Food and nutrition highlights 2021 : Helping you maintain and improve your health
Download in PDF format
(7.36 MB, 42 pages)
Organization: Health Canada
Published: 2022-06-06
Cat: H161-12E-PDF
ISSN: 2563-3376
On this page
- Welcome to Health Canada's annual highlights report
- Food and nutrition: 2021 in brief
- Ensuring the safety of Canada's food supply
- Improving the nutritional quality of food
- Regulatory innovation
- Promoting healthy eating
- Canada's food guide
- Additional healthy eating initiatives
- International collaboration
- Annex A: Approvals
- Healthy clicks: Food and nutrition at a glance
Welcome to Health Canada's annual highlights report

Assistant Deputy Minister

Associate Assistant Deputy Minister
As the COVID-19 pandemic continued through 2021, Health Canada worked to support the people of Canada with food and nutrition information, analysis and regulation to keep them safe and healthy.
In a time of increased stress, a safe, healthy and nutritious diet is more important than ever. The pandemic highlighted the importance of creating environments that support healthy eating to help decrease chronic diseases rates, as identified in our Healthy Eating Strategy. In 2021, we continued our work to promote Canada's food guide and developed additional materials to make it accessible and useful for people of different ages and backgrounds. It continues to be one of our most popular pieces of guidance.
Canada has one of the safest food supplies in the world, supported by the hard work of our food safety scientists. In 2021, we conducted scientific research and advanced key regulatory modernization initiatives to respond to innovation and new technologies while maintaining the safety of the food we eat.
This report will provide you with an overview of our work in 2021 to support healthy eating, food safety, and innovation.
Food and nutrition: 2021 in brief

Director General,
Office of Nutrition Policy and Promotion

Director General,
Food Directorate
Health Canada strives to create a supportive food environment for people in Canada. We do this by promoting healthy eating, assessing the health risks and benefits of food, and by developing policies, regulations, and guidelines as well as providing trusted information for the safety and nutritional quality of all food sold in Canada.
In 2021, our scientists conducted research on foodborne pathogens such as Salmonella, Listeria, Norovirus, parasites and E. coli to inform our policies and guidance on the safe handling and preparation of foods. We also studied different bacteria and parasites to better understand their presence in our food supply. An updated risk assessment for caffeinated energy drinks was undertaken to determine their risk to human health, and ultimately supported the risk management measures currently in place for these products. To ensure that dietary exposure to food contaminants is as low as possible, we proposed new and updated maximum levels for inorganic arsenic in foods that are significant primary sources of exposure in infants and children in Canada. To help people in Canada increase their vitamin D intake, a marketing authorization came into force in December 2021, which permits manufacturers to increase vitamin D levels in cow's milk, goat's milk and margarine. Vitamin D helps our bodies absorb calcium, and build and maintain strong bones and teeth.
Health Canada's healthy eating strategy, which includes Canada's food guide, aims to make it easier for people in Canada to make healthier food choices. In 2021, to support the updated Canada's food guide, published in 2019, we formed youth groups to engage and promote healthy eating among their peers. In parallel, we launched a marketing campaign to encourage families with children to make healthier eating choices. We also continued work on tools to assess adherence to the food guide and developed a food literacy questionnaire to measure people's food and nutrition knowledge, skills, attitudes and behaviours.
International collaboration helps us to deliver on our mandate. In 2021, Health Canada worked closely with a range of international partners on food safety and nutrition to establish global standards and share scientific information. For example, we hosted the 46th Session of the Codex Committee on Food Labelling from September 27 to October 1, 2021, attended by more than 600 representatives from more than 75 countries. This session of the committee focused on discussing food labelling and in particular, labeling of allergens, front-of pack labelling, e-commerce and the use-of technology in labelling. We also participated in a workshop in collaboration with the Government of India to help educate food exporters about Canada's food allergen labelling regulations, and in another series of workshops to develop an Asia Pacific Economic Cooperation (APEC) Food Safety Risk Communication Framework and Supporting Guidelines.
In 2021, we continued to make progress to ensure food safety and promote healthy eating choices at a time where protecting people's health is more important than ever. Unhealthy diets increase the risk of obesity, type 2 diabetes, cardiovascular diseases and these conditions are risk factors for COVID-19 severity and mortality in adults.
While responding to the COVID-19 pandemic in 2021, Health Canada continued to ensure that people in Canada had access to safe and nutritious food. We established policies, regulations, guidelines and standards related to the safety and nutritional quality of all food sold in Canada. This work was done in consultation with other parts of the federal government, provincial and territorial governments, consumers, academia, experts and industry.
Ensuring the safety of Canada's food supply
Health Canada's role is to protect people in Canada from risks to health and the spread of disease. We achieve this by using both preventative and responsive methods including:
- conducting health-risk assessments in support of food safety investigations;
- providing information about food safety;
- undertaking scientific research to identify, characterize and respond to new and emerging food safety risks;
- setting food safety standards and addressing emerging risks; and
- conducting food monitoring and surveillance.
The Canadian Food Inspection Agency plays an equally important role in monitoring compliance with the regulations and taking enforcement action such as food recalls.
People in Canada are also important players in maintaining an effective food safety system. There are safe food practices we can all follow including:
- washing our hands with soap and water for at least 20 seconds, before and after handling food and food packaging;
- following food handling & cooking instructions;
- properly storing food; and
- reporting a food safety or labelling concern.
Focus on…
Enhancing outreach and educations activities
In 2021, a new food safety marketing campaign was launched. Several videos were released to encourage parents of kids under 13 to practice and integrate food safety behaviours as norms into everyday life, and to provide easy and simple actions to handle and prepare food safely (using the four core food safety practices clean, separate, cook, chill).
Assessing the safety of new products or product uses
If a company plans to sell a novel food, a new infant formula, or a food containing a food additive used in a new way, they need approval from Health Canada. In 2021, 10 novel foods, 20 food additives and 7 infant formulas were approved (see Annex A).
For example, in 2021, Health Canada assessed the safety of a new preservative, modified vinegar. After conducting a rigorous scientific safety assessment and consultations, Health Canada collaborated with the Canadian Food Inspection Agency to amend the List of Permitted Preservatives to allow modified vinegar to be used as a preservative in some meat and poultry products. Modified vinegar is an alternative to other preservatives, which are already permitted in certain meat and poultry products. Unlike common vinegar, which is acidic, modified vinegar is manufactured to be less acidic and, therefore, less likely to affect the taste or texture of foods. The change became effective on November 8, 2021.
Considering that novel foods, food additives and infant formula are consumed by people in Canada after approval, a product is not approved for sale if it is found to be unsafe. It is thus crucial that their safety be assessed before they reach the public in Canada.
Setting safety standards and developing science-based policies
Food contamination occurs when food contains something that makes it unsafe to eat. Contaminants can include bacteria, pests and chemicals. The Government of Canada's Chemicals Management Plan aims to reduce the risks posed by chemicals. As part of this plan, Health Canada and the Canadian Food Inspection Agency conduct regular surveillance of the levels of certain contaminants in the Canadian food supply. Health Canada also establishes maximum levels for contaminants to minimize dietary exposure to food contaminants.
For example, we recently published food contaminant proposals to add a maximum level for inorganic arsenic in rice-based foods for infants and children and to lower the maximum level for total arsenic in fruit juice and fruit nectar. Health Canada will be working over the next year to consider comments received during the consultation and to publish any changes. We also updated the arsenic in food webpage, providing more information to people in Canada on steps they can take to reduce exposure. Canadian maximum levels for chemical contaminants in foods are set out in the List of Contaminants and Other Adulterating Substances in Foods, as well the List of Maximum Levels for Various Chemical Contaminants in Foods.
Conducting risk assessments
Health Canada conducts food-related health risk assessments to determine if the presence of a certain substance or microorganism in food (for example, a chemical contaminant, allergen, bacteria, virus or parasite) poses a health risk to consumers. If we find that a substance or microorganism in food poses a human health risk, risk management actions are taken to reduce, and if possible, eliminate the risk. When requested by the food industry, Health Canada conducts safety assessments of food packaging materials and incidental additives that have the potential to leave chemical residues in food or substances used in the production of food. In 2021, Health Canada issued 668 of these opinions. Maintaining transparency is key to this assessment process and Health Canada has previously published detailed information regarding this process for stakeholders.
In 2021, Health Canada conducted many risk assessments:
- In May 2021, the Canadian Food Inspection Agency recalled 15 products that exceeded Health Canada's maximum level for cyanide in apricot kernels. Health Canada provided critical support to the Canadian Food Inspection Agency to provide a single health risk assessment that captured all the recalled products.
- In May 2021, Health Canada published a risk assessment on Escherichia coli O157 in Gouda cheese made from raw milk in Canada. Gouda cheese made with raw milk was implicated as the source of Escherichia coli (E. coli) O157 outbreaks in 2002 and 2013 in Canada, causing 41 illnesses and 1 death. Following the 2013 outbreak, Health Canada conducted a risk assessment on E. coli O157 in Gouda cheese made from raw milk in Canada. The risk assessment evaluates the potential for E. coli O157 to be present and multiply in Gouda cheese made from raw milk produced in a farmstead type environment in Canada. It identifies points in the cheese making process where interventions may mitigate potential bacterial contaminations in Gouda cheese from raw milk, and it assesses the risk of illness caused by E. coli O157 due to the consumption of this type of cheese. The risk assessment published in 2021 identified options, which could be effective in reducing the risk of illness caused by E. coli O157 in Gouda cheese made from raw milk, and used to inform future decision-making.
- In June 2021, partners within the Government of Canada and the Government of the United States published a paper on the risk assessment of Norovirus illness linked to the consumption of raw oysters in the United States and in Canada in the journal Risk Analysis. This work is important to help regulators understand the impacts of preventative control practices in the shellfish industry, ensuring that our regulatory guidance effectively supports the production of oysters that are safe to eat.
- In July 2021, Health Canada issued a request for information and data from stakeholders to support the risk analysis of Shiga toxin-producing Escherichia coli, a type of E. coli that is an important cause of foodborne illness. The data collected will inform our analysis of the current food safety approach as it relates to Shiga toxin-producing Escherichia coli, support future advice to the public, industry and regulatory authorities, and assist in continuing to protect people in Canada from foodborne illnesses.
- In September 2021, Health Canada updated a health risk assessment for caffeinated energy drinks. The scientific article titled Caffeinated energy drinks in the Canadian context: health risk assessment with a focus on cardiovascular effects has been published and confirms the risk management measures currently in place for these products is health protective for consumers.
Focus on…
Acrylamide in food
Acrylamide is a chemical that can form from naturally present compounds in starchy foods during high-temperature processing or cooking (for example, baking, frying, roasting). In 2021, Health Canada updated the online information page on acrylamide in food, with updated advice for consumers on how to reduce exposure to acrylamide from foods as well as to provide resources to the food industry in support of best practices in food processing.
Food safety research
Health Canada conducts and invests in research to support strategic management of Canada's food system and the promotion of food safety, nutrition and healthy eating in Canada. Research and related scientific activities play an important role in many decision-making processes, and contribute to the development of regulations, policies and programs and the delivery of services and information. Research and surveillance activities at Health Canada are conducted to address knowledge gaps and provide essential scientific evidence to inform food programme activities in different fields such as food chemistry, microbiology and nutrition. Areas of focus for research and surveillance evolve over time to remain responsive to the ever-changing food and health environment in Canada. Highlights of key areas we are currently working on are provided in the following paragraphs.
Listeria
Listeria monocytogenes bacteria are commonly found in the environment, can contaminate food, especially ready-to-eat foods, potentially causing listeriosis. Listeriosis is an infection that mainly affects pregnant women, newborns, adults 65 years and older, as well as people with compromised immune systems. Symptoms of infection include fever, muscle aches and headaches.
The Listeriosis Reference Service is developing new methods for detecting Listeria species in foods, and in 2021, it helped Health Canada guide activities related to the control of Listeria in high-risk foods and supported the food industry in addressing environmental verification and monitoring.
Vibrio
Vibrios are toxin-producing bacteria found naturally in water, fish and shellfish. People infected with disease-causing Vibrio may develop foodborne illness and symptoms such as diarrhea. Health Canada's Vibrio research laboratory pursues surveillance work on seafood to detect and characterize Vibrio species. The diversity and characteristics of these bacteria species are used to determine the potential risks associated with climate change and international trade on seafood consumers. For example, the research laboratory monitors the antimicrobial resistance patterns of several species of Vibrio isolated from Canadian seafood. In 2021, 62 seafood samples were analysed for Vibrio, with results indicating that most samples are antimicrobial resistant. Further research is needed to better understand the impact of these Vibrio species on human health.
Toxoplasma gondii
The Parasitology laboratory has been examining the presence and transmission patterns of the food and waterborne parasite, Toxoplasma gondii, in wildlife harvested as country foods in Canada's North. The human blood levels of toxoplasmosis are considerably higher in the North than in the rest of Canada, likely due to the consumption of infected wildlife (e.g., beluga, narwhal, seals, fish) following traditional methods of food preparation (i.e., raw, dried, fermented). Human infections may result in serious complications, especially in immunosuppressed individuals. The data generated in this multi-year project will address the significant knowledge gaps regarding food safety and security in Canada's North. Furthermore, studies on the impact of climate change on the prevalence of this bacteria will be pursued in the future.
Salmonella
The Salmonella lab pursues research to understand how Salmonella adapts to grow and survive on foods. By studying the properties of foods implicated in outbreaks, such as profiteroles in 2021, and how Salmonella responds to various storage and cooking conditions, we are able to inform guidance on the safe handling and preparation of foods to prevent future outbreaks from occurring.
Food surveillance and toxicology research
The Canadian Total Diet Study is a food surveillance program that allows Health Canada to monitor the concentration of chemical contaminants in foods that are typically consumed by people in Canada. Since its inception in 1969, the Canadian Total Diet Study has enabled Health Canada to monitor the concentration of contaminants in food sold in Canada in order to determine the trends, support the development of food safety policies and regulations, and help ensure the safety of Canada's food supply. This study allows the measurement of trends not only over time, but also over regions as it rotates through cities across Canada. Overall, the Total Diet Study program provides data on levels of chemical contaminants in foods and on emerging food-borne chemicals. In 2021, the Department published 18 new datasets from Canada's Total Diet Study and other targeted surveillance data on contaminants in food (e.g. total Mercury, BPA in liquid infant formula products) to the Government of Canada's Open Data Portal.
The toxicology research program serves to identify, characterize and interpret food-based health hazards from chemicals or food additives. For example, in 2021, our researchers have been advising on Health Canada's interpretation of an international safety re-evaluation of titanium dioxide use as a food additive coloring agent after uncertainties and data gaps were identified by international food safety entities. In Canada, titanium dioxide can be used for various foods such as confectionaries, baked goods and condiments.
Focus on…
Information for people with a crustacean allergy
In 2021, Health Canada published information for people with a crustacean allergy to alert them that they may have an allergic reaction if they eat edible insects like crickets and mealworms.
Improving the nutritional quality of food
In addition to maintaining the safety of Canada's food supply, Health Canada is responsible for establishing policies and providing advice to improve the nutritional quality of food with the aim of promoting nutritional health and well-being.
Vitamin D fortification
Vitamin D is an essential nutrient for bone health, and many other functions in the human body. Vitamin D occurs naturally in very few foods, namely eggs and fatty fish. Most dietary vitamin D is from fortified foods, such as milks, fortified plant-based beverages and margarine.
Health Canada recognizes that it is challenging to consume the recommended amounts of vitamin D through the current food supply. To help improve the vitamin D intakes and bone health of people living in Canada, we have been working to expand vitamin D fortification of foods. As a first step, in December 2021, a marketing authorization came into force to permit increased vitamin D levels in cow's milk, goat's milk and margarine. As a next step, we plan to increase vitamin D levels of fortified plant-based beverages and permit the vitamin D fortification of yogurt.
Over the coming years, Health Canada will monitor the impact of these changes on the vitamin D status of people in Canada through the Canadian Health Measures Survey. If necessary, we will consider allowing the addition of vitamin D to more foods in the future.
Monitoring the food environment
In September 2018, the prohibition of partially hydrogenated oil (PHO) came into effect to reduce industrially produced trans fats in the food supply to the lowest level possible in Canada. The World Health Organization (WHO) recommends having less than one percent of total energy intake coming from trans fat. The prohibition will help achieve our public health objective of 90% or more of people in Canada meeting this WHO target. This is expected to help reduce the risk of coronary heart disease among the general population.
In order to determine the effectiveness of the prohibition, Health Canada researchers performed analyses to determine trans fat intakes and the blood levels of trans fat of people living in Canada before the prohibition came into effect, based on data from the Canadian Community Health Survey (CCHS) – Nutrition 2015 and from the Canadian Health Measures Survey (CHMS) 2012-2015, respectively.
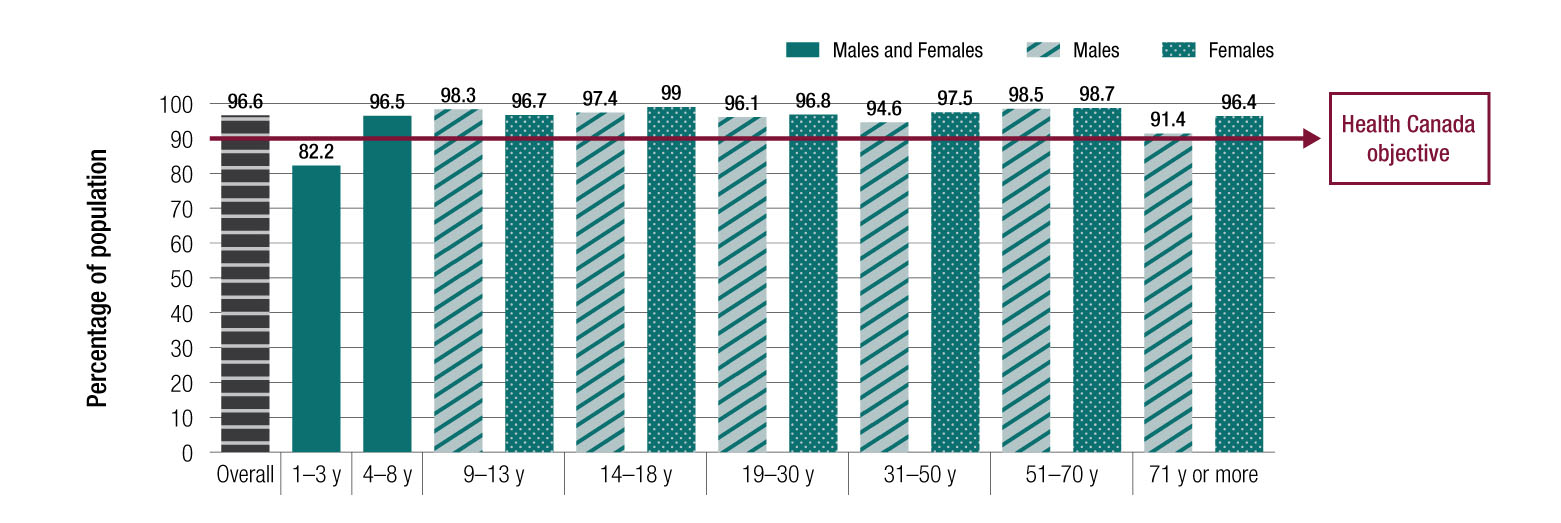
Figure 1: Text description
| Proportion of the population (%) | |
|---|---|
| Overall | 96.6 |
| 1-3 y | 82.2 |
| 4-8 y | 96.5 |
| 9-13 y – males | 98.3 |
| 9-13 y – females | 96.7 |
| 14-18 y – males | 97.4 |
| 14-18 y – females | 99 |
| 19-30 y – males | 96.1 |
| 19-30 y – females | 96.8 |
| 31-50 y – males | 94.6 |
| 31-50 y – females | 97.5 |
| 51-70 y – males | 98.5 |
| 51-70 y – females | 98.7 |
| 71 y or more – males | 91.4 |
| 71 y or more – females | 96.4 |
All age and sex groups had average intakes of trans fat that met the WHO recommendation before the prohibition. In addition, the target of 90% of the population having total trans fat intakes that meet the WHO recommendation had already been achieved in most groups, except children 1-3 years old (Figure 1). Even though less than 90% of children in that age group had intakes that met the WHO recommendation, it is important to note that most of the trans fats they consumed were naturally occurring in food (e.g. cow milk). Furthermore, their proportion of trans fat intake coming from foods containing industrially produced trans fats was the lowest of all groups.
The blood levels of trans fat of people living in Canada before the prohibition were similar for all age and sex groups. The levels observed have not been linked with increased cardiovascular disease risk.
The relatively low intakes and blood levels of trans fats are likely due to previous initiatives to reduce trans fats in foods starting with labelling of the amount of trans fat on most pre-packaged foods, which became mandatory in 2007. These analyses will be repeated using data from future cycles of CHMS and CCHS for determining further improvement after the PHO prohibition. Health Canada scientists will be submitting these results for publication in scientific journals in 2022.
Regulatory innovation
With factors such as growing populations, scientific advancements and climate change, the food system is continually evolving. This affects the way food is produced, processed, distributed, packaged, consumed and disposed of. As the food industry adapts and innovates, our traditional approaches to regulating food must evolve as well. Over time, our regulations have become difficult to navigate, understand and apply. Complex and outdated regulations make it challenging to respond to advances in science, technology or new product development. As a result, companies can face difficulties introducing new and innovative food products, or adopting new methods designed to make products safer or better for consumers. We are working to modernize our regulations so that they can accommodate the latest scientific or technological advances without lowering our safety standards. To support these efforts, we are also strengthening our communications to give people in Canada and the food industry the information they need about our requirements and processes. This includes making guidance we provide clearer and improving our consultation processes.
In 2021, we advanced several modernization initiatives in support of our Regulatory Innovation Agenda.

Chief Regulatory Officer
"While some of our regulatory modernization efforts were delayed because of the pandemic, modernizing outdated food rules and frameworks continues to be a high priority for Health Canada. Our goal is to build a regulatory framework for food that can accommodate evolution and innovation in the Canadian and global food environment."
David K. Lee
Chief Regulatory Officer
New regulations for human milk fortifiers
Human milk fortifiers (HMFs) are infant foods that are added to human milk to increase its nutritional value. They provide supplementary nutrients for infants who are born prematurely or have certain medical conditions, and are most commonly used in hospital neonatal intensive care units. The nutrients include added vitamins, mineral nutrients and amino acids, as well as protein, fat and carbohydrates, which are essential for these infants to develop and grow properly.
In April 2021, Health Canada updated regulations to better align them with evolving science and ensure the safety and efficacy of these products. Part of this work included consulting on and amending two marketing authorizations to allow Health Canada to authorize the safe uses of certain food enzymes and preservatives not previously permitted in human milk fortifiers. A modification to two food additive lists was published, which will allow these food additives to be used. Along with the new human milk fortifier regulations coming into force, technical guidance documents were made available to assist industry in preparing submissions.
Food labelling coordination
The Canadian Food Inspection Agency and Health Canada are the governing bodies responsible for food labelling and may update requirements for food products for several reasons. For example, amendments may be proposed to improve the clarity or availability of nutrition information. In 2021, we published a Joint Policy on Food Labelling Coordination which maintains our ability to take timely action to address consumer deception and acute risks to health and safety while still providing industry stakeholders with greater predictability around the timing of future labelling changes. This will allow them to plan and better prepare for future changes to food labelling.
Regulations for supplemented foods
Unlike conventional foods, supplemented foods are prepackaged foods containing one or more added supplemental ingredients, such as vitamins, minerals, amino acids, caffeine or herbal ingredients, which have historically been marketed as providing specific physiological or health effects. These ingredients can pose a risk to health if overconsumed by the general population or if consumed by certain vulnerable populations such as children or pregnant women. Given their potential risks, it is not suitable for supplemental ingredients to be added to food unless carried out according to specific safety requirements.
There is currently no regulatory framework for supplemented foods in the Food and Drug Regulations. As a temporary measure, Health Canada has been using Temporary Marketing Authorizations since 2012 to permit the sale of these products on a case-by-case basis and under specific conditions. Since the transition in 2012, Health Canada has issued over 1400 Temporary Marketing Authorizations for supplemented foods. In 2021 alone, we issued Temporary Marketing Authorizations for 136 new supplemented food products. Industry stakeholders have raised concerns that the Temporary Marketing Authorization framework involves a significant amount of administrative burden and is a barrier to innovation.
In 2021, Health Canada published a proposed regulatory framework for supplemented foods. The proposed regulations will establish detailed conditions for the use of supplemental ingredients in food to protect the health and safety of people in Canada while providing a predictable regulatory environment for industry. The proposal will also establish additional requirements on the labelling and advertising of supplemented foods beyond the general requirements for prepackaged foods.
Development of clinical trials framework for food for a special dietary purpose
Foods for a special dietary purpose have been specially processed or formulated to meet the particular requirements of people with a health condition or to be the sole or primary source of nutrition for an individual. Examples include infant formula, human milk fortifiers, formulated liquid diet products and foods to address special dietary needs for a medical condition, such as inborn error of metabolism. Clinical trial data is needed before these foods can be authorized for sale in Canada, but currently there is no regulatory framework in place enabling clinical trials on non-compliant foods for a special dietary purpose. We are developing new regulations to enable these types of clinical trials to be conducted in Canada, which will allow people in Canada to have early access to new products by removing the current barrier where a clinical trial has to be conducted outside of Canada before the product can be submitted to Health Canada for review. A pre-consultation with targeted stakeholders, from academia, industry and hospitals, along with a technical webinar, was conducted between April and June 2021 to solicit their input on the proposed regulatory framework. Draft regulations on clinical trials for foods for a special dietary purpose are planned for publication in late 2022. These proposed regulations are part of a broader regulatory modernization initiative for clinical trials, as set out in the Forward Regulatory Plan 2022-2024: Modernization of the Regulation of Clinical Trials.
Promoting healthy eating
An unhealthy diet is a major risk factor for chronic diseases such as diabetes and heart disease and these conditions are risk factors for COVID-19 severity and mortality in adults. While healthy eating is one of the best ways to protect and promote good health, a wide variety of factors influence our ability to make healthy food choices. Not only are our choices shaped by the food available in our homes, schools, restaurants and grocery stores, they are also influenced by social media and advertising. To help people in Canada make better choices, Health Canada developed a healthy eating strategy, which aims to make it easier to make healthier food choices by:
- improving the nutritional quality of foods;
- improving healthy eating information; and
- protecting populations considered vulnerable (pregnant women, children and people over the age of 65).
One of the tools used to promote our healthy eating strategy and give people in Canada healthy eating information is Canada's food guide.
Focus on…
Canada's food guide by the numbers
Number of:
- Food guide snapshots accessed*: 554,514
- Translated snapshots accessed*: 150,860
- e-newsletter subscribers: 61,196
- Website visits: 2,942,286
- Times content featured on social media: 911,117
- Food guide recipes accessed*: 1,345,989
* accessed = ordered + downloaded + viewed
Top three recipes:
Canada's food guide
Canada's food guide is the national authoritative resource on healthy advice and is widely integrated into policies and programs across the country. The 2019 food guide encourages people in Canada to eat a variety of nutritious foods each day, be mindful of eating habits, cook more often, and enjoy healthy food. It also encourages all sectors to take actions within their sphere of influence to help create supportive environments for healthy eating.
Increasing awareness and use of Canada's food guide
To ensure that Canada's food guide is accessible to all people in Canada, the food guide snapshot is available in 31 languages, including nine Indigenous languages. Over the past year, we have continued our work to provide food guide resources and content relevant to all. For example, Health Canada worked with the organization Quell, which is dedicated to reducing inequities in the food and hospitality industry and creating opportunities that reflect Canada's diverse and multicultural population, to develop 15 recipes for Canada's food guide.
In March 2021, we also launched a Healthy Eating At Home social media advertising campaign. The campaign encouraged families with children to make healthy eating choices at home during the pandemic using resources from Canada's food guide on YouTube, Facebook, Instagram and Pinterest. The campaign advertisements were displayed 21.5 million times, the video was viewed over 2 million times, and there was a 54% increase in visits to the food guide website during the campaign.
As many people have yet to discover the versatility of plant-based proteins or are unsure how to use them in everyday cooking, Health Canada worked in collaboration with the Guelph Family Health Study and the Food Innovation and Research Studio at George Brown College to develop 30 recipes using plant-based protein foods. We hope that these recipes will help support people in Canada in eating more plant-based protein foods as part of their everyday diet. Plant-based protein foods can provide more fibre and less saturated fat than other types of protein foods which can be beneficial for heart health.
To promote Canada's food guide, Health Canada also sends out a monthly e-newsletter to approximately 60,000 subscribers. The newsletter provides healthy eating tips, recipes and updates on food guide content, tools and resources. In 2021, we conducted a survey of subscribers to learn more about them and get feedback on the newsletter. Survey responses indicated that subscribers would like to see more recipes, engagement content (e.g., polls or quizzes) and appealing graphics. This feedback will help us improve the newsletter to better meet the needs of people in Canada.
Reaching youth
To encourage healthy eating habits from youth and into adulthood, teens and young adults are a key target audience for Canada's food guide awareness and education activities.
In January 2021, we established two youth engagement groups to engage their peers on Canada's food guide healthy eating messages, provide advice to Health Canada on education and awareness activities, and support the use and integration of the food guide across their campuses and in their communities both in-person and online. The Canada's food guide Youth Engagement Team, established in partnership with the Students Commission of Canada, is made up of 20 teens and young adults ages 12-25 from across Canada. The Canada's food guide Student Ambassador Network, established in partnership with Meal Exchange, is made up of 30 post-secondary students ages 19-25 from different institutions across the country. To date, the groups have provided input to Health Canada on a variety of topics, such as national advertising campaign messages, food guide brand extensions for children and teens, and diversity and inclusion in Canada's food guide. Youth have also started implementing activities in their communities, including virtual cooking workshops, social media posts and challenges, student surveys, town halls, and collaborations with campus stakeholders.
Health Canada also developed two distinct Canada's food guide brands for children (8-12 years) and teens (13-18 years) to inform the look and feel of resources targeted to them and ensure that they are relevant and relatable to these younger audiences. Brand concepts were tested directly with children and teens through public opinion research and were discussed with members of the Canada's food guide Youth Engagement Team for additional input.

Chief Medical Advisor
"In 2021, to continue our work to support the most vulnerable people in Canada, we provided information on healthy eating and advanced initiatives to promote healthier food choices. Our goal is to make healthier food choices easy and ultimately, positively affect their lives by decreasing certain risk factors, including obesity, that are associated with chronic diseases like diabetes and heart disease."
Dr. Supriya Sharma
Chief Medical Advisor
Assessing use of Canada's food guide
In collaboration with academic experts, Health Canada worked to develop tools to help researchers and policy makers. Assessing the extent to which people in Canada are following Canada's food guide recommendations is important to help inform future actions to support healthy eating. To do this, Health Canada collaborated with academic experts to develop tools that can be used by researchers and policy makers to consistently assess adherence to the food guide recommendations. The 2019 Healthy Eating Food Index is one tool that uses information about what a person ate in the last 24 hours to assess the extent to which they are following the 2019 Canada's food guide guidance on food choices. Two articles describing the development and the evaluation of this index have been published in the scientific journal Applied Physiology, Nutrition, and Metabolism.
Focus on…
Promoting healthy eating on campuses
Health Canada funded the City and University of Guelph to engage a network of university administrators, researchers, students, staff and food service providers to promote healthy eating as part of their Food Secure Campus Communities initiative. Working in partnership with the University of British Columbia and Meal Exchange, this included identifying and documenting promising programs, policies and projects that promote food security, including healthy eating initiatives on Canadian campuses. An interactive online map developed through this initiative enables organizations to collaborate, advance their efforts and help food insecure students locate local services. Through this collaboration, a set of principles was developed to provide a framework for promoting food security on campus. Principles under the health and wellbeing component of the framework include "healthy eating is promoted and accessible on campus" and "campus promotes student awareness and skills for healthy eating." These principles help to encourage alignment of campus food security efforts with Canada's food guide recommendations.
Additional healthy eating initiatives
Nutrition labelling
In 2016, Health Canada amended regulations to make improvements to the nutrition facts table and list of ingredients on food labels. These amendments make them easier to understand, thereby helping people in Canada make informed choices. The transition period for the 2016 nutrition labelling changes ended on December 14, 2021. However, given the challenges imposed by the COVID-19 pandemic, the Canadian Food Inspection Agency will focus its efforts on education and compliance promotion for the first year. As of December 15, 2022, until December 14, 2023, they will verify compliance and apply enforcement discretion in cases where non-compliant companies have detailed plans showing how they intend to meet the new requirements at the earliest possible time. However, based on food label data for over 6500 new products collected by Mintel in 2021, 69% had updated labels showing the new nutrition facts table.
In June 2021, Health Canada also launched the Nutrition Labelling Online Course to provide information to health professionals and educators about nutrition labelling. Topics include food labelling in Canada, the nutrition facts table, the serving size, the % daily value, the list of ingredients and nutrition claims. This tool contains interactive content, quizzes, and ready-to-use resources activities. Completing this course will increase understanding of the latest nutrition labelling information and assist health care professionals share this information with people in Canada.
Amendments to Table of daily values and Table of reference amounts for foods
In June 2021, Health Canada published two Notices of Proposal to update the Table of Daily Values and Table of Reference Amounts for Foods.
Health Canada proposed to update the Table of Daily Values by modifying the daily values of potassium and sodium for specific age groups to better align with the latest dietary reference intakes for these nutrients. The table sets out the reference points upon which the % daily values in the nutrition facts table are based.
Health Canada also proposed amendments to the Table of Reference Amount for Foods based on new data and to address gaps in certain food categories. The Table sets out reference amounts for different food categories and provides detailed instructions on how to declare the serving size in the nutrition facts table. Publication of the Notices of Proposal was followed by a 75-day consultation period. Based on consultation feedback, Health Canada plans to finalise these amendments in 2022.
Monitoring food and beverage advertising to children
Food and beverage marketing is all around us and many of the foods and drinks that are frequently advertised contribute too much sodium, sugars, or saturated fat to our eating patterns. Health research has conclusively linked food advertising to children's food preferences, requests, purchases and eating behaviors, and can therefore be a key contributor to childhood obesity and chronic diseases. Throughout 2021, Health Canada continued to advance an evidence-based monitoring strategy. The collective results from planned and ongoing monitoring activities will provide invaluable data and information about the extent and nature of food advertising aimed at children in Canada.
To support this work, we have developed strong partnerships with recognized subject matter experts in the field, and supported the collection of new data on the state of food advertising in a range of media and settings, including recreation facilities. Around 75% of children in Canada participate in organized sports, and many recreation facilities offer activities and events aimed at children. While recreation facilities are a great resource for parents to help children socialize and stay active, finding healthy food options can sometimes be challenging and food advertising is often found in this setting. This makes recreation facilities a critical area to monitor because they are a popular gathering place for children and because they can influence children's dietary intake.
In January 2021, facility managers and directors of recreation centres were canvassed to learn more about food promotion in recreation facilities. The majority of respondents (89%) said that up to 75% of their visitors in the last year were children under 13 years old. The survey findings also suggest that many common activities (for example, facility food service contracts or sports team sponsorship) may result in children's exposure to food advertising in recreation facilities. In 2022, a second phase of this study will be launched, consisting of audits of recreation facilities to document food promotion on the ground.
In last year's highlights report, we reported on how Health Canada has supported the expansion of the International Food Policy Study to include children and youth between the ages of 10 and 17 years. The study evaluates the impact of national-level food policies by conducting surveys across Canada, the United States, Australia, Chile, the United Kingdom and Mexico. Having data on this age group helps us assess how different factors or policies such as food advertising, the food guide, nutrition labelling, and the nutrition environment at schools influence the eating habits of children in Canada.
The study found that among youth in Canada, about 70% of respondents reported seeing or hearing advertisements for certain food categories in 2020, particularly for fast food. Only about 25% reported seeing advertisements for fruit or vegetables. Supporting the survey in 2020 allowed us to observe a unique time in the lives of children and youth around the world, by comparing data from 2019 and 2020. Between 2019 and 2020, youth in Canada reported less exposure to food and beverage advertising potentially because of the pandemic. This finding was consistent across all countries surveyed. For youth in Canada, advertising exposure tended to occur in TV/movies, on websites/social media, and in stores. Cartoons/characters made by food companies were the most commonly reported marketing strategy for unhealthy food or drinks by youth in Canada.
In the 30 days, how often did you see or
hear advertisements for these kind of foods or drinks?
% ≥ once per week
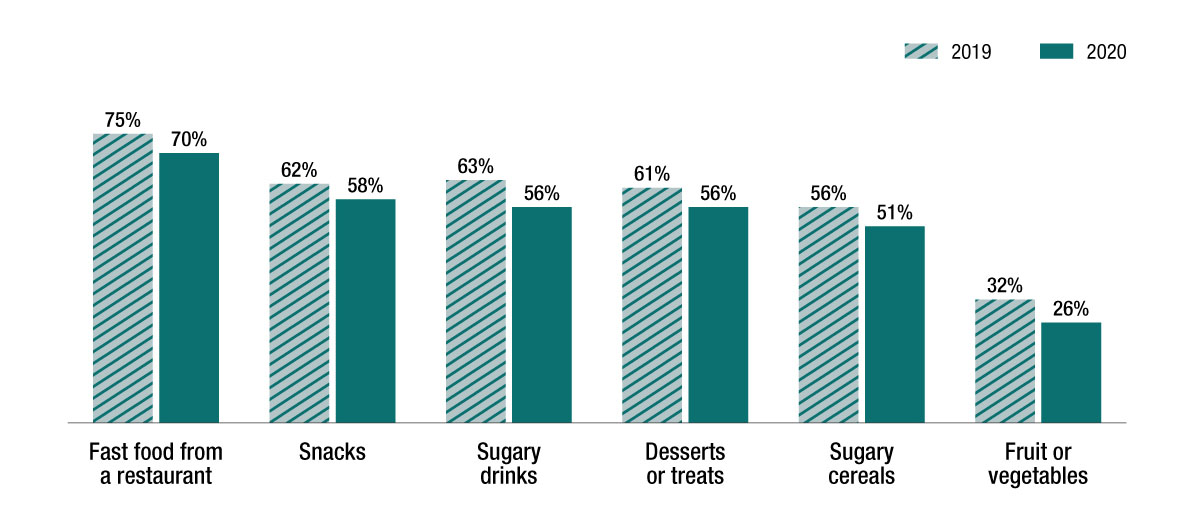
Among all Canadian respondants
(2019 N=3682; 2020 N=3897)
Reference: Hammond D, White CM, Vanderlee L, Acton RB, Rynard VL, on behalf of the IFPS research team. International Food Policy Study Youth Survey: 6 Country 2020 Data Summary. November 2021.
Figure 2: Text description
| 2019 (n=3682) | 2020 (n=3897) | |
|---|---|---|
| Fast food from a restaurant | 75 | 70 |
| Snacks | 62 | 58 |
| Sugary drinks | 63 | 56 |
| Desserts or treats | 61 | 56 |
| Sugary cereals | 56 | 51 |
| Fruit or vegetables | 32 | 26 |
The survey also indicated that youth awareness of Canada's food guide remains very high. Over 80% of youth in 2019 and 2020 reported learning about the food guide in school. A majority of respondents in 2020 reported making a special effort to follow food guide recommendations by drinking water, eating vegetables and fruits, and eating less sugar or fewer sugary drinks. Other positive trends include reported decreases in consumption of fast food and sugary drinks between 2019 and 2020, which may be a result of the COVID-19 pandemic and youth spending less time away from their homes. Ongoing participation in the survey will allow us to see whether these changes are temporary or carry over into future years. While these are all positive trends, the reported intake of fruits and vegetables remained low as youth reported eating fewer than two fruits or vegetables a day, which appears to be a consistent finding across all six countries surveyed.
Focus on…
Health Canada's Nutrition Science Advisory Committee
Established in 2020, Health Canada's Nutrition Science Advisory Committee is a group of external expert advisors that provide scientific and technical advice on nutrition in a timely and independent manner. The committee held four meetings in 2021 and provided advice in several areas including emerging scientific trends that affect the nutritional health of people of Canada. This group of external expert advisors has provided us with scientific and technical advice on the evidence review cycle for dietary guidance, and "what" and "how" we can be more responsive in food and nutrition monitoring. The Nutrition Science Advisory Committee's advice will support Health Canada's efforts to use the best available evidence in fulfilling its mandate to support the nutritional health of people of Canada, and to support building a stronger food and monitoring infrastructure to understand new and emerging trends impacting the nutritional health of people in Canada.
International collaboration
Health Canada fosters international partnerships and collaborations in a wide range of sectors including food and nutrition. Collaboration with international partners significantly increases the information available, thereby enhancing the rigour of our decisions. Health Canada works with a wide range of bilateral partners and international organizations to advance issues related to food safety and nutrition. In 2021, Health Canada participated in a number of international committees, liaison groups and working groups.
Building international relationships
In 2021, Health Canada regularly met with counterparts from the United States Food and Drug Administration & Food Standards Australia New Zealand, established relationships with counterparts in the United Kingdom and participated in various international liaison groups with the objective of enhancing our understanding of current and emerging issues of mutual interest and concern related to food pathogens, food chemicals and nutritional safety. Health Canada acted as secretariat of the International Food Chemical Safety Liaison Group from 2019 – 2021; the third time it has done so since the group was formed in 2005. This group is a forum for discussion and collaboration amongst government organizations involved in the risk assessment, risk management, and communication of food chemical safety issues.
Codex Alimentarius Commission and the Codex Committee on Food Labelling (CCFL46)
Established in 1963 by the World Health Organization, and the Food and Agriculture Organization of the United Nations, the Codex Alimentarius Commission develops international food standards to protect health and to facilitate fair practices in the food trade. Its standards, developed through its subsidiary bodies, contribute to the safety of the global food supply and help to protect the people in Canada. Canada has been the permanent host of the Codex Committee on Food Labelling (CCFL) since its inception in 1965; hosting this event is one of Canada's most long-standing and significant international engagement activities related to food and nutrition. Most recently, in 2021, Health Canada successfully hosted the 46th Codex Committee on Food Labelling, attended by more than 600 representatives from more than 75 countries. This session of the committee focused on discussing food labelling and in particular, labeling of allergens, front-of pack labelling, e-commerce and the use-of technology in labelling. In addition to this work, Health Canada's Food Directorate led eight Canadian delegationsFootnote 1 to technical and horizontal Codex Committees and participated in supporting the development of many international food safety and nutrition standards which will help to maintain the safety of the food system at home and abroad.
International allergen labelling regulatory workshops
Health Canada has hosted a series of workshops organized in Korea, Vietnam, Indonesia, and Taiwan to help avoid or reduce the number of allergen labelling issues in imported products to Canada. We recently participated in a workshop organized by the Canadian Food Inspection Agency with the Government of India to educate food exporters about Canada's food allergen labelling regulations. The training provided a better appreciation of Canadian ingredient labelling compliance considerations with focus on the Food and Drugs Act and Regulations. Participation in these workshops will help ensure that products imported from these countries will be appropriately labeled for allergens according to our regulations.
Communicating food safety risks
Health Canada also participated in a series of workshops to develop an Asia Pacific Economic Cooperation (APEC) Food Safety Risk Communication Framework and Supporting Guidelines, and played a leadership role in the drafting of certain supporting guidelines. This framework and its supporting guidelines focus on harmonizing best practices for effective communication about food safety matters, to the public and industry.
Dietary reference intakes
Dietary Reference Intakes are a set of reference values for nutrients that form an important scientific basis for food and nutrition programs, policies, and regulations. For example, they inform the daily value on nutrition labels and ensure foods and supplements contain safe levels of nutrients. Canada and the United States have collaborated since the mid-1990s to provide joint support for the development of Daily Reference Intake values. In 2021, Health Canada and several U.S. government agencies jointly commissioned a review of the values for energy, which were last published in 2002. The review and establishment of Daily Reference Intake values are overseen by the independent National Academies of Science, Engineering, and Medicine. The work will include assessment of human requirements for energy intake as well as examination of the evidence of relationships with chronic disease and other health outcomes.
Annex A: Approvals
Approved Novel Foods
- Napin-rich canola protein isolate: This alternative source of plant-based protein can be used in unstandardized foods.
- Plum Pox Virus (PPV) Resistant C5 Plum: This plum was developed to be resistant to plum pox virus, kanamycin antibiotics, and β-glucuronidase-based visual marker.
- Quizalofop-tolerant rice (RTA1): This rice line demonstrates tolerance to aryloxyphenoxypropionate (FOP) herbicides, such as quizalofop-p-ethyl (Quizalofop) which is used to control grass weeds.
- 2'Fucosyllactose produced via fermentation of a genetically modified E.coli K12 MG1655 strain: This carbohydrate derived from genetically modified E.coli is intended for use in infant formula, foods for young children and other foods.
- D-tagatose: This enzymatically synthesized D-tagatose is intended for use as a sweetening agent to fully or partially replace existing added sugars, but can be used as a flavor enhancer, humectant, texturizer, stabilizer, and food ingredient in various foods.
- High Oleic Soybean: This soybean line was developed to produce oil with a high oleic acid content, and decreased saturated fat compared to commodity soybean oil.
- 2'-fucosyllactose (2'-FL) to foods for children 1 to 3 years of age: This carbohydrate derived from genetically modified E.coli is intended for use in toddler formulas (liquid, or powder to be reconstituted) for children 1 to 3 years of age, formulated as nutritional supplements.
- MON 95379 lepidopteran protected corn: This genetically modified corn has been developed to be resistant to feeding damage caused by targeted lepidopteran pests.
- Event EF2-114 Pineapple: This genetically modified pineapple variety exhibits pink-coloured fruit flesh through increased expression of lycopene is intended for use in human foods.
- Camelina Oil derived from Thifensulfuron-Tolerant Camelina line 14CS0851-01-14: This genetically modified camelina (Camelina sativa L. Crantz) line displays tolerance to a sulfonylurea (SU) herbicide.
Approved Infant Formula and Human Milk Fortifiers
- Enfamil®A+ Concentrate 385 mL Metal Can to include a new source of DHA (DHASCO-B) and reduced levels of Arachidonic Acid (ARA)
- Similac®Alimentum®Powder, with an addition of 2'-fucosyllactose and lutein for a hypoallergenic infant formula for term infants
- Addition of a resealable closure (DreamCap™ 26) to Nestlé Good Start concentrated liquid infant formulas
- Metabolic Level 1 Products: Nine powdered formulas (Cyclinex®-1, Glutarex®-1, Hominex®-1, Ketonex®-1, I-Valex®-1, Phenex®-1, Propimex®-1, Tyrex®-1, ProPhree®) packaged in 400 g metal cans for the nutritional management of infants with inborn errors of metabolism
- Enfamil®A+®Powdered Infant Formula in a Multi-Serve Pouch in Carton: 750 g pouch
- New whey protein concentrate for Similac®Pro-Advance®Powder Step 1 & 2
- Nutramigen A+ Ready-to-Feed Liquid in a 946 mL Aseptic Plastic Bottle with a new source of DHA (DHASCO-B) reduced levels of Arachidonic Acid (ARA) and increased levels of inositol
- Similac Human Milk Fortifier Special Care 30
- Similac Human Milk Fortifier Extensively Hydrolyzed Protein Concentrated Liquid
- Similac Human Milk Fortifier Hydrolyzed Protein Concentrated Liquid for Preterm Babies for the reduction of potassium level
Approved Food Additives
- Modified Vinegar: Part 2 of the List of Permitted Preservatives was modified to enable its use as a preservative in various standardized meat and poultry products as well as in unstandardized preparations of meat and poultry products.
- Maltogenic alpha-Amylase: the List of Permitted Food Enzymes was modified to enable its use from three sources in bread, flour, whole wheat flour and unstandardized bakery products
- Glutaminase: the List of Permitted Food Enzymes was modified to enable its use from Bacillus licheniformis SJ13263 in various Foods
- Milk Coagulating Enzyme: the List of Permitted Food Enzymes was modified to enable its use from Rhizomucor miehei CBS 146319 in yogurt
- Lysophospholipase: the List of Permitted Food Enzymes was modified to enable its use from Trichoderma reesei RF7206 in the production of sugars and dextrin from starch
- Methylparaben, Propylparaben and their Sodium Salts: Part 2 of the List of Permitted Preservatives was modified to enable their use in various foods
- Dimethyl Dicarbonate: Part 3 of the List of Permitted Preservatives was modified to enable its as a preservative in fruit wine, cider, and perry (a product of the alcoholic fermentation of pear juice)
- Steviol Glycosides: the List of Permitted Sweeteners was modified to enable their use in a variety of foods
- Three Food Additives to Certain Plant Protein Isolate-based Products: The List of Permitted Emulsifying, Gelling, Stabilizing or Thickening Agents, the List of Permitted Food Enzymes and Part 2 of the List of Permitted Preservatives were modified to enable the use of gellan gum, nisin and transglutaminase from Streptoverticillium mobaraense S-8112 in certain plant protein isolate-based products that resemble egg products
- Potassium Aluminum Sulphate: the List of Permitted Firming Agents was modified to enable its use in salted jellyfish
- Sodium Phosphate, Tribasic: the List of Permitted Food Additives with Other Accepted Uses was modified to enable its use to reduce processing losses and thaw drip in frozen shrimp and frozen cooked shrimp
- Chymosin from Aspergillus niger DSM 32805: the List of Permitted Food Enzymes was modified to enable its use in certain dairy-based foods
- Certain Food Additives to Human Milk Fortifier: the List of Permitted Emulsifying, Gelling, Stabilizing or Thickening Agents and the List of Permitted pH Adjusting Agents, Acid-Reacting Materials and Water Correcting Agents were modified to enable the use of carrageenan, gellan gum, lecithin, monoglycerides, potassium citrate, potassium hydroxide, and sodium citrate for use in human milk fortifier
- Hydrochloric Acid: the List of Permitted pH Adjusting Agents, and the list of Acid-Reacting Materials and Water Correcting Agents were modified to enable its use as a pH adjusting agent in specific unstandardized foods
- Potassium Polyaspartate: the List of Permitted Food Additives with Other Accepted Uses was modified to enable its use in wine
- β-amylase from Bacillus flexus AE-BAF: the List of Permitted Food Enzymes was modified to enable its use is various foods
- Spirulina Extract: the List of Permitted Colouring Agents was modified to enable its use in certain unstandardized foods
- Xylanase from Aspergillus acidus RF7398: the List of Permitted Food Enzymes was modified to enable its use in bread, flour, whole wheat flour, pasta, and unstandardized bakery products
- Caramel: the List of Permitted Colouring Agents was modified to enable its use as a colouring agent in a flavouring preparation used in the manufacture of flavoured cheddar cheese
- Pectinase from Aspergillus niger: the List of Permitted Food Enzymes was modified to enable its use in unstandardized fruit and vegetable products
Healthy clicks: Food and nutrition at a glance
Stay informed about our activities:
Follow us on Facebook
Follow us on Twitter
Follow us on Instagram
Follow us on YouTube
Find more information on healthy eating on our Food and Nutrition page
See the latest news from Health Canada on our website
Find other Health-related information on the Government of Canada website
Sign up to our Stakeholder Registry
Embrace healthy eating:
Consult Canada's food guide
Take inspiration from the Food guide snapshot
Check out Canada's Dietary Guidelines
Subscribe to the food guide's e-newsletter
Try one of our recipes
Seek out our food safety advice:
Protect yourself with our food safety tips
Refer to our safe cooking temperatures
Footnote
- Footnote 1
-
Including Codex Committee on General Principles, Codex Committee on Contaminants in Foods, Codex Committee on Food Additives, Codex Committee on Food Hygiene, Codex Committee on Methods of Analysis and Sampling, Codex Committee on Nutrition and Foods for Special Dietary uses, Executive Committee of the Codex Alimentarius Commission, and the FAO/WHO Coordinating Committee for North America and South West Pacific.
