Recommendations for tuberculosis infection screening among returned travellers
Published: May 15, 2026
On this page
- Preamble
- Key points for the healthcare provider
- Traveler risk assessment framework
- Definitions
- Threshold for important absolute benefits and harms
- Recommendations
- Background
- Guideline development methods
- Evidence assessment and recommendations on TB prevention in travellers
- Infection prevention measures
- Bacillus Calmette-Guérin (BCG) vaccine
- Conclusions and research needs
- Abbreviations
- Acknowledgements
- Conflicts of interest
- Appendix 1: Sample search strategy
- Appendix 2: Logic model and model structure
- Appendix 3: Supplemental tables and sample calculations
- Appendix 4: Evidence to decision framework for question 1
- Appendix 5: Assessment of good practice statement
- References
Preamble
The Committee to Advise on Tropical Medicine and Travel (CATMAT) provides the Public Health Agency of Canada (PHAC) with ongoing and timely medical, scientific, and public health advice relating to tropical infectious disease and health risks associated with international travel. The Agency acknowledges that the advice and recommendations set out in this statement are based upon the best current available scientific knowledge and medical practices and is disseminating this document for information purposes to the medical community caring for travellers.
Persons administering or using drugs, vaccines, or other products should also be aware of the contents of the product monograph(s) or other similarly approved standards or instructions for use. Recommendations for use and other information set out herein may differ from that set out in the product monograph(s) or other similarly approved standards or instructions for use by the licensed manufacturer(s). Manufacturers have sought approval and provided evidence as to the safety and efficacy of their products only when used in accordance with the product monographs or other similarly approved standards or instructions for use.
Key points for the healthcare provider
- Tuberculosis (TB) is a respiratory disease transmitted through inhalation of bacilli, which can be cleared, contained in a controlled and dormant “TB infection”, or may progress or reactivate to “TB disease”.
- Canadians may be exposed to TB through living, travelling, or working in a TB-endemic country.
- The risk of TB infection acquired during travel is influenced by the incidence of TB in the country visited, duration of travel, and individual risk activities, such as health care work.
- Canadian travellers born in TB endemic countries carry both the risk of TB exposure from travel as well as TB exposure from living in their country of birth.
- The risk of progression from TB infection to TB disease is influenced by time since infection and underlying medical co-morbidities.
- TB disease can be prevented through detecting TB infection (diagnostic testing with the tuberculin skin test [TST] or interferon-gamma release assays [IGRA]) and providing suitable preventive therapy.
- Diagnostic tests (TST and IGRA) are sensitive and specific for TB infection, but cannot differentiate between infections that will progress to TB disease and those that will not.
- Screening for TB infection among travellers should be targeted to those at highest risk for developing TB disease who, if determined to be infected, would accept treatment and who have not previously tested positive for TB infection (refer to Recommendations).
- We acknowledge that our guidance differs from that for travellers in Chapter 13 of the Canadian Tuberculosis Standards (CTS), 8th edition. This reflects differences in methodologic approaches, evidence sources, judgement and values (Appendix 3, Table 9).
- The objective of this statement is to assess the risk of TB infection and disease among travellers, and provide recommendations for travellers returning from travel to a higher TB incidence country (see Definitions) who would benefit from TB infection screening and preventive treatment. Populations considered are travellers born in or outside of Canada and those providing healthcare while travelling.
Traveller risk assessment framework
To inform estimation of benefits and harms related to TB screening, CATMAT suggests assessing each traveller’s:
- Likelihood of TB infection: risk factors include previous residence (e.g., born outside of Canada) and/or travel to a higher TB incidence country, travel to provide health care, prolonged travel duration.
- Likelihood of developing TB disease if infected: low, moderate, high or very high (Table 1).
Definitions
- Higher TB incidence countries: countries with a higher incidence relative to Canada, with an annual reported TB incidence of more than 10 cases per 100,000 population (Summary of the evidence). This threshold is the same cut-off value used in the systematic review and meta-analysis that was used to estimate travel-associated TB infection likelihoodFootnote 1.
- Non-Canadian-born residents who have immigrated to Canada from a higher TB incidence country are a travelling subpopulation of interest for TB screening and will be a focus in this guideline. This subpopulation is abbreviated as “non-Canadian-born residents” in the remainder of this document. CATMAT chose to use this definition over “visiting friends and relatives (VFR)”, as it more specifically captures the pre-arrival TB exposure risk of this subpopulation.
| Risk group | Risk factors |
|---|---|
| Very high | HIV, close TB contact, silicosis |
| High | Chronic kidney disease, transplant, fibronodular disease, receiving immunosuppressing medications, cancer |
| Moderate | Granuloma on chest x-ray, diabetes, heavy alcohol/ tobacco use |
| Low | General adult population |
Threshold for important absolute benefits and harms
CATMAT decided on an absolute benefit threshold of 1 per 1,000 (0.1%) for screening as a moderate important decrease in developing TB disease.
CATMAT decided on an absolute harm threshold of 1 per 1,000 (0.1%) serious adverse events (SAE) resulting from treatment as a moderate important absolute increase in risk (refer to Outcomes and thresholds).
Recommendations
1. CATMAT suggests that Canadian-born adult travellers returning to Canada after travel in a higher TB incidence country(ies) for 6 consecutive months or more and who are at very high risk of developing TB disease be screened for TB infection after return.
(Discretionary recommendation, low-certainty evidence)
Rationale:
- CATMAT suggests screening because the estimated benefits are moderate and the estimated harms are small. Screening may result in a moderate overall absolute reduction of 0.11% of developing TB disease (low-certainty evidence) compared to absolute likelihood of 0.03% for TB-treatment associated SAE (low-certainty evidence).
- Shorter travel duration and travellers with low to high risk of developing TB disease did not meet the threshold for a moderate benefit.
2. CATMAT suggests that Canadian-born adult healthcare providers returning to Canada after delivering patient care in higher TB incidence country(ies) for 3 consecutive months or more and who are at very high risk of developing TB disease be screened for TB infection after return.
(Discretionary recommendation, moderate-certainty evidence)
Rationale:
- CATMAT suggests screening because the estimated benefits are moderate and the estimated harms are small. Screening may result in a moderate absolute reduction of 0.12% of developing TB disease and with an absolute likelihood of 0.03% for TB-treatment associated SAE (moderate-certainty evidence).
- Shorter travel duration and travellers with low to high risk of developing TB disease did not meet the threshold for a moderate benefit.
3. CATMAT suggests that non-Canadian-born resident adult travellers (see Definitions), including healthcare workers, who are returning to Canada after travel in a higher TB incidence country(ies) for any duration of travel and who are at high to very high risk of developing TB disease be screened for TB infection after return.
(Discretionary recommendation, low-certainty evidence)
Rationale:
- CATMAT suggests screening for high or very high-risk populations because the estimated benefit is moderate to large (absolute reduction of 0.38-2.29% of developing TB disease) and is substantially more than the estimated likelihood for TB-treatment associated SAE (0.20%), which is also above our threshold for a moderate effect (low-certainty evidence).
- In making this recommendation, CATMAT placed higher value on the relatively larger estimates of benefit compared to harm (TB-treatment associated SAE).
Remarks:
Among all travellers
- People who meet criteria for TB infection screening—based on the CTS or local guidelines, and not solely due to travel-related exposure—should be assessed and managed according to those recommendations.
- Healthcare providers should consider individual likelihood of TB infection and risk of developing TB disease if infected in their decision to screen (refer to Figure 1, Figure 2, and Implementation considerations).
- A single post-travel test using TST, done 8 weeks or more after return is suggested.
- Individuals who screen positive should be assessed for active disease and referred to a TB specialist for preventive treatment of TB infection as outlined in the CTS (Chapter 6)
- These CATMAT recommendations do not apply to individuals who have previously screened positive and have been treated for TB infection. The risk of reinfection and recommendations for clinical care in these scenarios are outside the scope of this guidance.
Among travelling non-Canadian-born residents (see Definitions)
- In general, if documentation exists of a previous negative TB screening test since taking residence in Canada, the suggested approach to TB screening would follow the same as those for Canadian-born travellers.
- The risk of TB infection among non-Canadian-born residents is primarily associated with TB exposure from having lived in a higher TB incidence country.
- For individuals at moderate risk of developing TB disease, CATMAT emphasizes a need for discussion and shared decision making between the healthcare provider and patient regarding potential benefits and harms of TB infection screening and treatment (refer to Figure 2 and Implementation considerations). This is because estimates of benefits and harms are relatively balanced.
4. Healthcare providers should counsel travellers on TB infection prevention measures. (Good practice statement)
- Should travellers wish to undertake infection prevention measures, healthcare providers should advise them to consider wearing a N95 respirator if they will be in a high-risk setting, and/or to avoid these settings (e.g., crowded, poorly ventilated indoor environments).

Figure 1: Descriptive text
This image is a decision aid for the management of Canadian-born travellers who may be at risk of development of TB disease. The first consideration is whether the traveller has been previously screened for TB infection.
- If the traveller has previously screened positive and was treated, then screening for TB infection post-travel is not recommended.
- If the traveller has previously screened positive but was not treated, consider treatment as per Chapter 6 of the CTS.
If the traveller previously screened negative, or was not previously screened, then the second consideration is whether the traveller is returning from a TB endemic area with more than 10 cases per 100,000 population. Note, the risk of infection in travellers increases with increasing incidence of TB in the destination of travel. TB incidence estimates are available from the WHO.
- If not, then screening for TB infection post-travel is not routinely suggested.
- If yes, then consider whether the purpose of travel is for general travel (such as tourism) or to deliver patient care.
For Canadians travelling for the purpose of general travel, screening for TB infection post-travel is suggested only for individuals who are at very high risk of developing TB disease should they contract TB infection (for example, those living with HIV, silicosis, or who have had close TB contact), and who are travelling for at least 6 consecutive months.
For Canadians travelling for the purpose of delivering patient care, screening for TB infection post-travel is suggested only for individuals who are at very high risk of developing TB disease should they contract TB infection (for example, those living with HIV, silicosis, or who have had close TB contact), and who are travelling for at least 3 consecutive months.
A single post-travel test using TST, done 8-weeks or more after the traveller’s return is the suggested screening strategy. Individuals who screen positive should be assessed for active disease and referred to a TB specialist for preventive treatment of TB infection. Refer to Implementation considerations.
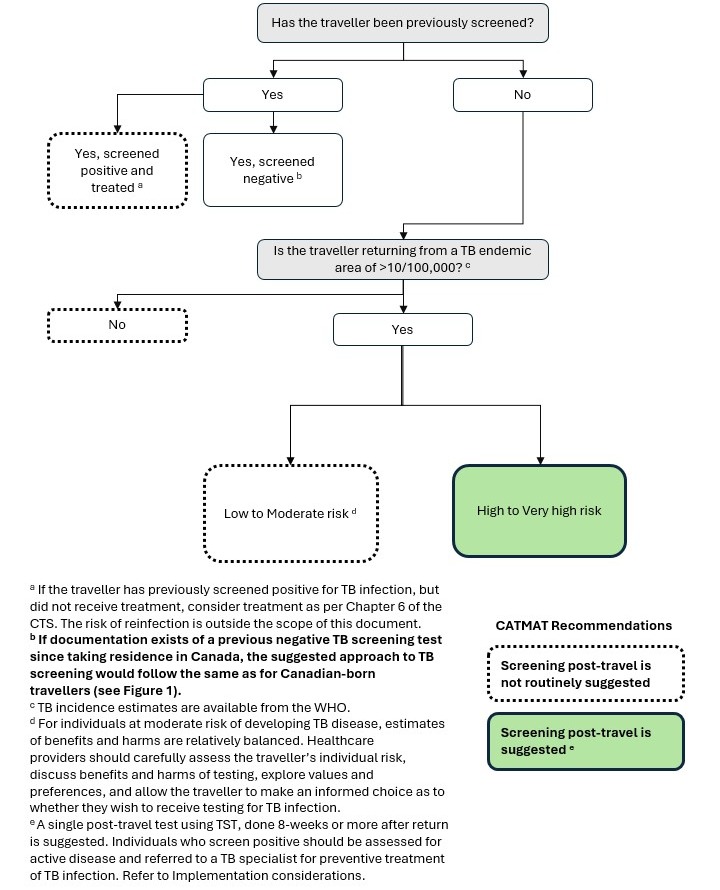
Figure 2: Descriptive text
This image is a decision aid for the management of non-Canadian-born resident travellers (see Definitions) who may be at risk of development of TB disease. The first consideration is whether the traveller has been previously screened for TB infection.
- If the traveller has previously screened positive and was treated, then screening for TB infection post-travel is not recommended.
- If the traveller has previously screened positive but was not treated, consider treatment as per Chapter 6 of the CTS.
If the traveller previously screened negative since taking residence in Canada, the suggested approach to TB screening would follow the same as for Canadian-born travellers outlined in Figure 1.
If the traveller was not previously screened, then the second consideration is whether the traveller is returning from a TB endemic area with more than 10 cases per 100,000 population. Note, TB incidence estimates are available from the WHO.
- If not, then screening for TB infection post-travel is not routinely suggested.
- If yes, then consider the traveller’s individual risk of developing TB disease should they become infected.
For travellers at high or very high risk of developing TB disease (for example those with kidney disease or fibronodular disease, transplant patients, those taking immunosuppressants, or those living with cancer, HIV, or silicosis, or who have had close TB contact), screening for TB infection post-travel is suggested.
For travellers at low or moderate risk of developing TB disease (for example, the general adult population or those living with diabetes, heavy alcohol or tobacco use, or a granuloma on a chest x-ray), screening post-travel is not routinely suggested. Estimates of benefits and harms are relatively balanced among travellers at moderate risk of developing TB disease. Healthcare providers should carefully assess the traveller’s individual risk, discuss benefits and harms of testing, explore values and preferences, and provide the traveller the opportunity to make an informed choice as to whether they wish to receive testing for TB infection.
A single post-travel test using TST, done 8-weeks or more after the traveller’s return is the suggested screening strategy. Individuals who screen positive should be assessed for active disease and referred to a TB specialist for preventive treatment of TB infection. Refer to Implementation considerations.
Implementation considerations
Who does this guidance apply to?
Adult travellers to countries where the annual reported TB incidence is >10 TB cases per 100,000 population.
Who is not considered by this guidance?
- Travellers <15 years of age. This population will be considered in a separate guideline.
- Travellers who have previously screened positive and were treated for TB infection and/or disease.
Why is TB infection testing suggested?
- TB infection testing (following return from travel) provides an opportunity to discuss and offer preventive TB treatment to certain at-risk populations of travellers. This includes, depending on circumstances, travellers who previously screened negative as well as travellers who were never screened for TB infection.
Who is suggested to receive TB infection testing in this guidance? (see Recommendations)
- Certain travellers at higher likelihood for progression to TB disease, if infected
- Certain travellers more likely to be infected with TB, such as previous residence in a higher TB incidence country (e.g., non-Canadian-born residents) or participating in higher risk activities (e.g., providing healthcare) while travelling
- Travellers who would accept treatment if it was recommended
- Acceptability of preventive treatment should be discussed with the traveller before screening
What is the duration of travel for which screening is recommended?
The duration of travel varies by the type of traveller and their exposure to TB infection (see Recommendations and Figure 1 and Figure 2). Baseline risk of infection generally increases with longer duration of travel.
Who is suggested NOT to receive TB infection testing?
All travellers who do not fall within CATMAT’s recommendations. Refer to Figure 1 and Figure 2. Please note this includes those who have previously screened positive and were treated, regardless of travel history or duration. The risk of reinfection and recommendations for clinical care in these scenarios are outside the scope of this guidance.
What TB infection test(s) are suggested?
The tuberculin skin test (TST) is suggested, as it is currently the most accessible screening option in Canada and highly sensitive and specific. Interferon-gamma release assays (IGRA) may be suitable for some travellers where available. Serial IGRAs are not recommended as conversions and reversions are common with IGRAs, and it is challenging to define a true “conversion” related to new TB infection. IGRA would not be suitable for travellers who may have several trips given the need for serial IGRA testing in this setting. Refer to Chapter 4 of the CTS for more information on TB infection diagnosis.
When is TB infection testing suggested related to travel?
A single post-travel test using TST, done 8 weeks or more after return from travel. While consultation may occur with the patient prior to travel, CATMAT suggests that individuals who meet criteria for screening return for TB infection testing post-travel.
What to do if a test is positive?
Individuals who screen positive should be assessed to rule out active disease and referred to a TB specialist for management of TB infection.
What TB infection treatment(s) are suggested?
Provide preventive treatment of TB infection as outlined in Chapter 6 of the CTS. Daily rifampin for 4 months (4R) is generally the treatment of first choice. For rifamycin-based regimens, close attention to drug interactions must be considered. Alternative treatments such as isoniazid may be used, but have a higher rate of drug-associated liver harm compared with rifampin (2.3% vs. 0.9%). The risk of isoniazid hepatotoxicity increases with age.
Background
Tuberculosis (TB) is a respiratory disease caused by Mycobacterium tuberculosis (MTB). TB remains a major global health challenge, causing an estimated 10.8 million cases and 1.25 million deaths worldwide in 2023Footnote 4. TB elimination is a priority in Canada, and although Canada is considered a low-incidence country for TB, non-Canadian-born residents are one of the populations that are most disproportionately affected by TB in CanadaFootnote 5. Inuit, First Nations and Métis populations are others that are disproportionately affected in Canada. For more information and clinical guidance for this population, refer to Chapter 12 of the CTS.
A key step to achieving TB elimination is identifying those with TB infection who are at high risk of developing TB disease. Canadians may be exposed to TB through living, travelling or working in a TB-endemic country with a subsequent risk of progression to TB disease.
Clinical features
Infection with MTB occurs when a susceptible individual inhales bacilli-containing droplet nuclei from an individual with active pulmonary TB. Bacilli may then be contained within granulomas, in a non-infectious state known as TB infection. In the absence of treatment for TB infection, a small proportion of individuals infected with MTB will ultimately develop TB disease. The highest risk of developing TB disease is within 2 years of infection however the risk of reactivation to TB disease persists throughout lifeFootnote 6Footnote 7.
While pulmonary TB is the most common presentation of TB disease, TB can infect any site including extrapulmonary infections of the brain, bones, abdomen, and lymph nodes. Symptoms of active pulmonary TB typically include a cough that lasts two weeks or longer; coughing blood or phlegm, chest pain, weakness or tiredness, weight loss, loss of appetite, fever and chills, and night sweats. Persons with active TB disease may also be asymptomatic, thus healthcare providers should keep a high index of suspicion for this condition.
Epidemiology of TB in travellers
Rates of TB disease vary widely among different geographic regions of the world (Global tuberculosis report 2024, WHO).
- Low-incidence settings (<10 cases per 100,000 population) generally include high-income countries such as in western Europe, Canada, the United States of America, Australia, and New ZealandFootnote 4.
- Intermediate TB incidence countries (10-100 cases per 100,000 population) are generally found in Central and South America, the Middle East, North Africa, eastern Europe, and central Asian countries.
- The highest TB incidence countries (>100 cases per 100,000 population) are in southern and south-eastern Asia and sub-Saharan AfricaFootnote 4.
Among the highest-incidence regions (>100 cases per 100,000 population), the most common travel destinations, in descending order, for Canadians are: India, Philippines, Vietnam, Thailand, South Africa, Tanzania, Cambodia, Malaysia and Indonesia (Source: Statistics Canada, National Travel Survey, 2023).
Travellers to countries with higher TB incidences may be at relatively increased risk of TB infection. This includes those who are born in high TB incidence countries or persons travelling to provide healthcare. Those at highest risk of progressing to TB disease are those with a recent TB contact, children less than 5 years of age or those with underlying medical conditions (Table 1)Footnote 1Footnote 8Footnote 9Footnote 10.
Among non-Canadian-born residents, TB disease often results from reactivation of a TB infection acquired in their country of origin. The top places of birth for Canadian immigrants from higher TB incidence countries between 2016-2021, in order of immigration volume to Canada (and their corresponding TB incidence as of 2023) were: India (195 cases per 100,000), Philippines (643 cases per 100,000), China (52 cases per 100,000), Pakistan (277 cases per 100,000), Hong Kong (50 cases per 100,000), Iran (11 cases per 100,000), and Vietnam (182 cases per 100,000)Footnote 11.
TB infection screening and preventive treatment
The tuberculin skin test (TST) and interferon gamma (IFN- γ) release assay (IGRA) are used to detect TB infection. Both are acceptable for TB infection screening; however, the TST is currently more accessible and is likely to be covered by provincial or territorial health insurance plans in Canada. Of note, serial IGRA testing is not recommended and is therefore not suitable for travellers who embark on multiple trips to countries with higher TB incidence.
The preferred treatments for TB infection are outlined in Chapter 6 of the CTS, which identifies two options for first-line therapies. The treatment of choice is daily rifampin for four months (4R). The alternative option, weekly rifapentine and isoniazid for three months (3HP) is also effective, however, rifapentine is not currently licensed for routine use in Canada, and can only be accessed for an urgent public health need through the Special Access Program or via the List of Drugs for an Urgent Public Health Need. If rifamycin-based regimens are not tolerated or contraindicated, the preferred treatment option is nine months of daily isoniazid (9H).
The accuracy of TB infection screening and efficacy and harms of preventive treatment are summarized in the Summary of the evidence section below. For current Canadian recommendations on which tests should be used to diagnose TB infection in specific clinical circumstances, the limitations of each test, and for TB preventive treatment regimens, refer to Chapter 4 and Chapter 6 of the CTS, 8th Edition (2022).
Guideline development methods
This statement was developed by the Committee to Advise on Tropical Medicine and Travel (CATMAT) Tuberculosis working group (WG). With support from the CATMAT secretariat, the WG formulated key questions, a logic model, conducted literature reviews, synthesized the evidence, drafted recommendations, and developed the statement.
Key decisions supporting the recommendation development process were discussed with and approved by the full CATMAT committee. The final version of the statement and its recommendations were approved by CATMAT.
Canadian TB experts, including Chair of the Canadian Tuberculosis Standards, were consulted on the methodology, and reviewed this statement as well as recommendations herein. The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) methodology was used to assess the evidence and develop recommendations.
Key questions
The key PICO (Population of interest, Intervention, Comparison, and Outcome) question for our systematic GRADE analysis was:
- Should Canadian (adult) travellers to higher TB incidence countries be screened post-travel and, for those found to be positive, offered preventive treatment? If so, which travellers, and for what duration of travel?
We also developed a good practice statement to address:
- Should healthcare providers advise travellers to undertake infection prevention measures while travelling (good practice statement)?
We developed a logic model for TB infection screening (Figure 3). Key research questions, as shown on the model, are:
- What is the baseline risk of TB infection in travellers? Are certain subpopulations at increased risk for TB infection?
- Which travellers are at the highest risk of developing TB disease if infected (during or before travel)?
- What is the sensitivity and specificity and operational characteristics of the diagnostic tests (TST and IGRA) for TB infection?
- What is the safety and efficacy of 4 months of rifampin for TB preventive treatment?
- What proportion of travellers complete TB screening post-travel? What proportion of travellers who screen positive complete preventive treatment? What is the acceptance of TB infection screening and treatment by travellers?
- What is the cost and cost-effectiveness of screening and treating TB infection among travellers to prevent TB disease?
Outcomes and thresholds
The decision to screen for TB infection requires weighing benefits and harms of the screening test and TB preventive therapy. Outcomes for screening include the likelihood of preventing the development of TB disease, risk of treatment-associated serious adverse events for individuals who screen positive, as well as the burden of unnecessary treatment (if the test is not accurate) and drug-drug interactions.
The committee assessed the importance of all screening outcomes and considered the prevention of TB disease and serious adverse events to be critical for clinical decision making. Unnecessary treatment and drug-drug interactions were rated as important but not critical outcomes (see Appendix 3, Table 10). Our analyses focused on critical outcomes, with consideration for unnecessary treatment (important outcome) (Appendix 4). Drug-drug interactions would likely not have a major impact on the traveller’s decision making and were therefore not considered further.
To support our assessment we defined, for each critical outcome, a range of absolute effects that we believe to be reflective of patient beliefs (Table 2). They ranged from trivial effects, which we believe would not be influential to decision-making, to small through large absolute effects, which we believe would affect patient choice. This GRADE-compatible approach facilitates comparisons across different outcomes. For example, a large critical harm is generally considered to outweigh a moderate benefit.
A threshold for a moderate effect size for each critical outcome reflected the committee’s judgement on a value that constitutes a level of benefit or harm from screening and treatment that patients would consider to be clinically meaningful. Where available, studies summarizing the values and preferences of patients and the public were used. However, when not available CATMAT members drew on their own experiences and observations when sharing decision-making with patients and the public. A scale for the anticipated absolute effect size relative to the threshold for a moderate effect was determined as the following (Table 2):
| Critical Outcome | Thresholds of absolute increase or decrease in benefits and harms (# per 1,000 [%]) | Key WG Judgements | |||
|---|---|---|---|---|---|
| Trivial | Small | Moderate | Large | ||
| Prevention of TB Disease | <0.5 per 1,000 (0.05%) | 0.5 per 1,000 (0.05%) | 1 per 1,000 (0.1%) | 5 per 1,000 (0.5%) | Both outcomes are similarly weighed in decision making as there is a range in severity for SAE, however, in most cases, travellers would place greater value on the reduction in TB disease as a priority over the risk of SAE. |
| Treatment-associated serious adverse event (SAE) | <0.5 per 1,000 (0.05%) | 0.5 per 1,000 (0.05%) | 1 per 1,000 (0.1%) | 5 per 1,000 (0.5%) | |
Systematic or rapid review process
A comprehensive systematic or rapid review was conducted to search for, select, analyze and assess the evidence to inform the recommendations in this statement. With the support of a reference librarian, a search strategy was developed and a systematic literature search for systematic reviews and primary studies was completed (Appendix 1):
- up to July 24, 2024 for research question 1,
- up to December 15, 2023 for question 5, and
- up to May 25, 2023 for question 6.
Five electronic databases (Medline, Embase, Global Health, ProQuest, and Scopus) were searched using the pertinent terms for each key question.
A systematic review of TB infection in travellers by Diefenbach-Elstob et al. (2021) was used to answer research question 1Footnote 1. A systematic review and meta-analysis by Campbell et al. (2020) examining risks of TB disease progression was used to inform research question 2Footnote 2. For research question 3, we identified a recent systematic review and meta-analysis by the US Preventive Services Task Force which assessed the accuracy of TB screening tests (TST and IGRA)Footnote 12. For research question 4, we referred to the systematic review and GRADE appraisal of clinical trials of TB preventive treatment regimens by the United States Centers for Disease ControlFootnote 13. The data for questions 5 and 6 are described in the section on Feasibility and acceptability and were not included in the assessment of the balance of benefits of harms.
Model and key assumptions
We developed a model to estimate the absolute benefit and harm associated with TB infection screening and treatment (Appendix 2, Figure 4). They key model parameters were (Table 3):
- baseline risk of TB infection among travellers and certain subpopulations,
- risk of TB disease if infected,
- sensitivity and specificity of TB infection testing,
- efficacy of TB infection treatment, and
- harms (SAE) of TB infection treatment.
See the section for full discussion of each of these steps. Risk of disease from travel was estimated for 10 years following exposure, and it was assumed that the annual risk of disease would decay by 50% after the first two years following exposureFootnote 14Footnote 15.
| Model input | Pooled estimate |
|---|---|
| Annual risk of TB infection in Canadian-born travellersFootnote 1 | 2.06% |
| Annual risk of TB infection in healthcare workersFootnote 1 | 4.30% |
| Prevalence of TB infection among non-Canadian-born residentsFootnote 16 | 25% |
| Annual risk of TB disease: | |
| Very high (HIV, silicosis)Footnote 3Footnote 2 | 3.00% for years 1-2; 1.50% for years 3-10 |
| High (chronic kidney disease, transplant, fibronodular disease, receiving immunosuppressing medications, cancer)Footnote 2Footnote 3 | 0.50% for years 1-2; 0.25% for years 3-10 |
| Moderate (Granuloma on chest x-ray, diabetes, heavy alcohol/ tobacco use)Footnote 3 | 0.15% for years 1-2; 0.08% for years 3-10 |
| Low (general adult population)Footnote 2Footnote 3 | 0.03% for years 1-2; 0.01% for years 3-10 |
| TB diagnostic accuracy of TST ≥ 10mm indurationFootnote 12 | Sensitivity: 81% |
| Specificity: 98% | |
| Treatment efficacy with rifampinFootnote 13 | Efficacy: 75% |
| SAE with rifampinFootnote 17Footnote 18 | Risk of SAE: 0.9% |
The balance of benefit and harms were judged by comparing the absolute risk reduction of TB disease with the absolute risk of serious adverse events.
The absolute risk reduction of TB disease (benefit) and absolute risks of serious adverse events (harms) across different travel durations for the three different travelling populations (Canadian-born general adult travellers, Canadian-born healthcare workers, and non-Canadian-born resident travellers) were estimated. Refer to Table 4, Table 5, Table 6 and Table 7 in the Summary of the evidence and sample calculations in Appendix 3.
The absolute risk reduction of TB disease was estimated from the baseline risk of TB infection in travellers (varying by traveller type and duration of travel) x the estimated number of true positives x the annual risk of disease progression x the efficacy of rifampin.
The absolute increase in risk of serious adverse events was estimated from the total number of travellers treated x Grade 3-5 SAE for rifampin.
The absolute increase in unnecessary treatment was estimated, by taking the difference between the estimated number of true positives and the product of the estimated number of true positives x the annual risk of disease progression, then taking the sum of this difference with the estimated number of false positives.
Although there are limitations with the modelled estimates, such as the assumption of one cumulative trip, the model structure generally reflects the standard pathway of care in Canada. These modelled estimates were used for the Evidence to decision process (see below).
Evidence to decision process
The committee discussed the evidence and developed the recommendations after considering benefits and harms of TB infection screening and treatment, patient values and preferences, resources, acceptability, and feasibilityFootnote 19. To evaluate the certainty of evidence from model outputs, the model structure and the level of certainty in the model inputs was assessed using GRADE (Appendix 4)Footnote 20. The overall level of certainty of the evidence of benefits and harms was presented as high, moderate, low or very low. The perspective taken and decision to recommend screening was made from that of the individual traveller, however the committee reviewed the evidence and acknowledges the implications of preventing disease transmission and resource allocation at the population level.
The assessment leading to the final recommendations was summarized in an Evidence to decision frameworkFootnote 21(see Appendix 4). This work was supported by a GRADE methodological consultant from McMaster University.
The good practice statement was developed because we believed there was accepted evidence of unequivocal net benefit with negligible harms and costsFootnote 22(see Appendix 5).
Evidence assessment and recommendations on TB prevention in travellers
-
CATMAT suggests that Canadian-born adult travellers returning to Canada after travel in a higher TB incidence country(ies) for 6 consecutive months or more and who are at very high risk of developing TB disease be screened for TB infection after return.
(Discretionary recommendation, low-certainty evidence)
CATMAT suggests that Canadian-born adult healthcare providers returning to Canada after delivering patient care in higher TB incidence country(ies) for 3 consecutive months or more and who are at very high risk of developing TB disease be screened for TB infection after return.
(Discretionary recommendation, moderate-certainty evidence)
CATMAT suggests that non-Canadian-born resident adult travellers (see Definitions), including healthcare workers, who are returning to Canada after travel in a higher TB incidence country(ies) for any duration of travel and who are at high to very high-risk of developing TB disease be screened for TB infection after return.
(Discretionary recommendation, low-certainty evidence)
Healthcare providers should counsel travellers on infection prevention measures if they may be at risk of TB exposure.
(Good practice statement)
Summary of the evidence
Baseline risk of infection
Canadian-born travellers
The risk of TB infection is affected by the individual’s type of exposure, including the TB incidence in the destination country, travel duration, travel purpose, and pre-travel exposure to TB. A systematic review of risk of travel-associated TB infection estimated overall incidence of TB infection among travellers visiting higher TB incidence countries was 2.32% (95% confidence interval [CI] 1.71-3.13) after 12-18 months of travel, with a rate of travel-associated active TB of 120.71 cases per 100,000 travellersFootnote 1. Moreover, certain subpopulations were at a relatively higher risk. For example, healthcare workers (HCW) travelling for work had a pooled cumulative incidence of 4.3% (95% CI 2.8-6.7) at a median of 6 months, which was significantly higher than for non-HCW travellers (2.06% CI 1.49-2.86)Footnote 1. This is consistent with another analysis which found that compared to the general population, stratified pooled estimates for annual TB incidence rate ratios in HCWs were 2.42 (95% CI 1.20–3.64), 2.45 (95% CI 1.05–3.84), and 3.68 (95% CI 2.89–4.48) times higher than the risk for that of the general population in for low, middle, and high incidence countries, respectivelyFootnote 23. This is consistent with the double increased risk of TB infection among travelling health care workers as compared to the general travellers found in the Diefenbach Estob systematic reviewFootnote 1. Given this documented higher risk of infection among HCWs, CATMAT stratified the baseline risk of infection by HCW status (4.3% vs non-HCWs, 2.06%).
Travel to a higher TB incidence country, relative to Canada, was defined as one with >10 TB cases per 100,000 population, corresponding with the inclusion criteria for studies in the systematic reviewFootnote 1. This study did not find a difference in TB infection among travellers to intermediate (>10-99 cases/100,000 population) compared to high (>100/100,000) TB incidence settings; this may have been reflecting that destination countries were not clearly reported in all the included studies. We also undertook sensitivity analyses by elaborating models based on infection likelihoods that were 30% and 50% of our baseline rates. Finally, while travel durations for the studies included in the systematic review varied from 6 to 24 months, we simplified our model by assuming that the risk of infection was attributable to one year of travel.
The risk of TB infection has previously been estimated in other settings with the Styblo rule, derived in 1985 using data from 16 countries, which linked annual TB mortality, the incidence of smear-positive TB results, and the annual risk of infectionFootnote 24. In high TB incidence countries, Styblo estimated that an annual incidence of 50 to 60 smear-positive TB cases per 100,000 population corresponds to a 1% annual risk of infectionFootnote 24. However, this ratio has been shown to overestimate TB transmissionFootnote 24 and was therefore not used in our analysis. Recent modelling studies have estimated updated annual risk of infection, including among non-Canadian-born residents from TB endemic countries, taking into consideration improvements in national TB disease treatment programs and infection controlFootnote 16Footnote 25. These models, based on a combination of direct estimates from TB infection surveys and indirect estimates calculated from the WHO smear-positive TB prevalence with the revised Styblo ratio, suggest that the annual predicted incidence of TB infection among Canadians travelling to TB endemic countries is less than 2%Footnote 16Footnote 25. Since these estimates were not based specifically on travellers, we opted to base our assessment on the baseline estimates of infection from the Diefenbach-Elstob et al. systematic review in the primary analysisFootnote 1 and conducted sensitivity analyses using estimates of risk of TB infection for varying rates of TB incidence derived from the Styblo ratio.
Non-Canadian-born resident adult travellers (see Definitions)
Canadian and U.S. surveillance data and other observational studies in low TB incidence settings suggest that specific traveller subpopulations – persons born in higher TB incidence countries, VFRs (those visiting friends and relatives), and extended-stay travellers - are at higher risk of travel-associated TB infection and diseaseFootnote 26Footnote 27Footnote 28Footnote 29Footnote 30Footnote 31Footnote 32Footnote 33. The prevalence of TB infection, based on a modelling study, among non-Canadian-born residents from higher TB incidence countries, is estimated to be 25%Footnote 16. This is consistent with other estimates in the literature ranging from 10-40%, though it varies depending on the TB incidence in the country of origin and age of the individual (a proxy for duration of exposure and in certain age groups increased risk of progression to TB disease)Footnote 25Footnote 34Footnote 35Footnote 36Footnote 37.
For persons born in higher TB incidence countries, estimates suggest that 15-50% of TB disease cases in some of these populations develop after VFR travel to their countries of originFootnote 28Footnote 32Footnote 38Footnote 39Footnote 40. Surveillance data from European travel clinics demonstrated a fifteen-fold higher risk of active TB in VFR travellers compared to short term or expatriate travellers (3.61% vs 0.23%)Footnote 28.
Non-Canadian-born resident travellers with prior negative screening test results may have similar risk profiles to Canadian-born travellers.
Risk of development of TB disease
Risk of progression from TB infection to TB disease is influenced by individual-level factors such as time since infection, age and concomitant medical co-morbidities (Table 3)Footnote 1Footnote 8Footnote 41. The risk of developing TB disease is highest within the first 2 years after infection and is much lower after this, dropping to about a third of the risk by the start of the fifth yearFootnote 4Footnote 15Footnote 42Footnote 43.
Among persons with no medical co-morbidities, only 5% to 10% with TB infection will develop TB disease during their lifetimeFootnote 7. The TST and IGRA cannot differentiate between infections that will progress to TB disease and those that will not. A large systematic review and meta-analysis that included cohort studies and randomized trials of untreated populations who screened positive with TST or IGRA developed TB disease at different rates depending on underlying medical co-morbidities and the magnitude of associated immunosuppressionFootnote 2. This data is summarized by risk group (low to very high) of the annual risk to develop TB disease (Table 3).
Although pediatric travellers are known to have a higher risk of progression to TB diseaseFootnote 10Footnote 14Footnote 44Footnote 45, this population will be considered in a separate guideline.
Accuracy of diagnostic tests for TB infection
A 2023 systematic review identified 101 studies of at least fair quality that aimed to synthesize performance characteristics of TST and IGRA assays; pooled sensitivity for TST across all thresholds (5mm, 10mm, 15mm) ranged from 0.60-0.81, pooled sensitivity for all IGRAs ranged from 0.81-0.90, and specificity for all tests ranged from 0.95-0.99Footnote 12. At the 10mm threshold, TST sensitivity was 0.81 and specificity was 0.98, while IGRA sensitivity for the QFT-Plus was 0.89 and specificity was 0.98Footnote 12. The committee decided to use the TST values in our model since this test is universally available across Canada and performs similarly to IGRA (Table 3). Further, IGRA is not recommended for repeat testing, which is more likely in the travel setting, e.g., for travellers who embark on multiple trips.
Safety and efficacy of preventive TB treatment
A 2020 systematic review of clinical trials of regimens to treat TB infection was conducted by the United States Centers for Disease Control (CDC) and included an evidence appraisal using GRADE criteriaFootnote 13. Since rifampin and placebo have not been directly compared in clinical trials, efficacy was evaluated by a network meta-analysis, which demonstrated an odds ratio of 0.25 (95% CI of 0.11-0.57) for 3-4 months of daily rifampin compared to no treatmentFootnote 13. We used the 75% efficacy for our model (Table 3).
Three clinical trials assessed the risk of serious adverse events of rifampin compared to isoniazid for TB infection treatmentFootnote 17Footnote 18Footnote 46. The initial phase 1 trial was a small study including only 58 patients per study arm and outcome assessors were not blinded, therefore subject to biasFootnote 46. Among the phase 2 and 3 trials, the risk of serious adverse events (grade 3-5) that were judged by the panel as possibly or probably related to the study drug was 0.9% for the rifampin arm, and 2.3% for the isoniazid armFootnote 17Footnote 18. We used the 0.9% value for rifampin in our model (Table 3).
Benefits and harms of screening and treatment of TB infection
The benefits and harms of screening and treatment of TB infection were estimated with a model that included values for each of the critical and some important outcomes and are presented separately for three groups of travellers: Canadian-born, Canadian-born healthcare workers and non-Canadian-born residents across different travel durations (1 to 12 months).
Canadian-born travellers
Prevention of TB disease (critical outcome): The greatest estimated absolute risk reduction in individual likelihood of TB disease from screening and treating post-travel is among individuals at very high risk of developing TB disease if infected (i.e. individuals with HIV, close TB contact, or silicosis). For Canadian-born travellers at very high risk, travelling for 6 months or more, the estimated absolute risk reduction in TB disease ranges from 0.1-0.2% overall, and or between 4-6% for those who screen positive. For healthcare worker travellers at very high risk, travelling for 3 months or greater, the estimated absolute risk reduction in TB disease ranges from 0.1-0.5% overall, or between 4-9% for those who screen positive (Table 4, Table 5, Table 11, Table 12 and Appendix 3).
The magnitude of the benefits largely did not change when we explored different assumptions about the baseline risk, or when using IGRA rather than TST. Using estimates of risk of TB infection based on the Styblo ratio demonstrated a similar magnitude of benefit among only individuals at very high risk of developing TB disease, suggesting that travel related risk of infection is not a primary driver of the likelihood of developing TB disease.
Serious adverse events (critical outcome): For Canadian-born adult travellers the risk of serious adverse events from treatment with rifampin was consistently below our threshold of 0.1% for all groups of travellers (Table 4 and Table 5). Sensitivity analyses demonstrated that use of isoniazid daily for 9 months may increase the absolute risk of serious adverse events between 2-3-fold compared to rifampin.
Unnecessary treatment (important outcome): Among Canadian-born adult travellers screened for TB infection there is a 2-6% risk of being treated unnecessarily due to false positive tests (Table 4 and Table 5). For rifamycin-based regimens, medication adjustment and additional monitoring may be required due to drug-drug interactions. This is common for individuals on antihypertensives, anticoagulants, antifungals, methadone, some immunosuppressive agents, antiretrovirals, and hormonal contraceptives.
Refer to Appendix 3 for sample calculations for estimated benefits and harms.
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 0.23 | 0.04 | 0.01 | 0.00 |
| 6 months | 0.11 | 0.02 | 0.01 | 0.00 |
| 3 months | 0.06 | 0.01 | 0.00 | 0.00 |
| 1 month | 0.02 | 0.00 | 0.00 | 0.00 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.03 | 0.03 | 0.03 | 0.03 |
| 6 months | 0.03 | 0.03 | 0.03 | 0.03 |
| 3 months | 0.02 | 0.02 | 0.02 | 0.02 |
| 1 month | 0.02 | 0.02 | 0.02 | 0.02 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 3.33 | 3.58 | 3.61 | 3.62 |
| 6 months | 2.66 | 2.79 | 2.81 | 2.81 |
| 3 months | 2.33 | 2.39 | 2.40 | 2.41 |
| 1 month | 2.11 | 2.13 | 2.13 | 2.13 |
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 0.47 | 0.08 | 0.02 | 0.00 |
| 6 months | 0.24 | 0.04 | 0.01 | 0.00 |
| 3 months | 0.12 | 0.02 | 0.01 | 0.00 |
| 1 month | 0.04 | 0.01 | 0.00 | 0.00 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.05 | 0.05 | 0.05 | 0.05 |
| 6 months | 0.03 | 0.03 | 0.03 | 0.03 |
| 3 months | 0.03 | 0.03 | 0.03 | 0.03 |
| 1 month | 0.02 | 0.02 | 0.02 | 0.02 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 4.77 | 5.29 | 5.37 | 5.39 |
| 6 months | 3.39 | 3.65 | 3.68 | 3.70 |
| 3 months | 2.69 | 2.82 | 2.84 | 2.85 |
| 1 month | 2.23 | 2.27 | 2.28 | 2.28 |
Non-Canadian-born resident travellers (see Definitions)
Prevention of TB disease (critical outcome): For non-Canadian-born resident travellers, individuals in the high and very high-risk groups met our threshold for a moderate important benefit, with an estimated absolute risk reduction in TB disease ranging from 0.4-2.8% among all those screened, and 1.8-10.9% for those who screen positive, depending on travel duration and purpose of travel (Table 6, Table 7, Table 13 , Table 14, and Appendix 3). The majority of the TB infection among non-Canadian-born resident travellers is due to prior residence in higher TB incidence countries (25%). The additional risk of TB infection for 12 months of general travel or healthcare travel is 2.06% or 4.3%, respectively.
Similar to Canadian-born travellers, the magnitude of the benefits largely did not change when we explored different assumptions about the baseline risk, or when using IGRA rather than TST.
Serious adverse events (critical outcome): For non-Canadian-born resident travellers, the threshold for a moderate effect was surpassed for all risk groups, at 0.2%, owing to the higher likelihood of being infected (and treated) (Table 6 and Table 7). However, CATMAT placed a higher value on the relatively large estimate of benefit of TB disease prevention compared to the moderate absolute estimate of treatment-related adverse events for the high and very high-risk groups. For the moderate-risk group, benefits and harms were closely balanced (see Judgement). Similar to Canadian-born travellers sensitivity analyses demonstrated that use of isoniazid daily for 9 months may increase the absolute risk of serious adverse events between 2-3 fold compared to rifampin.
Unnecessary treatment (important outcome): This risk of unnecessary treatment increases to 19-25% for non-Canadian-born resident travellers due to the higher likelihood of screening positive compared to Canadian-born travellers (Table 6 and Table 7). The majority of the TB infection among non-Canadian-born resident travellers is due to prior residence in higher TB incidence countries (25%). The additional risk of TB infection for 12 months of general travel or healthcare travel is 2.06% or 4.3%, respectively.
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 2.50 | 0.42 | 0.13 | 0.02 |
| 6 months | 2.39 | 0.40 | 0.12 | 0.02 |
| 3 months | 2.33 | 0.39 | 0.12 | 0.02 |
| 1 month | 2.30 | 0.38 | 0.11 | 0.02 |
| 2 weeks | 2.29 | 0.38 | 0.11 | 0.02 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.21 | 0.21 | 0.21 | 0.21 |
| 6 months | 0.20 | 0.20 | 0.20 | 0.20 |
| 3 months | 0.20 | 0.20 | 0.20 | 0.20 |
| 1 month | 0.20 | 0.20 | 0.20 | 0.20 |
| 2 weeks | 0.20 | 0.20 | 0.20 | 0.20 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 20.04 | 22.82 | 23.21 | 23.35 |
| 6 months | 19.38 | 22.03 | 22.40 | 22.54 |
| 3 months | 19.04 | 21.64 | 22.00 | 22.13 |
| 1 month | 18.82 | 21.37 | 21.73 | 21.86 |
| 2 weeks | 18.76 | 21.30 | 21.66 | 21.79 |
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 2.75 | 0.46 | 0.14 | 0.02 |
| 6 months | 2.51 | 0.42 | 0.13 | 0.02 |
| 3 months | 2.40 | 0.40 | 0.12 | 0.02 |
| 1 month | 2.32 | 0.39 | 0.12 | 0.02 |
| 2 weeks | 2.30 | 0.38 | 0.11 | 0.02 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.23 | 0.23 | 0.23 | 0.23 |
| 6 months | 0.21 | 0.21 | 0.21 | 0.21 |
| 3 months | 0.20 | 0.20 | 0.20 | 0.20 |
| 1 month | 0.20 | 0.20 | 0.20 | 0.20 |
| 2 weeks | 0.20 | 0.20 | 0.20 | 0.20 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 21.48 | 24.54 | 24.96 | 25.12 |
| 6 months | 20.10 | 22.89 | 23.28 | 23.42 |
| 3 months | 19.41 | 22.07 | 22.44 | 22.57 |
| 1 month | 18.94 | 21.52 | 21.88 | 22.01 |
| 2 weeks | 18.82 | 21.37 | 21.73 | 21.85 |
Feasibility and acceptability
Acceptability: Screening
Screening with TST requires two visits to the healthcare centre, the first to inject the Tubersol®, and a second 48 to 72 hours later to read the test. Although there are no studies specific to travellers, a 2016 systematic review and meta-analysis examined the cascade of care for the diagnosis and treatment of TB infection among the general population, drawing on data from 58 studies conducted across multiple countriesFootnote 47. Among individuals identified for TB infection testing, approximately 72% completed the screening step.
Acceptability: Treatment
Failure to initiate and complete TB preventive therapy has been consistently recognized as a missed opportunity in TB control. Treatment initiation has been shown to be higher among individuals with immunosuppression, estimated at 85%, as compared to healthcare workers, estimated at 6-48% initiationFootnote 47Footnote 48Footnote 49. Reported treatment completion rates in the general population range widely, from 46% to 95%Footnote 50.This is undoubtedly due to the variability in values and preferences relating to a relatively long course of antimicrobial treatment for an asymptomatic condition.
A systematic review among immigrants found that the proportion of immigrants testing positive who initiated treatment was only 69% (95% CI [51%, 84%]) and, of those, only 74% (95% CI [66%, 81%]) completed therapyFootnote 51.
Barriers
Multiple social and health system barriers contribute to losses at different steps along the care cascade, including medical contraindications, language barriers and self-perceived low risk of infectionFootnote 47. Socio-economic factors such as unemployment or lack of social support have also been shown to have an impact on missed screening and poor compliance with TB infection therapyFootnote 52.
Commonly reported barriers to treatment initiation and adherence include perception that TB is not a serious disease, self-perceived low risk of TB disease, concern about treatment toxicity and the long duration of therapy for some regimensFootnote 47. Lower rates of treatment completion have been reported among immigrants and healthcare workersFootnote 50Footnote 52Footnote 53. Among healthcare workers, lower rates of treatment initiation and completion have been attributed to certain groups such as physicians or technicians, longer duration of employment, and older age and concerns regarding adverse events due to treatmentFootnote 49.
In the travel medicine context, many high-risk travellers, such as immigrants, do not seek travel adviceFootnote 54. Among those who do, TB is often not prioritized by either travellers or healthcare providers. Practical obstacles such as the requirement for two visits to administer and read the tuberculin skin test (TST), along with language barriers and immigration-related concerns, may further reduce uptake.
Barriers at the healthcare provider level are also well documented. These include limited knowledge about the benefits and risks of TB preventive therapy, discomfort with treating asymptomatic individuals, administrative burden, and unclear referral pathwaysFootnote 47Footnote 55Footnote 56. In a nested case-control study, immigrants, men, and healthcare workers were less likely to be offered TB infection treatment – differences not supported by underlying risk, suggesting potential provider biasFootnote 57.
Facilitators
A consistent theme across multiple studies is that shorter treatment regimens are associated with a higher degree of adherence to and completion of therapyFootnote 50Footnote 52, with a 20% greater completion rate reported in rifamycin-based regimens compared with isoniazidFootnote 47. Rifampin is a treatment regimen taken for a duration of 4 months, requiring daily doses for 120 days. It is an oral capsule taken on an empty stomach. Other factors associated with screening and treatment initiation and fewer losses to follow up include the presence of pre-existing immune compromise and being part of a contact investigation. This has been speculated to be due to their high-risk status and resulting in more intensive health careFootnote 47.
There are mixed results on the effect of directly observed therapy, but social interventions such as adherence coaching, education and active case management have had positive effects in some studies. The impact of incentive-based interventions appears to vary depending on the population and settingFootnote 52.
Resource considerations
The TST and TB preventive therapy are covered by provincial health insurance in Canada when they are considered medically necessary. IGRA is covered by some provincial health insurance plans in Canada, while in some provinces there may be out of pocket expenses which are estimated to be around $100Footnote 58.
There are no published cost-effectiveness analyses on screening for TB infection post-travel where those who screened positive were treated with rifampin for 4 months. A decision analysis with isoniazid for 9 months as preventive therapy compared four strategies for screening travellers to Mexico, Haiti or the Dominican Republic, found a single post-travel test to be most cost effective across a broad range of assumptions, and cost saving for longer trips or with higher risk of exposure, although the individuals in the model were not representative of all potential Canadian travellersFootnote 59. Among a hypothetical cohort of 30-year-old expatriates travelling from low to high TB incidence countries where various screening strategies including TST, IGRA or chest X-ray followed by treatment with either 3HP or 9H were modeled, the screening and treatment strategy found to be most cost-effective was an IGRA (QuantiFERON-TB Gold In-Tube) with 3HPFootnote 60. However, 3HP is not currently a widely available treatment strategy in Canada.
From the population perspective, studies suggest that there may be cost savings associated with targeted preventive treatment. A retrospective chart review and cost analysis estimated the costs associated with TB management (TB infection and TB disease) at three major treatment centers in CanadaFootnote 61. When assessing the cost effectiveness of treating TB infection, the total cost of using 4 months of rifampin for 23 individuals infected with TB were found to be equal to the total cost of managing 1 individual with TB disease, suggesting that targeted treatment of TB infection could reduce higher costs associated with treating TB disease, particularly drug-resistant TBFootnote 61.
Judgement
When the reduction in TB disease was at least moderate and exceeded the estimated increase in treatment-associated SAE, the committee judged that post-travel testing screening for TB infection provided more benefit than harm. For Canadian-born travellers, the benefits were at least moderate and harms were low for individuals in the very high-risk group that travelled for 6 months or more. For Canadian-born healthcare providers, the benefits were at least moderate and harms were low for individuals in the very high-risk group that travelled for 3 months or more.
For non-Canadian-born resident travellers at high and very high risk of disease progression, we placed a higher value on the relatively large estimate of benefit of TB disease prevention compared to the moderate absolute estimate of treatment-related adverse events. For non-Canadian-born resident travellers with low to moderate risk of developing TB disease, the balance of effects did not clearly favor the intervention of TB screening. For non-Canadian-born resident travellers at moderate risk of disease progression, estimates for harms and benefits were both moderate and the estimated increase in SAE likelihood numerically exceeded the estimated reduction in TB disease likelihood. Because the estimates of benefits and harms are relatively balanced, healthcare providers should carefully assess the traveller’s individual risk, discuss benefits and harms of testing, explore values and preferences, and allow the traveller to make an informed choice as to whether they wish to receive testing for TB infection (Figure 2).
The committee judged that the cost of the intervention to the individual traveller was low, that the intervention was acceptable to most travellers, and that the feasibility was unknown (Appendix 4). Based on these considerations, we chose to make a discretionary recommendation for targeted post-travel screening among certain high-risk groups.
Infection prevention measures
Should travellers wish to undertake infection prevention measures, healthcare providers should advise them to consider wearing a N95 mask if they will be in a high-risk setting, and/or to avoid these settings (e.g., crowded, poorly ventilated indoor environments).
Small indoor spaces, with poor ventilation or air recirculation, can increase the concentration of bacteria inhaled by susceptible individuals. Crowded settings force closer proximity and an increase in the number of individuals exposed to an infectious case. Additionally, the longer the duration of exposure, the greater likelihood of disease transmissionFootnote 62. Health Canada-approved respirators with a filter class of N95 or higher are recommended, as medical masks are not designed to protect against M. tuberculosisFootnote 63. N95 masks use a filter to remove at least 95% of airborne particles, including mycobacteria, and as such are commonly used in healthcare settings to prevent TB infectionFootnote 64.
Individuals planning to work in a healthcare setting in a higher TB incidence country are advised to follow infection prevention and control guidelines applicable to TB. As TB is an airborne pathogen, the use of a fit-tested N95 respirator is advised. For more information on airborne precautions please refer to Routine practices and additional precautions for preventing the transmission of infection in healthcare settings, notably Part B subsection III, Recommendations for routine practices in all healthcare settings, or local guidelines in the country of work. Further information on infection prevention and control guidelines specific to TB can be found in Chapter 14 of the CTS.
Individuals with increased risk for TB and other infectious diseases (such as immune compromised individuals, people living with HIV, people with underlying lung disease) should consult with a healthcare provider specialized in travel medicine for recommendations for masking and other infection preventions during travel.
Bacillus Calmette-Guérin (BCG) vaccine
The Bacillus Calmette-Guérin (BCG) vaccine has been recommended in the past, selectively for travellers by the UKFootnote 65, and in exceptional cases, by the WHOFootnote 66 and in CanadaFootnote 67. In general, given the lack of evidence relating to prevention of disease in adults, BCG is not recommended for adult travellers, such as healthcare workers, before travel to high TB incidence settings. Screening for TB infection after travel and chemoprophylaxis for those with a positive test is the preferred approach to TB control in travellers.
Conclusions and research needs
Given the potentially long interval between infection and development of disease, the association of TB disease with travel-related exposure may be under-recognized. With an increase in migration and travel, many non-Canadian-born resident travellers are at risk of reactivation of undiagnosed TB infection post-arrivalFootnote 68.
Modeled estimates suggest that TB infection testing should be targeted to travellers at the highest risk of developing TB disease, regardless of travel destination. Additional studies are needed on the precise impact of travel duration and TB incidence in the country of travel, including the impact of cumulative trips, given that current baseline risk estimates are based on a range of consecutive travel duration and TB incidence in the country of travel. While the current statement does not include pediatric travellers, CATMAT acknowledges the knowledge gap and higher risk of progression to TB disease in this population, with future statement updates to follow in this area.
Abbreviations
- AE
- Adverse event
- 3HP
- Rifapentine and isoniazid treatment (weekly, 3-month duration)
- 4R
- Rifampin treatment (daily, 4-month duration)
- 9H
- Isoniazid treatment (daily, 9-month duration)
- BCG
- Bacillus Calmette-Guérin
- CATMAT
- Committee to Advise on Tropical Medicine and Travel
- CI
- Confidence interval
- CTS
- Canadian Tuberculosis Standards
- GPS
- Good practice statement
- GRADE
- Grading of Recommendations, Assessment, Development and Evaluation
- HCW
- Healthcare worker
- HIV
- Human immunodeficiency virus
- IFN- γ
- Interferon gamma
- IGRA
- Interferon-gamma release assays
- MTB
- Mycobacterium tuberculosis
- N95
- NIOSH-approved N95 filtering facepiece respirator
- NACI
- National Advisory Committee on Immunization
- PHAC
- Public Health Agency of Canada
- PICO
- Population of interest, Intervention, Comparison, and Outcome
- QFT-Plus
- QuantiFERON-TB Gold Plus
- SAE
- Serious adverse event
- TB
- Tuberculosis
- TST
- Tuberculin skin test
- US
- United States
- UK
- United Kingdom
- VFR
- Visiting friends and relatives
- WG
- Working group
- WHO
- World Health Organization
Acknowledgements
This statement was prepared by the CATMAT Tuberculosis Working Group: C Greenaway (Lead), S Kadykalo (Secretariat), A Khatib, J Pernica, K Plewes, S Schofield, and was endorsed by CATMAT.
Former member: T Audcent.
CATMAT gratefully acknowledges the contribution of: N Bruce, YG Bui, M Carew, P Deb-Rinker, S Desjardins, M Desroches, T Diefenbach-Elstob, H Feguery, LS Ho, M Laplante, T Lee, M Libman, T Locke, T Nguyen, N Santesso and the Public Health Agency of Canada Library.
In addition, CATMAT gratefully acknowledges the contribution of Canadian tuberculosis experts J Campbell, J Johnston, and K Schwartzman.
CATMAT Members: M Libman (Chair), YG Bui (Vice-Chair), K Plewes (Malaria Sub-Committee Chair), I Bogoch, A Khatib, P Lagacé-Wiens, J Lee, C Yansouni, K Kazmi, and C Hogan
Former member: C Greenaway
Liaison representatives: J Pernica (Association of Medical Microbiology and Infectious Disease Canada) and K O'Laughlin (Centers for Disease Control and Prevention, United States)
Former liaison: I Viel-Thériault (Canadian Pediatric Society)
Ex-officio representatives: V Donici (National Defence and the Canadian Armed Forces [DND-CAF]), C Jensen (National Advisory Committee on Immunization [NACI] Secretariat, PHAC), D Marion (DND-CAF), S Schofield (DND-CAF), M Tunis (NACI Secretariat, PHAC), and R Zimmer (Biologic and Radiopharmaceutical Drugs Directorate, Health Canada)
Former ex-officio representatives: E Ebert (DND-CAF)
Conflicts of interest
None declared.
Appendix 1: Sample search strategy
| Search number | Search strategy | Results |
|---|---|---|
| 1 | exp *Tuberculosis/ | 183922 |
| 2 | ([active or latent] adj3 [Tuberculosis or TB]).tw,kf. | 15202 |
| 3 | or/1-2 [Tuberculosis] | 187351 |
| 4 | exp Travel/ | 31349 |
| 5 | (travel* or touris*).tw,kw,kf. | 88669 |
| 6 | or/4-5 [Travel] | 101950 |
| 7 | 3 and 6 | 655 |
| 8 | limit 7 to yr=2020-current | 107 |
Appendix 2: Logic model and model structure

Figure 3: Descriptive text
This image illustrates the logic model which outlines key questions and decision points used by the CATMAT TB working group to develop recommendations for TB infection screening in returned travellers, under the assumption that those who screen positive would be offered preventative treatment with the end goal of TB disease prevention. The logic model is ordered as follows:
- First, what is the baseline risk of TB infection in travellers?
- Second, which travellers are at the highest risk of developing TB disease?
- Third, what tests should be used? Considerations include test characteristics (sensitivity and specificity) and the potential for unnecessary treatment or missed treatment, as well as acceptance, feasibility and cost of the tests.
- Fourth and lastly, what is the safety and efficacy of the preventive treatment? Considerations include risks of serious adverse events or drug interactions, as well as acceptance, feasibility and cost of the treatment.

Figure 4: Descriptive text
This figure illustrates the structure of the model used to estimate the outcomes from TB infection screening and treatment among travellers with the following characteristics:
- The traveller is returning from a TB endemic area where there are more than 10 cases per 100,000 population.
- The screening strategy is a single post-travel TST test, and rifampin is the treatment.
- The follow-up period is 10 years.
The model is split into two main branches, where travellers either screen positive (treated with rifampin) or negative for TB infection (not treated).
Among those who screen positive and are treated, some are infected cases that are correctly identified through screening (true positive) while others are not truly infected but did receive a positive screening test result (false positive).
Among those true positives, some would develop disease while others would not.
- Among those who would develop disease, some would be effectively treated, while others may not be, both of whom may be at risk of developing serious adverse events.
- Those who would not develop disease would receive unnecessary treatment and may be at risk of developing serious adverse events. The same is true for false positives.
Among those who screen negative and are not treated, those who are not infected and received a negative screening test result (true negative) would, appropriately, not receive treatment. Those who are infected, yet received a negative screening test result (false negative), could develop disease, but would not have received treatment (missed treatment).
Appendix 3: Supplemental tables and sample calculations
| Differences | CATMAT | CTS, 8th Edition |
|---|---|---|
| Methodology | Full GRADE process applied to assess the evidence and develop recommendations, in consultation with a GRADE methodologist, from McMaster University. | The CTS used a modified GRADE approach. |
| Thresholds used to develop TB infection screening recommendations among travellers | CATMAT decided on an absolute benefit threshold of 1 per 1,000 (0.1%) of those being screened for up to 10 years. The same value (1 per 1,000 [0.1%]) was used for an absolute harm threshold. |
The CTS used a threshold of 1% of those screened positive for up to 5 years. |
| Estimation of outcomes of TB infection screening | CATMAT included diagnostic test performance (false positive and false negative) in the calculations. | Not included. |
| Definition of ‘higher TB incidence country’ | Countries with a higher incidence relative to Canada, with an annual reported TB incidence of more than 10 cases per 100,000 population. This threshold is the same cut-off value used in the systematic review and meta-analysisFootnote 1 used by CATMAT to estimate travel-associated TB infection likelihood. |
High TB-incidence countries are defined as having more than 50 cases per 100,000 population. |
| Terminology for Canadian travellers born outside of Canada | CATMAT chose to use the term “non-Canadian-born resident travellers” over “visiting friends and relatives (VFR)”, as it more specifically captures the pre-arrival TB exposure risk of this subpopulation. This terminology was agreed upon through consultation with Canadian TB experts. |
Foreign-born persons/people. |
| OutcomeFootnote a | Rating (score)Footnote b | Rationale |
|---|---|---|
| Prevention of TB disease | Critical 8.5 |
Most travellers would consider prevention of TB disease as meaningful and a key benefit in their decision making. Undetected or missed TB cases (false negative screening results) will be included as part of the model structure (Figure 4) for estimating this benefit (see Methods). |
| Grade 3-5 adverse events (AEs) | Critical 8 |
Most travellers would consider avoiding adverse events of Grade 3 to 5 related to TB treatment, that would result in discontinuation of therapy, as meaningful and a key harm in their decision making. These AEs include hepatotoxicity, flu-like reaction, rash. |
| Unnecessary treatment | Important 6 |
Most travellers would consider avoiding unnecessary treatment as important in their decision making. This outcome considers consequences related to unnecessary treatment such as adverse events and inefficient use of resources including healthcare provider's and patient's time. Given that screening is poorly predictive of progression to disease, the number of cases of TB disease prevented will be lower than the number of persons screened positive who are treated. |
| Drug-drug interactions that would require adjusting medications | Important 4 |
Drug-drug interactions (often medications for hypertension) that would require adjusting medications were considered as potentially important to decision making as they could present additional challenges for healthcare providers to monitor patients and a burden on the patients' time, over a short time period. This outcome would not have major impact on the traveller's decision making. |
| Non-serious adverse effects (e.g. gastro-intestinal upset) | Not important 3 |
Non-serious AEs are not considered as meaningful in decision making to a traveller. |
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 6.21 | 1.03 | 0.31 | 0.05 |
| 6 months | 4.00 | 0.67 | 0.20 | 0.03 |
| 3 months | 2.34 | 0.39 | 0.12 | 0.02 |
| 1 month | 0.88 | 0.15 | 0.04 | 0.01 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 6 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 3 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 1 month | 0.90 | 0.90 | 0.90 | 0.90 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 91.72 | 98.62 | 99.59 | 99.93 |
| 6 months | 94.66 | 99.11 | 99.73 | 99.96 |
| 3 months | 96.88 | 99.48 | 99.84 | 99.97 |
| 1 month | 98.83 | 99.81 | 99.94 | 99.99 |
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 8.71 | 1.45 | 0.44 | 0.07 |
| 6 months | 6.36 | 1.06 | 0.32 | 0.05 |
| 3 months | 4.13 | 0.69 | 0.21 | 0.03 |
| 1 month | 1.71 | 0.29 | 0.09 | 0.01 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 6 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 3 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 1 month | 0.90 | 0.90 | 0.90 | 0.90 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 88.38 | 98.06 | 99.42 | 99.90 |
| 6 months | 91.52 | 98.59 | 99.58 | 99.93 |
| 3 months | 94.50 | 99.08 | 99.72 | 99.95 |
| 1 month | 97.72 | 99.62 | 99.89 | 99.98 |
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 10.71 | 1.78 | 0.54 | 0.09 |
| 6 months | 10.60 | 1.77 | 0.53 | 0.09 |
| 3 months | 10.54 | 1.76 | 0.53 | 0.09 |
| 1 month | 10.49 | 1.75 | 0.52 | 0.09 |
| 2 weeks | 10.48 | 1.75 | 0.52 | 0.09 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 6 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 3 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 1 month | 0.90 | 0.90 | 0.90 | 0.90 |
| 2 weeks | 0.90 | 0.90 | 0.90 | 0.90 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 85.72 | 97.62 | 99.29 | 99.88 |
| 6 months | 85.87 | 97.65 | 99.29 | 99.88 |
| 3 months | 85.95 | 97.66 | 99.30 | 99.88 |
| 1 month | 86.01 | 97.67 | 99.30 | 99.88 |
| 2 weeks | 86.02 | 97.67 | 99.30 | 99.88 |
| Duration of travel | Population (stratified by likelihood of developing TB disease if infected) | |||
|---|---|---|---|---|
| Very High | High | Moderate | Low | |
| Estimated absolute risk reduction in TB disease (%)Footnote a | ||||
| 12 months | 10.93 | 1.82 | 0.55 | 0.09 |
| 6 months | 10.72 | 1.79 | 0.54 | 0.09 |
| 3 months | 10.60 | 1.77 | 0.53 | 0.09 |
| 1 month | 10.52 | 1.75 | 0.53 | 0.09 |
| 2 weeks | 10.49 | 1.75 | 0.52 | 0.09 |
| Estimated absolute increase in risk of serious adverse events (%)Footnote a | ||||
| 12 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 6 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 3 months | 0.90 | 0.90 | 0.90 | 0.90 |
| 1 month | 0.90 | 0.90 | 0.90 | 0.90 |
| 2 weeks | 0.90 | 0.90 | 0.90 | 0.90 |
| Estimated absolute increase in unnecessary treatment (%)Footnote b | ||||
| 12 months | 85.43 | 97.57 | 99.27 | 99.88 |
| 6 months | 85.71 | 97.62 | 99.29 | 99.88 |
| 3 months | 85.87 | 97.64 | 99.29 | 99.88 |
| 1 month | 85.98 | 97.66 | 99.30 | 99.88 |
| 2 weeks | 86.01 | 96.67 | 99.30 | 99.88 |
Sample calculations for values from Table 4 Estimated benefits and harms of TB infection screening in Canadian-born adult travellers, for general travellers in the very high-risk group with a 12-month duration of travel
Note: the baseline risk of TB infection is 2.06% in Canadian-born travellers, 4.30% in healthcare workers, and 25% in non-Canadian-born resident travellers.

Figure 5: Descriptive text
This figure shows how the number of true positives was estimated. The total estimated travellers treated per 100 travellers screened, which has a value of 2.06, is multiplied by the sensitivity of the 10mm tuberculin skin test, which has a value of 81%. The product, thus the estimated number of true positives for travellers in the very high-risk group with a 12-month duration of travel is 1.67.

Figure 6: Descriptive text
This figure shows how the number of false positives was estimated. First, the total number of travellers screened, which has a value of 100, is multiplied by the difference of 1 minus the baseline risk of TB infection, where the baseline risk is 2.06%. The estimated number of true negatives, which has a value of 95.98 (derived using the specificity of the 10mm tuberculin skin test, 98%), is then subtracted from this product. The difference, thus the estimated number of false positives for travellers in the very-high risk group with a 12-month duration of travel is 1.96.

Figure 7: Descriptive text
This figure shows how the absolute risk reduction in TB disease was estimated, as a percentage. First, the sum of two products is found. The first product is calculated by multiplying the estimated number of true positives (1.67) by the annual risk of disease progression over the first two years (3%), which are then multiplied by 2 years. The second product is calculated by multiplying the estimated number of true positives (1.67) by the annual risk of disease progression over the following 8 years (1.5%), which are then multiplied by 8 years. The sum of these two products is then multiplied by the efficacy of rifampin (75%). Thus, the estimated absolute risk reduction in TB disease for travellers in the very-high risk group over a 12-month duration of travel is 0.23%.

Figure 8: Descriptive text
This figure shows how the absolute increase in risk of serious adverse events was estimated, as a percentage. The total estimated travellers treated per 100 travellers screened, which is the sum of true positives and false positives and has a value of 3.63, is multiplied by the risk of Grade 3-5 severe adverse events for rifampin, which is 0.9%. Thus, the estimated absolute increase in risk of serious adverse events for travellers in the very-high risk group with a 12-month duration of travel is 0.03%.

Figure 9: Descriptive text
This figure shows how the absolute increase in unnecessary treatment was estimated, as a percentage. First, the sum of two products is found. The first product is calculated by multiplying the estimated number of true positives (1.67) by the annual risk of disease progression over the first two years (3%), which are then multiplied by 2 years. The second product is calculated by multiplying the estimated number of true positives (1.67) by the annual risk of disease progression over the following 8 years (1.5%), which are then multiplied by 8 years. Next, a difference is calculated by subtracting the sum of these products from the estimated number of true positives, which is 1.67. This difference is then added to the estimated number of false positives, which is 1.96. Thus, the estimated absolute increase in unnecessary treatment for travellers in the very-high risk group with a 12-month duration of travel is 3.33%.
Appendix 4: Evidence to decision framework for question 1
Question: Should Canadian (adult) travellers to higher TB incidence countries be screened post-travel and, for those found to be positive, offered preventive treatment? If so, which travellers, and for what duration of travel?
Population: Canadian-born adults and non-Canadian-born residents travelling from Canada to higher TB incidence countries, defined here as an annual reported TB incidence of >10 cases per 100,000 population.
Intervention: TB screening post travel and preventive treatment for those who screen positive.
Comparison: No screening
Main outcomes: Prevention of TB disease, treatment-associated serious adverse events (SAE), unnecessary treatment, drug-drug interactions
Setting: International travel
Perspective: Individual
Background: TB is a respiratory disease caused by Mycobacterium tuberculosis (MTB). Among Canadian travellers, TB infection may occur following exposure and infection in TB-endemic countries, with subsequent risk of progression to TB disease.
Rates of active TB vary widely among different geographic regions of the world. Intermediate TB incidence countries (10-100 cases per 100,000 population) are generally found in Central and South America, the Middle East, North Africa, eastern Europe, and central Asian countries. Incidence rates are highest (>100 cases per 100,000 population) in southern and south-eastern Asia and sub-Saharan Africa.
In the absence of treatment for TB infection, it can progress to TB disease within 2 years of infection or reactivate to active disease many years later. TB disease in travellers can be prevented through detecting TB infection and providing preventive therapy if the individual’s risk is high enough and the balance of benefits and harms are in favor of this intervention.
The objective of this guidance was to assess the risk of TB infection and disease among travellers, and provide recommendations on which travellers would benefit from TB infection screening and preventive treatment, with a focus on the adult Canadian-born and non-Canadian-born resident travellers.
Conflict of interests: None
Assessment
| Domain | Judgement | Research evidence and additional considerations |
|---|---|---|
Problem: Is the problem a priority? |
No Probably no Probably yes Yes Varies Don't know |
TB remains a major global health challenge, causing an estimated 10.8 million cases and 1.25 million deaths worldwide in 2023Footnote 4. TB elimination is a priority in Canada, and although Canada is considered a low-incidence country for TB, non-Canadian-born residents are one of the populations that are disproportionately affected by TB in CanadaFootnote 5. |
Desirable effects: How substantial are the desirable anticipated effects? |
Trivial Small Moderate Large Varies Don’t know |
The anticipated absolute reduction in risk of TB disease varies based on the population, risk of TB disease progression, and travel duration. For Canadian-born travellers at very high risk, going for a duration of 6 months or greater, the estimated absolute risk reduction in TB disease is moderate (from 0.1-0.2%). For health care worker travellers at very high risk, going for a duration of 3 months or greater, the estimated absolute risk reduction in TB disease is also moderate (0.1-0.5%) (Table 4, Table 5 and Table 16). For non-Canadian-born resident travellers, individuals in the high and very high-risk groups had a moderate-large, and large estimated absolute risk reduction, respectively, ranging from 0.4-2.8% among all those screened (Table 6, Table 7and Table 16). |
Undesirable effects: How substantial are the undesirable anticipated effects? |
Trivial Small Moderate Large Varies Don’t know |
The anticipated absolute risk of SAE with TB preventive treatment varies based on the population, risk of TB disease progression, and travel duration. For Canadian-born adult travellers, the risk of SAE from treatment with rifampin was small (less than 0.1%) for all groups of travellers (Table 4 , Table 5 and Table 16). Although not a critical outcome, there is an overall 2-6% risk of being treated unnecessarily if a Canadian-born traveller were to be screened for TB infection (Table 4 and Table 5). For non-Canadian-born resident travellers, the risk of serious adverse events was moderate for all risk groups, at 0.2%, owing to the higher likelihood of being infected (and treated) (Table 6, Table 7 and Table16). The risk of unnecessary treatment increases to 19-25% for non-Canadian-born resident travellers due to the greater likelihood of screening positive compared to Canadian-born travellers (Table 6 and Table 7). |
Certainty of evidence: What is the overall certainty of the evidence of effects? |
Very low Low Moderate High No included studies |
The overall certainty of evidence for the critical outcomes (likelihood of preventing TB disease and SAE) is low. Important outcomes such as unnecessary treatment and drug-drug interactions were not considered in the overall certainty of evidence. To evaluate the certainty of evidence, we assessed both the model structure and the evidence in the model (model inputs). For the model structure, the working group generally agreed that the model structure was sound and that it reflected the standard pathway of care in Canada. Inconsistency and publication bias were not a concern with a single model. For indirectness, our model outputs generally provided the critical outcomes needed. Limitations were noted in that the model assumes only one trip and that baseline risk estimates are representative of one year of travel. The impact of sequential travel was not considered. To assess imprecision, we conducted sensitivity analyses using different assumptions about the baseline risk (i.e. travel duration, risk of infection by TB incidence), and assessed results using alternative inputs including sensitivity and specificity of IGRA. The sensitivity analyses demonstrated model robustness, as our recommendations largely did not change with varying model inputs (Benefits and harms of screening and treatment of TB infection). To assess evidence in the model, we assessed the certainty of our model inputs. Our lowest certainty of evidence was for the baseline risk of infection among Canadian-born travellers and for the annual risk of disease progression among the ‘High’ risk group. These were assessed as ‘Low’ certainty due to risk of bias and inconsistency (Table 17 and Table18). |
Importance of outcomes to/in/for/in relation to affected population: Is there important uncertainty about or variability in how much travellers value the main outcomes? |
Important uncertainty or variability Possibly important uncertainty or variability Probably no important uncertainty or variability No important uncertainty or variability |
The working group identified the following outcomes as critical or important to decision making: Desirable outcomes:
Undesirable outcomes:
|
Balance of effects: Does the balance between desirable and undesirable effects favour the intervention or the comparison? |
Favours the comparison Probably favours the comparison Does not favour either the intervention or the comparison Probably favours the intervention Favours the intervention Varies Don’t know |
The balance of effects varies based on the population, risk of TB disease progression, and travel duration (Table 16). When the reduction in TB disease was at least moderate and exceeded the estimated increase in treatment associated SAE, then the committee judged that post-travel testing for TB infection provided more benefit than harm. For Canadian-born travellers, the benefits were at least moderate, and harms were low for individuals in the very high-risk group, travelling for 6 months or more. For Canadian-born health care providers, the benefits were at least moderate, and harms were low for individuals in the very high-risk group, travelling for 3 months or more. For non-Canadian-born resident travellers at high and very high risk of disease progression, we placed a higher value on the relatively large estimate of benefit of TB disease prevention compared to the moderate absolute estimate of treatment-related adverse events. For non-Canadian-born resident travellers with low to moderate risk of developing TB disease, the balance of effects did not clearly favor the intervention of TB screening. For non-Canadian-born resident travellers at moderate risk of disease progression, estimates for harms and benefits were both moderate and the estimated increase in SAE likelihood numerically exceeded the estimated reduction in TB disease likelihood. Because the estimates of benefits and harms are relatively balanced, healthcare providers should carefully assess the travellers individual risk, discuss benefits and harms of testing, explore values and preferences, and allow the traveller to make an informed choice as to whether they wish to receive testing for TB infection (Figure 2). |
Resource considerations: How large are the resource requirements (costs) to the individual? |
Large costs Moderate costs Small costs Negligible costs Varies Don’t know |
The TST and TB preventive therapy are covered by provincial health insurance in Canada when they are considered medically necessary. IGRA is covered by some provincial health insurance plans in Canada, while in some provinces there may be out of pocket expenses which are estimated to be around $100Footnote 58. |
Equity: What would be the impact on health equity? |
Reduced Probably reduced Probably no impact Probably increased Increased Varies Don’t know |
CATMAT acknowledges the presence of equity-related issues at the population level, however these considerations do not impact the decision-making for recommendation development by the Committee. CATMAT’s focus on the perspective of supporting the individual traveller does not negate nor reduce the importance of population level challenges. |
Acceptability: Is the intervention acceptable to key stakeholders? |
No Probably no Probably yes Yes Varies Don’t know |
Screening: Screening with TST requires two visits to the healthcare centre, the first to inject the Tubersol®, and a second visit 48 to 72 hours later to read the test. A 2016 systematic review and meta-analysis including 58 studies from multiple countries, reported a completion rate of 72% for TB infection screeningFootnote 47. Treatment: Sandgren et al. found that completion rates for TB infection therapy ranged from 46-95% in the general population highlighting wide varianceFootnote 50. Rifampin is a treatment regimen taken for a duration of 4 months, requiring daily doses for 120 days. It is an oral capsule taken on an empty stomach. Observational and experimental studies have demonstrated higher treatment completion rates with shorter course rifamycin-based regimens than with the longer alternative therapy with 9 months of isoniazidFootnote 47Footnote 50Footnote 52. Recommendations for screening are predicated on patients who test positive accepting treatment. |
Feasibility: Is the intervention feasible to implement for the individual? |
No Probably no Probably yes Yes Varies Don’t know |
Multiple social and health system barriers contribute to losses at different steps along the care cascade, including medical contraindications, language barriers and self-perceived low risk of infectionFootnote 47. Socio-economic factors such as unemployment or lack of social support have also been shown to have an impact on missed screening and poor compliance with TB infection therapyFootnote 52.Commonly reported barriers to treatment initiation and adherence include perception that TB is not a serious disease, self-perceived low risk of TB disease, concern about treatment toxicity and the long duration of therapy for some regimensFootnote 47. |
Type of recommendation
Discretionary recommendation for the intervention (post-travel TB infection testing). See Recommendations section.
| Type of traveller | Risk of TB disease | Travel duration | Size of desirable effects | Size of undesirable effects | Balance of harms and benefits |
|---|---|---|---|---|---|
| Canadian-born traveller | Very high | 12 months | Moderate | Small | Probably favours the intervention (TB testing) |
| 6 months | Moderate | Small | Probably favours the intervention (TB testing) | ||
| 3 months | Small | Small | Probably favours the comparison (no testing) | ||
| Canadian-born healthcare worker | Very high | 12 months | Moderate | Small | Probably favours the intervention (TB testing) |
| 6 months | Moderate | Small | Probably favours the intervention (TB testing) | ||
| 3 months | Moderate | Small | Probably favours the intervention (TB testing) | ||
| Non-Canadian-born resident general traveller | Very high | 2 weeks | Large | Moderate | Probably favours the intervention (TB testing) |
| High | 2 weeks | Moderate-large | Moderate | Probably favours the intervention (TB testing) | |
| Moderate | 2 weeks | Moderate | Moderate | Does not favour either the intervention or the comparison. | |
| Non-Canadian-born resident healthcare worker | Very high | 2 weeks | Large | Moderate | Probably favours the intervention (TB testing) |
| High | 2 weeks | Moderate-large | Moderate | Probably favours the intervention (TB testing) | |
| Moderate | 2 weeks | Moderate | Moderate | Does not favour either the intervention or the comparison. |
| Number of studies | Certainty assessment | Effect | Certainty | Importance | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Study design | Risk of bias | Inconsistency | Indirectness | Imprecision | Other considerations | Number of events | Number of individuals | Rate (95% CI) |
|||
| General traveller | |||||||||||
| 8 | Non-randomised studies | SeriousFootnote a | SeriousFootnote b | Not serious | Not serious | None | 13,108 | 1,154,230 | Event rate 2.06 per 100 (1.49 to 2.86) |
LowFootnote aFootnote b | Critical |
| Healthcare workers | |||||||||||
| 3 | Non-randomised studies | SeriousFootnote a | Not serious | Not serious | Not seriousFootnote c | None | 19 | 443 | Event rate 4.30 per 100 (2.77 to 6.68) |
ModerateFootnote a | Critical |
| Number of studies | Certainty assessment | Effect | Certainty | Importance | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Study design | Risk of bias | Inconsistency | Indirectness | Imprecision | Other considerations | Number of events | Number of individuals | Annual Rate (range) |
|||
| Annual risk of disease: Very high | |||||||||||
| 20 | Non-randomised studies | SeriousFootnote a | Not serious | Not serious | Not serious | None | 230 | 2,210 | Median 3 % (1.7 to 3.7) | ModerateFootnote aFootnote bFootnote c | Critical |
| Annual risk of disease: High | |||||||||||
| 36 | Non-randomised studies | SeriousFootnote a | SeriousFootnote b | Not serious | Not serious | None | 60 | 2,081 | Median 0.5 % (0.1 to 1.2) | LowFootnote aFootnote bFootnote d | Critical |
Appendix 5. Assessment of good practice statement
Good practice statement: Should travellers wish to undertake infection prevention measures, healthcare providers should advise them to consider wearing a N95 respirator if they will be in a high-risk setting, and/or to avoid these settings (e.g., crowded, poorly ventilated indoor environments).
| Criteria | Supporting information | Verification |
|---|---|---|
| Population and intervention components are clear. | Not applicable. | Yes |
| Message is necessary in regard to actual health care practice. | Infection prevention measures are important interventions to prevent exposure to communicable infections. | Yes |
| Implementing the GPS results in a large net positive consequence. | Due to the route of transmission of Mycobacterium tuberculosis bacteria through the airborne route, minimizing exposure in settings where the risk of transmission is high greatly reduces risk of infection. | Yes |
| Collecting and summarizing the evidence is a poor use of a guideline panel's limited time, energy or resources. The opportunity cost of collecting and summarizing the evidence is large and can be avoided. | Yes, this recommendation is about advising travellers to protect themselves. | Yes |
| There is a well-documented clear and explicit rationale connecting the indirect evidence. | Yes, minimizing exposure and using a N95 respirator are reasonably expected to reduce likelihood of infection, particularly since aerosol droplets can remain suspended in air and be transported significant distances from the source case. | Yes |
| Clear and actionable. | Not applicable. | Yes |
Final judgement: Development of GPS is appropriate.
References
- Footnote 1
-
Diefenbach-Elstob TR, Alabdulkarim B, Deb-Rinker P, Pernica JM, Schwarzer G, Menzies D, et al. Risk of latent and active tuberculosis infection in travellers: a systematic review and meta-analysis. Journal of Travel Medicine. 2020.
- Footnote 2
-
Campbell JR, Winters N, Menzies D. Absolute risk of tuberculosis among untreated populations with a positive tuberculin skin test or interferon-gamma release assay result: systematic review and meta-analysis. Bmj. 2020;368:m549.
- Footnote 3
-
Campbell JR, Christopher P, Peter D, Madhukar P, and Menzies D. Chapter 4: Diagnosis of tuberculosis infection. Canadian Journal of Respiratory, Critical Care, and Sleep Medicine. 2022;6(sup1):49-65.
- Footnote 4
-
Global tuberculosis report 2024. Geneva: World Health Organization; 2024. Contract No.: Licence: CC BY-NC-SA 3.0 IGO.
- Footnote 5
-
Government of Canada's Tuberculosis Response (2025). Public Health Agency of Canada; 2025.
- Footnote 6
-
Global tuberculosis report 2022. World Health Organization; 2022.
- Footnote 7
-
Vynnycky E, Fine PEM. Lifetime Risks, Incubation Period, and Serial Interval of Tuberculosis. American Journal of Epidemiology. 2000;152(3):247-63.
- Footnote 8
-
Denholm JT, Thevarajan I. Tuberculosis and the traveller: evaluating and reducing risk through travel consultation. J Travel Med. 2016;23(3).
- Footnote 9
-
Freeman RJ, Mancuso JD, Riddle MS, Keep LW. Systematic review and meta-analysis of TST conversion risk in deployed military and long-term civilian travelers. J Travel Med. 2010;17(4):233-42.
- Footnote 10
-
Saiman L, Gabriel PS, Schulte J, Vargas MP, Kenyon T, Onorato I. Risk factors for latent tuberculosis infection among children in New York City. Pediatrics. 2001;107(5):999-1003.
- Footnote 11
-
Focus on Geography Series, 2021 Census of Population: Statistics Canada; 2021 [Available from: https://www12.statcan.gc.ca/census-recensement/2021/as-sa/fogs-spg/page.cfm?topic=9&lang=E&dguid=2021A000011124
- Footnote 12
-
Jonas DE, Riley SR, Lee LC, Coffey CP, Wang S-H, Asher GN, et al. Screening for Latent Tuberculosis Infection in Adults: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2023;329(17):1495-509.
- Footnote 13
-
Sterling TR. Guidelines for the treatment of latent tuberculosis infection: recommendations from the National Tuberculosis Controllers Association and CDC, 2020. MMWR Recommendations and Reports. 2020;69.
- Footnote 14
-
Menzies D, Alvarez G, Khan K. Chapter 6: Treatment of latent tuberculosis infection. Canadian Tuberculosis Standards. 7th ed. Ottawa: Public Health Agency of Canada; 2014.
- Footnote 15
-
Menzies NA, Swartwood N, Testa C, Malyuta Y, Hill AN, Marks SM, et al. Time Since Infection and Risks of Future Disease for Individuals with Mycobacterium tuberculosis Infection in the United States. Epidemiology. 2021;32(1):70-8.
- Footnote 16
-
Jordan AE, Nsengiyumva NP, Houben RM, Dodd PJ, Dale KD, Trauer JM, et al. The prevalence of tuberculosis infection among foreign-born Canadians: a modelling study. CMAJ. 2023;195(48):E1651-E9.
- Footnote 17
-
Menzies D, Long R, Trajman A, Dion M-J, Yang J, Al Jahdali H, et al. Adverse events with 4 months of rifampin therapy or 9 months of isoniazid therapy for latent tuberculosis infection: a randomized trial. Annals of Internal Medicine. 2008;149(10):689-97.
- Footnote 18
-
Menzies D, Adjobimey M, Ruslami R, Trajman A, Sow O, Kim H, et al. Four months of rifampin or nine months of isoniazid for latent tuberculosis in adults. New England Journal of Medicine. 2018;379(5):440-53.
- Footnote 19
-
Lotfi T, Hajizadeh A, Moja L, Akl EA, Piggott T, Kredo T, et al. A taxonomy and framework for identifying and developing actionable statements in guidelines suggests avoiding informal recommendations. Journal of Clinical Epidemiology. 2022;141:161-71.
- Footnote 20
-
Brozek JL, Canelo-Aybar C, Akl EA, Bowen JM, Bucher J, Chiu WA, et al. GRADE Guidelines 30: the GRADE approach to assessing the certainty of modeled evidence-An overview in the context of health decision-making. J Clin Epidemiol. 2021;129:138-50.
- Footnote 21
-
Alonso-Coello P, Schünemann HJ, Moberg J, Brignardello-Petersen R, Akl EA, Davoli M, et al. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. Bmj. 2016;353:i2016.
- Footnote 22
-
Dewidar O, Lotfi T, Langendam MW, Parmelli E, Saz Parkinson Z, Solo K, et al. Good or best practice statements: proposal for the operationalisation and implementation of GRADE guidance. BMJ Evidence-Based Medicine. 2023;28(3):189-96.
- Footnote 23
-
Baussano I, Nunn P, Williams B, Pivetta E, Bugiani M, Scano F. Tuberculosis among health care workers. Emerging Infectious Diseases. 2011;17(3):488-94.
- Footnote 24
-
van leth F. Prevalence of tuberculous infection and incidence of tuberculosis; a re-assessment of the Styblo rule. Bulletin of the World Health Organization. 2008;86(1):20-6.
- Footnote 25
-
Houben RM, Dodd PJ. The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLOS Medicine. 2016;13(10):e1002152.
- Footnote 26
-
Boggild AK, Geduld J, Libman M, Ward BJ, McCarthy AE, Doyle PW, et al. Travel-acquired infections and illnesses in Canadians: surveillance report from CanTravNet surveillance data, 2009-2011. Open Med. 2014;8(1):e20-32.
- Footnote 27
-
Boggild AK, Geduld J, Libman M, Yansouni CP, McCarthy AE, Hajek J, et al. Spectrum of illness in migrants to Canada: sentinel surveillance through CanTravNet. J Travel Med. 2019;26(2).
- Footnote 28
-
Gautret P, Cramer JP, Field V, Caumes E, Jensenius M, Gkrania-Klotsas E, et al. Infectious diseases among travellers and migrants in Europe, EuroTravNet 2010. Euro Surveill. 2012;17(26).
- Footnote 29
-
Leder K, Tong S, Weld L, Kain KC, Wilder-Smith A, von Sonnenburg F, et al. Illness in travelers visiting friends and relatives: a review of the GeoSentinel Surveillance Network. Clin Infect Dis. 2006;43(9):1185-93.
- Footnote 30
-
Leder K, Torresi J, Libman MD, Cramer JP, Castelli F, Schlagenhauf P, et al. GeoSentinel surveillance of illness in returned travelers, 2007-2011. Annals of Internal Medicine. 2013;158:456-68.
- Footnote 31
-
Warne B, Weld LH, Cramer JP, Field VK, Grobusch MP, Caumes E, et al. Travel-related infection in European travelers, EuroTravNet 2011. J Travel Med. 2014;21(4):248-54.
- Footnote 32
-
Wikman-Jorgensen P, López-Velez R, Llenas-García J, Treviño B, Pascual R, Molina I, et al. Latent and active tuberculosis infections in migrants and travellers: a retrospective analysis from the Spanish +REDIVI collaborative network. Travel Med Infect Dis. 2020;36:101460.
- Footnote 33
-
Brown AB. Travel-related diagnoses among US nonmigrant travelers or migrants presenting to US GeoSentinel sites—GeoSentinel Network, 2012–2021. MMWR Surveillance Summaries. 2023;72.
- Footnote 34
-
Khan A, Phares CR, Phuong HL, Trinh DTK, Phan H, Merrifield C, et al. Overseas Treatment of Latent Tuberculosis Infection in US–Bound Immigrants. Emerging Infectious Diseases. 2022;28(3):582.
- Footnote 35
-
Collins JM, Stout JE, Ayers T, Hill AN, Katz DJ, Ho CS, et al. Prevalence of latent tuberculosis infection among non-US-born persons by country of birth—United States, 2012–2017. Clinical Infectious Diseases. 2021;73(9):e3468-e75.
- Footnote 36
-
Ding C, Hu M, Guo W, Hu W, Li X, Wang S, et al. Prevalence trends of latent tuberculosis infection at the global, regional, and country levels from 1990–2019. International Journal of Infectious Diseases. 2022;122:46-62.
- Footnote 37
-
Yelk Woodruff R, Hill A, Marks S, Navin T, Miramontes R. Estimated latent tuberculosis infection prevalence and tuberculosis reactivation rates among non-US-born residents in the United States, from the 2011–2012 National Health and Nutrition Examination Survey. Journal of Immigrant and Minority Health. 2021;23:806-12.
- Footnote 38
-
McCarthy O. Asian immigrant tuberculosis—the effect of visiting Asia. British journal of diseases of the chest. 1984;78:248-53.
- Footnote 39
-
Ormerod L, Green R, Gray S. Are there still effects on Indian Subcontinent ethnic tuberculosis of return visits?: a longitudinal study 1978–97. Journal of Infection. 2001;43(2):132-4.
- Footnote 40
-
Kik S, Mensen M, Beltman M, Gijsberts M, Van Ameijden E, Cobelens F, et al. Risk of travelling to the country of origin for tuberculosis among immigrants living in a low-incidence country. The International journal of tuberculosis and lung disease. 2011;15(1):38-43.
- Footnote 41
-
Greenaway C, Khan K, Schwartzman K. Chapter 13: Tuberculosis surveillance and screening in selected high-risk populations. Canadian Tuberculosis Standards. 7th ed. Ottawa: Public Health Agency of Canada; 2014.
- Footnote 42
-
Menzies NA, Wolf E, Connors D, Bellerose M, Sbarra AN, Cohen T, et al. Progression from latent infection to active disease in dynamic tuberculosis transmission models: a systematic review of the validity of modelling assumptions. The Lancet Infectious Diseases. 2018;18(8):e228-e38.
- Footnote 43
-
Barnett GD, Grzybowski S, Styblo K. Present risk of developing active tuberculosis in Saskatchewan according to previous tuberculin and X-ray status. Selected Papers of the Royal Netherlands Tuberculosis Association. 1971;14:51–74.
- Footnote 44
-
Young J, O'Connor ME. Risk factors associated with latent tuberculosis infection in Mexican American children. Pediatrics. 2005;115(6):e647-53.
- Footnote 45
-
Bottieau E, Florence E, Clerinx J, Vlieghe E, Vekemans M, Moerman F, et al. Fever after a stay in the tropics: clinical spectrum and outcome in HIV-infected travelers and migrants. J Acquir Immune Defic Syndr. 2008;48:547-52.
- Footnote 46
-
Menzies D, Dion MJ, Rabinovitch B, Mannix S, Brassard P, Schwartzman K. Treatment completion and costs of a randomized trial of rifampin for 4 months versus isoniazid for 9 months. Am J Respir Crit Care Med. 2004;170(4):445-9.
- Footnote 47
-
Alsdurf H, Hill PC, Matteelli A, Getahun H, Menzies D. The cascade of care in diagnosis and treatment of latent tuberculosis infection: a systematic review and meta-analysis. The Lancet Infectious Diseases. 2016;16(11):1269-78.
- Footnote 48
-
O’Connell J, de Barra E, McConkey S. Systematic review of latent tuberculosis infection research to inform programmatic management in Ireland. Irish Journal of Medical Science (1971 -). 2022;191(4):1485-504.
- Footnote 49
-
Yang H, Park H. Factors influencing the initiation and adherence of LTBI treatment in healthcare workers: a systematic review. Archives of Environmental & Occupational Health. 2022;77(1):76-86.
- Footnote 50
-
Sandgren A, Vonk Noordegraaf-Schouten M, van Kessel F, Stuurman A, Oordt-Speets A, van der Werf MJ. Initiation and completion rates for latent tuberculosis infection treatment: a systematic review. BMC Infectious Diseases. 2016;16(1):204.
- Footnote 51
-
Rustage K, Lobe J, Hayward SE, Kristensen KL, Margineanu I, Stienstra Y, et al. Initiation and completion of treatment for latent tuberculosis infection in migrants globally: a systematic review and meta-analysis. The Lancet Infectious Diseases. 2021;21(12):1701-12.
- Footnote 52
-
Stuurman AL, Vonk Noordegraaf-Schouten M, van Kessel F, Oordt-Speets AM, Sandgren A, van der Werf MJ. Interventions for improving adherence to treatment for latent tuberculosis infection: a systematic review. BMC Infectious Diseases. 2016;16(1):257.
- Footnote 53
-
Hirsch-Moverman Y, Daftary A, Franks J, Colson P. Adherence to treatment for latent tuberculosis infection: systematic review of studies in the US and Canada. The International journal of tuberculosis and lung disease. 2008;12(11):1235-54.
- Footnote 54
-
Duval B, De Serre G, Shadmani R, Boulianne N, Pohani G, Naus M, et al. A Population-based Comparison between Travelers Who Consulted Travel Clinics and Those Who Did Not. Journal of Travel Medicine. 2006;10(1):4-10.
- Footnote 55
-
Pathak V, Harrington Z, Dobler CC. Attitudes towards preventive tuberculosis treatment among hospital staff. PeerJ. 2016;4:e1738.
- Footnote 56
-
Bhanot N, Haran M, Lodha A, Paul V, Goswami R, Chapnick E. Physicians' attitudes towards self-treatment of latent tuberculosis. The International journal of tuberculosis and lung disease. 2012;16(2):169-71.
- Footnote 57
-
Dobler CC, Luu Q, Marks GB. What patient factors predict physicians’ decision not to treat latent tuberculosis infection in tuberculosis contacts? PLoS ONE. 2013;8(9):e76552.
- Footnote 58
-
QUANTIFERON-TB GOLD Plus (FOR TB SCREENING): LifeLabs; [Available from: https://www.lifelabs.com/test/quantiferon-tb-gold/?myProvince=on.
- Footnote 59
-
Tan M, Menzies D, Schwartzman K. Tuberculosis screening of travelers to higher-incidence countries: A cost-effectiveness analysis. BMC Public Health. 2008;8(1):201.
- Footnote 60
-
Kowada A. Cost effectiveness of interferon-gamma release assay for tuberculosis screening using three months of rifapentine and isoniazid among long-term expatriates from low to high incidence countries. Travel Medicine and Infectious Disease. 2016;14(5):489-98.
- Footnote 61
-
Campbell JR, Nsengiyumva P, Chiang LY, Jamieson F, Khadawardi H, Mah HK, et al. Costs of Tuberculosis at 3 Treatment Centers, Canada, 2010-2016. Emerg Infect Dis. 2022;28(9):1814-23.
- Footnote 62
-
Long R, Divangahi M, Schwartzman K. Chapter 2: Transmission and pathogenesis of tuberculosis. Canadian Journal of Respiratory, Critical Care, and Sleep Medicine. 2022;6(sup1):22-32.
- Footnote 63
-
Johnston BL, Ogunremi T, Defalco K, Savard N, Smith SW. Chapter 14: Prevention and control of tuberculosis transmission in healthcare settings. Canadian Journal of Respiratory, Critical Care, and Sleep Medicine. 2022;6(sup1):205-28.
- Footnote 64
-
Jensen PA, Lambert LA, Iademarco MF, Ridzon R, Control CfD, Prevention. Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. 2005.
- Footnote 65
-
The Department of Health (UK). Tuberculosis: the green book, chapter 32. Immunisation against infectious diseases (The Green Book)2011.
- Footnote 66
-
World Health Organization. Chapter 6: Vaccine-preventable diseases and vaccines. International Travel and Health2017.
- Footnote 67
-
Behr M, Elwood K. Chapter 16: Bacille Calmette-Guérin (BCG) vaccination in Canada. Canadian Tuberculosis Standards. 7th ed. Ottawa: Public Health Agency of Canada; 2014.
- Footnote 68
-
Shamputa IC, Nguyen DTK, Mackenzie H, Gaudet DJ, Harquail A, Barker K, et al. Enhanced screening for tuberculosis infection among immigrants in southern New Brunswick: A cross-sectional pilot study. Canada Communicable Disease Report. 2025;51(5).