2021–22 Departmental Plan: Health Canada

Download the alternative format
(PDF format, 3.26 MB, 77 pages)
Organization: Health Canada
Published: 2021
The Honourable Patty Hajdu, P.C., M.P.
Minister of Health
© Her Majesty the Queen in Right of Canada,
represented by the President of the Treasury Board, 2021.
Catalogue No: H1-9/31E-PDF
ISSN: 2371-6576
Pub.: 200327
This document is available on the Treasury Board of Canada SecretariatEndnote i website.
This document is available in alternative formats upon request.
Table of contents
- From the Minister
- Plans at a glance
- Planned results and resources, and key risks
- Health Canada's Planned response to COVID-19
- Core Responsibility 1: Health Care Systems
- Core Responsibility 2: Health Protection and Promotion
- Internal Services
- Experimentation and GBA+
- Spending and human resources
- Corporate information
- Appendix: Definitions
- Endnotes
From the Minister

As the Minister of Health, I am pleased to present the 2021-22 Departmental Plan for Health Canada. The plan outlines the priorities for the year ahead and is a guide to the work that the Department will undertake on behalf of all Canadians.
As the COVID-19 pandemic continues to evolve, so does our response. Health Canada, the Public Health Agency of Canada (PHAC) and Canada's Chief Public Health Officer (CPHO) continue to lead a comprehensive, coordinated and multi-faceted whole-of-government response - one that not only involves other federal departments and agencies, but also provinces and territories, municipalities and Indigenous communities.
Thanks to innovative and agile regulatory measures, advances in science and technology, and an unprecedented level of global cooperation, Canada reached a critical milestone in its fight against the pandemic with the authorization of COVID-19 vaccines. Building on this momentum, my Department will continue to support the development of a diverse portfolio of COVID-19 vaccine agreements with world-leading candidates to optimize the deployment of safe, effective, and timely vaccines to Canadians in 2021, as well as support PHAC's rollout of approved vaccines across the country.
Health Canada will also continue to identify and acquire COVID-19 treatments and therapies; expedite regulatory reviews of drugs, medical devices and disinfectants; procure any needed medical and personal protective equipment; prioritize the review of COVID-19 tests, including new, innovative options and technologies; and pursue strategies to expand testing capacity. We will do so without compromising Canada's high standards for safety, efficacy and quality.
Our health care system is a symbol of our national identity and COVID-19 has also shown that our system needs to be more flexible and able to reach people at home. As such, my Department will further invest in mental health and virtual supports for Canadians.
Health Canada will also work with all of its partners in health - including provinces, territories and other stakeholders - to set new, national standards for long-term care to make sure seniors and those in care live in safe and dignified conditions. As part of this effort, we will establish a new Safe Long-term Care Fund that will provide up to $1 billion to help provinces and territories protect people in long-term care, and prevent and control infections in seniors and our most vulnerable.
We are also committed to solidifying the foundational elements of a national pharmacare program. This includes developing a national strategy on high-cost drugs for rare diseases, as well as establishing a transition office to oversee the creation of a Canadian Drug Agency. Drug shortages remain a global challenge and COVID-19 has increased demand for certain medications, adding to the complexity of ensuring an adequate supply where it is needed. My Department remains dedicated in its efforts to ensure Canadians have access to the medications they rely on.
A key priority for Health Canada continues to be addressing the unrelenting opioid overdose crisis. In 2021-22, we will further assist community-based organizations that respond to substance-use issues, so they can reinforce their frontline services in a COVID-19 context. In collaboration with provinces and territories and community partners, my Department will carry on the work to deliver evidence-based treatments and harm reduction and prevention programs - recognizing and promoting the concept that everyone must be treated with compassion and without stigma.
In collaboration with partners and stakeholders, Health Canada continues to implement the Cannabis Act as well as monitor, collect and analyze scientific evidence and data to better understand its impact. The Department will strengthen ongoing efforts to suppress the illegal market and to help Canadians understand the health effects of cannabis use and make informed decisions to protect their health.
Health Canada will also redouble its efforts to protect young Canadians from inducements to vape by restricting the promotion of vaping products to youth; raising awareness of the potential harms; and taking additional compliance and enforcement actions as necessary. The Department continues to advance Canada's Tobacco Strategy - a modernized approach for successful tobacco control with the goal of achieving the target of less than 5% tobacco use by 2035.
I would be remiss not to mention the dedicated Health Canada employees who continue to work tirelessly on Canada's response to COVID-19. I am grateful for their work to steer us through this pandemic while continuing to deliver on other priorities. I am also grateful to all Canadians for following public health advice and making the necessary sacrifices in their day-to-day lives. We will continue to support each other as we navigate through this crisis and emerge stronger and more resilient.
I look forward to working closely with provincial and territorial governments, Indigenous partners, key stakeholders and communities across the country to advance priorities that promote and protect the health of all Canadians.
The Honourable Patty Hajdu, P.C. M.P.
Minister of Health
Plans at a glance
Health Canada is the federal department responsible for helping Canadians maintain and improve their health. In keeping with the Department's commitment to making this country's population among the healthiest in the world, its main responsibilities are as a regulator, a catalyst for innovation, a funder, and an information provider.
Health Canada also administers the Canada Health Act, which embodies national principles to ensure a universal and equitable publicly-funded health care system. In addition to working closely with provincial and territorial governments, the Department works with partners in the Health Portfolio (PHAC, Canada Food Inspection Agency, and Canadian Institutes of Health Research), other federal departments and agencies, non-governmental organizations, other countries, Indigenous partners and the private sector.
From coast to coast to coast, Health Canada employees - scientists and researchers, inspectors, doctors and nurses, policy analysts and administrative professionals, and many others - are working to help Canadians maintain and improve their health.
Core Responsibilities
Health Canada's Departmental Results Framework outlines two core responsibilities for the Department: Health Care Systems and Health Protection and Promotion. This reporting framework provides the structure for planned activities, which are organized according to these core responsibilities and their corresponding results.

Core responsibilities - Text description
The figure illustrates Health Canada's two core responsibilities as outlined in its Departmental Results Framework.
- Core Responsibility 1: Health Care Systems
- Core Responsibility 2: Health Protection and Promotion
Under the Health Care Systems core responsibility, Health Canada provides national leadership to foster sustainable health care systems that ensure access for Canadians to appropriate and effective health care. This is mainly achieved through partnerships with provincial and territorial governments and support through targeted funding agreements to organizations and key pan-Canadian health partners that are contributing to health system improvements.
Within the Health Protection and Promotion core responsibility, Health Canada works with domestic and international partners to assess, manage and communicate the health and safety risks and benefits associated with health and consumer products, food, chemicals, pesticides, environmental factors, tobacco and vaping products, cannabis, and controlled substances. These risks are managed through rigorous regulatory frameworks and by communicating risks and benefits to Canadians so that they can make informed decisions.
Key Plans
In 2021-22, among the many others detailed in this report, Health Canada plans to achieve the following key results that contribute to the health of Canadians and respond to the government's priorities as articulated in the 2020 Speech from the Throne.
Health Canada's Planned Response to COVID-19
- In 2021-22, Health Canada will continue to work alongside PHAC and the CPHO to lead the ongoing whole-of-government response to this unprecedented public health crisis, in close collaboration with other federal departments and agencies, and provinces and territories (P/Ts) - implementing Canada's comprehensive, coordinated and multi-faceted approach to protect all Canadians from COVID-19.
- The COVID-19 Task Force will continue to lead priorities to ensure coordination and collaboration across the federal government and work with P/Ts and other stakeholders to advance priorities, such as: contributing to the development of sound and evidence-based policies and programs to protect vulnerable populations and high-risk communities, including residents of long-term care; supporting the diverse portfolio of COVID-19 world-leading vaccines to enable the deployment of safe, effective and timely vaccines to Canadians; as well as collaborating with other federal departments to identify and acquire COVID-19 treatments and therapies.
- The pandemic response cuts across each of Health Canada's Core Responsibilities and all 5 associated Departmental Results. This report groups the Department's planned 2021-22 initiatives under the following overarching focus areas: providing timely, trusted, and evidence-based information; promoting the Canada COVID-19 app and online self-assessment tool; supporting mental health; supporting virtual care; facilitating access to health products (including authorizations of clinical trials, vaccines, treatments, medical devices and disinfectants); stabilizing the supply of critical COVID-19 drugs and managing drug and medical device shortages; facilitating access to treatment services for substance use; adapting compliance and enforcement approaches; and enhancing internal services.
Key Plans: Core Responsibility 1: Health Care Systems
- To ensure that Canadians have access to appropriate and effective health services, Health Canada will focus on the following priorities, as referenced in the 2020 Speech from the Throne: access to mental health and substance use services and to home and community care; primary care; national pharmacare; palliative care; medical assistance in dying (MAID); diagnostic services; thalidomide survivors support; patient safety; combatting cancer; organ, tissues and blood donation and transplantation; as well as supporting access to health services for specific populations.
- Health Canada will work with all of its partners in health - including P/Ts and other stakeholders - to set new national standards for long-term care and take additional action to help people stay in their homes longer, so that seniors get the best support possible. The Department will also undertake to establish national standards for access to mental health services so Canadians can get access to the support they need, when they need it.
- The Department will work towards expanding access to community-based mental health and substance use services for children and youth, enhancing supports for family caregivers at home, and making home care and palliative care more available across the country.
- Our collective response to COVID-19 has led to the development of a new stream of collaborative work with P/Ts around virtual care, and this will remain a key focus for 2021-22.
- Health Canada will continue to work with its partners in health to implement a national, universal pharmacare system so that Canadians have the drug coverage they need. This includes developing a national strategy on high-cost drugs for rare diseases and establishing a transition office to support the creation of the Canada Drug Agency, and work on a national formulary.
- The Department will also make important investments to support pan-Canadian health organizations that directly contribute to health system improvements in key areas such as digital health, health information, drugs and technologies, mental health and addictions, cancer prevention and control, patient safety, and service delivery innovation.
Key Plans: Core Responsibility 2: Health Protection and Promotion
- The opioid overdose crisis continues to devastate a number of Canadian communities and families. Health Canada will work closely with all of its partners in health to respond with a comprehensive, collaborative, coordinated, and compassionate public health approach. Specific initiatives range from examining root causes in order to better prevent problematic substance use at the outset, through to increasing access to a safer supply of prescription opioids, addressing potential gaps in the regulatory controls for precursors, working with the newly-created People with Lived and Living Experience Council, and scaling up the most effective programs.
- Health Canada will focus on the ongoing implementation of the cannabis legislative and regulatory framework. This will include initiating a mandated review of the administration and operation of the Cannabis Act. The Department will continue to implement strategies aimed at promoting a diverse commercial cannabis industry and deterring the illicit market.
- Health Canada will take additional action to address the rapid rise in youth vaping, and advance implementation of Canada's Tobacco Strategy - a modernized approach for successful tobacco control with the goal of achieving the target of less than 5% tobacco use by 2035. This will include finalizing regulations to further reduce the appeal of vaping products to young Canadians and launching the first legislative review of the Tobacco and Vaping Products Act.
- The Department will continue to play a leadership role in addressing critical national drug and medical device shortages, including advancing new regulatory tools to help alleviate and prevent shortages from happening.
- Recognizing that antimicrobial resistance (AMR) is an urgent public health issue, Health Canada will take additional measures to safeguard the use of available antimicrobial drugs for human use, raise awareness, promote innovation, and monitor and support the prudent use of antimicrobials in animals.
- The Department will advance its Regulatory Innovation Agenda arising from the Health and Biosciences Sectoral Regulatory Review Roadmap and the Agri-food and Aquaculture Regulatory Review Roadmap, with a goal of making the federal regulatory framework more agile and responsive to an innovative environment, while ensuring the system remains science and safety-based.
Experimentation
Building on its Beyond2020 commitments, Health Canada continues to increase employee capacity for experimentation and innovation via its Innovation and Experimentation Policy Framework 2020. Initiatives planned for 2021-22 fall under the banners of Learn and Act, Explore and Test, and Measure and Share. The Department also announced multiple calls for proposals under its Solutions Fund seeking projects that respond to COVID-19, as well as policy, program and workplace improvements. Approved for 2020-21 are initiatives under Project Cipher, Project Hummingbird, and PRODigy.
Sex and Gender-Based Analysis Plus (SGBA+/GBA+)
Health Canada renewed its Sex and Gender Action Plan for another 3 years starting in 2020-21. The Plan provides a framework to strengthen the integration of sex, gender and other intersectional factors (such as age, race and income level) in externally and internally facing work of the Department. Under the renewed Action Plan, priorities are to: increase departmental capacity to apply SGBA+; collaborate with internal and external partners to strengthen the Department's sex, gender and diversity-related evidence base and expertise; and strengthen accountability and transparency in implementing SGBA+.
For more information on Health Canada's plans, priorities and planned results, see the "Core responsibilities: planned results and resources, and key risks" section of this report.
Planned results and resources, and key risks
Health Canada's planned response to COVID-19
The COVID-19 pandemic has dominated the lives of Canadian and global citizens for more than a year. From the beginning, Health Canada has used every tool available and done everything in its power to protect the health of Canadians. Working with all of its partners in health - including other federal government departments and agencies, provinces and territories (P/Ts), municipalities, Indigenous communities, private sector organizations and other stakeholders - the Department has sought to grow healthcare capacity, scale up testing and tracing, and ensure that Canadians stay safe and healthy and receive the support they need. It has been a true Team Canada effort.
The pandemic continues to affect our collective health, social and economic well-being, from mental health and primary care, to long-term care, to access to health care and health products. During this unprecedented time, Health Canada has mobilized partners, regulators, researchers and experts, resources and volunteers. Collaboration between all jurisdictions has been a cornerstone of Canada's approach to this global challenge, and to date, the Government of Canada has invested $322 billion to support Canadians, including direct support to P/Ts. $52 billion of this funding has supported coordinated action to strengthen critical health care systems, purchase PPE and supplies, and support critical medical research and vaccine development.
In 2021-22, Health Canada will continue to work alongside PHAC and the CPHO to lead the ongoing whole-of-government response to this unprecedented public health crisis, in close collaboration with other federal departments and agencies, and P/Ts - implementing Canada's comprehensive, coordinated and multi-faceted approach to protect all Canadians from COVID-19.
What's new?
In December 2020, Health Canada authorized the first two COVID-19 vaccines using the expedited review pathway provided under the Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19. Following thorough, independent reviews of the evidence, Health Canada determined that the Pfizer-BioNTech and Moderna vaccines met the Department's stringent safety, efficacy and quality requirements for use in Canada. The manufacturers are required to continue providing safety, efficacy and quality information to Health Canada to ensure the benefits of their vaccines continue to be demonstrated through market use.
Dedicated COVID-19 Task Force and governance structures
In the early days of the pandemic, Health Canada, in partnership with PHAC, established a dedicated COVID-19 Task Force and governance structures to ensure coordination and collaboration across the federal government and work with P/Ts and other stakeholders. In 2021-22, this Task Force will continue to lead on priorities such as: enhancing the Government-wide ability to monitor the evolution of the pandemic; contributing to the development of sound and evidence-based policies and programs to protect vulnerable populations and high-risk communities, including residents of long-term care; supporting greater testing and contact-tracing capacity across the country; collaborating to enhance data management and infrastructure; ongoing implementation, monitoring and assessing the national exposure notification app (COVID Alert); overseeing proper supply of PPE and medical supplies; supporting the diverse portfolio of COVID-19 world-leading vaccines to enable the deployment of safe, effective and timely vaccines to Canadians; as well as collaborating with other federal departments to identify and acquire COVID-19 treatments and therapies.
In 2021-22, the COVID-19 Task Force will continue to identify needs, build capabilities and inform decisions, by:
Developing policy to guide the federal response to COVID-19
- Assess and develop policy responses to the effects of the pandemic on the health and well-being of Canadians.
- Contribute to the safe and practical management of Canada's borders from a health perspective, including the appropriate application of quarantine and testing.
- Support the government-wide effort to distribute safe and effective vaccines to Canadians in a timely manner.
- Continue to collaborate with other federal government departments and external partners to identify and fill key research gaps in order to develop and/or acquire targeted science-based COVID-19 treatments and therapies for Canadians.
- Anticipate and strategically plan for the longer-term effects of the pandemic through research and analysis.
Refining PPE and other medical supplies modelling tools to better identify national needs and inform future procurement plans
- Develop models that consider PPE, medical equipment and supplies at both the national and P/T level.
- Strengthen PPE and other medical supplies data-sharing and collaboration across the federal government and work with P/Ts to share accurate and timely information.
Supporting testing capacity and developing innovative approaches to testing and screening
- Develop and implement strategies to ensure sufficient diagnostic testing and screening capacity is available.
- Advance options for rapid screening technologies and other innovative approaches to testing that improve efficiency and performance in settings such as workplaces and schools.
- Draw on external science and policy experts to develop and refine existing and innovative approaches to testing, including through the Testing and Screening Expert Advisory Panel.
- Collaborate with the private sector to help align their testing practices with public health advice and systems, and to grow testing capacity through partnerships.
- Support comprehensive testing and contact tracing in surge situations ("hot spots") by allocating emergency supplies and supporting laboratory capacity.
- Collaborate with P/Ts to improve and modernize data management across Canada, to help all orders of government coordinate efforts to contain the virus.
Building surge capacity to support P/Ts in managing the pandemic
- Establish the humanitarian workforce (civil workforce of volunteers) to maintain a strong surge response capacity for COVID-19 until end of March 2023.
- Enhance collaboration with P/Ts to develop new health worker recruitment solutions to support pandemic response.
- Establish, in collaboration with P/Ts and other partners, a COVID-19 Critical Drug Reserve to complement conventional F/P/T drug shortage management efforts.
Refining the COVID Alert App (exposure notification) and assessing emerging technologies
- Continue to work with and support P/Ts in the implementation of the app, and raise its benefits to help limit the spread of the virus.
- Develop tools to measure performance and adoption of the app.
- Establish, in collaboration with P/Ts and other partners, a COVID-19 Critical Drug Reserve to complement conventional F/P/T drug shortage management efforts.
- Develop new functionalities to further increase its effectiveness.
- Assess new technologies to prepare for and respond to emerging issues.
- Continue to implement marketing and outreach activities to promote the app to increase adoption and use by Canadians.
Given Health Canada's mandate, every action the Department took in 2020-21 and plans to take in 2021-22 considered COVID-19 impacts - the pandemic response cuts across each of its Core Responsibilities and all 5 associated Departmental Results. The overarching initiatives highlight Health Canada's coordinated response to protect the health of Canadians in these challenging times.
Branches within the Department will continue to work collaboratively and creatively across the organization, the health portfolio, the federal government, P/Ts, and the private sector, from coast to coast to coast, to ensure that Canada's response is appropriate and adaptable, based on the latest science, research and the evolving situation. In 2021-22, these initiatives will include:
Providing timely, trusted, and evidence-based information
Health Canada will continue to provide Canadians with useful, timely, evidence-based information to protect themselves, their families, their communities, and their businesses during the COVID-19 pandemic, using a range of online and traditional communications tools and channels. This includes the central information hub at Canada.ca/coronavirus, as well as digital applications such as COVID Alert, Canada COVID-19 app and the Wellness Together Canada portal. By January 2021, COVID Alert had been adopted by 9 P/Ts and downloaded more than 6 million times.
For those who may not have access to or use the Internet, Health Canada and PHAC will continue to provide regular media briefings and other outreach using a variety of channels such as radio, print and television advertising, traditional print mail outs, and a toll-free telephone information line available in multiple languages, 7 days a week. The Department will also continue to update Canadians on its regulatory work, such as efforts related to vaccines, and testing and screening.
Did you know?
The COVID-19 pandemic has had a significant impact on the lives of Canadians. Fear of the virus and the associated social disruption means that access to mental health supports has become more difficult at a time when these are most needed. In April 2020, Health Canada launched a free, online portal (Wellness Together Canada) to connect Canadians with mental health and substance use resources during COVID-19. By end of 2020, over 765,000 individuals across all P/Ts had accessed the portal in over 2 million distinct web sessions. This includes access to self-assessment, educational content, self-guided programming, peer-to-peer support, and one-to-one counseling by text and phone.
Promoting the Canada COVID-19 App and online self-assessment tool
Health Canada formed a digital surge team to explore, assess and support the procurement of digital tools and solutions in response to COVID-19. The response from Canadian researchers and innovators in the early stages of the pandemic was extremely strong, and the Department was able to rapidly establish an online self-assessment tool to help Canadians assess their symptoms and make informed decisions. By end of January 2021, this service had been accessed more than 8.9 million times. Health Canada also launched a mobile version of this tool ("Canada COVID-19") to create a hub for Canadians to access resources and remain informed about COVID-19. The Department will continue to manage and support these tools in 2021-22.
Supporting virtual care
In spring 2020, P/Ts made tremendous strides to put in place tools and supports for virtual services. To further accelerate this work so that Canadians could access necessary care during the pandemic, the Government announced in May 2020 a $240.5 million investment towards further implementing, maintaining and expanding these virtual care and mental health tools.
Of this funding, $150 million will be provided directly to P/Ts through bilateral agreements to support their efforts in the following shared priority areas:
- Secure messaging and information-sharing;
- Secure video-conferencing technology;
- Remote patient monitoring tools;
- Patient access to COVID-19 and other lab results;
- Integration and alignment of new platforms, tools and approaches into existing digital health systems.
In addition, Canada Health Infoway will receive $50 million to develop pan-Canadian standards for secure messaging and video-conferencing and to support P/Ts with their implementation and expansion of virtual care.
Facilitating access to health products
Health Canada will continue to work closely with domestic and international partners to anticipate and meet the needs ofCanadians for health products. Additionally, the Department will: provide scientific advice; expedite the review of clinical trial applications; authorize the sale of drugs and medical devices designed to diagnose, prevent and treat COVID-19 infections and related illnesses; assess the health risks of products; as well as address drug and medical device shortages.
Did you know?
In March 2020, during the early days of the pandemic, a COVID-19 Tiger Team was formed to address industry's needs, questions and concerns about drugs and medical devices. The focus quickly shifted to include healthcare professionals such as pharmacists, medical associations, nurses and hospitals. As a result of this early engagement, ongoing mechanisms to disseminate information were established and will continue to expand throughout 2021-22.
Authorizing clinical trials
In 2020, Health Canada implemented regulatory flexibilities associated with clinical trial applications for medical devices, drugs and vaccines relating to COVID-19 and prioritized approval of these applications. The Department will continue to assess the need for the accommodations put in place to address the urgent demand for these products. As of end of 2020, Health Canada had authorized over 81 clinical trials for potential treatments and vaccines.
Drug and vaccine authorizations
On September 16, 2020, the Minister of Health approved an Interim Order to import, sell and advertise drugs during the pandemic. This order helped expedite the authorization of drugs and vaccines for COVID-19. In late 2020, Health Canada authorized the treatment bamlanivimab and approved the first two COVID-19 vaccines, from Pfizer-BioNTech and Moderna. As part of its continued commitment to openness and transparency, the Department published a number of documents related to these decisions, including high-level summaries of the evidence that it reviewed to support authorization. Health Canada will continue to provide detailed information, such as scientific summaries and the full clinical trial data packages. In 2021-22, the Department will continue to expedite its scientific review of these products, without compromising Canada's high standards for safety, efficacy and quality. It will also prioritize the environmental assessment of vaccines and biologic drugs as well as the health assessment of all new chemicals or polymers used in drugs or medical devices related to COVID-19.
Did you know?
The Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19 allows Health Canada to respond to urgent public health needs by streamlining the authorization of COVID-19 treatments and vaccines. Under this expedited review process, Health Canada assesses data submitted by manufacturers as it becomes available during the product development process, while maintaining Canada's high regulatory standards.
The Department is working closely with PHAC to monitor the safety and effectiveness of vaccines and other products as they are deployed across the country and will rapidly investigate and mitigate any risks. If necessary, industry may be required to provide additional evidence to address any uncertainties. Furthermore, Health Canada will introduce new regulations in 2021-22 to ensure the continued availability of products approved under the Interim Order.
Did you know?
Health Canada conducts research in partnership with other federal agencies and international experts to address issues related to infectious diseases and their associated vaccines. This work has led to peer-reviewed publications supporting the advancement and regulation of vaccines for influenza, Zika and COVID-19, including a recent publication in the prestigious journal Cell that improved our understanding of variant strains of SARS-CoV-2.
Medical device authorizations
In March of 2020, Health Canada temporarily expedited the authorization of COVID-19 related medical devices, including testing devices, so that they could be imported and sold in Canada at an accelerated pace via the Interim Order Respecting the Importation and Sale of Medical Devices for Use in Relation to COVID-19. The Department has engaged with COVID-19 device manufacturers since the onset of the pandemic and has published guidance on several topics, including serology tests, ventilators, swabs, and PPE (including face coverings and respirators). As of end of 2020, Health Canada had authorized more than 500 medical devices under this temporary measure, including 332 PPE and 51 testing devices.
Health Canada intends to maintain these temporary regulatory measures until at least fall 2021, at which time the Department will need to amend its regulations to allow the products approved under the temporary measures to continue to be sold. In addition, Health Canada will prioritize the review of incident reports involving these medical devices to identify potential problems early, should they occur.
Sanitizers, disinfectant and cleaning products
From the onset of the pandemic to end of 2020, Health Canada authorized over 4,400 new hand sanitizer products, approximately 200 new disinfectants, and issued over 2,000 new site licences as per interim expedited service standards for over-the-counter drugs and natural health products. These requests are expected to increase significantly over 2021-22 as novel products are developed. The Department has also developed an online list of hard-surface disinfectants effective against COVID-19 (over 500 products as of December 2020).
In addition, to address a shortage during the COVID-19 pandemic, Health Canada introduced an interim measure to allow importation and sales of hand sanitizers (over 600 as of December 2020) that do not compromise safety because they are registered or authorized in other jurisdictions with similar regulatory regimes. The Department will continue to: conduct health and environmental risk assessments of these products; assess the risk and efficacy of disinfectants; liaise with international regulatory partners on novel approaches to regulation; and facilitate timely access to safe, effective, and high-quality products, registered under the Pest Control Products Act, that are used as sanitizers.
In April 2020, Health Canada implemented temporary policies to facilitate access to certain workplace and household cleaning products and soaps in the event of a supply shortage. In 2021-22, the Department will continue to monitor the situation and take appropriate action to balance the need to access key products during the pandemic, while complying with regulatory requirements.
Stabilizing the supply of critical COVID-19 drugs and managing drug and medical device shortages
In March 2020, the Minister of Health signed the Interim Order Respecting Drugs, Medical Devices and Foods for a Special Dietary Purpose in Relation to COVID-19. This temporary measure allows Health Canada to permit the exceptional importation and sale of products that are manufactured to comparable standards in foreign jurisdictions. The measure also requires that any shortages of medical devices related to COVID-19 be reported to Health Canada and publically communicated.
In October 2020, the Minister signed the Interim Order respecting the prevention and alleviation of shortages of drugs in relation to COVID-19. This temporary measure provides Health Canada with two new tools that can be applied to address shortage situations: the authority to compel information; and, the ability to add Terms and Conditions to an authorization or drug identification number.
In November 2020, the Minister signed the Interim Order Respecting Drug Shortages (Safeguarding the Drug Supply), which introduces new measures to help prevent bulk importation programs, such as the one recently established by the United States, from causing or worsening a drug shortage in Canada. Under the interim order, drugs intended for the Canadian market are prohibited from being distributed or sold out-of-country if this would cause or exacerbate a drug shortage. It also provides authority for the Minister to require information from a Drug Establishment License holder or manufacturer, related to a shortage or the risk of a shortage.
Health Canada intends to maintain these flexibilities until at least fall 2021, at which time the Department will consider regulatory amendments to allow the products approved under the temporary measures to continue to be sold.
Additionally, Health Canada will continue to monitor supply and demand of certain drugs and medical devices critical to the COVID-19 pandemic response to help mitigate and prevent shortages where possible. The Department is working with P/Ts and other partners to establish a COVID-19 Critical Drug Reserve, which will consist of up to a 6-month 'safety net' supply of drugs used in treating the symptoms of COVID-19. The initiative, which started in 2020-21 and will continue into 2021-22, complements current F/P/T efforts to manage the risk of shortages of key drugs. It will serve as an additional tool in the country's collective pandemic response efforts to help protect the health of Canadians.
Facilitating access to treatment services for substance use
Until September 2021, the Department will continue to issue a short-term subsection 56(1) exemption from the Controlled Drugs and Substances Act. This allows practitioners to verbally prescribe, and authorizes pharmacists to prescribe, sell, or provide controlled substances in limited circumstances, or transfer prescriptions for controlled substances. The exemptions also allow individuals to deliver controlled substances to those in isolation.
To further support people who use drugs during the COVID-19 pandemic, Health Canada proactively facilitated the rapid establishment of overdose prevention sites in community shelters for vulnerable individuals (e.g., persons who are homeless or housing unstable) exposed to COVID-19. In 2021-22, the Department will support increased access to evidence-based treatment services and expanded access to a safer drug supply by assessing these measures so that effective models can be scaled up.
Adapting compliance and enforcement approaches
In response to the pandemic, Health Canada adapted inspection activities to focus on areas of highest risk to public health and safety. The Department's hybrid approach to regulatory compliance and enforcement takes advantage of remote and virtual tools where the risk level permits, while focusing onsite inspections on situations involving higher risk to human health and safety. In 2021-22, Health Canada will continue to refine the use of remote and virtual tools to monitor compliance with regulatory requirements.
Enhancing Internal Services
All of Health Canada's Internal Services have been supporting the Department in response to the technological, financial and human resources needs created by the COVID-19 pandemic, in order to maintain operations and transition to a new working environment, without interruptions. Support was provided to the workforce, the workplace and the work itself so that Health Canada could continue to deliver on its mandate, whether employees were working remotely or on site.
In 2021-22, Health Canada will continue to: implement simplified and expedited staffing processes to support COVID-19 roles; respect public health guidelines and established safety protocols for onsite employees at all its facilities; and support employees working remotely through activities such as new Virtual Ergonomic Assessments.
The Department will also continue to: engage employees and seek feedback to evaluate and adjust communications and engagement strategies so they remain useful and relevant; check in with employees via virtual engagements to stay apprised on how the pandemic affects their professional and personal lives; and share information and resources to protect their mental and physical health and to support their work and productivity during the pandemic, through a variety of tools and platforms, including new web-based digital channels.
Key Risk for Health Canada's Response to COVID-19
Risk: Health Canada's ability to achieve its mandate may be at risk due to challenges posed by major health events (like COVID-19).
| Provide timely, trusted and evidence-based information | Continue to provide Canadians and health care providers with the timely, clear and evidence-based information they need to protect themselves. |
|---|---|
| Authorize clinical trials, and facilitate access to health products |
Invest efforts in the prevention and treatment of COVID-19 via clinical trials and flexible measures. For example:
|
| Foster engagement and collaboration |
Continue to work alongside PHAC and the CPHO via the COVID-19 Task Force to advance a whole-of-government approach by engaging and collaborating with partners and stakeholders to adapt to, and meet, the needs of Canadians. For example:
|
| Enhance internal services |
Continue the delivery of critical services and commitments during a major health crisis, by focussing on key activities such as:
|
Core Responsibility 1: Health Care Systems
Core Responsibility 1: Health Care Systems Description
Health Canada provides national leadership to support and encourage sustainable and adaptable health care systems that ensures access for Canadians to appropriate and effective health care services.
Core Responsibility 1: Health Care Systems Plans
Modern and sustainable health care systems are vital to addressing the health needs of Canadians. Although health care delivery is primarily under provincial and territorial jurisdiction, the federal government has an ongoing role in supporting health care innovation and collaboration across the country, maintaining the core principles of the Canada Health Act, and providing financial support through fiscal transfers to P/Ts. Health Canada will contribute to improving the quality and sustainability of health care as the systems continue to evolve in a context of technological and social changes, demographic shifts and fiscal pressures.
The United Nations 2030 Agenda for Sustainable Development and UN Sustainable Development Goals
Health Canada's planned activities under Core Responsibility 1: Health Care Systems directly support Canada's efforts to address the UN 2030 Agenda, particularly the Sustainable Development Goal 3 (SDG), promoting the good health and well-being of Canadians. For example:
- Promoting health care system and service delivery innovation, including e-prescribing and access to virtual care; improving patient safety and quality care; and strengthening Canada's health care systems with a focus on improving the capacity to protect vulnerable populations and high-risk communities.
- Improving access to appropriate and effective health care services (including MAID, home and community care, mental health and substance use services, and cancer care); supporting health human resources; and creating a national, universal system of pharmacare.
These planned activities also support the ongoing response to address the direct and indirect health impacts of COVID-19.
Departmental Result 1: Canada has modern and sustainable health care systems
Health Canada works closely with provincial and territorial governments, domestic and international organizations, health care providers and other stakeholders to develop and implement innovative approaches that improve the efficiency and sustainability of Canadian health care systems.
In support of its mandate, the Department conducts research and analysis and develops policy on such health care systems issues as: health expenditures and funding; primary care; supportive care (including home care, palliative and end-of-life care, and long-term care); medical assistance in dying; access to sexual and reproductive health services; affordability and accessibility of pharmaceuticals; health human resources and the impacts of health care systems modernization on the health workforce; quality of care; health care systems and service delivery innovation; and health technology. Since the beginning of the COVID-19 pandemic in January 2020, Health Canada also applies a COVID lens to all of its research, analysis and policy activities.
In 2021-22, Health Canada will make important investments to support several organizations that directly contribute to health system improvements. As part of this support, the Department will provide funding for: Canada Health Infoway; the Canadian Institute for Health Information; the Canadian Agency for Drugs and Technologies in Health; the Canadian Foundation for Healthcare Improvement; and the Canada Brain Research Fund. Highlights include:
- $85.5 million to Canada Health Infoway to advance digital health innovation, including the development of a pan-Canadian e-prescribing system and virtual care initiatives. As well, the funding will continue to support the adoption and use of electronic medical records, to help patients access their own health records electronically, and to better link electronic health record systems, improving access for all providers and institutions. An additional $50 million, started in 2020-21, will support P/Ts as they implement virtual care initiatives in their jurisdictions, and for the advancement of pan-Canadian virtual care initiatives.
- Up to $101.4 million to the Canadian Institute for Health Information (CIHI) to deliver actionable data analysis and information that accelerates improvements in health care, health system performance and population health across the continuum of care. This includes closing data gaps in such priority areas as: home care; mental health and addictions; pharmaceuticals; as well as organ donation and transplantation. In 2020-21, CIHI received funding up to $3.5 million under the Safe Restart Agreement to support health systems to address pressing data needs given the COVID-19 pandemic.
- $29.1 million to the Canadian Agency for Drugs and Technologies in Health (CADTH) towards strengthening the management of drugs and non-drug technologies. This funding will continue to support CADTH's core business activities, such as the Common Drug Review, health technology assessments and optimal use projects. It will also help facilitate the creation and dissemination of evidence-based information to support decision makers such as public drug plans and healthcare practitioners.
- $26.3 million to the Canadian Foundation for Healthcare Improvement (CFHI) towards accelerating the identification, spread and scale-up of health care innovations and to improve patient safety and quality care.Footnote 1 CFHI will focus the funding on: finding and promoting health care innovators and innovations; driving rapid adoption of proven innovations; enabling improvement-oriented systems; shaping the future of healthcare; safety improvement projects that evaluate and implement scalable, evidence-based best practices; using the patient voice in reporting and campaigns to raise awareness; as well as targeting policy, standards and regulations to influence Canada's health care systems towards safer care.
- CFHI is also receiving additional, time-limited funding to help expand its Long Term Care Plus (LTC+) initiative which is helping long-term care and retirement homes strengthen their pandemic preparedness and response. Health Canada provided an additional $2.05 million in 2020-21. The Government committed a further $6.4 million over two years, carrying through 2021-22, in the Fall Economic Statement 2020.
- $20 million, as per the Budget 2019 commitment, to the Brain Canada Foundation (Brain Canada) to support those affected by neurological injury and diseases. As well, the Department will continue augmenting innovative neuroscience discovery research through the Canada Brain Research Fund, which is administered by the Foundation. Health Canada is matching private and charitable contributions raised by Brain Canada, up to $160 million by 2022.
What's new?
Health Canada's support to Brain Canada will be invested in 3 key areas: projects across the research continuum from basic to clinical science; funding early-career researchers seeking to enter the brain research field; and strengthening critical neuroscience research platforms. Investments span diverse brain health conditions, including brain injury, brain cancer, mental illness, and neurodegenerative disorders. Brain Canada expects that a number of research proposals will address the longer-term impacts of COVID-19 and its connection to neurological health.
Through these investments, Canadians will gain greater access to their own medical records and health care providers will be able to coordinate patient care using digital technologies. Policy makers will have access to better information on the performance of the health care system and cost-effectiveness of drugs and technologies to support evidence-based decision-making. Promising innovations and best practices in service delivery will become more readily identified and shared across jurisdictions. Progress on these initiatives will continue to be monitored through indicators on drug spending and health expenditure at the system-level.
Did you know?
To enable Canadians to assess progress on shared health priorities, the Canadian Institute for Health Information (CIHI) led a process whereby Federal-Provincial-Territorial Ministers of Health agreed to a set of 12 common indicators for home and community care and mental health and addiction services. The Institute has now released its second annual report on the results for first 6 Pan-Canadian indicators.
The Government of Canada is working closely with P/Ts to expand virtual health services so that Canadians can continue to access the care they need during the pandemic. Based on the prime ministerial commitment in May 2020 of $240 million, Health Canada will implement virtual approaches and digital tools in 2021-22 to help Canadian health systems meet health care needs.
Departmental Result 2: Canadians have access to appropriate and effective health services
Through Health Canada, the federal government is responsible for promoting and defending the core principles of the Canada Health Act - public administration, comprehensiveness, universality, portability and accessibility - and ensuring provincial and territorial health care insurance plans provide reasonable access to health services without financial or other barriers, such as patient charges for insured services.
The federal government also plays an important role in providing financial contributions to P/Ts to support publicly funded health care services through the Canada Health Transfer (more than $41.9 billion for 2020-21Footnote 2) and through targeted funding to support improved access to home and community care, and mental health and addiction services (Budget 2017: $11 billion over ten years).
In 2021-22, to ensure that Canadians have access to appropriate and effective health services, Health Canada will focus on the following priorities, detailed below: access to mental health and substance use services and to home and community care; primary care; national pharmacare; palliative care; medical assistance in dying (MAID); diagnostic services; thalidomide survivors support; combatting cancer; organ, tissues and blood donation and transplantation; as well as supporting access to health services for specific populations.
Access to mental health and substance use services and to home and community care
Following agreement by Federal-Provincial-Territorial Ministers on a Common Statement of Principles on Shared Health Priorities in 2017, the federal government negotiated and signed bilateral agreements with all 13 P/Ts, for two 5-year periods (2017-18 to 2021-22 and 2022-23 to 2026-27). These agreements detail how each jurisdiction is using federal funding of $11 billion over 10 years to improve access to home and community care and mental health and substance use services. By March 2022, approximately $5 billion of the commitment will have flowed through to address these priorities. In 2021-22, the government is committed to renewing these agreements for the remaining 5 years of the 10-year commitment.
Key initiatives for 2021-22 involve expanding access to community-based mental health and substance use services for children and youth and enhancing supports for family caregivers at home. In addition, Health Canada will work with all its partners in health to: set national standards for access to mental health services to improve access to the support Canadians need, when they need it; make home care and palliative care more available across the country; and set new, national standards for long-term care and take additional action to help people stay in their homes longer, so that seniors get the best support possible.
Furthermore, the Department intends to direct $14.2 million to the Mental Health Commission of Canada to advance specific priorities in the areas of substance use, suicide prevention, population-based initiatives, and engagement. It will also provide up to $47 million (including $22.6 million for the Terry Fox Research Institute and Ovarian Cancer Canada) in fiscal year 2021-22 to address federal priorities under the Health Care Policy Strategies Program, including home and community care, mental health, palliative and end-of-life care, and other legacy and emerging priorities.
Primary care
Health Canada will explore innovative approaches with its partners in health - including new service delivery models, digital and virtual care solutions - to help ensure that every Canadian has access to a primary care provider or team.
Our collective response to COVID-19 has led to the development of a new stream of collaborative work with P/Ts around virtual care - new bilateral agreements are providing $150 million to help jurisdictions meet health care needs through virtual services. Over 2021-22, Canada Health Infoway will develop pan-Canadian standards and provide support to P/Ts to implement virtual care initiatives under the bilateral agreements, up to an additional $50 million in federal investments.
National pharmacare
Over the course of 2021-22, Health Canada will continue to work with its partners in health to implement a national, universal pharmacare system so that Canadians have the drug coverage they need, by establishing the Canadian Drug Agency transition office and developing a national strategy for high-cost drugs for rare diseases. As stated in the Speech from the Throne in September 2020, Health Canada will accelerate steps to achieve this system, including: establishing a national formulary to keep drug prices low, as well as working with P/Ts to move forward without delay.
These efforts will be supported by funding proposed in Budget 2019, including $35 million over 4 years for the transition office, and up to $1 billion over two years (starting in 2022-23), and up to $500 million per year ongoing, to develop the national strategy for high cost drugs for rare diseases.
Palliative care
Health Canada will continue to implement the 5-year Action Plan on Palliative Care (2019-20 to 2023-24). Various activities that were under development pivoted to incorporate pandemic palliative care into their objectives. For example, certain projects will provide patients with the support they need to stay out of acute care settings for as long as possible, such as: training volunteer navigators who help tap into local home care supports and services; and developing a remote symptom monitoring system that connects patients in the home and community with palliative care experts via a daily reporting application.
Various Action Plan activities that were delayed in 2020-21 by prioritization of the COVID response - such as developing a public awareness campaign and collaborating with Indigenous Services Canada on Indigenous engagement towards a distinctions-based Framework on palliative care - will continue in 2021-22.
Medical assistance in dying (MAID)
Health Canada will continue to support implementation of MAID as well as the Government's response to the 2019 Quebec Superior Court ruling regarding the MAID framework. In 2021-22, activities will include:
- Supporting Parliamentary consideration of Bill C-7 [ An Act to amend the Criminal Code (medical assistance in dying) ] and, if passed, supporting P/Ts in implementing the legislative changes;
- Supporting a Parliamentary review of the MAID law provisions and the state of palliative care in Canada as required by MAID legislation;
- Ongoing implementation of the MAID Monitoring Program (data collection about requests for, and provision of MAID across Canada) including release of the second federal Annual Report on MAID in Canada;
- Preparing and consulting on a set of revised federal Regulations for the MAID Monitoring Program.
Diagnostic services
On April 1, 2020, the Diagnostic Services Policy, which formalized the application of the Canada Health Act to diagnostic services, came into force. The policy confirmed the longstanding federal position that medically-necessary diagnostic services, such as Computerized Tomography Scan (CT Scan) and Magnetic Resonance Imaging (MRI), are insured services, regardless of the venue where the services are delivered. The Department will continue to work with jurisdictions to ensure compliance with the new policy.
Canadian thalidomide survivors support
The purpose and long-term outcome of the Canadian Thalidomide Survivors Support Program (CTSSP) is to contribute to meeting the lifetime needs of Canadian thalidomide survivors, allowing them to age with dignity. The CTSSP provides a fair and comprehensive approach to identifying thalidomide survivors that is based on international best practices. The Program remains committed to identifying opportunities for service delivery improvement.
Combatting cancer
In 2021-22, Health Canada will continue to support the expansion of a network of Canadian cancer centres to advance precision medicine in cancer research by providing over $20.3 million to the Terry Fox Research Institute.
The Department will also provide ongoing support for ovarian cancer research via $2.25 million in funding to Ovarian Cancer Canada to address gaps in knowledge about effective prevention, screening, and treatment options.
In addition, Health Canada will invest $52.5 million in funding to the Canadian Partnership Against Cancer (CPAC) to continue its work on key health issues in cancer control. CPAC will accelerate the uptake of new knowledge and coordinate approaches to advance cancer control across the country.
What's new?
Health Canada funding will help The Terry Fox Research Institute expand its national Marathon of Hope Cancer Centres Network. The network will facilitate the participation of thousands of Canadians in a cancer research project that will improve therapeutic decision-making related to the application of precision medicine. For example, the research could help to identify which patients will respond to new drug treatments or guide future investigation of new treatments. The Institute is matching the Department's funding through a combination of its own resources and contributions from other non-federal government sources.
Organ, tissues and blood donation and transplantation
Health Canada will continue to support collaboration among its partners in health and Canadian Blood Services to establish a coordinated organ and tissues donation and transplantation system that gives Canadians more timely and effective access to care. This includes developing a decision-making and accountability framework and a data and performance system that promotes a pan-Canadian approach to organ donation and transplantation. The Department will also build on existing progress to maximize safety and non-discrimination in blood and plasma donation policies.
Supporting access to health services for specific populations
Over the course of 2021-22, Health Canada will also make important investments in additional programs and organizations that directly support access to appropriate and effective health services for Canadians. For example, investments will help to address the health care needs of specific populations such as those living in Canada's territories and official language minority communities, taking into account findings from the 2017 Evaluation of the Official Languages Health Contribution Program (now called the Official Languages Health Program).
Did you know?
Health Canada provides $37.6 million per year under the Official Languages Health Program to community-based organizations, governments and academic institutions to improve access to health services for English-speaking communities in Quebec and French-speaking communities elsewhere in Canada. Funded activities will include the integration of bilingual health personnel through postsecondary training initiatives, the development of strategies and partnerships with health system stakeholders through community networking, and the implementation of innovative projects aimed at improving access to bilingual health services and fostering knowledge development and dissemination in official languages minority communities.
Key Risk for Core Responsibility 1: Health Care Systems
Risk: Health Canada's ability to effectively uphold the Canada Health Act (CHA) could be put at risk by challenges in administering the Act.
Key examples of Health Canada's planned risk responses:
- Implement new policies
- Work with P/Ts to ensure compliance with the new Diagnostic Services Policy, which took effect in April 2020.
- Work to resolve issues with P/Ts
- Evaluate provincial and territorial Reimbursement Action Plans to ensure the elimination of patient charges.
- Monitor litigation
- Monitor litigation that may impact the CHA, and support federal involvement as required.
- Monitor changes in health care delivery
-
Monitor changes in health care delivery for their effect on access to insured services. For example:
- Monitor and assess virtual access to physician services, which has been rapidly growing in Canada and around the world.
Planned results for Core Responsibility 1: Health Care Systems
| Departmental Result Indicators | Target | Date to achieve target | Actual Results |
|---|---|---|---|
|
National health expenditure as a percentage of Gross Domestic Product (GDP) (Baseline: 10.9% in 2014-15) |
Between 10.4% and 12.8% | March 31, 2022 |
2017-18: 11.5%Tablenote 1 |
|
Real per capita health expenditure (Baseline: $4,049 per person in 2014-15Footnote 2) |
Between $3,913 and $4,782 | March 31, 2022 |
2017-18: $4,245Tablenote 1 |
|
Drug spending as a percentage of Gross Domestic ProductFootnote 3 (Baseline: 1.7% in 2014-15) |
Between 1% and 2% | March 31, 2022 |
2017-18: 1.79%Tablenote 1 |
|
Percentage of family physicians using electronic medical records (Baseline: 73% in 2015) |
At least 95% | March 31, 2022 |
2017-18: 73% |
Tablenotes
|
|||
| Departmental Result Indicators | Target | Date to achieve target | Actual Results |
|---|---|---|---|
|
Percentage of Canadians (aged 15+) with a mental disorder who have expressed that they have an unmet mental health care need (Baseline: 26% in 2012) |
At most 22% | March 31, 2022 |
2017-18: 26.0%Result 2 Tablenote 1 |
|
Percentage of Canadians (aged 18+) who have expressed that they have an unmet need for access to home care services (Baseline: 1.6% in 2015-16) |
At most 1.0% | March 31, 2027 |
2017-18: 1.5%Result 2 Tablenote 4 |
|
Percentage of Canada Health Act compliance issues addressed within 24 months of identification (Baseline: 53% in 2016-17) |
At least 95% | March 31, 2022 |
2017-18: 94% |
|
Percentage of Canadians (aged 12+) who did not fill a prescription for medicine or skipped doses of medicine because of the cost (Baseline: 7.1% in 2014) |
At most 5% | March 31, 2022 |
2017-18: 7.1%Result 2 Tablenote 6 |
Tablenotes
|
|||
Planned budgetary financial resources (dollars) for Core Responsibility 1: Health Care Systems
| 2021-22 budgetary spending (as indicated in Main Estimates) |
2021-22 Planned spending |
2022-23 Planned spending |
2023-24 Planned spending |
|---|---|---|---|
| 2,456,807,897 | 2,456,807,897 | 1,558,123,313 | 1,566,896,000 |
| Note: The decrease in planned spending is mainly due to funding level decreases in 2022-23 for Long Term Care and other Supportive Care settings in the context of COVID-19; Strengthening Canada's Home and Community Care and Mental Health and Addiction Services Initiative; as well as the expiry of budgetary spending authorities in 2021-22 for Canada Health Infoway. The Department would have to request funding for this initiative for future years. | |||
Planned Human resources (full-time equivalents) for Core Responsibility 1: Health Care Systems
| 2021-22 Planned full-time equivalents |
2022-23 Planned full-time equivalents |
2023-24 Planned full-time equivalents |
|---|---|---|
| 276 | 272 | 266 |
|
Note: The decrease in planned FTEs is mainly due to the expiry of budgetary authorities in 2022-23 related to Long Term Care and other Supportive Care settings in the context of COVID-19. The Department would have to request funding for this initiative for future years. |
||
Financial, human resources and performance information for Health Canada's Program Inventory is available in the GC InfoBase.
Core Responsibility 2: Health Protection and Promotion
Core Responsibility 2: Health Protection and Promotion Description
Health Canada works with domestic and international partners to assess, manage and communicate the health and safety risks and benefits associated with health and consumer products, food, chemicals, pesticides, environmental factors, tobacco and vaping products, cannabis, and controlled substances.
Core Responsibility 2: Health Protection and Promotion Plans
The Department will continue to advance the Regulatory Innovation Agenda arising from the Health and Biosciences Sectoral Regulatory Review Roadmap and the Agri-food and Aquaculture Regulatory Review Roadmap. The Agenda cuts across multiple Departmental Results that make up Core Responsibility 2. It consists of several projects designed to make the federal regulatory framework more agile and responsive to an innovative environment, while ensuring the system remains science and safety-based. Its implementation will result in health product and food regulatory frameworks that protect the health and safety of Canadians with proportional oversight relative to any associated risks, while encouraging innovation. Some of the key activities Health Canada will advance over 2021-22 include:
- Continuing to develop proposals to modernize clinical trial regulations and make them more agile for the licensing of drugs and medical devices;
- Completing underlying policy and operational work required to implement a new regulatory pathway for advanced therapeutic products;
- Modernizing the Department's compliance and enforcement regulatory frameworks for health products;
- Maximizing international partnerships to share the oversight of foreign facilities;
- Continuing to modernize processes and regulations relating to pest control products, including facilitating the implementation of an integrated pesticide program;
- Creating modern and agile food regulations, including: establishing a regulatory framework for foods marketed through supplementation (i.e., foods containing added vitamins, minerals, and herbal ingredients) and modernizing oversight of microbiological criteria, official methods of analysis, and food additives;
- Improving the way we communicate with Canadians and industry about health products and food, including regulatory requirements and processes.
These efforts will be informed by the Department's experience in responding to COVID-19, which leveraged many of the regulatory improvements envisioned in the Agenda. These temporary measures put in place to respond to the pandemic provided an opportunity to pilot many of the planned improvements. For example, Health Canada launched temporary emergency measures to help companies bring urgently needed health products and medical supplies for COVID-19 to market. These measures included broadening access to clinical trials, expediting the review of health products and improving management of product shortages, while maintaining robust approaches to compliance and enforcement.
What's new?
Health Canada will develop in 2021-22 a Health Product Consumer Information application to provide Canadians with accurate and unbiased information. This will empower consumers to make informed decision on health products.
The United Nations 2030 Agenda for Sustainable Development and UN Sustainable Development Goals
Health Canada's planned activities under Core Responsibility 2: Health Protection and Promotion directly support Canada's efforts to address the UN 2030 Agenda and the related Sustainable Development Goals (SDGs). For example:
- Providing information to Canadians on healthy eating, prioritizing vulnerable populations at risk of food insecurity, and undertaking international collaboration on food safety and nutrition standards support (SDG 2).
- Ensuring access to safe, effective and quality health products through an innovative regulatory framework; and supporting healthier living and protection from unsafe substances through the Canadian Drugs and Substances Strategy and Substance Use and Addictions Program; to promote the good health and well-being of Canadians (SDG 3).
- Establishing Canadian drinking water guidelines to improve drinking water quality (SDG 6).
- Working with partner departments to support the safe and sustainable use of pesticide products, contributing to Canada's efforts towards sustainable consumption and production patterns (SDG 12).
- Building capacity through initiatives with P/Ts to support the health sector in preparing for and adapting to the impacts of climate change (SDG 13).
These planned activities also support the ongoing response to address the direct and indirect health impacts of COVID-19.
Departmental Result 3: Canadians have access to safe, effective and quality health products
Over the course of 2021-22, Health Canada will continue to make investments towards ensuring Canadians have timely access to safe, effective and quality health products (including prescription and non-prescription drugs, biologic and radiopharmaceutical drugs, natural health products, and medical devices) and to meet the needs of the health care system. Improvements will help accelerate market access for innovative, breakthrough products along with cost effective alternatives, such as generic drugs. The Department will focus on the following priorities, detailed below: addressing drug and medical device shortages; promoting timely access to health products; modernizing the way we provide access to drugs not readily available; applying real-world evidence to support regulatory decision-making; strengthening regulatory oversight; modernizing compliance and enforcement; combatting antimicrobial resistance (AMR); fostering international collaboration and coordination; as well as promoting access to new and renewed technologies.
Addressing drug and medical device shortages
Health Canada continues to take action on drug and medical device shortages to ensure that Canadians have access to the medicines and devices they need. In partnership with P/Ts, industry, and patient/health care groups, the Department will continue to play a leadership role in addressing critical national drug and medical device shortages, including advancing new regulatory tools to help alleviate and prevent shortages from happening, where possible.
Promoting timely access to health products
In 2021-22, Health Canada will continue to provide Canadians with timely access to safe health products by reviewing the safety, efficacy and quality of pharmaceutical and biologic drugs, medical devices and natural health products.
What's new?
Nitrosamines are compounds that can form in certain drugs during manufacturing. Some nitrosamines may increase the risk of cancer if people are exposed to them over long periods of time. In 2021-22, Health Canada will work with manufacturers and international partners to manage nitrosamine impurities in drugs. The Department will communicate with Canadians regarding affected drugs, and collaborate internationally to establish impurity limits, better understand the causes of contamination, and determine how to prevent these impurities in the future.
The Department will continue to improve access to generic drugs by permitting the filing of certain different physical or chemical forms of the medicinal ingredient as a generic submission. This initiative will help provide Canadians with access to a wider range of low-cost generic medicines.
To address challenges and barriers that can hinder access to safe and effective medicines for infants, children and adolescents, the Department developed a Pediatric Drug Action Plan in 2020-21. Under this initiative in 2021-22, Health Canada will improve access to essential medicines, including age-appropriate formulations, for pediatric populations in Canada, by working with various stakeholders, including manufacturers and academia.
Modernizing the way we provide access to drugs not readily available
Health Canada will propose amendments to the Food and Drug Regulations to allow for the authorization of human and veterinary drugs that fulfill an unmet medical need. Such drugs must already be approved by a trusted foreign regulator and have been on the market long enough in those countries to demonstrate their safety. This will facilitate access to medically-necessary drugs that are not currently available on the Canadian market. It will also enable animal owners and food producers to access veterinary drugs not authorized in Canada for minor uses or minor species. The Department will introduce these proposed regulations as part of the Agile Licensing for Drugs initiative from the Regulatory Innovation Agenda, which it intends to pre-publish in Canada Gazette, Part I in spring 2022.
Furthermore, Health Canada intends to finalize a new regulatory framework to enable public health officials responsible for public and military health emergencies to request access to drugs that are unavailable in Canada, for emergency preparedness and response activities.
Applying real-world evidence (RWE) to support regulatory decision-making
The Department will continue to apply RWE in support of regulatory decisions, in order to improve the extent and rate of access to prescription drugs in Canada. It will develop additional guidance on using RWE and will finalize a strategic plan with Health Technology Assessment partners.
Health Canada will continue to participate on the Drug Core Action Team along with the Canadian Agency for Drugs and Technologies in Health, l'Institut national d'excellence en santé et en services sociaux, the Canadian Institute for Health Information, and industry. This group is an advisory body that guides and supports the development of a pan-Canadian approach to the use of RWE and identifies where RWE can add value to regulatory decision-makers throughout a drug's lifecycle.
Strengthening regulatory oversight
In 2021-22, the Department will advance its efforts to modernize the regulatory framework for self-care products (natural health products and non-prescription drugs). In spring 2021, the Department plans to broadly consult on a regulatory proposal to improve the labelling of natural health products, including introducing label disclaimers for homeopathic products. These proposed changes will support Canadians in selecting and safely using these products.
Furthermore, Health Canada intends to introduce: a simplified pre-market authorization process for lower-risk non-prescription drugs; strengthened post-market authorities to permit the Department to act swiftly when a risk is identified; and a risk-based approach to Establishment Licensing and Good Manufacturing Practices, including for active pharmaceutical ingredients.
Did you know?
Health Canada regulates natural health products, so that Canadians can have confidence that the products they use are safe, effective and of high quality. Health Canada is committed to regulating NHPs under the Natural Health Products Regulations, which means that NHPs are not subject to non-prescription and prescription drug requirements. Labels are an important tool to assist Canadians in making informed health choices when selecting and using NHPs.
The Department will extend and evaluate a pilot project to provide early scientific advice to drug sponsors, in collaboration with the Canadian Agency on Drugs and Technologies in Health. It also intends to advance regulatory proposals to modernize the framework for licensing establishments related to health products and medical devices, with the goal of increasing regulatory decision-making efficiency and predictability. The revised approaches will: better support new and innovative companies by allowing them to begin operations sooner; facilitate industry compliance; maintain Canada's international reputation; as well as create a level playing field for Canadian pharmaceutical and medical device sectors.
Health Canada regulates new and emerging medical device technologies in part by assessing post-market data and encouraging patient involvement throughout the regulatory lifecycle. In 2021-22, the Department will continue to improve scientific advice for medical device sponsors and increase stakeholder engagement through a stronger web presence targeted to an expanded audience. It will also encourage all parties to contribute towards a shared responsibility for post-market surveillance of medical devices. Health Canada will also:
- Update guidance regarding mandatory reporting of medical device incidents;
- Modernize the process for industry to report medical device incidents to the Department, as well as the associated online database;
- Continue to solicit feedback from the Canadian Medical Devices Sentinel Network to validate safety signals (early indications of product-related safety issues);
- Implement regulations to support the surveillance of device safety and effectiveness;
- Improve access to data through RWE initiatives in collaboration with federal partners;
- Improve access to information on the medical devices that Canadians use, such as providing information online regarding high-risk implantable devices.
Health Canada assesses biocides (e.g., disinfectants and sanitizers) for their safety, efficacy and quality under the Food and Drug Regulations and the Pest Control Products Regulations. The Department proposes to continue leveraging foreign authorizations during the COVID-19 pandemic and to develop a framework for biocides in 2021-22. Consultations with key stakeholders regarding both the proposal and lessons learned from the pandemic began in the fall of 2020.
Modernizing compliance and enforcement
As part of its Compliance and Enforcement (C&E) Modernization and Transformation priority, Health Canada has committed to becoming more agile, assertive, consistent, innovative, proactive, and risk-based in its C&E approach. To guide this transformation, the Department has initiated work encompassing 6 critical areas: regulatory framework development; quality management systems; training for C&E designation; IT systems and tools; data analytics; and risk management tools. In 2021-22, Health Canada will:
- Develop a Risk-Based Decision Making Policy for inspection programs;
- Continue to modernize compliance programs by adopting virtual and remote tools for inspections where appropriate to do so;
- Improve risk management tools to focus on highest risk regulated parties.
Combatting antimicrobial resistance (AMR)
Recognizing that AMR is an urgent public health issue, and in accordance with the Federal Action Plan on Antimicrobial Resistance and Use in Canada and the Pan-Canadian Framework for Action on Tackling Antimicrobial Resistance and Antimicrobial Use, Health Canada continues to take a One Health approach to addressing the associated risks. A number of initiatives are underway to safeguard the use of available antimicrobial drugs for human use, raise awareness, promote innovation, and monitor and support the prudent use of antimicrobials in animals. The Department will continue to engage international partners, including the International Coalition of Medicines Regulatory Authorities, the Transatlantic Task Forces on Antimicrobial Resistance and other bilateral/multilateral partnerships towards this end. Key initiatives for 2021-22 include:
- Updating the human Pathogens of Interest List, to ensure it continues to capture emerging threats (such as fungal pathogens) requiring new therapeutic and diagnostic options in Canada;
- Encouraging manufacturers to submit for review innovative antimicrobials for human use, as appropriate during the dynamic pandemic situation;
- Begin targeted re-evaluations of medically important antimicrobials (MIAs) being used in animals, following the publication of the post-market re-evaluation of veterinary drugs, to support their prudent use;
- Sharing of reported antimicrobial sales data on MIAs for use in animals to support the AMR surveillance program and to inform policy decisions that strengthen antimicrobial stewardship;
- Continuing to facilitate access to low-risk veterinary health products that improve health and wellness in animals so that there is less need for antimicrobials. This includes working with the Canadian Food Inspection Agency on an interim pilot project for incorporating such products in livestock feed, with gradual expansion until amendments to the Feeds Regulations are implemented;
- Continuing efforts with partners and stakeholders to raise awareness about the growing threat of AMR.
Did you know?
Health Canada's approach to combatting AMR is based on the World Health Organization's concept of One Health. It entails a collaborative, multisectoral, and transdisciplinary approach - working at the local, regional, national, and global levels - with the goal of achieving optimal health outcomes recognizing the interconnection between people, animals, plants, and their shared environment.
Fostering international collaboration and coordination
Health Canada will continue to support access to drugs via bilateral arrangements with foreign regulators, international multilateral initiatives/networks, a set of quality management tools, rigorous regulatory project management practices, and transparency initiatives.
The Department will strengthen its international collaborations in the review of drug submissions, risk management plans and surveillance through initiatives such as Project Orbis and will promote international alignment and greater efficiencies in the drug review process via the newly expanded Australia-Canada-Singapore-Switzerland Consortium. The United Kingdom's Medicines and Healthcare Products Regulatory Agency joined this existing collaboration in 2021, providing a broader partnership to support global availability of medicines and international scientific alignment. Health Canada will also exchange knowledge and cooperate on drug reviews with the European Medicines Agency as an observer at the Committee for Medicinal Products for Human Use, and collaborate on reviews of COVID-19 vaccines and treatments.
The Department will continue to be active as a standing member of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. In this role, it will contribute to decision-making and participate in developing internationally-harmonized guidelines to align technical requirements for human drugs.
Also in 2021-22, Health Canada and the U.S. Food and Drug Administration, under the auspices of the International Medical Device Regulators Forum, will conduct a pilot project towards developing a Medical Device Single Review Program in order to improve patient access to medical devices, support innovation, and strengthen the development of standards.
What's new?
Health Canada and the U.S. Food and Drug Administration (FDA) will share information about Amyotrophic lateral sclerosis (ALS) drugs in development to facilitate the Canadian review process. Furthermore, the FDA has invited Health Canada to participate in its "Rare Disease Cures Accelerator" initiative to develop common standards related to rare diseases, find ways to incorporate the patient's perspective in clinical outcome assessments, and build clinical trial readiness. This will provide an opportunity to work jointly to expedite the development of treatments for rare disorders.
Promoting access to new and emerging technologies
Scientific and technological advances are accelerating the pace of innovation in the healthcare system, leading to the development of innovative health products that use emerging technologies such as advanced artificial intelligence (AI) and machine learning (ML) algorithms, telerobotics, 3D printing and gene editing. Increasingly, health products are becoming personalised, developed at point of care, and manufactured, distributed, and used in significantly new and untraditional ways.
To keep pace with these emerging technologies, Health Canada established, in 2018, the Digital Health Division within the Medical Devices Directorate. In 2021-22, the Department will develop guidance on pre-market requirements for locked AI/ML algorithms in medical devices. In addition, Health Canada will continue to hold meetings of the Scientific Advisory Committee in Digital Health, and participate as an active member of both the World Health Organization (WHO)/International Telecommunication Union (ITU) AI4Health Focus Group and the International Medical Devices Regulators Forum AI Working Group.
The Department will also implement a new regulatory pathway to accommodate advanced therapeutic products (defined as drugs or devices that are innovative, complex and distinct). As a key component of Health Canada's Regulatory Innovation Agenda, this work will support timely access for patients while optimizing safety and benefits, as well as strengthening innovation in the health and biosciences sector. The Department will consult stakeholders regarding potential advanced therapeutic product candidate(s) to pilot and on the concept of a new 'concierge service' to support them in navigating the new pathway.
Departmental Result 4: Canadians are protected from unsafe consumer and commercial products and substances
Helping Canadians lead healthier lives and providing protection from unsafe consumer and commercial products and substances will remain an important focus of Health Canada's work. Over the course of 2021-22, the Department's efforts in this regard will focus on the following priorities, detailed below: a comprehensive approach to substance use-related harms; regulating cannabis; chemicals management; improving workplace safety through the safe use of chemicals; environmental assessment of drugs; supporting the safety of consumer products and cosmetics; protecting Canadians from radiation; as well as strengthening pesticide regulation.
A comprehensive approach to substance use-related harms
Substance use-related harms continue to cause devastating health and social effects on Canadians from every walk of life. For many people, and in many jurisdictions, the COVID-19 pandemic has exacerbated these harms.
The opioid overdose crisis in particular continues to have significant impacts on Canadian communities and families. Between January 1, 2016 and June 30, 2020, opioid overdoses claimed the lives of 17,602 Canadians, making it one of the most serious public health concerns in a generation. The scale of this crisis is so severe that in 2019, life expectancy in Canada stopped increasing for the first time in more than 40 years.
As a result of considerable efforts among all levels of government, front-line workers, researchers, and advocates, opioid-related harms and deaths were actually trending downward at the end of 2019 and beginning of 2020. Tragically, several jurisdictions across Canada saw an alarming reversal in the months following the onset of COVID-19. Likely contributing factors include increased feelings of isolation, stress and anxiety, and limited availability or accessibility of services for those who use drugs during the outbreak. In addition, the illegal drug supply in Canada, already contaminated with highly potent fentanyl or fentanyl-like drugs, has grown more unpredictable and toxic as supply chains are disrupted by COVID-related travel restrictions and border measures.
The Government is deeply concerned about the number of drug-related overdoses and deaths in Canada, and recognizes the serious consequences this crisis is having for individuals and communities across the country. It is especially troubled by the disproportionate impact of the opioid overdose crisis on young-and middle-aged men and Indigenous Peoples. In 2021-22, Health Canada will continue to respond with a public health approach that is comprehensive, collaborative, coordinated, and compassionate. This involves working closely with all of its partners in health - other federal departments, P/Ts, Indigenous communities, private sector organizations, and a wide range of other stakeholders, including substance use experts, service providers, and people with lived and living experience.
Making real progress requires the Department to examine root causes in order to better prevent problematic substance use at the outset, while continuing to support efforts to prevent overdose and death, as well as treatment and recovery options for people who use drugs. The key to success will be collaborating with partners to support a wide range of community-led initiatives and interventions. The end goal is for fewer Canadians to face substance-use related harms.
Did you know?
The Canadian Drugs and Substances Strategy is delivered by the Minister of Health, in collaboration with 14 other federal departments and agencies, and includes 4 pillars - prevention, treatment, harm reduction and enforcement.
The Strategy enables the Government to: better identify significant health risks and trends associated with substance use; effectively develop and target interventions to prevent and reduce harms and promote treatment and recovery; monitor and report on impacts; and support overall development of drug and substance use policies.
Unmanaged pain and the trauma and complexity that often accompany it is a primary driver of substance use and a barrier to successful treatment of substance use disorders. As such, the Department will continue to support the Canadian Pain Task Force (including a $1 million financial contribution in 2021-22) in their updated mandate to recommend priority actions that ensure people with pain are recognized and supported and that pain is understood, prevented and effectively treated throughout Canada. This will include: continuing engagement with stakeholders across Canada; disseminating information about chronic pain and best practices; and publishing the Task Force's final report.
Health Canada will continue to work with domestic and international partners to build the evidence base and improve access to treatment services for substance use disorder. This includes developing integrated approaches to treating mental health, chronic pain, and other disorders (for example, through the Emergency Treatment Fund). It will also focus on finding new ways of better reaching young and middle-aged men to help improve education and awareness of substance use and harms and reduce stigma in order to increase help-seeking.
Did you know?
Stigma and discrimination form one of the biggest barriers to seeking and receiving treatment for drug use. Research conducted in 2019 showed that substance use stigma persists in Canada, despite increased agreement that more money should be spent on supporting those with substance use disorders. The majority of overdose deaths in Canada occur at home alone. Reducing stigma can help lessen people's likelihood of using drugs alone, increase their connection to treatment and harm reduction services, and increase public support for life-saving initiatives (e.g., safe consumption sites). Reducing stigma continues to be a priority for Health Canada.
The Canadian Drugs and Substances Strategy (CDSS) outlines the Government's comprehensive, collaborative, compassionate, and evidence-based approach to problematic substance use, with the goal of minimizing harms from substance use on individuals, families and communities. The CDSS covers a broad range of substances, including alcohol, cannabis, illegal drugs, and the problematic use of prescription drugs. In 2021, the Department will publish an updated and fully articulated Strategy, based on advice from the Ministerial Expert Task Force on Substance Use (created in January 2021) and on public input from 2018 related to federal drug policy. This input also identified several barriers that make it difficult for people to access appropriate substance use services, particularly as they relate to those who provide training and care. Health Canada is working with P/Ts to better understand these barriers. Furthermore, the CDSS will continue to guide the development of initiatives in response to the COVID-19 pandemic.
Through the Substance Use and Addictions Program (SUAP), Health Canada will continue to support a range of prevention, harm reduction, and treatment initiatives that respond to drug and substance use issues in Canada. The Department will provide up to $115.4 million towards the following initiatives:
- Strengthening the response to the opioid crisis and other emerging issues, including: harm reduction, community-led and frontline initiatives; increasing access to pharmaceutical-grade medications; and new approaches to problematic methamphetamine use;
- Supporting community-level initiatives, including those led by Indigenous communities, for cannabis public education and awareness or that address use of tobacco or vaping products;
- Improving access to opioid overdose training and naloxone distribution in underserved communities;
- Supporting the Canadian Centre on Substance Use and Addiction (CCSA) in fulfilling their national mandate and advancing research and evidence on cannabis use;
- Supporting research by the Mental Health Commission of Canada (MHCC) to assess the impacts of cannabis use on the mental health of Canadians.
In 2021-22, Health Canada will continue to scale up the most effective and evidence-based of these programs, such as increasing access to a safer supply of prescription opioids in order to better protect people with substance use disorder. The Department will also fund pilot projects designed to provide pharmaceutical-grade medications as safer alternatives to the contaminated illegal drug supply in Canada (referred to by some stakeholders as "safer supply").
Did you know?
Health Canada's SUAP provides grants and contributions funding to other levels of government, community-led and not-for-profit organizations, as well as individuals, to respond to drug and substance use issues across Canada. Funding supports a wide range of initiatives at the community, regional and national levels. Initiatives target a range of psychoactive substances, including opioids, stimulants, cannabis, alcohol, nicotine and tobacco. Over $115 million will be disbursed to selected recipients in 2021-22.
While the effectiveness of using a "safer supply" in treatment programs is well supported by domestic and international studies, there is less evidence available regarding its application from a harm reduction perspective. Health Canada will support an assessment of the pilot projects to capture early findings on effective implementation strategies, lessons learned and issues faced in navigating the establishment of lower-barrier, safer supply pilot projects in the current health care system. This will complement a multi-year evaluation of 5 long-term pilot projects funded by the Canadian Institutes of Health Research (CIHR).
Additionally, Health Canada will strengthen its support of such evidence-based harm reduction measures as access to supervised consumption sites, including improved access to naloxone and drug checking services. In 2021-22, the Department will develop proposed regulations to modernize the framework for supervised consumption sites and services.
The Department will continue to collect data on substance use through drug use surveys that provide information on patterns of use and associated harms related to opioids, alcohol and other substances, such as methamphetamine. Health Canada will also conduct a survey of people with lived and living experience, focusing on street-involved youth aged 17 to 25 years old, and people who attend Supervised Consumption Sites. This data will support the development of strategies to address emerging drug threats.
What's new?
Health Canada launched a new online survey to monitor the demand and use of new psychoactive substances in Canada that will be ongoing to March 2023. The survey will help: identify new substances; improve international reporting; inform risk assessments; inform potential health warning and prevention initiatives; and inform law enforcement about new substances they can expect to meet in clandestine laboratories.
Furthermore, Health Canada will work with partners to identify potential gaps in the regulatory controls for precursors used in the illegal manufacturing of methamphetamine, and to identify areas where additional action can be taken to address problematic methamphetamine use. The Department will also improve its monitoring of the legal domestic drug supply chain by broadening its ability to capture electronic submissions of loss and theft reports for controlled substances, as well as seizure and disposition reports from law enforcement.
Health Canada will also improve its capacity to provide analytical services and intelligence on illegal controlled substances for public health purposes. This includes providing timely and reliable information such as Drug Alerts, Drug Summary Reports and raw data tables to Canadian law enforcement agencies and public health partners. The Department will foster greater collaboration with these partners and agencies in support of harm reduction initiatives such as the analysis of drug checking techniques, and will continue to promote, monitor, verify and enforce compliance with the Controlled Drugs and Substances Act using a risk-based approach to oversee the supply chain and address gaps of potential diversion at licensed dealers and pharmacies.
Health Canada will: continue to collaborate with international partners such as the United Nations Office on Drugs and Crime and Pompidou Group to better understand how stigma affects people who use drugs and related drug policy interventions; promote the use of non-stigmatizing approaches during discussions with other countries at the Commission on Narcotic Drugs; and participate in the development of a World Health Organization Global Action Plan for the implementation of the Global Strategy to Reduce the Harmful Use of Alcohol (2010).
Did you know?
Health Canada established the People with Lived and Living Experience Council to strengthen its engagement with Canadians who have experienced substance use issues. The Council's advice will deepen understanding of this community and help inform the Department's policies and programs.
Regulating cannabis
The purpose of the Cannabis Act is to protect public health and public safety - in particular, the health of young persons by restricting their access to cannabis - while providing adults with legal access to regulated products and reducing illicit activities with cannabis. In 2021-22, Health Canada will focus on the ongoing implementation of the cannabis legislative and regulatory framework. This will include initiating a mandated review of the administration and operation of the Act.
In collaboration with P/Ts, Indigenous communities, the regulated industry, public health organizations, academics, federal partners, international partners, and law enforcement, the Department will continue to support the effective implementation of the Act as well as monitor, collect and analyze scientific evidence and data to better understand its impact. In addition, Health Canada will work closely with CIHR, CCSA and MHCC on cannabis research to inform policy and regulatory decisions and the development of more targeted public education campaigns.
The Department's Science Advisory Committee on Health Products Containing Cannabis will review available evidence regarding the use of cannabis and health products containing cannabis, address specific scientific and clinical questions, and review current and emerging scientific and clinical data. The Committee's advice will be reflected in a final report that will inform future deliberations regarding a potential path forward for these products.
Health Canada will continue to issue licences under the Act, including for: cannabis cultivation and processing, and sale of cannabis for medical purposes; hemp; research and analytical testing; and for the development of pharmaceuticals that include cannabis. The Department also facilitates reasonable access to cannabis for medical purposes by registering individuals who have the support of their health care practitioner to produce a limited amount of cannabis for their own medical purposes.
What's new?
Health Canada will launch a legislative review of the Cannabis Act in 2021-22. Under the provisions of the Act, the Minister must cause a review to be initiated 3 years following its coming into force, and table a report to both Houses of Parliament within 18 months, and no later than April 2023.
As mandated by the Act, the review will examine: the impact of the Act on public health and, in particular, on the health and consumption habits of young persons; the impact of cannabis on Indigenous persons and communities; and the impact of the cultivation of cannabis plants in a home setting.
The Department will also increase collaboration with partners to strengthen the integrity of the medical access framework by addressing the potential public health and public safety risks posed by individuals who may misuse the personal production of cannabis for medical purposes. For example, information will be shared with health care practitioners regarding appropriate dosing of cannabis as well as with P/T regulatory bodies responsible for licencing health care practitioners.
Health Canada will continue to implement strategies aimed at promoting a diverse commercial cannabis industry and deterring the illicit market. This includes reducing barriers for small-scale applicants, improving guidance, and enhancing outreach and intervention strategies with cannabis micro-class applicants.
To support Indigenous communities seeking to participate in the legal cannabis market, the Department will continue to provide advice and guidance to applicants regarding the application process. It will also continue to engage in trilateral/bilateral discussions with Indigenous communities and P/Ts related to public health and safety.
Health Canada will continue to manage the national Cannabis Tracking System, which provides information on cannabis production, inventories, distribution and sales. Tracking the movement of cannabis throughout the supply chain in this manner helps to prevent illegal cannabis from moving into or out of the legal market. The Department will also continue publishing market data on its website to keep Canadians informed of industry trends and progress towards the objective of displacing the illegal market.
Health Canada will continue to promote, monitor, verify and enforce compliance with legislative and regulatory requirements. In 2021-22, compliance promotion activities will proceed on a risk-based approach, with a particular focus on providing guidance to stakeholders regarding Good Production Practices requirements and promotion prohibitions for cannabis, cannabis accessories and services related to cannabis. The Department will also closely monitor the increasingly diverse range of new products entering the market through its mandatory new product notification requirement.
Health Canada will propose regulatory amendments to facilitate non-therapeutic cannabis research involving human participants. The Department will continue to closely monitor the use of vaping products among youth and will take further actions as necessary, such as new regulatory proposals, to reduce the appeal of these cannabis products to young Canadians.
Did you know?
Health Canada is closely monitoring all classes of cannabis products, including edible cannabis and cannabis extracts intended for inhalation (such as vaping products) entering the market to ensure they comply with regulations aimed at minimizing risks of injury to human health. Monitoring of severe adverse reactions, such as vaping-associated lung injury, continues to be a priority for the Department. Information collected in 2021-22 via the new product notification requirement and other sources will be used to support risk assessments, regulatory research, and development of additional guidance and standards.
Chemicals management
As part of its ongoing commitment to chemicals management, Health Canada will conduct further research, monitoring and surveillance (including biomonitoring) and risk assessments regarding chemical substances and human health, and will take appropriate action to help protect the health of Canadians. The Department will also continue to inform the public, including the most vulnerable Canadians, about the potential risks from chemicals and the actions they can take in and around their homes to protect their health through the Healthy Home Campaign.
Improving workplace safety through the safe use of chemicals
To mitigate risks posed by workplace hazardous products, the Department will continue to conduct hazard assessments, enforce the appropriate labelling and communication of hazards and undertake outreach activities. These efforts will be informed and supported by international cooperation, including through implementation of revised editions of the Globally Harmonized System of Classification and Labelling of Chemicals (GHS) and participation in the United Nations Sub-Committee of Experts on the GHS and in the Canada-U.S. Regulatory Cooperation Council.
Health Canada will also continue to develop and modernize policy and operational procedures pertaining to workplace hazardous products under the Hazardous Products Act and the Hazardous Materials Information Review Act and their Regulations. In addition, the Department will continue to implement its Management Response and Action Plan to address the recommendations made in the Evaluation of the Workplace Hazardous Products Program 2014-15 to 2018-19, which identified areas for improvement around compliance and enforcement activities.
What's new?
Health Canada is modernizing how it reviews confidential business information claims under the Hazardous Materials Information Review Act. This includes updating the application process, streamlining the review process, allowing electronic delivery of documents and updating the publication process. This will result in more efficient program delivery and ultimately benefit workers and claimants alike.
Environmental assessment of drugs
In 2021-22, Health Canada intends to continue consulting with the public and stakeholders towards modernizing the Food and Drug Regulations with regard to the environmental risk assessment of active ingredients in drugs. This new regulatory regime would require industry to provide environmental data for new drugs at different stages in the drug development process (e.g., when applying for a Drug Establishment Licence, a clinical trial application, or market authorization). This approach will better align with the U.S. Food and Drug Administration and the European Medicines Agency. Once these new regulations come into force, active ingredients in drugs would no longer fall under the purview of the Canadian Environmental Protection Act, 1999 and the New Substances Notification Regulations (Chemicals and Polymers) and (Organisms).
Supporting the safety of consumer products and cosmetics
The Department will continue to mitigate risks posed by unsafe consumer products and cosmetics by conducting risk assessments, compliance and enforcement, and outreach activities. International cooperation will play an ongoing role in supporting these efforts, for example, through coordinated joint recalls and awareness campaigns.
Health Canada will also continue to develop and update policy and operational procedures pertaining to consumer products under the Canada Consumer Product Safety Act and to cosmetics under the Food and Drugs Act, as well as their associated regulations, to ensure Canada maintains a modern regime.
Did you know?
Canadians are encouraged to report incidents related to consumer products or cosmetics to Health Canada. If a product is determined to be unsafe, the Department will work with industry to implement corrective actions (for example, a product recall). These incident reports also help determine trends regarding product types or emerging technologies that may present a potential hazard. This allows Health Canada to direct resources towards activities that lead to lower risks for all Canadian consumers.
Protecting Canadians from radiation
Health Canada will continue to monitor, advise, and report on exposure to radiation that occurs both naturally and from man-made sources under the authority of the Department of Health Act, the Comprehensive Nuclear-Test-Ban Treaty Implementation Act, the Emergency Management Act and the Nuclear Safety and Control Act and Regulations. As part of the Federal Nuclear Emergency Plan, the Department will participate in nuclear emergency trainings, drills and exercises and will coordinate with federal, provincial and international partners to ensure that emergency preparedness plans are ready for execution in the event of a nuclear emergency.
Additional ongoing activities for 2021-22 include:
- Carrying out research, conducting exposure assessments, and promoting compliance with relevant regulatory requirements to support the assessment and management of risks from devices that emit radiation;
- Exploring the modernization of the Radiation Emitting Devices Act and Regulations, giving consideration to stakeholder input;
- Developing relevant information and science-based advice for Canadians and stakeholders on the safety of these devices, particularly on the topic of health effects from radiofrequency electromagnetic fields and emerging 5G technologies.
Did you know?
November is Radon Action Month, a national initiative to bring together stakeholders and decision-makers to raise awareness of radon across Canada. Radon is an invisible radioactive gas that comes from the ground and is the second leading cause of lung cancer in non-smokers. Through outreach and engagement activities, Health Canada continues to inform Canadians about the health risks posed by radon in indoor air and how to reduce those risks.
Strengthening pesticide regulation
Health Canada will continue to promote, monitor and enforce compliance with the Pest Control Products Act and its Regulations. The Department will also work with Agriculture and Agri-Food Canada and Environment and Climate Change Canada to ensure that timely science-based decisions are made to support the safe and sustainable use of effective pesticide products.
Health Canada has launched a multi-year program renewal project in order to build a stronger and sustainable pesticide regulatory program that strengthens health and environmental protection and leads to improved quality and timeliness of scientific decisions. This proactively addresses the increased workload, increasing complexity of the scientific assessments and availability of key data when undertaking assessments. These efforts are guided by recent consultations toward a new risk-based continuous oversight program delivery model. In 2021-22, the Department will:
- Develop a risk-based framework for continuous oversight of registered pest control product over the course of the products lifecycle by identifying and addressing risk sooner with ongoing risk determination information;
- Develop new and leverage existing processes to improve the timing for the identification, collection and analysis of data, and engagement on the subsequent assessments in order to better define areas of risk, thereby resulting in smaller and more focused reviews;
- Advance and implement new risk management tools to prioritize pest control products for scientific review and risk mitigation.
In order to accelerate its program transformation, Health Canada will implement new technology and solutions to support the renewed program delivery model. These investments will respond to the Minister's mandate commitment to make science-based regulatory decisions in a timely manner.
Finally, the Department will continue to review remaining priority pesticides. Completing these large and complex re-evaluations will be an ongoing priority as new re-evaluations and special reviews are initiated every year.
Departmental Result 5: Canadians make healthy choices
Helping Canadians make healthy choices in their day-to-day lives is an important part of Health Canada's Health Protection and Promotion core responsibility. Over the course of 2021-22, the Department will focus on the following priorities, detailed below: promoting healthy eating; improving food packaging and labelling; fostering international collaboration and cooperation; reducing tobacco use and responding to the increase in youth use of vaping products; as well as supporting Canadians in making informed decisions about cannabis use through public education, research and surveillance.
Promoting Healthy Eating
In 2021-22, Health Canada will continue to advance initiatives under the Healthy Eating Strategy, which aims to curb the rising burden of obesity and chronic disease by making healthier choices easier for all Canadians.
As part of the strategy, the Department will continue to work towards restricting advertising to children of foods that contribute to excess consumption of sodium, sugars and saturated fat. Nearly 1 in 3 Canadian children is overweight or obese, and advertising of foods with nutrients of concern is considered a major contributor to childhood obesity globally. Health Canada maintains an open and ongoing engagement with stakeholders on this issue and, in 2021-22, will implement a monitoring strategy to collect and analyse data in order to report on the state of food advertising to children in Canada.
The Department will continue to promote healthy eating using Canada's Food Guide with a focus on increasing food skills. This includes new web pages and adapting the Canada Food Guide monthly e-newsletter to address topics related to eating healthy during the COVID-19 pandemic. In 2021-22, Health Canada will develop new resources, such as recipes, videos, articles and digital marketing campaigns to support Canadians in their understanding and use of the Food Guide, and will collaborate with stakeholders to further integrate the guide into policy, programs and resources.
The Department will prioritize and direct its policy and promotion efforts to vulnerable populations, including children and youth, young adults, and families who are experiencing or at risk of food insecurity. For example, the Food Guide Friendly Initiative will support actions that organizations take to make it easier for people to follow the Guide, with an initial focus on settings where vulnerable populations eat. These include recreational centres and arenas, colleges and universities, schools, as well organizations that serve them, such as community food centres.
Health Canada plans to publish its revised "Voluntary Sodium Reduction Targets for Processed Foods, 2020-25". These targets were established to address challenges identified by industry stakeholders on the 2012-16 targets and are intended to encourage continued progress regarding the saltiest products in each food category. The Department also plans to consult on new proposed sodium reduction targets for foods sold in restaurants and food services, focusing on those foods Canadians consume most often when eating out. These new targets will address a current gap in the approach to sodium reduction.
Did you know?
To demonstrate leadership and commitment toward the health of its employees, Health Canada is working on improving its own food environment. This includes developing a departmental hospitality policy and vending policy consistent with Canada Food Guide recommendations.
Improving Food packaging and labelling
Health Canada will advance new regulations that introduce a mandatory front-of-package nutrition symbol on prepackaged foods with levels of saturated fat, sugars and/or sodium that meet or exceed specific thresholds. This new symbol will help consumers quickly and easily identify such foods, allowing consumers to make healthier and more informed food choices for themselves and their families.
Health Canada will continue to coordinate with the Canadian Food Inspection Agency (CFIA) to establish a standardized cycle for implementing regulations that require a change to food labels, in order to provide greater predictability for the food industry.
Fostering international collaboration and coordination
Health Canada is an active participant in developing science-based international standards and risk management standards for food safety and nutrition and will continue to co-lead Canada's participation at the Codex Alimentarius Commission with the CFIA. These engagements allow Canada to influence the development of standards that are consistent with the country's needs and objectives while achieving global public health objectives regarding food safety and nutrition. In the coming fiscal year, the Department will plan and host the 46th session of the Codex Committee on Food Labelling.
Reducing tobacco use and responding to the increase in youth use of vaping products
In 2021-22, Health Canada will take additional action to address the rapid rise in youth vaping, and advance implementation of Canada's Tobacco Strategy - a modernized approach for successful tobacco control with the goal of achieving the target of less than 5% tobacco use by 2035. The Department will undertake the first legislative review of the Tobacco and Vaping Products Act, scheduled to begin no later than May 2021 and culminating with a report to Parliament in May 2022.
In cooperation with other F/P/T partners and key stakeholders, new or ongoing measures relating to youth vaping will include:
- Finalizing the proposed Concentration of Nicotine in Vaping Products Regulations that were pre-published in Canada Gazette Part I in December 2020, and proposed regulations that would restrict flavours in vaping products, to help further reduce the appeal of these products to young Canadians;
- Raising awareness and public education of the potential harms associated with vaping, particularly among youth and their parents, through public education campaigns;
- Research to understand vaping product composition and emissions, to conduct toxicity testing, and surveillance to monitor youth vaping trends;
- Additional qualitative research and surveillance activities, including public opinion research to better understand the increase in youth vaping to inform future regulations;
- Online inspections of websites where advertising or promotion of vaping products is accessible to youth; conducting inspections of vaping products and activities, as well as product sampling and testing; and publishing the results of key compliance and enforcement activities.
What's new?
To address the rapid rise in youth vaping, Health Canada will take further action to reduce the appeal of and access to vaping products among young Canadians - including new regulatory proposals, public education, and compliance and enforcement actions. The Department will continue to closely monitor and study the use of tobacco and vaping products among youth and take additional actions as necessary.
In 2021-22, new or ongoing measures relating to tobacco will include:
- Enforcing plain packaging requirements on all tobacco products to reduce their appeal;
- Developing new health warnings, health information messages and statements about toxicity as part of renewed tobacco product labelling requirements;
- Promoting compliance, inspecting tobacco products, and other activities related to new requirements;
- Renewing the Pan-Canadian Quitline Initiative and developing voluntary smoking cessation standards for health care organizations, in coordination with P/Ts and other stakeholders;
- Supporting community-level initiatives that address tobacco or vaping products through SUAP. A total of $3.5 million is planned in grants ($100,000) and contribution agreements ($3.4 million).
Supporting Canadians in making informed decisions about cannabis use through public education, research and surveillance
As the legal cannabis industry increasingly displaces the illegal market and adults are provided with greater access to regulated cannabis products, Health Canada will closely monitor changes in knowledge, attitudes and behaviors through population-based surveys (e.g., the Canadian Cannabis Survey) and adapt its public education and awareness activities to ensure that Canadians are able to make informed decisions to protect their health. The Department will inform the public of any health risks.
Furthermore, Health Canada will provide ongoing support to community-based and Indigenous organizations in educating their communities on the health effects of cannabis use through SUAP. A total of $13.4 million is planned in grants ($100,000) and contribution agreements ($13.3 million).
The Department recognizes that ongoing research is fundamental to understanding the benefits and harms of cannabis. The new fiscal year will see Health Canada providing $5.6 million to the CCSA and the MHCC to continue their work supporting research on cannabis use and its impact on mental health.
The Department will also undertake regulatory research to address information gaps, such as the potential health hazards associated with inhalation of cannabis-derived vaping aerosols. The evidence gathered will inform future regulatory and compliance efforts, risk assessment and public education and awareness.
Key Risks for Core Responsibility 2: Health Protection and Promotion
1. Risk: Canadians may lose confidence in Health Canada's ability to help protect their health if the Department is not regarded as a trusted regulator and used as a credible source of information.
Key examples of Health Canada's planned risk responses:
- Implement informative initiatives
-
Expand the amount of regulatory health and safety information made available to Canadians in a simple and accessible way through innovative initiatives and communication activities. For example:
- Publish information on authorized clinical trials for drugs and vaccines for COVID-19, as well as information on health products authorized under Interim Orders for COVID-19 use.
- Publish reports regarding industry non-compliance on the Health Canada website outlining the summary results of vaping compliance and enforcement activities conducted under the Tobacco and Vaping Products Act and/or the Canada Consumer Product Safety Act.
- Offer more engagement opportunities to Canadians and stakeholders
-
In line with the Government of Canada's Open Government initiative and Health Canada's Forward Regulatory Plan, Canadians and stakeholders have greater opportunity to be involved in decision-making processes, including the regulatory process throughout its development. For example:
- Consult broadly with stakeholders, including patient groups, health partners, and industry on continuing initiatives, such as making use of foreign decisions to support access to products otherwise not available in Canada as part of improving the regulatory review of drugs and devices.
- In the context of the COVID-19 outbreak, engage with industry, federal partners, P/Ts, and other stakeholders to better support them in a time of crisis.
- Improve communication tools
-
Continue to acquire, develop and improve the tools, processes and resources needed to effectively communicate to, and engage, Canadians on Health Canada's digital platforms, including Canada.ca and Health Canada social media channels. For example:
- Improve and expand tools such as the interactive map to show the locations of opioid-related activities taking place in communities across the country.
- Proactively release valuable COVID-19-related data and information to inform the public while employing digital tools to improve the reach of and dissemination of information.
- Use digital performance measurement tools, surveys and analysis to support continuous improvement, to encourage user-focused digital content that is findable and accessible to support increased trust of government web and social media content.
- Collaborate with federal partners to innovate, leverage and/or optimize content and digital tools for Canadians and streamline and automate the publishing of data to Canada.ca allowing internal stakeholders to manage publishing and update in real time.
- Implement a new, user-focused recalls and safety alerts system that will improve the ability of Canadians to take action to protect themselves from unsafe products.
2. Risk: Health Canada's ability to help protect the health of Canadians may be weakened due to the increasing complexity of the global supply chain and the rapid pace of innovation.
Key examples of Health Canada's planned risk responses:
- Strengthen oversight
-
Develop strategies and tools to further strengthen market surveillance and oversight of emerging products. For example:
- Monitor compliance with the regulations for the manufacture, sale, labeling and promotion of vaping products.
- Promote, verify and enforce compliance with the Cannabis Act and its regulations consistent with the approach outlined in Health Canada's Compliance and Enforcement Policy for the Cannabis Act.
- Collaborate internationally
-
Collaborate with international regulatory organizations, and align where appropriate with foreign regulators. For example:
- Working with international partners on COVID-19 issues, including clinical trials, product market authorizations, risk assessments and product shortages.
- Communicating joint recalls of products and sharing information regarding non-compliant health product inspections, where appropriate.
- Increase use of regulatory and non-regulatory activities
-
Increase the use of regulatory and non-regulatory activities that address changing business models in the supply chain, specifically for foreign sites. For example:
- Share information with other trusted regulators regarding non-compliance.
- Carry out foreign on-site Good Manufacturing Practices inspections, to increase Health Canada's oversight of drug production earlier in the supply chain.
- Exchange information on inspections outcomes for pharmaceutical products between Health Canada and international partners conducted in foreign jurisdictions.
Planned results for Core Responsibility 2: Health Protection and Promotion
| Departmental Result Indicators | Target | Date to achieve target | Actual Results |
|---|---|---|---|
|
Percentage of new drug decisions issued within service standardsResult 3: Tablenote 1 (Baseline: 88% in 2017-18) |
At least 93% | March 31, 2022 |
2017-18: 88% |
|
Percentage of Risk Management Plan reviews for new drug decisions completed within service standardsResult 3: Tablenote 2 (Baseline: 91% in 2017-18) |
At least 90% | March 31, 2022 |
2017-18: 91% |
| Percentage of drug companies deemed to be compliant with manufacturing requirements under the Food and Drugs Act and associated regulations | Between 85% and 95%Result 3: Tablenote 3 | March 31, 2022 |
2017-18: 96% |
|
Tablenotes
|
|||
| Departmental Result Indicators | Target | Date to achieve target | Actual Results |
|---|---|---|---|
|
Percentage of domestic consumer product recalls communicated to Canadians in a timely manner (Baseline: 86% in 2016-17) |
At least 90%Result 4: Tablenote 1 | March 31, 2022 |
2017-18: 87% |
|
Percentage of actions taken in a timely manner to protect the health of Canadians from substances found to be a risk to human health (Baseline: 85% in 2016-17) |
Exactly 100%Result 4: Tablenote 2 | March 31, 2022 |
2017-18: 86%Result 4: Tablenote 3 |
|
Percentage of pre-market submission reviews that are completed within service standardsResult 4: Tablenote 4 (Baseline: 95% in 2019-20) |
At least 90% | March 31, 2022 |
2017-18: 92% |
|
Tablenotes
|
|||
| Departmental Result Indicators | Target | Date to achieve target | Actual Results |
|---|---|---|---|
|
Percentage of Canadians (aged 15+) who are current cigarette smokersResult 5: Tablenote 1 (Baseline: 16% in 2017) |
At most 5% | March 31, 2035 |
2017-18: 18% |
|
Percentage of Canadians (aged 15-24) who have used cannabis in the last 12 months (Baseline: 26.9% in 2018-19 [14.2% of Canadians aged 15-17 and 31.4% of Canadians aged 18-24]) |
At most 29%Result 5: Tablenote 2 | March 31, 2025 |
2017-18: 25.5%Result 5: Tablenote 3 |
|
Percentage of Canadians who use dietary guidance provided by Health Canada (Baseline: 41% in 2012) |
At least 50% | March 31, 2022 |
2017-18: 47%Result 5: Tablenote 5 |
|
Tablenotes
|
|||
| 2021-22 budgetary spending (as indicated in Main Estimates) |
2021-22 Planned spending |
2022-23 Planned spending |
2023-24 Planned spending |
|---|---|---|---|
| 1,104,086,037 | 1,104,086,037 | 553,041,838 | 532,743,413 |
| Note: The decrease in planned spending is mainly due to the expiry of budgetary authorities in 2021-22 related to the Creation of a Critical Drug Reserve in collaboration with provinces and territories to support access to drugs which treat COVID-19 symptoms for Canadians; the Federal Framework for the Legalization and Regulation of Cannabis; Funding for Substance Use in the context of COVID-19; as well as Funding to support regulatory and operational critical COVID-19 focused functions. The Department would have to request funding for these initiatives for future years. | |||
| 2021-22 Planned full-time equivalents |
2022-23 Planned full-time equivalents |
2023-24 Planned full-time equivalents |
|---|---|---|
| 5,933 | 4,962 | 4,898 |
| Note: The decrease in planned FTEs is mainly due to the expiry of budgetary authorities in 2021-22 for the Federal Framework to Legalize and Regulate Cannabis; as well as Funding to support regulatory and operational critical COVID-19 focused functions. The Department would have to request funding for these initiatives for future years. | ||
Financial, human resources and performance information for Health Canada's Program Inventory is available in the GC InfoBase.
Internal Services
Internal Services Description
Internal Services are those groups of related activities and resources that the federal government considers to be services in support of Programs and/or required to meet corporate obligations of an organization. Internal Services refers to the activities and resources of the 10 distinct services that support Program delivery in the organization, regardless of the Internal Services delivery model in a department.
The 10 service categories are: Management and Oversight Services; Communications Services; Legal Services; Human Resources Management Services; Financial Management Services; Information Management Services; Information Technology Services; Real Property Management Services; Materiel Management Services; and Acquisition Management Services.
Internal Services Plans
Health Canada's greatest strength is an engaged, empowered and well-equipped workforce with employees that have the competencies (including science and regulatory skill sets), tools and opportunities to succeed in the pursuit of excellence in program and service delivery.
The Clerk of the Privy Council noted in his letter to the Prime Minister in the Twenty-Seventh Annual Report on the Public Service of Canada, that during these uncertain times, certainty is found in the Public Service's commitment to draw on all of its creativity and resourcefulness to implement Government plans aimed at protecting and supporting Canadians. According to the Clerk, our collective hard work on diversity and inclusion, mental health, and harassment - as well as our experimentation with new ideas - has given momentum to renewal of the public service. Health Canada supports the government-wide goals of Public Service Renewal, through initiatives that foster a more inclusive, agile and equipped workforce.
The Department is committed to ensuring that all employees feel safe and are treated with respect, dignity and fairness in the workplace, including instilling positive values and tackling racial bias, harassment and discrimination. Health Canada is committed to eliminating barriers to success and career progression through more inclusive recruitment practices.
Did you know?
In 2020, Health Canada was recognized as one of Canada's Best Diversity Employers (for the 6th year running), one of Canada's Top Employers of Young People (for the 10th year running), and in 2021, for the first time, as one of Canada's Top 100 Employers.
In 2021-22, the Department will focus on the following priorities:
Building a healthy, diverse and inclusive workforce
Health Canada is committed to ensuring a workplace that is free of racism and discrimination, and where all employees feel safe and are treated with respect, dignity and fairness. These values are the foundation of who we are, what we do, and how we do our work. To foster a workplace free of racism and discrimination, the Department will:
- Continue to implement its Multi-Year Diversity and Employment Equity Plan;
- Complete an employment equity review of internal policies, procedures and practices relevant to employment systems;
- Leverage the newly created Leadership Council on Diversity and Inclusion and Employee Networks to combat racism and discrimination;
- Develop an action plan to address findings from internal anti-racism listening sessions and identify concrete actions in creating a more diverse and inclusive workplace, free from racism and discrimination;
- Create a safe space to support Black, Indigenous, People of Colour, Persons with Disabilities and LGTBQ2+ employees facing racism or experiencing discrimination in the workplace within the independent Centre for Ombudsman, Resolution and Ethics.
What's new?
In 2020-21, Health Canada established a Leadership Council on Diversity and Inclusion, co-chaired by the Deputy Ministers, comprising of volunteers from various employment equity groups, disciplines and regions across Canada. It provides a forum for employees to discuss, validate, promote and enable departmental strategies, policies, and activities that strengthen an organizational culture of diversity and inclusion.
Additionally, in order to attract and retain a diverse, inclusive, and bilingual workforce within a healthy, accessible and high performing workplace, Health Canada will:
- Develop an Accessibility Plan to ensure that the requirements of the Accessible Canada Act and the Public Service Accessibility Strategy are met;
- Continue to implement mental health and wellness strategies;
- Modernize recruitment, classification, and staffing practices in support of greater efficiency;
- Facilitate tools for the virtual work environment, such as continuing the virtual onboarding program for new employees and adapting Official Languages training for remote learning;
- Ensure compliance with Bill C-65, creating a healthy, safe, harassment free, and violence free work environment for all employees.
The Department will continue to build a culture of resolution anchored in values and ethics where employees can raise concerns and seek the help they need through informal approaches without fear of speaking up. The Centre for Ombudsman, Resolution and Ethics will support both individual and organizational wellbeing by promoting a collaborative approach to managing conflict. Most importantly, Health Canada will continue to promote the benefits of a values-driven organization at all levels, with the emphasis of evidence-based decisions guided by ethics, fairness, inclusiveness and conflict competence.
Modernizing the workplace to enable a safe and productive workforce with access to modern tools and facilities
Health Canada's plan for 2021-22 includes modernizing the workplace to enable a safe and productive workforce with access to up-to-date tools and facilities. The Department will improve workplace safety and productivity and modernize the delivery of its financial management services through such initiatives as:
- Implementing the National Accommodation Strategy, which ensures modernization of office facilities to meet specific program requirements, providing an innovative, flexible and modern working environment in accordance with health and safety guidelines;
- Upgrading and improving its infrastructure and tools to stabilize and support digital modernization of the applications and systems that deliver programs to Canadians;
- Continuing to participate in Labs Canada to support modern and collaborate laboratories;
- Completing a financial systems strategy and roadmap in order to migrate to an updated departmental financial and material system (the SAP S/4HANA version) by 2025.
Communication Services
In 2021-22, Health Canada will continue to provide Canadians with the timely and relevant information they need to take action on their personal and collective health and safety. This will be accomplished through an array of digital and traditional communication methods. For example, the Department will continue to provide communications in support of the Government's response to the COVID-19 pandemic, while also communicating on other important issues, such as improving health systems, virtual care, mental health, and access to prescription medications.
Health Canada will continue to deliver evidence-based and innovative public awareness campaigns and to work with P/Ts and other stakeholders to inform Canadians about priority topics such as opioids and other controlled substances, tobacco, vaping, cannabis, healthy eating, food and product safety, and environmental health.
| 2021-22 Budgetary spending (as indicated in Main Estimates) |
2021-22 Planned spending |
2022-23 Planned spending |
2023-24 Planned spending |
|---|---|---|---|
| 301,904,724 | 301,904,724 | 267,393,165 | 272,186,639 |
| Note: The decrease in planned spending is mainly due to the expiry of budgetary spending authorities in 2021-22 for Funding to support regulatory and operational critical COVID-19 focused functions; as well as the Federal Framework to Legalize and Regulate Cannabis. The Department would have to request funding for these initiatives for future years. | |||
| 2021-22 Planned full-time equivalents |
2022-23 Planned full-time equivalents |
2023-24 Planned full-time equivalents |
|---|---|---|
| 1,804 | 1,636 | 1,658 |
| Note: The decrease in planned FTEs is mainly due to the expiry of budgetary spending authorities in 2021-22 for Funding to support regulatory and operational critical COVID-19 focused functions; as well as the Federal Framework to Legalize and Regulate Cannabis. The Department would have to request funding for these initiatives for future years. | ||
Experimentation and GBA+
Experimentation
Innovation and experimentation are more important than ever to help drive Health Canada's response to the COVID-19 pandemic and continue to meet its mandate to protect the health and safety of Canadians.
Building on its Beyond2020 commitments, the Department continues to increase employee capacity for experimentation and innovation via its Innovation and Experimentation Policy Framework 2020. Highlights of initiatives planned for 2021-22 include:
- Learn and Act - Bi-monthly webinars will bring private and public service experts together with Health Canada employees to learn how to apply innovation and experimentation to their work.
- Explore and Test - New and ongoing investments in employee-led innovation and experimentation through the Department's Solutions Fund will continue to improve operations and functionality in order to deliver better services and greater value to Canadians.
- Measure and Share - Working with Statistics Canada's online platform for sharing results of innovation and experimentation projects (the Radar Innovation Database), Health Canada will connect teams and share the outcomes of its projects and lessons learned.
Since the launch of the Solutions Fund in 2018, the Department has funded 11 projects. In late 2020, it announced two consecutive calls for proposals under the Fund seeking projects that respond to COVID-19, as well as policy, program and workplace improvements. Health Canada will continue to invest in experimentation projects in 2021-22, such as:
- Project Cipher: Experimenting with the use of machine learning to improve inspection processes. In 2021-22, the project will be testing the application of AI and machine learning to process and draw actionable information from unstructured data contained within inspection reports.
- Project Hummingbird: continue exploring the use of drone technology with advanced satellite imagery to support outdoor cannabis inspections. In 2021-22, the Department will be conducting experiments on proof of concept.
- PRODigy: Streamlining the process of submitting incident reports to Health Canada related to consumer products and cosmetics, and supporting follow-up action by the Department. The project's second phase focuses on modifying the report into a more user-friendly format, in an effort to increase submission rates. The project will conclude in early 2021-22 once the improvements are fully implemented.
What's new?
While exploring drone technology to conduct virtual cannabis inspections, Health Canada discovered that advanced satellite imagery could also be a useful tool. The Department is expanding its initial experiments combining the use of drone technology with advanced satellite imagery with the goal of providing inspectors with new information on crop inventory, crop anomalies, perimeter requirements, and safety concerns to support on-site assessments.
Other examples of experimentation and innovation initiatives that will begin or continue in 2021-22 include:
- Under the Innovative Solutions Canada program, Health Canada will collaborate with the National Research Council and Environment and Climate Change Canada to support two projects related to Canada's response to COVID-19. The projects address the feasibility of compostable disposable surgical masks and respirators, as well as recycling technologies for disposable (single-use) PPE, used in the Canadian healthcare sector.
- Virtual Inspections of Cannabis Licence Holders (Project Eagle Eye) - Supported by the Greening Government Fund Initiative and aligned with Project Hummingbird (described above), this project explores how to replace some physical inspections that are a significant source of carbon emissions with virtual equivalents. If successful, the cannabis program's carbon footprint could be reduced while still maintaining (and perhaps augmenting) regulatory oversight. There is also significant potential for broader application within Health Canada and beyond.
- Health Canada's ongoing Micro-class Engagement Strategy aims to promote a diverse cannabis industry and deter activity in the illicit market. The strategy focuses on reducing barriers for small-scale applicants, improving guidance, and enhancing outreach and intervention strategies with the micro community.
- The Department's Pest Management Regulatory Agency will test and validate new processes related to the implementation of an integrated pesticide program, focusing on workload prioritization and management, continuous oversight and improved post-market evaluations.
- Health Canada's Microbiology Laboratory is developing a novel method for the rapid identification of visible impurities by FTIR spectroscopy that will allow for the quick and accurate compliance verification of cannabis, health products and food samples with high discriminatory power and almost no delay. This will eventually replace the current multi-step analysis and support prompt investigations by the Department's regulatory inspectors.
Sex and Gender-Based Analysis Plus (SGBA+/GBA+)
Health Canada renewed its Sex and Gender Action PlanFootnote 3 for another 3 years starting in 2020-21. The Plan provides a framework to strengthen the integration of sex, gender and other intersectional factors (such as age, race and income level) in externally and internally facing work of the Department. As Health Canada continues to advance equality and improve its approaches to diversity and inclusion, priorities will be guided by the results of a formal evaluation and by lessons learned from 2017 to 2020, including those derived over the course of the COVID-19 pandemic.
Priorities currently identified under the renewed Action Plan are to:
- Increase departmental capacity to apply SGBA+;
- Collaborate with internal and external partners to strengthen the Department's sex, gender and diversity-related evidence base and expertise;
- Strengthen accountability and transparency in implementing SGBA+.
In collaboration with CIHR, Health Canada established research-policy partnerships that support researchers who have both subject matter and SGBA+ expertise, to engage with departmental staff on priority Health Canada initiatives. In 2021-22, key initiatives in support of the renewed SGBA+ Action Plan include:
Improving capacity building
The Department will continue to provide employee training in several sex and gender related areas: increasing awareness of how SGBA+ could and should influence the development of policies and guidelines; the application of SGBA+ to project management and risk communication; and the integration of both SGBA+ and Indigenous lenses in the development and delivery of programs.
Health Canada will continue to include gender-neutral washrooms in the modernization of its workplace and will be diligent in upholding accessible design standards whenever possible.
Health Canada's Employee Assistance Program (EAP) provides services to employees in many federal departments and agencies, as well as to members of the Royal Canadian Mounted Police, members of the Canadian Armed Forces, and veterans of these organizations. This Program applies an SGBA+ lens to its policies, procedures and services. Based on findings from research conducted in 2018-19 and progress made on key deliverables in the past year, the following will serve as a focus for 2021-22:
- Optimizing outreach to those who tend to underuse EAP (such as males, LGBTQ2+ persons, Indigenous Persons, and victims of intimate partner violence) and/or who might be experiencing increased mental health impacts because of COVID-19 (e.g., women, Indigenous persons, LGBTQ2+ persons);
- Continuing to expand the use of resources and technologies (primarily video counselling and real-time chat) to enhance outreach to groups who access services at lower rates;
- Improving the capacity to appropriately match clients with diverse backgrounds or those belonging to LGBTQ2+ groups, with a counsellor who has "lived experience" or other relevant expertise.
Applying sex and gender-related evidence and expertise
Health product labelling: Health Canada will advance the policy-research partnership with McGill University to investigate consumer perceptions and behaviours in relation to health product labelling for cosmetics, natural health products and non-prescription drugs. In 2021-22, McGill researchers will host training sessions with the Department on applying SGBA+ to biomedical research as well as increasing the integration of sex and gender related concepts in future public policies.
Medical devices and drugs regulations: In partnership with CIHR's Institute of Gender and Health, Health Canada has funded two initiatives, to be implemented in 2021-22, to identify how the Department currently accounts for diversity, sex, and gender while regulating drugs and medical devices and recommend how to further integrate these concepts.
Cannabis: Health Canada will continue to include questions on sex, gender, age and other variables in its Canadian Cannabis Survey. The 2021 Survey will collect information on sex, gender, age, sexual orientation, country of birth and ethnicity, to better understand the interactions between cannabis use and priority populations. This information will help to inform public education and awareness activities in 2021-22.
Healthy Eating Policies: The Department considers diversity, sex and gender early on when developing healthy eating policies to help identify those who may be most affected. For example, Health Canada will continue to consult and conduct research with those who face challenges accessing, understanding and evaluating nutrition labelling information when making food choices. This information will inform labelling policies and related regulations.
Implementing SGBA+ across Health Canada programs
Canada's Food Guide: The Department will continue to develop targeted healthy eating resources and work with stakeholders to reach Canadians across various settings, ages, and population groups. For example, it will create a new, modern, user-centric food guide recipe gallery that will include new culturally-diverse recipes and articles. In addition, Health Canada will work with diverse groups of youth and young adults to inform the development of new resources and tools as well as to promote peer-to-peer engagement on healthy eating.
Chemicals Management: In 2021-22, the Department will strengthen application of SGBA+ considerations within its chemicals management activities; for example, developing training materials tailored to scientists, as well as tools to enhance SGBA+ considerations more systematically. In addition, The Pest Control Products Act requires that the needs of 'at risk' or vulnerable Canadians - including pregnant women, infants, children and seniors - be considered when regulating pesticides. Diversity, sex and gender will play an increasingly important role in these considerations.
What's new?
As Health Canada develops materials to communicate risks with regard to chemicals, messages will be increasingly customized for specific populations to address their interests and needs. For example, in 2021-22, a social marketing campaign related to chemicals management will be tailored to pregnant women, parents of young children, youth, new Canadians, and other at-risk populations.
Spending and human resources
This section provides an overview of the department's planned spending and human resources for the next three consecutive fiscal years, and compares planned spending for the upcoming year with the current and previous years' actual spending.
Planned spending
The following graph presents planned (voted and statutory) spending over time.
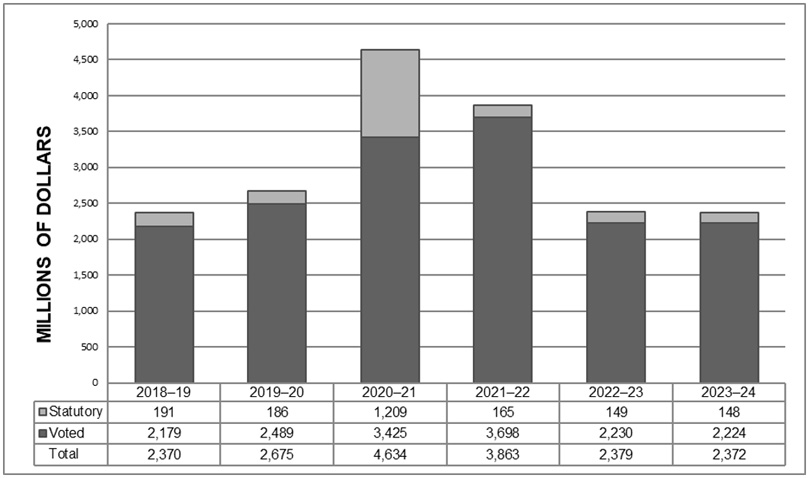
Departmental spending 2018-19 to 2023-24: Text description
The figure illustrates Health Canada's spending trend from fiscal year 2018-19 to fiscal year 2023-24 where spending, in millions of dollars, is shown on the vertical axis and time period, in fiscal years, is shown on the horizontal axis.
Health Canada's actual spending for fiscal year 2018-19: $2,370 million (Voted: $2,179 million, Statutory: $191 million); 2019-20: $2,675 million (Voted: $2,489 million, Statutory: $186 million).
Health Canada's forecast spending for fiscal year 2020-21: $4,634 million (Voted: $3,425 million, Statutory: $1,209 million).
Health Canada's planned spending for fiscal year 2021-22: $3,863 million (Voted: $3,698 million, Statutory: $165 million); 2022-23: $2,379 million (Voted: $2,230 million, Statutory: $149 million); 2023-24: $2,372 million (Voted: $2,224 million, Statutory: $148 million).
Budgetary planning summary for Core Responsibilities and Internal Services (dollars)
The following table shows actual, forecast and planned spending for each of Health Canada's core responsibilities and to Internal Services for the years relevant to the current planning year.
| Core Responsibilities and Internal Services | 2018-19 expenditures |
2019-20 expenditures |
2020-21 forecast spending |
2021-22 budgetary spending (as indicated in Main Estimates) |
2021-22 planned spending |
2022-23 planned spending |
2023-24 planned spending |
|---|---|---|---|---|---|---|---|
| Core Responsibility 1: Health Care Systems | 1,289,851,245 | 1,601,069,150 | 2,783,957,458 | 2,456,807,897 | 2,456,807,897 | 1,558,123,313 | 1,566,896,000 |
| Core Responsibility 2: Health Protection and Promotion | 726,841,710 | 728,899,756 | 1,315,119,997 | 1,104,086,037 | 1,104,086,037 | 553,041,838 | 532,743,413 |
| Subtotal | 2,016,692,955 | 2,329,968,906 | 4,099,077,455 | 3,560,893,934 | 3,560,893,934 | 2,111,165,151 | 2,099,639,413 |
| Internal Services | 353,056,297 | 345,420,163 | 535,013,976 | 301,904,724 | 301,904,724 | 267,393,165 | 272,186,639 |
| Total | 2,369,749,252 | 2,675,389,069 | 4,634,091,431 | 3,862,798,658 | 3,862,798,658 | 2,378,558,316 | 2,371,826,052 |
|
Note: The 2018-19 to 2020-21 fiscal years total expenditures and forecast spending include all Parliamentary appropriation sources: Main Estimates, Supplementary Estimates, and funding from various Treasury Board votes. For the 2021-22 to 2023-24 fiscal years, total planned spending does not include funding through Supplementary Estimates and carry forward adjustments. The planned spending increases in 2020-21 are mainly due to Health Canada's response to the COVID-19 pandemic; mainly for the Creation of a Critical Drug Reserve in collaboration with provinces and territories to support access to drugs which treat COVID-19 symptoms for Canadians; Long Term Care and other Supportive Care settings, the Safe Restart Agreement for the federal investments in testing, contact tracing and data management; Drugs, medical devices and virtual care; as well as Substance Use in the context of COVID-19. The planned spending decreases in 2021-22 are mainly due to funding level decreases related to the Creation of a Critical Drug Reserve in collaboration with provinces and territories to support access to drugs which treat COVID-19 symptoms for Canadians; as well as the as, the expiry of budgetary authorities in 2020-21 for Safe Restart Agreement for the federal investments in testing, contact tracing and data management; Chemical Management Plan and, virtual care. The planned spending decreases in 2022-23 are mainly due to funding level decreases for Long Term Care and other Supportive Care settings in the context of COVID-19; Strengthening Canada's Home and Community Care and Mental Health and Addiction Services Initiative; as well as the expiry of budgetary authorities in 2021-22 for the following: Creation of a Critical Drug Reserve in collaboration with provinces and territories to support access to drugs which treat COVID-19 symptoms for Canadians; Federal Framework for Legalization and Regulation of Cannabis; Canada Health Infoway; and, Substance use in the context of Covid-19. The Department will have to request funding for these initiatives for future years. |
|||||||
Planned human resources
The following table shows actual, forecast and planned full-time equivalents (FTEs) for each core responsibility in Health Canada's departmental results framework and to Internal Services for the years relevant to the current planning year.
| Core Responsibilities and Internal Services | 2018-19 actual FTEs |
2019-20 actual FTEs |
2020-21 forecast FTEs |
2021-22 planned FTEs |
2022-23 planned FTEs |
2023-24 planned FTEs |
|---|---|---|---|---|---|---|
| Core Responsibility 1: Health Care Systems | 210 | 215 | 306 | 276 | 272 | 266 |
| Core Responsibility 2: Health Protection and Promotion | 5,193 | 5,785 | 6,075 | 5,933 | 4,962 | 4,898 |
| Subtotal | 5,403 | 6,000 | 6,381 | 6,209 | 5,234 | 5,164 |
| Internal Services | 2,268 | 2,164 | 2,018 | 1,804 | 1,636 | 1,658 |
| Total | 7,671 | 8,164 | 8,399 | 8,013 | 6,870 | 6,822 |
|
Note: The 2018-19 and 2019-20 fiscal years full-time equivalents (FTEs) are based on actual expenditures on personnel. The 2020-21 fiscal year is based on total authorities from all Parliamentary appropriation sources: Main Estimates and Supplementary Estimates. For the 2021-22 to 2023-24 fiscal years, total FTEs do not include FTEs funded through Supplementary Estimates and carry forward adjustments. The calculation of the planned FTE figures is based on programs using their full revenue authority. The increase in actual FTEs in 2019-20 is mainly due to the Federal Framework to Legalize and Regulate Cannabis, and improving the accessibility, affordability and appropriate use of prescription drugs and medical device. The increase in forecasted FTEs in 2020-21 is mainly due to funding for the Safe Restart Agreement for the federal investments in testing, contact tracing and data management; the Creation of a Critical Drug Reserve in collaboration with provinces and territories to support access to drugs which treat COVID-19 symptoms for Canadians; as well as Funding to support regulatory and operational critical COVID-19 focused functions. The decrease in 2021-22 is mainly due to funding level decreases related to the Creation of a Critical Drug Reserve in collaboration with provinces and territories to support access to drugs which treat COVID-19 symptoms for Canadians; as well as the as, the expiry of budgetary authorities in 2020-21 for Safe Restart Agreement for the federal investments in testing, contact tracing and data management; and Chemical Management Plan. The decrease in planned FTEs in 2022-23 is mainly due to the expiry of budgetary authorities in 2021-22 for the Federal Framework to Legalize and Regulate Cannabis; as well as Funding to support regulatory and operational critical COVID-19 focused functions. The Department would have to request funding for these initiatives for future years. |
||||||
Estimates by vote
Information on the Health Canada's organizational appropriations is available in the 2021-22 Main Estimates.Endnote ii
Future-oriented condensed statement of operations
The future-oriented condensed statement of operations provides an overview of Health Canada's operations for 2020–21 to 2021–22.
The amounts for forecast and planned results in this statement of operations were prepared on an accrual basis. The amounts for forecast and planned spending presented in other sections of the Departmental Plan were prepared on an expenditure basis. Amounts may therefore differ.
A more detailed future-oriented statement of operations and associated notes, including a reconciliation of the net cost of operations to the requested authorities, are available on Health Canada's website.
| Financial information | 2020-21 forecast results |
2021-22 planned results |
Difference (2021-22 planned results minus 2020-21 forecast results) |
|---|---|---|---|
| Total expenses | 4,318,882,666 | 4,201,583,642 | (117,299,024) |
| Total revenues | 220,686,473 | 222,924,854 | 2,238,381 |
| Net cost of operations before government funding and transfers | 4,098,196,193 | 3,978,658,788 | (119,537,405) |
Health Canada is projecting $4,201.6 million in expenses based on 2021-22 Main Estimates and accrual information. This amount does not include future supplementary estimates. It represents a decrease of $117.3 million from 2020-21 forecast results.
The 2021-22 planned expenses by core responsibility are as follows:
- Health Care Systems $2,453.8 million;
- Health Protection and Promotion $1,390.3 million; and,
- Internal Services $357.8 million.
Health Canada receives most of its funding through annual Parliamentary appropriations. Health Canada's revenue is generated by programs that support the above-noted core responsibilities. Health Canada projects total revenues in 2021-22 to be $222.9 million, representing a modest increase of $2.2 million from 2020-21 projections.
The 2021-22 main sources of revenues by type are as follows:
- Services of a regulatory nature $76.8 million;
- Rights and privileges $120.6 million; and,
- Services of a non-regulatory nature $91.3 million.
Corporate information
Organizational profile
Appropriate Minister: The Honourable Patty Hajdu, P.C., M.P.
Institutional Head: Dr. Stephen Lucas
Ministerial portfolio: Health
Enabling instrument[s]: Assisted Human Reproduction ActEndnote iii, Canada Health ActEndnote iv, Canada Consumer Product Safety ActEndnote v, Cannabis ActEndnote vi, Controlled Drugs and Substances ActEndnote vii, Department of Health ActEndnote viii, Food and Drugs ActEndnote ix, Hazardous Materials Information Review ActEndnote x, Hazardous Products ActEndnote xi, Pest Control Products ActEndnote xii, Radiation Emitting Devices ActEndnote xiii, Tobacco and Vaping Products ActEndnote xiv.
List of Acts and RegulationsEndnote xv
Year of incorporation/commencement: 1913
Raison d'être, mandate and role
Raison d'être, mandate and role: who we are and what we do is available on the Health Canada website.
For more information on the department's organizational mandate letter commitments, see the "Minister's mandate letter".
Operating context
Information on the operating context is available on the Health Canada website.
Reporting framework

Health Canada's approved Departmental Results Framework and Program Inventory for 2021-22 are as follows: Text description
The figure illustrates Health Canada's approved Departmental Results Framework and Program Inventory for 2021-22.
The Departmental results Framework groups Health Canada's Core Responsibilities into two categories, all supported by Internal Services. These categories are (1) Health Care Systems and (2) Health Protection and Promotion, each of which are delivered through multiple programs in the Program Inventory. Each core responsibility has departmental results and several indicators associated with it.
Departmental Results Framework
Core Responsibility 1: Health Care Systems
- R 1: Canada has modern and sustainable health care systems
- I1: National health expenditure as a percentage of Gross Domestic Product
- I2: Real per capita health expenditure
- I3: Drug spending as a percentage of Gross Domestic Product
- I4: Percentage of family physicians using electronic medical records
- R 2: Canadians have access to appropriate and effective health services
- I5: Percentage of Canadians (aged 15+) with a mental disorder who have expressed that they have an unmet mental health care need
- I6: Percentage of Canadians (aged 15+) who have expressed that they have an unmet need for access to home care services
- I7: Percentage of Canada Health Act compliance issues addressed within 24 months of identification
- I8: Percentage of Canadians who did not fill a prescription for medicine because of the cost
Program Inventory under core responsibility one (from one - 15) as follow:
- 1. Health Care Systems Analysis & Policy
- 2. Access, Affordability, & Appropriate Use of Drugs & Medical Devices
- 3. Home, Community & Palliative Care
- 4. Mental Health
- 5. Digital Health
- 6. Health Information
- 7. Canada Health Act
- 8. Medical Assistance in Dying
- 9. Cancer Control
- 10. Patient Safety
- 11. Blood Systems, Organs, Tissue & Transplantation
- 12. Promoting Minority Official Languages in the Health Care Systems
- 13. Brain Research
- 14. Thalidomide
- 15. The Territorial Health Investment Fund (THIF)
Core Responsibility 2: Health Protection and Promotion
- R 3: Canadians have access to safe, effective and quality health products
- I9: Percentage of new drug decisions issued within service standards
- I10: Percentage of Risk Management Plan reviews for new drug decisions completed within service standards
- I11: Percentage of drug companies deemed to be compliant with manufacturing requirements under the Food and Drugs Act and associated regulations
- R 4: Canadians are protected from unsafe consumer and commercial products and substances
- I12: Percentage of domestic consumer product recalls communicated to Canadians in a timely manner
- I13: Percentage of actions taken in a timely manner to protect the health of Canadians from substances found to be a risk to human health
- I14: Percentage of actions taken in a timely manner to protect the health of Canadians from pesticides found to be a risk to human health and the environment
- R 5: Canadians make healthy choices
- I15: Percentage of Canadians (aged 15+) who have used any tobacco product in the past 30 days
- I16: Percentage of Canadians (aged 15-24) who have used cannabis in the last 12 months
- I17: Percentage of Canadians who use dietary guidance provided by Health Canada
Program Inventory under core responsibility two (from 16-32) as follow:
- 16. Pharmaceutical Drugs
- 17. Biologics & Radiopharmaceutical Drugs
- 18. Medical Devices
- 19. Natural Health Products
- 20. Food & Nutrition
- 21. Air Quality
- 22. Climate Change
- 23. Water Quality
- 24. Health Impacts of Chemicals
- 25. Consumer Product Safety
- 26. Workplace Hazardous Products
- 27. Tobacco Control
- 28. Controlled Substances
- 29. Cannabis
- 30. Radiation Protection
- 31. Pesticides
- 32. Specialized Health Services & Internationally Protected Persons Program
Internal Services
Changes to the approved reporting framework since 2020-21
The Substance Use and Addictions grants and contribution program supports the Canadian Drugs and Substances Strategy and Canada's Tobacco Strategy. As such, instead of a stand-alone Program, the activities, funds and performance indicators associated with this grants and contribution program has been dispersed between the Tobacco Control, Controlled Substances and Cannabis Programs.
| Structure | 2021-22 | 2020-21 | Change | Rationale for change |
|---|---|---|---|---|
| Core Responsibility 1 | Health Care Systems | Health Care Systems | No change | Not applicable |
| Program | Health Care Systems Analysis & Policy | Health Care Systems Analysis & Policy | No change | Not applicable |
| Program | Access, Affordability, & Appropriate Use of Drugs & Medical Devices | Access, Affordability, & Appropriate Use of Drugs & Medical Devices | No change | Not applicable |
| Program | Home, Community & Palliative Care | Home, Community & Palliative Care | No change | Not applicable |
| Program | Mental Health | Mental Health | No change | Not applicable |
| Program | Not applicable | Substance Use and Addictions | Program ended | The Program was integrated into other Programs to streamline and strengthen corporate planning, performance measurement and reporting |
| Program | Digital Health | Digital Health | No change | Not applicable |
| Program | Health Information | Health Information | No change | Not applicable |
| Program | Canada Health Act | Canada Health Act | No change | Not applicable |
| Program | Medical Assistance in Dying | Medical Assistance in Dying | No change | Not applicable |
| Program | Cancer Control | Cancer Control | No change | Not applicable |
| Program | Patient Safety | Patient Safety | No change | Not applicable |
| Program | Organs, Tissues and Blood | Organs, Tissues and Blood | No change | Not applicable |
| Program | Promoting Minority Official Languages in the Health Care Systems | Promoting Minority Official Languages in the Health Care Systems | No change | Not applicable |
| Program | Brain Research | Brain Research | No change | Not applicable |
| Program | Thalidomide | Thalidomide | No change | Not applicable |
| Program | The Territorial Health Investment Fund | The Territorial Health Investment Fund | No change | Not applicable |
| Structure | 2021-22 | 2020-21 | Change | Rationale for change |
|---|---|---|---|---|
| Core Responsibility 2 | Health Protection & Promotion |
Health Protection & Promotion |
No change | Not applicable |
| Program | Pharmaceutical Drugs | Pharmaceutical Drugs | No change | Not applicable |
| Program | Biologics & Radiopharmaceutical Drugs | Biologics & Radiopharmaceutical Drugs | No change | Not applicable |
| Program | Medical Devices | Medical Devices | No change | Not applicable |
| Program | Natural Health Products | Natural Health Products | No change | Not applicable |
| Program | Food & Nutrition | Food & Nutrition | No change | Not applicable |
| Program | Air Quality | Air Quality | No change | Not applicable |
| Program | Climate Change | Climate Change | No change | Not applicable |
| Program | Water Quality | Water Quality | No change | Not applicable |
| Program | Health Impacts of Chemicals | Health Impacts of Chemicals | No change | Not applicable |
| Program | Consumer Product Safety | Consumer Product Safety | No change | Not applicable |
| Program | Workplace Hazardous Products | Workplace Hazardous Products | No change | Not applicable |
| Program | Tobacco Control | Tobacco Control | No change | Not applicable |
| Program | Controlled Substances | Controlled Substances | No change | Not applicable |
| Program | Cannabis | Cannabis | No change | Not applicable |
| Program | Radiation Protection | Radiation Protection | No change | Not applicable |
| Program | Pesticides | Pesticides | No change | Not applicable |
| Program | Specialized Health Services & Internationally Protected Persons | Specialized Health Services & Internationally Protected Persons | No change | Not applicable |
Supporting information on the Program Inventory
Supporting information on planned expenditures, human resources, and results related to the Health Canada's Program Inventory is available in the GC InfoBase.Endnote xvi
Supplementary information tables
The following supplementary information tables are available on Health Canada's website:
- Departmental Sustainable Development Strategy
- Details on transfer payment programs
- Gender-based analysis plus
- Horizontal initiatives
- Up-front multi-year funding
Federal tax expenditures
Health Canada's Departmental Plan does not include information on tax expenditures that relate to its planned results for 2021–22.
Tax expenditures are the responsibility of the Minister of Finance, and the Department of Finance Canada publishes cost estimates and projections for government wide tax expenditures each year in the Report on Federal Tax Expenditures.Endnote xvii This report provides detailed information on tax expenditures, including objectives, historical background and references to related federal spending programs, as well as evaluations, research papers and gender-based analysis. The tax measures presented in this report are solely the responsibility of the Minister of Finance.
Organizational contact information
Edward de Sousa
Director General, Health Canada
Director General's Office
200 Eglantine Driveway, Tunney's Pasture
Ottawa, Ontario K1A 0K9
Telephone : 613-948-6358
Telephone : 613-948-6358
edward.desousa@canada.ca
Health Canada website
Appendix: Definitions
- appropriation
- Any authority of Parliament to pay money out of the Consolidated Revenue Fund.
- budgetary expenditures
- Operating and capital expenditures; transfer payments to other levels of government, organizations or individuals; and payments to Crown corporations.
- Core Responsibility
- An enduring function or role performed by a department. The intentions of the department with respect to a Core Responsibility are reflected in one or more related Departmental Results that the department seeks to contribute to or influence.
- Departmental Plan
- A report on the plans and expected performance of an appropriated department over a three-year period. Departmental Plans are tabled in Parliament each spring.
- departmental priority
- A plan or project that a department has chosen to focus and report on during the planning period. Departmental priorities represent the things that are most important or what must be done first to support the achievement of the desired departmental results.
- Departmental Result
- Any change that the department seeks to influence. A Departmental Result is often outside departments' immediate control, but it should be influenced by Program-level outcomes.
- Departmental Result Indicator
- A factor or variable that provides a valid and reliable means to measure or describe progress on a Departmental Result.
- Departmental Results Framework
- A framework that consists of the department's core responsibilities, departmental results and departmental result indicators.
- Departmental Results Report
- A report on the actual accomplishments against the plans, priorities and expected results set out in the corresponding Departmental Plan.
- experimentation
- The conducting of activities that seek to first explore, then test and compare, the effects and impacts of policies and interventions in order to inform evidence-based decision-making, and improve outcomes for Canadians, by learning what works and what doesn't. Experimentation is related to, but distinct form innovation (the trying of new things), because it involves a rigorous comparison of results. For example, using a new website to communicate with Canadians can be an innovation; systematically testing the new website against existing outreach tools or an old website to see which one leads to more engagement, is experimentation.
- full-time equivalent
- A measure of the extent to which an employee represents a full person-year charge against a departmental budget. Full-time equivalents are calculated as a ratio of assigned hours of work to scheduled hours of work. Scheduled hours of work are set out in collective agreements.
- gender-based analysis plus (GBA+)
- An analytical process used to assess how diverse groups of women, men and gender-diverse people experience policies, programs and services based on multiple factors including race, ethnicity, religion, age, and mental or physical disability.
- government-wide priorities
- For the purpose of the 2020-21 Departmental Plan, government-wide priorities refers to those high-level themes outlining the government's agenda in the 2020 Speech from the Throne, namely: Protecting Canadians from COVID-19; Helping Canadians through the pandemic; Building back better - a resiliency agenda for the middle class; The Canada we're fighting for.
- horizontal initiative
- An initiative where two or more federal organizations are given funding to pursue a shared outcome, often linked to a government priority.
- non-budgetary expenditures
- Net outlays and receipts related to loans, investments and advances, which change the composition of the financial assets of the Government of Canada.
- performance
- What an organization did with its resources to achieve its results, how well those results compare to what the organization intended to achieve, and how well lessons learned have been identified.
- performance indicator
- A qualitative or quantitative means of measuring an output or outcome, with the intention of gauging the performance of an organization, program, policy or initiative respecting expected results.
- performance reporting
- The process of communicating evidence-based performance information. Performance reporting supports decision-making, accountability and transparency.
- plan
- The articulation of strategic choices, which provides information on how an organization intends to achieve its priorities and associated results. Generally a plan will explain the logic behind the strategies chosen and tend to focus on actions that lead up to the expected result.
- planned spending
-
For Departmental Plans and Departmental Results Reports, planned spending refers to those amounts presented in the Main Estimates.
A department is expected to be aware of the authorities that it has sought and received. The determination of planned spending is a departmental responsibility, and departments must be able to defend the expenditure and accrual numbers presented in their Departmental Plans and Departmental Results Reports.
- Program
- Individual or groups of services, activities or combinations thereof that are managed together within the department and focus on a specific set of outputs, outcomes or service levels.
- Program Inventory
- Identifies all of the department's programs and describes how resources are organized to contribute to the department's core responsibilities and results.
- result
- An external consequence attributed, in part, to an organization, policy, program or initiative. Results are not within the control of a single organization, policy, program or initiative; instead they are within the area of the organization's influence.
- statutory expenditures
- Expenditures that Parliament has approved through legislation other than appropriation acts. The legislation sets out the purpose of the expenditures and the terms and conditions under which they may be made.
- strategic outcome
- A long-term and enduring benefit to Canadians that is linked to the organization's mandate, vision and core functions.
- target
- A measurable performance or success level that an organization, program or initiative plans to achieve within a specified time period. Targets can be either quantitative or qualitative.
- voted expenditures
- Expenditures that Parliament approves annually through an Appropriation Act. The Vote wording becomes the governing conditions under which these expenditures may be made.
Endnotes
Endnotes
- Endnote i
-
Treasury Board of Canada Secretariat, https://www.canada.ca/en/treasury-board-secretariat.html
- Endnote ii
-
2020-21 Main Estimates, https://www.canada.ca/en/treasury-board-secretariat/services/planned-government-spending/government-expenditure-plan-main-estimates.html
- Endnote iii
-
Assisted Human Reproduction Act https://laws-lois.justice.gc.ca/eng/acts/a-13.4
- Endnote iv
-
Canada Health Act https://laws-lois.justice.gc.ca/eng/acts/C-6/
- Endnote v
-
Canada Consumer Product Safety Act https://laws-lois.justice.gc.ca/eng/acts/c-1.68/
- Endnote vi
-
Cannabis Act https://laws-lois.justice.gc.ca/eng/acts/C-24.5/
- Endnote vii
-
Controlled Drugs and Substances Act https://laws-lois.justice.gc.ca/eng/acts/c-38.8/
- Endnote viii
-
Department of Health Act https://laws-lois.justice.gc.ca/eng/acts/H-3.2/index.html
- Endnote ix
-
Food and Drugs Act https://laws.justice.gc.ca/eng/acts/F-27/
- Endnote x
-
Hazardous Materials Information Review Act https://laws-lois.justice.gc.ca/eng/acts/H-2.7/
- Endnote xi
-
Hazardous Products Act https://laws-lois.justice.gc.ca/eng/acts/H-3/index.html
- Endnote xii
-
Pest Control Products Act https://laws-lois.justice.gc.ca/eng/acts/P-9.01/
- Endnote xiii
-
Radiation Emitting Devices Act https://laws-lois.justice.gc.ca/eng/acts/R-1/
- Endnote xiv
-
Tobacco and Vaping Products Act https://laws-lois.justice.gc.ca/eng/acts/T-11.5/
- Endnote xv
-
List of Acts, https://www.canada.ca/en/health-canada/corporate/about-health-canada/legislation-guidelines/acts-regulations/list-acts-regulations.html
- Endnote xvi
-
GC InfoBase, https://www.tbs-sct.gc.ca/ems-sgd/edb-bdd/index-eng.html
- Endnote xvii
-
Report on Federal Tax Expenditures, https://www.canada.ca/en/department-finance/services/publications/federal-tax-expenditures.html
- Footnote 1
-
In late 2020, the Canadian Foundation for Healthcare Improvement (CFHI) and the Canadian Patient Safety Institute (CPSI) amalgamated their organizations, now operating under the name of CFHI. As such, 2021-22 will be a transition year for the new organization as it amalgamates into one program. The Department's annual funding to each organization ($17 million to CFHI and $7.6 million to CPSI) has been combined and is flowing to the amalgamated organization under CFHI's name. For more detailed information, please refer to the Supplementary Information Table "Details on transfer payment programs - Contribution to improve health care quality and patient safety."
- Footnote 2
-
The CHT is an ongoing annual transfer to P/Ts that grows each year in line with nominal GDP growth, or a floor of 3%.
- Footnote 3
-
The terms Gender-Based Analysis Plus (GBA+) and Sex and Gender-Based Analysis Plus (SGBA+) refer to the same concept. Health Canada has chosen to use SGBA+ to emphasize the fact that differences between women, men and gender-diverse individuals can be biological (sex related) and/or socio-cultural (gender related).