Laboratory exposure to human pathogens and toxins, Canada 2021

 Download this article as a PDF
Download this article as a PDFPublished by: The Public Health Agency of Canada
Issue: Volume 48-10, October 2022: Equity, Diversity and Inclusion in Public Health
Date published: October 2022
ISSN: 1481-8531
Submit a manuscript
About CCDR
Browse
Volume 48-10, October 2022: Equity, Diversity and Inclusion in Public Health
Surveillance
Surveillance of laboratory exposures to human pathogens and toxins, Canada, 2021
Emily R Thompson1, Maryem El Jaouhari1, Nadine Eltayeb1, Christine Abalos1, Megan Striha1, Rojiemiahd Edjoc2, Collins Ayoo2, Samuel Bonti-Ankomah1
Affiliations
1 Health Security Regional Operations Branch, Public Health Agency of Canada, Ottawa, ON
2 Health Promotion and Chronic Disease Prevention Branch, Public Health Agency of Canada, Ottawa, ON
Correspondence
Suggested citation
Thompson ER, El Jaouhari M, Eltayeb N, Abalos C, Striha M, Edjoc R, Ayoo C, Bonti-Ankomah E. Surveillance of laboratory exposures to human pathogens and toxins, Canada, 2021. Can Commun Dis Rep 2022;48(10):484–91. https://doi.org/10.14745/ccdr.v48i10a08
Keywords: laboratory exposures, laboratory incidents, laboratory-acquired infections, human pathogens and toxins, surveillance, Laboratory Incident Notification Canada, Centre for Biosecurity
Abstract
Background: The Laboratory Incident Notification Canada surveillance system monitors laboratory incidents that are mandated to be reported under the Human Pathogens and Toxins Act and the Human Pathogens and Toxins Regulations. This article describes laboratory exposure incidents that occurred in Canada in 2021 and individuals affected in these incidents.
Methods: We extracted all laboratory incidents occurring in licensed Canadian laboratories in 2021 from the Laboratory Incident Notification Canada system and analyzed them using the software R. We calculated the rate of exposure incidents and performed descriptive statistics by sector, root cause, activity, occurrence type and type of pathogen/toxin. Analysis of the education level, route of exposure, sector, role and laboratory experience of the affected persons was also conducted. We conducted seasonality analysis to compare the median monthly occurrence of exposure incidents between 2016 and 2020 to monthly incidents in 2021.
Results: Forty-three exposure incidents involving 72 individuals were reported to Laboratory Incident Notification Canada in 2021. There were two confirmed laboratory-acquired infections and one suspected infection. The annual incident exposure rate was 4.2 incidents per 100 active licenses. Most exposure incidents involved non-Security Sensitive Biological Agents (n=38; 86.4%) and human risk group 2 (RG2) pathogens (n=27; 61.4%), with bacteria (n=20; 45.5%) and viruses (n=16; 36.4%) as the most implicated agent types. Microbiology was the most common activity associated with these incidents (n=18; 41.9%) and most incidents were reported by the academic sector (n=20; 46.5%). Sharps-related (n=12; 22.2%) incidents were the most common, while human interaction (e.g. workload constraints/pressures/demands, human error) (n=29, 28.2%) was the most common root cause. Most affected individuals were exposed through inhalation (n=38; 52.8%) and worked as technicians or technologists (n=51; 70.8%). Seasonality analyses revealed that the number of exposure incidents reported in 2021 were highest in September and May.
Conclusion: The rate of laboratory incidents was slightly lower in 2021 than in 2020. The most common occurrence type was sharps-related while issues with human interaction was the most cited root cause.
Introduction
Working with human pathogens and toxins (HPTs) in laboratory settings poses a risk of exposure for personnel. The risk of laboratory-acquired infections (LAIs) has driven the development of biosafety oversight measures around the world, such as regulated laboratory safety practices and mandatory reporting of exposures in various settings. In the United States, Belgium and the United Kingdom, LAIs are monitored by a number of different agencies and systems with various levels of regulatory authority Footnote 1 Footnote 2 Footnote 3. In comparison, Canada has a mandated comprehensive exposure reporting system at the federal level, established to collect all laboratory incidents related to HPTs.
The Public Health Agency of Canada's (PHAC) Centre for Biosecurity administers and enforces the Human Pathogens and Toxins Act (HPTA) and the Human Pathogen and Toxins Regulations (HPTR), which were enacted to promote safety and security of the public against the risks of working with HPTs in hospital, academic, public and private laboratories. The HPTA came into effect in 2015, fulfilling its directions through the HPTR. The HPTA classifies HPTs into risk groups (RG) based on their potential to cause harm to the health of both an individual and the community Footnote 4. The RG1 pathogens are those that are not capable of or are unlikely to cause disease in humans and are associated with low risk to both individuals and the community. The RG2 pathogens can cause serious disease in an individual but is unlikely to do so, and the risk of community spread is low. The RG3-classified pathogens are likely to cause serious disease in an infected individual, but the risk of spread to the public is low. The RG4 category is reserved for HPTs that are likely to cause harmful and serious disease in an individual and are of concern for spread to others in the community. A subset of HPTs has been determined to pose increased biosecurity risk due to their potential in being used as a biological weapon, and are classified as Security Sensitive Biological Agents (SSBAs). Under the HPTA, all Canadian laboratory facilities are required to obtain a license if conducting any controlled activities with RG2, RG3, or RG4 HPTs Footnote 5.
The HPTA outlines actions to be undertaken by laboratories, such as the mandatory timely reporting of incidents involving RG2 pathogens or above in the following instances Footnote 6: exposures and suspected or confirmed laboratory-acquired infections/intoxication; inadvertent possession, production and/or release of an HPT; missing, lost, or stolen biological agent, including SSBAs not received within 24 hours of expected arrival; and changes affecting biocontainment, including changes to the physical structure of the facility, to any equipment or to the standard operating procedures.
In December 2015, the Laboratory Incident Notification Canada (LINC) Surveillance System was launched to implement and oversee requirements of reporting incidents involving RG2 HPTs and above, as outlined in the HPTR. The LINC receives reports from all licensed laboratories in Canada describing exposure and non-exposure laboratory incidents that involve HPTs. A total of 279 exposure incidents were reported between 2016 and 2020, involving a total of 596 individuals in private, public, hospital and academic laboratories Footnote 7 Footnote 8 Footnote 9 Footnote 10 Footnote 11. In light of the ongoing coronavirus disease 2019 (COVID-19) pandemic, there has been a heightened awareness and public interest in biosafety and biosecurity.
The objective of this annual report is to describe the distribution of laboratory incidents reported to LINC in 2021 and the associated factors, including activity, sector, agent, occurrence type, root causes and the number and characteristics of people exposed.
Methods
Data sources
Since the launch of the LINC surveillance system, regulated parties are required to submit their notification reports for laboratory exposure incidents, including suspected or confirmed LAIs, via the PHAC's Biosecurity Portal. Forms are standardized to capture select information for exposure incidents. Data are captured via the Microsoft Customer Relationship Management system and reviewed for accuracy and completeness by LINC team members. Data from exposure incidents that occurred between January 1, 2021, and December 31, 2021, including incidents reported in this period without a specified incident date, were used in this analysis. If more than one follow-up report was submitted, data from the most recent report were used. Extracted data were cleaned by investigating any outliers, correcting for spelling errors, and removing duplicate entries. Reporting is voluntary when the agent or incident is not under the purview of the HPTA. This includes incidents involving agents in their natural environment or classified as RG1. These reports are sometimes submitted to LINC at the discretion of the reporter, and are often incomplete as a follow-up is not required. Consequently, these reports are excluded from the analysis (ruled out).
Within the scope of the HPTA/HPTR, an exposure incident is defined as a laboratory incident that could have resulted in intoxication/infection or did result in a suspected or confirmed LAI Footnote 4 Footnote 5 Footnote 6. A non-exposure incident refers to any of the following: 1) the inadvertent possession, production or release of a pathogen or toxin; 2) a missing, lost or stolen pathogen or toxin; or 3) an SSBA not being received within 24 hours of expected arrival.
Analysis
We extracted LINC surveillance data to Microsoft Excel on February 28, 2022, and performed descriptive statistics in R 4.0.2. Cross validation of all findings were performed in SAS EG 7.1. Exposure incidents, including suspected and confirmed LAIs, were classified as either confirmed or ruled out incidents after the investigation was detailed in follow-up reports. The affected persons were ruled out if the event itself was ruled out, or if the person was otherwise determined not to be exposed. Annual numbers of exposure incidents between 2016 and 2020 were updated using the most recent data to account for changes in status of historic reports as LINC continuously receives new data; therefore, it should be noted that some discrepancies may exist between the values for this and past years' annual reports.
Among confirmed exposure incidents, we analyzed the number of incidents at the level of the report and the affected individual, of which there may be more than one per report. For each incident report, we examined the distribution of the sector, main activity, root cause(s) of the incident, occurrence type, and implicated pathogen/toxin involved, along with reporting delays. For each affected person, we examined the distribution of the highest level of education, years of laboratory experience, route of exposure, sector and regular role. In light of the ongoing COVID-19 pandemic, investigations specific to severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) were highlighted in the analysis.
We compared the number of exposure incidents over time from 2016 to 2021, and calculated the exposure incident rate per 100 active licenses, which has been described in detail in previous reports Footnote 8 Footnote 9. Finally, we performed an analysis of seasonality trends to compare the monthly occurrence of exposure incidents from between 2016 and 2021. The monthly exposure incidents for the five-year period were calculated by obtaining the median number of exposures for each month. The median was chosen over the mean as it provides a better measure of central tendency of exposure incidents by dampening noise from outlier data.
Results
Between January 1 and December 31, 2021, LINC received 106 reports on laboratory incidents: 53 exposure reports; 34 non-exposure reports; and 19 other reports involving changes affecting biocontainment (Figure 1). Among the exposure incidents, two resulted in confirmed LAIs, one involved a suspected LAI and ten were ruled out (Figure 1). One non-exposure report was ruled out. Initially, a total of 83 people were reportedly exposed in these laboratory incidents, but upon further investigation, 11 people were ruled out.
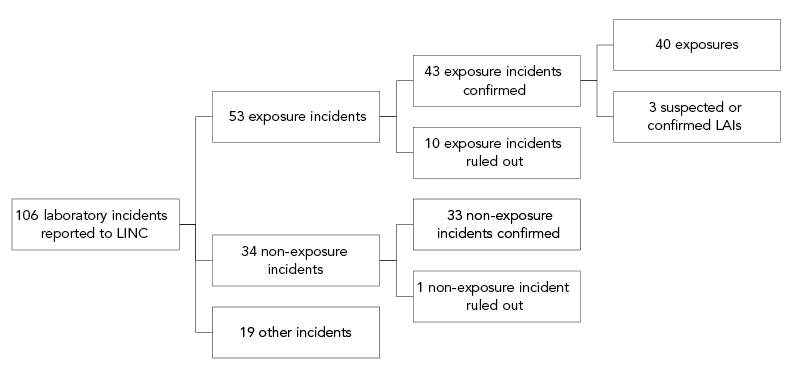
Figure 1 - Text description
| Incident type | Count |
|---|---|
| Total incidents | 106 |
| Total exposure incidents | 53 |
| Confirmed | 43 |
| Exposures | 40 |
| LAIs | 3 |
| Ruled out | 10 |
| Total non-exposure incidents | 34 |
| Confirmed | 33 |
| Ruled out | 1 |
| Total other incidents | 19 |
In 2021, there were 1,027 active licenses in Canada permitting the use of HPTs; therefore, the exposure incident rate was 4.2 incidents per 100 active licenses in 2021 (Figure 2).
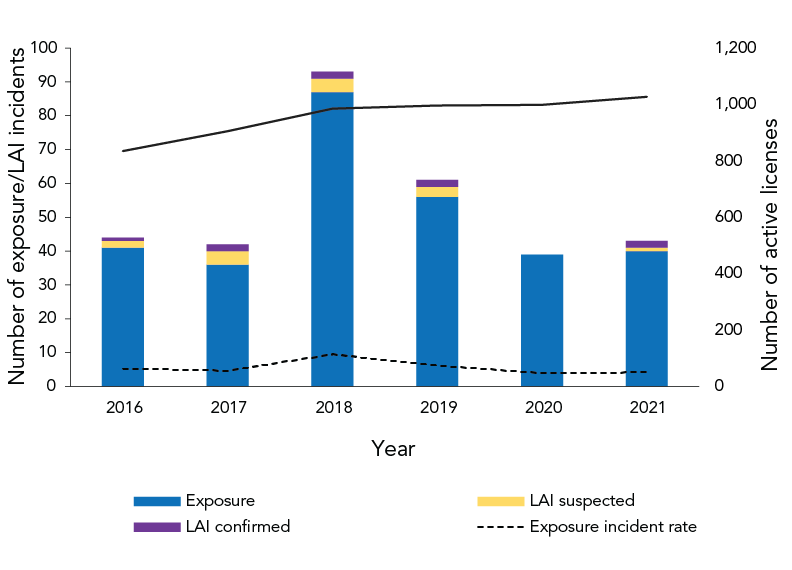
Figure 2 - Text description
| Year | Type | Number of exposures/LAI incidents |
|---|---|---|
| 2016 | Exposure | 41 |
| 2016 | LAI suspected | 2 |
| 2016 | LAI confirmed | 1 |
| 2017 | Exposure | 36 |
| 2017 | LAI suspected | 4 |
| 2017 | LAI confirmed | 2 |
| 2018 | Exposure | 87 |
| 2018 | LAI suspected | 4 |
| 2018 | LAI confirmed | 2 |
| 2019 | Exposure | 56 |
| 2019 | LAI suspected | 3 |
| 2019 | LAI confirmed | 2 |
| 2020 | Exposure | 39 |
| 2020 | LAI suspected | 0 |
| 2020 | LAI confirmed | 0 |
| 2021 | Exposure | 40 |
| 2021 | LAI suspected | 1 |
| 2021 | LAI confirmed | 2 |
| Year | Number of active licenses | Exposures | Exposure incident rate |
|---|---|---|---|
| 2016 | 835 | 44 | 5.3 |
| 2017 | 905 | 42 | 4.6 |
| 2018 | 985 | 93 | 9.4 |
| 2019 | 996 | 61 | 6.1 |
| 2020 | 999 | 39 | 3.9 |
| 2021 | 1,027 | 43 | 4.2 |
Between 2016 and 2020, the median number of exposure incidents per month varied with the lowest being 2.5 incidents per month in June and August, and the highest being 6.5 incidents per month in September (Figure 3). In comparison, the number of exposure incidents in 2021 also varied with the lowest in August (one incident per month) and the highest in February, May and October (six incidents per month).
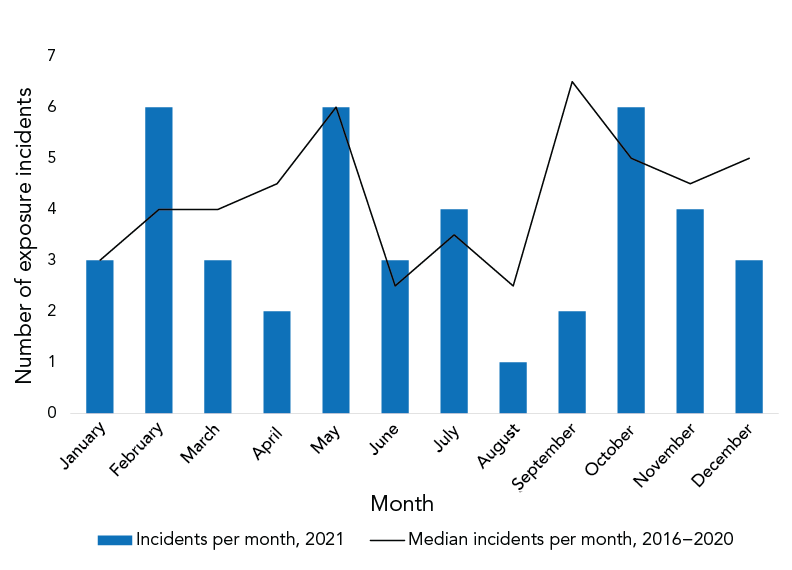
Figure 3 - Text description
| Month | Mean per month 2016–2020 | 2021 per month |
|---|---|---|
| January | 3 | 3 |
| February | 4 | 6 |
| March | 4 | 3 |
| April | 4.5 | 2 |
| May | 6 | 6 |
| June | 2.5 | 3 |
| July | 3.5 | 4 |
| August | 2.5 | 1 |
| September | 6.5 | 2 |
| October | 5 | 6 |
| November | 4.5 | 4 |
| December | 5 | 3 |
- Footnote a
-
Exposure incidents include those that involved a suspected or confirmed laboratory-acquired infection
Exposure incidents by main activity and sector
Among the exposure incidents reported, microbiology was the most common activity being performed during the incident (n=18; 41.9%), followed by in vivo animal research (n=12; 27.9%). Less frequently reported activities included animal care, cell culture, autopsy/necropsy, molecular investigation, microscopy or other (n=13; 30.2%). Definitions of the main activities are included in Appendix Table A1.
Most exposure incidents occurred in the academic sector (n=20; 11.2 per 100 active licenses), followed by the hospital sector (n=14; 9.0 per 100) (Figure 4). The veterinary/animal health sector had the highest rate of exposure incidents per 100 active licenses (21.4 per 100), followed closely by the public health sector (11.8 per 100). The environmental health sector had no reported incidents in 2021.
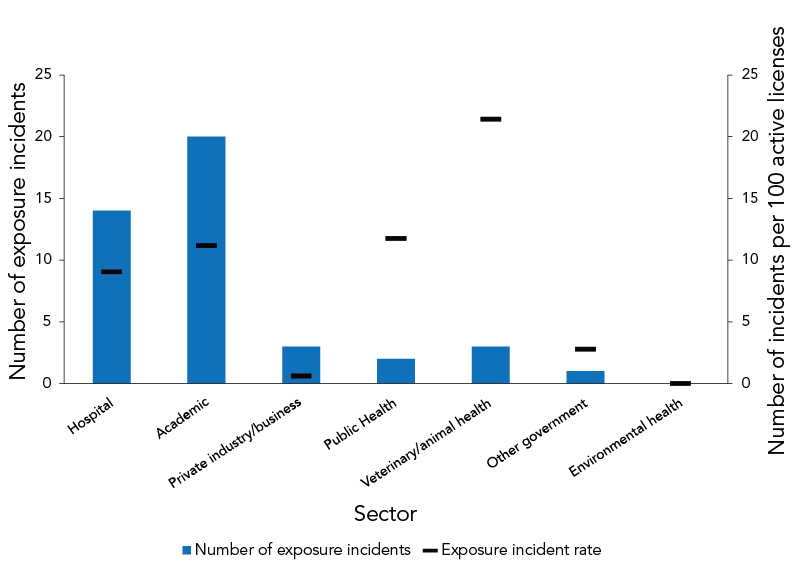
Figure 4 - Text description
| Sector | Number of exposure incidents | Percent | Number of active licenses | Exposure incident rate |
|---|---|---|---|---|
| Hospital | 14 | 32.6% | 155 | 9.0 |
| Academic | 20 | 46.5% | 179 | 11.2 |
| Private industry/business | 3 | 7.0% | 503 | 0.6 |
| Public health | 2 | 4.7% | 17 | 11.8 |
| Veterinary/animal health | 3 | 7.0% | 14 | 21.4 |
| Other government | 1 | 2.3% | 36 | 2.8 |
| Environmental health | 0 | 0.0% | 36 | 0.0 |
| Sum | 43 | 100% | 940 | 4.6 |
Implicated human pathogens and toxins
A total of 44 pathogens and toxins were implicated in the 43 exposure incidents; one incident implicated two pathogens. Of the 44 pathogens and toxins, most exposure incidents involved non-SSBA (n=38; 86.4%) and human RG2 pathogens (n=27; 61.4%) (Table 1). Bacteria (n=20; 45.5%) and viruses (n=16; 36.4%) were the most implicated agent types, while no submitted reports involved parasites. The most common RG2 agents involved in exposure incidents were Neisseria meningitides and Streptococcus agalactiae (n=3; 6.8% each). The most common RG3 agent was SARS-CoV-2 (n=6; 13.6%). Vaccinia virus was the agent involved in the suspected LAI and Salmonella enterica and Staphylococcus aureus were involved in the two confirmed LAIs.
| Biological agent type by risk group | Non-SSBA | SSBA | Unknown | Total | ||||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | |
| RG2 | 27 | 61 | 0 | 0 | 0 | 0 | 27 | 61 |
| Bacteria | 15 | 34 | 0 | 0 | 0 | 0 | 15 | 34 |
| Fungus | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Parasite | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Prion | 2 | 5 | 0 | 0 | 0 | 0 | 2 | 5 |
| Toxin | 1 | 2 | 0 | 0 | 0 | 0 | 1 | 2 |
| Virus | 9 | 20 | 0 | 0 | 0 | 0 | 9 | 20 |
| Unknown | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| RG3 | 11 | 25 | 5 | 11 | 0 | 0 | 16 | 36 |
| Bacteria | 1 | 2 | 3 | 7 | 0 | 0 | 4 | 9 |
| Fungus | 2 | 5 | 2 | 5 | 0 | 0 | 4 | 9 |
| Parasite | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Prion | 1 | 2 | 0 | 0 | 0 | 0 | 1 | 2 |
| Toxin | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Virus | 7 | 16 | 0 | 0 | 0 | 0 | 7 | 16 |
| Unknown | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| BacteriaFootnote a | 0 | 0 | 0 | 0 | 1 | 2 | 1 | 2 |
| Total | 38 | 86 | 5 | 11 | 1 | 2 | 44 | 100 |
- Table 1 - abbreviation
-
Abbreviations: RG2, risk group 2; RG3, risk group 3; SSBA, security sensitive biological agents
- Footnote a
-
This agent was identified as Brucella spp., but was not given a risk group as no species was identified
Occurrence types
Figure 5 presents the 54 types of occurrences reported in the 43 exposure incident reports. Sharps-related incidents were the most common (n=12; 27.9% of reports), followed by procedure-related incidents (n=10; 23.3% of reports) and personal protective equipment (PPE)-related incidents (n=9; 20.9% of reports). Definitions of occurrence types are provided in Table A2.
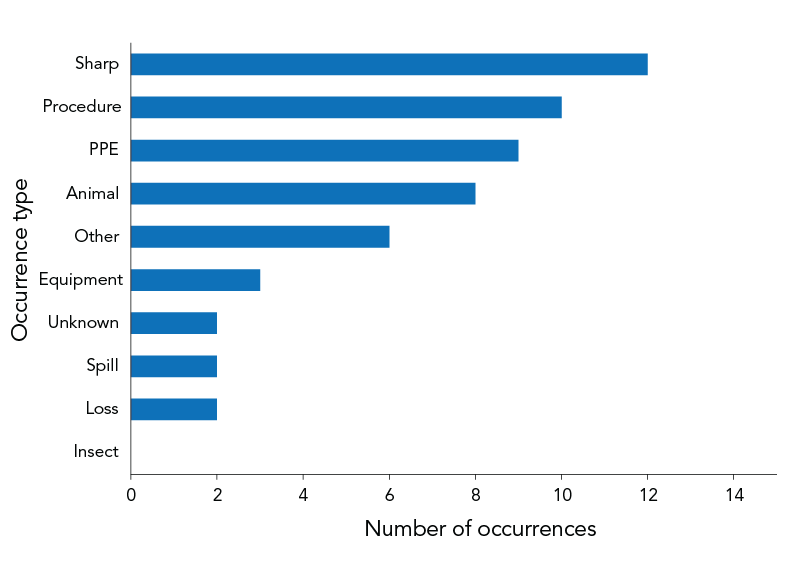
Figure 5 - Text description
| Occurrence type | Number of occurrences |
|---|---|
| Insect | 0 |
| Loss | 2 |
| Spill | 2 |
| Unknown | 2 |
| Equipment | 3 |
| Other | 6 |
| Animal | 8 |
| PPE | 9 |
| Procedure | 10 |
| Sharp | 12 |
Root causes and areas for laboratory safety improvement
In total, 103 root causes were identified in the 43 exposure reports through investigation of their follow-up reports (Table 2), giving an average of 2.4 root causes per report. The most cited root causes included issues with human interaction (n=29, 67.4%) and standard operating procedures (n=20, 46.5%), followed by issues with equipment (n=16, 37.2%) and training (n=14, 32.6%).
| Root cause | Examples of areas of concern | Citations | |
|---|---|---|---|
| n | %Footnote a | ||
| Human interaction | A violation (cutting a corner, not following correct procedure, deviating from standard operating procedure) | 29 | 67.4% |
| An error (a mistake, lapse of concentration, or slip of any kind) | |||
| Standard operating procedure | Documents were followed as written but not correct for activity/task | 20 | 46.5% |
| Procedures that should have been in place were not in place | |||
| Documents were not followed correctly | |||
| Equipment | Equipment quality control needed improvement | 16 | 37.2% |
| Equipment failed | |||
| Equipment was not appropriate for purpose | |||
| Training | Training not in place but should have been in place | 14 | 32.6% |
| Training not appropriate for task/activity | |||
| Staff were not qualified or proficient in performing task | |||
| Communication | Communication did not occur but should have | 10 | 23.3% |
| Communication was unclear, ambiguous, etc. | |||
| Management and oversight | Supervision needed improvement | 10 | 23.3% |
| Lack of auditing of standards, policies and procedures | |||
| Risk assessment needed improvement | |||
| Other | Not applicable | 4 | 9.3% |
- Footnote a
-
Denominator for percentage calculations is the total number of reports (n=43). The percentages add to more than 100% because each report may have multiple root causes. The percentages here represent the proportion of reports that indicate the given root cause, for example, 67.4% of reports selected human interaction as one of their root causes
Exposed individuals
In total, 72 individuals were exposed through the 43 confirmed exposure incidents reported to LINC. Most exposed individuals had a technical/trades diploma (n=47; 65.3%) or Bachelor's degree (n=10; 13.9%). Other highest reported education levels included Master's degree (n=4; 5.6%) and MD/PhD (n=3; 4.2%).
Among exposed individuals, most worked as a technician or technologist (n=51; 70.8%), student (n=10; 13.9%) or researcher (n=5, 6.9%). Other roles reported included supervisor/manager (n=2; 2.8%) and animal handler (n=2; 2.8%). The median number of years of experience was six years among technicians/technologists and two years among students (Figure 6).
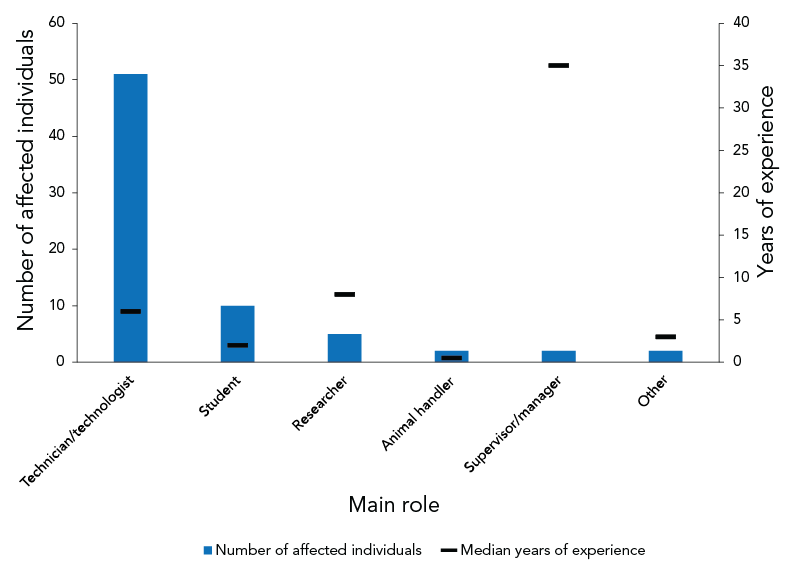
Figure 6 - Text description
| Main role | Median years of experience | Number of affected individuals |
|---|---|---|
| Technician/technologist | 6 | 51 |
| Student | 2 | 10 |
| Researcher | 8 | 5 |
| Animal handler | 0.5 | 2 |
| Supervisor/manager | 35 | 2 |
| Other | 3 | 2 |
- Footnote a
-
Other roles are those which reporters feel are not captured in the other categories, such as clinical veterinarians
Among the 72 exposed individuals, most were exposed to HPTs through inhalation (n=38; 52.8%) or through needle/sharps (n=12; 16.7%) (data not shown). Other reported routes of exposure included absorption through contact with mucous membranes or skin, and injection/inoculation.
Time between the incident and the reporting date
In 2021, 58.1% (n=25) of all exposure reports (n=43) were submitted to LINC within one week of the incident. The median number of days from incident occurrence to LINC reporting was six days in 2021, the same as in 2020 (Figure 7).
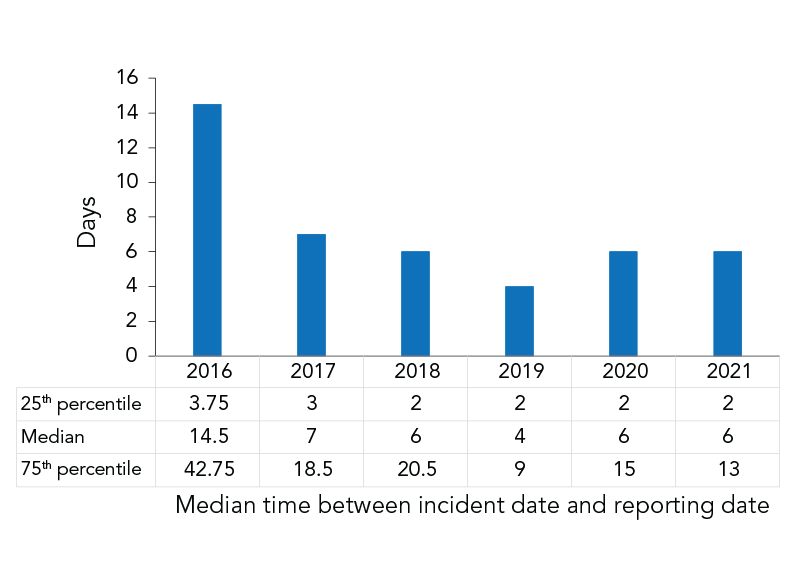
Figure 7 - Text description
| Days between incident date and reporting date | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 |
|---|---|---|---|---|---|---|
| 25th percentile | 3.75 | 3 | 2 | 2 | 2 | 2 |
| Median | 14.5 | 7 | 6 | 4 | 6 | 6 |
| 75th percentile | 42.75 | 18.5 | 20.5 | 9 | 15 | 13 |
Discussion
The LINC received 43 mandatory incident exposure reports in 2021, of which two resulted in a confirmed LAI and one led to a suspected LAI; a slight increase from 2020. As with previous years' reports, exposures occurred primarily while performing microbiological and in vivo animal research and mostly across hospital and academic sectors Footnote 7 Footnote 8 Footnote 9 Footnote 10 Footnote 11. Most incidents involved non-SSBAs and RG2 organisms, with bacteria and viruses being the most implicated agent type. Exposures mainly occurred as a result of breaches in operating procedures, sharps-related events, and failure of or inadequate PPE.
SARS-CoV-2 exposures
The year 2021 is the first full year of data for exposure incidents that occurred during the COVID-19 pandemic. The SARS-CoV-2 was the most commonly implicated agent across all pathogen groups, which may be explained, in part, by the heightened laboratory activities focused on COVID-19 treatment, vaccination and understanding of SARS-CoV-2 pathogenicity. It should be noted that as per the HPTA, reported exposure incidents involving SARS-CoV-2 did not include exposure incidents occurring during diagnostic activities.
Comparison of 2021 data with pre-pandemic levels shows some reduction in the number of laboratory exposure incidents and LAIs, suggesting that reduced laboratory personnel and laboratory work due to lockdowns and workplace closures, increased use of PPEs and renewed focus on biosafety may have had an impact on the occurrence of laboratory incidents. Data from exposure incidents once laboratory activities have fully resumed will be key in identifying further trends in laboratory incident occurrences.
Seasonal variation in exposure incidents
An analysis of the median exposure incidents per month from 2016 to 2020 demonstrates marked seasonal variation, with the highest number of incidents occurring in September and May, and the lowest number in January, June and August (Figure 3). We posit that these dips in exposure incident activity may be explained by lower staff levels during summer holidays, followed by an increase in September with lab members returning to the office as well as students returning to academic labs. Similarly, the peak in May might be representative of an influx of new lab members during the summer hiring period, as well as students taking on summer work terms.
Trends in the occurrence of exposure incidents observed in 2021 were similar to the median number of incidents per month between 2016 and 2020, with exception to September 2021. Only two incidents were reported in September, as compared to an expected 6.5 incidents. Investigation into the roles of the individuals involved, their years of experience, and the licence sector provided no further information on why fewer incidents were reported in September 2021. A lack of student presence on campuses as well as reduced staffing capacities in labs as a result of the COVID-19 pandemic may have impacted these numbers.
Human interaction as a root cause of incidents
Human interaction was cited as a root cause of exposure incidents in nearly 70% of reports, representing a 20% increase as compared to 2019. These incidents are frequently cited with other root causes, such that human interaction may equally impact other areas of concern such as training, equipment, standard operating procedures and communication. Consequently, human interaction represents an important area for improvement in laboratory biosafety. Indeed, a 2017 biosafety risk assessment of laboratory accidents in the United States identified human error as the key driver of laboratory biosafety accidents, but there are limited data quantifying the extent to which human interaction plays a role in laboratory biosafety Footnote 12. Additional details on root causes from exposure reports submitted to PHAC may provide an opportunity to further explore these trends and bridge this knowledge gap.
Strengths and limitations
The main strength of this study is the collection of laboratory incident data through a standardized and mandatory reporting system across Canada. Public Health Agency of Canada's Biosecurity Portal provides an accessible and easy-to-use method for reporting key information regarding laboratory exposure incidents. Consequently, this allows for near real-time assessment of biosafety risks and trends in HPT exposures. Efficient communication with other members of the Centre of Biosecurity, such as the inspections team, allows for rapid identification of trends in reporting and the potential for risk mitigation of incidents at licensed facilities.
As with previous years, under-reporting of laboratory exposure incidents remains a possible limitation for this analysis, and the magnitude is currently unknown. To mitigate these issues, the Centre for Biosecurity offers alternate methods of incident declaration, including fax, email and telephone calls, hosts training sessions and sends out quarterly newsletters to regulated parties alerting them of changes to the Canadian Biosafety Standards and their duty to report laboratory exposure incidents, in an effort to improve reporting compliance. In addition, inspectors check for and encourage reporting of incidents when inspecting regulated laboratories.
It is also important to note that the dearth of available information regarding laboratory incidents in other jurisdictions makes it particularly difficult to compare reporting trends observed outside of Canada. Laboratory incidents are frequently reported as case studies in other countries, so there is a lack of centrally located or easily accessible data on this subject. Canada is a unique example of a country with a centrally developed and standardised system to consistently collect information on laboratory incidents.
Finally, the LINC system does not currently support the collection of the total number of personnel or distribution of roles within laboratories. The number of active licenses is used as a proxy for workforce size, which limits the opportunity to provide more comprehensive analyses of exposure incidence rates. As such, caution should be used when interpreting this data at the laboratory level.
Conclusion
The rate of laboratory exposure incidents in 2021 was similar to that reported in 2020. Disruptions to laboratory work in light of the ongoing COVID-19 pandemic may have contributed to lower rates of laboratory exposure incidents as compared to previous years. Despite this, analysis of reports from regulated parties has shown sustained trends in the characteristics of laboratory exposure incidents in Canada, including occurrence type, root causes and sectors involved, which may help to further inform guidelines to improve biosafety and biosecurity in Canada.
Authors' statement
ERT and MEJ contributed equally to this article.
Competing interests
None.
Acknowledgements
We would like to express our gratitude to our regulated parties for their continued support and contribution regarding incident reporting across Canada. We would also like to say a special thanks to the staff of the Centre of Biosecurity for their continued input, support and expertise.
Funding
None.
References
- Footnote 1
-
Wurtz N, Papa A, Hukic M, Di Caro A, Leparc-Goffart I, Leroy E, Landini MP, Sekeyova Z, Dumler JS, Bădescu D, Busquets N, Calistri A, Parolin C, Palù G, Christova I, Maurin M, La Scola B, Raoult D. Survey of laboratory-acquired infections around the world in biosafety level 3 and 4 laboratories. Eur J Clin Microbiol Infect Dis 2016;35(8):1247–58. https://doi.org/10.1007/s10096-016-2657-1
- Footnote 2
-
Singh K. Laboratory-acquired infections. Clin Infect Dis 2009;49(1):142–7. https://doi.org/10.1086/599104
- Footnote 3
-
Willemarck N, Van Vaerenbergh B, Descamps E, Brosius B, Thi CDD, Leunda A, Baldo A. Institut Scientifique de santé publique. Laboratory-Acquired Infections in Belgium (2007–2012). An online survey. Brussels, BE: ISSP; 2015; (accessed 2021-02-25). https://www.bioveiligheid.be/sites/default/files/2015_willemarck_lai_report_belgium_2007_2012_final.pdf
- Footnote 4
-
Public Health Agency of Canada. Canadian Biosafety Standard (CBS) Second Edition. Ottawa, ON: PHAC; (accessed 22021-02-25; modified 2021). https://www.canada.ca/en/public-health/services/canadian-biosafety-standards-guidelines/second-edition.html
- Footnote 5
-
Public health Agency of Canada. Notification and Reporting Under the HPTA and HPTR Using the Reporting Module of the Biosecurity Portal. Ottawa, ON: PHAC; (accessed 2021-03-10; modified 2021). https://www.canada.ca/en/public-health/services/canadian-biosafety-standards-guidelines/guidance/notification-reporting-human-pathogens-toxins-act-regulations.html
- Footnote 6
-
Government of Canada. Human Pathogens and Toxins Act. S.C. 2009, c. 24. June 23, 2009. https://lois-laws.justice.gc.ca/eng/acts/H-5.67/FullText.html
- Footnote 7
-
Bienek A, Heisz M, Su M. Surveillance of laboratory exposures to human pathogens and toxins: Canada 2016. Can Commun Dis Rep 2017;43(11):228–35. https://doi.org/10.14745/ccdr.v43i11a04
- Footnote 8
-
Pomerleau-Normandin D, Heisz M, Tanguay F. Surveillance of laboratory exposures to human pathogens and toxins: Canada 2017. Can Commun Dis Rep 2018;44(11):297–304. https://doi.org/10.14745/ccdr.v44i11a05
- Footnote 9
-
Choucrallah D, Sarmiento L, Ettles S, Tanguay F, Heisz M, Falardeau E. Surveillance of laboratory exposures to human pathogens and toxins: Canada 2018. Can Commun Dis Rep 2019;45(9):244–51. https://doi.org/10.14745/ccdr.v45i09a04
- Footnote 10
-
Lien A, Abalos C, Atchessi N, Edjoc R, Heisz M. Surveillance of laboratory exposures to human pathogens and toxins, Canada 2019. Can Commun Dis Rep 2020;46(09):292–8. https://doi.org/10.14745/ccdr.v46i09a07
- Footnote 11
-
Atchessi N, Striha M, Edjoc R, Thompson E, El Jaouhari M, Heisz M. Surveillance of laboratory exposures to human pathogens and toxins, Canada 2020. Can Commun Dis Rep 2021;47(10):422–9. https://doi.org/10.14745/ccdr.v47i10a04
- Footnote 12
-
Ritterson R, Casagrande R. Basic Scholarship in Biosafety Is Critically Needed To Reduce Risk of Laboratory Accidents. mSphere 2017;2(2):e00010-17. https://doi.org/10.1128/mSphere.00010-17
Appendix
| Main activity | Description |
|---|---|
| Animal care | Activities such as attending to the daily care of animals and providing animals with treatment |
| Autopsy or necropsy | Post-mortem surgical examinations for purposes such as determining cause of death or to evaluate disease or injury for research or educational purposes |
| Cell culture | The process of growing cells under controlled conditions; it can also involve the removal of cells from an animal or plant |
| Education or training | Education or training of students and/or personnel on laboratory techniques and procedures |
| In vivo animal research | Experimentation with live, non-human animals |
| Maintenance | The upkeep, repair and/or routine and general cleaning of equipment and facilities |
| Microbiology | Activities involving the manipulation, isolation, or analysis of microorganisms in their viable or infectious state |
| Molecular investigations | Activities involving the manipulation of genetic material from microorganisms or other infectious material for further analysis |
| Serology | Diagnostic examination and/or scientific study of immunological reactions and properties of blood serum |
| Hematology | Scientific study of the physiology of blood |
| Occurrence type | Description |
|---|---|
| Spill | Any unintended release of an agent from its container |
| Loss of containment | Includes malfunction or misuse of containment devices or equipment and other types of failures that results in the agent being spilled outside of, or released from containment |
| Sharps-related | Needle stick, cut with a scalpel, blade or other sharps injury (i.e. broken glass) |
| Animal-related | Includes animal bites or scratches, as well as other exposure incidents resulting from animal behaviour (i.e. animal movement resulting in a needle stick) |
| Insect-related | Includes insect bites |
| PPE-related | Includes either inadequate PPE for the activity or failure of the PPE in some way |
| Equipment-related | Includes failure of equipment, incorrect equipment for the activity, or misuse of equipment |
| Procedure-related | Includes instances when written procedures were not followed, were inadequate or absent, or were incorrect for the activity |
|
|
