Canadian Biosafety Guideline Pathogen Risk Assessment

Download the alternative format
(PDF format, 1.09 MB, 46 pages)
Organization: Public Health Agency of Canada
Date published: April 2018
Table of Contents
- Preface
- Abbreviations and acronyms
- Chapter 1 - Introduction
- Chapter 2 - Principles of pathogen risk assessment
- Chapter 3 - Components of a pathogen risk assessment
- Chapter 4 - Glossary
- Chapter 5 - References and resources
Preface
In Canada, facilities where Risk Group 2, 3, and 4 human pathogens or toxins are handled and stored are regulated by the Public Health Agency of Canada (PHAC) under the Human Pathogens and Toxins Act (HPTA) and the Human Pathogens and Toxins Regulations (HPTR). The importation of animal pathogens, infected animals, animal products or by-products (e.g., tissue, serum), or other substances that may carry an animal pathogen or parts thereof (e.g., toxin) are regulated by the PHAC or the Canadian Food Inspection Agency (CFIA) under the Health of Animals Act (HAA) and Health of Animals Regulations (HAR).
The following figure depicts the document hierarchy used by the PHAC to oversee biosafety and biosecurity operations. Each tier of the pyramid corresponds to a document type, with documents increasing in order of precedence moving upwards. Acts and regulations are found at the top of the pyramid, as they are the documents that convey the PHAC's legal authorities. Guidance material and technical pieces are found at the bottom of the pyramid, as they are intended to summarize recommendations and scientific information only.
Figure 1: The Public Health Agency of Canada's Biosafety and Biosecurity Document Hierarchy
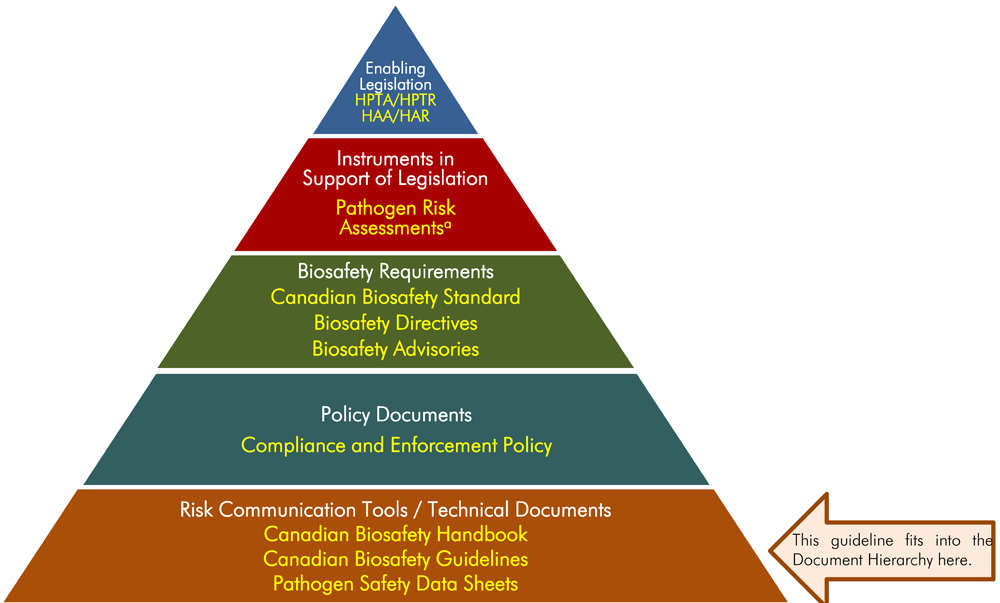
Figure 1: The Public Health Agency of Canada's Biosafety and Biosecurity Document Hierarchy
Figure in the form of a pyramid depicting the document hierarchy used by the PHAC to oversee biosafety and biosecurity operations. Each of the five tiers of the pyramid corresponds to a document type, with documents increasing in order of precedence moving upwards.
At the top sits the Enabling Legislation, that is, the HPTA, HPTR, HAA, and HAR, that convey the PHAC’s legal authorities. Below the acts and regulations sit Instrument in Support of Legislation, which are the Pathogen Risk Assessments. The next tier down are the Biosafety Requirements, which include the Canadian Biosafety Standard, Biosafety Directives, and Biosafety Advisories. The second lowest tier are the Policy Documents, the Compliance and Enforcement Policy. Guidance material and technical pieces found at the bottom of the pyramid, under the Risk Communication Tools and Technical Documents heading are intended to summarize recommendations and scientific information only. These include the Canadian Biosafety Handbook, Canadian Biosafety Guidelines, and Pathogen Safety Data Sheets.
a) Pathogen risk assessments determine a pathogen's risk group. This guideline provides instruction on how to perform a pathogen risk assessment.
The Pathogen Risk Assessment guideline was developed by the PHAC and the CFIA as part of a series of electronic publications that expand upon the biosafety and biosecurity concepts discussed in the current edition of the Canadian Biosafety Handbook (CBH), the companion document to the Canadian Biosafety Standard (CBS). It describes how to conduct a pathogen risk assessment to evaluate the risks associated with human or animal pathogens, toxins, or other regulated infectious material. This guideline is intended to assist organizations in meeting the requirements specified in the CBS, but should not be interpreted as requirements. Organizations may choose alternate approaches to meet the requirements specified in the CBS.
This guideline is continuously evolving and subject to ongoing improvement. The PHAC and the CFIA welcome comments, clarifications, and suggestions for incorporation into the future versions. Please send this information (with references, where applicable) to:
PHAC e-mail: PHAC.pathogens-pathogenes.ASPC@canada.ca
Abbreviations and acronyms
- CBH
- Canadian Biosafety Handbook
- CBS
- Canadian Biosafety Standard
- CFIA
- Canadian Food Inspection Agency
- HAA
- Health of Animals Act
- HAR
- Health of Animals Regulations
- HPTA
- Human Pathogens and Toxins Act
- HPTR
- Human Pathogens and Toxins Regulations
- LRA
- Local risk assessment
- PHAC
- Public Health Agency of Canada
- RG
- Risk group (i.e., RG1, RG2, RG3, RG4)
- SSBA
- Security sensitive biological agent
Chapter 1 - Introduction
The words in bold type are defined in the glossary found in Chapter 4.
1.1 Scope
Pathogens are classified by risk group based on the inherent characteristics of the pathogen. Risk Group 2 (RG2), RG3, and RG4 human pathogens are regulated under the Human Pathogens and Toxins Act (HTPA) and Human Pathogens and Toxins Regulations (HPTR).Footnote 1,Footnote 2 All animal pathogens are regulated under the Health of Animals Act (HAA) and Health of Animals Regulations (HAR).Footnote 3,Footnote 4 The risk group helps determine the operational practice requirements, minimum physical containment requirements, and applicable performance and verification testing requirements; however, there may be additional or different requirements based on the specific activities planned and, in some cases, based on the unique characteristics of the pathogen.
The operational practice requirements, physical containment requirements, and applicable performance and verification testing requirements for regulated containment zones are specified in the Canadian Biosafety Standard (CBS).Footnote 5 The Canadian Biosafety Handbook (CBH) provides guidance on how to meet the requirements.Footnote 6
1.2 Overview
The Pathogen Risk Assessment guideline provides comprehensive guidance on how to conduct a pathogen risk assessment on pathogens that are uncharacterized or that may have been modified, to determine the likelihood and consequences of exposure to the pathogen, or its release. This guideline is meant to be used in conjunction with the CBS, the CBH, and the Pathogen Risk Assessment Template available on the Government of Canada website.Footnote 7
The information provided in this document is meant as guidance only and should not be interpreted as requirements. Regulated parties may choose alternate approaches to meet the requirements specified in the CBS.
The pathogen risk assessment process serves to document the inherent risks associated with a pathogen and will inform the development of risk mitigation strategies. The end result of a pathogen risk assessment is the pathogen's risk group for both humans and animals. The information obtained from the pathogen risk assessment will help determine the appropriate containment level for working with the pathogen, and will be an input to the local risk assessment (LRA). It is in the LRA that the inherent risks associated with the pathogen are evaluated in the context of the activities being undertaken.
1.3 Risk analysis and risk assessment
The purpose of the risk assessment methodology presented in this guideline is to identify the critical risk factors for determining a pathogen's risk group, identify the key questions to be answered to fully assess each risk factor, and reduce inter-operator and intra-assessment variability. A solid risk assessment methodology ensures that a standardized approach is used so that the outcome (i.e., risk group) is consistent regardless of who is performing the assessment. Risk assessment can be highly subjective, particularly when data is limited or variable in nature. While analyses of the same data by different experts can lead to different interpretations, technical expertise remains the foundation of any risk assessment process.
1.3.1 Hazard
A hazard is a thing, or its property, that can be a danger to people or other living things. In a pathogen risk assessment, the hazard is a microorganism, protein, or nucleic acid with the potential for harm or adverse health effects (e.g., infection, disease, death) to humans or animals.
1.3.2 Risk
Risk is a function of the probability of an undesirable event occurring (e.g., exposure to a pathogen) and the severity of the consequences of that event (e.g., infection, disease, death). Both the probability of the event occurring and the consequence must exist for there to be a "risk"; if either is absent, there is no risk.
1.3.3 Risk assessment basics
Risk assessment is a mechanism for determining the likelihood that a hazard will cause harm and the degree of that harm. Risk assessments are based on science, policy, and expert judgement, and can evolve as the level of scientific understanding progresses.
In a biosafety context, the hazard is often a pathogen, and the risk assessment aims to characterize the likelihood that exposure would lead to disease, and the severity of the disease. The assessment may consider the activities performed, the characteristics of the pathogen, and the prevalence of the pathogen in the environment or community. Risk assessment is an iterative process comprised of four components: (1) hazard identification, (2) hazard characterization, (3) exposure assessment, and (4) review and continual improvement (Figure 1-1).
Figure 1-1: Basic risk assessment model. The basic risk assessment model involves four iterative steps: hazard identification, hazard characterization, exposure assessment, and review and continual improvement.
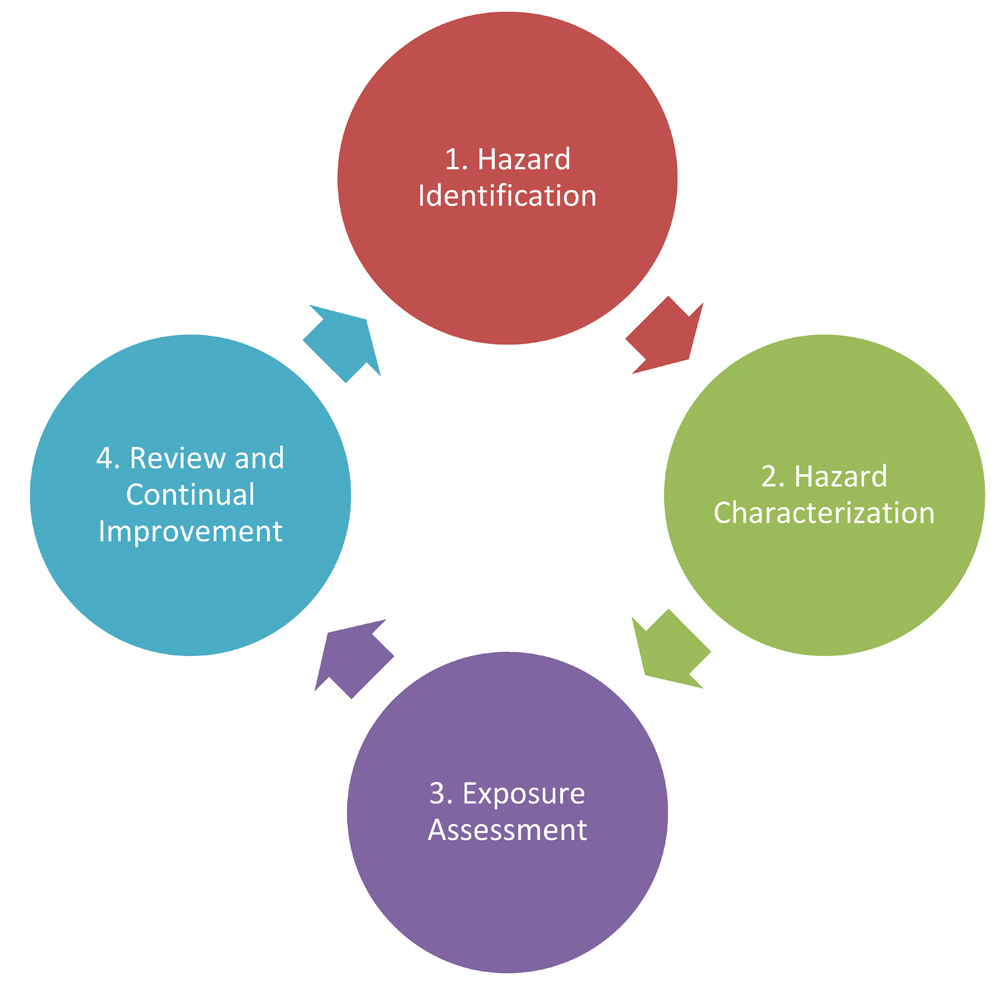
Figure 1-1: Basic risk assessment model. The basic risk assessment model involves four iterative steps: hazard identification, hazard characterization, exposure assessment, and review and continual improvement.
Figure depicting the relationship between the four iterative steps of a basic risk assessment model, starting with hazard identification, followed by hazard characterization, which is followed by exposure assessment then review and continual improvement, which completes the cycle by once again linking to hazard identification.
Several types of risk assessment are performed to support the biosafety program within an organization. The broadest is the overarching risk assessment, which encompasses risk management and mitigation strategies that apply to the biosafety program as a whole. The pathogen risk assessment determines the inherent risk of the pathogen, expressed in terms of risk group, which helps determine the minimum containment requirements, which are set out in the CBS. The LRA considers not only the pathogens or toxins in use, but also the activities being undertaken in the facility, and is used to determine whether the existing mitigation is sufficient and appropriate. The biosecurity risk assessment broadens these assessments to also determine the risks associated with the loss, theft, misuse, diversion, or intentional unauthorized release of pathogens, toxins, and related assets. These risk assessments are closely related to each other, but consider different aspects of risk. Figure 1-2 illustrates the relationship between the key elements of the biosafety program and the corresponding types of risk assessment.
Figure 1-2: Relationship between the key elements of the biosafety program and the corresponding types of risk assessment.
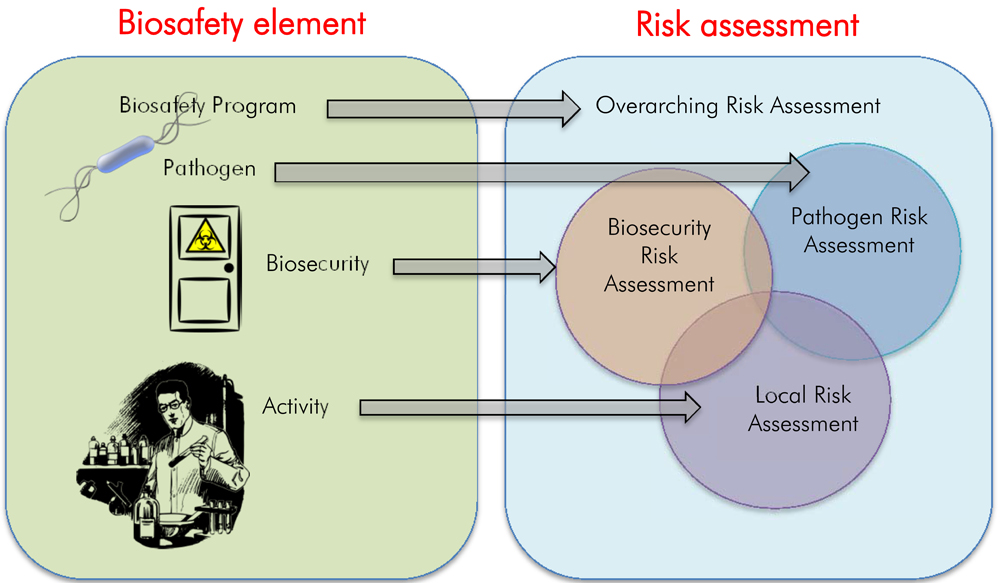
Figure 1-2: Relationship between the key elements of the biosafety program and the corresponding types of risk assessment.
Figure demonstrating the relationship between the risks associated with the key elements of the biosafety program and the corresponding types of risk assessment. These are the Biosafety Program linked to Overarching Risk Assessment; the Pathogen linked to Pathogen Risk Assessment; Biosecurity linked to the Biosecurity Risk Assessment; and the Activity linked to Local Risk Assessment. The figure also presents an overlap between pathogen risk assessment, biosecurity risk assessment, and local risk assessment showing that there are overlapping considerations. In addition, these three are all under the overarching risk assessment.
1.3.4 Risk groups
Risk groups range from RG1 pathogens, which pose a low risk to the individual and community, to RG4 pathogens, which pose a high risk to the individual and community (see Table 1-1). The risk group is a key factor in the determination of how a pathogen must be handled within the facility. All pathogens can be classified into risk groups based on the outcome of the pathogen risk assessment. The pathogen risk assessment characterizes the risks associated with the pathogen through the thorough examination of four risk factors:
- pathogenicity;
- communicability;
- pre-exposure measures and post-exposure measures; and
- impact on animal population (i.e., host range, natural distribution, and economic impact).
Most pathogens will clearly fall into one of the four risk groups. The single most important risk factor is pathogenicity (i.e., ability of a pathogen to cause disease). If an organism is unable to cause disease in humans or animals, it is irrelevant that it is not susceptible to current medical treatments. The remaining risk factors are important for understanding the community risk, and are particularly important for differentiating between RG3 and RG4 pathogens. More information on risk groups and risk factors can be found in Chapter 4 of the CBH.
| Risk Group | Individual Risk | Community Risk | Example |
|---|---|---|---|
| RG1 | No or Low | Low | Commensal bacteria |
| RG2 | Moderate | Low | Pathogenic Escherichia coli |
| RG3 | High | Low | Bacillus anthracis |
| RG4 | High | High | Ebola virus |
1.4 How to use the Pathogen Risk Assessment guideline
A detailed list of all abbreviations and acronyms used throughout this guideline is located at the beginning of this document. Each word or term is spelled out upon first use in the guideline, with the abbreviation immediately following in brackets. After its initial definition, the abbreviation is used exclusively throughout the remainder of the document. A comprehensive glossary of definitions for technical terms is located in Chapter 4 of this document. Terms defined in the glossary appear in bold type upon first use in the guideline. A list of references and other resources is provided in Chapter 5.
Chapter 2 - Principles of pathogen risk assessment
The pathogen risk assessment closely examines specific risk factors that reflect the inherent characteristics of a pathogen that contribute to the risk it poses to humans and animals. The outcome of a pathogen risk assessment is the risk group, which helps determine the minimum physical containment requirements, operational practice requirements, and performance and verification testing requirements for the safe handling and storing of the pathogen. To ensure the integrity of the pathogen risk assessment, it is essential to identify and disclose knowledge gaps and any assumptions that were used in the assessment. In addition, all decisions and factors taken into account when making those decisions need to be documented in order to maintain transparency in the pathogen risk assessment process.
2.1 Quantitative versus qualitative risk assessment
Quantitative risk assessment involves assigning fixed numerical values to probability and consequence. Quantitative risk assessment has high requirements for data and a comprehensive understanding of how probability and consequence relate to overall risk. A quantitative approach is often used to address things like the safe level of exposure to a chemical or the safe dosage for a drug, within a certain margin of error.
Qualitative risk assessment uses descriptive language or interval scales to rate probability and consequences, and may apply a mathematical or matrix style relationship to determine overall risk. Qualitative risk assessment is useful for screening a number of risks to determine whether they require further analysis or for categorizing risks into discreet groups (e.g., risk groups). Unlike quantitative risk assessments, qualitative risk assessments are possible even when there are significant data gaps. Pathogen risk assessments are generally qualitative because they are typically based on published literature, where information may be variable or even contradictory, and there are often significant data gaps. Furthermore, because the outcome of the pathogen risk assessment is one of four discrete risk group values (i.e., RG1 to RG4), a qualitative approach is suitable.
2.2 Acceptable risk
Zero risk is generally recognized as an unachievable goal and, thus, many risk assessment models incorporate the concept of acceptable risk (also known as tolerable risk). Acceptable risk is related to the willingness of a person, organization, or society to accept or avoid risk, and may vary depending on the circumstances. Acceptable risk is determined by weighing the potentially adverse outcomes against factors that determine whether individuals or groups within society are willing to be subjected to the risk. For example, a risk might be deemed "acceptable" if it is outweighed by a significant benefit, or if the cost of reducing the risk would be excessive when compared to the benefit. In contrast, a risk might be deemed "unacceptable" if the risks outweighed the benefit, or if the risk was excessively high.
The Public Health Agency of Canada (PHAC) has incorporated the concept of acceptable risk into the pathogen risk assessment methodology, including in the criteria for rating each risk factor, and the decision tree for how these criteria determine the risk group.Footnote 7 In addition to determining a pathogen's risk group, the PHAC considers scenario-specific factors on a case-by-case basis. For example, during an outbreak the PHAC may issue a Biosafety Advisory that permits certain lower-risk activities to be performed under lower containment requirements, even though the pathogen's risk group does not change (e.g., diagnostic activities with an RG3 pathogen may be performed at CL2 with specific additional requirements). This may lead to more rapid diagnosis, which would ultimately benefit the overall public and/or animal health response.
2.3 Evidence-based risk assessment
Wherever possible, pathogen risk assessments are based on a rigorous evaluation of information that is empirical and current. The pathogen risk assessment seeks to maximize objectivity through use of reasonably obtainable data, established methodology, and the systematic and defensible application of expert judgement. Quantitative information should be used when available, and qualitative information should be obtained from trusted, peer-reviewed sources, such as respected literature sources (e.g., with a high citation index in the field) and other domestic or international jurisdictions or experts. Every pathogen risk assessment must include a full citation of all sources used; this is foundational for demonstrating the scientific integrity of the assessment.
2.3.1 Data quality
The quantity and quality of information available about a pathogen will impact the confidence in the decisions made during the pathogen risk assessment. High quality data includes information obtained from clinical trials and standardized studies (e.g., evidence from systematic reviews of randomized control trials and meta-analyses). Lower quality data includes expert opinion, and non-referenced literature, such as web sources or independent communications. Risk assessments should be routinely reviewed as new information becomes available, especially if the data quality is low. Appropriate attribution of the sources (i.e., references) will help when evaluating the quality and sufficiency of the data used in the risk assessment.
Based on the types of data sources used and the sufficiency of the information available to conduct a pathogen risk assessment, data quality (i.e., confidence level of the information) may be estimated to be:
- High – sufficient for a thorough analysis of all elements of the risk assessment.
- Medium – sufficient for a thorough analysis of some elements of the risk assessment; some data gaps; minor assumptions made.
- Low – insufficient for a thorough risk assessment; major data gaps; major assumptions made.
2.4 Uncertainty and assumptions
In the course of a pathogen risk assessment, it is inevitable that the presence of uncertainties will necessitate assumptions to be made. Pathogen risk assessments should include a clear outline of knowledge gaps and uncertainties, and of the assumptions made to bridge these gaps.
2.5 Precautionary principle
The application of the precautionary principle in science-based decision making for the purposes of protecting human or animal health means, simply, that a lack of scientific certainty must not be used to justify a delay in decision-making if there is a risk of serious or irreversible harm.Footnote 8
In terms of pathogen risk assessment, the application of the precautionary principle means that a risk group decision must always be made, even when there is little-to-no information to support a full assessment of all risk factors. In practical terms, this may require the use of surrogate data or indirect evidence to support conclusions. In addition, a clear understanding of one's risk tolerance may impact how the precautionary principle is applied, as a lower risk tolerance could lead to the application of more stringent controls.
Chapter 3 - Components of a pathogen risk assessment
All risk assessments essentially follow the same systematic approach that includes the following four components, updated to reflect the pathogen risk assessment:
Hazard identification:
Identification of the microorganism, protein, or nucleic acid capable of causing adverse human or animal health effects. This may include the historical background, physical characteristics, genome structure, taxonomy, and other identifying characteristics.
Hazard characterization:
A qualitative or quantitative evaluation of the nature of the adverse human or animal health effects associated with the microorganism, protein, or nucleic acid. Infectious agents can be categorized into a series of clearly defined risk groups.
Exposure assessment:
A qualitative or quantitative evaluation of the likelihood of exposure to the infectious substance from direct use, including dose-response assessment where possible. For pathogen risk assessments, the exposure assessment is directly related to the containment zone (e.g., laboratory, animal room, and animal cubicle) environment and examines the relationship between consequence and likelihood of exposure, which are the foundation for determining the appropriate containment requirements.
Review and continual improvement:
The pathogen risk assessment should be regularly reviewed to identify new information regarding the pathogen or mitigation measures that may affect the outcome of the assessment (e.g., new host, pathogen evolution, availability of new treatment). It should also be reviewed whenever the pathogen is modified (e.g., by genetic manipulation) to determine the impact of the modification.
3.1 Hazard identification
In a pathogen risk assessment, the hazard is the biological agent under investigation. The assessment starts with the pathogen's taxonomy and accepted name, which can be used to determine whether there are specific legal requirements associated with its use. For example, work involving zoonotic pathogens that also cause foreign animal diseases require approval from both the PHAC and the Canadian Food Inspection Agency (CFIA).
A brief description of the pathogen should highlight the physical characteristics that might be relevant to the interpretation of the risk assessment or overall risk. Potentially relevant factors include:
General information
- Taxonomy and a brief description of subgroupings, previous nomenclature, and changes;
- Historical background;
- Size, shape, and structure;
- Ideal growth conditions;
- Genome structure/information; and
- Modifications (e.g., CRISPR; gene drives).
Bacteria
- Motility;
- Sporulation;
- Toxin production;
- Oxygen requirements;
- Gram staining, acid fast (AF) staining; and
- Enzymatic activity.
Viruses
- RNA/DNA genome;
- single/double stranded;
- positive-sense or negative-sense; and
- other classifications (e.g., arboviruses).
Other (e.g., fungi, prions, parasites)
- Life cycle;
- Reproduction;
- Morphology;
- Growth and physiology; and
- Toxin production.
Reconstructed, engineered, or modified pathogens can be assessed by comparing the newly created pathogen to the wild type or previously assessed variant. This way, the various modifications can be linked to anticipated effects on the different risk factors (e.g., pathogenicity, communicability). The assessment of a modified or engineered system should consider the risks posed by the wild-type pathogen, the risks of the inserted material, and the combined risks of the product.
3.1.1 Biosecurity considerations
Biosecurity refers to security measures designed to prevent unauthorized access to pathogens, toxins, and other related assets (e.g., personnel, equipment, non-infectious material, and animals), or their loss, theft, misuse, diversion, or unauthorized release. Certain pathogens and toxins pose increased biosecurity risks due to their potential for use as a biological weapon. In Canada, these are referred to as security sensitive biological agents (SSBAs). SSBAs are identified as prescribed human pathogens and toxins by Section 10 of the HPTR.Footnote 2 However, even if the pathogen is not an SSBA, it should be noted if it appears on other lists of biological agents of potential concern for biosecurity. These include the Australia Group Common Controls List and the United States Select Agents and Toxins List.Footnote 9,Footnote 10
3.2 Hazard characterization
Hazard characterization involves an analysis of the four key risk factors that are the critical determinants of the risk group. The risk factors detail inherent characteristics of the pathogen that contribute to the risk to the individual and/or community. These factors may vary between humans and animals. For example, while the availability of an effective vaccine may dramatically impact the risk to the human population, routine vaccination is not common amongst animals and, thus, is not a principle risk factor for animals. Table 3-1 provides an overview of the individual risk factors.
| Risk Factor | Humans | Animals |
|---|---|---|
| Pathogenicity | Individual risk | Individual risk |
| Pre- and post-exposure measures | Community risk | Risk factor not applied |
| Communicability | Community risk | Community risk |
| Impact on the animal population (i.e., host range, natural distribution, and economic impact) | Risk factor not applied | Community risk |
Each risk factor is evaluated by answering a series of questions and documenting the supporting data and associated references. These are the indicators of the risk factor. For example, indicators of pathogenicity are (1) whether the pathogen can produce infection, (2) whether the pathogen can produce acute disease, (3) whether the pathogen can cause serious sequelae or mortality, and (4) whether there are specific at-risk populations. These indicator questions support the overall assessment of the risk factor (e.g., low, moderate, or high pathogenicity). In the Pathogen Risk Assessment Template available online, the indicator questions are in white boxes and the overall risk rating is in a blue box.Footnote 7 The overall risk ratings are used to determine the risk group using the decision tree at the end of the template.
3.2.1 Pathogenicity
The level of risk associated with pathogenicity is the strongest determinant of risk group. For the purposes of the pathogen risk assessment, pathogenicity is defined as the proportion of those exposed who experience significant morbidity or mortality. Pathogenicity is composed of two separate elements: (1) infectivity, or the proportion of those exposed who become infected, with or without disease, and (2) virulence, or the proportion of those infected who experience significant morbidity or mortality. Some of the common terminology used to reflect pathogenicity is described below.
When assessing pathogenicity indicators in animals, it is important to consider the natural rather than experimental animal host(s), or the host in which the pathogen is commonly found. Information relating to infection of uncommon hosts and, to a lesser extent, experimentally infected hosts should be reviewed critically to determine whether the information reflects what may occur in nature. Section 3 of the Pathogen Risk Assessment Template illustrates how the pathogenicity risk factor indicators and overall risk rating are tabulated.Footnote 7
3.2.1.1 Infection
Infection refers to the situation where an organism (e.g., bacteria) grows and sustains itself within another organism (e.g., humans). Infection may or may not be associated with signs of disease (i.e., illness). For example, the rate of infection for commensal bacteria is extremely high, yet many are completely harmless to their hosts. Exposure to the organism, along with its ability to infect and subsequently cause disease, are critical considerations when assessing pathogenicity. For example, a few cases of illness for a ubiquitous commensal bacterium (i.e., high exposure, low infection) may be evidence of low pathogenicity; whereas, a few cases of illness for a very rare pathogen (i.e., low exposure, low infection) may be evidence of moderate or high pathogenicity.
3.2.1.2 Mortality
Mortality simply means death. Mortality rate is the number of deaths in a given population during a specified period (=deaths/population/time). To fully assess mortality rate, it is important to consider the incidence (=new cases/population/time), prevalence (=cases/population/time), attack rate (=infected/exposed), death rate (=deaths/infected), and population-specific mortality (e.g., young/old, pregnant, elderly, severely ill, or immunocompromised). Population-specific mortality is sometimes expressed as case fatality rate (i.e., deaths in a given population of cases, such as those with a comorbid condition) and in some cases, mortality is exclusively associated with at-risk populations, such as the severely ill or immunocompromised.
3.2.1.3 Morbidity
Morbidity is the condition of being diseased. Many of the terms used to understand mortality can also be used to evaluate morbidity. The pathogen risk assessment separates signs of disease into two categories: (a) acute (i.e., immediate/short-term impacts), and (b) chronic (i.e., sequelae/long-term impacts), that are presented below.
Acute impact
Immediate impact refers to acute conditions, or the signs and symptoms of disease that appear in the short term (e.g., within days/weeks). Low immediate impact means there are minimal signs of disease; those affected show symptoms, but are able to function and symptoms may resolve on their own (e.g., cough, sore throat, low-grade fever). Moderate immediate impact means there are clear signs of disease; those affected are able to function in a limited manner (e.g., may require bed rest, missed work). High immediate impact means there are significant signs of disease; those affected are not able to function (e.g., may require hospitalization, extended periods of missed work or, in extreme cases, mechanical assistance may be required or death may be imminent). In some cases, immediate impact is exclusively associated with at-risk populations, such as the severely ill or immunocompromised.
Chronic impact
Long-term impact refers to the signs and symptoms of disease that either appear in or persist for the long-term (e.g., months/years). Low long-term impact means there are mild long-term signs or symptoms that do not impede the hosts ability to function normally (e.g., cold sores, mild scarring). Moderate long-term impact means there are signs and symptoms that impede the host's ability to function normally (e.g., some degree of immobility, severe migraines, memory loss). High long-term impact means there are long-term signs or symptoms that render the host unable to function normally (e.g., irreversible liver or kidney damage, loss of one of the senses, cancer). In some cases, long-term impact is exclusively associated with at-risk populations, such as the severely ill or immunocompromised.
3.2.1.4 Overall risk rating
The overall risk rating for pathogenicity is based on the severity of disease in the host. "None" refers to biological agents that are determined to be non-pathogenic. In this case, no other risk factors need be assessed, and the risk assessment outcome will be RG1. "Low" refers to opportunistic pathogens that have rarely been associated with disease in healthy individuals or animals as a result of exceptional circumstances (e.g., introduction of a commensal gut bacterium into the blood stream as a result of a penetrating injury). "Low" may also refer to pathogens that exclusively cause disease in severely ill or immunocompromised humans and animals. "Moderate" refers to pathogens that are able to cause serious disease, but are unlikely to do so, whereas, "High" refers to pathogens that are likely to cause serious disease.
3.2.1.5 Assessing pathogenicity with surrogate data
Since pathogenicity is the most important determinant of the risk group, it must be estimated using surrogate data if there is insufficient information for its evaluation. This may include evidence from animals, genetically related pathogens, and indirect evidence based on exposure. There are two common scenarios that arise where it is unclear whether a biological agent is non-pathogenic or simply has not had the opportunity to produce disease in susceptible hosts:
- A microorganism is identified in an environment in which humans and/or animals are highly likely to have been exposed (e.g., fungi found on plants consumed by humans and animals); or,
- A microorganism is identified in an environment where human and/or animal exposure is unlikely (e.g., lab adapted strains of pathogens exclusively used under containment, or environmental isolates from remote areas).
In the first case, the absence of cases of disease coupled with likely exposure can be used as indirect evidence of non-pathogenicity. Whereas in the second case, the possibility that the microorganism could cause disease but that susceptible hosts simply have never been exposed cannot be ruled out, and pathogenicity cannot be estimated to be "Low". When estimating rather than assessing pathogenicity, it is critical to also outline the uncertainties and assumptions made. This will facilitate routine review as new information becomes available.
3.2.2 Pre- and post-exposure measures
The availability of effective preventive and therapeutic treatments may reduce the risks posed by a pathogen to the individual and the community. Pre-exposure measures refer to medical interventions given before exposure occurs for the purposes of preventing or lessening the impact of infection and disease (e.g., pre-exposure prophylaxis or vaccines). Post-exposure measures refer to medical interventions given after exposure for the purposes of preventing or lessening the impact of infection/disease (e.g., post-exposure prophylaxis, therapeutic treatments). The pre- and post-exposure measures risk factor is most relevant for assessing the human community risk, as widespread medical intervention to prevent or treat infectious diseases is not common in the animal population. Although there is no overall risk rating for protection of the animal population, information about the availability of effective pre- and post-exposure measures should be noted, and may be valuable for the LRA. Section 4 of the Pathogen Risk Assessment Template illustrates how pre- and post-exposure measures are tabulated to assess community protection.Footnote 7
3.2.2.1 Herd immunity
Most medical interventions vary in efficacy and availability, leaving the community only partially protected. In theory, if a preventative measure were completely effective and universally applied, the overall risk to the population would be significantly reduced or even eliminated. To determine whether the community would be completely protected if the pathogen were introduced into the population, the simple threshold concept of herd immunity can be applied.
A completely effective preventive measure would be almost 100% effective at preventing infection in previously immunized (i.e., vaccinated) individuals, even if multiple immunizations were required to achieve this level of protection. A universally applied preventive measure would be administered at least to the percentage of the population needed to achieve herd immunity. See the polio example in the text box below.Footnote 11
The simple threshold concept of herd immunity, by which a population can be protected based on the number of immune individuals, can be calculated as the critical vaccination level, Vc, where:Footnote 12
Vc = (1-1/R0)/E, where R0 is the basic reproduction number, and E is the vaccine effectiveness against transmission. The basic reproduction number is the number of secondary cases generated by a typical infectious individual when the rest of the population is susceptible (i.e. at the start of novel outbreak).
The Polio Example
An example of a disease that could be considered "completely preventable" is polio. The Canadian polio vaccination rate in 2013 was 91% of appropriately aged children receiving the recommended 4 vaccine doses.Footnote 11 The vaccine effectiveness (E) is 95% after 3 doses, and 100% after 4 doses. The basic reproduction number (R0) is estimated at between 4 and 7.
A conservative calculation of herd immunity is the following:
Vc = (1 - 1/7)/0.95
= 0.90 or 90%
Herd immunity is 90%. Since 91% of the population was vaccinated, herd immunity has been achieved.
Since vaccination rates fluctuate over time, waning herd immunity may require this risk factor to be re-evaluated periodically to determine whether polio still meets these criteria.
3.2.3 Communicability
Communicability is the most important community risk factor. Communicability not only impacts the risk group of the microorganism, but also many of the physical and operational requirements for handling it within the facility. It involves examining the routes of infection, or how the pathogen gains entry into the host (e.g., inhalation, ingestion, injection, mucous membrane contact), in order to determine the likelihood of direct or indirect transmission. Whether the pathogen can transmit between humans and animals is also evaluated, although human-to-human and animal-to-animal transmission are likely to have the greatest impact on the human or animal community risk, respectively.
The primary route through which the pathogen naturally transmits between individuals is referred to as the "preferred" route. In the pathogen risk assessment, only one "preferred" route of infection should be selected, but a pathogen may have many other "possible" routes. The ease of transmission via the different routes of infection must be analyzed in combination to determine the likelihood of the agent transmitting between hosts via direct or indirect transmission. For example, if the only (i.e., preferred) route of infection is injection, there would likely be little community risk. However, the risk to the community could be higher if it was possible for the pathogen to also be transmitted via vectors or inhalation. This risk factor refers specifically to human-to-human and animal-to-animal transmission, rather than transmission between humans and animals, or from the environment. Zoonosis and infection from environmental sources should be noted and considered when determining effective containment, but are not included in the communicability risk factor output.
New and emerging pathogens may pose unique risks to the community and to pandemic preparedness and response. For example, a highly transmissible emerging pathogen with pandemic potential could have a significant impact on pandemic preparedness and response. If these pathogens were released into the community, there would be an expectation of a high case count, high health care burden, and significant costs associated with medical intervention and health impacts. This indicator must be revisited routinely, particularly if the pathogen becomes established in the population, since the risks before or during the early stages of an outbreak of an emerging pathogen are considerably different from the risks associated with an established pathogen, as show in the following example.Footnote 13
The Influenza Example
When the 2009 Influenza A/H1N1 virus first emerged, it had an immediate and significant impact on human health and the economy. It spread rapidly from where it originated, causing a global pandemic and costing billions of dollars in health care response. Although the novel virus generally caused flu symptoms similar to seasonal influenza, younger aged groups tended to have more severe symptoms.Footnote 13 As such, it was initially assessed as an RG3 pathogen. Once this strain became established in the population, and the pandemic strain was incorporated into seasonal flu vaccine campaigns around the globe, the impact diminished and A/H1N1 (2009) was re-assessed as an RG2 pathogen.
The PHAC or the CFIA should be informed of any emerging pathogen that have not already been assessed by them, as further communication may be needed. If an emerging pathogen is identified and not on the Biosecurity Portal pathogen list, the PHAC can be notified by email at PHAC.pathogens-pathogenes.ASPC@canada.ca.
3.2.4 Impact on the animal population (i.e., host range, natural distributions, and economic impact)
Introducing a pathogen into the animal population could have a significant impact on animal health and the economy. Host range, natural distribution, and economic impact are indicators that relate to the impact of releasing a pathogen into the animal population.
3.2.4.1 Host range
Host range is the number and variety of hosts that a pathogen can infect. Only natural hosts should be included in the species count. Factors that increase the host range include transmission strategies that increase contact with new hosts (e.g., vectors), high genetic variability, and rapid replication. A pathogen restricted to several closely related host species may not be as adaptable as a pathogen able to infect species in multiple distantly related taxonomic families or orders. A pathogen able to infect distantly related host species will require diverse mechanisms for gaining entry into the host and producing pathogenic effect; whereas a pathogen with a high degree of host specificity may have very specialized mechanisms of infection. Francisella tularensis, for example, is considered to have one of the broadest host ranges among bacterial pathogens, and is found in more than 300 species, including mammals, invertebrates, birds, and amphibians.Footnote 14 Many viruses are able to infect host species in multiple orders, and this is strongly correlated with the use of vectors as a mode of transmission.Footnote 15 Conversely, viruses transmitted primarily by close contact are more likely to be restricted to a few specific species for which this contact can occur.
3.2.4.2 Natural distribution
Natural distribution of a pathogen is important in determining the impact on the animal community if it were released from containment. The release of a pathogen that is not present in Canada and whose host species are present in Canada would pose a significantly greater risk to the population than a pathogen already circulating in Canada.
Natural distribution, or endemicity, considers whether a pathogen is already in circulation (i.e., natural transmission) in a particular country, region, or human or animal population. The affected population will usually have some level of resistance or immunity to endemic pathogens. Should a non-endemic pathogen enter into an area or population where it has not established itself, it would have the potential to enter a new (i.e., naïve) host population and pose a serious risk if the new host provided favourable conditions for the reproduction and transmission of the pathogen.Footnote 7
Of course, endemic pathogens can also cause serious outbreaks in livestock. They can be transmitted through various modes, so it is important to characterize the frequency of contact and methods through which they can be transmitted (e.g., direct contact between live animals, or indirect contact from transport vehicles, deadstock collectors, veterinarians, and animal technicians) in order to determine the level of risk associated with the release of the pathogen.Footnote 16
In addition, a change in the environment that is favourable to an endemic pathogen (e.g., that allows it to increase its host range or pathogenicity) can lead to the emergence of new infectious diseases that result from a change in the immunological, ecological, and/or the behavioural parameters of the host or pathogen.Footnote 17
3.2.4.3 Economic impact
Within Canada, the infection of certain animal populations would result in greater economic consequences. The CFIA has classified animals in terms of their economic value to Canada as follows:
- Highest value livestock industries: bovine, equine, porcine, poultry, crustaceans, and wild and farmed finfish.
- Medium value livestock industries: small ruminants (e.g., sheep and goats), bees, molluscs, and other farmed ruminants (e.g., cervids, bison).
- Lowest value livestock industries and non-livestock animals: lagomorphs (e.g., rabbits), companion animals (e.g., dogs, cats), reptiles, amphibians, rodents, and primates.
Note: Diseases that are of significant importance to animal health or the Canadian economy are usually designated foreign animal diseases or reportable diseases (i.e., infection with a non-indigenous animal pathogen), and may be under the authority of the CFIA.
3.2.5 Risk group classification
With the assessments of the risk factors discussed above (i.e., pathogenicity, pre- and post-exposure measures, communicability, impact on the animal population), the overall risk group of the organism can be determined using the Risk Group Decision Trees for humans and animals found in Section 8 of the Pathogen Risk Assessment Template.Footnote 7
3.3 Exposure assessment
In the context of biosafety, the exposure assessment takes place: (1) when establishing the minimum physical and operational containment requirements, which are outlined in the CBS; and (2) when performing LRAs. Both aim to prevent exposure of those working with pathogens and release of the pathogen from the containment zone.
3.3.1 The classical exposure assessment
The classical health risk assessment typically involves an assessment of exposure at the population level and may involve dose-response modeling, data monitoring, models, and estimates or data about persistence and accumulation of a substance. These types of exposure assessments are instrumental in many health risk assessments, especially when determining the "safe" level of exposure to a particular substance. In the case of a pathogen risk assessment, classical exposure-response relationships typically do not exist, for the following reasons:
- The "safe" level of exposure to a pathogen is a level below the infectious dose. Often, a pathogen's infectious dose is not known, and for pathogens, the impact of exposure is generally "all or none". Either infection occurs or it does not.
- The endpoint of a pathogen risk assessment is the determination of the risk group, which helps determine the minimum containment requirements for safely handling and storing the pathogen. This limits the utility of dose-response analysis, as the purpose of the pathogen risk assessment is to prevent exposure in the containment zone, based on the risks posed by the pathogen, using specific physical and operational controls.
- While there is an array of harmonized testing guidelines for assessing the toxic effects of chemicals (e.g., those available from the Organization for Economic Cooperation and Development and Office of Pollution Prevention and Toxics), standardized studies on the pathogenic effects of organisms are generally lacking. There are few harmonized guidelines for assessing pathogens, and those that do exist do not capture the full spectrum of potential pathogenic effects.
3.3.2 Incorporating the concepts of exposure in the risk assessment process
Exposure assessment occurs not only when establishing the minimum physical and operational containment requirements, but also when performing LRAs. The risk group of the pathogen helps determine the containment level at which it should be handled; however, the LRA is critical for further characterizing and mitigating risks that are specific to a given activity. In this way the exposure assessment for a pathogen is reflected in the containment level and associated requirements set out in the CBS, as well as LRAs performed within the facility.
In some cases, the PHAC or the CFIA may pro-actively reduce the containment requirements for a given pathogen. For example, a pathogen that is able to cause serious human disease and meets the definition of an RG3 pathogen, but is poorly transmissible or not airborne, may be safely handled with lower physical containment requirements, but with additional operational requirements. The PHAC has developed Biosafety Directives for specific pathogens to outline such exceptions and any additional biosafety requirements. A regulated party who determines, through an LRA, that certain activities with a pathogen can be safely performed at a lower containment level must receive approval by the PHAC or the CFIA prior to making the change.
The majority of the operational practice requirements and some of the physical containment requirements in the CBS are dependent on the activities being performed or the nature of the pathogens in use (i.e., based on an LRA). In this way, specific exposure scenarios can be considered that warrant specific physical or operational controls to be used to prevent exposure and release (e.g., the use of a biological safety cabinet [BSC] when working with pathogens transmissible via the airborne route or for procedures that may generate infectious aerosols).
Activities within a containment zone can be largely divided into in vivo, which represents the greater risk, and in vitro activities; each of these presents different risks, such as the added risk of bites and scratches, and pathogens shedding, when working with animals. In vitro activities can be further subdivided into propagative (e.g., culturing) and non-propagative (e.g., extraction of DNA) activities. Non-propagative in vitro activities pose the lowest risk and are the activities that are the most likely to be permitted to be performed with reduced containment requirements.
Should the physical containment requirements for some or all activities with a pathogen be reduced or modified, the information would be communicated in the form of a Biosafety Directive or Biosafety Advisory, available on the Government of Canada website.Footnote 18
The human immunodeficiency virus (HIV) example
HIV is capable of causing serious and, ultimately, fatal disease in humans. It is classified as an RG3 human pathogen. HIV is a bloodborne pathogen that can be transmitted by exposure of mucous membranes through intimate contact. It is not airborne and does not survive very long outside of the host. The most likely scenario for exposure to HIV in a facility is through accidental percutaneous exposure (e.g., needle stick injury). Given that the added physical and engineering controls present at Containment Level 3 (CL3) do not provide added protection against HIV, a Biosafety Directive has been developed to outline how HIV can be safely handled at CL2.Footnote 18
3.4 Other considerations
3.4.1 International classification
A good practice for a pathogen risk assessment is to evaluate how the outcome aligns with the international landscape. While there is some variability in risk group ratings of pathogens by international governing bodies, they often align. If the risk group determination differs significantly internationally, it may be worth revisiting the assessment to determine the reason. In many cases, the risk group designation may reflect the local jurisdiction in which the assessment is performed. For example, a country where a specific pathogen is not endemic may classify the pathogen in a higher risk group, than a country where the pathogen is endemic. Similarly, if the vector for a particular pathogen is not present or cannot survive in Canada, the risk may be lower than in countries with a more permissible climate.
3.5 Review and continual improvement
Although a specific time frame does not exist for when a pathogen risk assessment should be reviewed, it is highly recommended that they be reviewed regularly, since a pathogen risk assessment is a continuously evolving process.
The pathogen risk assessment must be reviewed and updated whenever:
- new information becomes available;
- the pathogen or community has changed (e.g., natural attenuation, lab attenuation, herd immunity, vaccination, genetic alteration);
- the conditions of use have changed (e.g., new animal model, first use of a lab strain in an animal);
- a new vector appears in a region; or
- a disease becomes rare or eradicated.
Any of these scenarios has the potential to change the risk group, the containment requirements, the LRA, or all three.
Chapter 4 - Glossary
The following list is an excerpt from the CBH pertaining to terms referenced in this guideline. It is important to note that while some of the definitions provided in the glossary are universally accepted, many of them were developed specifically for the CBS and the CBH; therefore, some definitions may not be applicable to facilities that fall outside of the scope of the CBS and CBH. A comprehensive list of terms and their definitions can be found in the glossary of the CBH.
- Animal pathogen
- Any pathogen that causes disease in animals; including those derived from biotechnology. In the context of the Canadian Biosafety Standard, "animal pathogen" refers only to pathogens that cause disease in terrestrial animals; including those that infect avian and amphibian animals, but excluding those that cause disease in aquatic animals and invertebrates.
- Biosafety
- Containment principles, technologies, and practices that are implemented to prevent unintentional exposure to infectious material and toxins, or their accidental release.
- Biosecurity
- Security measures designed to prevent the loss, theft, misuse, diversion, or intentional release of pathogens, toxins, and other related assets (e.g., personnel, equipment, non-infectious material, and animals).
- Biosecurity risk assessment
- A risk assessment in which pathogens, toxins, infectious material, and other related assets (e.g., equipment, animals, information) in possession are identified and prioritized, the threats and risks associated with these materials are defined, and appropriate mitigation strategies are determined to protect these materials against potential theft, misuse, diversion, or intentional release.
- Community
- Encompasses both human (i.e., the public) and animal populations.
- Containment
- The combination of physical design parameters and operational practices that protect personnel, the immediate work environment, and the community from exposure to biological material. The term "biocontainment" is also used in this context.
- Containment level (CL)
- Minimum physical containment and operational practice requirements for handling infectious material or toxins safely in laboratory, large scale production, and animal work environments. There are four containment levels ranging from a basic laboratory (Containment Level 1 [CL1]) to the highest level of containment (Containment Level 4 [CL4]).
- Containment zone
- A physical area that meets the requirements for a specified containment level. A containment zone can be a single room (e.g., Containment Level 2 [CL2] laboratory), a series of co-located rooms (e.g., several non-adjoining but lockable CL2 laboratory work areas), or it can be comprised of several adjoining rooms (e.g., Containment Level 3 [CL3] suite with dedicated laboratory areas and separate animal rooms, or animal cubicles). Dedicated support areas, including anterooms (with showers and "clean" and "dirty" change areas, where required), are considered to be part of the containment zone.
- Disease
- A disorder of structure or function in a living human or animal, or one of its parts, resulting from infection or intoxication. It is typically manifested by distinguishing signs and symptoms.
- Economic impact
- The financial cost of a release from the containment zone and includes, but is not limited to, costs associated with loss of food crops and livestock, and economic impacts related to trade restrictions.
- Endemic
- Regularly found in a defined geographic location or in a particular human or animal population.
- Exposure
- Contact with, or close proximity to, infectious material or toxins that may result in infection or intoxication, respectively. Routes of exposure include inhalation, ingestion, inoculation, and absorption.
- Facility (plural: facilities)
- Structures or buildings, or defined areas within structures or buildings, where infectious material or toxins are handled or stored. This could include individual research and diagnostic laboratories, large scale production areas, or animal housing zones. A facility could also be a suite or building containing more than one of these areas.
- Hazard
- Any source (thing or situation) with the potential for damage, harm, or adverse health effects to people or other living things. In a pathogen risk assessment, the hazard is a micro-organism, protein, or nucleic acid that can produce infection and lead to disease in humans or animals.
- Herd immunity
- The reduction in the risk of infection among susceptible individuals in a population caused by the proportion of immune individuals.
- Host
- The organism that harbours, is infected with, or is fed upon by another organism. These can include the definitive (i.e., primary) host, intermediate (i.e., secondary) hosts, reservoir host, and dead-end host.
- Host range
- A measure of the number and variety of hosts that a pathogen can infect.
- Laboratory (plural: laboratories)
- An area within a facility or the facility itself where biological material is handled or stored for scientific or medical purposes.
- Local risk assessment (LRA)
- Site-specific risk assessment used to identify hazards based on the infectious materials or toxins in use and the activities being performed. This analysis provides risk mitigation and risk management strategies to be incorporated into the physical containment design and operational practices of the facility.
- Mode of transmission
- The means by which an infectious agent travels to the host (i.e., direct contact, indirect contact, casual contact, contact with aerosolized droplet, airborne, injection, vector borne, and inhalation).
- Natural distribution
- The geographical area within which a pathogen is normally present.
- Non-indigenous animal pathogen
- A pathogen that causes an animal disease listed in the World Organisation for Animal Health's OIE-Listed diseases, infections and infestations (as amended from time to time) and that is exotic to Canada (i.e., foreign animal disease agents that are not present in Canada). These pathogens may have serious negative health effects to the Canadian animal population.
- Operational practice requirements
- Administrative controls and procedures followed in a containment zone to protect personnel, the environment, and ultimately the community, from infectious material or toxins, as outlined in Chapter 4 of the Canadian Biosafety Standard.
- Overall risk rating
- The level of risk assigned to each of the four key risk factors based on indicator questions for a specific pathogen. These are used to determine the risk group.
- Overarching risk assessment
- A broad risk assessment that supports the biosafety program as a whole and may encompass multiple containment zones within an institution or organization. Mitigation and management strategies reflect the type of biosafety program needed to protect personnel from exposure and to prevent the release of pathogens and toxins.
- Pathogen
- A microorganism, nucleic acid, or protein capable of causing disease or infection in humans or animals. Examples of human pathogens are listed in Schedules 2 to 4 and in Part 2 of Schedule 5 of the Human Pathogens and Toxins Act, but these are not exhaustive lists. Examples of animal pathogens can be found on the Canadian Food Inspection Agency website.
- Pathogen risk assessment
- The determination of the risk group and appropriate physical containment and operational practice requirements needed to safely handle the infectious material or toxins in question.
- Pathogenicity
- The ability of a pathogen to cause disease in a human or animal host.
- Physical containment requirements
- Physical barriers in the form of engineering controls and facility design used to protect personnel, the environment, and ultimately the community, from pathogens or toxins, as outlined in Chapter 3 of the Canadian Biosafety Standard.
- Post-exposure measures
- Medical interventions given after exposure for the purposes of preventing infection or lessening the impact of infection or disease (e.g., post-exposure prophylaxis, therapeutic treatments).
- Pre-exposure measures
- Medical interventions given before exposure occurs for the purposes of preventing infection or lessening the impact of infection or disease (e.g., pre-exposure prophylaxis or vaccines).
- Release
- The discharge of infectious materials or toxins from a containment system.
- Risk
- A function of the probability of an undesirable event occurring (e.g., exposure to a pathogen) and the severity of the consequences of that event (e.g., infection, disease, death).
- Risk factor
- A characteristic of a pathogen that relates to the risk it poses to individuals or the public. Four key risk factors are considered in the course of a pathogen risk assessment.
- Risk group
- The classification of biological material based on its inherent characteristics, including pathogenicity, virulence, risk of spread, and availability of effective prophylactic or therapeutic treatments, that describes the risk to the health of individuals and the public as well as the health of animals and the animal population.
- Route of infection
- The path by which an infectious substance enters the host. Route of infection is related to, but distinct from, the mode of transmission, which is the means by which a pathogen travels to the host.
- (Microbial) Toxin
- A poisonous substance that is produced or derived from a microorganism and can lead to adverse health effects in humans or animals. Human toxins are listed in Schedule 1 and Part 1 of Schedule 5 in the Human Pathogens and Toxins Act.
- Vector
- Organism that serves as vehicles for carrying and transmitting an infectious agent from one host to another, and is required for part of the infectious agent's developmental cycle. Most commonly, vectors are arthropods or small mammals.
- Zoonoses (singular: zoonosis)
- Diseases that are transmissible between living animals and humans. Zoonoses include anthropozoonoses (i.e., diseases transmitted from animals to humans) and zooanthroponoses, also known as reverse zoonoses (i.e., diseases transmitted from humans to animals).
Chapter 5 - References and resources
Footnotes
- Footnote 1
Human Pathogens and Toxins Act (S.C. 2009, c. 24). (2015).
- Footnote 2
Human Pathogens and Toxins Regulations (SOR/2015-44). (2015).
- Footnote 3
Health of Animals Act (S.C. 1990, c. 21). (2015).
- Footnote 4
Health of Animals Regulations (C.R.C., c. 296). (2015).
- Footnote 5
Government of Canada. (2015). Canadian Biosafety Standard, 2nd ed. Ottawa, ON, Canada: Government of Canada.
- Footnote 6
Government of Canada. (2016). Canadian Biosafety Handbook, 2nd ed. Ottawa, ON, Canada: Government of Canada.
- Footnote 7
Government of Canada. Pathogen Risk Assessment Template. Available from https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/pathogen-safety-data-sheets-risk-assessment/pathogen-risk-assessment-template.html
- Footnote 8
The Australia Group. List of Human and Animal Pathogens and Toxins for Export Control. Retrieved 08/10, 2017 from http://www.australiagroup.net/en/human_animal_pathogens.html
- Footnote 9
United States Federal Select Agents Program. Select Agents and Toxins List. Retrieved 08/10, 2017 from https://www.selectagents.gov/SelectAgentsandToxinsList.html
- Footnote 10
Government of Canada. (2016). Vaccine coverage in Canadian children: Highlights from the 2013 childhood National Immunization Coverage Survey (cNICS). Retrieved 10/24, 2017 from https://www.canada.ca/en/public-health/services/publications/healthy-living/vaccine-coverage-canadian-children-highlights-2013-childhood-national-immunization-coverage-survey.html
- Footnote 11
Fine, P., Eames K., and Heymann, D.L. (2011). 'Herd Immunity'': A Rough Guide. Clinical Infectious Diseases, 52(7):911–916
- Footnote 12
Van Kerkhove, M., Vandemaele, K., Shinde, V., Jaramillo-Gutierrez, G., Koukounari, A. et al. (2012). Risk factors for severe outcomes following 2009 Influenza A (H1N1) infection: A Global Pooled Analysis. PLOS Medicine, 8(7). Retrieved 10/24, 2017 from http://www.who.int/influenza/surveillance_monitoring/Risk_factors_H1N1.pdf
- Footnote 13
Peterson, J.M., and Molins, C.R. (2010). Subpopulations of Francisella tularensis ssp. Tularensis and holarctica: identification and associated epidemiology. Future Microbiology, 5(4):649-61
- Footnote 14
Pederson, A.B., Altizer, S., Poss, M., Cunningham, A.A., and Nunn, C.L. (2005). Patterns of host specificity and transmission among parasites of wild primates. International Journal of Parasitology, 35(6):647-57
- Footnote 15
Sternberg Lewerin, S., Osterberg, J., Alenius, S., Elvander, M., Fellstrom, C., et al. (2015). Risk assessment as a tool for improving external biosecurity at farm level. BMC Veterinary Research, 11:171
- Footnote 16
Rachowicz, L.J., Hero, J.M., Alford, R.A., Taylor, J.W., Morgan, J.A.T., et al. (2005). The novel and endemic pathogen hypotheses: Competing explanations for the origin of emerging infectious diseases of wildlife. Conservation Biology, 19(5):1441-1448
- Footnote 17
Government of Canada. Biosafety Directives, Advisories and Notifications. Available from https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/biosafety-directives-advisories-notifications.html
- Footnote 18
Government of Canada. Biosafety Directives, Advisories and Notifications. Available from https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/biosafety-directives-advisories-notifications.html