Health Product InfoWatch: September 2021
Download in PDF format
(1.13 MB, 9 pages)
Health Products and Food Branch
Marketed Health Products Directorate
Health Product InfoWatch Editorial Team
ISSN: 2368-8025
Cat.: H167-1E-PDF
Pub.: 210000
Contents
- Health products mentioned in this issue
- Coronavirus disease (COVID-19)
- Drug and vaccine authorizations and communications for COVID-19
- Monthly recap
- New information
- Scope
- Helpful links
- Suggestions?
- Copyright
Health products mentioned in this issue
Pharmaceuticals and biologics
Comirnaty (Pfizer-BioNTech COVID-19 Vaccine)
Fluoroquinolones
Losartan
pms-PROGESTERONE
Spikevax (COVID-19 Vaccine Moderna)
Vaxzevria (AstraZeneca COVID-19 Vaccine)
Veklury (remdesivir)
Medical devices
Ultrasound Gels and Lotions Manufactured by Eco-Med Pharmaceuticals, Inc.
Natural and non-prescription health products
Eco-Med Prevent + Hand Sanitizer
Hand sanitizers that may pose health risks
Other
Ivermectin
Unauthorized health products
Coronavirus disease (COVID-19)
For the most up-to-date information on COVID-19, please visit the Government of Canada Coronavirus disease (COVID-19) website Canada.ca/coronavirus, which includes a dedicated section for healthcare professionals, and for the health product industry.
The COVID-19 vaccines and treatments portal provides information for consumers, healthcare professionals and researchers on vaccines and treatments authorized for COVID-19.
For information about adverse events following immunization that individuals have reported after receiving a COVID-19 vaccine in Canada, new safety signals or other safety updates, please visit the COVID-19 vaccine safety in Canada webpage. This page is updated weekly.
Drug and vaccine authorizations and communications for COVID-19
Recent communications related to authorized COVID-19 vaccines and treatments are highlighted in this section.
Notice of Compliance for COVID-19 drugs and vaccines
On September 16, 2020, the Minister of Health signed the Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19 (Interim Order) to allow for the expedited authorization of COVID-19 drugs and vaccines. The Interim Order expired on September 16, 2021, and products authorized under the Interim Order can now be transitioned to the Food and Drug Regulations (FDR), after the market authorization holder submits a New Drug Submission to Health Canada for review. The FDR were amended to permit the same flexibilities provided by the Interim Order (e.g., rolling submissions, terms and conditions for monitoring and pre-positioning). Authorization under the FDR will provide the companies with some additional benefits, such as data protection. New COVID-19 products are eligible to apply directly to the FDR.
Pfizer-BioNTech COVID-19 Vaccine and COVID-19 Vaccine Moderna, originally authorized under the Interim Order, have now been issued Notice of Compliance (NOC) under the FDR. These authorizations do not reflect any change in the safety or efficacy profile of these products or in how they are administered, but are a transition in the regulatory mechanism to allow continued access for Canadians to products that provide protection against and treatment for COVID-19.
Brand name change for COVID-19 drugs and vaccines
The following COVID-19 vaccines have undergone a brand name change in September 2021 upon NOC issuance or labelling update under the Interim Order:
New Brand name |
Date of NOC issuance |
Former brand name (as authorized under the Interim Order) |
Date of Interim Order authorization |
| Comirnaty (koe-mir'-na-tee) | September 16, 2021 | Pfizer-BioNTech COVID-19 Vaccine |
December 9, 2020 |
Spikevax |
September 16, 2021 | COVID-19 Vaccine Moderna |
December 23, 2020 |
Vaxzevria |
PendingFootnote * |
AstraZeneca COVID-19 Vaccine |
February 26, 2021 |
|
|||
Canadian product monographs for authorized vaccines and treatments for COVID-19 can be accessed through the COVID-19 vaccines and treatments portal or Health Canada's Drug Product Database.
Veklury (remdesivir)
This safety review evaluated the risk of sinus bradycardia associated with Veklury (remdesivir). Health Canada's review of the available information concluded that a link is possible. The Department will work with the manufacturer of Veklury to update the Canadian product monograph to include the potential risk of sinus bradycardia.
Summary Safety Review - Veklury (remdesivir)
Monthly recap of health product safety information
The following is a list of health product advisories, type I recalls and summaries of completed safety reviews published in August 2021 by Health Canada.
For health product advisories related to COVID-19 vaccines and treatments, please see the Drug and vaccine authorizations and communications for COVID-19 section.
Certain hand sanitizers that may pose health risks
Health Canada advised Canadians that certain hand sanitizers were recalled because they either contained ingredients that were not permitted by Health Canada, were not properly labelled, were unauthorized, or were missing important information.
Advisory - Certain hand sanitizers that may pose health risks
Fluoroquinolones
This safety review evaluated the risk of heart valve regurgitation associated with systemic or inhaled fluoroquinolone antibiotics (ciprofloxacin-, levofloxacin-, moxifloxacin- and norfloxacin-containing products). Health Canada's review of the available information did not establish a link. Health Canada will continue to monitor safety information involving fluoroquinolones.
Summary Safety Review - Fluoroquinolones
Health products manufactured by Eco-Med Pharmaceuticals Inc.
Health Canada advised Canadians to stop using and discard all products manufactured by Eco-Med Pharmaceuticals Inc. (ultrasound gels, transmission and massage lotions, hand sanitizers and first aid antiseptics) due to potential bacterial contamination with Burkholderia stabilis. Health Canada has also communicated this information to healthcare professionals.
Advisory - Health products manufactured by Eco-Med Pharmaceuticals Inc.
Health Professional Risk Communication - Ultrasound Gels and Lotions
Manufactured by Eco-Med Pharmaceuticals Inc.
Medical Device Recall - EcoGel 200 Ultrasound Gel - MediChoice
(M500812)
Drug Recall - Eco-Med Prevent + Hand Sanitizer
Ivermectin
Health Canada received concerning reports of the use of veterinary ivermectin to prevent or treat COVID-19. Health Canada advised Canadians not to use either the veterinary or human drug versions of ivermectin to prevent or treat COVID-19.
Losartan
Teva Canada Ltd. and Sivem Pharmaceuticals ULC recalled 7 lots and Sandoz Canada Inc. and Sanis Health Inc. recalled all lots of prescription losartan tablets, in 25 mg, 50 mg and 100 mg strengths, after tests found an azido impurity above the acceptable limit.
pms-PROGESTERONE
Pharmascience Inc. recalled one lot (1318593, expiry 10/2022) of pms-PROGESTERONE 100 mg capsules because the bottles may contain Akorn 100 mg progesterone gelatin capsules in addition to, or instead of the intended pms-PROGESTERONE 100 mg gelatin capsules. The Akorn capsules contain peanut oil, an ingredient that is not listed on the label of pms-PROGESTERONE 100 mg capsules.
Unauthorized health products
Health Canada advised Canadians about various unauthorized health products being sold at retail locations across Canada or online that may pose serious health risks.
Advisory - Various unauthorized health products
New health product safety information
The following topic has been selected to raise awareness and, in some cases, to stimulate reporting of adverse reactions.
Adverse reactions to health products - annual report 2020
Introduction
Post-market proactive and spontaneous reporting systems help in the
identification and analysis of new safety information for health products
so that appropriate action can be taken to minimize risks to human health.
In Canada, adverse reactions (ARs) or medical device incidents (MDIs)
suspected of being associated with the use of health products can be reported to the Canada Vigilance Program (CVP). This article summarizes information about domestic AR cases reported
for pharmaceuticals, natural health products, biologics, Footnote * radiopharmaceuticals, disinfectants, sanitizers with disinfectant claims,
and cannabis products received by the CVP in 2020 and posted on the Canada Vigilance AR online database. Although foreign AR cases are included in the internal CVP database,
these are not included in this article.
Domestic adverse reaction reports and cases
In 2020, Health Canada received 186,099 domestic post-market AR reports.
These reports represented 79,345 AR cases. A case consists of all
information describing the AR(s) experienced by one patient at one time and
suspected of being related to the use of one or more health products; thus,
a case may include an initial AR report and possibly several follow-up
reports that provide additional information about the AR(s). Duplicate
cases may exist if an AR report about the same AR event was received from
different reporters (e.g., from a health professional, consumer, hospital,
and/or manufacturer). Domestic AR cases reported in 2020 by product type
are provided in Table 1.
Product type |
No. (%) of reports |
Pharmaceuticals |
46,567 (58.7) |
BiologicsFootnote * |
31,111 (39.2) |
Natural health products |
488 (0.6) |
Radiopharmaceuticals |
466 (0.6) |
Medical cannabis |
121 (0.2) |
Non-medical cannabis |
40 (0.0) |
OtherFootnote † |
552 (0.7) |
Total |
79,345 (100) |
|
|
In Canada, Market Authorization Holders (MAHs) and hospitals are required to submit AR reports to the CVP in accordance with the requirements of the Food and Drugs Act and its Regulations. For serious ARs that have occurred in Canada (domestic ARs), MAHs are required to send a report within 15 days of becoming aware of the incident. Hospitals are required to send, within 30 days of documentation, all reports of serious ARs to therapeutic products that have occurred in their facility. Community members (consumers, patients and non-hospital-based health professionals) can voluntarily submit AR reports at any time.
In 2020, MAHs submitted 86.8% of all domestic AR cases received by the CVP, while hospitals submitted 6.9% (Figure 1). The remaining cases were mainly received directly from community members (6.1%). Most of the domestic AR cases reported to Health Canada directly or via a MAH originated from health professionals, including physicians, pharmacists, nurses, dentists, etc. (Figure 2).

Figure 1 - Text description
The figure shows the percentage of number of domestic AR cases reported in 2020 by source.
Report source |
Number of cases by percentage |
Market Authorization Holder |
86.8% |
Hospital |
6.9% |
Community |
6.1% |
Other |
0.3% |
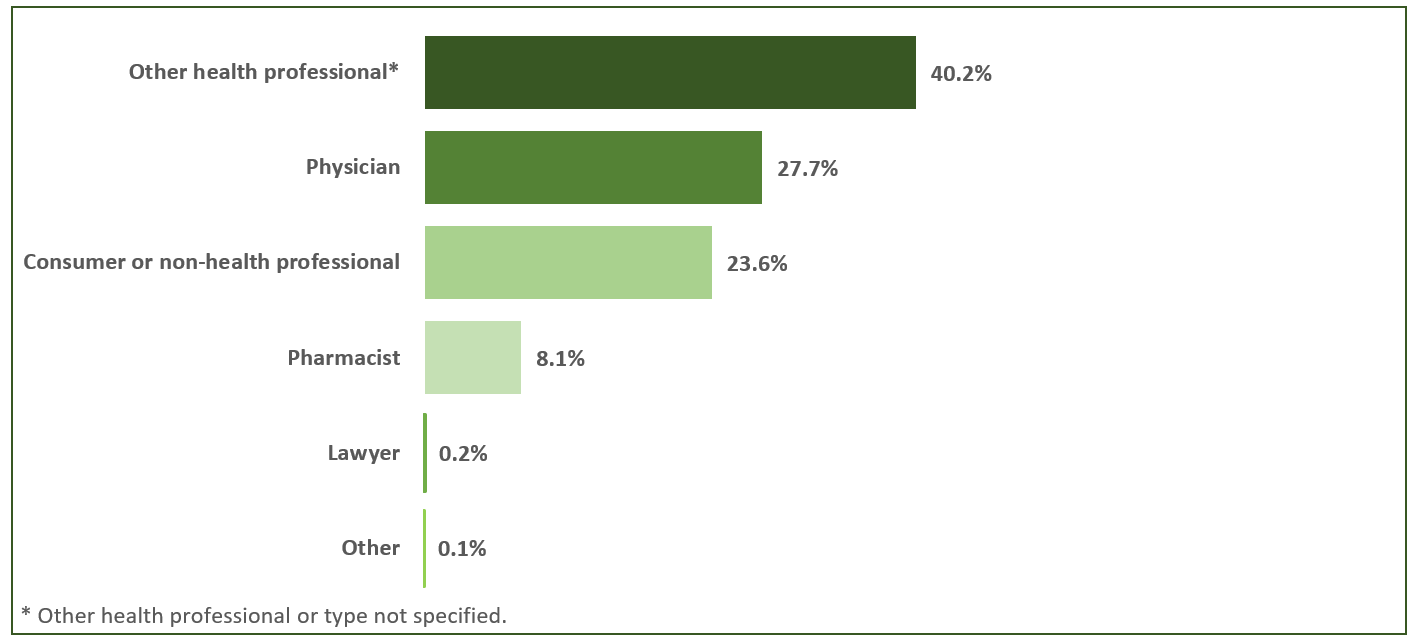
Figure 2 - Text description
The figure shows the percentage of number of domestic AR cases reported in 2020 by type of originating reporter.
Originating reporter |
Number of cases by percentage |
Other health professionalFootnote * |
40.2% |
Physician |
27.7% |
Consumer Or Other Non-Health Professional |
23.6% |
Pharmacist |
8.1% |
Lawyer |
0.2% |
Other |
0.1% |
|
|
Sex and age distribution
The distribution for the 79,345 cases by sex was 58.5% female, 37.1% male
and 4.3% sex unknown or unreported, which reflects the sex distribution
seen globally. Footnote † The distribution by age group was 3.9% pediatric (< 19 years), 51.6%
adult (19-64 years), 24.6% elderly (≥ 65 years) and 19.9% age unknown or
unreported.
Suspect products and adverse reactions
The top 10 groups of suspect products most commonly identified in domestic
AR cases are listed in Table 2. The groups are classified according to the World Health Organization's Anatomical Therapeutic Chemical (ATC)
classification system.
Health product (ATC group) |
No. (%) of times reportedFootnote † |
Immunosuppressants (L04) |
49,119 (31.9) |
Antineoplastic agents (L01) |
21,613 (14.0) |
Drugs for obstructive airway diseases (R03) |
10,829 (7.0) |
Analgesics (N02) |
7,591 (4.9) |
PsycholepticsFootnote ‡ (N05) |
5,500 (3.6) |
Antiepileptics (N03) |
4,325 (2.8) |
Corticosteroids for systemic use (H02) |
3,869 (2.5) |
Antidiarrheals, intestinal antiinflammatory/antiinfective agents (A07) |
3,560 (2.3) |
Antibacterials for systemic use (J01) |
2,979 (1.9) |
Antiinflammatory and antirheumatic products (M01) |
2,723 (1.8) |
|
|
Table 3 displays the top 10 domestic ARs reported to the CVP based on System Organ Class codes. The ARs are coded using Medical Dictionary for Regulatory Activities (MedDRA) terminology. The most commonly reported ARs were general disorders and administration site conditions, which include disorders that affect several body systems or sites (e.g., drug ineffective, fatigue, fever, edema, pain, reactions at the administration site). The second most common ARs were gastrointestinal disorders.
System Organ Class |
No. (%) of times reportedFootnote † |
General disorders and administration site conditions |
77,209 (22.7) |
Gastrointestinal disorders |
32,661 (9.6) |
Injury, poisoning and procedural complications |
30,268 (8.9) |
Investigations |
27,926 (8.2) |
Musculoskeletal and connective tissue disorders |
25,904 (7.6) |
Respiratory, thoracic and mediastinal disorders |
22,326 (6.6) |
Infections and infestations |
21,896 (6.5) |
Nervous system disorders |
20,913 (6.2) |
Skin and subcutaneous tissue disorders |
16,377 (4.8) |
Psychiatric disorders |
12,209 (3.6) |
|
|
Reason for seriousness
Of the 79,345 AR cases, 69.6% were considered serious. Footnote ‡ A case can have more than one reported reason for seriousness. In 2020,
22.5% of all AR cases indicated that hospitalization was required, 2.8%
indicated a life-threatening condition, and 6.8% indicated a death had
occurred.
Investigation of reported adverse reactions
It is difficult to compare the risk of health products based solely on the
volume of submitted reports. Several factors may influence AR reporting,
such as the known risks associated with a product, the length of time a
product has been on the market, volume of use, publicity about an AR,
regulatory actions taken to minimize risks, and/or method of data
collection. For example, rare and serious ARs may be reported more
frequently in organized data collection systems Footnote § compared to voluntary reporting, which may affect the pattern of reporting.
In general, there is underreporting of adverse events to spontaneous
reporting programs like the CVP.
AR reports are reviewed as part of a routine monitoring strategy. When a reported AR is known and included in the product monograph, it is not considered to be a new signal unless there is a change in the frequency or severity of the AR. Post-market ARs may be attributed to a variety of factors, including previously unrecognized pharmacological effects of the product, idiosyncratic effects, drug interactions (e.g., drug-drug, drug-disease, drug-natural health product interactions), or factors related to specific patient populations, individual patient factors (e.g., pharmacogenomics factors), medication incidents, or other factors that may have been too infrequent to be identified in clinical trials.
AR reports, along with other sources of information from domestic and international sources, help in the identification and analysis of new safety information and support Health Canada's decisions to take action. For example, a causal association between the product and the AR may prompt an action from Health Canada. The same applies if new risks are determined from a cluster of similarly reported ARs, or from AR reports suggesting labelling gaps or product quality issues.
In 2020, drug-related information received and assessed by Health Canada resulted in 1,034 actions, which included informing Canadians and health professionals about new safety information, recommending label changes, or removing a drug product from the market. The important new safety information for health professionals and Canadians is communicated via the Recalls and Safety Alerts Database on the Healthy Canadians Web Site. The new safety information is also further distributed through the MedEffect™ e-Notice email notification system. In addition, Health Canada published 20 summaries of its safety reviews, which describe Health Canada's findings and risk management actions related to potential safety issues.
Domestic adverse reactions related to COVID-19 health products including
vaccines
In 2020, Health Canada received 7 AR cases related to authorized and unauthorized
products used to treat COVID-19 (remdesivir, authorized on July 27, 2020,
and drugs used off-label for COVID-19, including hydroxychloroquine
sulfate, tocilizumab, immune globulin, azithromycin and ceftriaxone, and
methylprednisolone and prednisone). Health Canada also received 2 AR cases
related to a COVID-19 vaccine (Pfizer-BioNTech COVID-19 vaccine, authorized
on December 9, 2020). An up-to-date summary of reported ARs following
COVID-19 vaccination in Canada, including those reported to the Canadian
Adverse Events Following Immunization Surveillance System managed by the
Public Health Agency of Canada, is available online.
Conclusion
Each year, the CVP receives thousands of reports that contribute to a better understanding of the safety associated with marketed health products. Health Canada would like to thank all who have contributed information and encourage the continued support of post-market surveillance through AR and MDI reporting. Any ARs or MDIs suspected of being associated with the use of health products should be reported to the CVP. Every report counts, and together, they tell a story.
Scope
This monthly publication is intended primarily for healthcare professionals and includes information on pharmaceuticals, biologics, medical devices and natural health products. It provides a summary of key health product safety information published in the previous month by Health Canada, as well as a selection of new health product safety information meant to raise awareness. New information contained in this issue is not comprehensive but rather represents a selection of clinically relevant items warranting enhanced dissemination.
Reporting Adverse Reactions
Canada Vigilance Program
Telephone: 1-866-234-2345
Fax or mail: Form available on MedEffect Canada
For more information on how to report an adverse reaction, visit the Adverse Reaction and Medical Device Problem Reporting page.
Helpful links
- MedEffectTM Canada
- Recalls and Safety Alerts Database
- New Safety and Effectiveness Reviews
- Canada Vigilance Adverse Reaction Online Database
- Drug Product Database
- Medical Devices Active Licence Listing
- Licensed Natural Health Products Database
- The Drug and Health Product Register
- Drug Shortages Canada
- Stop Illegal Marketing of Drugs and Devices
- List of drugs for exceptional importation and sale
- Drug and vaccine authorizations for COVID-19: List of authorized drugs, vaccines and expanded indications
- Reported side effects following COVID-19 vaccination in Canada
Suggestions?
Your comments are important to us. Let us know what you think by reaching us at infowatch-infovigilance@hc-sc.gc.ca
Health Canada
Marketed Health Products Directorate
Address Locator 1906C
Ottawa ON K1A 0K9
Telephone: 613-954-6522
Fax: 613-952-7738
Copyright
©2021 Her Majesty the Queen in Right of Canada. This publication may be reproduced without permission provided the source is fully acknowledged. The use of this publication for advertising purposes is prohibited. Health Canada does not assume liability for the accuracy or authenticity of the information submitted in case reports.
Adverse reactions (ARs) to health products are considered to be suspicions, as a definite causal association often cannot be determined. Spontaneous reports of ARs cannot be used to estimate the incidence of ARs because ARs remain underreported and patient exposure is unknown.
Due to time constraints relating to the production of this publication, information published may not reflect the most current information.
Footnotes
- Footnote *
-
Biologics include biotechnology products; vaccines; fractionated blood products; human blood and blood component products; human cells, tissues, and organs; and sperm and ova products.
- Footnote †
-
Watson, S., Caster, O., Rochon, P.A. & den Ruijter, H. (2019). Reported adverse drug reactions in women and men: Aggregated evidence from globally collected individual case reports during half a century. EClinicalMedicine, 17, 100188, ISSN 2589-5370. https://doi.org/10.1016/j.eclinm.2019.10.001.
- Footnote ‡
-
In the Food and Drugs Act and Regulations, a serious AR is defined as "a noxious and unintended response to a drug that occurs at any dose and that requires in-patient hospitalization or prolongation of existing hospitalization, causes congenital malformation, results in persistent or significant disability or incapacity, is life-threatening or results in death". Other situations may also warrant a designation as serious, "such as medically important events that may not be immediately life-threatening or result in death or hospitalization but may jeopardize the patient or may require intervention to prevent one of the other outcomes listed in the definition from the Regulations".
- Footnote §
-
Organized data collection systems include patient registries, surveys, and patient support or disease management programs.
