Guidance Document - Health Canada and Advertising Preclearance Agencies' Roles Related to Health Product Advertising
Date Created: 2010-11-03
HC Pub.: 100624
Cat.: 978-1-100-17539-3
ISBN: H164-132/2010E-PDF
Table of Contents
- 1 Introduction
- 1.1 Purpose
- 1.2 Scope
- 1.3 Background
- 1.4 Definitions
- 2 Advertising Preclearance Overview
- 3 Guidance for Implementation
- Appendix A: List of Abbreviations
- Appendix B: Legislative and Regulatory Framework
- Appendix C: Canadian Advertising Preclearance Agencies
- Appendix D: Roles of Advertising Preclearance Agencies
- Appendix E: Advertising Complaint Adjudication
Foreword
Guidance documents are meant to provide assistance on how to comply with governing statutes and regulations. Guidance documents also provide assistance to staff on how Health Canada's mandates and objectives should be implemented in a manner that is fair, consistent and effective.
Guidance documents are administrative instruments not having force of law and, as such, allow for flexibility in approach. Alternative approaches to the principles and practices described in this document may be acceptable provided they are supported by adequate justification. Alternative approaches should be discussed in advance with the relevant program area to avoid possibly finding that applicable statutory or regulatory requirements have not been met.
As a corollary to the above, it is equally important to note that Health Canada reserves the right to request information or material, or define conditions not specifically described in this document. Health Canada is committed to ensuring that such requests are justifiable and that decisions are clearly documented.
This document should be read in conjunction with the relevant sections of other applicable guidance documents and policies. This guidance document supersedes the two policies entitled "Pharmaceutical Advertising Advisory Board and Health Canada Roles and Consultation Related to Advertising Review" (1996) and "Advertising Standards Canada and Health Canada's Roles and Consultation Related to Advertising Review and Complaint Adjudication" (1997).
1 Introduction
1.1 Purpose
Health Canada and the Canadian Advertising Preclearance Agencies (APAs) share the common goal of maintaining integrity in health product advertising. The purpose of this guidance document is to inform staff and stakeholders of the different roles these organizations play in achieving this goal.
1.2 Scope
This guidance document clarifies the roles of Health Canada and the Canadian APAs with respect to the review of health product advertising, the adjudication of complaints and processes to submit requests for clarifications. Health products include prescription and non-prescription pharmaceuticals, biologicals and natural health products intended for human use. Advertising of medical devices is not included in the scope of this document.
1.3 Background
Health Canada is the national regulatory authority for health product advertisements and is responsible for administering and directing compliance with the Acts and Regulations. It is the responsibility of all advertisers to ensure that health product advertisements comply with the requirements of the Food and Drugs Act (F&DA) and its
Regulations, and the Controlled Drugs and Substances Act (CDSA).
When required by the F&DA andits Regulations, market authorization holders of health products will file, prior to marketing, a submission containing information and evidence to establish the safety, efficacy and quality of a health product for its intended use, and will subsequently receive marketing authorization in the form of a Notice of Compliance (NOC) and/or a Drug Identification Number (DIN), a Natural Product Number (NPN) or a Drug Identification Number for Homeopathic Medicine (DIN-HM).
For the purposes of the F&DA, advertising is defined as including any representation by any means whatever for the purpose of promoting directly or indirectly the sale or disposal of any food, drug, cosmetic or device. If a message regarding a drug is not considered to promote the sale or use of the drug, it is not subject to the advertising provisions of the F&DAand Regulations. The Health Canada policy The Distinction between Advertising and Other Activities clarifies the distinction between advertising to promote the sale of a drug and activities that are not primarily intended to promote the sale of a drug.
The preclearance of advertising for marketed health products is administered through an independent, self-regulatory and voluntary system. Advertising preclearance agencies are independent entities which review and preclear advertising material to help interested parties ensure compliance with the regulatory provisions of the F&DA, CDSA and respective Regulations, the Natural Health Products Regulations (NHPR), and consistency with the various Health Canada guidance documents and their own codes of advertising. The agencies also offer mechanisms to resolve complaints on advertising for authorized health products.
Numerous provisions of the F&DAand its Regulations (e.g.,the Food and Drug Regulations [F&DR]and the NHPR) and the CDSA apply to drug advertising, and are included as reference in appendix B.
1.4 Definitions
Some of the following terms may be interpreted more broadly in other, non-advertising contexts; however, for the purpose of this guidance document, they are defined as follows:
- Advertisement:
- Any representation by any means whatever for the purpose of promoting directly or indirectly the sale or use of any food, drug, cosmetic or device.
- Advertising Preclearance Agencies:
- Advertising preclearance agencies are independent entities which review and preclear advertising material to help interested parties ensure compliance with the advertising provisions of federal legislation, the various Health Canada guidance documents, as well as their own codes of advertising. The agencies also offer mechanisms to resolve complaints on advertising for authorized health products. The board of directors or advisory bodies of these agencies may include stakeholders from academia, consumer groups, the media, advertising agencies, the pharmaceutical industry, and healthcare professional associations. Health Canada acts as an ex-officio observer and advisor to these boards and advisory bodies, without relinquishing any part of its authority under the F&DA and Regulations. Although Health Canada works in collaboration with these agencies, it does not endorse them.
- Appeal:
- This refers to a request submitted to an APA under an appeal process and procedure stipulated by the agency for review of the agency decision to deny approval of advertising material submitted for preclearance.
- Claim:
- Any representation made on behalf of a health product, including the indication for use and marketing claims. A marketing claim may be a statement that is designed to promote the sale of a health product and which highlights a specific product attribute e.g., "longer lasting" or "tastes great".
- Comparative claim:
- A comparative claim is a statement that compares an identified attribute of one drug product/ingredient to that of another/other drug product(s)/ingredients(s) in terms of comparability or superiority.
- Complaint:
- Complaint refers to a written submission by any party, alleging that an advertisement is non-compliant, in whole or in part, with a provision of the F&DA, CDSA, respective Regulations, Health Canada advertising guidance documents or policies, and/or with advertising codes of the APAs.
- Compliance verification:
- Actions taken to verify compliance in response to information regarding known or suspected non-compliance with the applicable requirements of the F&DA and its associated Regulations. This includes actions such as information gathering either off-site or via on-site visits.
- Health Risk Assessment:
- Health risk assessment (HRA) can be defined as a systematic and scientific characterization of potential adverse health consequences that have or can result from human exposure to agents and/or situations. It is the process by which the probability and severity of harm due to a chemical substance or product (i.e., combination of substances) is assessed and involves:
- issue identification
- hazard identification
- hazard characterization
- exposure assessment
- risk characterization
- risk classification
- risk management strategy
An HRA may be required in circumstances such as when there are safety concerns about a product and/or its advertising (e.g. products being promoted in such a way that it is used outside the recommended conditions of use [i.e., off-label use] and thus, could cause a serious adverse health consequence).
- Health Risk Classification:
- The risk classification is dependent on the seriousness of the adverse health consequence and the likelihood the event will occur. The numerical designation, assigned by the Health Products and Food Branch to a particular product to indicate the relative degree of health risk presented by the product, is as follows:
- TYPE I: A situation in which there is a reasonable probability that the use of or exposure to a product will cause the serious adverse health consequence(s) in question or death.
- TYPE IIA: A situation in which there is a reasonable probability that the use of or exposure to a product will cause the moderate adverse health consequence(s) in question or death.
- TYPE IIB: A situation in which there is a reasonable probability that the use of or exposure to a product will cause the mild adverse health consequence(s) in question or death.
- TYPE III: A situation in which there is a reasonable probability that the use of or exposure to a product will not cause the adverse health consequence(s) in question or death.
- Indication for Use:
- A statement that describes the limitations for use of a health product, including the disease state, condition(s) or symptom(s) and the target population, if specified, for which the health product is intended and authorized to be used by Health Canada. The indication for use is part of the Terms of Market Authorization, as identified in the Product Monograph (PM) accompanying the Notice of Compliance (NOC) or in the document that assigns a Drug Identification Number (DIN), a Natural Product Number (NPN) or a Drug Identification Number for Homeopathic Medicine (DIN-HM) and any related labelling material.
- Market Authorization Holder (MAH):
- The Market Authorization Holder (MAH) is also referred to as Sponsor or Manufacturer. The MAH is the legal entity that holds the Notice of Compliance, the Drug Identification Number (DIN), the Natural Product Number (NPN), the Homeopathic Medicine Number (DIN-HM), the medical device licence number, or that has received approval to initiate clinical trials in Canada.
- Marketed Health Products Directorate (MHPD):
- The Marketed Health Products Directorate (MHPD) works to assure that the Health Products and Food Branch (HPFB) programs take a consistent approach to post-approval safety surveillance, assessment of signals and safety trends and risk communications concerning all regulated marketed health products. Activities of MHPD include: monitoring and collecting adverse reaction and medication incident data; reviewing and analyzing marketed health product safety data; conducting risk/benefit assessments of marketed health products; communicating product related risks to healthcare professionals and the public; overview of regulatory advertising activities; providing policies to effectively regulate marketed health products; and active surveillance and drug effectiveness projects.
- Non-compliance:
- The state of non-conformity of a regulated party or a product with a legislative or regulatory requirement or a recognized standard.
- Precautionary Principle/Approach:
- A precautionary approach to decision-making emphasizes the need to take timely and appropriate preventative action, even in the absence of a full scientific demonstration of cause and effect.
- Preclearance:
- Process through which advertising materials are reviewed by the APAs prior to dissemination, to ensure that health product advertising, in all media, complies with the F&DA , CDSA, respective Regulations, Health Canada guidance documents, and the APA codes of advertising. Advertising approved by the APA is assigned an approval number and/or authorization to use the APA logo or seal/mark, which signifies that the advertising has been reviewed and, in the view of the APA involved, complies with the applicable legislation, regulations, guidance documents, and the APA codes.
- Risk:
- A measure of both the potential harm to human health that may result from being exposed to a product under specific conditions of use, together with the likelihood that the harm will occur.
- Terms of Market Authorization:
- The Terms of Market Authorization (TMA) are comprised of all labelling information (e.g., PM, prescribing information, inserts, etc.) that accompanies the NOC and/or in the document that assigns a DIN, NPN or DIN-HM and any related labelling material for health products. This information is derived from the review of information on the health product that is submitted for regulatory review and authorization, as required by the F&DA, and respective Regulations, (and as interpreted by guidance documents and policies).
- Unauthorized Product:
- This refers to a health product (e.g., drug, vaccine, natural health product) for which the TMA has not been established by Health Canada. The F&DAand associated Regulations contain provisions applicable to the advertising of unauthorized health products (Appendix B).
2 Advertising Preclearance Overview
Canadian APAs provide advertising material review services to advertisers and advertising agencies. They use Health Canada's guidance documents and their own codes of advertising to ensure that advertising material submitted to them, in all media, complies with the F&DA, CDSA, and respective Regulations. The APAs review advertising for consistency with the TMA and to verify that the advertising is accurate, balanced and evidence-based, and does not expand upon or conflict with the TMA. Advertisers should submit a copy of their advertising material to an APA for review prior to production to avoid costly changes to final executions. Advertising approved by the APA is assigned an approval number and/or authorization to use the APA logo or seal/mark, which signifies that the advertising has been reviewed and, in the view of the APA involved, complies with the applicable legislation, regulations, guidance documents, and the APA codes.
Canadian APAs are committed to work towards the integrity and accuracy of advertising through voluntary preclearance to achieve compliance with the F&DA, CDSA, respective Regulations, Health Canada guidance documents, and the APA codes of advertising.
Health Canada supports and encourages the use of a voluntary health product advertising preclearance system that supports adherence to federal legislative requirements.
During the preclearance review of a health product advertisement, or during the processing of an appeal or complaint, the APAs may request further clarification from Health Canada. Conversely, Health Canada may wish to bring an advertising issue to the attention of the APAs. These requests for clarification are described in this guidance document, along with complaint-handling procedures.
The APAs also offer independent complaint-resolution and appeal procedures, and administer related sanctions and remedial measures according to their internal policies, procedures and standards.
The APAs should monitor trends in health product advertising and promotion and adjust their codes and practices as required to fulfill their mandate.
The APAs may evaluate marketing claims such as comparative claims that are not included in the TMA, provided the data submitted by the advertising sponsor does not expand upon or conflict with the TMA and does not require submission of an application to Health Canada.
Some agencies also provide advisory opinions on messages directed to consumers for prescription drugs, Schedule D drugs (biologics, including vaccines) and on material discussing a medical condition/disease to ensure that they meet the regulatory requirements.
Additional information about the health product advertising preclearance system in place in Canada, including the list of Canadian APAs, is available on Health Canada Web site.
3 Guidance for Implementation
3.1 Role of Health Canada
3.1.1 National Regulatory Authority
Health Canada establishes the TMA of health products and is the national regulatory authority for health product advertising. To this effect, Health Canada sets standards for health product advertising material and:
- provides policies to effectively regulate marketed health products advertising,
- develops guidance documents that govern its interpretation of the regulatory framework to be incorporated in the APA's various codes of advertising, where applicable, and
- oversees the enforcement of the advertising prohibitions.
Health Canada uses the following approach in performing its roles:
- Transparency: Health Canada is responsible for promptly informing the APAs of any new standards, policies or guidance documents to ensure the accurate review and preclearance of advertising material.
- Risk-Based Enforcement: Health Canada uses a risk-based approach in its enforcement of federal legislation.
3.2 Roles of the Advertising Preclearance Agencies
The APAs review and preclear health product advertisements to determine, in their view, whether the advertisement complies with the provisions of the F&DA, the CDSA, respective Regulations, Health Canada's guidance documents, as well as with the agencies' own codes of advertising.
The List of Canadian Advertising Preclearance Agencies includes groups with distinct roles (Appendices C and D).
3.2.1 Preclearance of Nonprescription Drugs and Natural Health Products Advertising Directed to Consumers
An agency in this category has notified Health Canada that it has publicly self-attested to meeting Health Canada's recommended criteria for the preclearance of advertising material for non-prescription drugs including natural health products directed to consumers.
The services provided would generally include the review of advertising copy for radio, television, Internet, mass print (e.g., newspapers, magazines, flyers, point of purchase), social media, and out-of-home (e.g., billboards, transit) targeting consumers. Consultation service for new product launches and advertising concepts may also be offered in this category.
3.2.2 Preclearance of Health Product Advertising Directed to Healthcare Professionals
An agency in this category is recognized by Health Canada for preclearance of advertising material for all health products directed to healthcare professionals. The services provided would generally include the review of advertising pieces targeting healthcare professionals in all media such as print publications (e.g., medical journals, information brochures, pamphlets, and direct mail/detail aid materials), audio (e.g., radio), audio/visual (e.g., television), electronic means of communication (e.g., the Internet, including social media), as well as any novel media not yet contemplated. Consultation service for new product launches and advertising concepts may also be offered in this category.
3.2.3 Advisory Opinions on Direct-to-Consumer Advertisement of Prescription Drugs, Schedule D Drugs and Disease Awareness Campaigns
An agency in this category provides advisory opinions on messages directed to consumers for prescription drugs (e.g., reminder ads), material discussing a medical condition or disease (e.g., help seeking messages), Schedule D drug (biologics, including vaccines) advertisements, and other messages or materials (e.g., clinical trial recruitment announcements, press releases, institutional messages, etc.). Agencies in this category are required to forward copies of advisory opinions to Health Canada for information purposes. Advisory opinions are assessments as to whether materials, based on Health Canada's policy The Distinction between Advertising and Other Activities, are promotional, in which case they must be in accordance with the F&DAandRegulations, CDSA, and associated policy and guidance documents. Consultation service for new product launches and advertising concepts may also be offered in this category.
Note: An agency can provide services that are categorized in more than one of the above categories/groups.
3.3 Role of the Market Authorization Holder (MAH)
The MAH, or sponsor, has a responsibility to develop advertising material that is compliant with the F&DA and associated regulations, to cooperate with complaint resolution procedures, and to discontinue or amend non-compliant advertising material in order to bring it to compliance.
3.4 Requests for Clarifications
The request for clarification process between The APAs and Health Canada shall be as follows:
3.4.1 APAs Requesting Clarifications from Health Canada
When there is a question or concern about a health product advertising or TMA, the enquiry should be directed to Health Canada in writing, setting out:
- the type of issue (see 3.4.1.1),
- the APA's interpretation and analysis of the issue, and
- the decision of the APA on the issue.
The following information should also be provided where applicable:
- the sponsor's interpretation of the issue, and
- the decision of the APA's complaint resolution panel.
The dialogue will be between the APA and Health Canada. The advertising sponsor will be apprised of the decision through the APA.
Health Canada will make every effort to conduct a preliminary review of the request for clarification and issue an acknowledgement letter to the APA within 15 business days. If a resolution has not been achieved within this period, the APAs will be provided with an estimated time frame for resolution and will be informed of the reason for delay. Requests that are brought to Health Canada's attention are often complex and may require several internal consultations within the department. For example, if it is necessary to consult with a product-specific directorate of Health Canada, it may take up to 30 days to provide a response. Health Canada will strive to provide the APA with a final assessment in a timely manner.
3.4.1.1 Types of Issues
Clarification of Terms of Market Authorization
Issues raised with Health Canada include clarification of the advertiser's interpretation of the TMA, and any claims or statements being made which would necessitate a change to the TMA (such as changes requiring a Supplementary New Drug Submission (Division 8, Part C of the F&DR), a DIN submission (Division 1, Part C of the F&DR), or a Notifiable Change Submission).
Health Canada should not be consulted on other issues (such as therapeutic comparative claims), unless the contentious claim conflicts with, relates to, or expands upon the TMA, or if the APAs require clarification as to whether the claim is consistent with the TMA.
Regulatory/policy interpretations and emerging advertising trends
The APAs may consult Health Canada for advice on application of federal regulatory requirements and emerging advertising trends. Such issues may be posed in writing or discussed at the APA bilateral meetings with Health Canada.
3.4.2 Health Canada Requesting Clarifications from the APAs
When Health Canada has a concern about the review of an advertising piece or campaign, it will bring this matter to the attention of the APA providing the review and will identify:
- the issue,
- Health Canada's interpretation and analysis of the issue, and
- suggestions for addressing the issue.
The dialogue will be between Health Canada and the APA. If applicable, the advertising sponsor will be apprised of the decision through the APA.
The APA will make every effort to address the request for clarification and provide Health Canada with a written response and account of the actions taken, where applicable, within 15 business days.
3.5 Complaint Adjudication
The first route for advertising complaints adjudication of all Health Canada authorized health products is through the APAs, except as specified below. The APAs will process complaints pursuant to the provisions and time frames expressed in their internal complaint resolution procedures.
The following types of advertising complaints should be reported directly to Health Canada, bypassing the APAs:
- Complaints related to direct-to-consumer-advertising (DTCA) of prescription drugs and Schedule D drugs (biologics, including vaccines) should be submitted to the Regulatory Advertising and Risk Communications Section of MHPD.
- Complaints related to advertising of unauthorized health products should be submitted to the Health Products and Food Branch Inspectorate (HPFBI) for compliance verification. HPFBI is the branch of Health Canada responsible for compliance and enforcement activities relating to contraventions of the advertising provisions of the F&DA, CDSA and respective Regulations
The procedures for handling complaints received by Canadian APAs and Health Canada shall be as follows (Appendix E):
3.5.1 Complaints submitted to APAs
3.5.1.1 Referrals between APAs
Complaints submitted to the APAs should be referred as follows:
- Advertising of non-prescription drugs including natural health products to consumers should be submitted to the APAs that have publicly self-attested to meeting Health Canada's recommended criteria.
- Advertising of prescription drugs and other health products to healthcare professionals should be submitted to an agency recognized by Health Canada for preclearance of advertising material for all health products directed to healthcare professionals.
3.5.1.2 Referrals to Health Canada
The APAs are expected to refer to Health Canada (MHPD serves as point of contact):
- any complaints that relate to advertising which, in the APA's judgement, contravenes the F&DA, the CDSA, and respective Regulations and is suspected to present an imminent and/or significant health risk;
- any complaints that relate to advertising which, in the APA's judgement, contravenes the F&DA, the CDSA, and respective Regulations, and for which the agency has been unable to achieve compliance, due to wilful nonparticipation or non-compliance with the standards and procedures of the APA;
- advertising complaints related to health products unauthorized for sale in Canada; or
- advertising complaints related to prescription drugs (Schedule F drugs) and Schedule D drugs (biologics, including vaccines) directed to the general public.
3.5.2 Complaints submitted to Health Canada
For all complaints received, whether referred or directly submitted, MHPD determines non-compliance (by evaluating the advertisement against the requirements of the Acts and their Regulations) and assesses the health risk posed by the advertising material at issue. The outcome of the risk assessment is the determination of the level of health risk, which could increase or decrease at any time, given new information surrounding the circumstances of the non-compliance.
3.5.2.1 Conducting a health risk assessment
The MHPD will perform a rudimentary health risk assessment to determine the level of risk to human health posed by exposure to the advertising, prior to taking risk management actions. This will involve the use of a Risk Matrix below.
| Seriousness of Health Consequences | ||||
|---|---|---|---|---|
| Low | Medium | High | ||
| Probability/Likelihood of occurrence of Health Consequences | High | High | ||
| Medium | Medium | |||
| Low | Low | |||
Since the determination of the level of risk is based on evidence available to date, and given that the adverse health consequence and its seriousness is most often "unknown", the MHPD would utilize the precautionary principle/approach in risk assessment and management. When the rudimentary risk assessment suggests a "medium" or "high" level of health risk, an HRA may be requested from the applicable product-specific directorate.
For the purpose of the rudimentary risk assessment performed at the MHPD:
High risk: High probability/likelihood of the health consequence occurring with a high seriousness ( symptoms are life threatening or disabling).
Medium risk: Medium probability/likelihood of the health consequence occurring with a medium seriousness ( symptoms are mild and non-life threatening. Usually some form of treatment is indicated. No residual disability following treatment).
Low risk: Low probability/likelihood of the health consequence occurring with a low seriousness ( symptoms are minimally bothersome, no therapy is necessary).
3.5.2.2 Risk Management
Once non-compliance and the health risk level of an advertisement is determined, immediate risk management actions may be taken by Health Canada and may include but is not limited to:
- warning letter to the advertising sponsor and/or broadcaster,
- requesting immediate cessation of the advertisement,
- contacting and/or referral to the APAs,
- issuance of a risk communication,
- suspension or cancellation of marketing authorisation/product licence,
- prosecution.
Risk management actions can be taken alone or in combination, and sequentially or simultaneously. Further information on regulatory measures available to Health Canada in order to achieve compliance by regulated parties are described in Health Canada's Compliance and Enforcement Policy (POL-0001)
3.5.2.3 Complaints submitted directly to Health Canada by the Complainant in error
Complaints pertaining to advertising for authorized health products are sometimes submitted in error directly to Health Canada by the complainant. These will first be assessed for non-compliance with the Acts and Regulations and the level of health risk they pose, as described in section 3.5.2, and then handled as follows:
- When no health risk or a low level of risk is determined, Health Canada will forward the complaint to an APA according to their preclearance role (e.g., the APAs which preclear only consumer-directed materials should not adjudicate complaints for advertising to healthcare professionals. Refer to section 3.2 for the roles of the APAs). Health Canada will seek consent from the complainant to disclose their identity to the APA and will ask for their preference regarding an APA. Health Canada will subsequently forward the complaint with a summary of the findings (if applicable) to that agency for further processing according to the time frames and procedures expressed in the agency's internal complaint resolution processes.
- When a high or medium level of health risk is determined, Health Canada will take the appropriate risk management approach which may involve a request for immediate cessation of the advertising from the sponsor and simultaneously informing the APAs of the actions taken. Other possible actions are listed in section 3.5.2.2.
3.5.2.4 Complaints referred to Health Canada by the APAs
Complaints referred to Health Canada by the APAs will first be assessed for non-compliance with the Acts and Regulations and the level of health risk they pose, as described in section 3.5.2, and then handled as follows:
- Contravention of the F&DA, the CDSA, respective Regulations, and a suspected health risk: this pertains to an advertising complaint which, in the APA's judgement, contravenes the F&DA, CDSA, respective Regulations, and may present a health risk. Where an advertisement is found to contravene the legislation and pose a low or no health risk, the complaint will be returned to the APA for further processing. Where a medium to high health risk is determined, Health Canada will take direct action with the sponsor and simultaneously inform the relevant APA of that action.
- Contravention of the F&DA, CDSA, and respective Regulations without a resolution by the APA: this pertains to an advertising complaint which, in the APA's judgement, may not present a health risk but contravenes the F&DA, CDSA, and respective Regulations, and for which it has been unable to come to a resolution due to wilful non-participation in, or non-compliance with its standards and procedures. When Health Canada is of the view that a contravention has occurred, Health Canada will take direct action corresponding to the assessed level of risk with the sponsor. Health Canada will simultaneously inform the relevant APA of that action.
- Advertising of unauthorized health products: When a complaint is pertaining to advertising for health products unauthorized for sale in Canada, Health Canada will first confirm the health product's status. Advertising of unauthorized health products is considered to pose a health risk and is processed by Health Canada's HPFBI for compliance verification.
The HPFBI utilizes a risk-based approach to compliance and enforcement and has a set of regulatory measures in its disposal in order to achieve compliance by regulated parties, as described in Health Canada's Compliance and Enforcement Policy (POL-0001)
Once the compliance verification is completed, the Inspectorate will notify MHPD and the relevant APA of the outcome.
- DTCA of prescription drugs and Schedule D drugs: When a complaint pertains to the advertising of Schedule F drugs (prescription drugs) or Schedule D drugs (biologics, including vaccines) directed to the general public, Health Canada will determine whether the advertising material is in accordance with the F&DA, CDSA, respective Regulations, and related guidance documents. Where Health Canada is of the view that there is a contravention, Health Canada will notify the sponsor of the non-compliance and request immediate corrective actions. Should the non-compliance continue, the complaint shall be forwarded to the HPFBI for further compliance verification. Once the compliance verification is completed, HPFBI shall notify MHPD and the APA.
3.5.2.5 Health Canada involvement in APA complaint and appeal processes
Health Canada will not otherwise become involved in the APAs complaint and appeal processes.
3.5.2.6 The Food and Drugs Act Liaison Office
The Food and Drugs Act Liaison Office (FDALO) provides an impartial and confidential resource for individuals, businesses and organizations when they experience problems with how Health Canada administers the F&DA. It acts as an intermediary to help the parties arrive at a mutually agreed upon resolution. Complainants or advertisers who are not satisfied after having first attempted to resolve their issues through mechanisms described above, may wish to take advantage of the conflict resolution services offered by the FDALO.
Appendix A: List of Abbreviations
- APA
- Advertising Preclearance Agencies
- ASC
- Advertising Standards Canada
- CDSA
- Controlled Drugs and Substances Act
- DIN
- Drug Identification Number
- DIN-HM
- Drug Identification Number for Homeopathic Medicine
- DTCA
- Direct-To-Consumer-Advertising
- F&DA
- Food and Drugs Act
- FDALO
- Food and Drugs Act Liaison Office
- F&DR
- Food and Drug Regulations
- HPFB
- Health Products and Food Branch
- HPFBI
- Health Products and Food Branch Inspectorate
- HRA
- Health Risk Assessment
- MAH
- Market Authorization Holder
- MHPD
- Marketed Health Products Directorate
- MIJO
- Formerly Broadcast Clearance Advisory (MIJO is an acronym of the company founders respective names)
- NHP
- Natural Health Product
- NHPD
- Natural Health Products Directorate
- NHPR
- Natural Health Products Regulations
- NHP-UPLAR
- Natural Health Products (Unprocessed Product Licence Applications) Regulations
- NOC
- Notice of Compliance
- NPN
- Natural Product Number
- PAAB
- Pharmaceutical Advertising Advisory Board
- PM
- Product Monograph
- Rx Drug
- Prescription Drug
- TMA
- Terms of Market Authorization
Appendix B: Legislative and Regulatory Framework
1. Sections of the Food and Drugs Act:
Section 3(1): No person shall advertise any food, drug, cosmetic or device to the general public as a treatment, preventative or cure for any of the diseases, disorders or abnormal physical states referred to in Schedule A.
Exemption: Refer to Sections A.01.067 and A.01.068 of the F&DR, and Sections 103.2 and 103.3 of the Natural Health Products Regulations (NHPR), which exempt natural health products (NHPs) and non-prescription drugs from the F&DA's Section 3 general prohibition on labelling and advertising of preventative claims for Schedule A diseases. Therefore, as of June 1, 2008, authorized claims for the prevention of Schedule A diseases may appear on the labels of the NHPs and non-prescription drugs. Claims on product labels are to match the authorized wording. Claims used in advertising may deviate in their wording as long as the claims are consistent with the product's TMA and do not directly or indirectly exceed the scope of the TMA.
Section 9(1): No person shall label, package, treat, process, sell or advertise any drug in a manner that is false, misleading or deceptive or is likely to create an erroneous impression regarding its character, value, quantity, composition, merit or safety.
In order to make informed decisions about their health, consumers should be provided with fair and balanced information about the benefits and the risks associated with the use of advertised health products. Examples of contraventions to Section 9(1) include:
- omitting or downplaying risks,
- overstating the effectiveness,
- promoting unauthorized health products or indications (off-label): promotion of a health product prior to market authorization is not permitted because the terms of such authorization have not been established and the proposed indication(s) for use have not been verified,
- misleading drug comparisons
Section 14(1): No person shall distribute or cause to be distributed any drug as a sample.
Exception: (2) Subsection (1) does not apply to the distribution, under prescribed conditions, of samples of drugs to physicians, dentists, veterinary surgeons or pharmacists.
2. Sections of the Food and Drug Regulations:
Section A.01.067: A drug is exempt from subsection 3(1) of the Act with respect to its advertisement to the general public as a preventative, but not as a treatment or cure, for any of the diseases, disorders or abnormal physical states referred to in Schedule A to the Act.
Section A.01.068: A drug is exempt from subsection 3(2) of the Act with respect to its sale by a person where the drug is represented by label or is advertised by that person to the general public as a preventative, but not as a treatment or cure, for any of the diseases, disorders or abnormal physical states referred to in Schedule A to the Act.
Section C.01.007: No reference, direct or indirect, to the Act or to these Regulations shall be made upon any label of or in any advertisement for a drug unless such reference is a specific requirement of the Act or these Regulations.
Section C.01.044: Where a person advertises to the general public a Schedule F Drug (prescription drugs), the person shall not make any representation other than with respect to the brand name, proper name, common name, price and quantity of the drug.
Section C.08.002: No person shall sell or advertise a new drug unless
- the manufacturer of the new drug has filed with the Minister a new drug submission or an abbreviated new drug submission relating to the new drug that is satisfactory to the Minister;
- the Minister has issued, pursuant to section C.08.004, a notice of compliance to the manufacturer of the new drug in respect of the new drug submission or abbreviated new drug submission;
- the notice of compliance in respect of the submission has not been suspended pursuant to section C.08.006; and
- the manufacturer of the new drug has submitted to the Minister specimens of the final version of any labels, including package inserts, product brochures and file cards, intended for use in connection with that new drug, and a statement setting out the proposed date on which those labels will first be used.
Section G.01.007 No person shall
- advertise a controlled drug to the general public; or
- issue or publish any other written advertisement respecting a controlled drug unless that advertisement carries the symbol in a clear and conspicuous colour and size in the upper left quarter of the first page of the advertisement.
3. Sections of the Narcotic Control Regulations:
Section 70: No person shall
- publish or cause to be published or furnish any advertisement respecting a narcotic unless the symbol "N" is clearly and conspicuously displayed in the upper left-hand quarter thereof or, if the advertisement consists of more than one page, on the first page thereof;
- publish or cause to be published or furnish any advertisement to the general public respecting a narcotic; or
- advertise in a pharmacy a preparation referred to in section 36.
4. Sections of the Natural Health Products Regulations:
Section 92: No reference, direct or indirect, to the Act, the Food and Drug Regulations or to these Regulations shall be made on any label of or in any advertisement for a natural health product unless the reference is specifically required by law.
Section 103.2: A natural health product is exempt from subsection 3(1) of the Act with respect to its advertisement to the general public as a preventative, but not as a treatment or cure, for any of the diseases, disorders or abnormal physical states referred to in Schedule A to the Act.
Section 103.3: A natural health product is exempt from subsection 3(2) of the Act with respect to its sale by a person where the drug is represented by label or is advertised by that person to the general public as a preventative, but not as a treatment or cure, for any of the diseases, disorders or abnormal physical states referred to in Schedule A to the Act.
5. The Natural Health Products (Unprocessed Product Licence Applications) Regulations
The Natural Health Products (Unprocessed Product Licence Applications) Regulations (NHP-UPLAR) promulgated on August 4, 2010, allow for the legal sale of a category of natural health products for which Health Canada has not yet issued a product licence but has completed an initial assessment to ensure that information supporting the safety, quality and efficacy of the product has been provided, and that specific safety criteria have been met.
The Guidance Document Health Canada and Advertising Preclearance Agencies' Roles Related to Health Product Advertising does not address exempt NHPs. Please refer to the Natural Health Products Compliance and Enforcement Policy (POL-0044) which is available on the Compliance and Enforcement section of Health Canada's Web site.
Health Canada has developed a position statement that applies to exempt NHPs in relation to preclearance and complaint adjudication of advertising materials: Health Canada's Position Statement on the Preclearance and Complaint Adjudication of Exempted Natural Health Product Advertising Materials.
Appendix C Canadian Advertising Preclearance Agencies
- Preclearance of Non-prescription drugs and Natural Health Products Advertising Directed to Consumers
- Advertising Standards Canada (ASC)
- MIJO (formerly Broadcast Clearance Advisory)
- Preclearance of Health Product Advertising directed to Healthcare Professionals
- Pharmaceutical Advertising Advisory Board (PAAB)
- Advisory opinions on Direct-to-Consumer Advertisement of Prescription drugs, Schedule D drugs and disease awareness campaigns
- Advertising Standards Canada (ASC)
- Pharmaceutical Advertising Advisory Board (PAAB)
Appendix D Roles of Advertising Preclearance Agencies
| Advertising Preclearance Agency | Review Advertising for: | Target Audience |
|---|---|---|
| Pharmaceutical Advertising Advisory Board (PAAB) | Rx drugs Schedule D drugs (Biologics, including vaccines) Non-Rx drugs NHPs |
Healthcare professionals |
| Advertising Standards Canada (ASC) |
Non-Rx drugs NHPs |
Consumers |
| MIJO (formerly Broadcast Clearance Advisory) |
Non-Rx drugs NHPs |
Consumers |
| Advertising Preclearance Agency | Advisory Opinions (Consumer-directed: Rx & Schedule D) | Target Audience |
|---|---|---|
| Pharmaceutical Advertising Advisory Board (PAAB) | Medical condition / disease Rx drugs Schedule D drugs (including vaccines) |
Consumers |
| Advertising Standards Canada (ASC) |
Medical condition / disease Rx drugs Schedule D drugs (including vaccines) |
Consumers |
Appendix E Advertising Complaint Adjudication
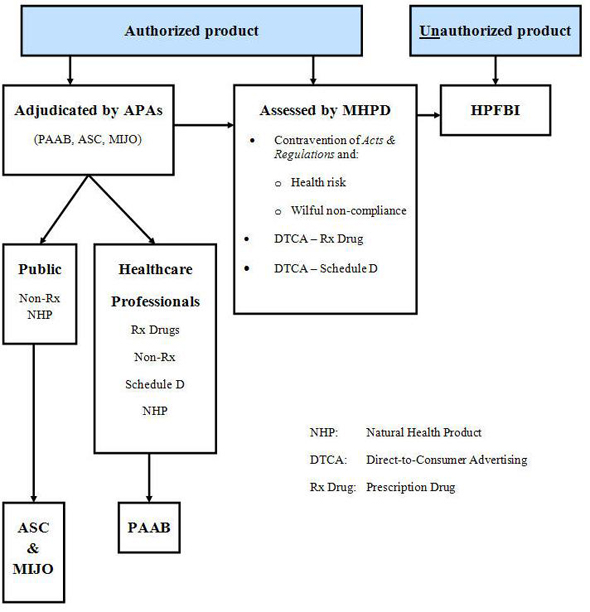
Text description
Authorized product:
- Adjudicated by APAs (PAAB, ASC, MIJO)
- Public: Non-Rx, NHP
- ASC & MIJO
- Healthcare Professionals: Rx Drugs, Non-Rx, Schedule D, NHP
- PAAB
- Public: Non-Rx, NHP
- Assessed by HMPD
- Contravention of Acts and Regulations and:
- Health risk
- Wilful non-compliance
- DTCA-Rx Drug
- DTCA-Schedule D
- Contravention of Acts and Regulations and:
- PAAB
Unauthorized product:
- HPFBI