Pan-Canadian Action Plan on Antimicrobial Resistance: Year 2 Progress Report (June 2024 to May 2025)
Download in PDF format
(5.32 MB, 38 pages)
Related links
Contents
- Introduction
- Pillar 1: Research and innovation
- Pillar 2: Surveillance
- Pillar 3: Stewardship
- Pillar 4: Infection, Prevention and Control
- Pillar 5: Leadership
- Look Ahead
- Annex A: Strengthening progress monitoring
- Annex B: Additional Materials
- Glossary
Introduction
Canada is taking concrete actions to reduce the development and spread of antimicrobial resistance (AMR). The Pan-Canadian Action Plan (PCAP) on Antimicrobial Resistance (AMR) applies a collaborative One Health approach with federal, provincial and territorial (FPT) governments, to implement the ten shared priority actions in response to the growing threat of AMR. These committed actions span across five pillars: 1) Research and Innovation, 2) Surveillance, 3) Stewardship, 4) Infection Prevention and Control (IPC), and 5) Leadership.
In September 2024 Canada published its first progress report on the PCAP entitled Year 1 Progress Report: June 2023–May 2024, which outlined key FPT accomplishments such as Canada's Genomics Research and Development Initiative (GRDI) on AMR (AMR-One Health) and Nova Scotia's AMR Action Plan. The Year 1 Report was released ahead of the United Nations General Assembly (UNGA) High-Level Meeting (HLM) on AMR, and was supported by a joint written statement from Canada's Chief Public Health Officer and Chief Veterinary Officer which reaffirmed Canada's ongoing commitment to addressing this global health threat.
The Year 2 Progress Report provides an update on the continuing progress made in implementing the PCAP from June 1, 2024, to May 31, 2025. Across all pillars, FPT partners are strengthening domestic and global leadership on AMR in collaboration with stakeholders across One Health sectors.
Canada is committed to continuing to develop subsequent progress reports which will capture and reflect new evidence, projects, timelines, resource context as well as challenges. It is important to note that key PCAP activities have varying timelines and resources.
Pillar 1: Research and innovation
Research and innovation play a crucial role in tackling the growing threat of AMR in Canada. The PCAP highlights the need for multi-disciplinary research and innovation to secure new antimicrobials for human use, facilitate sustainable access, develop diagnostics and alternatives, including vaccines, and continue to strengthen a One Health approach to research and development.
Priority Actions:
- 1.1 Develop and implement economic and/or regulatory incentives to support innovation and facilitate sustainable access to new and existing antimicrobials, diagnostics, and alternatives to antimicrobials.
- 1.2 Develop a One Health, national research strategy for combatting AMR across all Action Plan pillars.
The Public Health Agency of Canada (PHAC), in collaboration with Health Canada (HC) and other federal partners workedto develop a pilot project with a goal of increasing access to high-priority antimicrobials for humans that have not yet been granted market authorization in Canada. To support this, PHAC consulted with drug manufacturers, FPT counterparts, hospital clinicians, health authorities, professional associations and networks to inform the development and design of the Antimicrobial Economic Incentives Pilot Project for Canada.
As announced in 2023, PHAC provided funding to Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator (CARB-X) until March 2025. Over the last year, this investment supported CARB-X's 2025 request for proposals to support projects that address critical gaps in AMR. A total of 333 Expressions of Interest were submitted across the specified themes, and projects are expected to be published later in 2025.
Spotlight: One Health AMR Research Strategy
The Canadian Institutes of Health Research (CIHR) and Agriculture and Agri-Food Canada (AAFC), with support from PHAC and Environment and Climate Change Canada (ECCC), continue to develop a National One Health AMR Research Strategy (NOHARS). The purpose of the strategy is to help prioritize and coordinate AMR research, including supporting a collaborative One Health approach that integrates human, animal, agriculture and environmental research priorities. Over the past year, a list of potential priorities was developed and refined through a pan-Canadian survey and other engagement activities, including conference workshops and focus groups. The next step is to consult with Indigenous partners and create a strategy document that will include the results of a federally-funded AMR research mapping exercise, and a list of research priorities.
Additional Research and Innovation activities underway include:
- AAFC, the Canadian Food Inspection Agency (CFIA), Department of Fisheries and Oceans Canada (DFO), ECCC, HC, PHAC and the National Research Council Canada (NRC) participated in the Genomics Research and Development Initiative Shared Priority Project (GRDI SPP) on AMR One Health (2022-2027), which aims to continue research on risks associated with the emergence and dissemination of AMR across the One Health continuum. This includes research on novel activities, the environmental dimensions of AMR, and the integration of genomics data into risk assessment models. Key achievements include:
- Releasing 88 new peer-reviewed publications during the first three years of the project. Additionally, 140 public communication activities, including conferences and presentations took place; and
- Launching the GRDI-AMR National Centre for Biotechnology Information (NCBI) BioProject to facilitate public access to genomic data generated through the GRDI SPP project on AMR One Health. The project developed 15 tools to process samples, analyze data, and coordinate data submission to the public repository.
- CIHR announced that it will provide $5.4M in funding over 6 years (2024-2030) to support the creation of two training platforms focused on building One Health research capacity: 1) the Canadian Antimicrobial Resistance Network; and 2) the Canadian One Health Training program on Emerging Zoonoses. The training platforms will bring together mentors from a variety of disciplines, sectors and jurisdictions to equip trainees and early career researchers with the foundational skills and knowledge required to apply One Health approaches to combat AMR and respond to emerging zoonoses.
- Innovation, Science and Economic Development Canada (ISED), as part of the Biomanufacturing and Life Sciences Strategy and in partnership with HC, supported the domestic biomanufacturing and life sciences sector which could be leveraged to produce vaccines and therapeutics against AMR threats.
- NRC continued its research and development (R&D) efforts to develop innovative alternatives to antibiotics through:
- Funding a new 3-year research project (total project value $4.3M) to support the development of new phage therapies for combatting AMR;
- Providing scientific expertise to support Canadian small-medium enterprises in designing, developing and testing novel antimicrobial products; and
- Collaborating with PHAC to advance phage therapy and susceptibility testing.
- AAFC supported several research projects and technologies that aim to develop antimicrobial alternatives to combat diseases in poultry, swine, beef, crops as well as research on environmental transmission of AMR. This past year, research was conducted on the control of necrotic enteritis; bovine respiratory disease; phage-carrier biopesticides in orchards; and a genomic assessment of AMR transmission in farms.
- The International Development Research Centre (IDRC) continued to deliver the Innovative Veterinary Solutions for Antimicrobial Resistance (InnoVet-AMR) 2.0 (2023 – 2027) and provided $26.3M in funding to support 16 projects. Of these international projects, 14 are focused on developing alternatives to antimicrobials that can target multiple pathogens for livestock such as ruminants, pigs, poultry, and aquaculture.
- DFO conducted research on AMR in aquaculture environments and identified antibiotic resistance genes (ARG) in both farm-impacted and non-impacted sediments, including resistance to antimicrobials not used in salmon farming. Progress was made in implementing cost-effective ARG detection methods and facilitating improved AMR surveillance, which contributed to multiple scientific publications and presentations.
- The Government of British Columbia (BC) contributed by:
- Providing funding to researchers from BC and Manitoba who created an e-Book summarizing evidence that reducing unnecessary antibiotic exposure during infancy and promoting breastfeeding can contribute to reversing the global pandemic of asthma and atopic disease; and
- Establishing a Special Access Program for drug stockpiles in Lower Mainland BC, primarily to treat Carbapenemase-producing organism (CPO) infections. This stockpile is supported by CPO treatment guidelines.
- Public Health Ontario's (PHO) Comprehensive Ontario Microbiology laboratory Administrative data for Antimicrobial Resistance (COMBAT-AMR) research program continued to facilitate the updating of a province-wide microbiology database, linked to administrative datasets at ICES. The program advances knowledge about the prevalence and outcomes of AMR, and this year's research publications included topics such as:
- The relative increase in mortality associated with antimicrobial resistant bloodstream infections;
- Identifying risk factors and outcomes of persistent gram-negative bloodstream infections;
- Identifying the prevalence and predictors of concomitant bacterial co-infections in patients with respiratory viruses;
- Quantifying patient harms and benefits associated with prolonged antibiotic therapy and
- Reviewing the evidence for the efficacy of short vs long duration antibiotic treatment for urinary tract infections in companion animals.
- The Government of Prince Edward Island (PEI) funded a number of AMR research projects including:
- A collaborative project between The Atlantic Veterinary College (AVC) at University of Prince Edward Island (UPEI) and the University of Groningen to expand and increase the accessibility of an open-source R package Footnote 1 that helps with AMR analysis and prediction, entitled AMR for R. With the support of UPEI and AVC, the package now supports veterinary microbiology data analysis.
- Collecting baseline data on parasites of PEI sheep flocks and their patterns of AMR. The project will collect data over 2 years and enroll 10 flocks and 450 lambs. The results of the project will be presented to sheep farmers to improve understanding of resistance and appropriate parasite prevention and treatment strategies.
- The Government of PEI also contributed funding for an investigation by UPEI researchers working with The Canadian Dairy Network for Antimicrobial Stewardship and Resistance into the AMR of zoonoses (E. coli, Salmonella, Campylobacter) and bovine respiratory disease pathogens. This research aims at offering evidence-based tools and resources to help reduce AMU, prevent or reduce AMR, and enhance animal health on dairy farms.
Pillar 2: Surveillance
The Surveillance pillar is the foundation of Canada's ability to detect and understand AMR threats. It is closely linked to other pillars such as Research and Innovation, Stewardship, and Infection Prevention and Control and Leadership, and supports the development of evidence-based decisions and policies that address AMR. Access to reliable surveillance data improves Canada's ability to effectively respond to AMR threats across all One Health threats.
Priority Actions:
- 2.1 Maintain sources, coverage and integration of AMR and antimicrobial use (AMU) surveillance data, including the use of modern laboratory technologies and standardized reporting, to help monitor AMR/AMU across One Health sectors, with specific focus on: improving data from the environment; transmission pathways between sectors; and population groups disproportionately impacted by AMR and inappropriate AMU.
- 2.2 Work with partners to:
- establish baselines and targets for national, provincial and territorial levels of AMR and appropriate AMU in human health.
- establish baselines, goals and measures of progress for increasing appropriate AMU and reducing AMR in the agriculture and agri-food sectors.
PHAC released an updated AMR Priority Pathogen List which replaced the original 2015 list. Prioritizing AMR pathogens helps focus resources used for strengthening human surveillance and stewardship activities, improving IPC measures and supporting the development of new treatment and diagnostic strategies. Of the 155 pathogens assessed, 68 (44%) showed evidence of AMR, and 29 were selected for detailed analysis using a multi-criteria decision analysis framework based on nine weighted criteria Footnote 2 . This methodology, aligned with international best practices, was tailored to Canadian priorities and used national data from 2017 to 2022. Notably, Canada is the first country to include health equity as a prioritization criterion, reinforcing its commitment to addressing health disparities and ensuring that disproportionately affected populations are considered in future AMR strategies.
In November 2024, HC and PHAC jointly published the 2023 Veterinary Antimicrobial Sales Highlights Report, and updated the existing Veterinary Antimicrobial Sales Reporting (VASR) dashboard. The report summarizes key findings from the VASR system, which is a component of the Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS), and provides a summary of medically important antimicrobial sales intended for use in animals. For the first time, sales of medically important antimicrobials for cattle were stratified by production type (dairy cattle, beef cattle, veal calves) using biomass estimates (population and weight measures) specific to each group. In addition, provincial-level sales of medically important antimicrobials for animals were adjusted by province-specific animal biomass estimates. This adjustment more accurately reflects provincial antimicrobial sales by taking into account the different animal industries in each province. PHAC's CIPARS also published a new dashboard for farm AMU with up to 2023 data. CIPARS also hosted its annual CIPARS stakeholder webinar to share key findings from the most recent surveillance data. The webinar included a main presentation (PDF) of integrated data across surveillance components, followed by five concurrent presentations of sector-specific data in humans, poultry, dairy cattle, feedlot cattle and grower-finisher pigs. Additional data shared included:
- data on retail seafood;
- data on AMR from irrigation and surface water samples (i.e., from the environment) from PHAC's FoodNet Canada surveillance system;
- data on AMR in Salmonella from environmental samples collected for diagnostic purposes;
- information on the prevalence of AMR in feed ingredients and mixed feed based on data routinely collected and provided by CFIA; and
- as well as prevalence of AMR in sick animals, including CIPARS data on bovine respiratory disease pathogens in healthy feedlot cattle and AMRNetVet data on bovine respiratory disease in sick cattle.
Spotlight: Public Health Agency of Canada’s Environmental Surveillance Strategic Framework
To advance environmental surveillance of AMR, PHAC developed the Environmental Surveillance Strategic Framework (ESSF) for AMR. This framework will guide PHAC's efforts to enhance access to data on AMR from environmental sources, including drivers that can contribute to its emergence and spread, and transmission pathways among One Health sectors. The ESSF proposes a series of short, medium, and long-term actions aimed at strengthening data availability and integration through collaboration with partners. These actions support PHAC's goal of implementing a One Health approach to detect, understand and mitigate the impacts of AMR in Canada. Notably, the ESSF is among the first frameworks, at the international level, that focuses on environmental AMR surveillance and reinforcing Canada's leadership in this emerging field.
Spotlight: Public Health Ontario's – Strengthening Ontario Antimicrobial Resistance Surveillance (SOARS) initiative
PHO's Strengthening Ontario Antimicrobial Resistance Surveillance (SOARS) initiative aims to improve access to comprehensive AMR and AMU data by leveraging technical expertise and capacity developed through the COMBAT-AMR initiative and operationalizing the use of Ontario Laboratories Information System (OLIS) and other provincial data assets. Examples of work that has been undertaken to support this initiative include:
- An enhanced epidemiological summary of Candida auris (C. auris) cases in Ontario (PDF), in response to the January 1, 2025 designation of C. auris as a Disease of Public Health Significance;
- A re-designed interactive dashboard that will better support AMR and AMU surveillance, and will replace the existing Ontario Antimicrobial Stewardship Program & AMR Comparison Tool; and
- Additionally, a new state-of-the-art genomics laboratory at PHO will enable ongoing innovative research, including continuous development of novel genomics testing methodologies that will support pathogen surveillance and enhance public health outcomes across Ontario.
Spotlight: Québec's Ministry of Agriculture, Fisheries and Food (MAPAQ)
Monitoring antibiotic use and bacterial resistance to antibiotics is essential in animal health and for combatting AMR. The Government of Québec's Ministry of Agriculture, Fisheries and Food (French only) (MAPAQ) continues to implement activities that explore antibiotic use in Québec including:
- Continuing to collect sales records (French only) for medicated premixes and medicated feeds from feed mills that prepare and distribute feed under the Regulation respecting medicated premixes and medicated feed for animals (chapter P-42, r. 10);
- Collaborating with feed mills that prepare and distribute animal feed to ensure validity and quality of the data submitted; and
- Improving the interactive report (French only) of theQuébec Veterinary Antibiotic Surveillance Program (French only) by adding the results of antibiotic resistance obtained in dogs, cats, and horses. These species are added to those already covered in the report, namely poultry, pigs, cattle, small ruminants, fish, and bees.
Additional Surveillance activities underway include:
- PHAC supported surveillance efforts by:
- Releasing the 2024 Canadian Antimicrobial Resistance Surveillance System, presenting data and findings on AMR and AMU from 10 surveillance programs based at PHAC, covering both humans and animals; and updating the CARSS AMR and AMU dashboards;
- Collaborating with the Privy Council Office of Canada to develop a national survey to assess the knowledge, beliefs and behaviours of AMU and AMR. This work helped to break down socio-demographic data, improve Northern data, and led to new findings on self-medication with antibiotics;
- Developing population-level data on AMR in smaller and larger municipalities in Canada through the National Wastewater Survey to contribute to a One Health Surveillance System;
- These efforts are complemented by a PHAC-HC partnership to generate data on antibiotics detected in wastewater, which will help monitor an important driver of AMR in natural and built environments.
- Additionally, a research pilot of four central Canadian communities revealed a rural and urban divide in the profile of AMR factors that were otherwise found to be stable over the study period.
- To further strengthen AMR surveillance in Canada, the Canadian Nosocomial Infection Surveillance Program (CNISP) commenced a national surveillance protocol for long-term care homes (LTCH) to address a critical information gap in this high-risk sector that exists across jurisdictions both nationally and globally. This pilot aims to generate actionable data to guide infection prevention and control efforts, support antimicrobial stewardship, and reduce infections and antimicrobial resistance among vulnerable LTCH residents.
- Expanding the National Microbiology Laboratory's Antimicrobial Resistance Network (AMRNet) to include surveillance coverage from eight provinces and territories (PTs) for human data, and from six PTs for the veterinary pilot, AMRNet-Vet. This expansion aims to capture more information on the antimicrobial susceptibility of clinical isolates from both people and animals;
- Expanding the ability to conduct AMR prediction testing for Neisseria gonorrhoeae directly from clinical specimens, which can now be done in 11 PTs. This complements the Gonococcal Antimicrobial Surveillance Program – Canada (GASP-Canada) genomics program which provides genomic analysis for all Neisseria gonorrhoeae cultures;
- Updating the AMU in the Canadian Community Sector dashboard to include public data on AMU from retail pharmacies from January 2018 to June 2024, including a 72-month trend and an Access, Watch, Reserve (AWaRE) category breakdown;
- Conducting the Canadian National Antimicrobial Prescribing Survey
(NAPS) from 2019-2024 to provide insight into prescribing practices for antimicrobials in hospital settings.- From 2018 to 2023, 66 Canadian hospitals conducted NAPS audits on 19,916 prescriptions for 11,510 patients. The survey results found that nearly one-fifth (18.4%) of prescriptions were deemed inappropriate or suboptimal in participating Canadian healthcare facilities.
- Indigenous Services Canada (ISC) co-developed AMR/AMU surveillance and reporting protocols for emerging AMR in First Nations and Inuit communities in collaboration with the National Microbiology Laboratory of PHAC, and implemented community-led AMR surveillance efforts (including patient testing) in Indigenous communities.
- Additionally, ISC is currently in the planning stage of re-engaging various partners to expand surveillance protocol development and AMR patient testing. This work will build on the N. gonorrhoeae AMR protocol development and urine specimen testing conducted with First Nations clients between 2017 and 2019.
- In June 2024, AAFC, PHAC, HC and CFIA, delivered a presentation on AMU baselines and goal setting to the animal health and agriculture industries. The presentation provided global context on AMR and showcased approaches to establishing baselines and goals used by other countries.
- AAFC provided support to Animal Health Canada and the Canadian Animal Health Surveillance System (CAHSS) to continue collaborative work with FPT governments, industry and animal health partners to maintain and expand an animal health surveillance network of networks, which includes an AMU/AMR network focused on promoting collaboration and sharing of information to safeguard the effectiveness of antimicrobials.
- The Government of PEI provided funding to the AVC to pilot a surveillance program to monitor Veterinary Teaching Hospital patients (cats, dogs and horses) for fecal carriage of carbapenem-resistant Enterobacterales (CRE). The aim of this study was to pilot a screening program to identify patients carrying CRE and to estimate the prevalence of this organism within these patients. An information sharing agreement has been developed with the Canadian Animal Health Surveillance System to include the AVC Diagnostic Services laboratory data in CAHSS's repository.
- AVC also launched a survey on CRE mitigation strategies for the academic veterinary community, with plans to publish both the survey and the above-mentioned pilot surveillance program.
- The BC Centre for Disease Control (BCCDC) continued to track community AMU and AMR rates via two publicly available surveillance dashboards through data partnerships with the BC Ministry of Health and LifeLabs. These dashboards present decades of AMU in community settings, providing transparency to the public and important contextual information for practitioners in the province.
- The Provincial Infection Control Network of BC continued to advance surveillance efforts by:
- Monitoring and reporting on Clostridioides difficile infections, MRSA, CPO and Candida auris (C. auris) through quarterly and annual surveillance reports. These public-facing reports include both provincial-level trends and health authority-stratified data;
- Expanding internal CPO surveillance in partnership with BCCDC's Public Health Laboratory, and providing a quarterly internal report for IPC partners in BC that integrates epidemiologic information (e.g., travel history, known exposure) with whole genome sequencing data.
- Newfoundland Health Services (NLHS) has focused on AMU and AMR monitoring through:
- Inpatient (Pyxis) AMU monitoring, which is conducted in most hospitals in the province;
- A 3-year contract 2021-2023 for AMR and AMU data sharing with the National Microbiology Laboratory and a 5-year contract for 2024-2029; and
- Monitoring and reporting rates of antimicrobial resistant hospital acquired infections to the Canadian Nosocomial Infection Surveillance Program (CNISP). The participating hospitals account for 87% of all acute care beds.
Pillar 3: Stewardship
Antimicrobial Stewardship (AMS) utilizes a systems approach, recognizing the role of patients, prescribers, producers and the public in the promotion, monitoring, improvement and evaluation of appropriate AMU. Efforts have been made to ensure the prudent and responsible use of antimicrobials across both human and animal health to slow the emergence and spread of AMR. These crucial educational efforts allow for increased confidence in prescribing and supporting the preservation of the effectiveness of antimicrobials.
Priority Actions:
- 3.1 Develop, implement and promote guidelines/standards for appropriate AMU in humans and animals through policy and regulatory initiatives, monitoring and educational interventions/accreditation requirements for health professionals and prescribers.
- 3.2 Foster understanding of the risks of AMR and the importance of appropriate use of antimicrobials in humans and animals amongst the public, patients and producers through awareness/education campaigns, feedback mechanisms and policy and regulatory initiatives.
In 2024, HC published new educational materials for patients on the appropriate use of antimicrobials. The materials included a poster and a fact sheet that aimed to improve the understanding of the risks of AMR and the crucial role patients play in supporting AMS and preserving the effectiveness of antimicrobials. HC and PHAC collaborated on opportunities for dissemination to Canadian patients and AMS stakeholders, such as health care professionals and patient partners groups.
PHAC continued to support a national antimicrobial prescribing guideline project with the Association of Medical Microbiology and Infectious Disease (AMMI) Canada and Spectrum (Firstline). The first set of guidelines were finalized in November 2025, and will be distributed to health care providers at the point-of-care through the Firstline digital platform.
Additionally, with the financial support of CFIA and PHAC, the Canadian Academy of Health Sciences (CAHS) published an assessment of AMR and AMU in food-producing animals in Canada (PDF). The report highlighted strategic interventions that can be implemented to further strengthen the prudent use of antimicrobials in food-producing animals in Canada to help mitigate the risk of AMR to human health, and emphasized the importance of CIPARS surveillance of AMR and AMU in food-producing animals in measuring and demonstrating the success of policy changes.
Spotlight: Québec's Ministry of Agriculture, Fisheries and Food (MAPAQ)
MAPAQ proposed, and passed, a bill to amend the Animal Health Protection Act (chapter P-42). The amendments came into force on October 8, 2024 and enabled the Québec government to respond more effectively to health emergencies, adopt improved response methods to address new animal health issues, and better combat antibiotic resistance, thereby ensuring better protection of animal and public health, and preserving the economic health of the bio-food sector.
In addition, MAPAQ supported various stewardship projects to combat AMR such as:
- Funding for the Faculty of Veterinary Medicine at the University of Montreal to ensure that other species could be added to the Firstline application for prescribing veterinarians in Québec. Guidelines for dogs, cats, and horses are currently available in the application, while recommendations for livestock species are being developed;
- Providing a total of $452.6K in financial assistance to 11 veterinary clinics to strengthen their ability to attract and retain workers and ensure continuous service delivery to agricultural producers. This measure is aimed at vulnerable veterinary establishments that provide eligible services to agricultural producers in their region;
- Granting $3.8M in financial assistance for veterinary support for animal production farms, as part of its Québec Integrated Animal Health Program. This support includes raising awareness among producers about the judicious use of antibiotics, conducting health assessments, and implementing biosecurity plans for livestock farming; and
- Establishing a tripartite committee comprising the Ministry of Health and Social Services (MSSS), the Ministry of the Environment, Climate Change, Wildlife and Parks (MELCCFP) and MAPAQ to provide a forum for information sharing and exchange between the three sectors in order to explore opportunities for collaboration in the field of antibiotic stewardship.
Spotlight: PACE in BC
The Provincial Antimicrobial Clinical Expert committee (PACE) in BC was established in 2014 and oversees a wide array of antimicrobial stewardship activities across the province including drug reviews and the development of best practice guidance. It is comprised of medical (infectious disease physicians) and pharmaceutical representatives from each of the BC health authorities and programs (e.g., BC Centre for Disease Control). Recent accomplishments include the development of:
- A penicillin allergy de-labeling Toolkit (PDF);
- CPO treatment guidance (PDF); and
- A provincial antimicrobial prioritization scheme, based on the AWaRe methodology. Footnote 3
PACE's public-facing guidance can be found at: Antimicrobial Stewardship for Healthcare Professionals.
Additional Stewardship activities underway include:
- Various federal, provincial and territorial organizations celebrated World Antimicrobial Awareness Week (WAAW) from November 18–24, 2024 by raising public awareness about AMR and AMU in humans, animals, food, plants (sales of antimicrobials), and water. Some key highlights include:
- PHAC published the revised AMR website to ensure all AMR resources are accessible and user-friendly; developed social media products promoting the Year 1 Progress Report, a patient story, dental AMU, the updated Enhanced Surveillance of Antimicrobial-Resistant Gonorrhea system, and the Gonococcal Antimicrobial Surveillance Program Canada dashboards onInfoBase; and led a webinar series showcasing AMS supported projects including in long-term care settings and the development of the national antimicrobial prescribing guidelines.
- The National Collaborating Centre for Infectious Diseases (NCCID), in partnership with a network of over 50 collaborators from more than 20 organizations across Canada, launched a renewed campaign and website named AMR Aware Canada. This supports increased awareness of human health AMR amongst clinicians and the general public, and provides curated tools to help mitigate AMR and campaign resources for World AMR Awareness Week.
- CFIA launched a digital AMR veterinary toolkit on World AMR Awareness Week to support conversations with pet and livestock owners on how to help minimize the risk of AMR in animals. It was promoted through social media including a LinkedIn advertisement targeted at veterinarians.
- Nunavut distributed AMR-related "Practice Pearls" (resource handouts) to nurses, nurse practitioners, pharmacists and physicians in the territory.
- PHAC supported stewardship efforts by:
- Working with PTs and AMR stakeholders to complete the "Using Behavioural Science to Decrease Overprescribing in Long-Term Care" project which was implemented in eight long-term care homes within five provinces. This project promotes AMS best practice when testing for urinary tract infections, and data analysis is currently underway;
- Integrating AMR and AMS content in the Centre for Biosafety Curriculum resources for K-12 teachers which raises awareness of the importance of microbes and AMR related pathogens that can cause infection. This project integrates key concepts of AMS in engaging and practical learning experiences for students;
- Collaborating with the University Health Network to develop recommendations from Canadian AMS and infectious disease experts to support a standardized approach to evaluating and reporting judicious use of antimicrobials in Canadian hospitals.
- HC published a summary of its Compliance Monitoring Project, which looked at how Commercial Feed Mills (CFM) that use prescription veterinary drugs to make medicated feed are complying with the prescription rules for medically important antimicrobials (i.e., those important in human medicine, as per List A).
- HC published a notice encouraging label updates for veterinary antimicrobials important in human medicine with unspecified or prolonged durations of use, to better align with prudent use.
- BCCDC launched multiple media and education campaigns including:
- Conversations with practitioners and members of the public on the topic of antibiotic use. These conversations were presented in various media across the province to engage residents and to help them better understand the role they play in combatting AMR; and
- Continuing the implementation of "Do Bugs Need Drugs?", a public education program which has reached more than 150K people across the province since 2005 (5.6K in 2024/25). The curricula focuses on a wide range of topics including handwashing, illness prevention and antibiotics.
- The Saskatchewan Health Authority Antimicrobial Stewardship Program continued to promote awareness and interventions throughout the province to improve AMU in acute care, long-term care, and primary healthcare. This included:
- A new initiative promoting the use of Methicillin-resistant Staphylococcus aureus (MRSA) nasal swabs in Intensive Care Units to reduce unnecessary broad-spectrum antibiotic use in patients with pneumonia;
- Collaborating with IPC staff to build off the PHAC-led study in long-term care to focus on improving appropriate urine culture use and reducing antibiotic treatment for asymptomatic bacteriuria; and
- Collaborating with oral health practitioners in the province, to provide educational information and tools to promote AMS in dentistry.
- PHO's AMS program continued to advance knowledge on best practices in stewardship interventions by:
- Conducting a process evaluation of PHO's previously published Audit and Feedback (A&F) trial to better understand factors influencing family physicians' use of A&F reports.
- Performing an economic evaluation which found that for every $1 spent on the A&F program in Ontario for family physicians, there was a return on investment of $8.82.
- Conducting a systematic review of A&F interventions for antibiotic prescribing in primary care which found A&F is an effective method to reduce and improve antibiotic prescribing in primary care.
- Developing facility-specific antibiograms as part of PHO's Efficient Facility Feedback of Existing Culture Testing is Able to Mitigate Resistance (EFFECT-AMR) project by using OLIS data and leveraging technical expertise and capacity developed through the COMBAT-AMR initiative. The design of the antibiogram for over 600 long-term care homes was also informed by feedback from more than 350 LTC practitioners as well as a study of variability in hospital antibiograms. Distribution and evaluation of the antibiogram reports are planned for the upcoming year; and
- Publishing studies on the impact of hospital antimicrobial stewardship funding on broad-spectrum antibiotic use and participation in provincial AMU surveillance.
- Nunavut's Territorial Antimicrobial Stewardship Program Committee has developed Urinary Tract Infection (UTI) Treatment Guidelines. These guidelines are geared towards healthcare providers (nurses, nurse practitioners, pharmacists and physicians) working in the territory.
- In addition, an evergreen document on the Antimicrobial Stewardship Program has been launched on the Department of Health website (PDF).
- In May 2025, the Government of Alberta announced an investment of $9M over three years to the University of Calgary's Faculty of Veterinary Medicine to support ongoing operations of the Diagnostic Services Unit. This investment will support the growing needs of producers and veterinarians in the province by providing timely, high quality diagnostic services to implement appropriate treatment plans and avoid unnecessary use of antimicrobials.
- The Government of PEI launched quarterly newsletters for veterinarians and livestock producers summarizing provincial livestock necropsy findings, including pathogens cultured and if there was any AMR noted. Each newsletter contains educational articles to improve knowledge and understanding of disease prevention and appropriate treatments.
Pillar 4: Infection, Prevention and Control
Infection, Prevention and Control (IPC) strategies for human and animal health such as washing hands, staying home when sick, vaccination, veterinary oversight and biosecurity measures are a few effective strategies for controlling the spread of infection. In healthcare settings, IPC measures can prevent and mitigate the risk of healthcare-associated infections. These are crucial activities that help to prevent the spread of infections, decreasing the need for antimicrobials, thereby limiting the threat of AMR.
Priority Actions
- 4.1 Increase effective implementation of infection prevention measures, particularly for populations disproportionately impacted by AMR such as remote, northern and isolated communities, First Nations, Inuit and Métis populations, long-term care residents, and hospitalized patients by developing, updating and promoting uptake of guidelines/best practices for human health.
- 4.2 Support the increased implementation of enhanced IPC, biosecurity, and food safety protocols across the agriculture and agri-food sectors, prioritizing sound animal husbandry, access to veterinary care, and access to additional health and nutritional aids to promote animal health.
Efforts have been made to address information gaps, update and promote infection prevention guidelines, and scale up best practices to ensure the protection of disproportionately impacted populations along with the community at large. Notably, with support from the National Advisory Committee on Infection Prevention and Control (NAC-IPC), PHAC published IPC guidelines for Candida auris – a multidrug-resistant fungal pathogen that can cause healthcare associated invasive infections and outbreaks, posing a serious threat to Canadians and global health. In addition, PHAC has developed an infographic (PDF) summarizing IPC best practices to help control the spread of C. auris in healthcare settings.
Ontario's Ministry of Health continued to support and strengthen the Infection Prevention and Control Hub program (PDF) ("Hub program"). The Hub program is a recognized network of IPC expertise for congregate living settings (CLS) that provides tailored supports that protect vulnerable populations within CLS. By increasing IPC capacity in CLS, the Hub program aims to decrease rates of infection, decrease frequency, duration and severity of outbreaks, as well as decrease the transmission of antibiotic-resistant organisms in these settings.
The Hub program also supports the broader health system by ensuring there is capacity for these CLS to respond to new and emerging infectious disease threats. IPC Hubs are a key driver in strengthening sustainable IPC capacity across Ontario to improve resident and population health outcomes and continue to build a skilled, adaptable, and resilient health workforce, including certified infection control practitioners.
There are currently 37 IPC Hubs that are hosted both within hospitals and public health units in every region across the province, including rural and remote regions of the province. In Spring 2025, the Hubs were able to successfully pivot their resources and expertise to support the measles outbreak:
- IPC Hubs provided critical IPC support and resources to settings experiencing the highest needs for measles-related support including in childcare centers, primary care, hospital settings, allied health providers and international agriculture workers accommodations.
- Their support included offering education and training opportunities, on-site assessments to provide measles mitigation recommendations; and working with system partners to prepare for and manage outbreaks in international agriculture worker living accommodations.
Additional infection prevention and control activities:
- PHAC implemented a targeted communication plan aimed at key audiences to support the effective dissemination of IPC materials, including guidelines, interim recommendations, infographics, and webinars. As part of this effort, two webinars were delivered on Candida auris.
- ISC continued to advance:
- Revising IPC manuals for Indigenous health facilities as required;
- Developing a national IPC program to support ISC regions and Indigenous communities; and
- Developing Integrated Oral Disease Prevention and Management training modules.
- CFIA supported IPC efforts by:
- Supporting Animal Health Canada, in collaboration with the National Poultry Groups to develop biosecurity resources for smallholders of poultry and swine, as well as backyard flocks and herds. These resources enhance biosecurity measures to reduce the risk of disease introduction, therefore decreasing the need for AMU. These resources can be found on the AHC webpage;
- Updating and supporting the development of the National Biosecurity Standards and Biosecurity Principles for both plants/crops and animals to support industry implementation of infection prevention strategies; and
- Publishing modernized livestock feed regulatory changes to the Feeds Regulations, 2024 in Part II of the Canada Gazette on July 3, 2024 reduces regulatory burden for and facilitates access to feed and nutritional aids for animal health including veterinary health products.
- Continuing to facilitate access to animal health products and nutritional aids as alternatives to keeping animals healthy and reducing reliance on routine use of antimicrobial use through:
- Approved 15 new veterinary biologics and consulted on or approved innovative feed products – including gut modifiers, mycotoxin detoxifiers, and fee mitigants – to help keep animals healthy and reduce antimicrobial use.
- HC facilitated access to low-risk Veterinary Health Products (VHPs), including a 2024 program expansion for VHPs to be mixed in livestock feed:
- HC and CFIA developed policies and guidance to allow the addition of VHPs in livestock feeds.
- In November 2024 the CFIA added 16 new VHPs that are now allowed to be mixed with livestock feed to the Compendium of Non-Feed Products.
- AAFC maintained support for industry-led research under the AgriScience Program (Clusters Component), which is aimed at developing innovative approaches to supporting animal health while reducing AMU across major livestock and poultry sectors.
- The Provincial Infection Control Network of BC (PICNet) continued to advance IPC efforts by launching a multi-year (2024–2027) CPO Strategic Action Plan which aims to enhance provincial surveillance protocols for CPO and Candida auris, and update IPC guidelines for the prevention and management of CPO in health settings in BC.
- PHO continued to support front-line practitioners across health care sectors to make improvements to their IPC programs, in response to needs identified through direct interactions with stakeholders in the field, by providing new or updated knowledge products and support for implementation. This included:
- The Antibiotic Resistant Organism Risk Factor-Based Screening Guidance for All Health Care Settings (PDF);
- A Candida auris case management algorithm (PDF); and
- Several resources related to avian influenza including a fact sheet Prevention and Management of Avian Influenza in Health Care Settings (PDF) to assist with identifying and managing suspected or confirmed cases of avian influenza; an updated Highly Pathogenic Avian Influenza (HPAI): Infection Prevention and Control Guidance for Veterinary Clinics (PDF) that provides care for animals at risk for HPAI infection; the Infection Prevention and Control Organizational Risk Assessment for Clinical Office Practice (PDF) to support health care organizations to identify and address potential internal and external infection risks; and the Infection Prevention and Control for Home and Community Care: Quick Reference (PDF) to support health care workers delivering in-home care.
Pillar 5: Leadership
Canada continues to demonstrate strong leadership both domestically and internationally in fostering interjurisdictional coordination and global collaboration to combat AMR. Through sustained efforts, Canada is advancing key initiatives that support the effective implementation of the PCAP, reinforcing its commitment to a cohesive One Health response to AMR.
Priority Actions
- 5.1 Build on existing One Health AMR governance structures to create a "network of networks" with inclusive representation to support action plan implementation and share progress and lessons learned within and across the five pillars of action, prioritizing strengthened FPT, First Nations, Inuit and Métis collaboration to co-develop AMR actions.
- 5.2 Increase Canada's contributions to global efforts to advance key bilateral and multilateral commitments by prioritizing:
- Generating improved data/evidence on AMR/AMU and strengthening surveillance systems and data standards
- Maintaining efforts to support low- and middle-income countries by advancing equitable access, stewardship and IPC initiatives
Domestically, key governance groups such as the Federal, Provincial and Territorial AMR Steering Committee (FPT SC), PHAC's Expert Advisory Group on AMR and the federal Interdepartmental Director Generals' Group on AMR have enabled coordinated engagement and collaboration across all One Health Sectors. Regular meetings of PCAP governance tables have enabled partners to advance several key priorities and have provided an opportunity to engage with experts across a range of One Health sectors and senior officials from 13 federal departments, which ensures a comprehensive One Health approach to PCAP implementation. In addition, since the renewal of the FPT SC's mandate in July 2024 the committee's membership grew to include representation from both human and animal/agricultural sectors across all PTs.
Last year marked a pivotal year for AMR in the international landscape. Two capstone international events took place: the UNGA HLM on AMR in September 2024; and the Fourth Global High-Level Ministerial Conference on AMR in November 2024. For the UNGA HLM Political Declaration on AMR (PDF), Canada ensured that its priorities and perspectives were incorporated allowing for clear alignment with PCAP and successful participation of Canadian officials at the HLM. Last year's G7 Health Ministers Meeting in Italy and G20 Health Ministers Meeting in Brazil both included dialogue related to AMR in which Canada participated. In March 2025, Canada engaged with G7 partners for a technical working group conversation related to access and innovation in the AMR space. These international efforts have been instrumental in revitalizing global momentum and advancing efforts to address AMR, and it is critical that this attention is sustained moving forward.
Spotlight: Alberta's Action Plan on AMR
In November 2024, the Government of Alberta publicly released Alberta's One Health Antimicrobial Resistance Framework for Action (the Framework). This Framework lays the groundwork for Alberta's response to AMR and guides their efforts to effectively address this growing threat. The Framework was developed through extensive engagement with AMR partners, including human and animal health professionals, professional associations and licensing bodies, healthcare delivery partners, agriculture and agri-food industries, and academics, to provide crucial direction for Alberta's approach to AMR. It acknowledges a One Health approach for coordinating efforts among the ministries of Health, Agriculture and Irrigation, and Environment and Protected Areas, and will be implemented through a phased approach with an investment of $5M over the next two years for stewardship initiatives from the Government of Alberta. As successes and best practices are learned through each phase of implementation, the Government of Alberta will continue to engage with partners and stakeholders to identify priority areas for action for implementation.
Additional leadership activities underway include:
- Canada continued to support global AMR and AMU surveillance efforts by contributing data to three international surveillance systems. This included the WHO's Global Antimicrobial Resistance and Use Surveillance System (GLASS), the World Organisation for Animal Health's (WOAH) global database on ANImal antiMicrobial USE (ANIMUSE), and the Food and Agriculture Organization (FAO)'s InFARM database.
- For the first time, Canadian data submitted to ANIMUSE was made publicly available through ANIMUSE.
- These, and other global monitoring efforts, are important ways in which Canada can support international efforts to monitor AMR and AMU and share data that can be of use to other countries.
- Canadian experts have provided technical expertise internationally:
- Canadian experts engaged with the Quadripartite Joint Secretariat regarding technical aspects of integrated surveillance (Quadripartite Technical Group on Integrated Surveillance) and on webinars for implementing the One Health Priority Research Agenda for Antimicrobial Resistance; supporting international capacity-building.
- A CFIA expert participates as a member of WOAH's Terrestrial Animal Health Standards Commission (May 2024 -May 2027) and an expert from PHAC participates in WOAH's AMR Working Group (starting in February 2024). These groups support WOAH members through the development and revision of standards and guidance on animal health AMU and AMR and the sustainability of WOAH's Global Strategy on Antimicrobial Resistance and Prudent Use.
- An expert from PHAC served as chair for the WOAH AMR ad hoc Group tasked with producing technical guidance to support interested countries to develop AMU surveillance programs in aquaculture. This guidance is anticipated to be published in December 2025.
- An expert from CFIA served as Chair of the International Plant Protection Convention's (IPPC) Commission on Phytosanitary Measures (CPM) for a two-year term (2023–2025). During the 19th Session of the CPM in March 2025 a focus group was tasked with evaluating the potential value and scope of best practices for minimizing the emergency of AMR associated with AMU in crop protection.
- An expert from PHAC is a member of WOAH's Electronic Technical Group on AMU and has provided assistance in capacity-building activities in Africa and Southeast Asia.
- Continuing to provide technical collaboration with member countries/regions through the Transatlantic Taskforce on Antimicrobial Resistance (TATFAR). TATFAR work packages cover areas such as surveillance, risk assessment, communication, policy and innovation. Canada currently chairs or co-chairs two work packages related to risk analysis for foodborne AMR and One Health AMR Surveillance.
- IDRC participates as a committee member with the International Research Consortium on Animal Health (STAR-IDAZ), WOAH and the International Veterinary Vaccinology Network (IVVN) to determine gaps for vaccines and alternatives to determine gaps for vaccines and alternatives to define priorities to improve aquaculture health.
- IDRC has funded various Joint Programming Initiative on Antimicrobial Resistance (JPIAMR) projects with components in LMICs. In total, 7 projects were funded ($2.5M), following two calls for proposals, one on surveillance and the other on One Health. The surveillance projects aimed at developing strategies, technologies, and methods for the detection, monitoring, profiling and/or surveillance of antimicrobial resistance and AM resistant pathogens.The One Health funded projects aimed at designing, implementing, evaluating, and comparing interventions with true impact on reducing the development and transmission of antibiotic resistance in and between the different One Health settings.
- CIHR invested $500K over 3 years to the JPIAMR project called SHIELD which aims to enhance trained immunity (TI) against fungal threats, a solution for combatting AMR.
- HC's Assistant Deputy Minister of the Health Products and Food Branch co-chaired a session on AMR at the annual summit in November 2024 of the International Coalition of Medicines Regulatory Authorities (ICMRA). The session focused on how regulators can work together to address the challenges of AMR.
- HC also continued its participation in the Quadrilateral Anti-Infective Drug Cluster, with the U.S. Food and Drug Administration, European Medicines Agency, and Japan's Pharmaceuticals and Medical Devices Agency, which meets quarterly to discuss ways to increase scientific alignment between regulatory agencies surrounding antibacterial and antifungal drug development and other regulatory topics related to AMR.
- The Council of Chief Veterinary Officers Subcommittee on AMU in Food Animals provides a regular forum for information exchange and collaboration between federal, provincial and territorial representatives regarding AMU / AMR / AMS initiatives; this is done through a roundtable discussion at each meeting, and through targeted presentations on key topics of interest.
- CFIA participated in a number of international efforts including:
- Actively providing feedback on new and revised chapters for the Terrestrial and Aquatic Animal Health Codes. This included extensive comments on the new Biosecurity chapter for the Terrestrial Animal Health Code which will support countries in preventing the introduction of disease and therefore reduce the need to use antimicrobials;
- Supporting Canada's Chief Veterinary Officer (CVO), in their participation in the G7 CVO Forum in Padua Italy, in October 2024. The meetings focused on zoning and biosecurity as tools for infection prevention and control as well as the facilitation of trade in animals and animal products.
- The BCCDC organized the third edition of the Western Canada One Health Antimicrobial Stewardship Conference, held in Vancouver in October 2024. The event brought together over 90 colleagues from across the animal, human and environmental health sectors to collaboratively address AMR.
- Inspired by Nova Scotia's work on AMR, the Saskatchewan Health Authority's Antimicrobial Stewardship Program, in collaboration with the Saskatchewan Ministry of Health, initiated the process of developing a committee to review the current state of activities in Saskatchewan that address the PCAP pillars. Work is underway to identify successes and gaps to ensure that Saskatchewan is doing its part in addressing the goals outlined in the PCAP.
- PHO continued to lead efforts for the Canadian ANtibiotic prescribing feedback initiative: Building a national framework to combat AntiMicrobial Resistance in primary care (CANBuild-AMR). The aim of this program is to increase the capacity to conduct A&F and standardize feedback interventions which will allow for more effective evaluation of AMU at a national level. This collaboration is supporting the implementation and/or redesign of antibiotic A&F interventions in eight provinces.
- PHO also led a scoping review to better understand opportunities for local public health to advance AMS implementation and mitigate AMR on collaboration with colleagues at the United States Centers for Disease Control and Prevention (CDC), the UK Health Security Agency (UKHSA) and local public health experts
- Contributed to the development of World Health Organization (WHO) research priorities for AMR in Human Health and published standards for High Quality AMS Research with international colleagues.
- Governance tables were instrumental in advancing PCAP implementation during this period:
- FPT SC was renewed and expanded to include representatives from both the animal and human health sectors from all PTs. The committee met twice;
- PHAC's Expert Advisory Group, comprised of reputed AMR experts from all One Health Sectors met 5 times;
- The federal Interdepartmental Directors General AMR Committee comprised or representatives from 13 departments met 5 times; and
- A federal Policy and Coordination Group was created to enhance One Health approaches and coordination related to PCAP implementation. This new committee met monthly.
Look Ahead
Governments and partners are committed to continuing to advance priority actions across the five PCAP pillars. PCAP is an evergreen plan, and therefore this coming year will be an opportunity to continue to build upon efforts and activities that are informed by research, analysis, lessons learned and engagement. The following highlights some key areas that will continue to be advanced.
Research and innovation:
- PHAC will deliver the economic pull incentive pilot project (implementation, data collection and analysis, evaluation).
- The National One Health AMR Research Strategy (NOHARS) will establish a set of AMR research priorities to build scientific knowledge across the five pillars of the PCAP.
Surveillance:
- PHAC will:
- Collect, analyze and report on data for priority organisms through the AMRNet's One Health surveillance. Data analysis for 2023-2024 is in progress and is expected to be released by March 2026.
- Work to maintain the collection of AMR and AMU data in sentinel primary care settings for humans. This will be used to establish baseline information on AMR diagnoses, prescribing patterns and, if possible, the impact on some vulnerable populations (e.g., elderly people living in the community); and
- CIPARS will further enhance reporting of surveillance data with new figures on the CIPARS AMR interactive data webpage with dashboards featuring data on:
- Salmonella serovars and Campylobacter species,
- bacterial recovery, and
- key and integrated findings.
- CFIA will explore options to facilitate access to annual sales data for livestock vaccines. By gaining better insights into vaccine usage, CFIA and PHAC can more effectively track and analyze patterns that impact animal health and AMU.
Stewardship:
- HC will:
- Make revisions to its Categorization of Antimicrobial Drugs Based on Importance in Human Medicine. This document assists in the regulation of veterinary antimicrobial drugs and provides the list of medically important antimicrobials. Consultation on the proposed revised version closed in August and the comments are being considered before moving forward.
- Expand the Canadian Reserve List for antimicrobial drugs, originally published in March 2024, to develop a full AWaRe classification for Canada by March 2027.
- CFIA will continue to facilitate access to alternative health management products by way of pre-market authorizations of innovative feed products and veterinary vaccines which helps keep animals healthy and reduce routine AMU.
- Newfoundland and Labrador is implementing a new health information system that will be used by all facilities run by NLHS. The anticipated launch date is April 2026 and will include antimicrobial stewardship activities such as:
- Antimicrobial orders having documented indication for use;
- Restrictions on antimicrobial ordering based on the World Health Organization AWaRE categories with mechanisms in place for permitted use;
- More timely reporting of AMR patterns through the provincial antibiogram; and
- Direct link to the Firstline app to improve accessibility to treatment guidelines.
Infection prevention and control:
- ISC will:
- Support the development of a national IPC program to assist ISC regions and Indigenous partners in ensuring the IPC manual for Indigenous health facilities remains current; and
- Support the development of Integrated Oral Disease Prevention and Management training modules.
- CFIA will:
- Support farmers and veterinarians through the development of biosecurity guidance and education modules on medicated livestock feed that can be shared with clients on AMR and antimicrobial alternatives; and
- Work with veterinary professions and the feed industry to help improve and promote awareness about the quality of prescriptions for medicated feeds.
Leadership:
- CFIA will:
- Host a G7 Chief Veterinary Officers (CVO) meeting in November 2025, under its G7 Presidency, which will include a discussion on One Health and other infection, prevention and control measures such as biosecurity and vaccination; and
- Continue to chair the QUADS alliance (Australia, Canada, New Zealand, United Kingdom and United States of America), and will host a meeting of the CVOs to discussion infection prevention and control and prudent and responsible AMU.
- Continue technical collaboration with international partners through TATFAR.
- ISC is exploring a new AMS steering committee in collaboration with National Indigenous Organizations. They intend to engage with National Indigenous Organizations in Winter 2025 to discuss the feasibility of the steering committee.
- PHAC will continue to coordinate with AMR partners on the FPT Steering Committee, the External Advisory Group and the federal interdepartmental governance tables to support PCAP implementation.
Annex A: Strengthening progress monitoring
The PCAP committed PHAC to working with partners to "develop and implement an approach for monitoring and reporting on progress." Progress reporting supports continuous improvement and helps partners inform adjustments to activities over time. PCAP progress monitoring and reporting has, to-date, focused on summarizing the key activities advancing implementation. To bolster this process moving forward, a performance management approach has been developed that includes a logic model and proposed approach to measures of progress.
Logic model
The logic model, presented in Figure 1, illustrates the "cause and effect story" of the PCAP, including the desired pathways linking PCAP implementation outputs and their intended outcomes. The outputs and outcomes depicted in the logic model closely align to the PCAP's 10 priority actions and desired outcomes.
The logic model also includes an ultimate outcome: "The health of humans, animals, plants/crops and the environment are protected through comprehensive and coordinated actions that mitigate the threat of AMR and facilitate responsible antimicrobial use." This ultimate outcome builds on the vision for a pan-Canadian approach to combatting AMR endorsed by FPT Ministers of Health and Agriculture in 2017 Footnote 4 .
Horizontal connections across the logic model outputs and outcomes help visualize how the PCAP's priority actions are designed as a suite of mutually reinforcing activities that will collectively advance desired outcomes across One Health sectors. The outcomes further underscore that actions and progress monitoring is a joint One Health effort. The logic model is not intended to determine or measure causation/attribution of specific implementation actions to outcomes.
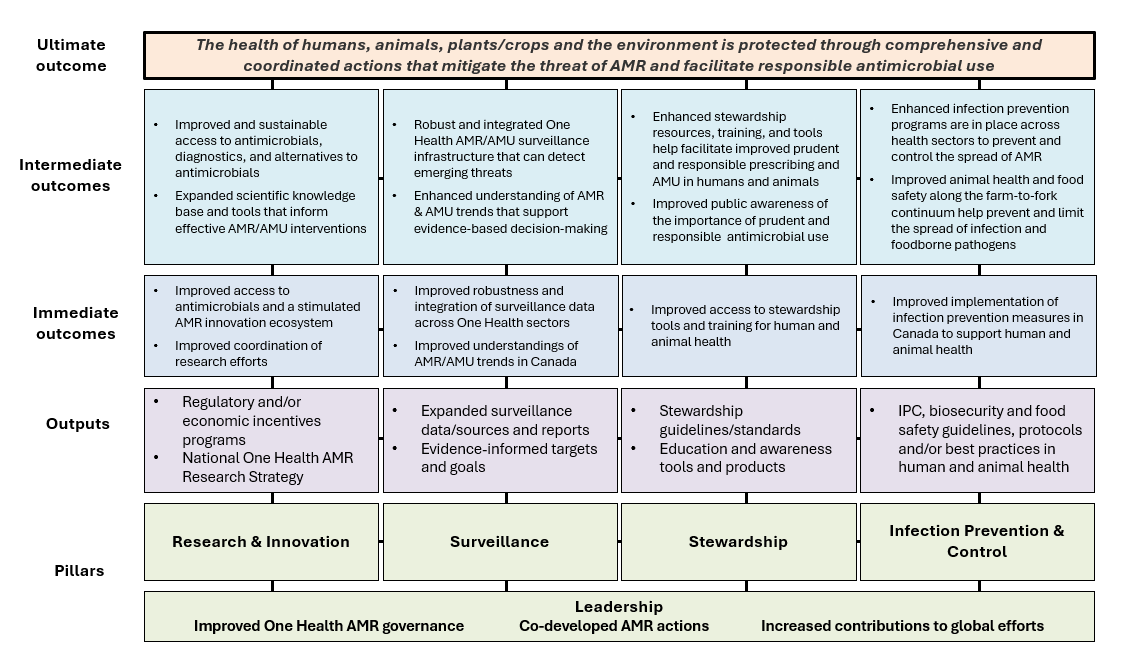
Figure 1 – Text description
- Ultimate outcome
- The health of humans, animals, plants/crops and the environment is protected through comprehensive and coordinated actions that mitigate the threat of AMR and facilitate responsible antimicrobial use
- Intermediate outcomes
- Research and innovation
- Improved and sustainable access to antimicrobials, diagnostics, and alternatives to antimicrobials
- Expanded scientific knowledge base and tools that inform effective AMR/AMU interventions
- Surveillance
- Robust and integrated One Health AMR/AMU surveillance infrastructure that can detect emerging threats
- Enhanced understanding of AMR/AMU trends that support evidence-based decision making
- Stewardship
- Enhanced stewardship resources, training, and tools help facilitate improved prudent and responsible prescribing and AMU in humans and animals
- Improved public awareness of the importance of prudent and responsible antimicrobial use
- Infection prevention and control
- Enhanced infection prevention and control programs are in place across health sectors to prevent and control the spread of AMR
- Improved animal health and food safety along the farms-to-fork continuum help prevent and limit the spread of infection and foodborne pathogens
- Research and innovation
- Immediate outcomes
- Research and innovation
- Improved access to antimicrobials and a stimulated AMR innovation ecosystem
- Improved coordination of research efforts
- Surveillance
- Improved robustness and integration of surveillance data across One Health sectors
- Improved understanding of AMR/AMU trends in Canada
- Stewardship
- Improved access to stewardship tools and training for human and animal health
- Infection prevention and control
- Improved implementation of infection prevention measures in Canada to support human and animal health
- Research and innovation
- Outputs
- Research and innovation
- Regulatory and/or economic incentives programs
- National One Health AMR Research Strategy
- Surveillance
- Expanded surveillance data/sources and reports
- Evidence-informed targets and goals
- Stewardship
- Stewardship guidelines/standards
- Education and awareness tools and products
- Infection prevention and control
- IPC, biosecurity and food safety guidelines, protocols and/or best practices in human and animal health
- Research and innovation
- Pillars
- Research and innovation
- Surveillance
- Stewardship
- Infection prevention and control
- Leadership
- Improved One Health AMR governance
- Co-developed AMR actions
- Increased contributions to global efforts
Measures of progress
The PCAP includes a commitment to establish measures of progress in the form of baselines (i.e., the reference indicator), goals and/or targets for AMR and AMU in Canada Footnote 5 . This work is in development, will be sector-specific, and will be incorporated in future progress reporting. It is expected that the logic model and measures of progress will together improve monitoring and understanding of the desired collective impact of PCAP activities.
While work is underway to develop domestic measures of progress, Canada regularly reports on AMR and AMU indicators through international surveillance reporting systems (GLASS, ANIMUSE, InFARM). These surveillance systems often align with indicators identified in international commitments such as the Sustainable Development Goals (SDGs).
A snapshot of available global indicators is presented in Table 1 as a useful window into Canada's progress on AMR and AMU relative to other countries and to related global commitments, including those established in the 2024 Political Declaration of the HLM on AMR Footnote 6.
In 2024, the FAO launched their first year of data collection for a global database on AMR in bacteria from animals and food (InFARM). Canada participated in the piloting of the FAO's INFARM system and provided data for the recent first year of data collection. Indicators for reporting changes in AMR frequencies over time have yet to be developed for InFARM.
| Source | Indicator | Year Footnote 7 | Canada profile | Related global commitments |
|---|---|---|---|---|
| GLASS-AMU Footnote 8 | Use of antibiotics by AWaRe classification (Relative use: Access, Watch, Reserve) (human health) | 2022 | Access: 73.2% Watch: 26.6% Reserve: 0.2% | The 2024 Political Declaration included a commitment2 for countries to achieve at least 70% of overall human antibiotic use in the Access category |
| GLASS-AMR Footnote 9 (SDG Indicator 3.d.2) | Proportion of bloodstream infections (BSIs) caused by Staphylococcus aureus that are resistant to methicillin (%) (human health) | 2022 | 21% | There are no established targets for the two SDG indicators. However, BSIs associated with bacterial AMR contribute to global mortality. The 2024 Political Declaration included a commitment to reduce global deaths associated with bacterial AMR by 10% by 2030 against the 2019 baseline of 4.95 million deaths |
| GLASS-AMR Footnote 10 (SDG Indicator 3.d.2) | Proportion of BSIs caused by Escherichia coli that are resistant to third-generation cephalosporins (%) (human health) | 2022 | 23.4% | |
| ANIMUSE Footnote 11 | mg of antimicrobial agents / kg estimated animal biomass (food-producing animals) | 2023 | 78.16 mg/kg biomassWOAH7 (CIPARS-VASR data reported to WOAH) 86 mg/kg biomass Footnote 12 (CIPARS-VASR) | The 2024 Political Declaration included a commitment to meaningfully reduce, by 2030, the quantity of antimicrobials used globally in the agri-food system from the current level |
| Tracking AMR Country Self-Assessment Survey (TrACSS) Footnote 13 | Country progress with development of a national action plan on AMR (Q.2.3) | 2023 | National AMR action plan approved by government and is being implemented | The 2024 Political Declaration included a commitment to sustainable financing and budgeted activities for effective implementation of national action plans |
Path forward
Moving forward, Canada will continue to report data on global AMR and AMU indicators and summarize Canada's progress in future reporting. Domestic indicators are in development and will supplement the global indicators with baselines, targets and goals Footnote 14 adapted to the Canadian context. This approach will help governments and partners inform, adjust and evaluate interventions over time. The PCAP's guiding principles (One Health, equity, collaboration, and momentum) will continue to inform Canada's strengthened approach to progress monitoring.
The logic model and indicators will remain flexible to new contexts and evidence.
Annex B: Additional Materials
Pillar 2 – Surveillance
Veterinary Antimicrobial Sales Highlights Report
CIPARS 2024 Stakeholder Webinar Integrated deck EN_FINAL.pdf (PDF)
Veterinary Antimicrobial Sales in Canada — Canada.ca
CIPARS Annual Webinar
- Human:
CIPARS human surveillance component salmonella and campylobacter AMR – 2023 results (PDF)
- Poultry:
CIPARS AMU and AMR surveillance Farmphotos.ca Poultry 2023 results (PDF)
- Dairy cattle:
CIPARS farm surveillance component: Dairy cattle (PDF)
- Feedlot Cattle:
Canadian feedlot antimicrobial use and antimicrobial resistance surveillance program (PDF)
- Grower-Finisher Pigs:
CIPARS Farm Surveillance Component: Grower-Finisher Pigs (PDF)
Glossary
- A&F
- Audit and Feedback
- AAFC
- Agriculture and Agri-Food Canada
- AHC
- Animal Health Canada
- AMR
- Antimicrobial Resistance
- AMRNet
- Antimicrobial Resistance Network
- AMU
- Antimicrobial Use
- AMS
- Antimicrobial stewardship
- ANIMUSE
- ANImal antiMicrobial USE
- AWaRe
- Access, Watch, Reserve
- AVC
- Atlantic Veterinary College
- BC
- British Columbia
- BCCDC
- British Columbia Centre for Disease Control
- CAHS
- Canadian Academy of Health Sciences
- CAHSS
- Canadian Animal Health Surveillance System
- CARB-X
- Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator
- CARSS
- Canadian Antimicrobial Resistance Surveillance Systems
- CFIA
- Canadian Food Inspection Agency
- CIHR
- Canadian Institute of Health Research
- CIPARS
- Canadian Integrated Program for Antimicrobial Resistance Surveillance
- CLS
- Congregate Living Settings
- CNISP
- Canadian Nosocomial Infection Surveillance Program
- COMBAT-AMR
- Comprehensive Ontario Microbiology laboratory Administrative data for Antimicrobial Resistance
- CPO
- Carbapenemase-producing organisms
- CVO
- Chief Veterinary Officer
- DFO
- Fisheries and Oceans Canada
- ECCC
- Environment and Climate Change Canada
- FAO
- Food and Agriculture Organization
- FPT
- Federal, Provincial and Territorial
- GDP
- Gross Domestic Product
- GLASS
- Global AMR and Use Surveillance System
- GRDI
- Genomics Research and Development Initiative
- HC
- Health Canada
- HLM
- High Level Meeting
- IDRC
- International Development Research Centre
- InFARM
- International FAO Antimicrobial Resistance Monitoring
- IPC
- Infection Prevention and Control
- ISC
- Indigenous Services Canada
- JPIAMR
- Joint Programming Initiative on Antimicrobial Resistance
- LTCH
- Long-Term Care Homes
- MAPAQ
- Ministry of Agriculture, Fisheries and Food
- MRSA
- Methicillin-resistant Staphylococcus aureus
- NLHS
- Newfoundland Health Services
- NML
- National Microbiology Laboratory
- NOHARS
- National One Health AMR Research Strategy
- NRC
- National Research Council
- OLIS
- Ontario Laboratories Information System
- PACE
- Provincial Antimicrobial Clinical Expert committee
- PCAP
- Pan-Canadian Action Plan on AMR
- PEI
- Prince Edward Island
- PHAC
- Public Health Agency of Canada
- PHO
- Public Health Ontario
- PTs
- Provinces and Territories
- SDGs
- Sustainable Development Goals
- SOARS
- Strengthening Ontario Antimicrobial Resistance Surveillance
- SPP
- Shared Priority Project
- TATFAR
- Transatlantic Taskforce on Antimicrobial Resistance
- UPEI
- University of Prince Edward Island
- UNGA
- United Nations General Assembly
- UNGA-HLM
- United Nations General Assembly High-Level Meeting
- VASR
- Veterinary Antimicrobial Sales Reporting
- VHP
- Veterinary Health Products
- WAAW
- World Antimicrobial Awareness Week
- WOAH
- World Organisation for Animal Health
- WHO
- World Health Organization
