thus, situations in which ENDS are unavailable or available through prescription only are dominant over the status quo.
Conclusion: These results show that a policy change whereby ENDS were unavailable to the Canadian population or available through prescription only would likely increase population health and reduce health care costs.
Keywords: tobacco products, smoking, smoking cessation, electronic nicotine delivery systems, vaping, cost-benefit analysis
Highlights
- Vaping is commonly used by smokers to try to quit smoking.
- Vaping may increase smoking initiation in youth, resulting in negative long-term impacts on health.
- The results of this study show that, under our study assumptions, restricting access to vaping is likely to result in increased population health and reduced health care costs.
- Policy changes restricting access to vaping need to be examined with caution to avoid unintended consequences such as negative health impacts for current and former smokers who rely on vaping as a harm reduction strategy.
Introduction
Despite a considerable decrease in smoking prevalence over the last 50 years,Footnote 1 smoking continues to be the most common cause of preventable disease and mortality in Canada.Footnote 2 The costs of tobacco use in 2012 were CAD 16 billion, with the majority of indirect costs (CAD 9.5 billion) related to lost wages from long-term disability and premature mortality, and the largest portion of direct costs (CAD 6.5 billion) associated with health care costs.Footnote 2 Given the impact of smoking on the Canadian population, the Government of Canada has set a target to reduce smoking prevalence to below 5% by 2035.Footnote 3 Reaching that target requires a careful examination of smoking cessation strategies, as well as strategies preventing smoking initiation.
Vaping, the act of using electronic nicotine delivery systems (ENDS), is a common smoking cessation strategy. In Canada, almost one-third of current and former smokers report having vaped as a way to try to quit smoking.Footnote 4 There are, however, concerns that vaping could lead to increased cigarette smoking initiation in youth.Footnote 5Footnote 6 This association, though, has been debated, with some studies suggesting a negligible risk of youth vaping-related smoking initiation at the population level.Footnote 7Footnote 8 In Canada, 11% of youth aged under 25 years and 32% of teens aged 15 to 17 years who ever smoked daily used ENDS prior to initiating traditional cigarette smoking.Footnote 5 Also of concern is the rising prevalence of vaping over time. Between 2013 and 2019, the proportion of Canadian youth aged 15 to 19 years who endorsed vaping increased from 20% to 36%, while that of those aged 20 to 24 years jumped from 20% to 48%.Footnote 9Footnote 10
While ENDS can have a positive effect on smoking cessation,Footnote 11Footnote 12 their impact on youth smoking initiation is concerning. In order to determine whether the benefits incurred from ENDS through smoking cessation alone are sufficient to offset the negative health consequences of increased smoking initiation, we conducted a cost-utility analysis to determine the impact of ENDS on life expectancy, quality-adjusted life years (QALYs) and smoking-related health care costs in Canada under three different scenarios: (1) the status quo, i.e. current access to ENDS; (2) a complete ban of ENDS; and (3) limited access to ENDS for smoking cessation only, as prescribed by a medical professional. Sales of nicotine-containing vaping products are currently permitted to adults aged 18 years and older in Canada,Footnote 13 although some provinces have a higher minimum age.
Methods
We used gender-specific Markov models to examine smoking behaviours of nonsmokers and current and former smokers in relation to ENDS, as well as the impact of ENDS on mortality and smoking-related illnesses in cohorts of males and females aged 15 to 19 years. We estimated life years, QALYs and smoking-related health care costs in three different scenarios:(1) the status quo, in which ENDS are widely available; (2) a complete ban on ENDS; and (3) limited access to ENDS through prescription by health care professionals, for smoking cessation purposes. In the prescription-based scenario, we modelled the impact of ENDS as an increment on current smoking cessation tools, and not as a replacement for currently approved tools.
The perspective of the reference-case analysis was that of publicly funded health care system. While public health care costs are under the jurisdiction of provincial ministries, the results are generalizable to all ministries, since data were extracted from national population-based surveys. A lifetime horizon was adopted to take into consideration the overall lifetime costs and health effects.
Data collection
Data from population-based surveys of the Canadian population were used where possible and supplemented with data available through literature review. Although the Canadian Community Health SurveyFootnote 14 was initially considered because it collects information on a large number of Canadians (over 100 000 Canadians 12 years of age and over), specific data on smoking and vaping status were extracted from the Canadian Tobacco, Alcohol and Drugs Survey (CTADS), 2017.Footnote 4 While the sample size is smaller (approximately 16 000 Canadians 15 years and over), the CTADS offers more vaping- and smoking-specific data, and oversamples the 15 to 24 year age group, which comprises the population at highest risk for smoking initiation.Footnote 4 We stratified data by age groups and gender.
Model design
We extracted age-specific distributions for each of the smoking states (nonsmokers, current smokers and former smokers) from the 2017 CTADS,Footnote 4 and simulated how the cohort progresses through life, from 15 to 105 years of age (Figure 1).
In the scenarios in which ENDS are available, we examined their impact on smoking initiation between ages 15 and 24, and their impact on smoking cessation for ages 25 to 105. An age of 25 was chosen as a cut-off between youth and adults, since Canadian population-based survey dataFootnote 4Footnote 14 show that the number of nonsmokers initiating smoking or vaping after the age of 25 is extremely small.
In the scenario in which ENDS are available for smoking cessation through prescription only, we assumed a reduced access to ENDS for smokers, since almost 15% of Canadians over the age of 12 do not have access to a primary health care provider.Footnote 15 A cycle length of six months was used, since smoking cessation is traditionally defined as sustained abstinence of at least six months’ duration.Footnote 16 This timeframe therefore represents the minimum time period required to go from current to former smoker.
We extracted smoking status from the CTADS based on the traditional smoking categories used in the survey (current daily smoker; current occasional smoker; former daily smoker; former occasional smoker; experimental smoker; lifetime abstainer), and regrouped them into the following new categories:
- Current daily smokers: current daily smokers
- Former daily smokers: former daily smokers and current occasional smokers who were once daily smokers
- Never/experimental/occasional (never-daily) smokers: experimental smokers, lifetime abstainers, former occasional smokers and current occasional smokers who were never daily smokers
Current occasional smokers who were once daily smokers were grouped together with former daily smokers to allow for a conservative estimate of costs and QALY losses associated with smoking-related illnesses. Current occasional smokers who once were daily smokers are likely at higher risk of smoking-related illnesses than former daily smokers who are now completely abstinent, but are also likely at lower risk of smoking-related illnesses than current daily smokers. Grouping current occasional smokers who were once daily smokers with current daily smokers would inflate illness-related costs and QALY losses, which were based on risk estimates for current daily smokers.
Current occasional smokers who never were daily smokers were grouped together with the “Never/experimental/occasional (never daily) smokers,” since these individuals are also unlikely to experience the same costs and QALY losses associated with smoking-related illnesses that daily smokers experience.
These groupings therefore allow for the most conservative approach for estimating costs and QALY losses associated with smoking-related illnesses.
In addition to the smoking states, we also included the following:
- Ever ENDS users (15–24 years of age model): individuals who have ever vaped
- Dual users (25–105 years of age model): individuals who used e-cigarettes to quit smoking in the two years before the survey
- Death
Schematics of the models for each scenario are shown in Figure 1.
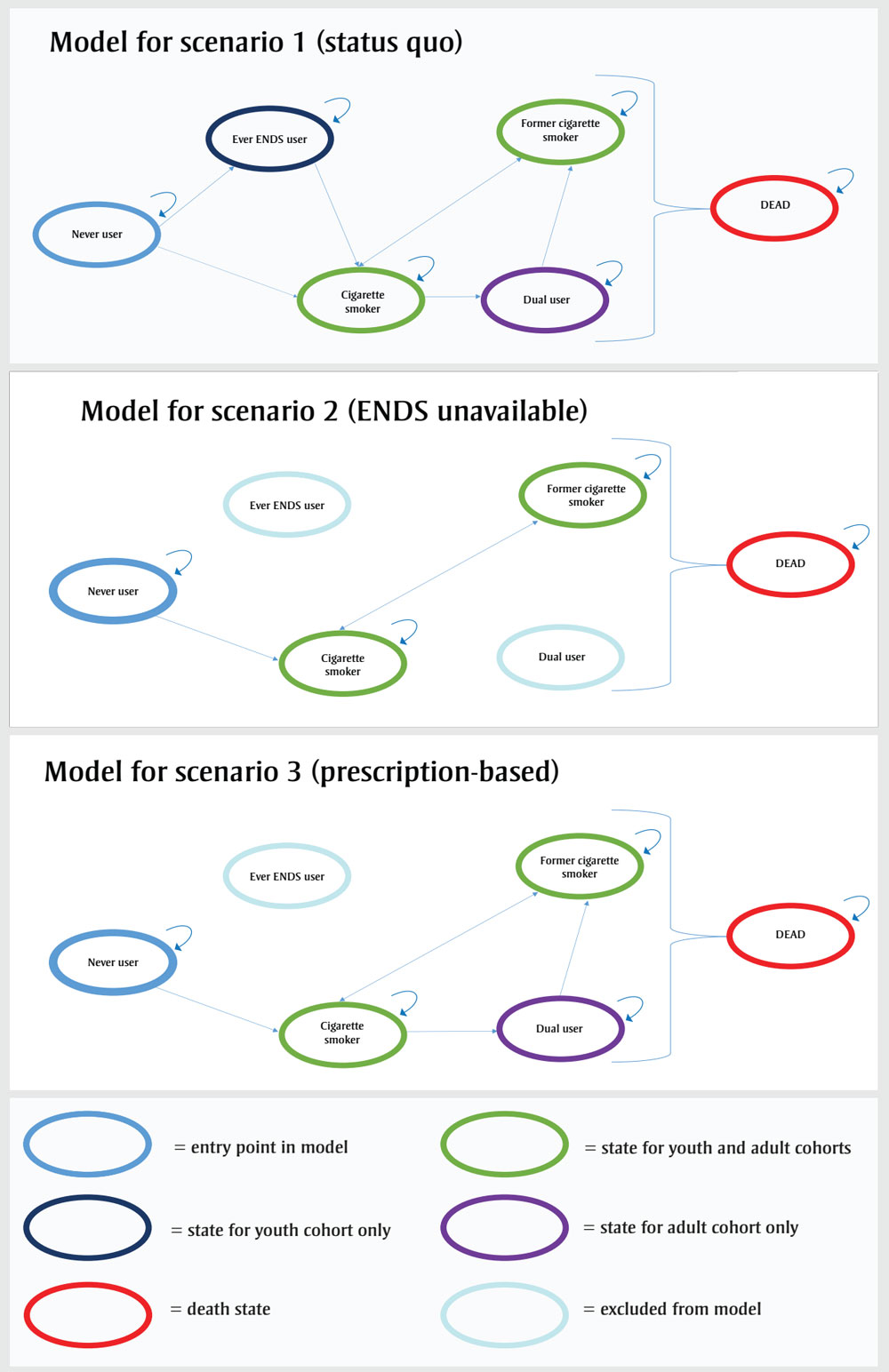
Figure 1 - Text description
- Markov Model for scenario 1 (status quo)
- Never user (Entry point in model) links to both Ever ENDS user (state for youth cohort only) and cigarette smoker (state for youth and adult cohorts only). Ever ENDS user links to cigarette smoker. Cigarette smoker links to former cigarette smoker (state for youth and adult cohorts only) and dual user (state for adult cohort only). Former cigarette links to cigarette smoker. Dual user links to former cigarette smoker. All of these behaviours lead to dead (death state).
- Markov Model for scenario 2 (ENDS unavailable)
- Never user (Entry point in model) links to cigarette smoker (state for youth and adult cohorts only). Cigarette smoker links to former cigarette smoker (state for youth and adult cohorts only). Former cigarette links to cigarette smoker. Dual user and ever ENDS users are behaviours that are not linked to other behaviours. All of these behaviours lead to dead (death state).
- Markov Model for scenario 3 (Prescription-based)
- Never user (Entry point in model) links to cigarette smoker (state for youth and adult cohorts only). Cigarette smoker links to former cigarette smoker (state for youth and adult cohorts only) and dual user (state for adult cohort only). Former cigarette links to cigarette smoker. Dual user links to former cigarette smoker. Ever ENDS users is a behaviour that is not linked to other behaviours. All of these behaviours lead to dead (death state).
Abbreviation : ENDS, electronic nicotine delivery system.
Data requirements
Transition probabilities
Age- and gender-specific transition probabilities were obtained for smoking initiation, continuation and cessation in all three main scenarios.
CTADS allows determination of the proportion of a given age-gender cohort by smoking status (current daily smoker, current occasional smoker, former daily smoker, former occasional smoker, experimental smoker and lifetime abstainer) at the time of the survey. One can also derive both the age at which a daily smoker started smoking and the time since a former daily smoker quit smoking. From this information, it was possible to determine the proportion of a given age-gender cohort by smoking status at the start of each age-gender cohort. By assuming that the transition from one smoking status to another (i.e. the probability of initiation of smoking and the probability of smoking cessation) remains constant within a specific age-gender group, we were able to obtain transition probabilities by calibration of the data by determining the probabilities that replicate the proportion in each smoking status category (current, former and never smoker) both at the time of beginning of age cohort and at the time of survey (calculation example available in transition probabilities document).
We used a relative risk (RR) for smoking initiation in ENDS users versus nonusers obtained from the literatureFootnote 17 (RR = 2.18, 95% CI: 1.65–2.83), as it was more conservative than the RR we extracted from CTADS-derived data (RR = 7.83 in males and 9.09 in females). The large RR difference was felt to be related to the small sample size of the CTADS. The RR obtained from the literature was applied to both male and female 15-to-24-years cohorts to determine transition probabilities relating to daily smoking initiation for ENDS users versus nonusers through a further calibration process. A slightly more conservative adjusted odds ratio (AOR) of 1.79 was described in an observational Canadian study;Footnote 18 however, the RR of 2.18 mentioned above was chosen for this study because the RR measure is more generalizable to populations other than the AOR. Finally, the RR of smoking cessation in ENDS users versus nonusers (RR = 1.05 in males and 1.08 in females) was obtained from CTADS data and applied to the 25-years-and-above cohort, so as to determine transition probabilities for ENDS users versus ENDS nonusers. This RR represents the additional smoking cessation benefits conferred by ENDS over and above those seen with other currently available smoking cessation tools.
Mortality by smoking status
Annual probabilities of mortality for each age-gender cohort were obtained from Statistics Canada.Footnote 19 We then estimated the six-month probabilities of dying for each age-gender cohort by smoking status, through a calibration process combining the Statistics Canada mortality data with the baseline CTADS smoking status data. We also estimated the age- and gender-specific RR of mortality by smoking status from the literatureFootnote 20 (sample calculation available in calculation of probabilities document).
Smoking-related diseases
We obtained the prevalence of age- and gender-specific smoking-related illnesses (chronic obstructive pulmonary diseaseFootnote 21 [COPD], coronary heart diseaseFootnote 22 [CHD], strokeFootnote 22) from Canadian population-based data (data available in supplementary tables). For lung cancer,Footnote 23 only the incidence was available, but given the short median survival of lung cancer (16.9 months),Footnote 24 incidence was felt to correlate closely with yearly prevalence. We chose to include these diseases as they represent approximately 75% of the smoking-related mortality in developed countries.Footnote 25 The RR of developing these diseases for each smoking status was obtained from the literature.Footnote 20 Age- and gender-specific probabilities of developing each condition were obtained through calibration.
Vaping-related diseases
Given the paucity of data about the long-term health impacts of the prolonged use of ENDS, we assumed there were none. This is unlikely to be true, as there is some emerging evidence that ENDS may be associated with the development of COPD,Footnote 26Footnote 27 independent of cigarette smoking. However, given the lack of clear evidence, this assumption allows for the most conservative approach.
Costs
Average six-monthly costs related to the treatment of individuals with lung cancer,Footnote 28 COPD,Footnote 29 CHDFootnote 30 and strokeFootnote 31 were obtained from the literature. Costs obtained from previous years were inflated to current-year costs using the Bank of Canada Inflation Calculator. All costs are presented in 2020 Canadian dollars (data available in supplementary tables).
Utility values
We obtained age- and gender-specific utility values for never smokers from Canadian population data,Footnote 14 to which we applied disutility data related to smoking status.Footnote 32 The disutility data for each of the smoking-related illnesses (lung cancer, COPD, myocardial infarction, CHD and stroke) was obtained from a study based in the United KingdomFootnote 33 (data available in supplementary tables). We had planned on using Canadian- disutility data; however, the available Canadian data did not report on lung cancer separately from other cancers.Footnote 34 Disutility values for the other smoking-related illnesses were comparable between the UK and Canadian studies.
Analysis
All analyses were in the form of a cost-utility analysis to capture monetary and utility costs and benefits. A cost-utility approach was chosen given the possibility that one scenario could lead to more QALYs but not necessarily to reduced health care costs, since health and economic impacts related to smoking initiation occur many years later than those related to smoking cessation.
Probability distributions were used to account for uncertainty around the parameters of interest. We used beta distributions for transition probabilities and utility values, lognormal distributions for relative risks and gamma distributions for disutility values and uncertain costs (data available in supplementary tables). The probabilistic analysis was performed using a Monte Carlo simulation with 10 000 replications to ensure stability of the data. We used a threshold value of willingness to pay CAD 50 000 per QALY for interpretation of the results. All outcomes were weighted equally, regardless of the characteristics of people affected by the intervention. As per the CADTH guidelines,Footnote 35 an annual discount rate of 1.5% was applied to all costs and utilities. Confidence intervals were not produced because they are not considered meaningful for economic evaluations, the purpose of which is to inform binary decisions. Confidence intervals in this setting are not considered best practice.Footnote 35Footnote 36 Research Ethics Board approval was not needed for this study, given its use of publicly available data.
Assumptions
Two main general assumptions underlie the analysis proposed in this framework. First, we assumed no vaping-related long-term impacts, as ENDS are still relatively new. As already discussed, this is unlikely to be true. However, since most long-term vaping health impacts are not yet known, this assumption allows for the most conservative approach. Second, we assumed a stable smoking relapse rate across age groups, as no age-stratified smoking relapse rate was identified in our literature review. However, as people age, they may be less likely to relapse as more time elapses since their quit date.
Sensitivity analyses
In order to account for the uncertainty of assumptions upon which the analysis is built, we performed multiple sensitivity analyses. These include
- a relapse rate increasing by 10% in each decade of life;
- a relapse rate decreasing by 10% in each decade of life;
- a RR of 1.79 for vaping-related smoking initiation;Footnote 18
- a 90% decrease in ENDS access (instead of 100%) for all individuals in scenario 2, and for youth in scenario 3, to account for black market access and online ordering;
- an 80% decrease in ENDS access (instead of 100%) for all individuals in scenario 2, and for youth in scenario 3, to account for black market access and online ordering;
- a 50% decrease in ENDS access (instead of 15%) because of difficulty in accessing health care in the scenario in which ENDS are only available through prescription;
- a 10% decrease ENDS access (instead of 15%) because health care access may be easier than anticipated in the scenario in which ENDS are only available through prescription;
- a discount rate of 0%;
- a discount rate of 5%;
- a scenario in which youth vaping increases by 50%;
- a scenario in which youth vaping does not increase risk of smoking initiation; and
- a scenario in which vaping does not increase quitting rates.
Results
Table 1 summarizes the impact of ENDS on life expectancy, quality-adjusted life years (QALYs) and smoking-related health care costs in Canada under our three different scenarios: (1) the status quo, with current access to ENDS; (2) a complete ban of ENDS; and (3) limited access to ENDS for smoking cessation only, as prescribed by a medical professional. Results are presented per 1000 individuals. Incremental cost effectiveness ratios (ICERs) were not relevant, as all other scenarios are dominant over scenario 1.
In the sensitivity analyses, scenarios 2 and 3 were cost-saving for women in all situations compared to the status quo scenario, except for the one in which we assumed no association between youth vaping and smoking initiation. For men, scenarios 2 and 3 were cost-saving in most sensitivity analyses (except, again, when no association was assumed between youth vaping and smoking initiation), and otherwise would be considered cost-effective based on a willingness to pay of CAD 50 000 (relapse rate decreasing by 10% in each decade of life: ICER CAD 628 for scenario 2 vs. 1; increased access to physician to 90%: ICER CAD 28 for scenario 2 vs. 1; discount rate of 0%: ICER CAD 285 for scenario 2 vs. 1; data available in sensitivity analyses).
| Category | ENDS widely available (scenario 1) | ENDS completely unavailable (scenario 2) | Difference between scenarios 2 and 1 | ENDS available through prescription for smoking cessation (scenario 3) | Difference between scenarios 3 and 1 |
|---|---|---|---|---|---|
| Males | |||||
| Life years | 41 553 | 41 568 | 15 | 41 570 | 17 |
| QALYs | 35 871 | 35 894 | 23 | 35 897 | 26 |
| Costs | CAD 79 645 964 | CAD 79 645 960 | CAD -3 | CAD 79 605 869 | CAD -40 095 |
| Females | |||||
| Life years | 43 596 | 43 610 | 14 | 43 611 | 15 |
| QALYs | 37 416 | 37 438 | 21 | 37 438 | 22 |
| Costs | CAD 69 242 856 | CAD 69 085 926 | CAD -156 930 | CAD 69 076 034 | CAD -166 821 |
Abbreviations: CAD, Canadian dollars; ENDS, electronic nicotine delivery systems; QALY, quality-adjusted life year. |
|||||
Discussion
Our results show that, based on the study assumptions, a scenario in which ENDS are completely unavailable to the Canadian population would result in an increase in population health, as well as a reduction in health care costs, when compared to the status quo. A scenario in which ENDS are restricted to a prescription-based system would lead to even greater benefits and reduction in health care costs as compared to the other two scenarios analyzed.
The significant health and economic benefits conferred by smoking cessation are the rationale for harm reduction strategies. The main purpose of harm reduction is to decrease the impact of behaviours that are typically associated with negative consequences,Footnote 37 which, in the case of smoking, involve inhalation of toxins related to the combustion of tobacco. Because ENDS do not require tobacco combustion,Footnote 38 they are felt to be safer than cigarettes, and can therefore act as harm reduction tools by helping smokers transition to a less harmful habit.
Smoking cessation is known to result in important health and economic gains. For instance, the European Study on Quantifying Utility of Investment in Protection from Tobacco (EQUIPT) showed that, for each smoker who does not quit within a 12-month period, the system incurs an additional lifetime cost of USD 6460, and that smoker experiences a decreased life expectancy of 0.66 years and a reduction in lifetime QALYs of 1.09, as compared to a smoker who quits within the same period of time.Footnote 39
However, the results of our study are concerning: the dominance of scenario 2 (ENDS completely unavailable) over the status quo suggests that long-term harms incurred through increased smoking initiation in vaping youth outweigh the smoking cessation benefits of ENDS. This is even more apparent in scenario 3, in which ENDS are unavailable to youth but remain accessible for smoking cessation, leading to the most significant positive outcomes.
Our results align with those of some previous studies. Soneji et al.Footnote 40 demonstrated that, although ENDS are associated with increased years of life gained through their smoking cessation effect, they disproportionately increase years of life lost through increased youth-related smoking initiation. A similar conclusion was reached by Kalkhoran et al.,Footnote 41 who, through modelling of various scenarios in the US and the UK, showed that net harms resulted from all situations in which ENDS increased smoking initiation.
However, other studies have shown conflicting results. Cherng et alFootnote 42 showed that, under multiple scenarios, ENDS seemed to affect smoking cessation more than smoking initiation. Their study, however, only examined smoking prevalence and did not take into account other health or economic measures. Finally, a study by Levy et al.Footnote 43 projected a 21% reduction in smoking-attributable death and a 20% decrease in life years lost based on projected patterns of ENDS use in the US at the time of their study. The study projected a greater than 35% decrease in cigarette smoking by age 25 when ENDS were available; however, the authors assumed that only 5% of never-smoking youth ENDS users would go on to become daily smokers, which is about half of the proportion seen in Canadians under 25 years old.Footnote 4
Although our results show that limiting the availability of ENDS to the Canadian population, either completely or through prescription, could result in population health benefits and reduced health care costs, this conclusion must be seen through a realistic lens. A complete ENDS prohibition would offer the greatest protection to the smoking-naïve population; however, it would negate health gains accrued by smokers who have reduced or eliminated their cigarette consumption by switching over to ENDS.
Additionally, there are concerns that banning ENDS could result in the emergence of a black market,Footnote 44 with a potential increase in unsafe products. Scenario 3, which would allow smokers to access ENDS through a physician, demonstrated the greatest positive outcomes. This scenario would allow smokers to continue benefitting from ENDS while limiting youth exposure. Provincial health insurance program coverage could be explored, potentially resulting in decreasing financial barriers. However, this scenario also raises significant equity issues. Low-income Canadians are much more likely to have unmet health care needsFootnote 45 and potential difficulties in accessing physicians, yet they are also more likely to smoke.Footnote 46 These factors need to be taken into account when considering policy options.
Strengths and limitations
Our study’s major strength is that it relies on population-based Canadian data. Our assumptions about the impact of ENDS on smoking initiation, their use and their long-term health impacts were very conservative, so as to ensure the potential benefits of ENDS were not understated. The consistency of our findings across sensitivity analyses also lends credibility to our results.
There are, however, some populations that are not represented by the CTADS (i.e. residents of the Yukon, Northwest Territories and Nunavut, full-time residents of institutions and people without access to a land or cellular phone,Footnote 47) limiting generalizability of our findings to these groups. Also, concerns have been raised that CTADS may underestimate smoking prevalence due to higher nonresponse rates felt to be related to the inclusion of alcohol- and drug-related issues, which tend to be strongly associated with cigarette smoking.Footnote 48 It is therefore plausible that this would result in an under-estimation of the costs and health care costs reported in this analysis. In addition, having been created in 2003, ENDS represent a fairly new technology, and long-term safety data are not yet available, making long-term assumptions difficult.Footnote 38
Finally, in the prescription-only scenario, we based our ENDS-related smoking cessation estimate on individuals who used ENDS to quit smoking in the last two years. A scenario in which ENDS are prescribed by health care professionals would likely change risk perceptions and social norms around vaping, as well as potentially reduce the cost of ENDS, because they might then become subsidized by provincial health programs. This could lead to an increase in the proportion of smokers using ENDS as a smoking cessation tool, as compared to the estimate used in this evaluation, making the prescription-based scenario even more cost-saving as compared to the status quo.
Conclusion
Studies such as this one examining the impact of ENDS on health care costs and outcomes provide valuable information. Although our results suggest that restricting access to ENDS, either through a complete ban or through a prescription-based system, would be cost effective, factors such as equity and feasibility need to be considered. Alternative policy tools such as increased taxationFootnote 49 and strict marketing regulations have been shown to impact ENDS use. Further research should focus on determining the ideal policy mix that would achieve a balance between reducing ENDS-associated smoking initiation and offering support as a smoking cessation tool. More data is also needed about the long-term impact of ENDS on health.
Conflicts of interest
The authors have no conflicts of interest to disclose.
Authors’ contributions and statement
CP conceptualized and designed the study, carried out the analyses, interpreted the data, drafted the initial manuscript, and reviewed and revised the manuscript. DC participated in the conceptualization and design of the study, reviewed the analyses, participated in the interpretation of the data, and reviewed and revised the manuscript. Both authors approved the final manuscript as submitted and are accountable for all aspects of the work.
The content and views expressed in this article are those of the authors and do not necessarily reflect those of the Government of Canada.
